Abstract
Background:
Adverse drug reactions (ADRs) contribute significant clinical and economic burden to the country’s healthcare system globally. Prompt reporting of ADRs by the community pharmacist is essential to the active pharmacovigilance program.
Objectives:
This study assesses private community pharmacists’ knowledge, attitude, and practice (KAP) about ADRs and reporting.
Design:
A cross-sectional, qualitative study was performed using a pre-validated self-administered questionnaire.
Methods:
This self-administered questionnaire was conducted at community pharmacies between March and July 2022. The data collected were analyzed using the Mann–Whitney and Kruskal–Wallis tests to examine the differences in overall KAP scores with a subgroup of sociodemographic characteristics of the study participants. Logistic regression analysis was used to analyze the predictors of practice.
Results:
In total, 156 fully completed questionnaires were collected by the community pharmacists. A positive association between the designation, qualification, and work experience with the total scores of the respondents was observed (p < 0.05). Among the predictors of ADR reporting practice, a significant association was observed with knowledge score (⩾6, p = 0.0219), designation (pharmacists, p = 0.0102), qualification (masters, p = 0.0002), and work experience (⩾11 years, p = 0.0184). Most community pharmacists had good knowledge and attitude but poor practice toward reporting ADRs. Uncertainty of how and where to report, lack of training, lack of reporting forms, and insufficient clinical knowledge were the practice-based barriers in the ADR reporting process.
Conclusion:
Though the study found sufficient understanding and favorable views on ADR reporting, participants reported poor practices and barriers to reporting ADR. Therefore, structured continuing professional development programs for community pharmacists are needed to overcome the barriers and enhance the practice of ADR reporting.
Plain language summary
Introduction:
Medicines aren’t safe because they can have bad side effects that can cause major health problems for the patient. These fears make it more important and show how valuable it is to report side effects. This study examines community pharmacists’ knowledge, attitude, and practices (KAP) in the United Arab Emirates about side effects and how often people tell them.
Methods:
A structured questionnaire was used to collect information on the profiles of community pharmacists and their knowledge, views, and practices regarding adverse effects. The purpose was to discover what stops pharmacists from reporting adverse effects and suggest ways to improve reporting. We used statistical tests to examine the total KAP results and information about the study subjects’ backgrounds.
Results:
Out of 177 community pharmacists who filled out the form, only 156 completed it. The study found that people had good understanding and attitude scores but poor behaviors when reporting side effects. The main things that kept pharmacists from reporting side effects were not knowing how and where to report them, not having a reporting form, and not having enough training and knowledge about reporting.
Conclusion:
The study found that private community pharmacists know a lot about drugs and have positive views. However, they don’t always follow through with good advice, and there aren’t many good reasons why people don’t report unpleasant effects. Education and training programs about side effects might help people overcome the problems and get more neighborhood pharmacists to write about them. This will improve the standard of patient care and the safe and effective use of drugs.
Introduction
Adverse drug reactions (ADRs) represent a worldwide health concern requiring attention due to their significant contribution to patients’ drug-related morbidity and mortality, increasing hospital admission and healthcare expenditure.1,2 Advanced age, comorbidities, poly medication, inappropriate prescribing, and suboptimal monitoring are some of the risk factors for the development of ADRs. 3 Of all hospitalizations, 0.2%–59.6% are attributed to ADRs, and 1.8% are known to be fatal. 4 A retrospective study evaluating ADRs has reported that systemic antimicrobial agents, drugs acting on the cardiovascular system, alimentary tract and metabolism, and musculoskeletal system were the most common pharmacological groups according to the World Health Organization (WHO)-Anatomical Therapeutic Chemical (ATC) classification implicated in the ADRs. 5 Although every drug undergoes various phases of safety screening during pre-clinical studies and clinical trials, not all ADRs are identified due to clinical trial constraints. 2 Therefore, it becomes essential that drug products continue to be monitored for safety and efficacy in practice settings even after approval. 6
At this stage, the role of pharmacovigilance (PV) in resolving concerns linked to drug safety becomes highly important in enhancing the safe use of medications. Establishing PV is ideal to ensure secure, rational, and pharmacoeconomics medication use to improve clinical outcomes and minimize adverse drug outcomes. 7 PV has been the foundation of numerous drug safety initiatives, including medication cessation, issuing safety alerts, revisions in drug labeling, and prescription limitations. Artificial intelligence has significantly contributed to PV signal identification and data mining techniques. 8 Among various methods of reporting ADR, healthcare professionals’ spontaneous reporting system of ADRs is the backbone of PV that will serve as the basis for a robust and comprehensive post-marketing drug safety surveillance program. 9
Most countries have joined the WHO’s international drug monitoring program to identify and report the ADRs in clinical practice through PV centers.10,11 The United Arab Emirates (UAE) established the national drug surveillance program in 2008 and later in 2013, with the WHO inter-drug monitoring program in collaboration with the Uppsala Monitoring Center. 12 The Ministry of Health (MOH)/Emirates Health Services, Department of Health (DOH)-Abu Dhabi, and Dubai Health Authority (DHA)-Dubai now governs healthcare in the UAE. 13
