Abstract
Background:
Idiopathic pulmonary fibrosis (IPF) is a chronic, progressive, life-threatening lung disease with a global incidence of 0.09–1.30 per 10,000 individuals. Pirfenidone and nintedanib are the approved treatments for IPF.
Objectives:
This study evaluated the real-world safety and tolerability profiles of pirfenidone and nintedanib in IPF patients treated at the Mediterranean Institute for Transplantation and Advanced Specialized Therapies (IRCCS ISMETT). A comparative analysis was conducted based on the number, types, and severity of adverse drug reactions (ADRs) and to identify potential predictors of treatment discontinuation or ADR onset based on patient characteristics.
Design:
A retrospective observational study was conducted on 531 IPF patients treated at IRCCS ISMETT with either pirfenidone or nintedanib.
Methods:
Eligible patients were selected based on the logged monthly dispensations provided by the pharmacy service for both therapies. Covariates were extracted from electronic medical records (age, sex, body mass index, smoking history, comorbidities, forced vital capacity (FVC) %, diffusing capacity of the lung for carbon monoxide (DLCO) %, 6-minute walk test (6-MWT), polytherapy, oxygen therapy, drug switch, etc.). ADRs were categorized by severity and follow-up status, and further classified according to the Medical Dictionary for Regulatory Activities, specifying the Preferred Terms and the related System Organ Classes. Chi-square or Fisher’s exact test was used for categorical variables, and univariate and multiple logistic regression identified potential risk factors for ADR onset. Backward Stepwise logistic regression (BSLR) was used to determine independent variables associated with ADR occurrence.
Results:
The nintedanib group had more frequent ADRs related to gastrointestinal and hepatobiliary disorders, with nausea, diarrhea, anorexia, and weight loss as the most common. The pirfenidone group had more ADRs related to skin, nervous system, and vascular disorders, such as rash, nausea, dizziness, and blood pressure imbalances. Significant baseline differences between groups included age, smoking status, FVC (%), DLCO (%), and 6-MWT, with the nintedanib cohort showing worse baseline characteristics. A total of 450 ADRs were reported: 59.6% for nintedanib and 40.4% for pirfenidone. Independent variables that significantly increased the likelihood of experiencing ADR were drug change, treatment type, gender, and age.
Conclusion:
Identifying ADR predictors is essential for personalizing treatment strategies. Both pirfenidone and nintedanib are crucial in managing IPF, highlighting the need for further research to optimize personalized therapies and patient outcomes.
Plain language summary
Idiopathic pulmonary fibrosis (IPF) is a chronic, progressive, interstitial and life-threatening lung disease classified as a rare disease. Two medications, pirfenidone and nintedanib, are approved to slow the progression of IPF, but both can cause side effects. This study aimed to compare these treatments and identify factors that might predict which patients are more likely to experience side effects. Researchers at IRCCS ISMETT analyzed the medical records of 531 patients with IPF treated with pirfenidone or nintedanib over a period of eight years starting from May 2014. They looked at factors such as age, smoking history, lung function, and other health conditions to understand how these might influence the likelihood of side effects. The study found that patients taking nintedanib were more likely to experience digestive issues like nausea, diarrhea, and weight loss. In contrast, patients on pirfenidone were more prone to skin rashes, dizziness, and changes in blood pressure. Overall, side effects were reported more frequently with nintedanib. Certain factors made side effects more likely: being older, taking nintedanib instead of pirfenidone, switching treatments, and gender, with men being less affected. The nintedanib group also had worse lung function at the start of treatment compared to the pirfenidone group. These findings underline the importance of tailoring treatment to each patient’s needs to reduce side effects and improve outcomes. Both pirfenidone and nintedanib remain vital for managing IPF, but further research is needed to enhance personalized care and improve quality of life for patients.
