Abstract
Background:
Currently, the factors influencing small bowel bleeding caused by nonsteroidal anti-inflammatory drugs (NSAIDs) remain unclear.
Objectives:
This study aimed to assess NSAID-associated small bowel bleeding and evaluate the impact of other drugs on it through a pharmacovigilance study, thereby providing valuable insights for clinical practice.
Design:
Data on NSAID-associated small bowel bleeding were retrospectively extracted from two public adverse drug reaction databases—the Food and Drug Administration’s (FDA) Adverse Event Reporting System (FAERS) and the Japan Pharmaceuticals and Medical Devices Agency’s Adverse Drug Event Reporting (JADER)—from 2004 to 2023 for further analysis.
Methods:
The reporting odds ratio (ROR), a pharmacovigilance technique, was employed to identify signals of adverse reactions, and the Chi-square test was utilized to assess differences between groups.
Results:
Multiple NSAIDs associated with small bowel bleeding were identified in both databases. In the drug combination analysis, no significant differences in the risk of small bowel bleeding were found between NSAIDs combined with proton pump inhibitors (PPIs) and NSAIDs alone in FAERS (all p > 0.05). Decreasing risks were found when multiple NSAIDs were combined with rebamipide or probiotics compared to NSAIDs alone in JADER (p < 0.05 and ROR < 1). In subgroup analyses of age and sex, older adults and males who used aspirin showed higher risk signals in both databases (all p < 0.05 and ROR > 1).
Conclusion:
PPIs did not show a significant impact on NSAIDs-associated small bowel bleeding, while rebamipide and probiotics may exhibited a preventive effect against NSAIDs-associated small bowel bleeding. Older adults and males constituted risk factors for aspirin-associated small bowel bleeding.
Plain language summary
This pharmacovigilance study analyzed U.S. and Japanese drug adverse event databases (2004–2023) to evaluate NSAID-related small bowel bleeding risks. Results showed that combining NSAIDs with proton pump inhibitors (PPIs) did not reduce bleeding risk, but pairing NSAIDs with mucosal protective agents (e.g., rebamipide) or probiotics significantly lowered risk in Japanese data. Elderly males emerged as a high-risk population for NSAID-related small bowel bleeding, requiring heightened clinical attention. These findings highlight the potential of mucosal protective agents for preventing NSAID-induced intestinal injury, with probiotics showing particular promise for further exploration. Meanwhile, the impact of PPIs in this risk warrants additional validation.
Introduction
Small bowel bleeding is defined as bleeding from the jejunum and ileum below the ligament of Treitz. Nonsteroidal anti-inflammatory drugs (NSAIDs) cause damage to the mucosa of the gastrointestinal tract by inhibiting the enzyme cyclooxygenase (COX), leading to a decrease in the synthesis of prostaglandin (PG) analogs. 1 Small bowel endoscopy has revealed that NSAIDs often damage the small intestinal mucosa, with approximately 50% of patients on long-term use experiencing mucosal breakdown. 2 In the consensus of the American College of Gastroenterology, it is also recognized that the use of NSAIDs constitutes a risk factor for small intestinal bleeding. However, there is currently no definitive conclusion regarding whether proton pump inhibitors (PPIs) or other medications can mitigate the risk of this adverse reaction. 3 Therefore, a pharmacovigilance analysis of NSAID-induced small bowel bleeding is necessary. This analysis can help guide clinicians in early prevention and intervention to improve patient outcomes.
In recent years, large real-world adverse drug reaction databases, such as the U.S. Food and Drug Administration’s (FDA) Adverse Event Reporting System (FAERS) and the Japan Pharmaceuticals and Medical Devices Agency’s (PMDA) Adverse Drug Event Reporting (JADER), have accumulated a vast number of reported adverse drug reaction events. These adverse event (AE) reports typically document reactions that occur during clinical drug therapy and are considered more authentic and accurate for reflective clinical practice. 4 These databases are now being recognized as viable alternatives to randomized controlled trials for preventive drug discovery.
The objective of this study was to evaluate the impact of the PPIs, rebamipide, probiotics, gender, and age on NSAID-associated small bowel bleeding using data from FAERS and JADER.
Materials and methods
Data collection and pre-processing
The current study collected FAERS data from the FDA website (http://www.fda.gov/) from Q1 2004 through Q3 2023 and from the PMDA website (https://www.pmda.go.jp/index.html) from Q1 2004 through Q2 2023 JADER data. AEs in both databases were coded according to MedDRA preferred terms (PTs). The PTs for AEs related to small intestinal hemorrhage (SIH) selected for this study were “small intestinal ulcer hemorrhage” and “small intestinal hemorrhage.” Drugs associated with AEs were assigned different roles in the AEs (primary suspicion, secondary suspicion, concomitant, interaction), and the case was only included if the drug was listed as a primary suspicion (PS) for SIH . The drug screening process is shown in Figure 1.