PV laws and legislation exist following the best standards worldwide. Health regulatory authorities continually work to raise awareness among healthcare practitioners about the necessity of monitoring and reporting ADR in the country; nonetheless, appropriate application remains a challenge. 8 Additionally, the UAE Ministry of Health and Prevention implemented an innovative UAE RADR application in 2019 to facilitate PV activities in the country. 14
Improving medication safety to reduce patient harm must be a joint responsibility among key stakeholders. Drug safety practices should include all pharmaceutical products and not be limited to newer medicines, monitored and reported for associated ADRs in the daily routine clinical practice. The pharmaceutical industry is primarily responsible for the continued collection of ADRs connected with medications; nevertheless, consumers have an essential role after marketing. 15 Pharmacists in different healthcare settings contribute significantly to identifying, monitoring, reporting ADRs, and optimizing drug therapy. Prompt reporting of ADRs by the community pharmacist is essential to the PV program, as they are the first point of contact for patients concerning drug safety issues. 16
There are no data published by the health authorities that manage healthcare services in different emirates of the UAE about the number of ADRs reported. However, as per the data from the DOH), which manages the healthcare services across the emirate of Abu Dhabi, an average of 595 ADRs were reported during 2013–2021, and there were no ADRs reported that caused hospitalization and/or mortality. Additionally, no structured studies have been conducted and/or published in the UAE to determine the number of ADRs reported. 17
Several studies have been undertaken in the UAE to establish the knowledge, attitude, and practice (KAP) of ADR reporting among healthcare professionals, identify their current tactics, and specify actions to strengthen ADR reporting.18–21 Even though most of the studies observed fairly better knowledge and positive attitudes toward ADR reporting, poor practice and low levels of ADRs were also revealed among them. In addition, determinants such as the complexity of the reporting process, insufficient information, inadequate training, and lack of reporting knowledge and skills were identified as the main barriers hindering ADR reporting. Research in the UAE detected inadequate KAP among healthcare practitioners. It indicated that 81%, 83%, and 83.3% of doctors, community pharmacists, and hospital pharmacists, respectively, were ignorant of the ADR reporting center in UAE, and 56%, 60%, and 72% did not know the processes involved in reporting ADRs. 22 Furthermore, another study has emphasized the need to reinforce and promote favorable public perceptions and attitudes regarding drug safety and ADR reporting in the UAE. 23
Not many studies have assessed the KAP of community pharmacists on the identification, monitoring, and reporting of ADRs in the UAE’s Ras Al Khaimah region. The main goal of this study is to thoroughly evaluate community pharmacists’ KAPs on ADRs and reporting in the UAE. This study sheds light on the shortcomings and challenges in KAP regarding ADR reporting by community pharmacists to benefit stakeholders, healthcare providers, and policymakers in the UAE. By providing a comprehensive overview of the present KAP, this research also aims to identify areas for improvement and inform targeted measures to strengthen pharmacovigilance efforts and patient safety initiatives in the UAE healthcare system.
Materials and methods
Study design, study settings, and study populations
This cross-sectional study was conducted using a validated questionnaire related to KAPs regarding ADRs and its reporting among community pharmacists in Ras Al Khaimah among community pharmacists for 5 months between March and July 2022 in the northern emirate of UAE.
Inclusion and exclusion criteria
The study included all registered and assistant pharmacists practicing at the private community pharmacies within Ras Al-Khaimah. Pharmacy technicians, trainee pharmacists, supporting staff, and pharmacists who worked in primary health centers, hospitals, and drug stores were excluded. The study did not include pharmacists who were unavailable while providing questionnaires or traveling for annual holidays.
Sample size and sampling method
A convenient sampling technique was involved to collect data from the study participants. The required sample size was calculated using qualtricsXM. 24 There are around 90 pharmacies within Ras Al-Khaimah, with at least one pharmacist and one assistant pharmacist in each pharmacy. Considering a total of 180 probable respondents from 90 pharmacies and a response distribution of 50%, the required number of respondents was 123, with a 5% error margin and a 95% confidence interval.
Data collection method
The Strengthening of the Reporting of Observational Studies in Epidemiology (STROBE) reporting guidelines for cross-sectional studies was adopted and followed as a protocol for conducting this study (Supplemental Material). 25 The study investigator met the pharmacist independently at their practice sites, explained the purpose of the study, and asked the pharmacist to fill out the validated structured self-administered questionnaire along with the informed consent provided in hard copy delivered by hand. Participants were informed that the study was optional, and we guaranteed their replies would be anonymous and confidential. Pharmacist ambiguity related to the questionnaire was answered verbally. Sufficient time (approximately 1 week) and reminders were provided to each participating pharmacist working in chain pharmacies and independent pharmacies to complete and submit the questionnaire, and their response was documented. The investigator thoroughly reviewed the submitted questionnaires to verify the integrity of the gathered data and to prevent any missing or inadequate information within the questionnaire. The tool did not include any questions that may potentially expose the personal identity of the study participant or the organization.
