Abstract
Background:
Pharmacovigilance is essential for ensuring patient safety, and hospital pharmacists play a key role in this system. However, their knowledge, attitudes, and practices (KAP) remain understudied in China.
Objectives:
To evaluate the KAP of hospital pharmacists toward pharmacovigilance in China.
Design:
Nationwide, cross-sectional study.
Methods:
This study evaluated the KAP of 9724 hospital pharmacists from 2145 hospitals across all 31 provinces of mainland China using a structured questionnaire and a multi-stage sampling approach. Descriptive statistics were used to summarize the KAP of the participants, and multivariable logistic regression was applied to explore the influencing factors of high KAP performance.
Results:
The proportions of respondents achieving high performance (score ⩾4 out of a maximum of 5) in knowledge, attitudes, and practices were 22.2%, 44.2%, and 68.0%, respectively, with significant correlations between these dimensions (p < 0.001). Misconceptions were prevalent, with 50.1% of pharmacists incorrectly attributing pharmacovigilance responsibility to medical institutions instead of marketing authorization holders (MAHs), and 36.2% misunderstanding pharmacovigilance as solely adverse drug reaction (ADR) reporting. A total of 95.5% of respondents expressed a willingness to participate in pharmacovigilance training. The reporting of ADEs/ADRs, medication errors, and drug quality problems to regulatory authorities is well-established nationwide, but 27.8% infrequently or never reported serious ADRs to pharmaceutical companies. Higher educational levels, hospital tiers, and leadership roles positively influenced knowledge and attitude, while male pharmacists showed superior practices compared to females. Clinical pharmacists and team leaders outperformed dispensing pharmacists in all KAP dimensions.
Conclusion:
Our study reveals a significant scope for enhancement in the KAP of hospital pharmacists pertaining to pharmacovigilance in China. Specifically, there is a critical need to improve the understanding of pharmacovigilance’s scope and responsibilities, and fostering stronger collaborative efforts between medical institutions and MAH is imperative, particularly within targeted pharmacist groups.
Plain language summary
Why is this study important?
Ensuring the safety of medicines is essential for protecting patients. Hospital pharmacists play a key role in identifying and reporting medicine-related safety issues. However, little is known about their knowledge, attitudes, and practices (KAP) in China.
We surveyed 9,724 hospital pharmacists from 2,145 hospitals across China to understand their KAP related to drug safety monitoring (pharmacovigilance).
What did we find?
• Many pharmacists had knowledge gaps:
○ About half mistakenly believed that hospitals—not pharmaceutical companies—were mainly responsible for pharmacovigilance.
○ Over one-third thought pharmacovigilance only involved reporting adverse drug reactions (ADRs), while in reality, it covers a broader range of medication safety issues.
• Nearly all pharmacists (95.5%) were willing to receive further training on pharmacovigilance.
• While many were actively reporting safety issues to regulatory authorities, some pharmacists did not consistently report serious ADRs to pharmaceutical companies.
• A pharmacist’s education level, hospital type, and job position influenced their knowledge and engagement with pharmacovigilance.
What does this mean?
Our findings suggest a clear need for:
• Better and targeted training for hospital pharmacists on pharmacovigilance.
• Stronger collaboration between hospitals and pharmaceutical companies to ensure safer medication use in China.
Introduction
Pharmacovigilance (PV) is defined by the World Health Organization as the “science and activities relating to the detection, assessment, understanding, and prevention of adverse effects or any other drug-related problems.”1,2 Pharmacovigilance encompasses activities such as assessing adverse drug reactions (ADRs), evaluating risk management plans, periodic safety update reports, and post-approval safety studies. 3 Pharmacovigilance systems are crucial for ensuring drug safety, particularly in analyzing ADRs and identifying safety signals. They safeguard patient safety and engage regulators, pharmaceutical companies, patients, healthcare professionals (HCPs), and pharmacists. In many countries, hospital pharmacists are vital to sustaining and overseeing ongoing pharmacovigilance initiatives.4,5 The revised “Drug Administration Law of the People’s Republic of China” 6 in 2019 specified the principles and objectives for the development of a robust pharmacovigilance framework, highlighting the critical role of hospital pharmacists in ensuring drug safety.
Effective pharmacovigilance requires collaboration among healthcare institutions, drug users, and marketing authorization holders (MAHs), supported by a robust technical system. In China, this system relies mainly on the passive surveillance of the China Adverse Drug Reaction Monitoring System,7–13 supplemented by active surveillance from the China Sentinel Hospital Alliance 14 and the China Hospital Pharmacovigilance System.7,12,14,15
In addition to monitoring adverse reactions, efforts are made to report medication errors. In September 2012, the Medication Safety Panel in the International Network for the Rational Use of Drugs (INRUD) established a National Monitoring Network for Clinical Safe Medication, enabling ME reporting, primarily by pharmacists (79.20%). 16 However, the lack of a comprehensive reporting system hampers the analysis and prevention of other medication-related risks, including natural (e.g., lack of efficacy, quality defects) and artificial risks (e.g., misuse, abuse).
