Abstract
Background:
Given the growing problem of antibiotic resistance, it is crucial to improve Helicobacter pylori (H. pylori) treatment interventions or provide adjunctive therapy. The objective of this meta-analysis was to evaluate whether Lactobacillus reuteri (L. reuteri) could improve H. pylori eradication rate, reduce the incidence of adverse events (AEs), and alleviate gastrointestinal symptoms.
Design:
A meta-analysis of randomized controlled trials (RCTs) comparing L. reuteri supplementation therapy with placebo was conducted.
Sources and methods:
We retrieved relevant studies from PubMed, Embase, and the Cochrane Library. The primary outcome was H. pylori eradication rate, and the scores on the Gastrointestinal Symptom Rating Scale and AEs were secondary outcomes.
Results:
Eight RCTs including 1087 patients were included in this analysis. The L. reuteri supplementation group showed significantly higher H. pylori eradication rates in both intention-to-treat (ITT) and per-protocol (PP) analysis [ITT: 80.0% versus 72.6%; p = 0.005, relative risk (RR): 1.10; 95% confidence interval (CI): 1.03–1.17; number needed to treat (NNT) = 14; PP: 81.8% versus 75.0%; p = 0.006, RR: 1.09; 95% CI: 1.03–1.16; NNT = 15]. Patients treated with L. reuteri showed greater improvements in gastrointestinal symptoms (pooled mean difference: −2.43, 95% CI: −4.56 to −0.29, p = 0.03). The incidence of AEs was significantly reduced in the L. reuteri supplementation group based on ITT and PP analysis (ITT: p < 0.00001, RR: 0.72, 95% CI: 0.67–0.78; PP: p < 0.00001, RR: 0.70, 95% CI: 0.65–0.77).
Conclusion:
The present meta-analysis demonstrated that supplementation with L. reuteri was beneficial for improving the eradication rate of H. pylori, reducing the overall incidence of side effects, and relieving gastrointestinal symptoms in patients during treatment. The findings provide new insights into clinical decision-making.
Trial registration (PROSPERO):
CRD42023424052.
Plain language summary
Given the growing problem of antibiotic resistance, it is crucial to improve Helicobacter pylori (H. pylori) treatment interventions or provide adjunctive therapy. Eight randomized controlled trials (RCTs) including 1087 patients were included in this analysis. The present meta-analysis demonstrated that supplementing with L. reuteri tends to increase the eradication rate of H. pylori, reduce the overall incidence of antibiotic-related side effects, and alleviate gastrointestinal symptoms in patients during treatment, providing new insights for clinical decision-making.
Background
Helicobacter pylori (H. pylori), as the main pathogenic bacteria of chronic gastritis, peptic ulcer, mucosa-associated lymphoid tissue (MALT) lymphoma, and gastric adenocarcinoma, has attracted significant attention worldwide since its discovery. 1 The treatment of H. pylori is challenged by the increasing prevalence of antibiotic resistance, and the impact of antibiotic therapy on the gut microbiome is an important consideration that must not be overlooked.2,3 Therefore, it is crucial to improve therapeutic interventions or add adjuvant treatments.
Probiotics are non-pathogenic living bacteria that confer health benefits to the host, including anti-oxidative and anti-inflammatory effects that may help prevent intestinal infections, cancer, and cardiovascular disease.4,5 Numerous studies have demonstrated that certain Lactobacillus strains, such as Lactobacillus GG, Lactobacillus acidophilus, and Lactobacillus reuteri (L. reuteri), possess anti-H. pylori properties. Several clinical trials have integrated specific probiotics into conventional regimens to reduce adverse effects (AEs), improve drug compliance, and increase eradication rates.6–9 According to a meta-analysis by Yu et al., 10 supplementing Lactobacillus during the treatment of H. pylori infection can effectively improve the eradication rate and reduce the incidence of therapy-related taste disturbance. Another study compared the effectiveness of various probiotics in H. pylori eradication therapy, suggesting that Lactobacillus and multiple strains are favorable choices among probiotics. Additionally, subjects in China exhibited higher eradication rate than those in other countries in this study. 11 It is worth mentioning that L. reuteri has demonstrated promising results in eradicating H. pylori by producing powerful antimicrobial compounds that inhibit the growth of H. pylori and strengthen the mucosal barrier against infection by increasing mucin secretion.12,13
Despite the promising results observed in clinical trials, there is currently no clear consensus on when to administer probiotics as an adjuvant therapy for H. pylori eradication, and the appropriate dosage and duration of treatment continue to be debated. To evaluate the potential benefit of L. reuteri supplementation in combination with standard therapy, this meta-analysis was conducted to determine whether it could significantly improve H. pylori eradication rate, reduce the incidence of AEs, and alleviate gastrointestinal symptoms.