Questionnaire development and validation
A self-administered questionnaire assessing KAP was developed by consulting relevant literature, recommendations, and similar research. It was subsequently adjusted to suit the requirements of the current investigation.17–20 The questionnaires were validated for content and criteria validity. The initial question was forwarded to three academic clinical pharmacists specializing in pharmacovigilance to assess the content validity. The questionnaire underwent a pilot test to evaluate internal consistency and reliability. This test was conducted with a convenient sample of six pharmacists and four assistant pharmacists, who were not included in the final participants of the study. The questionnaire’s Cronbach alpha value, determined using the reliability scale, was 0.697. No other alterations were made to the questionnaire.
The final version of the validated self-administered instrument consists of 31 questions divided into different sections covering the study objectives. The first section focused mainly on participant information and demographic details, age, gender, designation, qualification, work experience, nationality, country of graduation, number of prescriptions dispensed, patient served, and average time spent per prescription daily. The second segment has 10 questions assessing the prior knowledge (familiarity gained through understanding and experiences related to ADRs to medications) of the participants associated with the reporting of ADRs, including awareness of the reporting program in UAE, PV, reporting of ADRs related to over-the-counter products, herbal products, previously documented ADRs, medication safety, severe and life-threatening ADRs, and consulting the prescriber before reporting any ADRs.
The third section comprises seven questions that analyze participant attitudes (individual perceptions or views gained through experience related to ADRs) toward ADR reporting. This section includes questions about the importance of community pharmacists in ADR reporting, improving patient safety, beneficiary, mandatory reporting, part of professional practice, and time consumption. Finally, the fourth section contains the participants’ ADR reporting practices (individuals’ skills and actions/activities related to identification, monitoring, management, and reporting of ADRs at their practice sites), including encountered ADRs and the practice of reporting on their patients, training and attending workshops related to ADR, and the availability of reporting forms in their practice sites. The responses to the questionnaire section mentioned above were provided as “yes,” “no,” and “I do not know.”
In addition, the questionnaire also included free text questions corresponding to providing advice to patients on possible adverse effects of drugs during dispensing, asking about ADRs while serving their patients, and the percentage of patients reporting ADRs directly to community pharmacists. Furthermore, a few questions that determined how to confirm the occurrence of ADRs, exploring the factors that encourage the reporting of ADRs, the types of ADRs to be reported, and the barriers that discourage the reporting of any ADRs, were also included in the study.
Scoring system
The questionnaire was provided to the study participants in English and did not require language translation. The pharmacist’s response to the structured questionnaire about KAP was categorized as “correct” or “incorrect.” Each correct and positive response received a score of “1,” and the negative and incorrect responses received a score of “0.” The total score for the knowledge-related questions was “10”; the maximum score for attitudes and practice-related questions was 7 each. The knowledge component was scored on a scale of 0–10, with two categories: excellent knowledge (score ⩾6) and weak knowledge (score <5).
Regarding questions related to attitude, participants’ responses, such as “yes,” were considered positive, while “no” and “I do not know” were considered negative. A score equal to or greater than 4 was classified as a good attitude, while a score equal to or less than 3 was classified as a negative attitude. Similarly, for the practice-related questions, a total score of ⩽ 3 was considered inadequate practice, and a score of ⩾ 4 was regarded as adequate practice. The scoring system helped us understand study participants’ ADR reporting practices.
The final part of the questionnaire (Q No. 28–31) included four questions, and each question was further subdivided into different domains to assess the willingness, factors encouraging the nature of ADRs, and barriers of the respondents in reporting ADRs. The responses to questions related to willingness, factors encouraging, and nature of ADRs to be reported were assessed using five Likert items ranging from strongly agree 1, agree 2, neutral 3, disagree 4, and strongly disagree 5. The responses to the obstacles to reporting ADRs were categorized into three options: never, partially, and totally. These options were assigned scores of 1, 2, and 3, respectively.
Data analysis
The questions were encoded, inputted into the Excel spreadsheet, and examined using SPSS version 27 (IBM Corp., Armonk, NY, USA. Question numbers 8, 9, and 15 were negative-worded statements; hence, they were reverse-coded during analysis. Data normality was verified using the one-sample Kolmogorov–Smirnov test. Descriptive statistics were employed to examine the average and variability of continuous variables and the proportions and frequencies of categorical variables to describe the demographic characteristics.