Keywords
Introduction
Idiopathic pulmonary fibrosis (IPF) is a chronic, progressive, interstitial, and life-threatening lung disease of unknown etiology characterized by the irreversible decline in lung function, worsening dyspnea, and quality of life, with an estimated median survival of 3–5 years.1,2
Despite being classified as a rare disease, IPF is experiencing a rising global incidence ranging from 0.09 to 1.30 per 10,000 individuals, with prevalence estimates varying from 0.33 to 4.51 per 10,000 individuals, mostly in the elderly and male.3–5
The risk factors that can influence the onset of the pathology include age, sex, smoking history, environmental exposure, familial pulmonary fibrosis, and comorbidities (e.g., gastroesophageal reflux, chronic viral infections such as Epstein-Barr virus, hepatitis C, emphysema, lung cancer, pulmonary hypertension, sleep apnea, and coronary artery disease), and polytherapy.6–11
Lung transplantation can be considered an option for only a small percentage of patients, with the majority relying on antifibrotic therapy and supportive care.
Two drugs, pirfenidone and nintedanib, have been approved for the treatment of patients with IPF. Pirfenidone received a marketing authorization valid throughout the European Union (EU) on February 27, 2011, 12 and Nintedanib on January 14, 2015. 13
Nintedanib is a highly potent, orally available small-molecule tyrosine kinase inhibitor (TKI) that targets multiple receptor tyrosine kinases (RTKs), including VEGFR 1–3, PDGFR-α and -β, and FGFR 1–3.14,15 By competitively and reversibly binding to the ATP pocket of these receptors, nintedanib effectively disrupts key intracellular signaling pathways involved in the proliferation, migration, and transformation of lung fibroblasts.16,17
Pirfenidone is an orally bioavailable synthetic molecule that modulates TGF-β and TNF-α activity in vitro, inhibits fibroblast proliferation, reduces collagen synthesis, and attenuates fibrosis markers in animal models of lung fibrosis. 18
The marketing authorization of pirfenidone and nintedanib marked a significant advancement in the treatment of IPF, offering patients alternatives to supportive therapies and transplantation. 19 Clinical studies showed that both drugs can slow the decline of lung function, but with a certain degree of variability regarding safety and tolerability profiles.20–22
The CAPACITY and ASCEND trials assessed the impact of pirfenidone on the progression of IPF and found a favorable risk–benefit profile. The most prevalent adverse reactions reported were gastrointestinal events (such as nausea, dyspepsia, vomiting, and anorexia), skin disorders (such as rash and photosensitivity), and dizziness.18,23
The INPULSIS 1–2 studies were conducted to evaluate the efficacy and safety of nintedanib. Adverse reactions to nintedanib were commonly linked to gastrointestinal issues, with diarrhea being the most frequently reported symptom. 24
However, the length of observation and the characteristics of patients (including comorbidities, disease severity, use of concomitant medications, and mortality rate) eligible for pivotal randomized controlled trials differ substantially from those treated in clinical practice, often resulting in a distinct benefit–risk profile. To date, published data concerning the safety of pirfenidone and nintedanib in the post-marketing setting are still limited and suggest several safety issues occasionally associated with treatment discontinuation. 22 For this reason, further investigation into the real-world tolerability of both drugs is necessary.
The study aimed to evaluate the real-world safety and tolerability profiles of pirfenidone and nintedanib on the cohort of IPF patients treated at the Mediterranean Institute for Transplantation and Advanced Specialized Therapies (IRCCS ISMETT) through a comparative analysis between the two treatments based on the number, types, and severity of reported adverse drug reactions (ADRs) and a specific analysis aimed at identifying possible predictors of treatment discontinuation or onset of ADRs based on patients characteristics.
