Abstract
Background:
The association between dipeptidyl peptidase-4 inhibitors (DPP-4i) exposure and the risk of colorectal cancer (CRC) in patients with type 2 diabetes mellitus (T2DM) is unclear.
Objectives:
This meta-analysis aims to investigate the relationship between DPP-4i exposure and the incidence of CRC in patients with T2DM.
Design:
A systematic review and meta-analysis.
Methods:
A comprehensive search of electronic databases, including PubMed, Web of Science, EMBASE, and ScienceDirect, was conducted up to March 2024. The studies including randomized clinical trials (RCTs), cohort studies, and case–control studies were retrieved. The odds ratio (OR) was calculated using Stata 12.0 statistical software. The primary outcome assessed was the incidence of CRC.
Results:
This meta-analysis incorporated six retrospective cohort studies and two case–control studies. The findings indicate that the incidence of CRC in the DPP-4i exposure group was significantly higher than that in the control group (OR = 1.11, 95% CI: 1.02–1.21, p = 0.013). Subgroup analysis revealed that both male (OR = 2.07, p < 0.001) and female participants (OR = 1.49, p = 0.05) in the DPP-4i exposure group exhibited a significantly higher incidence of CRC compared to the control group. Among participants younger than 65 years, the incidence of CRC was markedly elevated in the exposure group (OR = 2.81, p < 0.001). Furthermore, when the exposure duration was less than 1 year, the CRC incidence in the exposure group surpassed that of the control group (OR = 1.24, p = 0.005). When sulfonylureas (SU) were used as control drugs, the incidence of CRC was higher in the exposure group (OR = 1.10, p = 0.017).
Conclusion:
There is a potential correlation between DPP-4i exposure and increased incidence of CRC in T2DM patients. This association appears to be influenced by gender, age, duration of exposure, and the choice of control medications. Therefore, attention should be paid to colorectal diseases when DPP-4i is employed in the clinic.
Trial registration:
The meta-analysis has been registered with the International Prospective Register of Systematic Reviews (PROSPERO). The registration number is CRD42024535292.
Plain language summary
This study investigated the relationship between dipeptidyl peptidase -4 inhibitor (DPP-4i), which is commonly used to control blood glucose in patients with type 2 diabetes, and the risk of colorectal cancer (CRC).
Our main concerned outcome is how many type 2 diabetic patients who use DPP-4i later developed colorectal cancer. The results showed that the incidence of colorectal cancer was higher in patients taking DPP-4i than in diabetic patients taking other drugs to control blood glucose.
Then, after further analysis, we find that the risk of CRC in patients taking DPP-4i is significantly increased in both men and women. Especially for patients under 65 years old, and those who have just started taking DPP-4i for less than a year, their risk of CRC is higher. In addition, when sulfonylurea drugs are compared, the incidence of CRC in patients who use DPP-4i is still high.
Therefore, patients with type 2 diabetes should pay more attention to their colon and rectum health while using DPP-4i to control blood glucose.
Introduction
According to the International Agency for Research on Cancer, colorectal cancer (CRC) has emerged as the third most common cancer globally, with nearly 1,930,000 new cases diagnosed in 2022. Significantly, CRC is the second leading cause of cancer-related deaths, with fatalities exceeding 900,000, representing a severe threat to public health. 1 Established risk factors for CRC include familial inheritance, obesity, smoking, excessive alcohol consumption, and high intake of red and processed meats. 2 Emerging evidence suggests that type 2 diabetes mellitus (T2DM) may also contribute to the risk of developing CRC.3,4 Notably, there are nearly 529 million individuals with diabetes worldwide, of whom 96.0% are diagnosed with T2DM. 5 Some research proposes that managing T2DM might mitigate the risk of developing CRC. 4
Currently, therapeutic options for T2DM encompass a range of drugs including insulin, metformin, sulfonylureas (SU), glinides, thiazolidinediones (TZDs), alpha-glucosidase inhibitors, glucagon-like peptide-1 receptor agonists (GLP-1 RAs), and sodium-glucose cotransporter 2 inhibitors (SGLT-2i). Dipeptidyl peptidase-4 inhibitors (DPP-4i) are prevalently employed as a common second-line therapy. 6 Some scholars posit that DPP-4i might reduce CRC incidence by modulating signaling pathways. For instance, in vitro studies have shown that DPP-4i can diminish the viability and migration capabilities of CRC cells. 7 However, retrospective cohort studies suggest an association between DPP-4i exposure in T2DM patients and an increased incidence of CRC. 8
