Abstract
Background:
Bibliometric analyses have been used previously to study the measures of quality and impact of research performed in several health-related areas such as adverse drug reactions (ADRs) and pharmacovigilance (PV), etc. This method can assess the research performance of publications quantitatively and statistically. There is no evidence of bibilometric studies analyzing ADRs and PV from Nepal. Therefore, the present study aimed to assess scientific output on ADRs and PV-related research activities in Nepal using a bibliometric analysis of publications from 2004 January to December 2018, that is, 15 years.
Methods:
A systematic search was conducted in PubMed, Web of Science, Google Scholar, Scopus and Nepal Journal Online (NepJOL) databases. ‘Adverse Drug Reactions‘ or ‘ADRs‘ or ‘ADR‘ or ‘Adverse drug reaction‘ or ‘AE‘ or ‘Adverse Event‘ or ‘Drug-Induced Reaction‘ or ‘Pharmacovigilance‘ or ‘PV‘ and ‘Nepal‘. The search covered 15 years (January 2004 to December 2018) of study on ADRs and PV in Nepal. Only articles retrieved from databases were included, whereas published/unpublished drug bulletins, pharmacy newsletters and thesis were excluded. The articles thus retrieved were recorded, and thereafter analyzed. Word count code was used for the analysis of keywords used in the retrieved articles.
Results:
A total of 124 articles were retrieved, with the highest rate of publications in 2006 and 2007, with 16 papers each. Among the articles, 10 (8.1%) were published in Kathmandu University Medical Journal (KUMJ). Single papers were published in 38 different journals. Brief reports (1.6%), case reports (31.2%), case series (0.8%), education forums (0.8%), letters to the editor (5.6%), original research articles (41.9%), review articles (9.7%), short communications and short reports (8.1%) on ADRs and PV were recorded. Out of 124 papers, 52 (41.9%) were original research publications. The majority (74.1%) of research was done in the category of ADR incidence, types, prevention, and management, followed by policy and suggestions for strengthening national and regional pharmacovigilance centers of Nepal (14.5%).
Conclusions:
During the study years, there was an increase in scientific publications on drug safety. A total of 124 published articles were found during bibliometric analysis of ADRs and PV research activities in Nepal.
Introduction
Adverse drug reactions (ADRs) are ‘a response to a medicine which is noxious and unintended, and which occurs at doses normally used in man’. 1 Globally, ADRs are significant cause underlying morbidity and mortality in hospital.2–4 The World Health Organization (WHO) has set up a pharmacovigilance (PV) unit and developed a PV strategy to coordinate ADR detection, reporting, and monitoring at a global level, which is managed through the Uppsala Monitoring Center (UMC) based in Sweden. 5 PV is defined as ‘the science and activities relating to the detection, assessment, understanding, and prevention of adverse effects or any other possible drug-related problems’. 6 For healthcare organizations and providers, PV is a program to monitor the adverse effects of medications and prevent its occurrence in patients, and to promote patient safety and wellbeing. 7 Nepal joined the WHO Pharmacovigilance program in July 2006. 8 A National Pharmacovigilance Center was set up at the Department of Drug Administration (DDA) to monitor PV activities in Nepal. Presently, there are 15 regional PV centers in Nepal. As of now, 831 ADR reports have been reported to the National Pharmacovigilance Center at the DDA. 9 Several studies have been performed in Nepal on ADRs and PV. Review and analysis of these studies will enhance the understanding of PV in Nepal, and suggest ways to improve PV practice.
As bibliometric analysis measures the quality and impact of research yields, 10 it might be helpful to obtain information on ADR reporting and the various activities run by PV centers in Nepal. Evaluation is generally performed by measuring different metrics such as citation counts, H-index, field-weighted citation impact, outputs in top percentiles, journal impact factor, cite score, SCImago journal rank, and Scopus SNIP. 11
Only a limited number of bibliometric analyses regarding medication errors and adverse drug events have been carried out. Hung-HC reported a bibliometric analysis of medication errors and adverse drug events studies from 1961 to 2013. 12 The database search revealed 3343 and 3342 documentations of medication errors and adverse drug events, respectively. Similarly, utilizing bibliometric visualization techniques, Rodrigues et al. 13 carried out a study that analyzed the pattern of literature in patient safety that suggests research directions for the future. The latter study showed the frequency of published articles and types of publications regarding PV and ADRs, 14 whereas our present study focuses on the impact of those published papers in different journals.
