Abstract
Background:
Tourniquet-related responses are a frequent clinical challenge during foot and ankle surgery under peripheral nerve block. Pharmacologic sedation and analgesia represent a common approach. Ciprofol, a novel intravenous sedative, remains underexplored in this surgical context.
Objective:
This study aimed to evaluate the impact of ciprofol-assisted sedation on tourniquet-related responses in patients undergoing foot and ankle surgery.
Design:
In total, 240 patients scheduled for foot and ankle procedures under peripheral nerve block with the use of a high thigh tourniquet were enrolled and randomized to undergo ciprofol (Group C) or propofol (Group P) sedation.
Methods:
In Group C, ciprofol was administered at an initial rate of 0.5 mg/(kg·h), with maintenance doses ranging from 0.5 to 2 mg/(kg·h) to maintain a BIS (bispectral index) between 65 and 80. Group P received an initial infusion of propofol at 2 mg/(kg·h), with maintenance between 2 and 8 mg/(kg·h). Primary outcomes included the incidence of intraoperative rescue analgesia and body movement. Secondary outcomes encompassed hemodynamic fluctuations, anesthesia success rate, injection-related discomfort, respiratory depression, blood pressure abnormalities, postoperative complications, and long-term complications.
Results:
There were no significant differences between groups in terms of the need for rescue analgesia or body movement during surgery. However, patients in the ciprofol group experienced significantly lower rates of injection pain and intraoperative hypotension relative to the propofol group (p < 0.05). No significant differences were found in postoperative adverse effects or long-term complications. In addition, both anesthesia completion rates and physician/patient satisfaction scores were similar between the two groups.
Conclusion:
BIS-guided sedation with ciprofol during foot and ankle surgery provides equivalent control of tourniquet-related responses compared to propofol. However, ciprofol demonstrates superior intraoperative hemodynamic stability and a more favorable safety profile, including reduced adverse event rates.
Trial Registration:
http://www.chictr.org.cn; ChiCTR2400083924; May 7, 2024.
Plain language summary
Introduction
Foot and ankle surgeries are frequently performed procedures that encompass interventions such as fracture fixation, soft tissue debridement, and removal of orthopedic implants following bone healing.1,2 Intraoperatively, a tourniquet is commonly employed in most of these procedures to minimize bleeding and enhance surgical visibility. 3 Although tourniquets are effective tools for achieving hemostasis, their use can provoke undesirable physiological reactions and complications, which may adversely influence patient outcomes and comfort. 4 Various strategies have been explored to mitigate these effects, including optimizing cuff size and inflation parameters, employing ischemic preconditioning techniques, and utilizing appropriate anesthetic regimens.5–7 Of the methods tested to date, pharmacologic sedation and analgesia remain key strategies.
Propofol is a widely employed intravenous anesthetic known for its rapid onset and brief recovery period. 8 Nevertheless, its clinical use is often limited by adverse effects such as respiratory depression, hypotension, and injection-associated pain. Ciprofol, a novel intravenous sedative within the alkylphenol class that includes propofol, 9 is a γ-aminobutyric acid (GABA) type A receptor agonist that enhances inhibitory neurotransmission.10,11 Preliminary studies suggest that ciprofol provides comparable, if not superior, anesthetic efficacy, with advantages including faster onset, reduced cardiovascular suppression, and lower incidence of injection-related discomfort. 12 These characteristics highlight ciprofol as an alternative to propofol for procedural sedation.
In light of these prior findings, the present randomized clinical trial was developed with the aim of comprehensively assessing the effects of ciprofol versus propofol sedation on intraoperative responses to tourniquet application in patients undergoing foot and ankle surgery under peripheral nerve block.
Methods
Study design
This prospective, randomized, controlled clinical trial was performed at the First Affiliated Hospital of the University of Science and Technology of China. The reporting of this study conforms to the CONSORT statement (Supplemental Material). 13 In addition, the trial adhered to the Declaration of Helsinki.
Study subjects
The study enrolled 240 adult patients undergoing elective foot and ankle surgery with a peripheral nerve block and the use of a high thigh tourniquet. This study was initiated in May 2024 and completed in May 2025.
Inclusion criteria
All enrolled patients met the following criteria: (1) 18–60 years old, (2) American Society of Anesthesiologists (ASA) class I and II, and (3) provided informed consent after being informed of the study intervention and evaluations.
