Abstract
Background:
In tissue regenerative trials, investigators operate in the intersection of routine clinical care and research. Often, clinical trials are the only option to introduce innovative treatments to disadvantaged populations in safety-net hospitals (SNHs). It is necessary to maintain a balance between efficient study conduct and patient safety. Social determinants play a role in medication adherence in clinical trials and chronic disease management that may increase risks if not managed correctly.
Objectives:
We aimed to assess the safety of patients in tissue regenerative clinical trials while examining underlying causes to develop proactive risk mitigation strategies for high-risk patients.
Design:
A single-center, retrospective study was conducted for clinical trials with tissue regenerative products for chronic wounds at an SNH.
Methods:
Data obtained from 186 subjects were analyzed retrospectively for correlation between social determinants of health and adverse events by using Spearman correlation and Kruskal–Wallis tests.
Results:
Wound healing was achieved in 41.94% of patients who received investigational products. Overall, the diabetic foot ulcer group was noted to have a higher prevalence of serious adverse events (SAEs; 22.8% of enrolled subjects) and adverse events (78.3% of enrolled subjects) as compared to the venous stasis ulcer group, with 12.4% of SAEs and 71.0% adverse events observed in the study population. Kruskal–Wallis test demonstrated statistically significant correlation between polypharmacy (⩾5 drugs) and a higher number of adverse events (p = 0.0016). A Spearman correlation test showed that a higher number of comorbidities was associated with a higher number of adverse events (p = 0.0007).
Conclusion:
These findings in polypharmacy and comorbidities being associated with a higher number of adverse events highlighted the importance of safety monitoring of patients with high disease burden in clinical trials. Understanding the frequency/types of adverse events can provide important insights for those conducting trials in a particular indication. In addition, monitoring can help to address social determinants that contribute to higher numbers of adverse events, and proactively address disease burden with appropriate medical management to minimize risks in tissue regenerative clinical trials.
Plain language summary
Why was this study done? People with chronic wounds, like diabetic foot ulcers and venous ulcers, often don’t have access to new treatments unless they join clinical trials, especially at hospitals that serve low-income populations. It’s important to make sure these treatments are safe, especially for patients who have other health problems or take many medications.
What did the researchers do? The researchers looked at data from 186 patients who took part in clinical trials using regenerative tissue products to treat chronic wounds. They wanted to find out if patients with more health problems or who took many medications had more side effects or complications during the study.
What did they find? The study found that patients with diabetic foot ulcers had more side effects than those with venous ulcers. People who took five or more medications or had several other medical conditions were more likely to have side effects. This means that certain patients may be at higher risk during these types of studies.
What does this mean? Doctors and researchers need to closely watch patients who have many health issues or take lots of medications when they are in clinical trials. Understanding who is at higher risk can help make these trials safer and improve care for vulnerable patients.
Introduction
The most common types of chronic wounds, diabetic foot ulcers (DFUs) and venous leg ulcers (VLUs), are a growing concern in healthcare due to their significant impact on both patients and the healthcare system. 1 Acute wounds typically resolve in 4–6 weeks, while chronic wounds persist after initial occurrence.2,3 Chronic wounds affect approximately 8.2 million Medicare beneficiaries (i.e., 2% of the US population), and are a significant burden on the healthcare system, which is estimated $28 billion each year. 4 These types of wounds are often difficult to treat and can lead to severe complications such as infection, amputation, and even death. The 5-year mortality rates for patients with chronic wounds can be comparable to those associated with certain types of cancer, which emphasizes the urgency of finding effective, evidence-based treatment options. 5
One of the key challenges in managing chronic wounds is the pain, odor, and reduced mobility that patients often experience, which can lead to social isolation and a decrease in quality of life. In this context, skin substitutes (e.g., human cells, tissues, and cellular and tissue-based products (HCT/Ps) and/or acellular, synthetic products) have emerged as an advanced therapy for chronic wounds that fail to show progress with standard care methods, such as various dressings, compression therapy, or wound debridement.6,7 Advanced therapies, such as the use of skin substitutes, are recommended for wounds that fail to decrease in size 4 weeks after initial onset. 8
Currently, there are 75 skin substitutes available, and this number is continuing to grow.8,9 Meanwhile, the healthcare system faces challenges in terms of selection, cost, and accessibility as each product comes with its own set of advantages and limitations, and the evidence supporting their use can be inconsistent, which makes it difficult to establish clear treatment guidelines for different types of chronic wounds.8,9 In addition, cost-effectiveness remains a critical issue, as most skin substitutes are expensive, and widespread adoption may be limited due to financial limitations.8,9
The Centers for Medicare & Medicaid Services (CMS), the federal agency overseeing major healthcare programs including Medicare, has introduced a significant policy change affecting HCT/Ps used in wound care. 10 Medicare, a government health insurance program for individuals 65 and older, now mandates proof of efficacy for HCT/Ps to qualify for reimbursement. This policy shift means that companies must conduct clinical trials to demonstrate the effectiveness of their products. 10
The upcoming Medicare policy for HCT/Ps is a substantial change aimed at ensuring that products used in wound care are both safe and effective. 10 This policy demands that manufacturers of HCT/Ps provide evidence from clinical trials to receive reimbursement coverage. The objective is to safeguard patient health by confirming that the products deliver the claimed benefits. 10
From the Food and Drug Administration’s (FDA’s) perspective, most HCT/Ps that meet specific criteria under 21CFR 1271.10 are regulated solely under Section 361 of the Public Health Service Act. This means they do not require premarket authorization or clinical trials. 11 These criteria include minimal manipulation of cells or tissues and homologous use. 12 However, with CMS’s new reimbursement requirements, companies producing HCT/Ps for wound care will need to navigate both the regulatory landscape and the new demands for clinical trial evidence.