Materials and methods
Patients and settings
A single-center retrospective observational study was conducted on 531 patients diagnosed with IPF, treated at IRCCS ISMETT over a period of 8 years starting from May 2014 with pirfenidone or nintedanib. The data collection for this study was completed by March 2024. As required by the Italian Drug Agency (AIFA), diagnosis of IPF was assessed according to the guidelines of the American Thoracic Society, European Respiratory Society, Japanese Respiratory Society, and Latin American Thoracic Society guidelines as described by Richeldi et al.23-26
The inclusion criteria for both pirfenidone and nintedanib were as follows: age ⩾40 years, pulmonary fibrosis from IPF, insidious onset or unexplained exertional dyspnea for ⩾3 months, presence of usual interstitial pneumonia (UIP) pattern on high-resolution computed tomography or open lung surgical biopsy, video-assisted thoracoscopic surgical biopsy showing probable or definite UIP, or transbronchial biopsy not suggestive of another diagnosis, normal liver function or falling within Child-Pugh A (Child-Pugh B was also included for pirfenidone), mild or moderate disease severity for pirfenidone and mild to severe disease severity for nintedanib, percent predicted forced vital capacity (FVC% pred) ⩾50% with a forced expiratory volume at 1 s/forced vital capacity ratio ⩾70%, a percent predicted diffusing capacity of the lung for carbon monoxide (DLCO% pred) ⩾35% at screening for pirfenidone, 25 while a DLCO% pred ⩾30% at screening for nintedanib. 26
The exclusion criteria for both pirfenidone and nintedanib were as follows: diagnosis of pulmonary fibrosis from other etiologies (i.e., fibrosing interstitial lung disease with progressive phenotype, interstitial lung disease associated with systemic sclerosis, etc.), lung transplantation, and drug discontinuation in the first 6 months of treatment.
Both pirfenidone and nintedanib were prescribed as per AIFA criteria and the manufacturer’s recommendations.
Pirfenidone was titrated to a dose of three 267 mg capsules taken three times daily (totaling 2403 mg/day), as tolerated. Nintedanib was prescribed at a dose of 150 mg twice daily. From the beginning of the therapy, patients are required to undergo monthly blood tests (liver and kidney function tests, as well as a complete blood count) for the first 3 months. They should also have a medical examination every 3 months for the first year and subsequently as needed based on clinical requirements.
Data source and analysis
Eligible patients were selected based on the logged monthly dispensations provided by the pharmacy service for both therapies. The following covariates were extracted from the electronic medical record for each patient: age at IPF diagnosis, sex, body mass index (BMI), smoking history (classified as smokers, ex-smokers, those who had already quit smoking before starting the treatment, and non-smokers), presence of other comorbidities, FVC%, DLCO%, 6-minute walk test (6-MWT), environmental exposure, familial pulmonary fibrosis, polytherapy, oxygen therapy, and nintedanib versus pirfenidone or pirfenidone versus nintedanib drug switch.
The ADRs included in this study were those reported in the Italian National Pharmacovigilance Network. These were defined according to Directive 84/2010 of the EU as “adverse reactions in human beings, arising from the use of the medicinal product within the terms of the marketing authorization, as well as from use outside the terms of the marketing authorization, and adverse reactions associated with occupational exposure.” The causality of the suspected ADRs was not assessed in this study, as regulations stipulate that it must be carried out after the report by AIFA through the regional pharmacovigilance centers of each country.
All the ADRs were categorized by severity (severe, not severe) and follow-up status (recovered, not recovered), and further classified according to the Medical Dictionary for Regulatory Activities, version 23.0, specifying the Preferred Terms and the related System Organ Classes (SOCs). According to European Medical Agency guidelines, an adverse reaction is considered severe if it is life-threatening or has a fatal outcome; it requires inpatient hospitalization or extended existing hospitalization, it results in persistent or significant disability or incapacity, or it determines a congenital anomaly/birth defect. 27
Statistical analysis
Statistical analysis was performed using SAS version 9.4, (SAS Institute Inc., Cary, NC, USA). Continuous data were expressed as mean and standard deviation, and categorical data were expressed as count or percentage, unless otherwise specified. Association between categorical variables was analyzed using the Chi-square test or Fisher’s exact test, as appropriate. Univariate and multiple logistic regression were used to determine potential risk factors associated with the onset of ADRs and to adjust for patients’ characteristics. A stepwise logistic regression (BSLR; SAS code: selection = stepwise, slentry = 0.20, slstay = 0.15) was used to identify the independent variables associated with the occurrence of ADR. Covariates were treated as independent variables, while ADRs and drug discontinuation were treated as dependent variables. All p-values ⩽0.05 were considered statistically significant.