The examination of differences between the pharmacist and assistant pharmacist in terms of each question related to KAP was carried out, and the comparison of the total overall KAP scores with a subgroup of sociodemographic features of the research participants was carried out using the Mann–Whitney U test (for two groups) and the Kruskal–Wallis test (for more than two groups). The association between the KAP scores of the study participants and the reporting of ADRs was assessed using Pearson’s correlation. The chi-square test was used to investigate the association between the mean scores of KAP reporting ADRs and the characteristics of pharmacists. The determinants of practice linked to ADR reporting were analyzed using multivariate logistic regression. p value less than 0.05 was deemed to indicate statistical significance (Figure 1–3).

Overall knowledge of the study participants regarding adverse drug reactions (n = 156); good knowledge (n = 140), poor knowledge (n = 16).

Overall attitude of the study participants regarding adverse drug reactions (n = 156); positive attitude (n = 151), negative attitude (n = 5).
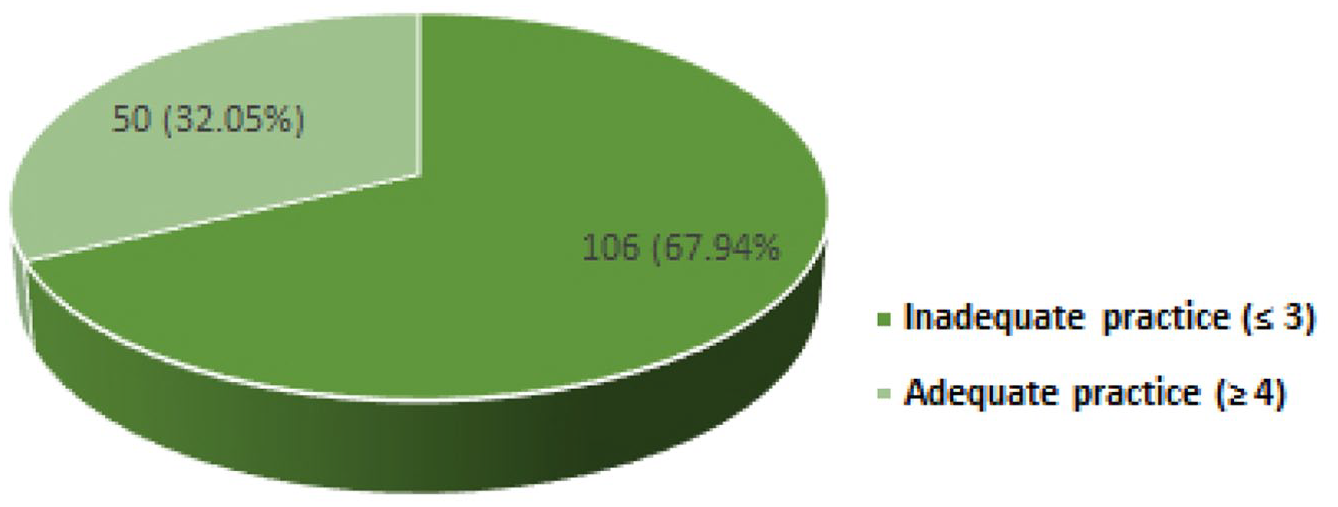
Overall practice of the study participants regarding adverse drug reactions (n = 156); inadequate practice (n = 106), adequate practice (n = 50).
Results
Demographic characteristics of study participants
In our study, 177 private community pharmacists were approached and given questionnaires. A total of 156 study participants completed the questionnaires with a response rate of 88% and were included for analysis. The majority of the respondents were males (61.53%), in the age group of 21–35 years (71.70%), and pharmacists (75%). In addition, most of the respondents had bachelor’s degrees (54.48%). The demographic details of the study participants are provided in Table 1.
Demographic characteristics of the study participants.

Overall KAP scores of study participants related to ADR
Based on the individual score, the participants’ knowledge was classified as good or poor. Among the total study participants, 140 respondents (89.74%) had a good knowledge score (⩾6), and 16 respondents (10.25%) had a poor knowledge score (⩽ 5).
An attempt was made to assess the overall attitude of the study participants using seven questions. As a result, of the total 156 participants, it was observed that 150 study participants (96.13%) showed positive attitudes (⩾ 4) and 6 (3.84%) showed negative attitudes (⩽ 3).
Seven questions were used to analyze the general practice of the study participants related to the ADR reporting process. The overall analysis of practice showed that a “good” ADR reporting practice was identified in only 50 respondents (32.05%), while 106 respondents (67.94%) have a poor ADR reporting practice (Table 2).
Comparison of mean knowledge, attitude, practice, and overall scores about reporting adverse drug reactions among study participants.

Statistically significant at 0.05 level (two-tailed)
Statistically significant at 0.01 level (two-tailed),
Interrelationship of the KAP scores with ADR reporting
The inter-association of the study participants’ KAP scores concerning their ADR reporting was analyzed. There was a significant positive correlation between knowledge and attitude and knowledge and practice, indicating that good knowledge positively imparted their attitude and practice (p < 0.01). Furthermore, attitude had a significant positive correlation with the practice of study participants concerning ADR reporting (p < 0.01). The details are presented in Table 3.