Therefore, in the current regulatory landscape and with high costs for wound care due to complex management of chronic wounds, it is necessary to maintain a balance between efficient study conduct and patient safety.10–13 The leg ulcers are indicative of potentially chronic systemic issues that may increase risks posed to patients during clinical trials.14,15 The medical community places a major focus on the overall safety and welfare of patients. 16 Due to the experimental nature, the safety aspect is further emphasized during the conduct of clinical trials, where continuous monitoring of benefit–risk ratio changes as investigational product moves through the lifecycle, and well-being of research subjects are not only requirements established by competent regulatory authorities but also part of professional and ethical norms for physician investigators and clinical researchers. 16
The two most common types of leg ulcers are VLUs and DFUs.1,3 VLUs are resulting from venous insufficiency, due to venous reflux or blockage in the venous system.3,17 In addition, diabetic neuropathy, which is often accompanied by peripheral arterial disease, results in diabetic wounds on the lower extremities in many patients with diabetes. Also, there are other complications of diabetic wounds that can increase the possibility of infection, such as decreased mobility of the joints in the lower extremities and issues with blood circulation due to microvascular disease. Often, non-healing DFUs can lead to amputation in diabetic patients. These wounds in routine clinical care are treated with offloading and sharp debridement. In the past few decades, leg ulcers have been under investigation for many innovative, experimental biomedical products, which are currently being tested in clinical trials. 18
A safety-net hospital (SNH) is a type of medical facility in the United States that, by legal obligation and/or its mission, provides healthcare for patients regardless of their insurance status. Unlike many European countries, the United States does not have a policy of universal healthcare or the ability to pay.19,20 This legal mandate reinforces SNHs to be more inclusive and serve populations, which often are disadvantaged both socially and financially. These hospitals serve a proportionately higher number of uninsured, Medicaid, Medicare, Children’s Health Insurance Program (CHiP), low-income, and other vulnerable populations than their “non-safety net hospital” counterparts.19–21 SNHs are not defined by their ownership terms; they can be either publicly or privately owned.20,21 The mission of SNHs is to provide the best possible care for those who are discouraged from interacting with the healthcare system or seeking medical attention due to various circumstances, such as the burden of financial payments, lack of insurance plans/coverage, potential to lose their job/missing work/lost wages, or high-burden health conditions.20,21
In addition, patients at SNHs have a high burden of diseases and are prone to multiple comorbidities while facing severe societal issues that prevent them from receiving adequate healthcare. Clinical trials with investigational products that target these disease states are at high risk for introducing confounding factors to safety profiles that may disrupt the overall conduct of the trial. Due to these high-risk disease states, it is important to manage the overall care of patients involved in clinical research.
According to the World Health Organization (WHO), social determinants of health (SDoH) have been linked to increased health risks. 22 By understanding and analyzing those social factors, risk mitigation strategies can be developed to possibly prevent issues related to safety and mitigate risks.
The purpose of this study was to evaluate patterns of adverse events in DFUs and VLUs populations with investigational tissue regenerative products, analyze how SDoH correlate with the safety profile to determine which patients are more susceptible to having adverse events, and then develop risk and safety mitigation strategies for tissue regenerative clinical trials.
Methods
Study design and settings
A single-center, retrospective study was conducted of 186 patients randomized into 18 clinical trials with investigational tissue regenerative products at an SNH, which is a type of medical facility in the United States that, by legal obligation and its mission, provides healthcare for patients regardless of their insurance status. These clinical trials were similar in eligibility criteria, study design, study procedures, endpoints, and involved investigational products aimed at evaluating the efficacy and safety of these products for the treatment of DFUs and VLUs.
Data sources and measurements
The de-identified, aggregate dataset was assembled based on data obtained from all 18 randomized wound care clinical trials with tissue regenerative products, which were conducted in the department of surgery during the period of time from July 2007 to October 2024. All clinical trials were registered through a public trials registry by industry sponsors and received an institutional review board (IRB) approval prior to the study initiation. Informed consent from each research subject was obtained prior to any research procedures being performed.
Variables and study size
Specifically, data obtained from 186 patients were assessed retrospectively for the correlation of SDoH variables and adverse events (e.g., categorized by relatedness, seriousness, and expectedness). The study dropout rates (e.g., loss of follow-up for any reason or due to adverse event, withdrawal by investigator, or death) were also assessed, and the connection with SDoH was examined.
Patient demographics at the time of enrollment were evaluated, including gender (male/female), race (White, Black, or other), Hispanic ethnicity, weight (kg), height (m), body mass index (BMI), and age (years). Polypharmacy was defined as concomitant medication ⩾5, and included the following therapeutic indications: antiglycemics, antiplatelets, anticoagulants, antihypertensives, statins, non-opioid/opioid analgesics, antidepressants, antibiotics, etc. Comorbidities/pre-existing conditions were recorded.
Socioeconomic variables were evaluated, including primary language spoken (English, Spanish, or other), type of insurance (Medicare, Medicaid, private/other, or uninsured), median income of patient zip code, and distance traveled to visits (miles).
As part of standard clinical research operations at our site and as a method of maintaining metrics in real time, a database of serious adverse events (SAEs) and non-SAE events was maintained, documenting event start and end dates, the date the event was reported to the sponsor, and regulatory groups. Each safety event was assessed by categories of severity, expectedness, and relatedness to the study products (e.g., medical device) and/or study procedures. The aggregate dataset from 18 clinical trials was used in this retrospective analysis.
Each adverse event was classified and stratified by relationship to the investigational tissue regenerative product and study procedures (e.g., relevant for surgical procedures required by the study protocol only, not routine clinical care) to better understand the correlation between the type of event and exposure to the investigational product. An assessment of the expectedness of the event was performed based on the Instructions for Use or User Manual, which is in clinical trials with tissue regenerative study products classified as medical devices, and is equivalent to the Investigators Brochure utilized in trials with cell or tissue-based products classified as biologics.