Ethics approval
This study was conducted in accordance with the Declaration of Helsinki and approved by the Institutional Review Board (IRRB/35/21) and Ethics Committee of IRCCS ISMETT. Informed written consent was obtained from all patients.
The reporting of this study conforms to the STROBE statement (Supplemental Material). 28
Results
Baseline characteristics
Our study included 531 IPF patients receiving antifibrotic drugs, of whom 252 (47.5%) were treated with pirfenidone and 279 (52.5%) with nintedanib.
In the pirfenidone-treated cohort, the mean age was 69.7 ± 6.8 years, with 75.4% (190/252) being male. Current smokers accounted for 5.2% of the group, 47.2% were ex-smokers, and 47.6% had never smoked.
In the nintedanib-treated cohort, the mean age was 71.6 ± 7.6 years, with 80.3% being male. Current smokers represented 10.4%, 36.2% were ex-smokers, and 53.4% had never smoked. The proportion of current smokers was double that of the pirfenidone group (10.4% vs 5.2%), and 53.4% had never smoked.
Furthermore, when comparing the two treatment groups (pirfenidone vs nintedanib) in relation to smoking status (current smoker, ex-smoker, non-smoker) in patients who experienced an ADR, the chi-square analysis did not reveal any statistically significant differences (p = 0.3916) between the three groups. Pre-treatment FVC, DLCO, and 6-MWT for the pirfenidone group were 81.9% ± 18.2, 54.6% ± 42.5, and 400.6 ± 111 meters, respectively.
For the nintedanib group, the pre-treatment FVC, DLCO, and 6-MWT were 77.02% ± 16.6, 43.5% ± 13.5, and 335.5 ± 175.3 meters, respectively.
Baseline characteristics revealed statistically significant differences between the pirfenidone and nintedanib groups in terms of age, smoking status, FVC (%), DLCO (%), and 6-MWT, indicating poorer baseline characteristics in the nintedanib cohort compared to the pirfenidone group.
Detailed patients’ baseline characteristics are shown in Table 1.
Patients’ baseline characteristics.

Data were analyzed using a t-test or a Chi-square test as appropriate (**p < 0.01; ***p < 0.001).
6-MWT, 6-minute walk test; BMI, body mass index; DLCO, diffusing capacity of the lung for carbon monoxide; FVC, forced vital capacity.
Adverse events
A total of 450 ADRs associated with pirfenidone and nintedanib were reported, of which 59.6% (268) referred to nintedanib and 40.4% (182) to pirfenidone. Among the 252 patients treated with pirfenidone, 29.76% (n = 75) experienced an ADR. Conversely, 47.3% (n = 132) of the 279 patients treated with nintedanib reported an ADR, suggesting a greater incidence of ADR with nintedanib. Regarding the severity of ADRs, the data suggest no significant difference between nintedanib and pirfenidone. Both drugs have a low incidence of severe ADRs, with pirfenidone showing a slightly higher, but not statistically significant, severity. A comprehensive summary of the total and severe ADRs by SOC is presented in Table 2.
Number and percentage of total and severe ADRs by SOC.

ADR, adverse drug reaction; SOC, System Organ Classes.
In particular, serious ADRs occurred in 3.77% (17/450) of cases: 8 in the nintedanib group and 9 in the pirfenidone group. Specifically, 2.99% (8/268) of patients in the nintedanib group experienced a serious ADR, compared to 4.95% (9/182) of patients in the pirfenidone group. The distribution of ADRs by SOC groups is reported in Figure 1.

Distribution of ADRs by SOC.
The nintedanib group showed a higher frequency of ADR for the gastrointestinal and hepatobiliary disorders SOC, with the most representative ADR being nausea, diarrhea, anorexia, and weight loss, respectively. By contrast, the pirfenidone group exhibited a higher frequency of ADR for the skin and subcutaneous tissue, nervous system disorders, and vascular disorders SOC (Table 2), with the most representative ADR being rash cutaneous, nausea and anorexia, dizziness and headache, and blood pressure imbalances, respectively.