Pharmacists are at the forefront of pharmacovigilance, playing crucial roles in the prevention, identification, documentation, and reporting of drug-related safety issues. In China, prior studies have assessed the knowledge, attitudes, and practices (KAP) related to pharmacovigilance, but with notable limitations. For example, a national survey of HCPs largely focused on ADR reporting and did not assess broader aspects of pharmacovigilance such as medication errors or off-label use. 11 Other studies were confined to specific regions—such as Yunnan Province, which compared HCPs with the general public, or Shanxi Province, which targeted public perceptions—and primarily examined ADR reporting behaviors.17,18 Earlier research in northern China also indicated very low ADR reporting rates among pharmacists over a decade ago. 19 To address these gaps, we conducted a nationwide cross-sectional survey targeting hospital pharmacists, aiming to provide a more comprehensive understanding of their KAP toward the broader concept of pharmacovigilance.
Materials and methods
Study design, settings, and participants
This was a nationwide, cross-sectional, questionnaire-based study conducted among hospital pharmacists across China. Eligible participants included pharmacists employed in public, private, and university-affiliated hospitals situated within mainland China. Between January 25th and January 30th, 2021, a closed electronic survey was administered online via the Questionnaire Star platform, with voluntary participation and no incentives provided. This study employed a multi-stage sampling approach. In the first stage, all provincial-level administrative regions in Mainland China were included in the survey scope. In the second stage, questionnaires were distributed to each province via the organizational networks of several academic societies, such as the Chinese Pharmacists Association, utilizing a convenience sampling method. To avoid duplicate data, each respondent was allowed to complete the questionnaire only once. The reporting of this study conforms to the Checklist for Reporting Results of Internet E-Surveys (CHERRIES) statement, 20 and the reporting checklist is provided in Supplemental Material 1.
Questionnaire development and pilot study
The questionnaire was developed based on the KAP model, which is widely used to assess HCPs’ behavior and decision-making,21,22 and was informed by a comprehensive review of existing literature.23–31 Its content and structure were rigorously evaluated by six experts in the subject area. Based on expert panel discussions, each specific question was categorized under one of the three primary dimensions of the KAP, which were further broken down into secondary dimensions to capture specific thematic areas: four for knowledge (concept, scope, responsibility, and surveillance of pharmacovigilance), two for attitude (responsibility and training), and four for practice (drug quality issues, adverse drug effects [ADEs]/ADRs, medication errors, and medication safety hazards; see details in Figure 4). Subsequently, a two-round Delphi method involving 20 experts was conducted to evaluate the contribution of each question item to its corresponding dimension, enabling weighted scoring with a maximum of 5 points assigned to both primary and secondary dimensions. To ascertain its reliability and validity, the questionnaire underwent assessment by two experienced researchers specializing in epidemiology and statistics. Furthermore, a preliminary pilot study was executed to initially evaluate the questionnaire prior to the main data collection phase. This pilot study involved 189 pharmacists from various hospitals. On average, completion of the questionnaire took approximately 10–15 min. Based on the feedback received, minor modifications were made to the wording of certain questions. Data from the pilot study were not included in the final analysis of the study results.
The finalized questionnaire consists of 42 questions, organized into 4 distinct sections. The initial section gathered demographic information of the respondents, including sex, education, job position, and years of experience, among other details. The subsequent three sections were dedicated to assessing KAP, respectively. To ensure validity, the questionnaire included control questions, with contradictory responses leading to disqualification.
The instrument incorporated various question types, including degree, frequency, and choice questions. A 5-point Likert scale (5 = strongly agree, 4 = partly agree, 3 = neutral, 2 = partly disagree, 1 = strongly disagree) was used for degree questions. The answers to the frequency questions were always (5 points), often (4 points), sometimes (3 points), occasionally (2 points), or never (1 point). Choice questions allowed for single or multiple selections. Additionally, the questionnaire concluded with an open-ended query, inviting participants to offer suggestions on pharmacovigilance. The complete questionnaire and the background of the experts involved in the Delphi process are provided in the Supplemental Material 2 for reference.
Statistical analysis
We assessed questionnaire reliability using Cronbach’s Alpha and analyzed validity with Kaiser-Meyer-Olkin (KMO) and Bartlett tests.32,33 Descriptive statistics summarized participants’ demographics and their KAP toward pharmacovigilance. Qualitative data were expressed as frequencies and proportions (%), while quantitative data were summarized as means and standard deviations (mean ± SD) for normal distributions, and medians with interquartile ranges (median, IQR) for non-normally distributed data. We used Pearson’s or Spearman’s correlation tests to assess interrelations among KAP scores. A multivariable logistic regression model using the direct entry method for covariate selection was applied to investigate determinants of high KAP levels (score ⩾4), calculating odds ratios (ORs) with 95% confidence intervals (CIs) for these associations. All statistical analyses were two-sided, with p values <0.05 indicating significance. Data analysis was performed using SAS 9.4 (SAS Institute Inc., Cary, NC, USA) and R 4.0.5 (R is an open-source programming language and software environment for statistical computing and graphics, The R Foundation for Statistical Computing, https://www.R-project.org/).