ADR reporting KAP of the study participants.

Correlation is significant at the 0.01 level (two-tailed)
Predictors of practice toward ADR reporting
A multivariate logistic regression analysis was conducted to evaluate the correlation between each variable and the practice of reporting ADRs. The multivariate exploration revealed a significant association was observed with knowledge score (⩾6, p = 0.0219), designation (pharmacists, p = 0.0102), qualification (masters, p = 0.0002), and work experience (⩾11 years, p = 0.0184) related to ADR reporting (Table 4).
Predictors of practice related to ADR reporting.
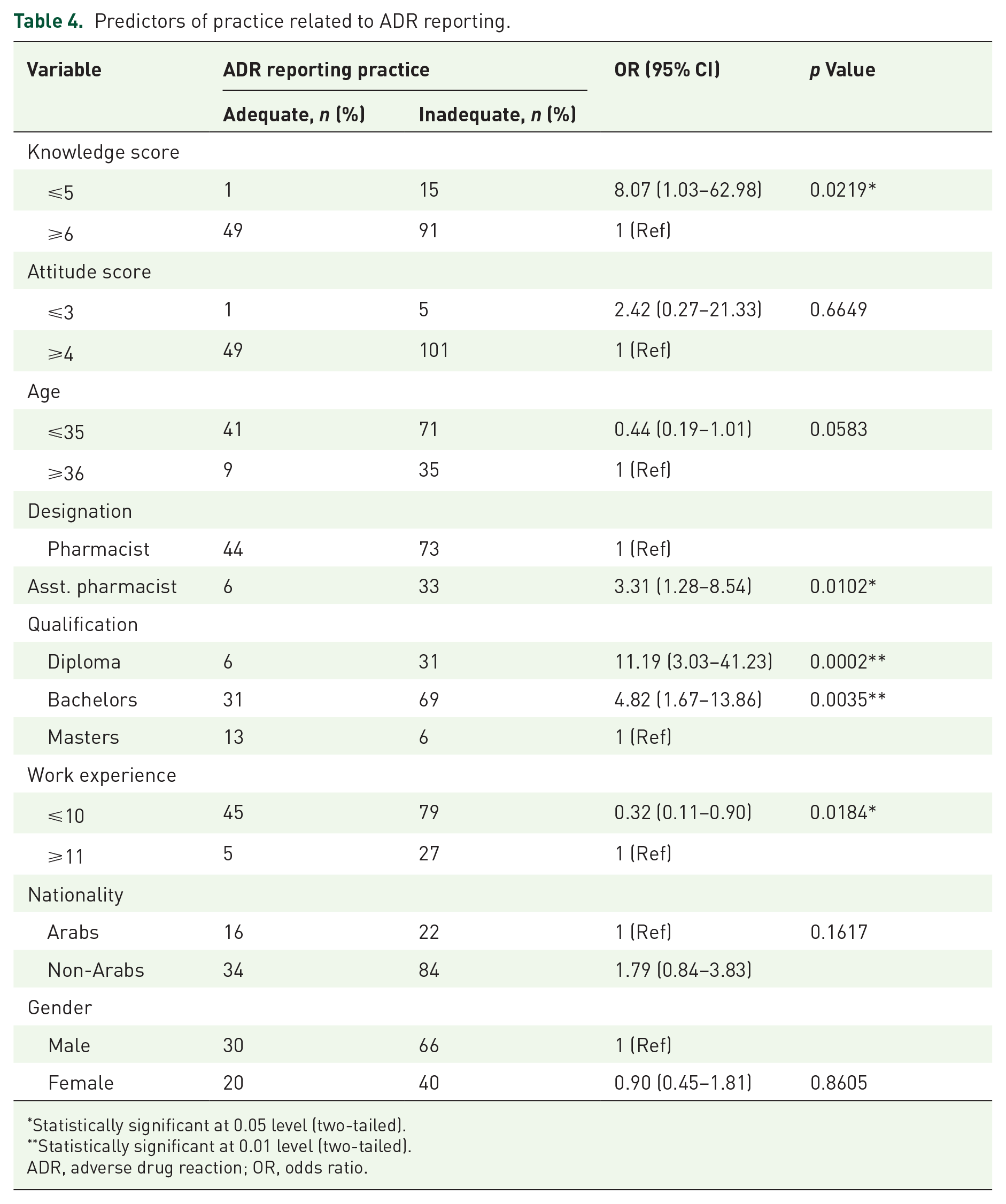
Statistically significant at 0.05 level (two-tailed).
Statistically significant at 0.01 level (two-tailed).
ADR, adverse drug reaction; OR, odds ratio.
KAP responses
The details of the study participants’ responses to questions related to KAPs are presented in Table 4. A statistically significant difference was observed in one question each on knowledge and attitude and two questions related to practices (p < 0.05) between pharmacists and assistant pharmacists.