Materials and methods
This meta-analysis was registered in PROSPERO (registration no.: CRD42023424052) and conducted following the Preferred Reporting Items for Systematic Review and Meta-analyses guidelines. 14
Search strategy
To identify relevant literature, we systematically searched the following databases: (1) PubMed, (2) The Cochrane Library, and (3) Embase. The cut-off date for inclusion in our study was 31 December 2023, and only English articles were included across all databases within our scope. Potentially relevant randomized controlled trials (RCTs) were retrieved using the following MeSH terms: ‘Helicobacter pylori or H. pylori’, ‘probiotic or probiotics’, and ‘Lactobacillus reuteri or L. reuteri’. Furthermore, references from the retrieved articles as well as relevant meta-analyses or reviews were manually screened and incorporated into the eligible literature. The search strategies for each database were presented in Supplemental Table S1.
Selection criteria
All studies included in this meta-analysis were based on the following PICOS principles: (1) P (population): H. pylori-infected adult patients; (2) I and C (intervention and comparison): articles evaluating the efficacy and safety of L. reuteri as an adjuvant therapy for H. pylori eradication compared with placebo; (3) O (outcomes): endpoints included the eradication rate, AEs, and symptom scores before and after treatment; and (4) S (study): RCTs published in English.
The exclusion criteria were as follows: basic studies, conference abstracts, reviews, meta-analyses, case reports, summary-only articles, and literature with insufficient data.
Data extraction
Two investigators independently extracted data from the included studies, and any discrepancies were resolved through discussion until a reasonable solution was reached. Otherwise, the decision was made by a third reviewer. The detailed data were as follows: first author, year of publication, country, numbers of enrolled patients, baseline characteristics (age and previous experience in H. pylori eradication), eradication regimen, types and dosage of probiotics, follow-up time, eradication rates (intention-to-treat analysis, ITT; per-protocol analysis, PP), scores of Gastrointestinal Symptom Rating Scale (GSRS), and AEs.
Quality assessment
The Cochrane Risk of Bias Assessment Tool was used to assess the quality of all eligible studies, which included the following main evaluation indicators: random sequence generation, allocation concealment, blinding of patients and personnel, incomplete outcome data, selective reporting, and other bias. 15 Two researchers independently evaluated the quality of each study and reached a consensus. Studies with a score of 3 or higher were considered to be of high quality.
Statistical analysis
This meta-analysis was performed using the following data management software: Review Manager (version 5.3; Copenhagen: The Nordic Cochrane Center, The Cochrane Collaboration, 2014) and STATA (version 14.0; Stata Corp, College Station, Texas, USA). The dichotomous outcomes were expressed as relative risk (RR) with corresponding 95% confidence intervals (CIs). The effect size of the continuous variable was expressed as the mean difference (MD) and 95% CIs. When the p-value was less than 0.05, it was considered as statistically significant. We calculated the number needed to treat (NNT), using the formula NNT = 1/(assumed control risk (ACR) × (RR − 1)). Summary estimates of the eradication rates and AEs were performed on the ITT and PP principles. Subgroup analysis of H. pylori eradication rate was performed to determine whether the results were stable according to nations, eradication regimens, treatment lines, and the timing and duration of L. reuteri supplementation. In addition, heterogeneity in our study was assessed by the I2 statistic and χ2 test with the random-effects or fixed models. Significant heterogeneity was indicated when p < 0.10 or I2 > 50%, then we used a random-effects model. Otherwise, the fixed-effects model was used. We also explored the potential publication bias through the funnel plot and Egger’ s linear regression test. 16
Results
Search results and quality assessment
Through a preliminary literature search, we identified 1826 potentially eligible studies. However, most studies were excluded because they did not meet the inclusion criteria regarding the intervention. Ultimately, only 8 RCTs, consisting of 1087 patients (L. reuteri group: 544 patients; placebo group: 543 patients), were included in this analysis6,17–23 (Figure 1). One of the included articles had a mean age of over 18 years and was also deemed eligible for analysis. 6 Additionally, one of the studies included two comparisons that met our inclusion criteria (7-day and 14-day eradication rates, respectively), and were included in our analysis as independent studies (Poonyam a and Poonyam b). 17 Table 1 summarized the baseline characteristics and main evaluation indicators. Furthermore, all the studies scored 3–5 points on the quality assessment scale, indicating high quality (Supplemental Table S2, Supplemental Figure S1).