Results
Demographics of respondents
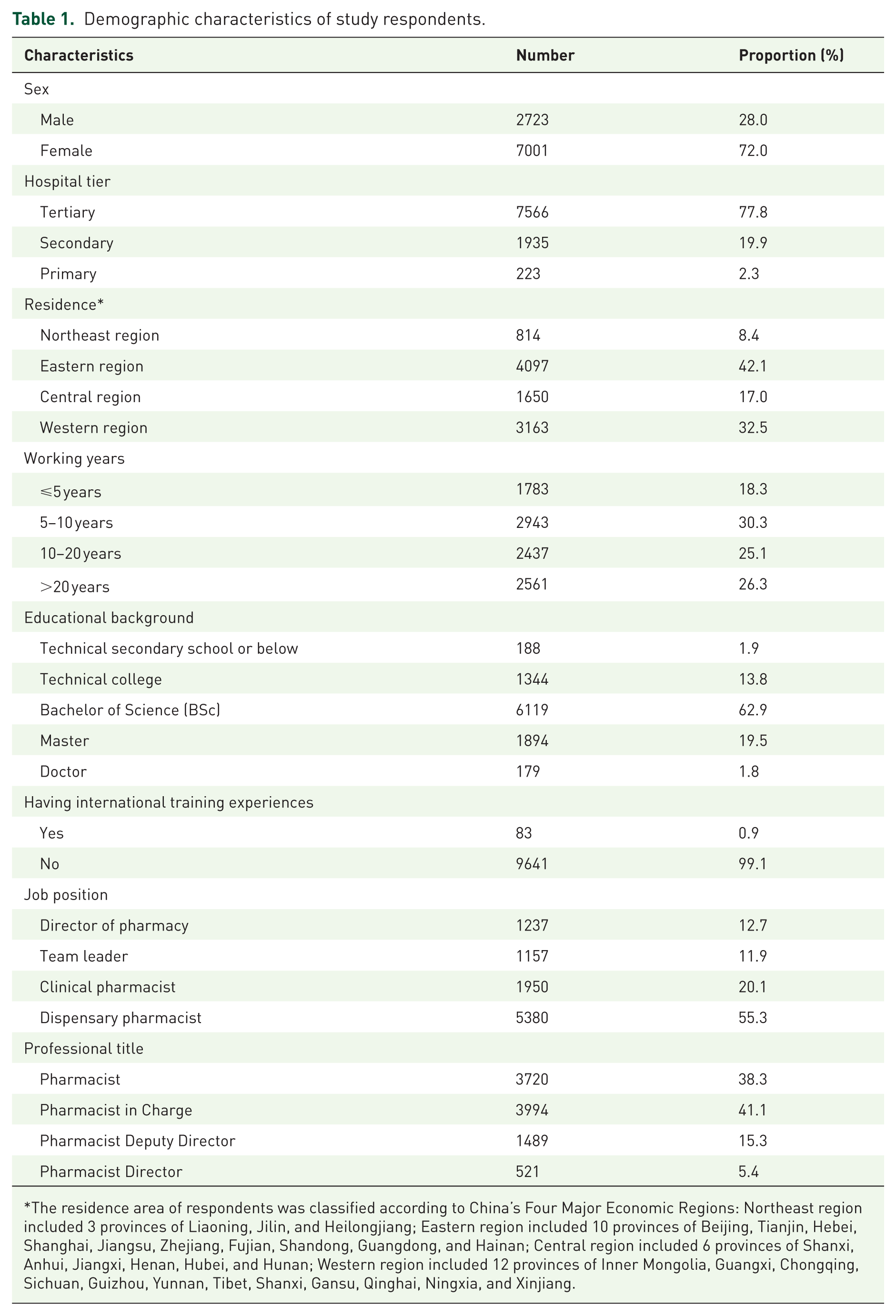
The survey elicited responses from 2145 hospitals across all 31 provinces, autonomous regions, and municipalities of mainland China, encompassing 188 cities. Among these, 1253 were tertiary hospitals (58.4%), 725 secondary hospitals (33.8%), and 167 primary hospitals (7.8%). From the 10,001 questionnaires collected, 9724 were deemed valid and included in our study, resulting in an effective response rate of 97.2%. The spatial distribution of respondents is shown in Figure 1.

Geographic distribution of study respondents.