According to regulatory and study protocol definitions, an adverse event is any undesirable experience associated with the use of a medical product in a patient. 23 The event is SAE and should be reported to FDA when the patient outcome is life-threatening, results in death, hospitalization (initial or prolonged), disability, congenital abnormality/birth defect, required intervention to prevent permanent impairment or damage (e.g., with medical devices); or other medically significant event that may jeopardize the patient and may require medical or surgical intervention (treatment) to prevent one of the other outcomes (e.g., significant blood abnormalities on laboratory tests, accidental finding on imaging tests, requiring immediate intervention, treatment in an emergency room).23,24
All other non-SAEs should be reported as part of continuing review per site organizational policies and procedures and captured and reported to the sponsor and IRBs in accordance with the study protocol, data safety monitoring plan, data safety monitoring board, if required, and site organizational policies and procedures. 24
We have compared the rate of adverse events for these two indications, and examined how safety signals relate to various SDoH, among them the ones which were investigated and selected by the WHO (e.g., income, education, housing security, access to healthcare services). 22
The adverse events were tracked by specific categories, ranked by seriousness, expectedness, and relatedness to the investigational products. The incidence rate of adverse events per subject randomized was monitored, and demographics were collected to capture and further assess factors related to SDoH. We also analyzed the type of adverse event that occurred based on the FDA safety reporting regulatory guidelines. 25 Once a patient was randomized to a specific treatment, the starting point of data collection was considered the subject enrolled in the study.
It was hypothesized that while conducting clinical trials at a tertiary care SNH, there were processes put in place to offset the burden of participating in trials, which may otherwise affect individuals struggling with SDoH. Adverse events were used to monitor the clinical trials’ safety profile and issues with adherence to study protocol and investigational product treatment regimens/study procedures due to overall subjects’ health concerns or attributed to SDoH. The following SDoH variables were collected and assessed in correlation with safety profile (e.g., rate of adverse events): race, gender, age, distance traveled to the clinical site, median household income by zip code, type of health insurance, primary language, etc.
Income was calculated by zip code associated with patients at the point the patient was randomized in the trial, and census data from 2022 were used to determine the median household income of that area. 26 Distance traveled calculations were made by using the subject’s address and calculating distance from their primary residence to the clinical site, where the study was conducted, as a consistent destination on the medical campus. Insurance was broken down into three categories based on the type. If an individual carried a government or state-serviced insurance plan, they fell under “government/state insurance,” if they had another form of insurance, they were categorized as “private insurance” coverage, and the last category, with the absence of insurance, was labeled as “underinsured or uninsured.”
BMI was divided into the following groups: BMI < 25, 25–29.99, and BMI ⩾ 30. According to the National Institutes of Health (NIH), a BMI below 18.5 was classified as underweight, a BMI between 18.5 and 24.9 is a normal weight, and an overweight individual has a BMI of 25–29.9. 27 In addition, obesity was defined as a BMI of 30 or above. 25
Statistical methods
Data obtained from 186 subjects were analyzed by using the SAS program, version 9.4 (SAS, Cary, North Carolina, USA). The distribution of Wilcoxon scores was examined, and Kruskal–Wallis tests were performed on data gathered to compare specific SDoH with the adverse events rate per subject enrolled. The Wilcoxon signed-rank test is a version of the dependent samples t test that can be performed on ordinal (ranked) data. When the sample size is greater than approximately 30, the Wilcoxon signed-rank statistic follows what is called the “z distribution.” The hypothesis testing of the Wilcoxon signed-rank test is based on knowing the critical z value for a 95% confidence interval (or a 5% level of significance, α = 0.05), which is z = 1.96 for a two-tailed test and directionality. Whenever a test is based on the normal distribution, the sample z value needed to be 1.96 or higher to reject the null hypothesis. Associations between social determinants and deviations were examined by utilizing the Spearman correlation test. 28
Spearman correlation coefficient (r) was utilized to assess relationships between variables. This statistical test is used for measuring a linear correlation. The Spearman coefficient is a number between (r) = −1 and (r) = 1 that measures the strength and direction of the relationship between two variables. 29 If Spearman correlation coefficient (r) = 0, there is no relationship between the variables, and no correlation is observed. If it is between 0 and 1, there is a positive correlation detected, which means that when one variable changes, the other variable changes in the same direction. If this coefficient is between 0 and −1, the correlation is negative, and when one variable changes, the other variable changes in the opposite direction. For example, when the Spearman correlation coefficient (r) > 0.5, a strong positive correlation between examined variables exists; if (r) is between 0.3 and 0.5, it is moderate; and between 0 and 0.3, a positive correlation is observed.28,29 In preparation of this manuscript, guidelines indicated on Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) checklist were followed (attached as a Supplemental Material). 30
Results
We analyzed retrospectively 18 clinical trials with investigational tissue regenerative products, in which 186 patients were randomized. For the demographics, 60.75% were female (Figure 1(a)); 73.12% were younger than 65 years old (Figure 1(b)). As shown in Figure 1(c), 38.17% of the patients were White, 41.4% were Black, and 20.43% identified as another race. In terms of primary languages, 91.94% spoke English, and 8.6% were non-English speaking (Figure 1(d)). As shown in Figure 2(a), 56.45% had a BMI of <25, 30.11% a BMI of 25–29.99, and the remainder of the patients’ BMI ⩾ 30. As demonstrated in Figure 2(b), 72.58% had fewer than three pre-existing conditions/comorbidities at the time of enrollment. Also, 63.44% of patients were taking five or more concomitant medications at the time of enrollment (Figure 2(c)).