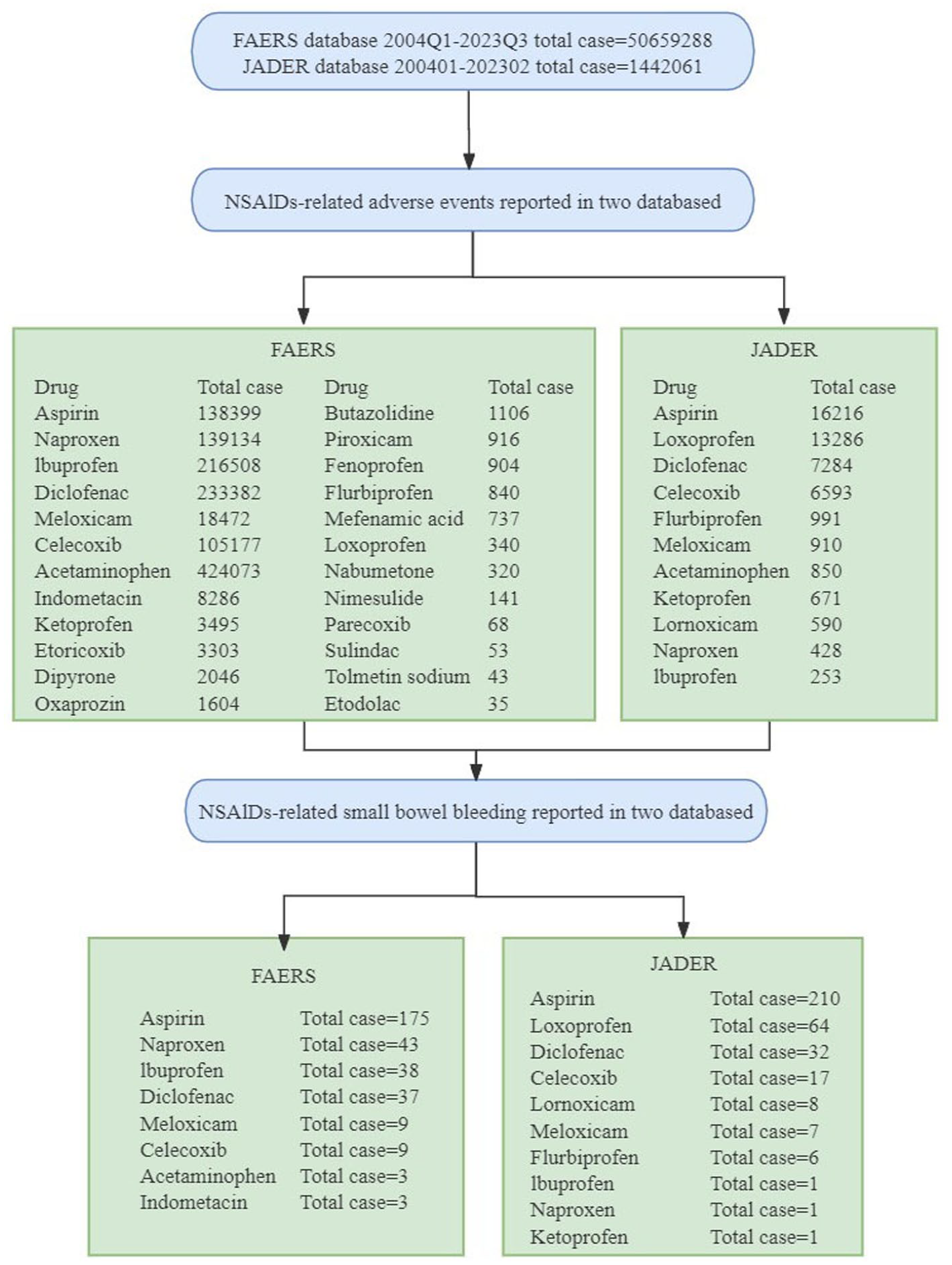
Adverse drug reaction screening flowchart.
Descriptive analysis
The current study undertook a descriptive analysis of the clinical characteristics of reported small bowel bleeding associated with NSAIDs, which included gender, age, outcomes, and other clinical features. AE outcomes in FAERS encompassed Death (DE), Life-threatening (LT), Hospitalization (HO), Disability (DS), and Others (OT), while JADER outcomes included Death, Unrecovered, Sequelae, Mild recovery, and Recovery.