Exclusion criteria
Participants were excluded if they (1) had significant dysfunction of the cardiovascular, respiratory, hepatic, renal, or immune systems, (2) had not discontinued antioxidant or anti-inflammatory medications at least 1 week before surgery, (3) had contraindications for peripheral nerve block, (4) had known allergies to propofol, ciprofol, or related anesthetic agents, (5) required a tourniquet application time of less than 40 min or more than 120 min, (6) required any change in the anesthesia plan during surgery, or (7) had a body mass index of 30 kg/m² or greater.
Randomization and blinding
Upon successful completion of the peripheral nerve block, patients were randomly assigned to ciprofol (Group C) or propofol (Group P) groups at a 1:1 ratio. Randomization was achieved using a computer-generated sequence and sealed opaque envelopes to ensure allocation concealment. Outcome assessors and data analysts were blinded to group assignments throughout the study period.
Anesthesia approach
After arriving in the operating room, all patients had a peripheral intravenous catheter placed, and lactated Ringer’s solution was initiated at a rate of 4–6 mg/(kg·h). Standard monitoring protocols were followed, including continuous electrocardiography (ECG), heart rate (HR), non-invasive blood pressure (BP), peripheral oxygen saturation (SpO2), and bispectral index (BIS) monitoring. Regional anesthesia was then performed using combined ultrasound-guided femoral and sciatic nerve blocks. For the sciatic nerve block, with the patient positioned laterally and the affected leg flexed at the hip and knee, a high-frequency linear ultrasound transducer (Sonosite M-TURBO; Sonosite Inc., USA; 6–13 MHz) was placed transversely at the popliteal fossa. The sciatic nerve was visualized in cross-section as a hyperechoic, oval structure with a honeycomb appearance. Under direct ultrasound guidance, 20 mL of 0.375% ropivacaine was injected via an in-plane needle approach to achieve a successful blockade. For the femoral nerve block, patients were then placed supine with the operative limb slightly abducted. The femoral nerve was identified 1 cm lateral to the femoral artery and 2 cm below the inguinal ligament using transverse ultrasound scanning. The nerve appeared as a flattened, hyperechoic oval structure. An in-plane approach was used to inject 20 mL of 0.375% ropivacaine around the nerve. All ultrasound-guided femoral and sciatic nerve blocks were performed by a supervising physician with over 5 years of experience in regional anesthesia. Twenty minutes after completing the block, sensory function was evaluated using a standardized pinprick assessment. Only patients who demonstrated complete sensory loss within both the femoral and sciatic nerve distributions, and who were expected to show no pain response to the surgical incision, were considered to have achieved a successful block and were eligible to continue in the study. Following confirmation of adequate sensory blockade, patients received supplemental oxygen via nasal cannula at a flow rate of 3 L/min. Sedation was then initiated using either ciprofol or propofol via a continuous infusion pump. After 10 min of sedative administration, a pneumatic tourniquet was applied at the proximal thigh of the surgical limb. After exsanguination, the cuff was inflated to a pressure of systolic BP + 100 mmHg (1 mmHg = 0.133 kPa). 14 Sedation infusion was discontinued immediately upon tourniquet release.
For hemodynamic management, if intraoperative changes in BP or HR exceeded ±20% of baseline values, titration of the sedative infusion rate was performed as a first-line measure. If hemodynamic instability persisted or showed no improvement within 5 min, pharmacologic interventions were initiated. For hypertension (BP >20% above baseline), 0.2 mg of nicardipine was administered. For hypotension (BP >20% below baseline), either 8 μg of norepinephrine or 6 mg of ephedrine was given. In cases of tachycardia (HR >100 bpm), esmolol 5–10 mg was administered, while bradycardia (HR <50 bpm) was treated with atropine 0.25–0.50 mg, with repeat dosing as needed. Respiratory depression, defined as SpO2 <95%, was managed by increasing oxygen flow, performing a jaw thrust maneuver, or delivering pressurized oxygen via a mask. If the patient exhibited ⩾2 episodes of involuntary body movement (e.g., limb or head movement) or if such movements interfered with the surgical procedure, a rescue dose of sufentanil (5 μg) was administered. In cases where sedation remained inadequate or tourniquet intolerance was unmanageable, the anesthesia plan was converted to general anesthesia with a laryngeal mask airway.