A meta-analysis has reported no correlation between DPP-4i usage and CRC incidence in T2DM patients; this study, however, is limited by the short follow-up duration of included studies and a lack of detailed subgroup analysis. 9 Therefore, we suppose that there is a correlation between exposure to DPP-4i and increased incidence of CRC in T2DM patients. Our meta-analysis seeks to comprehensively review the existing literature to assess the relationship between DPP-4i exposure and CRC incidence among patients with T2DM.
Materials and methods
Research registration
In accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, this meta-analysis has been registered with the International Prospective Register of Systematic Reviews (PROSPERO). The registration number is CRD42024535292.
Retrieval strategy
The search was conducted across PubMed, Web of Science, EMBASE, and ScienceDirect databases from their inception to March 2024, targeting studies related to DPP-4i and the incidence of CRC. A comprehensive search strategy was employed, utilizing both MeSH terms and free-text terms. References of all retrieved articles were also reviewed to ensure completeness. The search terms used included: ((dipeptidyl peptidase-4 inhibitors) OR (DPP-4i) OR (sitagliptin) OR (saxagliptin) OR (vildagliptin) OR (omarigliptin) OR (trelagliptin) OR (anagliptin) OR (gemigliptin)) AND ((colorectal cancer) OR (colorectal carcinoma) OR (CRC) OR (cancer) OR (carcinoma)).
Data inclusion and exclusion criteria
Eligibility criteria were based on the PICOS framework (participants, intervention, control, outcomes, study design). Inclusion criteria comprised the following: (1) participants diagnosed with T2DM; (2) intervention with DPP-4i; (3) control group not using DPP-4i or other hypoglycemic agents as control; (4) outcome measured as the incidence of CRC; (5) study design includes randomized clinical trials (RCTs), cohort studies, or case–control studies.
Exclusion criteria included the following: (1) studies not written in English; (2) unavailable data; (3) inaccessible full-text articles; (4) absence of relevant outcomes; (5) studies combining DPP-4i with other hypoglycemic drugs; and (6) redundant studies from the same cohort, where only the most recent or comprehensive data were retained.
Data extraction and quality evaluation
Data were systematically extracted using a pre-designed table that included details such as authors, publication date, study design, participant demographics, therapeutic regimens, incidence of CRC, and follow-up duration. Two independent researchers performed the data extraction and quality evaluation.
For RCTs, the Cochrane Risk of Bias tool was employed to assess potential biases within the trials. For cohort and case–control studies, the Newcastle–Ottawa Scale (NOS) was utilized. The NOS evaluates bias risk across three domains: selection, comparability, and outcome. Studies achieving a score >6 were deemed high quality and included in the analysis.
Statistical analysis
Statistical analyses were conducted using Stata software, version 12.0. The odds ratio (OR) and 95% confidence intervals (95% CI) were calculated to assess the association between DPP-4i exposure and the incidence of CRC. Given potential heterogeneity across studies—arising from differences in participant characteristics, study types, and variations in drug exposure—a random-effects model was applied to enhance the reliability of the results.
Heterogeneity among the studies was evaluated using the chi-square test, and the extent of heterogeneity was quantified with the I2 statistic. Interpretation of I2 values was as follows: 0%–25% indicated negligible heterogeneity, 26%–50% mild heterogeneity, 51%–75% moderate heterogeneity, and over 75% substantial heterogeneity. In addition, publication bias was assessed using Begg’s test, with statistical significance set at a two-sided p-value of <0.05.
Results
Description of study retrieval
A systematic search across five electronic databases yielded 1662 records. An additional three studies were identified from other sources. After the removal of duplicates, 1149 studies remained. Upon screening titles and abstracts, 1134 studies were excluded, leaving 15 studies for full-text assessment. Of these, two studies were excluded due to unavailable data, and five were excluded for lacking relevant outcomes. Ultimately, eight studies were included in the meta-analysis (Figure 1).