Signal detection
Disproportionate analysis is commonly utilized in pharmacovigilance as a tool to evaluate the potential association between specific AEs and particular drugs. Among these methods, the reporting odds ratio (ROR) is widely employed for detecting adverse drug reaction events. When the number of reported AEs is at least 3 and the lower limit of the 95% confidence interval (CI) of the ROR exceeds 1, it signifies that a specific drug has a more pronounced signal for the onset of a particularly adverse reaction compared to all other drugs. 5 The following formula was utilized in this study to calculate the ROR:
where “a” denotes target AEs of the target drug, “b” denotes other AEs of the target drug, “c” denotes target AEs of other drugs, and “d” denotes target AEs of other drugs.
Following this, NSAIDs with positive signals for small bowel bleeding were further scrutinized. Initially, these selected NSAIDs underwent drug combination analysis to examine the impact of NSAIDs on small bowel bleeding when combined with PPIs, rebamipide, and probiotics. In the comparative analysis of adverse reactions associated with combination therapy, “a” denotes the number of occurrences of the target adverse reaction for the drug when used in combination, “b” represents the number of occurrences of other adverse reactions for the same drug in combination therapy, “c” indicates the number of occurrences of the target adverse reaction for the drug when used alone, and “d” signifies the number of occurrences of other adverse reactions for the drug when used as a monotherapy. This stratification allows for a more nuanced understanding of the safety profile of the drug in different therapeutic contexts. Subsequently, subgroup analyses were carried out stratified by gender and age. Signal detection was still using the ROR methods and the Chi-square test or Fisher’s exact test was used to assess differences between groups. When the difference between groups was significant and the ROR was greater than 1, it suggested that a particular adverse reaction was more likely to occur. Conversely, when the between-group difference was significant and the ROR was less than 1, it suggested that a particular adverse reaction was less likely to occur. 6 The above methods are all done by R language (version 4.2.1). The reporting of this study conforms to the STROBE statement. 7
Results
Signal detection and descriptive characteristics
In this study, multiple common NSAIDs were individually selected and screened in the database, ultimately resulting in the identification of 8 NSAIDs with reported small bowel bleeding AEs in the FAERS database and 10 related NSAIDs in JADER (Figure 1).
We gathered baseline information on NSAIDs associated with small bowel bleeding AEs. In the FAERS database, 49.4% (n = 155) were male, 42.4% (n = 133) were female, 58.3% (n = 183) were elderly (⩾65 years), and 29.3% (n = 92) were under 65 years. In terms of reported years, the majority of reports, 42% (n = 132), were concentrated in the period 2014–2018. Among the outcomes of small bowel bleeding AEs, 11.8% (n = 37) resulted in death, 11.1% (n = 35) were life-threatening, 55.7% (n = 175) required hospitalization, 0.3% (n = 1) was disability, and 20.8% (n = 65) were classified as other (Table 1). Among the cases in JADER, 59.9% (n = 206) were male, 39.5% (n = 136) were female, 64% (n = 220) were elderly (⩾65 years), and 34% (n = 117) were under 65 years. The period with the highest number of reports was 2014–2018, accounting for 38.1% (n = 131). Regarding outcomes associated with small bowel bleeding AEs, 1.7% (n = 6) resulted in death, 1.5% (n = 5) showed no recovery, 33.7% (n = 113) experienced mild recovery, and 45.1% (n = 155) had a full recovery (Table 1).
Characteristics of reports with positive signal NSAID-related small bowel bleeding adverse events.

FAERS, Food and Drug Administration’s Adverse Event Reporting System; JADER, Japan Pharmaceuticals and Medical Devices Agency’s Adverse Drug Event Reporting; NSAID, nonsteroidal anti-inflammatory drug.
We then conducted a signal detection of the aforementioned drugs and further screened for NSAIDs with a positive signal for small bowel bleeding. In the FAERS database, aspirin has the highest number of cases and signal value (n = 175, ROR: 38.3, 95%CI: 32.54–44.44), followed by naproxen, ibuprofen, diclofenac, meloxicam, celecoxib, and indomethacin. In JADER, aspirin also has the highest number of cases and signal value (n = 210, ROR: 26.45, 95%CI: 22.65–30.88), followed by loxoprofen, diclofenac, celecoxib, lornoxicam, meloxicam, and flurbiprofen (Figure 2).

Signaling of NSAIDs-associated small intestinal bleeding adverse reactions in FAERS and JADER.
FAERS, Food and Drug Administration’s Adverse Event Reporting System; JADER, Japan Pharmaceuticals and Medical Devices Agency’s Adverse Drug Event Reporting; NSAID, nonsteroidal anti-inflammatory drug.
Combination therapy analysis
In the FAERS database, there were no reported small intestinal bleeding events in the groups where rebamipide was combined with aspirin (0/583) and diclofenac (0/1194). Similarly, no small bowel bleeding events were reported in the group where probiotics were combined with aspirin (0/277), diclofenac (0/269), ibuprofen (0/285), and naproxen (0/175). In addition, no significant group differences were found when comparing PPIs combined with aspirin (p = 0.264), diclofenac (p = 0.677), ibuprofen (p = 0.068), or naproxen (p = 0.430) to these NSAIDs alone.