Flow chart of studies included in the meta-analysis.
Characteristics of the studies included in this meta-analysis.
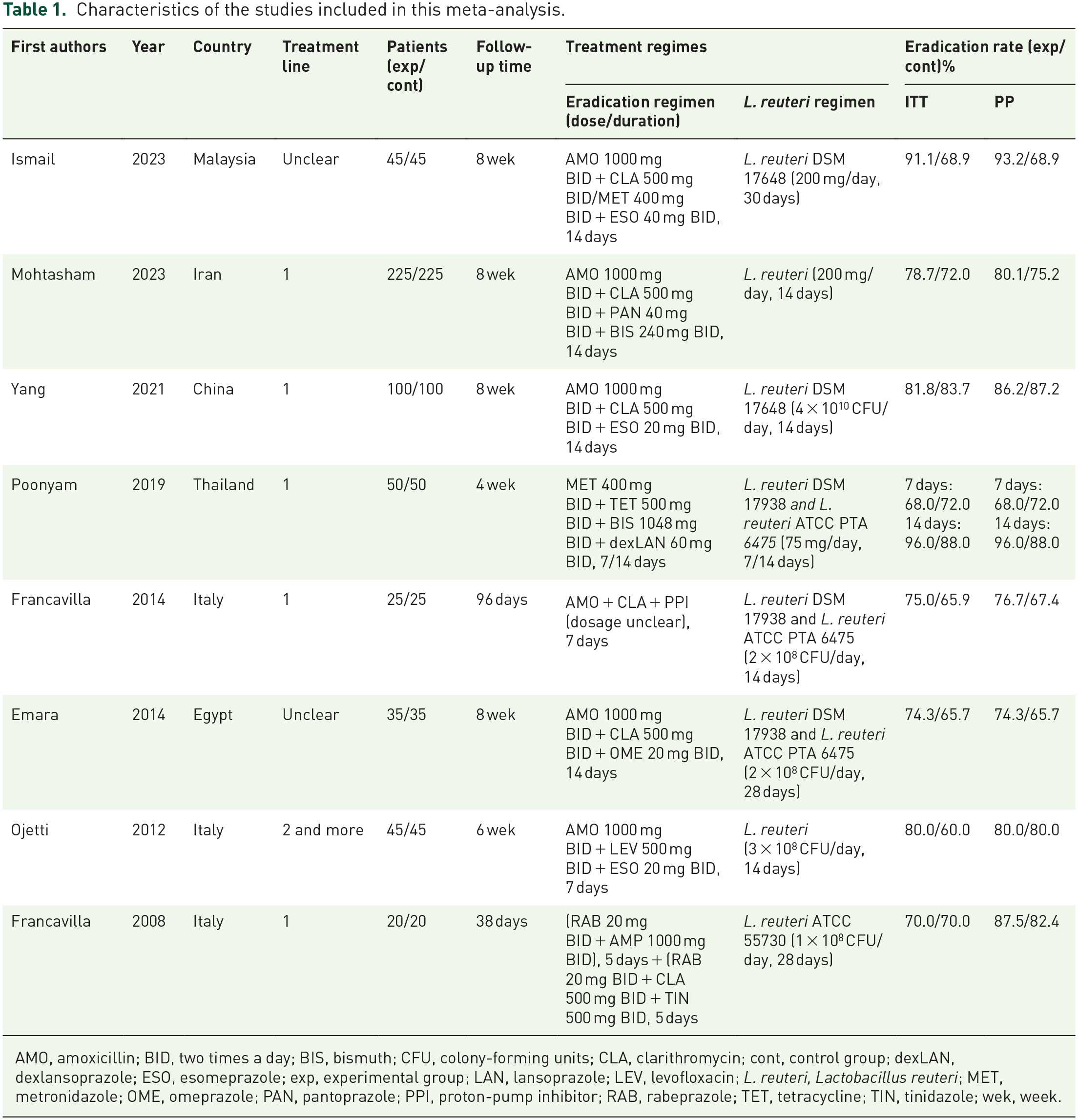
AMO, amoxicillin; BID, two times a day; BIS, bismuth; CFU, colony-forming units; CLA, clarithromycin; cont, control group; dexLAN, dexlansoprazole; ESO, esomeprazole; exp, experimental group; LAN, lansoprazole; LEV, levofloxacin; L. reuteri, Lactobacillus reuteri; MET, metronidazole; OME, omeprazole; PAN, pantoprazole; PPI, proton-pump inhibitor; RAB, rabeprazole; TET, tetracycline; TIN, tinidazole; wek, week.