The median onset of ADRs was 107 days, approximately 3.5 months after treatment initiation. Temporal distribution analysis revealed that 25% of ADRs occurred within the first 21 days, while 75% had emerged by 242 days (Figure 2).

Time of occurrence of ADR.
No significant difference was observed in the time to ADR onset between patients treated with nintedanib and those receiving pirfenidone. The median time to ADR occurrence was similar between the two treatments (109.5 days for nintedanib vs 107 days for pirfenidone). Moreover, the majority of ADRs were reported within the 75th percentile, with similar onset distributions (235 days for nintedanib vs 242 days for pirfenidone) (Figure 2).
For nintedanib, the most common ADRs, categorized under gastrointestinal disorders, were observed on average at 117 days, approximately 4 months after the introduction of therapy. By contrast, for pirfenidone, the most common ADRs, classified as skin and subcutaneous tissue disorders, were observed at around the fourth month.
A total of 88.43% of ADRs in the nintedanib group resulted in complete resolution of symptoms, compared to 98.90% in those treated with pirfenidone. The mean duration of ADRs until symptom resolution was 22 days for patients on nintedanib and 32 days for those receiving pirfenidone (Figure 3). Among the 75 patients who developed an ADR while on pirfenidone, 18.6% (14/75) underwent dose reduction, and 100% (14/14) of these patients experienced another ADR. For nintedanib, 72% (95/132) of patients who had an ADR underwent dose reduction, and 91.5% (87/95) experienced another ADR.

ADR duration.
The univariate logistic regression analyses for pirfenidone suggest that none of the examined covariates (age, gender, smoking status, BMI, DLCO, MWT6, FVC_p, polytherapy, exposure, family history, comorbidities, exercise-induced oxygen) significantly affect the likelihood of experiencing ADRs.
For nintedanib, univariate logistic regression analysis identified as statistical significant predictors of ADRs both for age (OR = 1.170 for a 5-year increase; 95%CI = 1.001–1.366; p = 0.0479) and gender (OR = 2.526; 95%CI = 1.365–4.675; p = 0.0032).