In the JADER database, combinations of PPIs with aspirin, loxoprofen, and celecoxib showed non-significant between-group differences compared to NSAIDs alone (p = 0.062, 0.156, 0.112 respectively), while diclofenac shows risk signal (ROR = 3.16, p = 0.005). Significant between-group difference and decreasing risks were observed in the combination group of rebamipide with aspirin (ROR = 0.28, p = 0.018) and loxoprofen (ROR = 0.27, p = 0.007) compared with the NSAIDs-alone group. However, no significant intergroup difference was found between diclofenac-rebamipide combination therapy and diclofenac alone (p = 0.777), while no small bowel hemorrhagic events were reported in the celecoxib-rebamipide combination group. No small bowel bleeding events were reported in the groups where probiotics were combined with diclofenac and celecoxib (0/85 and 0/181, respectively). When compared to these NSAIDs alone, the probiotic-combined groups of both aspirin and loxoprofen demonstrated significant between-group differences (p = 0.045 and 0.044, respectively) along with relative decreases in small bowel hemorrhagic risks (aspirin ROR = 0.17, loxoprofen ROR = 0.17) (Table 2). The occurrence of small bowel bleeding events for all NSAIDs and related combination therapies are been summarized in Table 3.
Small bowel bleeding adverse events occur in different combinations with NSAIDs in the FAERS and JADER databases.
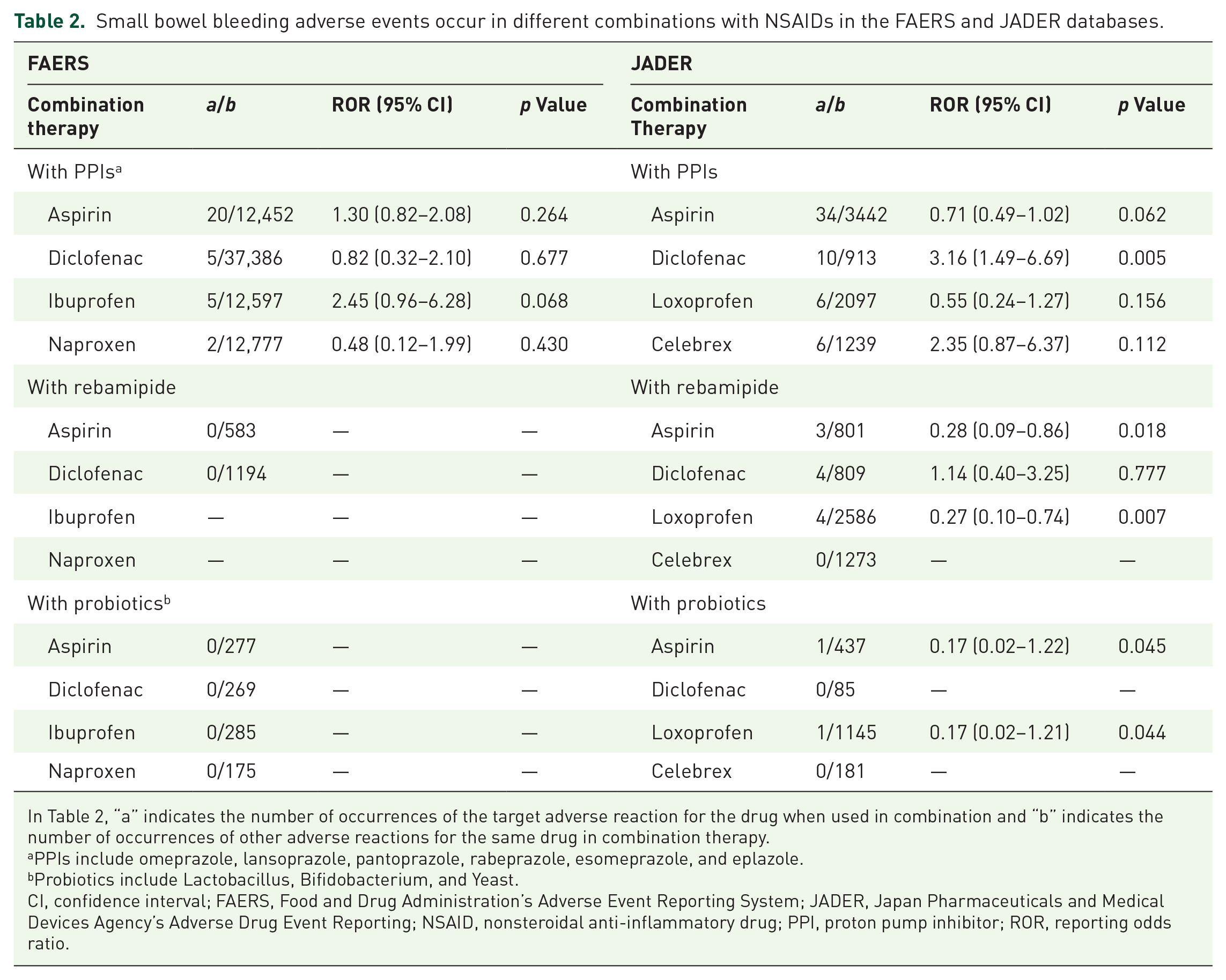
In Table 2, “a” indicates the number of occurrences of the target adverse reaction for the drug when used in combination and “b” indicates the number of occurrences of other adverse reactions for the same drug in combination therapy.