Eradication rates
Data on the eradication rates were available from all included studies. No significant heterogeneity was observed between them (ITT: p = 0.36, I2 = 10%; PP: p = 0.27, I2 = 19%). Compared with the placebo group, a significantly increased eradication rate was observed in the L. reuteri supplementation group with ITT analysis [p = 0.005, RR: 1.10; 95% CI: 1.03–1.17, Figure 2(a)] and an NNT of 14. In PP analysis, compared with the control group, the L. reuteri supplementation group also had better eradication rates, with the eradication rates of 75.0% and 81.8%, respectively [p = 0.06, RR: 1.09; 95% CI: 1.03–1.16, NNT = 15, Figure 2(b)].
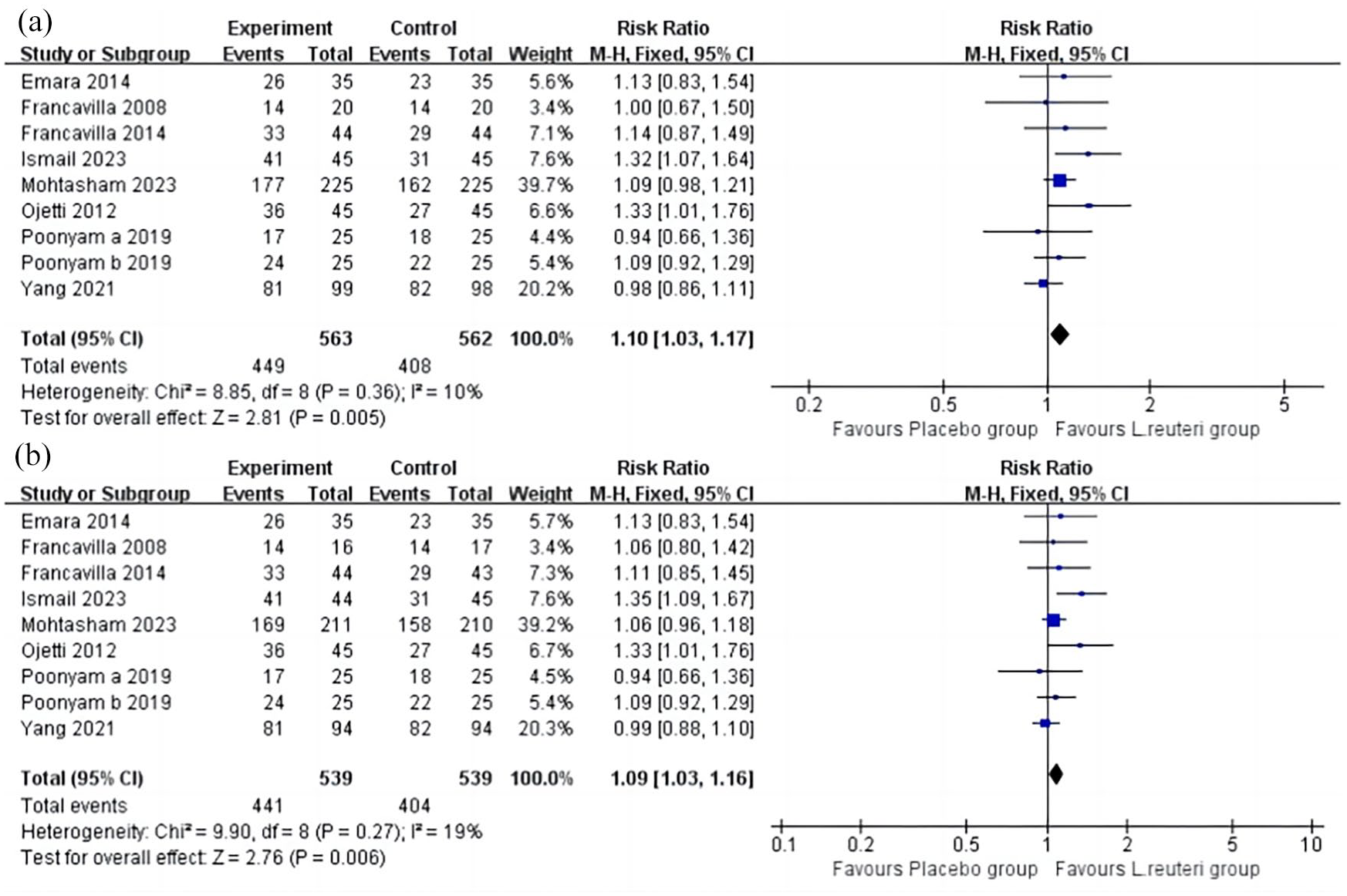
Forest plots of the pooled H. pylori eradication rates with ITT (a) and PP (b) analysis for the comparison of L. reuteri supplementation group versus placebo group.