The stepwise logistic regression was conducted to identify the factors that can significantly influence the likelihood of experiencing an ADR, considering as independent variables all the patient baseline characteristics reported in Table 1, and the treatment group (pirfenidone vs nintedanib).
The independent variables included in the final model that statistically significantly increased the odds of experiencing ADRs were as follows: drug switch (OR = 3.266, 95%CI = 2.043–5.220, p ⩽ 0.0001), treatment (OR nintedanib vs pirfenidone = 2.350, 95%CI = 1.605–3.439, p < 0001), gender (OR = 0.449, 95%CI = 0.288–0.701, p = 0.0004), and age (OR = 1.393 for a 10-year increase, 95%CI = 1.084–1.791, p = 0.0097) (Figure 4).
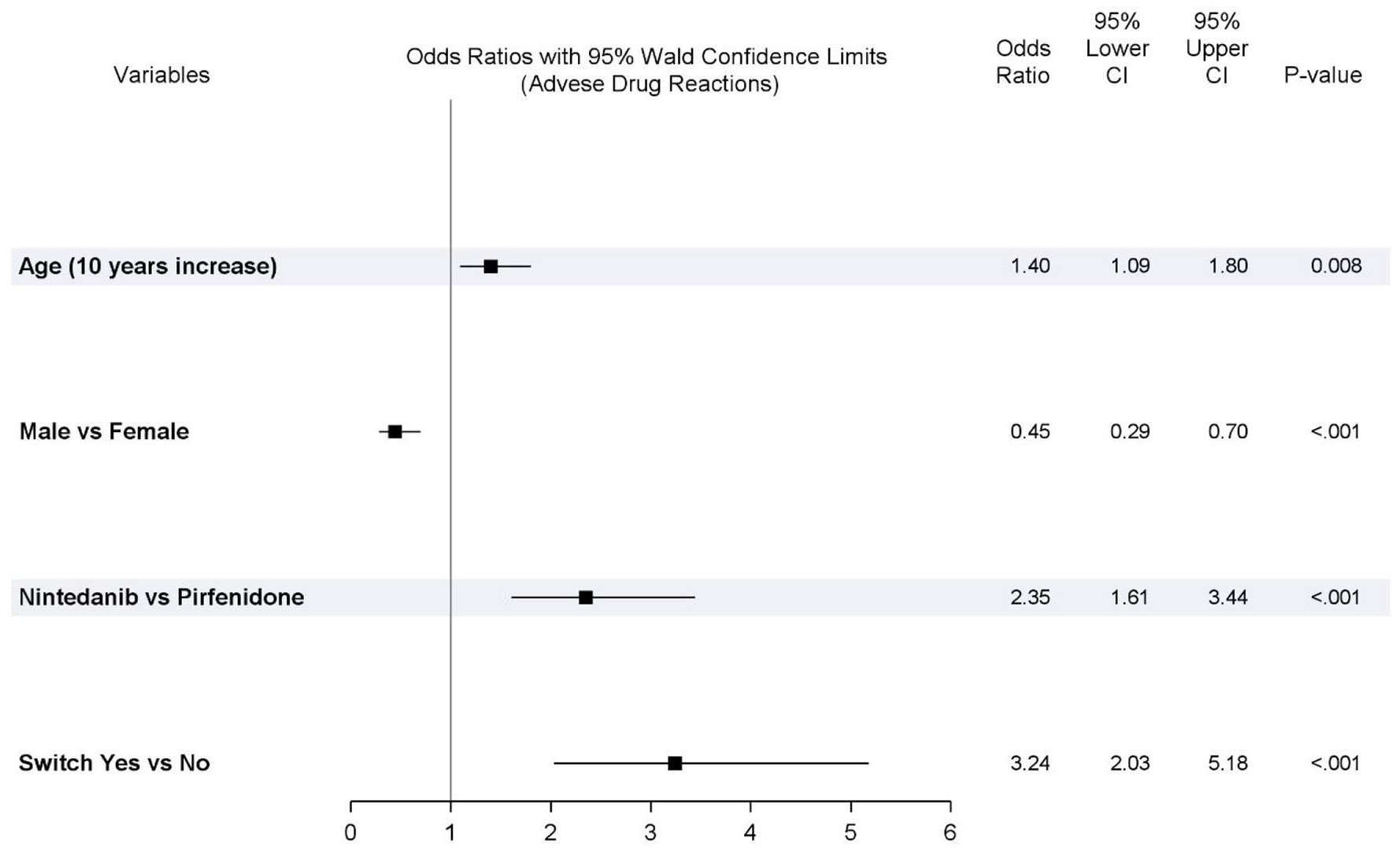
Factors affecting the probability of experiencing an ADR.
More in detail, the multiple analyses performed using stepwise logistic regression showed that, even when adjusting for differences in population baseline characteristics, treatment with nintedanib significantly increased the odds of experiencing an ADR (Figure 4).
Discussion
Our study assessed the safety and tolerability of pirfenidone and nintedanib in the treatment of IPF through insights derived from real-world data. Although the adverse effects of both medications are well-documented, there is still a need to establish a rational correlation between certain patient intrinsic variables and the onset of these effects. The two cohorts analyzed in this study reported adverse effects during pharmacological therapy that were consistent with those noted in the ASCEND and CAPACITY clinical trials for pirfenidone, as well as INPULSIS 1 and 2 for nintedanib.18,23,24 However, ADRs allowed for the identification of a non-significant difference between the two drugs in terms of causing serious adverse reactions. While pirfenidone appears to pose a higher risk of severe ADRs, the difference with nintedanib was not statistically significant. The incidence of gastrointestinal disorders was higher with nintedanib, whereas patients treated with pirfenidone exhibited a greater predisposition to skin and subcutaneous tissue disorders, which was an expected finding. By contrast, individuals on pirfenidone treatment experienced nervous system and vascular disorders, unlike those receiving nintedanib.