Interventions
Group C: Sedation commenced with a ciprofol infusion at 0.5 mg/(kg·h). The rate was increased by 0.1 mg/(kg·h) every minute until the BIS value dropped to ⩽80, at which point escalation ceased and a continuous infusion was maintained. During surgery, the infusion rate was kept between 0.5 and 2 mg/(kg·h), with adjustments in 0.1 mg/(kg·h) increments to maintain a target BIS range of 65–80.
Group P: Propofol infusion began at 2 mg/(kg·h) and was incrementally increased by 0.5 mg/(kg·h) per minute until the BIS value fell below 80. Once this threshold was reached, further increases were stopped. Intraoperatively, the infusion rate was maintained between 2 and 8 mg/(kg·h) and adjusted in 0.5 mg/(kg·h) steps to maintain BIS values within the target range of 65–80.
Outcomes
Hemodynamic parameters (BP, HR) and BIS values were recorded at seven predefined time points: before nerve block administration (T0), immediately before tourniquet inflation (T1), at the time of skin incision (T2), 30 min post-tourniquet inflation (T3), immediately before tourniquet deflation (T4), 10 min post-tourniquet deflation (T5), and 10 min after surgery completion (T6). Primary outcomes included the need for intraoperative rescue analgesia and body movement. Secondary outcomes included the following: (1) anesthesia success rate, defined as uninterrupted surgery without conversion or adjunct sedative use, maintaining BIS within 65–80 for ⩾70% of sedation duration.; (2) intraoperative adverse events, including injection pain, respiratory depression, hypertension, and hypotension; (3) recovery time as measured from cessation of sedative infusion to eye opening; (4) use of vasoactive agents during surgery; (5) total sedative dose (ciprofol or propofol); (6) postoperative complications, including nausea, vomiting, dizziness, drowsiness, delirium, and requirement for analgesics; (7) physician and patient satisfaction scores (out of 10); and (8) postoperative complications within 2 months, including chronic pain, swelling, infection, thrombosis, or nerve injury.
Statistical analyses and power calculations
Based on findings from a phase III clinical trial, the incidence of injection pain was estimated at 8.1% for ciprofol and 21.4% for propofol. 15 Assuming a 1:1 group allocation, a two-sided alpha of 0.05, 80% statistical power (1 − β), the preliminary calculation indicated that a minimum of 108 participants per group was required. Accounting for an estimated 10% dropout rate, a total of 240 patients (120 per group) was determined to be necessary for this study based on analyses conducted using PASS 11.0 (NCSS, LLC, Kaysville, UT, USA).
Data were analyzed in SPSS 26.0 (IBM Corp., Armonk, NY, USA). Continuous variables were reported as mean ± standard deviation (SD) if normally distributed, or as median (interquartile range) otherwise. Student’s t-test was applied for comparisons of normally distributed variables, while the Wilcoxon rank-sum test was used for skewed data. Categorical data were presented as frequencies with percentages, and compared with the χ² test. Repeated measures analyses of variance were employed for intragroup time-point comparisons. Statistical significance was defined as p < 0.05.
Results
Patient characteristics and surgical parameters
Out of the 240 patients initially enrolled, 223 were included in the final analysis after excluding individuals due to protocol deviations, such as unsuitable tourniquet application duration, changes in tourniquet type, or intraoperative deviation from the anesthesia plan (Figure 1). Group P consisted of 112 patients, and Group C included 111 patients. The demographic and surgical-related characteristics were comparable between the two groups (Table 1).

Flow chart of participants in the study.
Participant demographic and surgical-related characteristics.

Data are means (standard deviations), medians (interquartile range), or numbers (percentages).
ASA, American Society of Anesthesiologists; BMI, body mass index; F, female; M, male; ORIF, open reduction and internal fixation.
Comparative analysis of intraoperative parameters
As shown in Table 2, there were no significant differences in intraoperative rescue analgesia requirements, incidence of body movements, respiratory depression, hypertension, anesthesia completion rates, recovery times, vasoactive drug usage, or total administered doses of ciprofol or propofol between Groups P and C (p > 0.05 for all comparisons). However, Group C exhibited a significantly reduced incidence of both hypotension and injection pain relative to Group P (p < 0.05).
Intraoperative comparisons among patient groups.