Of the 9724 pharmacists included, the majority were female (72.0%) and from tertiary hospitals (77.8%). Pharmacists from the Eastern Region of China constituted the largest group (42.1%), followed by those from the Western (32.5%), Central (17.0%), and Northeastern regions (8.4%). Approximately one-third (30.3%) had 5–10 years of work experience, while more than half, accounting for 51.4%, had over 10 years of experience. The pharmacists predominantly held a Bachelor of Science degree (62.9%), were mostly dispensary pharmacists (55.3%) and with intermediate professional titles (Pharmacist in Charge, 41.1%). Very few had international training experience (0.9%). Detailed demographic information is presented in Table 1.
Demographic characteristics of study respondents.
The residence area of respondents was classified according to China’s Four Major Economic Regions: Northeast region included 3 provinces of Liaoning, Jilin, and Heilongjiang; Eastern region included 10 provinces of Beijing, Tianjin, Hebei, Shanghai, Jiangsu, Zhejiang, Fujian, Shandong, Guangdong, and Hainan; Central region included 6 provinces of Shanxi, Anhui, Jiangxi, Henan, Hubei, and Hunan; Western region included 12 provinces of Inner Mongolia, Guangxi, Chongqing, Sichuan, Guizhou, Yunnan, Tibet, Shanxi, Gansu, Qinghai, Ningxia, and Xinjiang.
Reliability and validity of the study questionnaire
The Cronbach’s Alpha coefficient of the questionnaire was 0.846, signifying a high level of reliability. In the KMO and Bartlett tests, the KMO measure was 0.864, indicating substantial correlation among the variables designed in the questionnaire. Furthermore, Bartlett’s test of sphericity yielded a significant result (p < 0.001), indicating compatibility with factor analysis and demonstrating strong structural validity.
Overall KAP at the primary dimension
The scores for KAP among hospital pharmacists concerning pharmacovigilance are presented in Figure 2, in both the first and second dimensions. In the primary dimension, the overall median scores (IQR) for knowledge, attitude, and practice were 3.74 (3.52–3.97), 3.92 (3.60–4.29), and 4.08 (3.77–4.81) out of a maximum of 5, respectively. The proportions of respondents achieving high performance (score ⩾4) in knowledge, attitude, and practice were 22.2%, 44.2%, and 68.0%, respectively. This data indicates that while the practice aspect shows a reasonable level of proficiency, there is room for enhancement in the pharmacists’ attitudes and especially knowledge regarding pharmacovigilance.

Scores of knowledge, attitude, and practice of hospital pharmacists regarding pharmacovigilance. (a) First dimension. (b) Second dimension of knowledge. (c) Second dimension of attitude. (d) Second dimension of practice.
Statistically positive associations were found between knowledge, attitude, and practice. The Pearson correlation coefficient between knowledge and attitude was 0.414, between knowledge and practice 0.274, and between attitude and practice 0.635, with all p values <0.001 (Figure 3). The data suggest that practice was more closely associated with attitude than with knowledge.

Scatter and density plots of the correlation between knowledge, attitude, and practice scores.
Responses to knowledge questions
In the second dimension of knowledge, the rankings of scores (median, IQR) were as follows: “Knowledge of Surveillance” (3.95, 3.58–4.21) > “Knowledge of Scope” (3.83, 3.09–4.61) > “Knowledge of Concept” (3.69, 3.58–4.12) > “Knowledge of Responsibility” (3.56, 3.25–3.92). Figure 4 shows the detailed questions in this aspect. The survey revealed that while 91.1% of pharmacists correctly understood the concept of pharmacovigilance, many found it relatively new, with only 37.8% familiar before the 2019 revision of the Drug Administration Law.