Advert effects
There were only two studies comparing the overall AEs.6,19 The incidence of total AEs in the placebo group was significantly higher than that in the L. reuteri supplementation group, and the difference was statistically significant by both ITT and PP analyses [ITT: p < 0.00001, RR: 0.72; 95% CI: 0.66–0.78, Figure 3(a); PP: p < 0.00001, RR: 0.70; 95% CI: 0.64–0.77, Figure 3(b)]. Furthermore, we performed a subgroup analysis of the top 10 AEs (nausea/vomiting, loss of appetite, taste disturbance, abdominal pain, heartburn, flatulence, diarrhea, headache, dizziness, cramp). Compared with the placebo group, the L. reuteri supplementation group showed superiority in reducing AEs in all outcomes except for dizziness (Table 2).

Forest plots of the total adverse events with ITT (a) and PP (b) analysis for the comparison of L. reuteri supplementation group versus placebo group.
Comparison of top 10 adverse effects between the L. reuteri group and the placebo group.

CI, confidence interval; L. reuteri, Lactobacillus reuteri; RR, relative risk.
Symptom assessment
Gastrointestinal symptoms before and after treatment were assessed and scored according to the 15-item GSRS in 5 studies.18–20,22,23 Results were expressed as mean ± standard deviation (SD) in four studies18,20,22,23 and as median ± SD in one study. 19 Therefore, we performed a meta-analysis of the score changes from baseline in the first four studies. By comparison, patients treated with L. reuteri showed greater improvements in gastrointestinal symptoms (pooled MD: −2.43, 95% CI: −4.56 to −0.29, p = 0.03, Figure 4).

Forest plots of the score changes from the baseline of Gastrointestinal Symptom Rating Scale with or without L. reuteri supplementation.
Subgroup analysis
To validate the consistency of the results, we conducted subgroup analyses on the eradication rates of H. pylori based on several categories of variables in our meta-analysis, including nations, eradication regimens, treatment lines, and the timing and duration of L. reuteri supplementation. As shown in Table 3, we performed subgroup analysis by nation and found that in the PP analysis, the L. reuteri supplementation group had higher eradication rates in both Europe and Asia. In the ITT analysis, there was no statistical significance in Europe. We observed that the eradication rate of H. pylori in the L. reuteri supplementation group was significantly higher than that in the placebo group when the supplementation was extended to a period after conventional eradication therapy. Both ITT and PP analyses displayed statistically significant differences (ITT: p = 0.03, RR: 1.20; 95% CI: 1.02–1.42; PP: p = 0.04, RR: 1.19; 95% CI: 1.01–1.41; Table 3). When patients received L. reuteri as a pretreatment or in combination with standard treatment, there was no significant difference in the eradication rates between the two groups (Table 3). Additionally, we found that the treatment duration of L. reuteri for 4 weeks or longer showed a significant advantage in improving the eradication rate compared with the placebo group (ITT: p = 0.02, RR: 1.18; 95% CI: 1.02–1.35; PP: p = 0.01, RR: 1.19; 95% CI: 1.04–1.36). This superiority was also significant in first-line treatment (ITT: p = 0.03, RR: 1.08; 95% CI: 1.01–1.16; PP: p = 0.04, RR: 1.07; 95% CI: 1.00–1.14). Although some subgroup analysis results indicated a favorable trend in the L. reuteri supplementation group, there was no statistical difference due to the limited number of articles included.