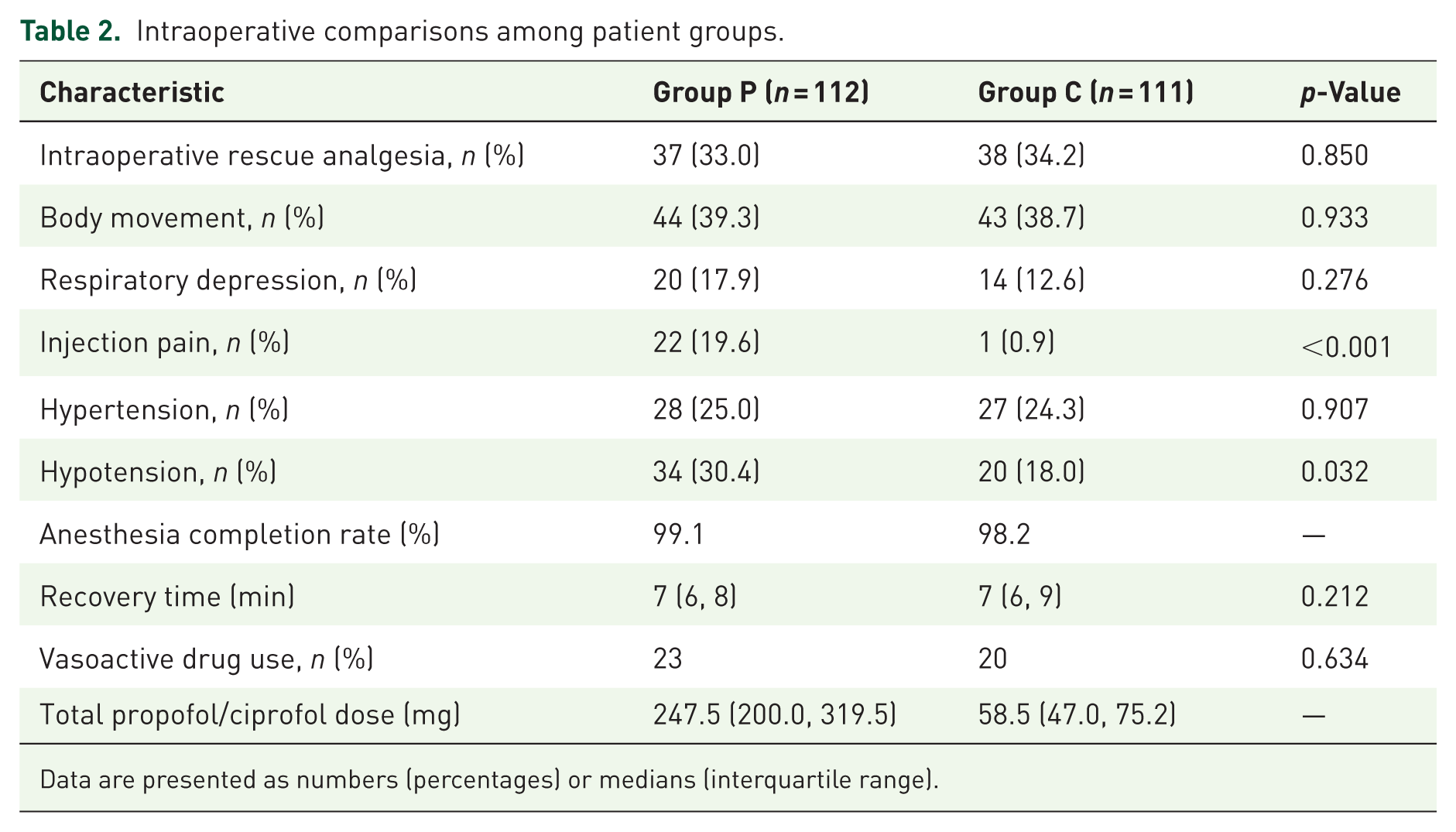
Data are presented as numbers (percentages) or medians (interquartile range).
Hemodynamic trends over time
Both groups experienced significant declines in mean arterial pressure (MAP) and HR at time points T2 and T3 compared to baseline (T0), indicating comparable hemodynamic responses during the initial surgical phases (p < 0.05). MAP and HR followed similar temporal patterns during tourniquet inflation in both cohorts. Notably, Group P displayed a significantly lower MAP than Group C at T3 (86.7 ± 14.2 mmHg vs 90.6 ± 14.8 mmHg, p < 0.05; Figure 2). In addition, BIS values at T5 were significantly lower in the ciprofol group compared to the propofol group (85.7 ± 6.1 vs 88.0 ± 6.0, p < 0.05; Figure 3), suggesting potential differences in sedative pharmacodynamics during recovery.