Bibliometric review is different from systematic review, and is considered a good tool for the evaluation of the pattern and advancement of research activities in a certain field over a period of time utilizing the literature system and available literature in certain area as research objects, and breaking down the literature quantitatively and qualitatively. 15 Bibliometric analysis examines bibliographical works within a specific field, theme, institution, or nation-state. It can depict changes and developments in a specific field over a period of time through the analysis of research publications in that field during that given time. 16 This can be useful for investigators in assessment of research results, finding gaps, and suggesting that more research be carried out in areas that have not yet been explored, or for future pathways. 17
In Nepal, not much information is available on PV research and other activities. One can find studies on PV in Nepal in different Nepalese and international journals. However, no information is available on the nature, pattern, outcomes, and other details of such studies. As a result, the state of PV research, research gaps, and practical recommendations to improve PV practice are lacking. To our knowledge, this is the first effort from the Nepalese pharmacy sector to use a bibliometric approach to explore PV- and ADR-related research activities in Nepal. This bibliometric analysis is important in the field of pharmacy practice since it will provide a comprehensive overview of at current research trends on PV and ADRs in Nepal.
Methods
Study design
This study is a bibliometric review of PV and ADRs in Nepal.
Search strategy and database used
A systematic search was conducted in PubMed, Web of Science (WoS), Google Scholar, Scopus, and Nepal Journal Online (NepJOL) databases using the search terms ‘Adverse Drug Reactions’ or ‘ADRs’ or ‘ADR’ or ‘Adverse drug reaction’ or ‘AE’ or ‘Adverse Event’ or ‘Drug-Induced Reaction’ or ‘Pharmacovigilance’ or ‘PV’ and ‘Nepal’. The search terms included terms describing and covering all fields in PV and ADRs using MeSH terms in PubMed (Table 1) combined with ‘Nepal’. A systematic search utilizing both Nepali and English databases was performed. The search covered the period from January 2004 to December 2018 using Medline/PubMed, Index Copernicus, Web of Science, and Scopus. Google and Google Scholar search engines were also used. The Nepalese electronic database included was NepJOL, where 150 journals are listed.
MeSH keywords (for search in electronic databases both in English).

Inclusion and exclusion criteria
Different article types, such as original research articles, case reports, review articles, short communications, letters to the editor, brief reports, case series, and education forums by Nepalese authors are included in this study. Articles in the English language, with publication dates between January 2004 and December 2018, relevant to PV and ADRs, and research (or review) based on the Nepalese scenario, were included.
Drug bulletins, articles not published in online journals, online pharmacy newsletters, conference presentations, etc. were excluded. No manual search was performed in libraries of different universities. Papers published in local language were also excluded. We excluded papers published before January 2004 and after December 2018. A flowchart of the operational framework of a bibliometric study is shown in Figure 1.