PPIs include omeprazole, lansoprazole, pantoprazole, rabeprazole, esomeprazole, and eplazole.
Probiotics include Lactobacillus, Bifidobacterium, and Yeast.
CI, confidence interval; FAERS, Food and Drug Administration’s Adverse Event Reporting System; JADER, Japan Pharmaceuticals and Medical Devices Agency’s Adverse Drug Event Reporting; NSAID, nonsteroidal anti-inflammatory drug; PPI, proton pump inhibitor; ROR, reporting odds ratio.
Occurrence of adverse reaction events for all NSAIDs and related combination therapies.

In the NSAIDs group, “a” denotes target adverse events of the target drug, “b” denotes other adverse events of the target drug, “c” denotes target adverse events of other drugs, and “d” denotes target adverse events of other drugs. In the combination therapy group, “a” indicates the number of occurrences of the target adverse reaction for the drug when used in combination, “b” indicates the number of occurrences of other adverse reactions for the same drug in combination therapy, “c” indicates the number of occurrences of the target adverse reaction for the drug when used alone, and “d” indicates the number of occurrences of other adverse reactions for the drug when used as a monotherapy.
FAERS, Food and Drug Administration’s Adverse Event Reporting System; JADER, Japan Pharmaceuticals and Medical Devices Agency’s Adverse Drug Event Reporting; NSAID, nonsteroidal anti-inflammatory drug; PPI, proton pump inhibitor.
In this study, we summarized the adverse reactions of NSAIDs and, after excluding irrelevant or duplicate reports, presented the top three reported adverse reactions for each NSAID. The results revealed that aspirin exhibited significant bleeding-related adverse reactions, particularly gastrointestinal bleeding, in both databases. For other NSAIDs, the FAERS database predominantly reported common drug adverse reactions, such as nausea and vomiting, while the JADER database showed a notable number of reports related to NSAID allergies and acute kidney injury. Notably, celecoxib demonstrated a significant association with cardiovascular and cerebrovascular AEs in the FAERS database, whereas the top three adverse reaction reports in the JADER database were all drug-related cutaneous adverse reactions (Supplemental Table 1).
Subgroup analysis
In the FAERS database, aspirin exhibited significant risk signals and differences between age (ROR = 1.87, p < 0.001) and sex (ROR = 1.68, p = 0.001) subgroups. On the other hand, ibuprofen, celecoxib, and meloxicam did not show significant differences in the between-group analyses in age and sex subgroups. Diclofenac and naproxen displayed significant between-group differences and risk signals in the age subgroup (ROR = 2.45, p = 0.017; ROR = 3.68, p < 0.001), while in the gender subgroup, these differences were not significant. In the JADER database, aspirin and loxoprofen showed significant differences between groups in age (p = 0.007, p < 0.001) and gender (p = 0.025, p = 0.001) subgroups, with risk signals evident in the signal calculations (aspirin age group ROR = 1.50, gender group ROR = 1.38; loxoprofen age group ROR = 5.82, gender group ROR = 2.36). By contrast, diclofenac, celecoxib, meloxicam, and clonoxicam did not show significant differences between groups in age and gender subgroups (Table 4).
Signal comparison and between-group differences in the occurrence of NSAID-associated small intestinal bleeding in FAERS and JADER.