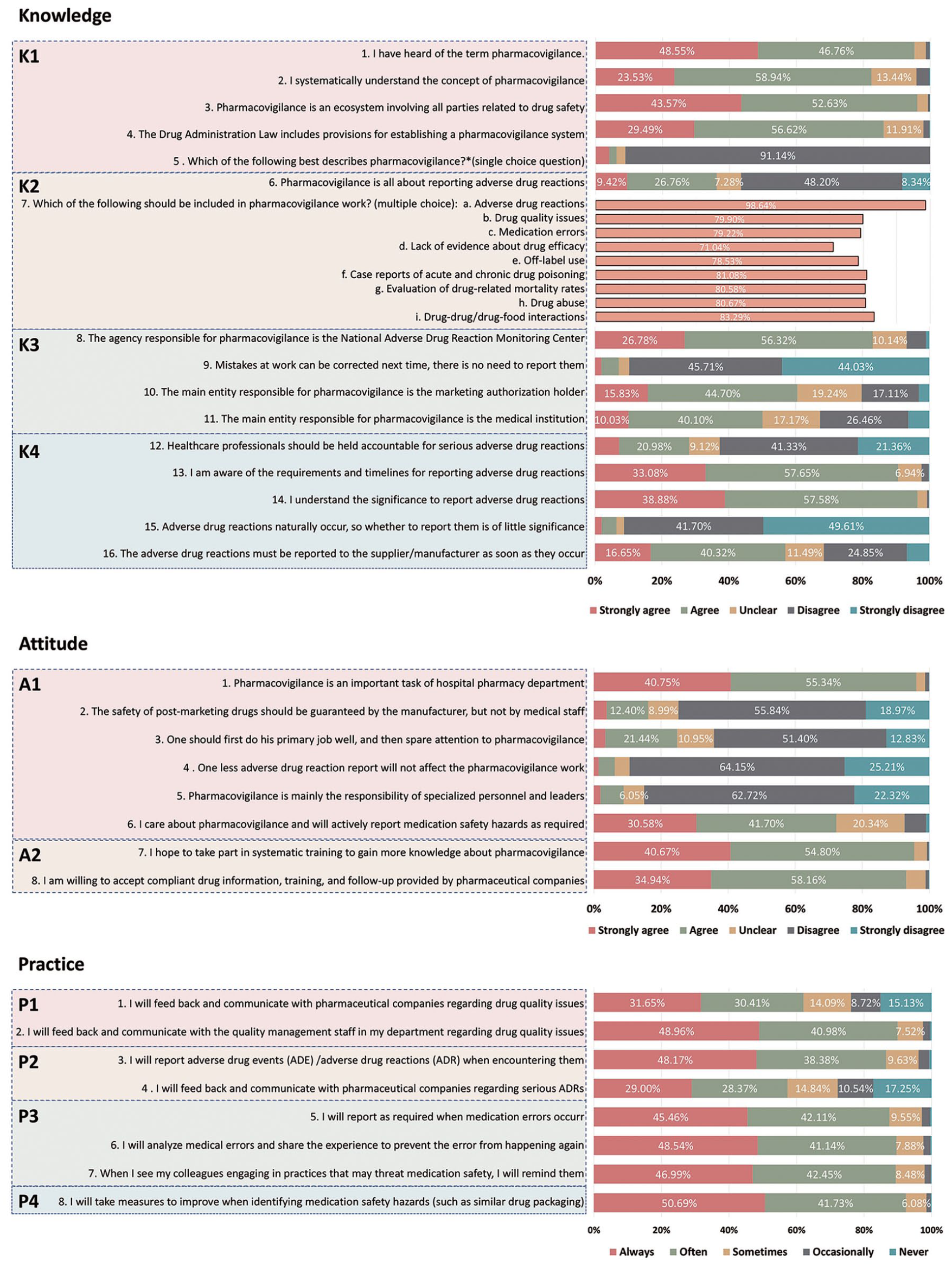
Detailed questions and responses on knowledge, attitude, and practice on pharmacovigilance.
A vast majority recognized that ADRs fall under pharmacovigilance (98.6%); however, many erroneously viewed it primarily as ADR reporting, with 9.4% strongly agreeing and 26.8% partly agreeing. Around 20%–28% did not consider aspects like medication errors, drug efficacy, and off-label use as part of pharmacovigilance.
“Knowledge of Responsibility” scored lowest, with many pharmacists mistakenly believing that the medical institution, not the MAH, was primarily responsible for pharmacovigilance. Notably, 975 (10.0%) strongly agreed and 3899 (40.1%) partly agreed with this view. Around 90% understood the requirements and timelines for ADR reporting, and 96.5% recognized its importance. However, concerningly, 24.9% and 6.7% partly or strongly disagreed with the immediate reporting of serious ADRs to suppliers/manufacturers.
Responses to attitude questions
In the second dimension of attitude, results (median, IQR) showed that “attitude towards training” (4.00, 4.00–5.00) scored higher than “attitude towards responsibility” (3.99, 3.62–4.25). As shown in Figure 4, a notable proportion (24.8%) placed low emphasis on pharmacovigilance responsibility, possibly due to a lack of clarity about its scope. A significant majority, 95.5%, expressed willingness to engage in systematic training on pharmacovigilance. Additionally, 93.1% were open to receiving drug information, training, and follow-ups provided by pharmaceutical companies.
Responses to practice questions
In the second dimension of practice, the score (median, IQR) rankings were: “Practice of Identifying Medication Safety Hazards” (5.00, 4.00–5.00) > “Practice of Addressing Medication Errors” (4.33, 4.00–5.00) > ”Practice for Managing ADEs/ADRs” (4.00, 3.32–5.00) > “Practice in Handling Drug Quality Issues” (4.00, 3.20–5.00). As shown in Figure 4, 62.1% of pharmacists “always” or “often” communicated with pharmaceutical companies about drug quality issues, and 89.9% liaised with their department’s quality management personnel. However, 10.5% only occasionally provided feedback on serious ADRs to MAH, and 17.3% never did. Regarding medication errors, 87.6% routinely reported them per regulations, 89.7% analyzed and shared information on medication errors to prevent recurrence, and 89.4% reminded colleagues about practices that could compromise medication safety.