Providing advice on ADRs to their patients
The respondents were also asked to rate community pharmacists’ advice about possible adverse drug effects while dispensing medications to patients in their practices. Only 43.58% generally provide advice about potential adverse effects while dispensing medications, whereas 55.76% report that they sometimes, and 0.64% never advise their patients about the adverse drug effects.
Enquiring the patients about ADRs
Regarding the inquiry about ADRs in patients, 41% of the study respondents inquired “often” whereas 40.38% did “rarely” and 15.34% “very rarely.” Only less than 2% of the study participants asked about the ADRs “very often” while serving their patients.
Patients reporting ADRs to community pharmacies
Furthermore, it was observed that a majority of the survey participants (61.54%) acknowledged that only 10%–20% of their patients disclose ADRs to the community pharmacy. In contrast, 30.76% of the respondents stated that patients never report any ADRs to the pharmacy.
Study participants mean KAP scores in reporting ADRs
A comparison of mean KAP scores on reporting ADRs of study participants was carried out concerning their demographic profiles. A statistically significant association was observed for the variables “designation” and “qualification” concerning the mean knowledge and practice scores among study participants except for age, gender, work experience, and nationality (p < 0.05). However, no association was observed between mean attitude scores and the study’s demographic variables.
The inter-association of the total KAP scores of the study participants was analyzed concerning their demographic profiles: A positive association was observed between the “designation,” “qualification,” and “work experience” with the KAP scores of the respondents, which was found to be statistically significant (p < 0.05). However, no such association has been found between total scores and the gender, age, and nationality of study participants (Table 5).
Response of the study participants to the KAP-related questions.

Statistically significant at 0.05 level (two-tailed).
Statistically significant at 0.01 level (two-tailed).
ADR, adverse drug reaction.
Perception of willingness, factors, and barriers in reporting ADR among study participants
Questions concerned with confirming the occurrence of ADR, factors that encourage the reporting of ADRs, types of ADRs to be reported, and barriers to ADR reporting were also analyzed in our study. Regarding the confirmation of the ADR, it was observed that the majority (80.12%) of community pharmacists verify through a patient interview, whereas 42.30% may refer to a physician to confirm the ADRs.
Among the factors that encourage the report of ADRs, most respondents (53.20%) affirm that it depends on the seriousness of the ADRs, and 21.79% would like to report ADRs if they involve any new drug. Regarding questions related to the nature of the ADRs reported by the respondents, more than 70% of the study participants claimed that they would like to report all ADRs irrespective of their severity, ranging from mild to severe to life-threatening. However, a small proportion (12.82%) of study participants stated that only severe and life-threatening ADRs should be reported.
Barriers that may hinder the reporting of ADRs observed that 61 respondents (18.04%) agreed that reporting forms are not available in the practice settings, while 52 respondents (15.38%) point of view about the uncertainty of how and where to report ADRs (Table 6).
Perceived willingness, factors, and barriers in reporting ADR among study participants.

The total number and percentage may not match 156 since respondents can select more than one to mark their level of agreement.
ADR, adverse drug reaction.
Correlation between demographic factors and scores related to perception, practice, barriers, and willingness among the participants of the research
A statistically significant association was observed between the study participants’ age group and the willingness score related to ADR reporting (p = 0.041). At the same time, nationality significantly affects the nature of ADRs (p = 0.028) and barrier score (p = 0.002). However, no statistically significant association was observed within the variables regarding factors related to ADR reporting among practicing community pharmacists (Table 7).
Association between demographic variables and perception, practice, barrier, and willingness scores among study participants.

Statistically significant at 0.05 level (two-tailed).
Statistically significant at 0.01 level (two-tailed).
Discussion
Drugs are one of the main interventional approaches for managing diseases, and ensuring patient medication safety and rational use of medicines is paramount. 26 The study aimed to assess community pharmacists’ KAP toward ADRs and identify the barriers that prevent reporting and factors that encourage reporting ADRs in the northern region of the UAE.