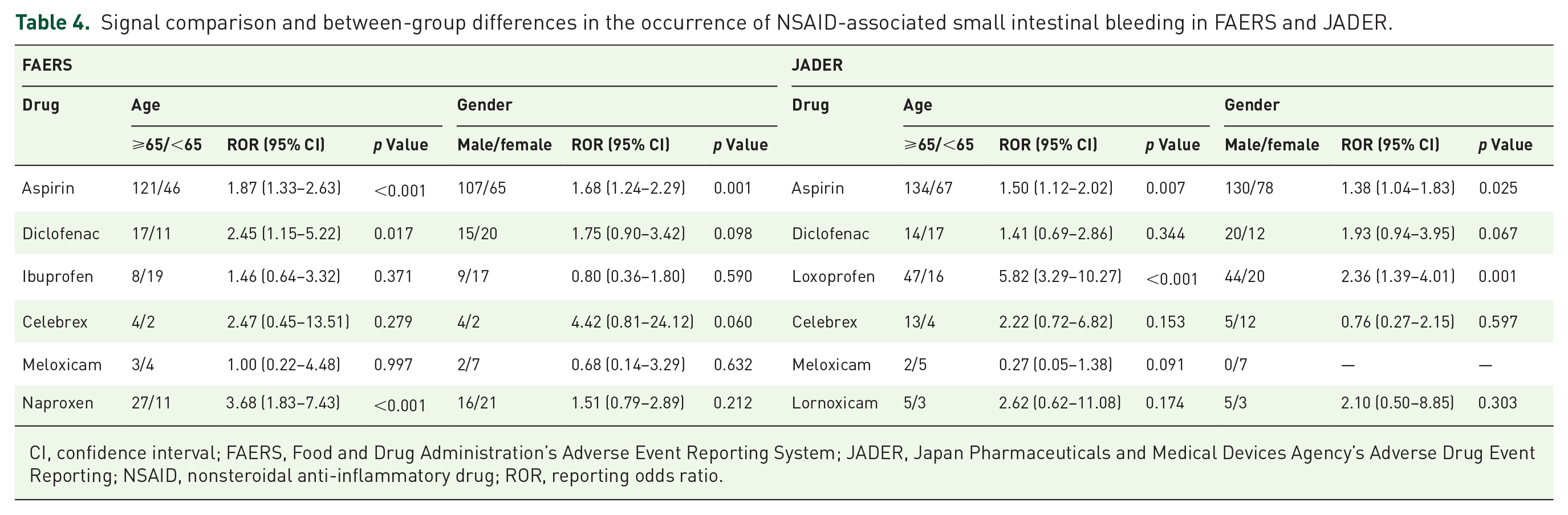
CI, confidence interval; FAERS, Food and Drug Administration’s Adverse Event Reporting System; JADER, Japan Pharmaceuticals and Medical Devices Agency’s Adverse Drug Event Reporting; NSAID, nonsteroidal anti-inflammatory drug; ROR, reporting odds ratio.
Discussion
NSAIDs-associated small bowel bleeding
Aspirin, as a non-selective inhibitor of COX, exerts a stronger inhibitory effect on COX-1 compared to other NSAIDs. This leads to the suppression of thromboxane A2 (TXA2), an antiplatelet aggregation and thrombosis agent, and an increase in prostaglandin E2 (PGE2) production, which reduces its gastroprotective effects. In addition, aspirin uniquely and irreversibly acetylates COX-1, further inhibiting platelet function and increasing the risk of small intestinal bleeding.8,9
Patients on long-term LDA therapy typically have cardiovascular and cerebrovascular-related primary diseases and may be prescribed additional antiplatelet and anticoagulant medications to manage these conditions. This further amplifies the risk of small intestinal bleeding, as demonstrated in a study on percutaneous coronary intervention (PCI) by He et al. 10 In a multicenter randomized clinical trial assessing the risk of gastrointestinal injury during post-procedural antiplatelet therapy for PCI, it was observed that the combined use of aspirin and clopidogrel increased the risk of small intestinal bleeding compared to single-agent antiplatelet therapy. These findings help to elucidate why LDA-associated small bowel bleeding is more common than other NSAID-related bleeding events.
Cases of diclofenac-induced small bowel hemorrhage have been reported in both the FAERS and JADER databases. A study conducted by Maiden et al. 11 revealed that 68%–75% of volunteers experienced varying degrees of small bowel injury after 2 weeks of diclofenac extended-release tablet use. These findings align with our database analysis. However, inconsistencies persist between the two databases. For example, while we identified 41 cases of naproxen-associated small bowel bleeding in the FAERS database, only 1 case was reported in the JADER database. Studies by Blackler et al.12,13 demonstrated the deleterious effects of naproxen on the small bowel in animal models, but the lack of relevant cases in the JADER database may reflect regional variations in reporting of adverse reactions to naproxen.
Another disparity between the databases emerged in the reporting of ibuprofen-associated small bowel hemorrhage compared to loxoprofen. While the FAERS database documented more cases of ibuprofen-related bleeding, the JADER database showed higher incidents with loxoprofen. It is noteworthy that the differences in selective and inhibitory activities on COX-1 and COX-2 between ibuprofen and loxoprofen were not significant,14,15 suggesting comparable risks of small bowel injury. Combining the reported cases from both databases in our study, regional disparities in medication practices may be influencing the observed differences in adverse reactions to these drugs.