Responses to open-ended questions
In response to the optional open-ended query, “Your opinions/suggestions on pharmacovigilance,” 2886 participants, representing a 28.9% completion rate, submitted their inputs. Among these, 587 responses (20.3%) advocated for enhanced training, while 710 responses (24.6%) emphasized the need for greater awareness and publicity of pharmacovigilance.
Influencing factors of KAP
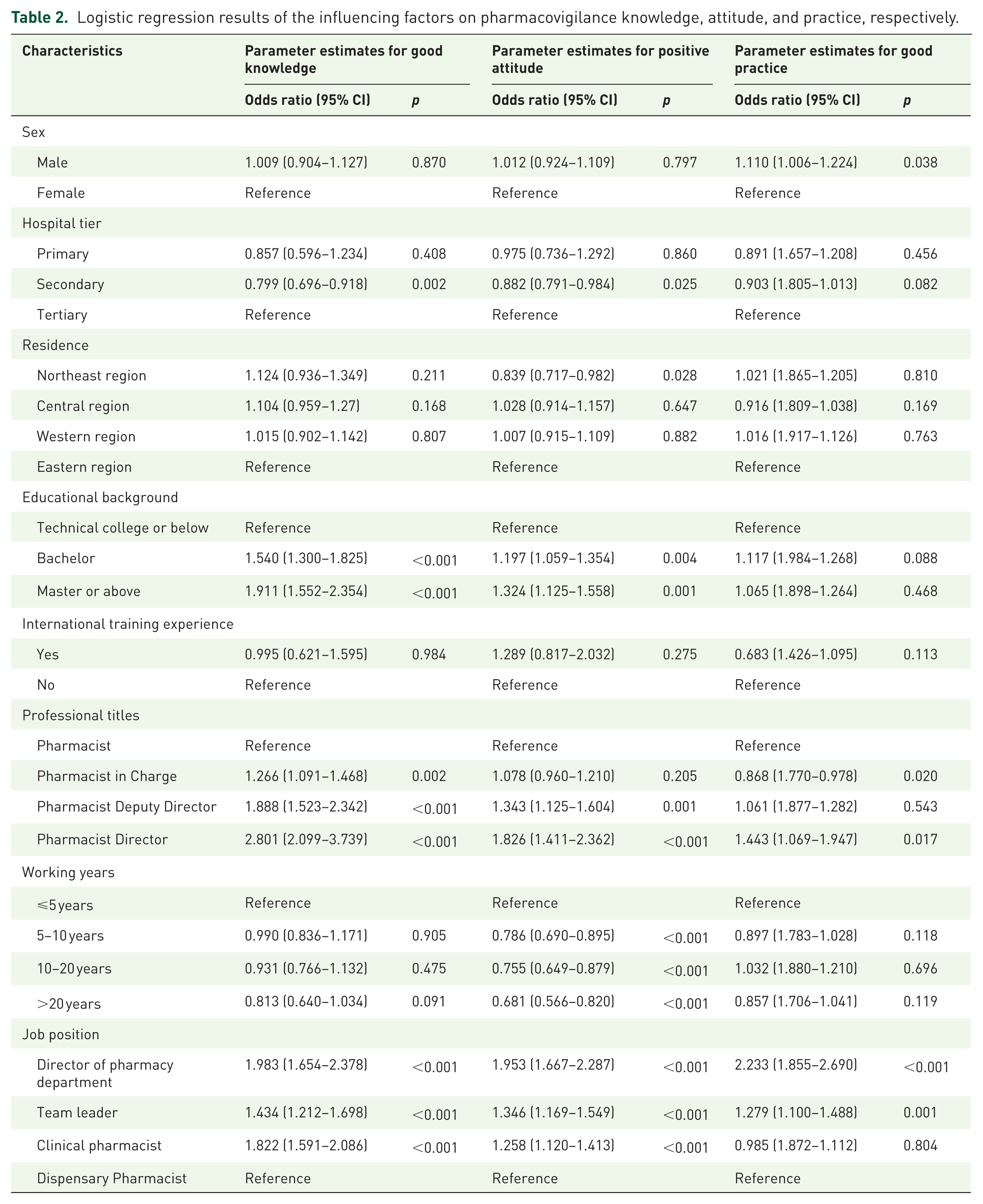
A Multivariable logistic regression model was applied to investigate the factors that correlate with the KAP of pharmacovigilance. The results are shown in Table 2. Male pharmacists outperformed their female counterparts in pharmacovigilance practice. Compared to tertiary hospitals, pharmacists in secondary hospitals were less likely to achieve high scores in knowledge and attitude. Compared with pharmacists living in the eastern region, those in the northeast region had lower odds of scoring high in attitude. Higher levels of education correlated positively with enhanced performance in knowledge and attitude. Having international training experience showed no significant correlation with KAP scores. Generally, higher professional titles were linked with improved knowledge and attitude, but intermediate-level pharmacists exhibited lower practice scores compared to their junior-level colleagues. Interestingly, pharmacists with longer tenure demonstrated lower scores in attitude than those with less than 5 years of working experience. Furthermore, clinical pharmacists, team leaders, and department directors outshone dispensing pharmacists in all aspects of KAP.