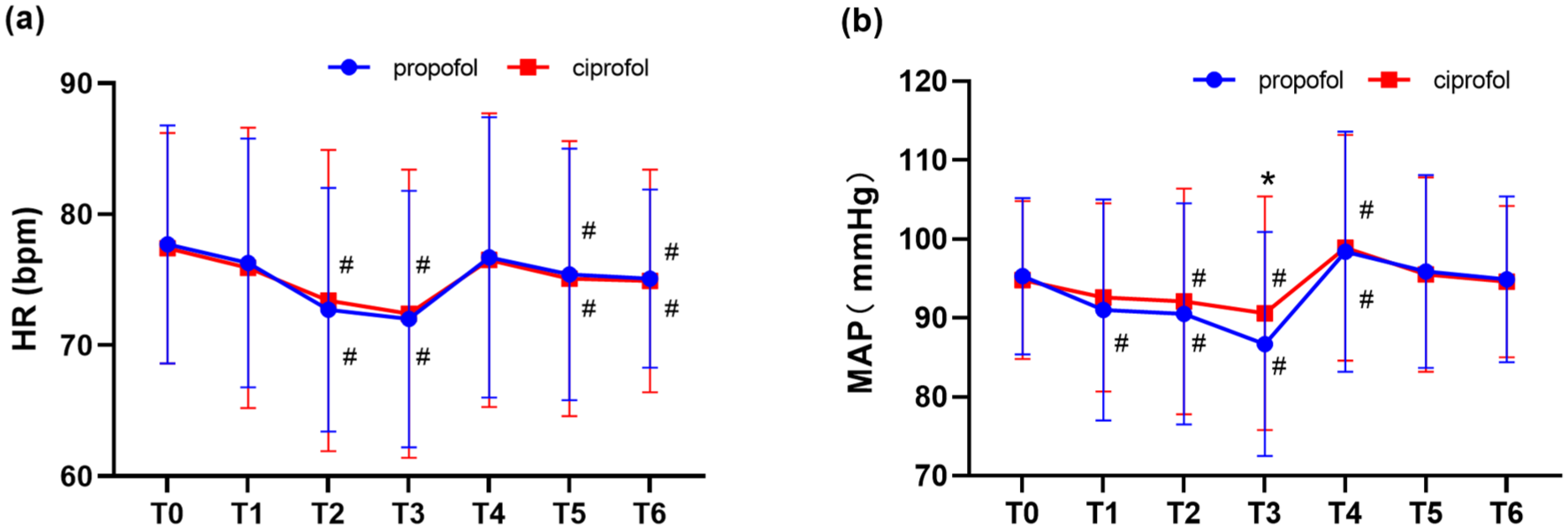
Time-dependent changes in HR and MAP. (a) HR and (b) MAP changes over time.
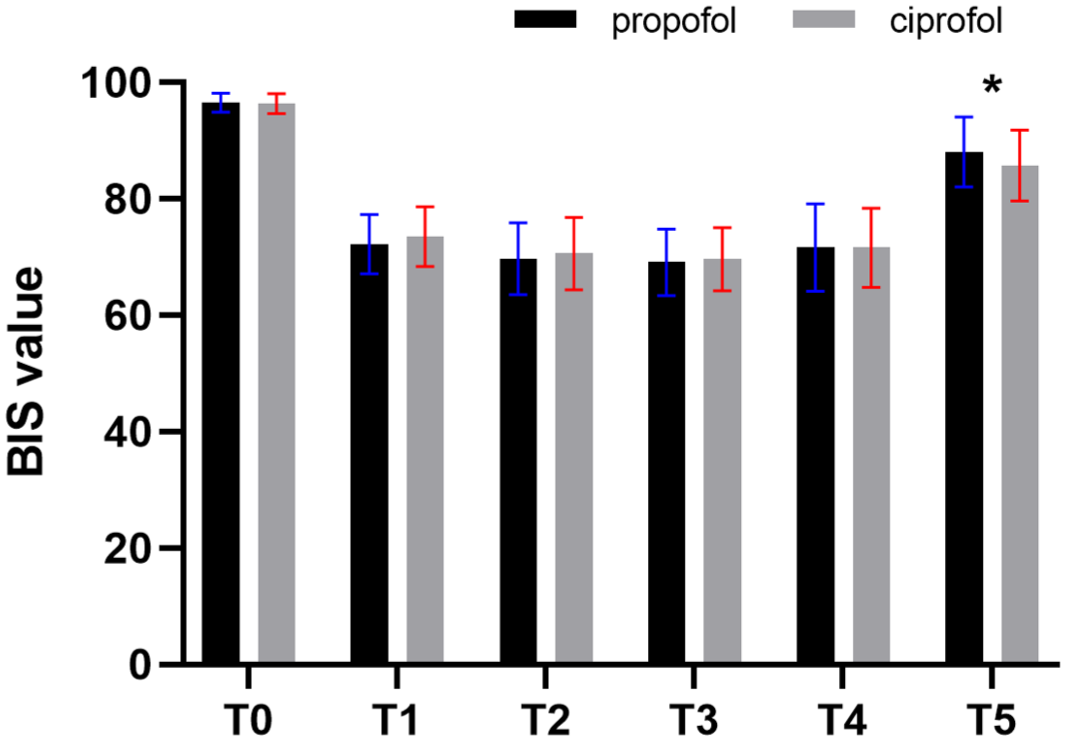
BIS values at different time points.