Previous studies have suggested that selective COX-2 inhibitors are generally safer for the small intestine compared to nonselective NSAIDs. 16 In a notable large-scale, double-blind, randomized clinical trial spanning 6 months, the group receiving celecoxib exhibited a lower risk of overall gastrointestinal mucosal injury when compared to the diclofenac-conjugated PPI group. 17 However, findings from two cross-sectional capsule endoscopy studies on NSAID-associated small bowel injury by Maiden et al.18,19 indicated that long-term use of both nonselective NSAIDs and selective COX-2 inhibitors had similar effects on small bowel injury.
Due to insufficient data volume of selective COX-2 inhibitor in two databases, we have not yet compared the signaling of AEs associated with nonselective COX inhibitors versus selective COX-2 inhibitors, it needs further investigation and validation through additional data collection, especially in assessing the long-term benefits and risks associated with this medication.
We conducted a preliminary analysis of the adverse reactions of NSAIDs and observed discrepancies in reporting between the two databases, which we attribute to their differing sizes. The FAERS database, being larger and encompassing data from beyond the United States, offers a more extensive dataset, potentially introducing bias when compared with the JADER database. In addition, regional medication practices may contribute to these disparities, for instance, we found scant data on loxoprofen in FAERS, in contrast to the numerous loxoprofen-related AEs, including small intestinal bleeding, recorded in JADER. Notably, there were also significant differences in AE reporting for selective COX-2 inhibitors. The FAERS database reported a substantial number of celecoxib-related cardiovascular and cerebrovascular AEs, a matter of considerable debate, particularly in populations with pre-existing cardiovascular conditions.20,21 Despite this, selective COX-2 inhibitors have demonstrated effective anti-inflammatory and analgesic properties in acute and chronic pain, as well as rheumatic diseases, with studies suggesting a relatively safe cardiovascular risk profile.22 –24 Therefore, patient-specific considerations are crucial for determining the appropriate treatment regimen.
Analysis of subgroups
Aspirin-induced small bowel bleeding was found to be significantly different between groups in both databases in age and gender subgroups. In comparative signal analyses of both databases, it was suggested that older adults and males are risk factors for aspirin-related small bowel bleeding. Another noteworthy aspect in this subgroup analysis was that the between-group differences in JADER for the age and gender subgroups of loxoprofen were equally significant, and the signal comparisons suggested that older adults (⩾65 years) and men were risk factors for loxoprofen-associated small bowel bleeding. In addition, the subgroup analysis in FAERS also suggested that older adults (⩾65 years) were also risk factors for small bowel bleeding caused by diclofenac and naproxen. A retrospective study showed that being male and aged >60 years were independent predictors of a positive diagnosis of occult gastrointestinal bleeding by capsule endoscopy (CE), 25 which is consistent with our findings. This therefore suggests that we need to pay more attention to early prevention and intervention in the elderly male population who are long-term NSAID users, especially those who have been taking aspirin for a long time.
Effect of NSAIDs in combination with other drugs on small bowel bleeding
NSAIDs with PPIs
In patients undergoing long-term NSAID therapy, clinicians often prescribe PPIs in combination to alleviate NSAID-induced damage to the upper gastrointestinal tract. However, a study by Wallace et al. 26 revealed that PPIs can impact intestinal flora by neutralizing gastric acid, leading to significant alterations in the ecology of the intestinal microbiota and ultimately resulting in small bowel injury, with notable changes in jejunal actinomycetes and bifidobacteria spp. Another randomized controlled trial by Washio et al. found that PPIs increased the short-term risk of small bowel injury induced by NSAIDs. 27 In addition, a retrospective cohort study by Miyake et al. highlighted that the combination of low-dose aspirin and PPIs elevated the risk of lower gastrointestinal bleeding. 28 This is also supported by some systematic reviews in recent years.29,30
Despite these findings from animal experiments and small-scale controlled trials, there remains a lack of large-scale, randomized, double-blind trials to further substantiate these conclusions. Contrary to previous studies, a large case–control study conducted by Nagata et al. 31 indicated that the use of NSAIDs alone or in combination with PPIs did not significantly impact the risk of lower gastrointestinal bleeding.
In our analysis utilizing data from the FAERS and JADER databases, comparing the risk signal of small bowel bleeding when NSAIDs were co-administered with PPIs versus NSAIDs alone, we found that in both databases, concomitant use of most NSAIDs with PPIs did not demonstrate a significant increase in small intestinal bleeding risk compared to monotherapy with these NSAIDs. Notably, diclofenac-PPI combination therapy exhibited a risk signal in the JADER database, however, this association was not statistically significant in the FAERS database. We hypothesize that this discrepancy may be attributable to differences in data volume between the two databases, potentially introducing a degree of bias. These findings derived from real-world clinical practice suggest that the concomitant use of PPIs with most non-selective COX-1 inhibitor NSAIDs is unlikely to significantly elevate the risk of small intestinal bleeding. Nonetheless, further validation through subsequent large-scale trials is imperative. Until such evidence is available, it may still be prudent to continue providing PPIs to long-term NSAID users to mitigate upper gastrointestinal tract damage when necessary.