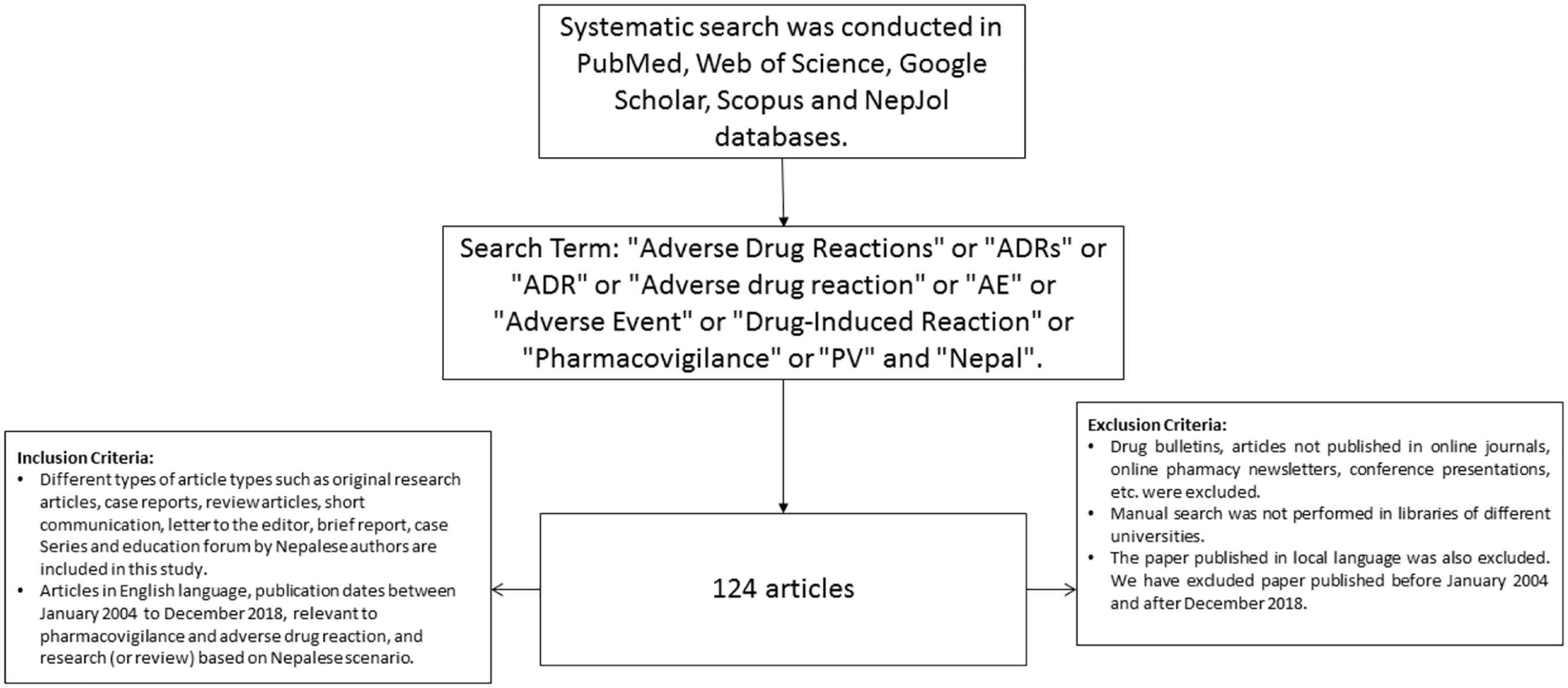
Flowchart of the operational framework of a bibliometric study.
Data collection and validation
On the basis of inclusion and exclusion criteria, two authors independently extracted data from all included publications, including titles, keywords, publication dates, authors, publishing journals, sum of citations, H-index, and so on. The information obtained from the different database were entered in MS-Excel 2010. All the information is again checked and screened by the other two authors. For the validity of the search strategy, two co-authors manually reviewed the 124 articles from the study period.
Data analysis and visualization
Databases were exported to Excel, and then to IBM SPSS Statistics 26 software package (IBM Corp. Release 2019. IBM SPSS Statistics for Windows, Version 26.0. Armonk, NY: IBM Corp.). Descriptive analyses of key descriptive study variables were also conducted and are summarized in the tables and figures.
The data exported included:
title of journal,
title of paper,
author’s name,
number of authors,
published year,
keywords,
journal impact factor (IF),
International Standard Serial Number (ISSN), and
H-index of the journals
On the basis of the main objectives(s) of the study, articles were classified by two independent reviewers into five categories:
ADRs (incidence, types, prevention and management);
policy and suggestions for strengthening national and regional PV centers of Nepal;
knowledge, attitude, and practice of PV and ADR among healthcare professionals and consumers;
PV and ADR educational interventions among health care professionals and consumers; and
drug withdrawal due to ADRs.