Baseline patient characteristics differed from those examined in other scientific studies, not only in individual intrinsic values within the cohort, such as age, sex, BMI, smoking status, and FVC, but also in several additional parameters. Environmental factors, such as the duration of exposure to asbestos in occupational and domestic settings, were considered alongside other important aspects such as concomitant polypharmacy, familial predisposition to IPF or other pulmonary diseases, comorbidities, and ongoing oxygen therapy. 29
In this study, the comparative analysis of data obtained from the two antifibrotic treatments aimed to identify potential predictors for treatment discontinuation or the onset of ADRs based on patient characteristics. Multiple logistic regression applied to the collected data yielded OR that revealed potential risk factors associated with the occurrence of ADRs. Factors such as age, gender, pharmacological switching (from pirfenidone to nintedanib and vice versa), and being in therapy with nintedanib, compared to pirfenidone, could be considered predictive elements for the emergence of certain adverse effects in patients, potentially leading to treatment discontinuation.
Some reflections on the study’s results can be made by comparing our data with those obtained from other scientific works. Notably, analyzing nintedanib often reveals a significant association between patient age and the occurrence of ADRs. In a retrospective review of clinical records for subjects prescribed the antifibrotic for IPF treatment, multiple logistic regression indicated that age ⩾70 years was one of two characteristics associated with treatment discontinuation, alongside congestive heart failure (OR = 2.21, 95% CI = 1.04–4.69, p = 0.04). 16 This significant finding was replicated in a recent monocentric observational retrospective study, where univariate analysis suggested that age ⩾75 years, baseline BMI ⩽25, and female sex were risk factors for antifibrotic therapy discontinuation. 20 In our work, we not only reaffirmed the average age at which patients may exhibit greater vulnerability to treatment discontinuation following the development of adverse events but also noted an increased probability in relation to advancing age (OR = 1.393 for a 10-year increase, 95%CI = 1.084–1.791, p = 0.0097). However, some studies failed to demonstrate a significant correlation between age and the development of ADRs, which could be attributed to the use of a cohort characterized by a lower mean age of enrolled patients (67.4 ± 7.8 vs 71.6 ± 7.6).30,31
Beyond age, another factor to consider as a potential predictor of ADRs is the patient’s sex. Multiple logistic regression revealed a statistically significant association for this parameter regarding the occurrence of adverse events in female subjects treated with nintedanib or pirfenidone. This variable is featured in most scientific works. A multiple analysis conducted in a monocentric observational retrospective study identified female sex as an independent predictor of treatment discontinuation within 12 months, based on a cohort with baseline characteristics similar to those of the patients enrolled in our study (76% male vs 80%). 20
Pharmacological switching refers to the therapeutic transition where one medication is replaced with another. This can occur due to a lack of therapeutic response following disease progression or, as in our case, due to the reporting of specific ADRs. This parameter was also classified in our work through stepwise logistic regression as an independent variable related to early discontinuation of treatment with nintedanib (OR = 3.266, 95%CI = 2.043–5.220, p ⩽ 0.0001). According to our analysis, these findings are supported by scientific literature. In a recent study, an increase in ADRs and discontinuation of second-line treatment were observed in patients who switched from pirfenidone to nintedanib, primarily due to adverse effects such as liver damage and anorexia with weight loss. 32 However, our results are inconsistent with another retrospective study in which the reverse switch from nintedanib to pirfenidone was well-tolerated by most patients (90.6%). Patients switched to pirfenidone due to gastrointestinal adverse effects from nintedanib showed stable %FVC values 6 months post-switch. 33