Study population demographic variables: (a) study participant gender, (b) study participant age, (c) study participant race group (White, Black, and other), and (d) study participant primary language (English, Spanish, and other).

Study participants’ medical history at the time of enrollment: (a) study population BMI assessment (BMI < 25, 25–29.99, >30), (b) number of participants’ pre-existing conditions (comorbidities), and (c) number of concomitant medications study participants were taking at the time of enrollment.
Regarding primary outcomes for efficacy, wound healing was achieved in approximately 41.94% of research subjects, who received investigational products, and their target wound closed at the end of treatment phase, which was defined as period between randomization to received first study product dose/treatment/application to the last dose/application received in the trial (mean duration = 12.05 weeks for the length of treatment for analyzed 18 clinical trials). Remarkably, two patients with venous stasis ulcers, who received cell-based investigational products containing keratinocytes and fibroblasts, discontinued the investigational product due to progressive disease defined by an increase of ⩾50% in the ulcer surface area at any time of treatment as compared to baseline. These patients in follow-up were prescribed antibiotics, followed by standard of care four-layer compression therapy. Subsequently, one of these patients underwent a thorough surgical intervention and received autologous skin grafts bilaterally. Several patients were withdrawn by principal investigator during treatment phase in diabetic ulcer trials due to fast progression of their disease, and alternative FDA-approved skin substitute therapy was applied such as bioengineered bilayer living cell constructs composed of keratinocytes and fibroblasts (e.g., Apligraf) and Integra graft, which consists of a silicone layer for initial wound coverage and a collagen/glycosaminoglycan matrix that encourages the growth of new dermal tissue. In all of these cases, the target wounds did not achieve closure during the follow-up phase.
At the follow-up phase (mean duration = 11.77 weeks), only standard-of-care wound dressings and FDA-approved therapies (e.g., four-layer compression dressings and/or compression stockings for venous ulcers, offloading boots) were applied. The target wounds’ status was monitored in follow-up, which was defined as the period since the last dose/application of investigational products received to the study exits/last visit per study protocol. In follow-up, 50% of wounds have remained closed since healing was achieved initially, and the other 50% did not heal upon completion of the entire study. Noteworthy, only 1% of patients who had healed initially upon completion of the treatment phase had experienced wound recidivism when the wound reopened at the follow-up phase. The main reasons for wound recidivism were trauma to the lower extremity or non-adherence to prescribed follow-up standard-of-care therapy (e.g., compression stockings).
In terms of safety profile, this study revealed that the rates of adverse events and SAEs, regardless of whether they were DFUs or VLUs, were 1.31 and 0.3 per enrolled patient (Figure 3(a)). In the analyzed study population, 245 adverse events and 55 SAEs were detected, as summarized in Figure 3(b). Out of 55 SAEs observed, only 1 was deemed as “related” to the study product or research procedures (Figure 3(c)). The DFU group was noted to have a higher prevalence of SAEs (22.8% of enrolled subjects) and adverse events (78.3% of enrolled subjects) as compared to the VLU group, with 12.4% of SAEs and 71.0% adverse events observed in the study population. Overall, 41.82% of safety events for all studies analyzed in both VLU and DFU indications were considered as “serious” and “expected” (Figure 3(d)), leaving 58.18.4%% of all observed SAEs as “unexpected.” Therefore, further analysis is needed to examine patterns of safety events to determine contributing factors/causes and develop robust and efficient risk-management plans.
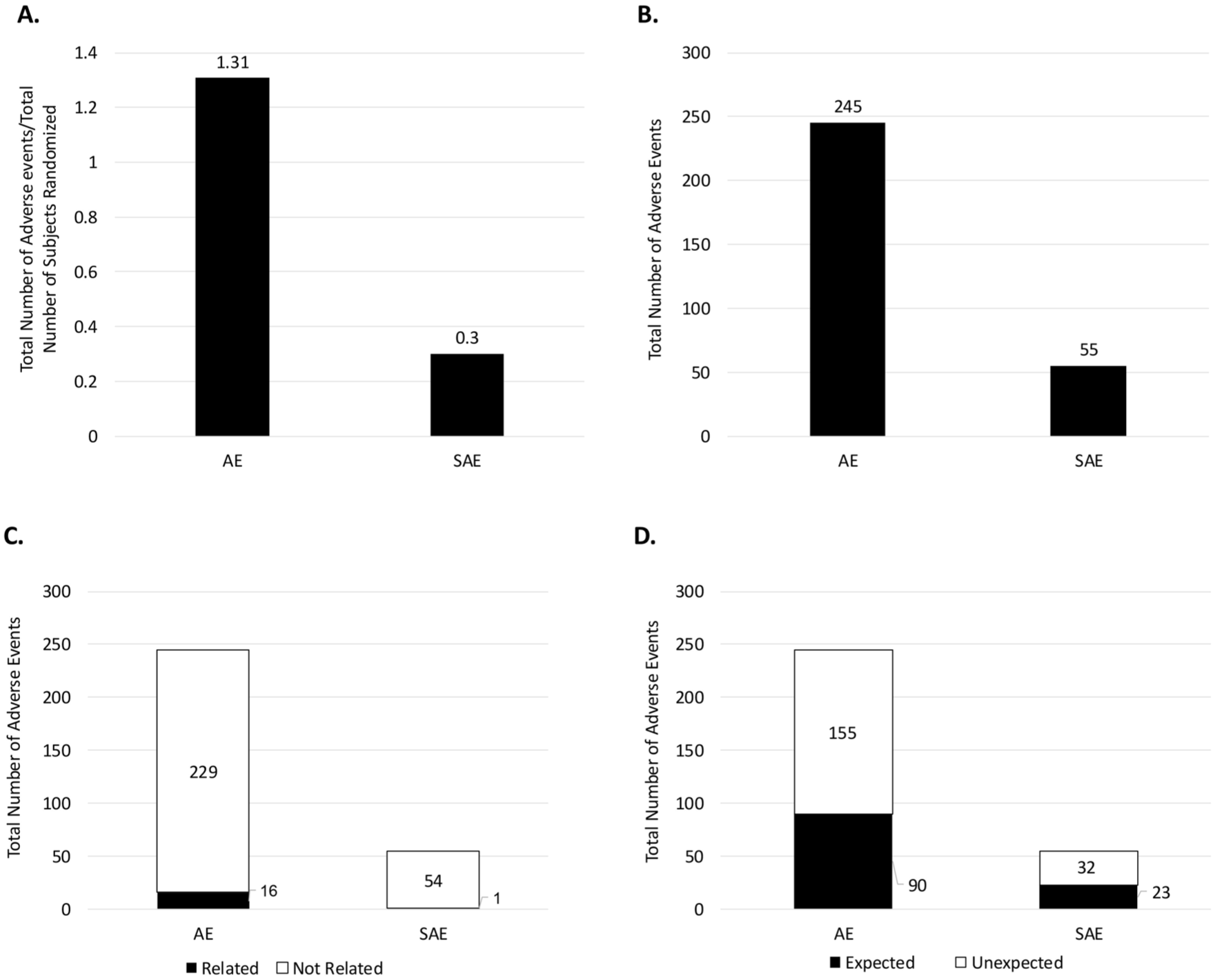
Adverse event assessments: (a) rate of non-serious and serious adverse events, (b) total number of non-serious and serious adverse events, (c) relatedness of the total number of non-serious and serious adverse events to the study product, and (d) expectedness of the total number of non-serious and serious adverse events.