Postoperative outcomes
As shown in Table 3, both groups exhibited comparable rates of postoperative nausea and vomiting, dizziness, drowsiness, delirium, use of postoperative analgesia, and incidence of complications within 2 months following surgery (p > 0.05). Furthermore, satisfaction ratings from both patients and physicians showed no meaningful differences between groups (p > 0.05).
Postoperative-related conditions in both groups of patients.

Data are numbers (percentages) or medians (interquartile range).
Discussion
This study focused on the perioperative management of patients undergoing foot and ankle surgeries involving high thigh tourniquet use, under regional nerve blockade. Both ciprofol and propofol were effective for sedation in this context. However, ciprofol demonstrated a more favorable profile in terms of hemodynamic stability and a reduced incidence of specific adverse effects, particularly hypotension and injection-related pain.
Previous studies have suggested comparable sedative efficacy between ciprofol (0.4–0.5 mg/kg) and propofol (2.0 mg/kg) at clinically relevant doses. 16 In line with these findings, this trial employed a starting infusion rate of 0.5 mg/(kg·h) for ciprofol with a maintenance dose of 0.1 mg/(kg·h), and a starting rate of 2 mg/(kg·h) for propofol with a 0.5 mg/(kg·h) maintenance dose. Based on a prior report, these BIS values were consistent with Richmond Agitation-Sedation Scale (RASS) trends. 17 The depth of sedation, as assessed by BIS values, was maintained within a target range of 65–80, corresponding to a RASS score of −1 to −3, indicating light to moderate sedation. A BIS value above 80 generally indicates a state of wakefulness, 18 which may lead to intraoperative awareness, increased anxiety, and patient movement in response to the tourniquet. discomfort. This often results in a greater need for additional pain relief medications. However, an excessively low BIS value reflects deep sedation, which is closely associated with a higher risk of complications such as hypotension, respiratory depression, and postoperative delirium.19–22 In the present study, both groups of patients could achieve the target range of BIS values throughout the procedure. Here, both agents maintained BIS values within the desired range throughout surgery. However, 10 min after drug cessation, BIS levels were lower in the ciprofol group than in the propofol group (p < 0.05), mirroring results reported by Deng et al. 23 This may be due to ciprofol’s higher lipophilicity, which enhances adipose tissue accumulation and delays redistribution and clearance.9,16,24 There was no significant difference in recovery time between the two groups. This suggests that although BIS monitoring detected pharmacokinetic differences, these variations were not substantial enough to produce a clinically meaningful delay in emergence. However, the finding may remain clinically relevant: in practice, particularly for patients receiving prolonged infusions or those with conditions such as obesity or advanced age that may alter drug metabolism, clinicians should be mindful of the potential for delayed awakening when using ciprofol.