Subgroup analysis for the eradication rate of H. pylori.

BQT, bismuth quadruple therapy; CI, confidence interval; ITT, intention-to-treat; L. reuteri, Lactobacillus reuteri; –, not available; PP, per-protocol; RR, relative risk.
Sensitivity analysis
By conducting a one-study-removed sensitivity analysis, we found that none of the studies had a substantial impact on the pooled risk of H. pylori eradication rates, indicating the reliability of our results (Supplemental Figure S2).
Publication bias
The funnel plot obtained by ITT and PP analyses of the eradication rates showed a roughly symmetrical distribution (Supplemental Figure S3). We found no significant publication bias in the pooled eradication rates as determined by Egger’s test. However, it should be noted that the number of studies included in the analysis was relatively small (Supplemental Figure S4).
Discussion
The management of H. pylori infection is becoming increasingly critical, particularly in countries with high antibiotic resistance. Despite the availability of different treatment approaches, concerns over treatment failure and side effects continue to grow in routine clinical practice. 2 Many researchers have explored whether probiotic supplementation can benefit H. pylori treatment, but the results are contradictory. Several studies showed that probiotics helped improve the eradication rate of H. pylori, but this benefit was only applicable to specific strains, including different strains of Lactobacillus spp., Bifidobacteria spp., and Saccharomyces boulardii. 3 In the absence of sufficient evidence, recent guidelines were conservative on the recommendation of probiotics in H. pylori eradication therapy and only suggested that supplementation with probiotics may reduce the incidence of side effects associated with eradication therapy.3,24 In our meta-analysis, we included eight RCTs comparing supplementation with L. reuteri versus placebo in the treatment with H. pylori and observed positive findings across several outcome measures, including an increased eradication rate of H. pylori, a reduced overall incidence of antibiotic-related side effects, and alleviation of gastrointestinal symptoms in patients during treatment. Notably, the probiotic group exhibited a significantly higher eradication rate than the placebo group, with a 7.4% improvement in the ITT analysis (80.0% versus 72.6%, p = 0.005) and a 6.8% improvement in the PP analysis (81.8% versus 75.0%; p = 0.006).
Probiotics may exert direct inhibition on H. pylori in a variety of ways. Primarily, the synthesis of antimicrobial peptides or organic acids may be one of the mechanisms through which specific probiotics, such as Lactobacillus and Bifidobacterial, combat H. pylori infections.25,26 Second, probiotics such as Lactobacillus spp. can exert anti-inflammatory effects by activating suppressor of cytokine signaling expression in patients with H. pylori infection. 27 Furthermore, some studies have demonstrated that L. reuteri can significantly reduce the bacterial load, 28 inhibit the colonization of H. pylori in the gastric mucosa, and eliminate the bacteria without the need for antibiotics, thus minimizing the damage to the human body. 29
A number of meta-analyses of RCTs have assessed the efficacy and side effects of probiotics in H. pylori eradication therapy and have shown that certain probiotics (certain strains of Lactobacillus, Bifidobacterium, and S. boulardii) can be effective in increasing the eradication rate and mitigating AEs associated with H. pylori eradication therapy.30–33 However, pooling data from studies of different probiotic strains may lead to bias. In the present study, we focused on the effects of L. reuteri on H. pylori therapy; however, there is a lack of studies that directly compare the efficacy of different probiotic strains in the adjuvant treatment of H. pylori. Therefore, more studies are needed to explore the impact of different probiotic strains on H. pylori eradication to provide new insights into clinical treatment.