While analyzing social determinants, there were statistically significant associations found between primary language spoken by clinical trial participants (p = 0.0047), male gender (p = 0.0358), and increased numbers of adverse events, as shown in Figure 4(a) and (c). The race of subjects was not statistically significant in correlation with the number of adverse events (p = 0.1342; Figure 4(b)). Noteworthy, the subjects with a high number of concomitant medications, defined as polypharmacy (⩾5 medications taken concurrently) at the enrollment point, were found to have a higher number of adverse events (p = 0.0049; Figure 4(d)).
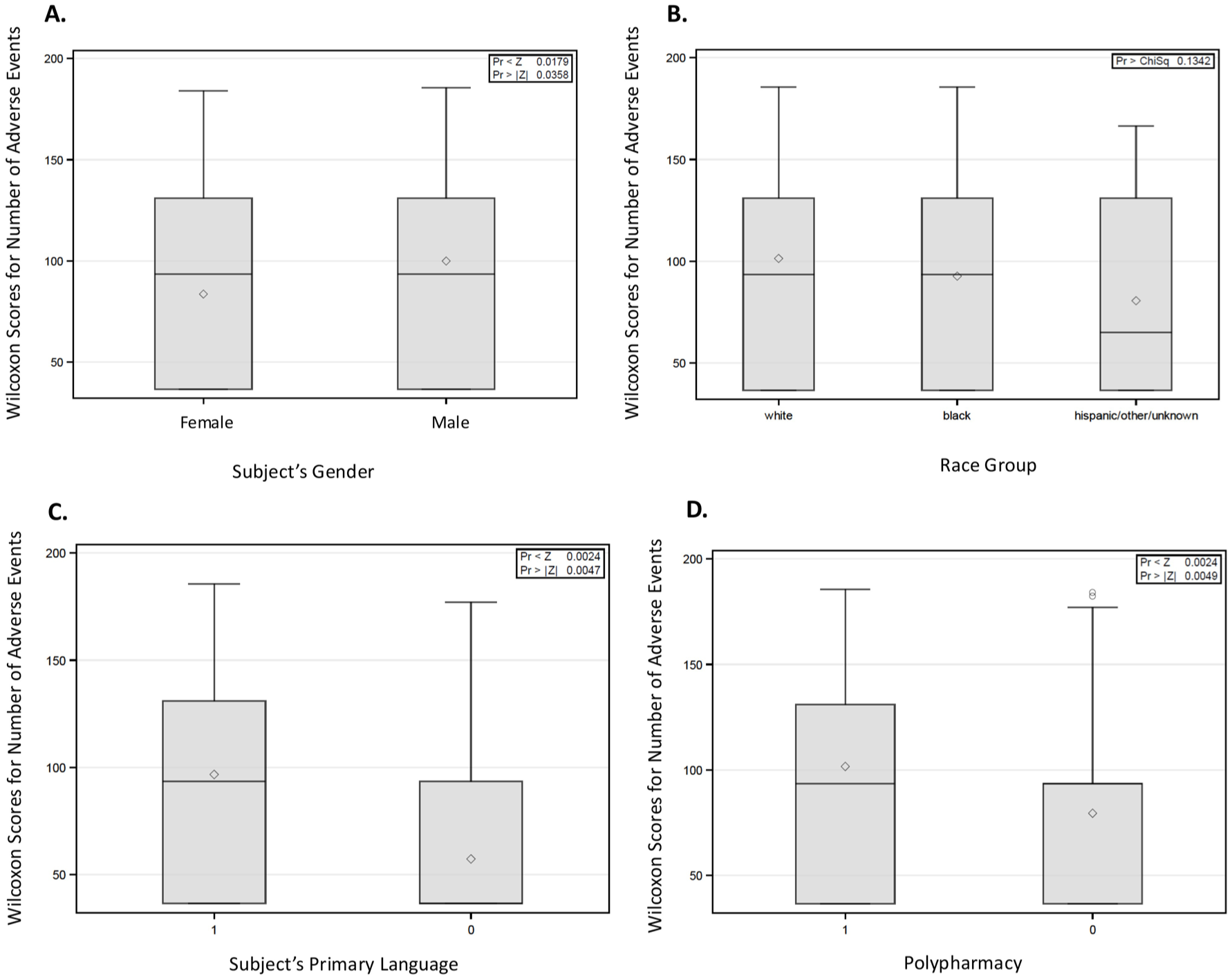
Wilcoxon score distributions for the number of adverse events based on (a) gender, (b) race group, (c) primary language, and (d) polypharmacy (number of concomitant medications at enrollment ⩾5).