Logistic regression results of the influencing factors on pharmacovigilance knowledge, attitude, and practice, respectively.
Discussion
In this study, we evaluated the KAP of hospital pharmacists in China regarding pharmacovigilance. To our knowledge, it represents the most extensive nationwide cross-sectional survey of pharmacists in the country. Overall, the KAP of hospital pharmacists in China regarding pharmacovigilance is reasonable but still requires enhancement, particularly in the knowledge and attitude aspects. Our study highlighted several areas that require further improvement. Before delving into these specific gaps, it is important to contextualize our findings against prior research in China.
Compared with previous studies, our findings provide a broader and more up-to-date perspective on hospital pharmacists’ KAP regarding pharmacovigilance. Earlier regional studies conducted in Yunnan and Shaanxi provinces either targeted the general public or included mixed HCPs, limiting their applicability to pharmacist-specific practice.17,18 A previous nationwide survey primarily concentrated on ADR reporting and did not include other key components of pharmacovigilance, such as medication errors, off-label use, or drug quality issues. 11 In contrast, our study adopted a wider pharmacovigilance framework, capturing multiple safety domains and uncovering additional knowledge gaps and practical barriers. Moreover, compared with an earlier study from northern China, where over 80% of pharmacists had never reported an ADR, our findings suggest significant progress in pharmacovigilance awareness and behavior over the past decade. 19 However, the persistence of fundamental misunderstandings—such as the role of MAHs—and the limited pharmacist–industry communication highlight that improvements in regulation and education have not yet fully translated into effective practice.
A key issue identified is the limited knowledge among hospital pharmacists regarding pharmacovigilance responsibility. The survey incorporated contradictory questions to gauge pharmacists’ understanding of the responsible entity in pharmacovigilance. The low scores (2.79 and 3.53 for reverse and forward questions, respectively) indicate a significant knowledge gap, with a notably 50.13% of pharmacists incorrectly identifying the medical institution, rather than the MAH, as the primary responsible party. The 2019 revision of the Drug Administration Law of the People’s Republic of China, which was the first law to address the pharmacovigilance system, explicitly states that the MAH is primarily responsible for pharmacovigilance. 6 Despite being in effect for 3 years, with considerable efforts by industry organizations and government agencies to disseminate information and organize educational initiatives for pharmacists, the actual impact on learning and understanding appears limited. This is evidenced by the pharmacists’ relatively poor comprehension of legal mandates assigning the MAH as the main responsible party in pharmacovigilance. Correcting this misconception is crucial for improving pharmacists’ practice in reporting drug safety issues to drug companies.
Another significant finding is the need for improved collaboration between medical institutions and MAHs. The survey revealed inadequate active information sharing between individual pharmacists and MAHs. Specific questions assessed pharmacists’ likelihood to communicate with pharmaceutical companies in cases of serious ADRs and drug quality issues, receiving modest index scores of 3.41 and 3.55, respectively. Conversely, pharmacists showed a greater propensity to engage with their departments in such scenarios, as indicated by higher index scores of 4.30 and 4.36. These survey findings align with the prevailing practices across diverse medical institutions at various levels. 12 Current regulations governing the reporting of adverse reactions do not mandate pharmacists in medical institutions to directly communicate with pharmaceutical companies or other stakeholders upon encountering adverse reactions. Instead, pharmaceutical companies primarily access pertinent data through the government’s adverse reaction reporting platform. This approach can inadvertently delay the MAH from receiving first-hand information promptly, highlighting a potential area for improvement in the pharmacovigilance communication process. The healthy development of a pharmacovigilance ecology requires timely, objective, and comprehensive data sharing and support from all involved parties, within legal and normative boundaries, to facilitate more effective scientific decision-making. 34
To address the above problems, we propose the establishment of a Pharmacovigilance Officer (PVO) role within healthcare institutions. While most Chinese hospitals have ADR liaisons, their responsibilities are typically limited to passive reporting of adverse reactions. The proposed PVO role builds upon and expands this existing mechanism by introducing a dedicated, system-level pharmacovigilance function within medical institutions. The PVO would oversee the identification, assessment, and management of a wide range of medication-related risks, including ADRs, medication errors, drug quality defects, and drug misuse or abuse. The conceptual design of the PVO also draws on the Medication Safety Officer (MSO) model advocated by the Institute for Safe Medication Practices in the United States. 35 However, unlike MSOs, who primarily focus on preventing medication errors, the PVO is intended to cover the full scope of pharmacovigilance. By institutionalizing this role, healthcare settings may achieve more proactive and coordinated pharmacovigilance practices, ultimately improving medication safety and overall care quality. It is also recommended that governmental bodies and hospital administrators intensify their efforts in raising awareness and providing comprehensive training. Such initiatives are pivotal for the progressive development and solidification of pharmacovigilance systems.