ADR incidence, types, prevention, and management is further divided into domains:
i. Cutaneous adverse drug reactions;
ii. ADRs due to generally prescribed drugs;
iii. ADRs related to antitubercular medicines;
iv. ADRs related to chemotherapy drugs;
v. ADRs due to antihypertensive medicines;
vi. ADRs related neuropsychiatric manifestations;
vii. ADRs due to antileprotic medicines;
viii. ADRs related to antileprotic medicines;
ix. ADRs related to oral hypoglycemic agents;
x. ADRs due to anti filarial medicines; and
xi. others [ADRs such as hematological reactions, gastrointestinal (GI) distress].
Analysis of keywords obtained from all articles included in this study was done using the Word Cloud generator.
Ethical approval
The data were downloaded from numerous databases such as PubMed, Scopus, and secondary data. The study did not involve any interactions with human subjects or human materials. There were no ethical questions about the data. Approval of an ethics committee was not necessary.
Results
A total of 124 articles were reviewed and analyzed.18–144
Annual number of ADRs and PV publications in Nepal
Figure 2 shows the distribution of papers on PV and ADRs by year of publication. Of the articles retrieved, it was found that the years 2006 and 2007 recorded the highest number of publications, that is, 16 publications, whereas in 2004 there were only two publications. The increase in the number of articles published in the period under consideration is shown in Figure 2.

Annual number of ADR and PV publications in Nepal.
Average author per article
The average number of authors per paper was 4.10 [standard deviation (SD) = 1.68]; 28 papers were authored by 3 authors, whereas 1 paper was written by 10 authors. Only three articles were written by a single author. Table 2 lists the average number of authors per article.
Average number of authors per article.

List of journals with frequency of papers published with IF, ISSN, and H-Index
The distribution of different journals (n = 63) in which the reviewed articles appeared, papers with journal impact factor, and ISSN are listed in Table 3. The highest number of articles were published in the Kathmandu University Medical Journal (KUMJ; 8.0%) followed by the Journal of Institute of Medicine (JIOM; 5.6%). A total of 38 single papers were published in different scientific journals. Online search was performed using Scientific Journal Ranking (SJR) for searching ISSN, H-index number, and SJR factor or impact factor of the journal. For those journals not found on SJR website, the journal homepage containing ISSN and impact factor was considered. Those not found in SJR or journal page were listed as not available (NA).
List of journals with IF and ISSN.

NA, not available; –, not given.
IF, impact factor; ISSN, international standard serial number; SJR, scientific journal ranking.
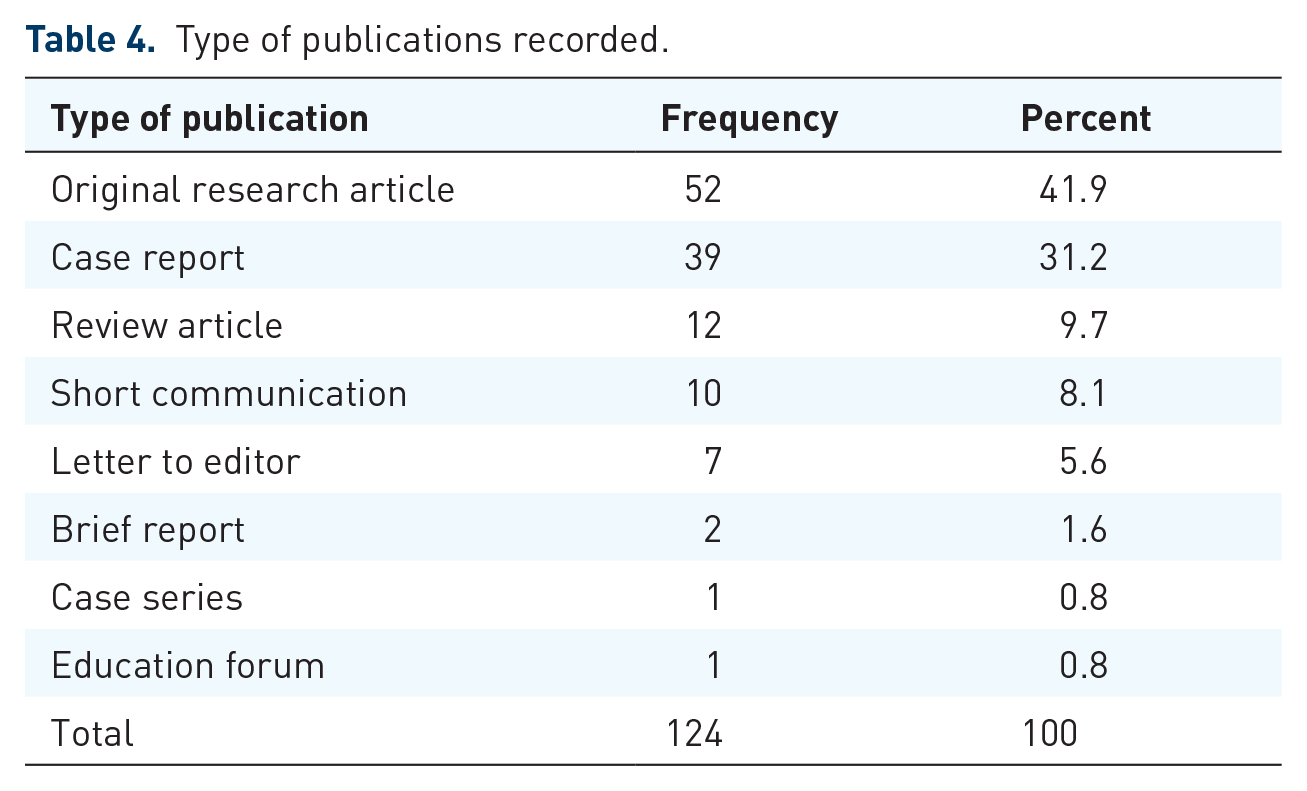
Different types of publications recorded