In the Kruskal–Wallis test, the distribution of Wilcoxon scores for the number of adverse events between different groups by type of insurance carrier (e.g., government/state provided, private, uninsured) was not statistically significant (p = 0.1057; Figure 5(a)). The Wilcoxon scores for BMI were higher for uninsured subjects as compared to BMIs of clinical trial participants who had government/state or private insurance, and these results were statistically significant with p = 0.0240 (Figure 5(b)).
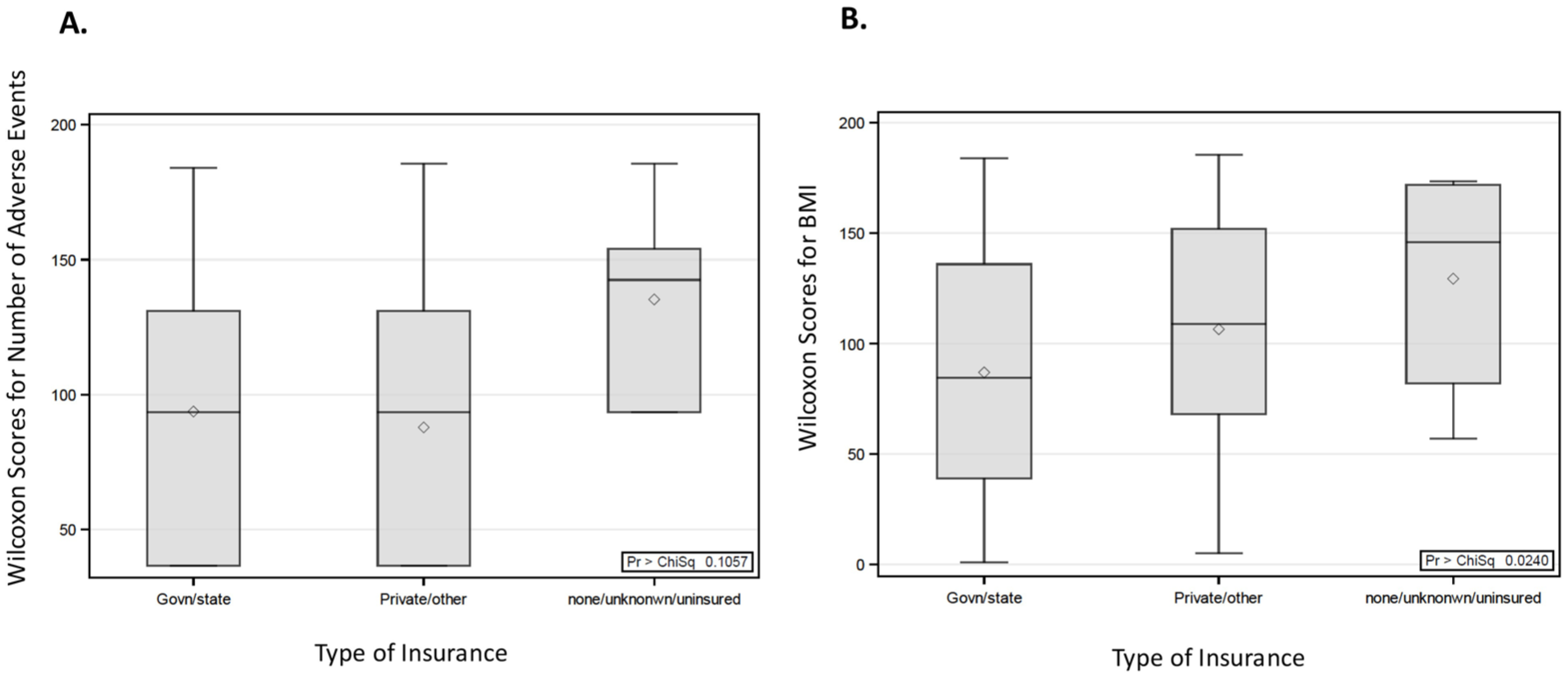
Wilcoxon score distributions for the number of adverse events based on type of insurance (a); Wilcoxon score distributions for BMI based on type of insurance (b).
In the Spearman correlation test, there was a weak correlation (r = 0.28) detected between a higher number of comorbidities at the time of enrollment and a higher number of adverse events, which was statistically significant (p = 0.0001).
A further analysis utilizing the Spearman correlation test demonstrated that socioeconomic status showed a weak negative association with statistical significance (r = −0.17; p = 0.0154) between median income and number of adverse events. Thus, subjects with higher median household income were less prone to experience an adverse event in the analyzed clinical trials.
In terms of retention of subjects in clinical trials and SDoH, in the Kruskal–Wallis test, there were no correlations observed between age (p = 0.1857), distance travelled to site (p = 0.7875), median household income (p = 0.3195), and comorbidities (p = 0.8029), when these variables were examined versus dropout status (Figure 6(a)–(c) and (e)). However, there was a statistically significant association between higher BMI and dropout status (p = 0.0304) as shown in Figure 6(d). In Fisher’s Exact test, there was no statistical significance detected when insurance carrier type and dropout status were examined (p = 0.8781). The findings of this project were partially presented at the 23rd International Society of Pharmacovigilance (ISoP) Annual Meeting. 31

Wilcoxon score distribution for dropout status based on (a) subject age, (b) distance traveled, (c) median household income, (d) BMI, and (e) number of comorbidities (pre-existing conditions) at the time of enrollment.