The multivariable logistic regression analysis revealed significant factors influencing pharmacovigilance KAP among hospital pharmacists in China. Notably, male pharmacists outperformed females in practice, suggesting sex-related differences in pharmacovigilance training or execution. Pharmacists in secondary hospitals and those from the Northeastern region demonstrated lower knowledge and attitude scores, indicating potential regional and institutional disparities. Expectedly, higher educational attainment was positively associated with better knowledge and attitude, underscoring the importance of academic background in shaping pharmacists’ perspectives and understanding of pharmacovigilance. Surprisingly, intermediate-level pharmacists had lower practice scores than junior colleagues, and more experienced pharmacists scored lower in knowledge and attitude, highlighting the need for ongoing education and practice-oriented training. Clinical pharmacists and those in leadership positions excelled in all KAP aspects, underscoring the importance of specialized roles in pharmacovigilance efficacy. These findings suggest the necessity of tailored training and interventions based on hospital level, geographical location, and professional rank to enhance pharmacovigilance among hospital pharmacists in China.
Strengths and limitations
Our study is the largest cross-sectional survey on the KAP regarding pharmacovigilance among hospital pharmacists in China to date. It provides a comprehensive view of their understanding and behaviors toward the pharmacovigilance system, offering valuable insights for its development and improvement. The questionnaire was developed with input from a multidisciplinary team and utilized the Delphi method to ensure scientific validity. Academic associations like the Chinese Pharmacists Association facilitated the survey process, achieving a highly effective response rate.
However, the study has limitations. Firstly, convenience sampling was used in the questionnaire distribution instead of strict random sampling methods, which may affect the representativeness of the results. For instance, pharmacists from tertiary hospitals were overrepresented. Despite this, the distribution of respondents across different regions broadly aligns with the population coverage of these areas. Secondly, although anonymity of the questionnaire was emphasized, the self-reporting format might be subject to reporting and recall biases, as participants might have misremembered past behaviors or provided socially desirable responses. In addition, despite the high effective response rate, the possibility of non-response bias cannot be excluded. Individuals who chose not to respond or submitted incomplete questionnaires may systematically differ from respondents, potentially introducing bias into our findings. Thirdly, being cross-sectional, the correlations identified do not imply causality, necessitating cautious interpretation of the findings.
Conclusion
Our study reveals a significant scope for enhancement in the KAP of hospital pharmacists pertaining to pharmacovigilance in China. Specifically, there is a critical need to improve the understanding of the scope and responsibilities inherent in pharmacovigilance. Furthermore, fostering stronger collaborative relationships between medical institutions and MAH is imperative. Strengthening these aspects is crucial for the advancement of pharmacovigilance in China, especially in targeted pharmacist groups, ultimately leading to improved patient safety and drug efficacy. This study underscores the importance of continuous education and the establishment of effective communication channels as key strategies in realizing these goals.
Supplemental Material
sj-docx-1-taw-10.1177_20420986251374963 – Supplemental material for Knowledge, attitude, and practice regarding pharmacovigilance among hospital pharmacists in China
Supplemental material, sj-docx-1-taw-10.1177_20420986251374963 for Knowledge, attitude, and practice regarding pharmacovigilance among hospital pharmacists in China by Yinchu Cheng, Tingting Qiu, Xiaole Zhang, Qi Zhou, Xiaojuan Li, Ailing Song, Qunhong Shen and Fang Liu in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-2-taw-10.1177_20420986251374963 – Supplemental material for Knowledge, attitude, and practice regarding pharmacovigilance among hospital pharmacists in China
Supplemental material, sj-docx-2-taw-10.1177_20420986251374963 for Knowledge, attitude, and practice regarding pharmacovigilance among hospital pharmacists in China by Yinchu Cheng, Tingting Qiu, Xiaole Zhang, Qi Zhou, Xiaojuan Li, Ailing Song, Qunhong Shen and Fang Liu in Therapeutic Advances in Drug Safety
Footnotes
Acknowledgements
The authors would like to thank the Partnership for Safe Medicines (PSM) Beijing Foundation, the Chinese Pharmacists Association, the Hospital Pharmacy Professional Committee of the Chinese Pharmaceutical Association, the Pharmacovigilance Professional Committee of the Chinese Pharmaceutical Association, the Pharmacogenetic Diseases Professional Committee of the Chinese Pharmacological Society, the Pharmaceutical Management Professional Committee of China Hospital Association, the Health and Development Institute of the Tsinghua University, the Adverse Drug Reactions Journal, the Medication Safety Panel in China Core Group of International Network for the Rational Use of Drugs (INRUD), and all the participants for supporting the study.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