Some previously published studies on the efficacy of probiotics for treating H. pylori have not yielded conclusions consistent with ours. For example, in the retrospective study of Zagari et al., 34 there was no difference in eradication rates in patients with and without probiotic supplementation. However, although various probiotics were used and evaluated in the study, only three patients used L. reuteri as the probiotic supplementation; the cure rate achieved with the bismuth quadruple therapy regimen was so high that any potential benefits of probiotic supplementation were not evident. In addition, the convenience of the three-in-one pill increased patient compliance, thus reducing the likelihood that probiotics enhanced eradication rate by improving patient adherence. In contrast, probiotics demonstrate a clear advantage in triple therapy with lower H. pylori eradication rate. 10
Our subgroup analysis based on different regimens found that L. reuteri supplementation had no statistically significant effect on the eradication rates in triple, quadruple, or sequential therapy, and high eradication rates (>90%) were not achieved in any of these regimens. However, the results of our subgroup analysis supported that supplementation with L. reuteri in first-line treatment regimen improved H. pylori eradication rates by 5.8% (80.1% versus 74.3%, p = 0.03) in ITT analysis and 5.5% (82.6% versus 77.1%, p = 0.04) in PP analysis. We also examined the effect of the timing and duration of L. reuteri supplementation on the eradication rates. In our meta-analysis, we found that treatment with L. reuteri for 4 weeks or longer had a significant advantage compared to the placebo group. Therefore, we supposed that appropriately extending the probiotic treatment course may be beneficial for improving the eradication rate. When considering the timing of L. reuteri supplementation, it would be advantageous to use it with an eradication regimen and to continue its use for a period of time after the withdrawal of treatment. These conclusions were consistent with some published studies,10,35 which reported higher eradication rates with Lactobacillus-supplemented triple therapy and indicated that a longer duration of Lactobacillus supplementation may improve the eradication efficacy. Furthermore, variations in the duration and species of supplementation with Lactobacillus and other factors, as well as the large sample size and multi-center nature of these studies, may have contributed to the different results observed.
When we assessed gastrointestinal symptoms using a self-assessment questionnaire, we observed significant improvements in both the frequency and severity of symptoms in patients who received L. reuteri supplementation. Similarly, several studies have explicitly reported the effectiveness of L. reuteri in improving symptoms such as functional constipation, abdominal distension, and diarrhea.36,37 The gut microbiota is in a delicate balance and can easily be disrupted by antibiotics, leading to a series of imbalances. We also investigated whether L. reuteri can reduce the incidence of antibiotic-related side effects and found that the group supplemented with probiotics had a lower incidence of AEs. In our meta-analysis, it was found that the three most common AEs were nausea or vomiting, loss of appetite, and taste disturbance. However, due to the chronic nature of H. pylori infection and the fact that L. reuteri may not be able to colonize sustainably in the human gut after stopping supplementation, we cannot afford the possibility that these benefits are temporary. There is evidence that patient compliance is a crucial factor in H. pylori eradication, which is significantly impacted by AEs of treatment.38,39 Therefore, our conclusions tend to favor supplementation with L. reuteri to improve the eradication rate by improving patient compliance. Nevertheless, the eradication rate of H. pylori was slightly improved, with an NNT of 14 in the ITT analysis and 15 in the PP analysis. Clinical supplementation of L. reuteri as an adjuvant for H. pylori eradication still requires individualized consideration and comprehensive evaluation.
Our meta-analysis had two main advantages. First, this was the first meta-analysis to evaluate the efficacy and safety of supplementing L. reuteri with standard H. pylori eradication therapy, providing a basis for its future clinical application. Second, all studies included were high quality RCTs, ensuring the reliability and validity of the overall results. However, there were also some limitations in our review. First, the sample size of our study was limited, especially in subgroup analyses; therefore, no conclusion can be drawn on the timing of L. reuteri supplementation. Second, we used GSRS to assess patients’ symptoms, which have strong individual perceptual differences and may increase the risk of information bias. Finally, studies that were published in languages other than English or did not mention relevant keywords in the title or abstract may have been missed.
Conclusion
In summary, this meta-analysis demonstrated that supplementation with L. reuteri was beneficial for improving the eradication rate of H. pylori, reducing the overall incidence of antibiotic-related side effects, and relieving gastrointestinal symptoms in patients during treatment. The findings provide new insights into clinical decision-making. Further larger-scale trials are necessary to verify our results.
Supplemental Material
sj-docx-1-tag-10.1177_17562848241258021 – Supplemental material for Lactobacillus reuteri compared with placebo as an adjuvant in Helicobacter pylori eradication therapy: a meta-analysis of randomized controlled trials
Supplemental material, sj-docx-1-tag-10.1177_17562848241258021 for Lactobacillus reuteri compared with placebo as an adjuvant in Helicobacter pylori eradication therapy: a meta-analysis of randomized controlled trials by Meng Li, Xiaolei Wang, Xinhong Dong, Guigen Teng, Yun Dai and Weihong Wang in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