Discussion
Despite the prevalence and severity of chronic wounds, they remain difficult to manage. The development of skin substitutes in the 1980s revolutionized the treatment of chronic wounds. Since then, numerous skin substitutes have been implemented, each boasting its respective advantages and disadvantages. Vecin and Kirsner, in their recent review, summarized randomized clinical trials (RCTs) data to support the use of skin substitutes, while also suggesting future directions in tissue engineering. 32 According to these authors, much of the RCT data available regarding skin substitutes compared a substitute to the standard of care but there was a lack of abundance of literature comparing the efficacy of skin substitutes to one another. 32 Additional RCT data are necessary to compare substitutes and gain understanding of which substitute best suits a specific clinical picture, disease progression, and individual characteristics of patient. The data presented in this review can serve to guide clinicians in their decision-making. 32 However, while available skin substitutes show improvement of healing outcomes in several chronic wound types, they provide barriers such as cost and accessibility. The authors of this review also introduced several potential future directions in tissue engineering that may serve to create skin substitutes that are effective and mitigate these challenges. 32
One of the directions to pursue in tissue engineering includes the implementation of stem cells in skin substitutes due to their potential for accelerated wound closure, reduced scar formation, and regeneration of skin appendages that are lacking in current skin substitutes. 33 Stem cells have been shown to promote the secretion of cytokines and growth factors that stimulate angiogenesis and extracellular matrix remodeling in the wound bed. 34 Several types of stem cells, including bone marrow-derived mesenchymal stem cells, bone marrow-derived endothelial progenitor cells, hematopoietic stem cells, and adipose-derived stromal cells, have been studied for their potential use in wound healing.35–38 A recent study has compared the use of human neonatal stem cells (hNSCs), amniotic epithelial stem cells (AECs), and mesenchymal stem cells (MSCs) derived from the placenta, and results show promise in wound healing. The hNSCs, AECs, and MSCs were isolated from the placenta, differentiated into keratinocytes and fibroblasts, and mixed in plasma to create a skin substitute, which was transplanted onto a severe rat thermal wound. The stem cells successfully promoted wound healing through re-epithelialization and improved skin architecture. 39 A 2019 study investigated the effectiveness of various types of stem cells in regenerating the epidermis of a wound. Adipose-tissue-derived stem cells, dental pulp stem cells, Wharton’s jelly stem cells, and bone marrow stem cells were placed on top of a dermal substitute, and parameters such as epidermal differentiation, matrix synthesis, and immunogenicity through HLA molecule expression were assessed. Of the various types of stem cells, Wharton’s jelly stem cells showed the greatest potential due to their epidermal differentiation and low immunogenicity. 40 While the use of stem cells provides an exciting potential new modality, the use of stem cells presents challenges such as identifying the optimal source of stem cells, refining processing and administration, elucidating the reprogramming process of stem cells, as well as future implications of stem cell-derived skin, such as immunogenicity and tumorigenicity. 41
Numerous studies have been conducted and demonstrated that mesenchymal stromal cells (MSCs) can induce re-epithelization and angiogenesis, decrease inflammation, contribute to immunological modulation, and promote DFUs healing, making them a promising approach to treat DFUs.33–41 Despite progress made, several authors have demonstrated that MSC therapy alone does not lead to complete restoration of skin parameters in diabetic foot patients within a 3-year follow-up period. 42 According to Maksimova et al., MSCs may present an important adjuvant to the therapy; however, other novel approaches are required to achieve better clinical outcomes. 42
However, recent experiences have accumulated numerous reports of adverse events and side effects associated with MSC therapy.43,44 Furthermore, the strategies and methods of MSC therapy did not change significantly in recent decades despite the clinical impact and awareness of potential complications.43,44 Cell processing and cell culture could dramatically influence cell population profile, change protein expression, and cell differentiation, paving the way for future negative effects. Long-term cell culture led to the accumulation of chromosomal abnormalities.43,44 Overdosed antibiotics in culture media enhanced the risk of mycoplasma contamination. Clinical trials reported thromboembolism and fibrosis as the most common adverse events of MSC therapy. 44 Their delayed manifestation generally depends on the patient’s individual phenotype and requires specific awareness during the clinical trials conducted with obligatory disclosure in the patient’s informed consents. 44
The ethical considerations regarding the utilization of stem cells as part of regenerative therapy were prompted by issues related to the Macchiarini trachea transplant case. Dr. Paolo Macchiarini gained international attention in 2011 when he performed the world’s first synthetic windpipe transplant at Sweden’s Karolinska University Hospital. 45 The procedure, which involved using a synthetic scaffold seeded with stem cells, was hailed as a potential breakthrough in regenerative medicine. However, subsequent surgeries on at least eight patients across Sweden, Russia, and the United States led to severe complications and patient deaths. In Sweden, three patients died following the procedures, though the deaths were not directly linked to the surgeries. 45
In 2022, Macchiarini was convicted by a Swedish court of causing bodily harm during the experimental surgeries, with all six papers published in high-profile scientific and medical journals, including the paper authored by him in the Lancet journal in 2011, ordered to be retrieved for fraudulent data and scientific misconduct. 45 The court found that, while two of the patients were critically ill and the surgeries could be justified, the third patient did not meet the eligibility criteria for such an experimental procedure. Initially, he received a suspended sentence, but in 2023, the Svea Court of Appeal increased his sentence to 2.5 years in prison, citing that he acted with indifference to the risks involved. 45 Macchiarini’s case underscores the critical importance of ethical standards, transparency, and rigorous peer review in medical research and practice. His actions have prompted widespread reforms in medical oversight and have served as a cautionary tale about the dangers of unchecked ambition in the medical field.