NSAIDs with probiotics
Intestinal flora are known to play a crucial role in NSAID-associated small bowel injury. 32 In our analysis of cases involving NSAIDs in combination with probiotics from two databases, we have collected only a few reported incidents of associated small bowel bleeding. Furthermore, when comparing signals from NSAIDs combined with probiotics versus NSAIDs alone, no signals of small bowel bleeding were detected. These findings collectively suggest a potential preventive effect of probiotics on NSAID-associated small bowel bleeding. However, it should be emphasized that all observed p-values demonstrated borderline statistical significance, necessitating further validation through additional studies with larger sample sizes to substantiate these findings.
Several studies have demonstrated the beneficial effects of specific probiotics, such as Lactobacillus casei and Lactobacillus sphaericus, in promoting healing of small intestinal injury in chronic low-dose aspirin users.33,34 In addition, research by Watanabe et al. indicated that NSAIDs trigger an inflammatory response and small intestine damage through activation of the LPS/TLR4 signaling pathway, while L. casei strain Shirota (LcS) inhibits this pathway, thereby exerting a preventive effect on indomethacin-induced intestinal mucosal injury. 35 Recent reviews have also discussed the negative impact of gut flora disruption on NSAIDs-induced small bowel injury, including some other important factors such as gender, age, and use of PPIs,36 –38 which were also examined in the current study. However, the intricate mechanisms of intestinal flora in the context of NSAID-associated small intestinal injury and repair remain incompletely defined, representing a significant area for further exploration.
Based on our study, we propose that probiotics may have a preventive role in NSAID-associated small bowel injury, and given their generally safe and well-tolerated nature, we recommend the combined use of probiotics for preventing small bowel injury in individuals on long-term NSAID therapy. However, we emphasize the necessity of large-scale trials to further evaluate and validate this proposition.
NSAIDs with rebamipide
Rebamipide is known for its gastroprotective properties, achieved through mechanisms such as the induction of COX-2, inhibition of inflammatory cytokine expression,39,40 and modulation of the intestinal microbiota. 41 Notably, two randomized, double-blind, placebo-controlled studies have provided envidence of its preventive effects on small bowel injury induced by aspirin and diclofenac, respectively.42, 43
Our analysis did not yield valid small bowel bleeding cases of NSAIDs combined with rebamipide in the FAERS database. However, in the JADER database, we found decreasing risks of small bowel bleeding when aspirin and loxoprofen were combined with rebamipide, compared with these NSAIDs alone. These findings suggest that rebamipide may indeed exert a preventive effect on NSAIDs-associated small bowel bleeding.
Our database analysis revealed that rebamipide may exhibited a preventive effect on NSAIDs-associated small intestinal bleeding, which corroborates the findings of the aforementioned randomized controlled studies. Given the protective effect on the upper gastrointestinal tract, we believe that may be valuable in alleviating the effects of long-term NSAID use, particularly for individuals with additional risk factors for gastrointestinal injury.
Limitations
This study has its limitations. First, the database is inherently subject to underreporting, incomplete reporting, selective reporting and non-uniform drug naming conventions, leading to the absence of some clinical information, 44 such as underlying diseases, comorbidities, and concomitant medication use, 45 which can introduce certain biases. Second, disproportionality analysis is limited to assessing signal strength and establishing statistical associations, and it cannot quantify risk or determine causality. 46 Third, small intestinal bleeding is a relatively rare adverse reaction, and the current database still lacks sufficient data on the impact of other types of drugs on this adverse reaction, which awaits further updates to enrich the research content. While this study represents an exploratory analysis utilizing real-world clinical practice data to provide reference value, we underscore the critical need for future rigorously designed research employing a unified pharmacovigilance framework to address these persistent methodological limitations.
Conclusion
In summary, a multimodal approach incorporating probiotics, rebamipide, and PPIs should be considered to manage the risk of small bowel bleeding in patients requiring long-term NSAID therapy, especially in older men. Continuous monitoring and further validation through large-scale studies are warranted to optimize the prevention and management of this adverse effect.
Supplemental Material
sj-docx-1-taw-10.1177_20420986251318848 – Supplemental material for Pharmacovigilance analysis of small bowel bleeding associated with NSAIDs
Supplemental material, sj-docx-1-taw-10.1177_20420986251318848 for Pharmacovigilance analysis of small bowel bleeding associated with NSAIDs by Ying-Han Deng, Meiting Jiang, Yun Chen and Hong-Bin Chen in Therapeutic Advances in Drug Safety
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