Other parameters that assume clinical relevance in predicting the development of ADRs from antifibrotics used in IPF include BMI, performance status, disease stage (III or IV), %FVC, and DLCO values.20,30,34–36 Nonetheless, in our study, these clinical predictors did not yield a significant difference, allowing correlation with the discontinuation of pharmacological treatment. In a retrospective study, univariate logistic regression analysis indicated that BMI and %FVC were risk factors for early treatment cessation. 34 However, contrasting our findings, the cohort in that particular case was characterized by lower BMI (21.4 vs 27.6 kg/m²) and a notable difference in %FVC values among enrolled subjects (67.9% vs 77.02%). A similar consideration can be made by analyzing another prospective observational study, where BMI was found to be a clinical predictor of ADRs if <20 kg/m². 29 Malnutrition, particularly in geriatric patients, may become a particularly relevant aspect to consider when administering antifibrotics. Scientific evidence indicates that the risk associated with malnutrition is significantly correlated with an increased risk of antifibrotic therapy discontinuation. 37 Although our study did not replicate these results, possibly due to the inclusion of a cohort with a higher average BMI, a similar trend was consistently observed in another study. 38
The predisposition to ADRs in patients with low %FVC using antifibrotics is frequently highlighted in scientific literature.34,35 However, in our study, this parameter was not selected as a clinical predictor of ADRs since the cohort treated with nintedanib exhibited a baseline mean %FVC of 77.02, higher than the predictive value obtained through multiple analyses in other studies (FVC < 70%). 35 Nevertheless, a somewhat inconsistent result emerged from a retrospective study indicating that clinical characteristics associated with disease severity, such as %FVC, %DLCO, and the use of home oxygen, showed no significant association with treatment discontinuation. 21 Despite the enrollment of subjects with %FVC values lower than the cutoff proposed in the studies, the development of ADRs did not depend on this variable. This aspect was also observed in treatment with pirfenidone. 38
An essential aspect of managing IPF involves selecting the appropriate antifibrotic treatment, either nintedanib or pirfenidone, based on their respective tendencies to cause ADRs and influence treatment continuation. A retrospective observational study conducted at a single center found that while nintedanib frequently leads to gastrointestinal issues such as diarrhea and nausea, these side effects rarely result in therapy discontinuation. 39 By contrast, pirfenidone is associated with more challenging ADRs, particularly skin reactions and behavioral changes, which can be difficult to manage despite existing treatment guidelines. Consequently, patients on pirfenidone demonstrated a higher likelihood of discontinuing treatment within 48 months. In our analysis, we observed that nintedanib and pirfenidone exhibit different tolerability profiles. Nintedanib tends to cause more frequent ADRs, though they are typically less severe, whereas pirfenidone, despite causing fewer ADRs overall, is linked to more severe reactions, although this difference was not statistically significant. However, the occurrence of severe ADRs remains low for both drugs. Given these findings, the selection of antifibrotic therapy should consider early identification of individual patient risk factors. Tailoring treatment to these factors could improve long-term disease management, enhance patient adherence, and reduce the incidence of adverse events in IPF therapy.
However, while this study effectively reflects a real-world population and provides valuable insights into the safety and tolerability of antifibrotic therapies, its observational design may carry inherent limitations. These include potential biases related to patient selection, reporting, and measurement. Differences in follow-up duration and unmeasured confounding factors may further impact the interpretation of results.
Nonetheless, these findings offer a valuable contribution to the current understanding of the adverse effects of these treatments in routine clinical settings.
Conclusion
This study highlights the need to evaluate the long-term tolerability of pirfenidone and nintedanib in the treatment of IPF. Identifying predictors of ADRs is essential for personalizing treatment strategies. Both medications play a significant role in the management of IPF, emphasizing the necessity for further research aimed at improving personalized therapy and optimizing patient outcomes.
Supplemental Material
sj-docx-1-taw-10.1177_20420986251341645 – Supplemental material for Real-world insights into safety, tolerability, and predictive factors of adverse drug reactions in treating idiopathic pulmonary fibrosis with pirfenidone and nintedanib
Supplemental material, sj-docx-1-taw-10.1177_20420986251341645 for Real-world insights into safety, tolerability, and predictive factors of adverse drug reactions in treating idiopathic pulmonary fibrosis with pirfenidone and nintedanib by Alessio Provenzani, Daniele Leonardi Vinci, Miriam Alaimo, Salvatore Di Maria, Fabio Tuzzolino, Gaetano Floridia, Roberta Di Stefano, Anna Carollo, Adriana Callari, Piera Polidori and Patrizio Vitulo in Therapeutic Advances in Drug Safety
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