Thus, further research in diverse study populations is warranted in establishing stem cell-based skin substitutes as a viable treatment for chronic wounds. Diverse patient populations provide a wide range of social, economic, cultural, and personal disparities that are crucial to the collection of data in clinical research to make recommendations that are generalizable to the real-world population.
Investigators in clinical trials face challenges of addressing SDoH. Assessing them in a systematic way can help to promote the safety and well-being of subjects enrolled in clinical trials with innovative tissue regenerative products, and also prevent exacerbation of their pre-existing condition.
Socioeconomic data are helpful to potentially improve the ability of a clinical trial program to assess efficacy and safety outcomes of new products in diverse populations. Barriers to access healthcare systems or the ability to participate in clinical trials need to be addressed, and a systematic review of site data can provide insights into the specific needs of different groups of patients. As a clinical site, we have a small percentage, 8.6%, of non-English speaking patients enrolled in analyzed clinical trials. However, it is an important aspect to examine the correlation with adverse events. The language barrier was pointed out by Clark et al. as an issue in clinical trials. 46 The association of adverse events and patients’ primary language was relevant, and it indicated that health literacy and education need to be addressed upfront for trial participants. 46
Monitoring of safety profiles in clinical trials and increasing numbers of adverse events are of particular concern due to the investigational nature and unknown risks. It is important to evaluate medication adherence in the context of SDoH as it can give insights into the overall status of patient health as pointed out previously by Wilder et al. 47 Drug–drug interactions are primary concern, when examining specific SDoH, managing potential and actual adverse events in a population that is approximately 70% defined by polypharmacy (⩾5 drugs taken concurrently; Figure 2(c)). Physician investigators must reconcile concomitant medications not only at the point of enrollment in the clinical trials but also at execution and follow-up phases. As pointed out by Hung et al., identifying unnecessary medication and reconciling prescription lists can help to avoid potential drug–drug interactions, reduce medication errors, and prevent adverse events. 48
As described by Beyene et al., comorbidities (e.g., diabetes, hyperlipidemia) can significantly delay wound healing. 14 In our project, out of 55 SAEs observed in 18 examined clinical trials with tissue regenerative products, only 1 was deemed as “related” to the study product or research procedures (Figure 3(c)). However, in the Spearman correlation test, a correlation was detected between a higher number of comorbidities and a higher number of adverse events, which was statistically significant (p = 0.0001). This observation highlighted an increased need for early identification of subjects with systemic health issues, multiple comorbidities, and the importance of medical management overall to improve the safety in tissue regenerative clinical trials, and avoid worsening of pre-existing diseases.
Insurance status is a major factor in individuals experiencing health disparities and is an important metric in providing equitable care, particularly relevant for individuals included in clinical trials. 49 There was no significant correlation between insurance and adverse events or dropout status, as shown in Figure 5(a) and through Fisher’s Exact test. It can be a potential indicator that at our site, we have appropriate resources and management of populations across different insurance types.
According to the NIH, an increased BMI has an adverse relationship with health and insurance status. 27 In the Kruskal–Wallis test, we observed that the Wilcoxon scores for BMI were higher for uninsured subjects as compared to BMIs of subjects who had government/state or private insurance, and these results were statistically significant with p = 0.0240 (Figure 5(b)). Therefore, a holistic approach to improve patients’ health and decrease BMI, which is associated with multiple comorbidities and poorer health prognosis, is needed.
Subject retention in clinical trials is very important both for efficacy and safety assessments. When various SDoH were examined versus dropout status, there was a statistically significant association detected between higher BMI and dropout status (p = 0.0304). Other social aspects (e.g., distance traveled, median household income, insurance type) were not associated with subjects’ dropout, suggesting that these issues were managed by the research team and administrative personnel at an SNH, and perhaps better subject retention was attributed to existing social programs. However, further studies are needed to explore SDoH and subjects’ retention in clinical trials.
Limitations
We have conducted a retrospective analysis on the safety and SDoH at a single, safety-net tertiary care hospital with some characteristics and practices that can be specific to our routine clinical care and clinical research practices. Therefore, large prospective studies at various sites in different settings (e.g., academic medical centers, rural clinics, private practices), with various patient populations, research practices are needed to further examine observed correlations and trends, increase the quality of research conducted, and improve safety profiles in clinical trials with tissue regenerative investigational products.
Conclusion
While conducting clinical trials, an understanding of the severity, frequency, and types of adverse events can help with the prediction of the safety profile and risk management in tissue regenerative clinical trials with specific wound etiology.
An understanding of the frequency and types of adverse events can provide an expectation for those conducting trials in a particular indication, for instance, a larger number of SAEs per patient on average can be expected for patients with DFUs, and these events will be more diverse as compared with VLU patients.
Polypharmacy and comorbidities were associated with a higher number of adverse events, which highlighted the importance of a holistic approach to medical management of patients’ health, and more focused, risk-based safety monitoring of patients with high disease burden in clinical trials. Male gender may preclude individuals to an increased number of adverse events and medical complications. Higher income had a significant, but weak correlation with less number of adverse events for participants of clinical trials with tissue regenerative products.
Safety monitoring can help to address social determinants that contribute to higher numbers of adverse events, and proactively address disease burden with appropriate medical management to minimize risks in tissue regenerative clinical trials.
Supplemental Material
sj-docx-1-taw-10.1177_20420986251385861 – Supplemental material for Safety and risk management in clinical trials for chronic wounds with tissue regenerative products
Supplemental material, sj-docx-1-taw-10.1177_20420986251385861 for Safety and risk management in clinical trials for chronic wounds with tissue regenerative products by Marina Alexandra Malikova, Connor Michael Roddy, Nolan Patrick Joyce and Katherine Nicole Cilley in Therapeutic Advances in Drug Safety
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