Different types of articles were recorded, as shown in Table 4. Brief reports, case reports, education forums, letters to the editor, original research articles, review articles, short communications, and short reports on ADR and PV were published. The number of original research articles published was 52 (41.9%), followed by 39 (31.2%) case reports.
Type of publications recorded.
Research categories and research domains
The different types of articles published in different categories and research domains are shown in Table 5. Most (74.1%) research was done in the category of ADRs (incidence, types, prevention and management) followed by policy, and suggestions for strengthening national and regional PV centers of Nepal (14.5%).
Research categories and research domains.

ADRs, adverse drug reactions.
Analysis of keywords using the Word cloud generator
The image generated based on the keywords extracted from the articles using Word cloud generator can be seen in Figure 3. As an example, keywords like ‘adverse drug reactions’, ‘pharmacovigilance’ and ‘Nepal’ appear larger than other words, demonstrating that they have been mentioned in the articles most commonly.

Word cloud generator of PV and ADR keywords.
Discussion
The results of this study show that 124 articles were published in different journals covering ADR and PV in the Nepalese context. This is the output from the past 15 years, covering 2004 to 2018, and is the first article analyzing the bibliometric of ADR and PV in Nepal.
Only 52 articles were original research articles based on the practice of PV in Nepal. The annual number of publications varied and was skewed over the years, with only two papers in the year 2004 and 16 publications in the year 2006 and 2007. Pattern of ADR reporting, especially to the regional PV centers and teaching hospitals, has been quite consistent.22,94 However, studies on different aspects of ADR and PV have not been carried out in a similar fashion. This may be because not all secondary and tertiary hospitals have PV centers of their own and PV has not received the research focus it should.
Most of the articles were descriptive in nature and were published in Nepalese journals, none of which have an impact factor. Furthermore, some of these journals were also non-indexed. None of the studies involved randomized controlled trials or were interventional studies. It shows that more rigorous studies with sound research designs are required to evaluate the overall state of PV and ADR in Nepal and improve it further.
Analyses of the keywords of these articles showed that the focus of the articles was on PV, ADR, Nepal, consumers, adverse effect, chemotherapy, antibacterial agents, casualty assessment, Naranjo algorithm, etc. The articles were focused on the PV programs and ADR assessment, reporting, ADRs due to antibiotics, chemotherapy and other medicines, and consumer and community involvement in the PV process.