The survey revealed that most participants knew the country’s ADR reporting scheme and were familiar with the PV program. The greater awareness exhibited by the pharmacists is likely due to more emphasis on topics related to ADRs and their reporting, which are incorporated into undergraduate and postgraduate pharmacy curricula by the universities. Additionally, attending Continuous Medical Education (CME) programs related to ADRs and their years of experience might have enhanced their awareness of PV programs. Similar findings were observed in Turkey, Saudi Arabia, and UAE studies.19,27,28 On the other hand, an Ethiopian study found a low level of knowledge about ADR reporting programs, but the majority have heard of PV. 29
The correlation between research participants with high levels of knowledge and favorable attitudes toward ADR reporting can enhance and promote the reporting of ADR by community pharmacists in the northern emirate region. The findings of our study are comparable with the responses of the community pharmacist in many other studies.1,18,30–32 In opposition to these findings, other previous studies reported poor or inadequate knowledge and positive attitudes toward reporting ADR among healthcare professionals and pharmacy students in hospitals, community pharmacies, and institutional settings.25,33–36 This disparity in knowledge and attitude among participants in different studies may be due to differences in the criteria determining the cut-off point to assess the study objectives and the gap in the study settings. In addition, in the various studies conducted in different countries, knowledge deficiency and poor attitudes regarding PV and ADR reporting have been reported as reasons for underreporting ADRs in both developed and developing countries.1,19,37,38
In general, the respondents in our study documented a poor practice with a low level of reporting ADR that was unsatisfactory, and they could not transform their solid knowledge and attitude into practice. There are two possible explanations for this. Initially, participants could lack knowledge about the existence of local and national ADR monitoring centers and the WHO Online ADR reporting database. Then, there is a perception that there is no legal requirement for community pharmacists to document and report ADRs to monitoring centers. This demonstrates the need to implement a nationwide photovoltaic program and ensure its successful attainment of goals and objectives, disseminate information to the local level, and deliver comprehensive training to the final users. These findings echo previous studies where the ADR reporting practice ranged from poor (11.1%–39.7%) to moderate/satisfactory (51.9%–74.8%).39–42 Fewer studies with a higher percentage of healthcare professionals, including pharmacists, reported no ADRs encountered during their practice.1,35,43 This finding is alarming and raises concern about events of drug safety problems. Poor practice may also be explained by various other factors such as the high number of patients being served by pharmacists daily, lack of time, and heavy workload, the pharmacist being busy with more focus on managerial activities, dealing with insurance coverage, and inventory control in community pharmacy and not providing honorarium leading to lack of motivation in reporting the ADRs.
Participants’ knowledge and practice of ADR reporting are unaffected by gender, age, work experience, and nationality. However, it was affected by their designation and qualification. The assistant pharmacist lacks expertise in ADR reporting, contrary to the pharmacist. Pharmacists can acquire their education through either a 4-year bachelor’s program or a 6-year program called Pharm D. The Pharm D program is specifically designed to train pharmacists who are scientifically and technically skilled, enabling them to provide the highest level of patient care services effectively. This program includes medication mistakes, drug-related issues, and the identification, monitoring, and reporting of PV (pharmacovigilance) and ADRs. These aspects are considered crucial components of patient care. Additionally, they receive extensive instruction and training at higher levels during their master’s programs, which makes them more understanding of this domain. This finding is similar to a study conducted in Sudan, which indicated no statistically significant difference in the average knowledge score between variables such as gender and age. It is also consistent with the findings of a study carried out in India, which showed that the individual’s qualification background influences the level of knowledge about reporting ADRs.36,44
Other key findings observed in our study are that a higher percentage of the study respondents suggested the patient interview, and fewer preferred to refer to the physician to confirm the occurrence of ADR in their patients. Referring to a physician for the management of an ADR is considered rational and good practice rather than suggesting a drug to treat the ADR since the act can result in negative consequences, especially when the exact cause of the reaction is unknown. The involvement of physicians and the benefit of patient support have been confirmed in many studies, which will help to improve the level of ADR reporting.37,45,46
Another element identified in this study was that a major percentage of study participants believed that all types of ADRs must be reported. The results of this study are similar to those of the study conducted in Nigeria, where most participants agreed that all ADRs need to be reported, including those already documented in the drug literature. 1 This enables them to be in a position and handle more responsibilities while dealing with prescriptions and working in close contact with patients. This observation greatly improves patient medication safety and contributes to national public health. Alternatively, a small number of study participants were misapprehension that only serious or life-threatening ADRs to drugs should be reported. Identical results were observed in a study that reported that two-thirds of healthcare professionals were only concerned about reporting severe ADRs. 47 This insight may be because minor ADRs are the regular ones that are unavoidable and do not cause any harm; however, severe and life-threatening ADRs can put a patient’s life in danger and must be reported. However, this assumption is not true. Even though some ADRs do not pose a direct threat to life, they can cause secondary injuries such as falls and fractures, especially in the elderly, leading to substantial mobility, cognition, and psychosocial impairment.