Tourniquet use remains a standard technique for reducing intraoperative bleeding but carries risks that increase with application time, including tourniquet-induced pain, reactive hypertension, ischemia-reperfusion injury, and neuromuscular damage.25–27 Tourniquet pain is characterized as dull, aching, or burning. The average duration of pain tolerance is 30 min.4,28 Thus, patients with expected tourniquet durations ⩾40 min were included in this study to ensure adequate observation of these effects. Tourniquet-induced hypertension, often occurring concurrently with pain, is also a common adverse effect of tourniquet application.28,29 Intraoperative body movements are predominantly triggered by discomfort related to tourniquet application. Numerous clinical investigations have shown that supplementing nerve block anesthesia with intravenous sedatives during foot and ankle surgeries can significantly reduce tourniquet-induced pain, alleviate anxiety, and enhance the overall surgical experience for patients.30–33 In this study, all participants received regional nerve blocks augmented by either propofol or ciprofol, in combination with low-dose opioids. Involuntary body movements were observed in 39.3% of patients in Group P and 38.7% in Group C. These episodes were effectively controlled through the administration of rescue doses of sufentanil, allowing over 98% of procedures to proceed to completion without the need for escalation. Nonetheless, conversion to general anesthesia was required in three patients, including one in Group P and two in Group C, due to severe intolerance to tourniquet-related discomfort. It is important to note that both groups showed a relatively high incidence of intraoperative rescue analgesia use. This is likely related to the unique nature of tourniquet-induced discomfort. While a well-executed nerve block can reliably eliminate incision-related pain, its ability to alleviate the centrally mediated and deep tissue discomfort caused by tourniquet application is comparatively limited. Hypertensive responses to tourniquet inflation were documented in approximately 25% of patients across both groups. Importantly, no cardiovascular complications occurred intraoperatively, indicating the relative safety of the sedation protocols used. Postoperative satisfaction levels, as reported by both patients and surgeons, were high and did not differ significantly between the two treatment groups.
A recent meta-analysis highlighted notable differences in the adverse effect profiles of ciprofol and propofol, particularly with respect to the incidence of injection-related pain, hypotension, and respiratory depression. 34 In the context of regional anesthesia, pain upon injection is a significant contributor to patient distress and anxiety. In the present study, ciprofol was associated with a markedly lower frequency of injection pain compared to propofol. This finding may be explained by the physicochemical properties of the oil-in-water ciprofol emulsion formulation, which enhances hydrophobicity and results in lower plasma concentrations.35,36 Many patients experience hypotension after sedation, which can lead to inadequate tissue perfusion and impaired organ function. These effects hinder postoperative recovery and increase the risk of cardiovascular and cerebrovascular complications.37,38 This study observed that intraoperative hypotension occurred less frequently in patients administered ciprofol than in those receiving propofol (p < 0.05), consistent with previous clinical observations. 12 As shown in Figure 2, MAP remained within an acceptable range in both groups, and the need for vasoactive medications did not differ significantly. This may be due to the inclusion of a relatively low-risk patient population. In comparison, in higher-risk patients who are more susceptible to hypotension, the influence of ciprofol on BP requires further investigation to be fully clarified. On the other hand, the incidence of respiratory depression (SpO2 < 95%) did not significantly differ between the two groups. This is likely attributable to the conservative, stepwise titration of sedatives and the maintenance of BIS values within the predefined target range of 65–80, thereby avoiding deep sedation and its associated respiratory risks.
Limitations
The study’s limitations include its single-center design and the relatively limited sample size, which may restrict generalizability. Moreover, the inclusion criteria focused on patients with lower surgical risk (ASA I–II), excluding those with comorbidities that may impact anesthetic response. Future research should expand on these findings by enrolling larger, more diverse patient populations, including those with higher perioperative risk. Finally, this study primarily used BIS as the measure of sedation depth and did not incorporate other clinical assessment tools, such as the RASS. Integrating clinical scoring scales in future research may provide additional insight into sedation quality and enhance the comprehensiveness of sedation evaluation.
Conclusion
In conclusion, both ciprofol and propofol, when administered under BIS-guided monitoring in combination with low-dose adjunctive analgesics, are effective in mitigating discomfort associated with tourniquet application during lower limb surgeries performed under nerve block. Ciprofol, in particular, offers enhanced hemodynamic stability and a more favorable adverse event profile, supporting its clinical potential as a sedative for use in this surgical context.
Supplemental Material
sj-doc-1-taw-10.1177_20420986251414580 – Supplemental material for Effect of ciprofol-assisted sedation on tourniquet-related responses in foot and ankle surgery: a randomized clinical trial
Supplemental material, sj-doc-1-taw-10.1177_20420986251414580 for Effect of ciprofol-assisted sedation on tourniquet-related responses in foot and ankle surgery: a randomized clinical trial by Yaoyang Ma, Bingqing Zhu, Shudong Wang, Danjun Lu, Wenlong Yan, Yi Yu, Ying Cao, Fang Kang and Juan Li in Therapeutic Advances in Drug Safety
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