By and large, there is a need to carry out more studies on ADR and PV in Nepal to improve the state of PV and promote quality use of medicines.
Strengths and limitations of this study
To the authors’ knowledge, this study is the first bibliometric research to assess research activity in the field of PV and ADRs conducted in Nepal. This research highlights trends in drug safety research patterns in Nepal. However, this study has a number of limitations, which need to be listed. First, this study is limited only to research activities of PV and ADRs conducted in the country of Nepal, and PV activities in Nepal are still in their infancy. We have not included drug bulletins or pharmacy newsletters, and we did not perform a manual search of thesis or projects undertaken by undergraduates, graduates, and PhD students in libraries of the different colleges and universities of Nepal. Second, the search strategy used in the current bibliometric study was held to be trustworthy and valid; however, the search strategy probably contained non-negligible errors that cannot be ignored completely. Third, the latest published articles, which were published after December 2018, have not been included in this study. It could have added to the study. Another significant limitation is that the keywords used might not be fully inclusive of possible keywords and there is the possibility of false-positive and false-negative outcomes. Nevertheless, the authors did their best to validate the data by reviewing manually, and tried to make an inclusive assessment of PV and ADRs research productivity in Nepal that ideally will be a positive and progressive expansion to the literature on PV and ADRs.
Take-home messages
As the significant focus of this study was Nepal only, our future research plan and our recommendations for the scientific community who are interested in ADRs and PV are as follows. First, a need to carry out in-depth systematic reviews of the literature by PV categories, ADR categories, by adverse events categories, by other categories (e.g. drug–drug interactions, medication errors), and by geographical demarcation (e.g. pharmacovigilance and ADR reporting activities within the South Asia or Asia region). Geographical demarcation need not be limited to any specific region; the PV activities of the world can be taken. Second, drug safety is an important area and Nepalese healthcare professionals should be aware of the noxious effects of drugs. Drug regulatory authorities like the DDA as well as consumers also need to be aware of safe drug use. Awareness of drug safety among all stakeholders, such as healthcare providers, regulatory authorities and consumers, aids in early detection and prevention of ADR. Furthermore, studies on different aspects of the ADR process, monitoring and outcomes can provide insights into the state of PV in Nepal and can provide recommendations for further improvements. Third, healthcare professionals and policymakers should focus on PV and ADR reporting so that the national PV system can be improved and the quality use of medicines can be promoted. Fourth, there is an increase in collaborative work in research and it is important to analyze links between scientific researchers in different countries. Further, investments in research should be increased in international research collaborations, and research networks should be encouraged to prioritize research in drug safety. Finally, the results of this study will form a useful baseline to be used by scientific researchers globally. During this bibliographic research, authors could not find articles related to transnational PV with information on new drug safety signals and regulatory actions. Hence, it is important for health professionals and policymakers in each country to enhance PV mechanisms that could potentially benefit regulatory decision making.
Conclusion
This study has summarized research trends in Nepal concerning PV and ADR. It shows that there is a need to study and evaluate ADR and PV in Nepal to improve PV practice and promote quality use of medicines. This study presents a bibliometric overview of publications on PV and ADRs in Nepal through the analysis of temporal research productivity, analysis of types of research papers, and providing a list of journals where these papers are published.
Footnotes
Acknowledgements
The author(s) would like to acknowledge João Batista Ferreira, Department of Management and Economics, Universidade Federal de Lavras (UFLA), Lavras/MG, Brazil for his contribution to the analysis of keywords.
Author contributions
SS and KD conceived the study. SS, KD, BKC, and SP developed the methods. SS, KD, and SP collected, checked and screened the obtained information. SS and KD analyzed the data. SS, KD, BKC, SP, and MIMI wrote the manuscript. All the authors edited and revised the manuscript, and read and approved the final version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Ethical approval and consent to participate
No ethical approval was needed as this was an analysis of available published research. No authors were communicated for additional information concerning their publications.
Consent for publication
Not applicable.