In this study, more community pharmacists were willing to report ADRs encountered during their practice. However, they identified numerous factors that prevent them from reporting ADR, including the inaccessibility of the reporting forms, the uncertainty of how and where to report, and not sure whether it is an ADR. Additionally, insufficient knowledge of drugs in detecting ADRs, time-consuming reporting, and fear that it may harm the confidence of patients were also observed as reasons that could discourage respondents from reporting ADRs. These results were consistent with previous studies, which documented barriers.46,48–50 Previous UAE studies have also reported similar observations.19,51 Pharmacists emphasize educating healthcare professionals on reporting ADRs. Multiple studies have demonstrated the positive impact of educational interventions on the KAPs of healthcare professionals in reporting ADRs.1,52–54 Certain participants may have concerns over potential reprisals from management or apprehension about legal accountability, which might act as obstacles. The same discovery was also documented in Kuwait and Iraq.55,56
In addition, inexperience in filling out forms, the complicated nature of ADRs, lack of easy access to report ADRs, increased workload, lack of information from patients, and absence of a national PV center were also reported as reasons for not reporting ADRs in a few other studies.15,18,21,42,48 These findings reveal that the study participants had inadequate training in ADR reporting and PV, which could have resulted in lower or underreporting ADRs. It is essential to consider the fact that any certified training geared toward conveying and acquiring knowledge requires employing an expert resource person who can execute a suitable mode of training by using quality resource materials with appropriate content that will facilitate learning to a great extent where the attendees can grab and internalize the content. So, while conducting PV- or ADR-related training, it may be necessary to confirm that all the above features are included so that the community pharmacist will benefit more from equipping them with the appropriate knowledge and skills.
It is essential to enhance the reporting of ADRs by implementing educational intervention strategies such as practical training programs, online modules, and a series of workshops and webinars for community pharmacists. This approach aims to improve their attitude, raise awareness, and increase ADR reporting rates. Reinforcing knowledge with a well-established and organized system and having a simplified, easy-to-use, standardized form for practicing healthcare professionals to report ADR is needed to improve drug safety. This finding contributes to good practice and will reflect positively on public health through the safe and effective use of medicines and advanced quality of patient care. The PV program should be considered the core component, and patient medication safety should be integrated into the pharmacy program and taught in depth in pharmacy schools to achieve program goals. Additionally, studies have also emphasized providing incentives for the pharmacist to report ADRs, which may help to enhance ADR reporting.38,57,58
Recommendations/practical implementations
Developing a standardized uniform PV center by integrating all the healthcare authorities Ministry of Health and Prevention, Emirates Health Services, Dubai Health Authority and Department of Health (MOHAP/EHS/DHA/DOH) with a simplified common reporting form with ease of access may help to enhance the monitoring and reporting of ADRs in the UAE. Furthermore, effective collaborations between health authorities and academic institutions strengthened by providing training related to ADR reporting to community pharmacists per the health authority regulations may help achieve objectives. To facilitate this, the health authority should establish explicit and comprehensive protocols and ensure effective implementation. This would promote reporting ADRs and improve UAE pharmacovigilance (PV) practices.
Limitations
Our study has a few limitations. The study relied on pharmacist self-reporting by providing questionnaires in which the data obtained reflected the study participants’ opinions. Nevertheless, self-reporting is a prevalent and pragmatic method of data collection. The information supplied may not accurately reflect real-world practice since some respondents may be reluctant to disclose problems in their approach. At the same time, the potential for recollection bias cannot be completely ruled out. Therefore, the study participants’ proper KAP cannot be fully explored. The findings of our study may provide useful insights as the survey was conducted in a single emirate. However, caution should be exercised when generalizing the findings to other regions of the UAE due to probable regional differences in practices and attitudes. Further research across different emirates is necessary to confirm the applicability of the findings. Adequate and periodic training related to ADR activities is one of several facets that could have impacted the study population’s KAPs. However, we could not assess that facet, one of our study’s limitations.
Conclusion
The current study observed that the practicing private community pharmacist had sufficient knowledge and a positive attitude toward ADR reporting. However, there are also poor practices and barriers to reporting ADR. The inaccessibility of the reporting forms, uncertainty of how and where to report, unsure whether it is an ADR, insufficient clinical knowledge, time consumption, and the fear that it may harm patients’ confidence were the identified barriers resulting in poor ADR reporting practices. This observation perhaps underscores the need to impart comprehensive training relevant to ADR, especially on detection, assessment, monitoring, reporting, and prevention, which will help impart more awareness of knowledge in good practice and may help overcome the reporting barriers. This survey-based study can serve as preliminary work, and the results can provide information on community pharmacists’ knowledge of reporting ADRs.
Supplemental Material
sj-docx-1-taw-10.1177_20420986241285930 – Supplemental material for Exploring the community pharmacist’s knowledge, attitude, and practices regarding adverse drug reactions and its reporting in the United Arab Emirates: a survey-based cross-sectional study
Supplemental material, sj-docx-1-taw-10.1177_20420986241285930 for Exploring the community pharmacist’s knowledge, attitude, and practices regarding adverse drug reactions and its reporting in the United Arab Emirates: a survey-based cross-sectional study by Javedh Shareef, Sathvik Belagodu Sridhar, Mullaicharam Bhupathyraaj, Atiqulla Shariff and Sabin Thomas in Therapeutic Advances in Drug Safety
Footnotes
Acknowledgements
The authors would like to thank the President of RAK Medical and Health Science University and the Dean of RAK College of Pharmacy for their encouragement and support. We would also like to express our special thanks and acknowledgment for the efforts of vetting committee members to provide necessary suggestions and guidance.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