Flow diagram of the selection process.
Characteristics of studies and quality evaluation
The eight studies included in the meta-analysis comprised six retrospective cohort studies and two nested case–control studies.8,10–16 The studies collectively involved more than 350,000 participants in the DPP-4i exposure group and nearly 330,000 participants in the control groups. In the control groups, three studies utilized SGLT-2i, three included TZDs, and the remaining three employed SU as control drugs.
Details of the included studies are provided in Table 1, with additional participant information available in Supplemental Table 1. The quality evaluation of the studies, assessed using the NOS, is detailed in Supplemental Table 2 (cohort studies) and Supplemental Table 3 (case-control studies). All studies met the meta-analysis quality criteria, with three cohort studies scoring nine points and the remaining three scoring eight points each. Both case–control studies scored nine points.
Characteristics of all the studies included in the meta-analysis.

DPP-4i, dipeptidyl peptidase-4 inhibitor; N/A, not applicable; SD, standard deviation; SGLT-2i, sodium-glucose cotransporter 2 inhibitor; SU, sulfonylureas; T2DM, type 2 diabetes mellitus; TZD, thiazolidinedione.
DPP-4i and the incidence of CRC
Eight studies investigated the relationship between exposure to DPP-4i and the incidence of CRC. Of that, the size of samples in the DPP-4i exposure group is more than 350,000, and the size of samples in the control group is nearly 330,000. The pooled analysis revealed that the incidence of CRC was significantly higher in participants exposed to DPP-4i compared to those in the control group (OR = 1.11, 95% CI: 1.02–1.21, p = 0.013; Figure 2).

Forest plot of the association between DPP-4i and the incidence of CRC in participants with T2DM (p = 0.013).
Analysis by tumor location divided CRC into rectal and colon cancer. Statistical results indicated no significant differences in the incidence of rectal cancer (p = 0.721; Figure 3(a)) or colon cancer (p = 0.965; Figure 3(b)) between the exposure and control groups. However, subgroup analysis by gender revealed that the incidence of CRC was significantly higher in the DPP-4i exposed group among both male participants (OR = 2.07, 95% CI: 1.54–2.78, p < 0.001; Figure 4(a)) and female participants (OR = 1.49, 95% CI: 1.00–2.22, p = 0.05; Figure 4(b)). Age-based analysis showed a higher incidence of CRC in participants under 65 years exposed to DPP-4i (OR = 2.81, 95% CI: 1.98–3.99, p < 0.001; Figure 5(a)), while no significant difference was found in participants 65 years and older (p = 0.244; Figure 5(b)). Geographic analysis did not show statistically significant differences in CRC incidence between participants from Eastern countries (p = 0.095; Figure 6(a)) and Western countries (p = 0.113; Figure 6(b)).

Forest plot of the subgroups between DPP-4i and the incidence of CRC across onset site. (a) Rectal cancer, p = 0.721. (b) Colon cancer, p = 0.965.

Forest plot of the subgroups between DPP-4i and the incidence of CRC across sex. (a) Male, p < 0.001. (b) Female, p = 0.05.

Forest plot of the subgroups between DPP-4i and the incidence of CRC across age. (a) <65 years, p < 0.001. (b) ⩾65 years, p = 0.244).

Forest plot of the subgroups between DPP-4i and the incidence of CRC across sources of participants. (a) The eastern country, p = 0.095. (b) The western country, p = 0.113.
Regarding the duration of drug exposure, the incidence of CRC was significantly higher in the DPP-4i exposure group compared to the control group for exposure periods of <1 year (OR = 1.24, 95% CI: 1.07–1.45, p = 0.005; Figure 7(a)). However, for longer exposure durations—ranging from over 1 year to 2 years and beyond—no statistically significant differences were observed (1–2 years: OR = 0.98, p = 0.854; >2 years: OR = 1.01, p = 0.924; Figure 7). In the subgroup analysis by type of control drug, a higher incidence of CRC was noted in the DPP-4i group when SU was used as the control (OR = 1.10, 95% CI: 1.02–1.19, p = 0.017; Figure 8(a)). Conversely, there were no significant differences in CRC incidence when TZDs (p = 0.518; Figure 8(b)) or SGLT-2i (p = 0.155; Figure 8(c)) was used as control.

Forest plot of the subgroups between DPP-4i and the incidence of CRC across exposure time. (a) <1 year, p = 0.005; (b) 1–2 years, p = 0.854; >2 years, p = 0.924.
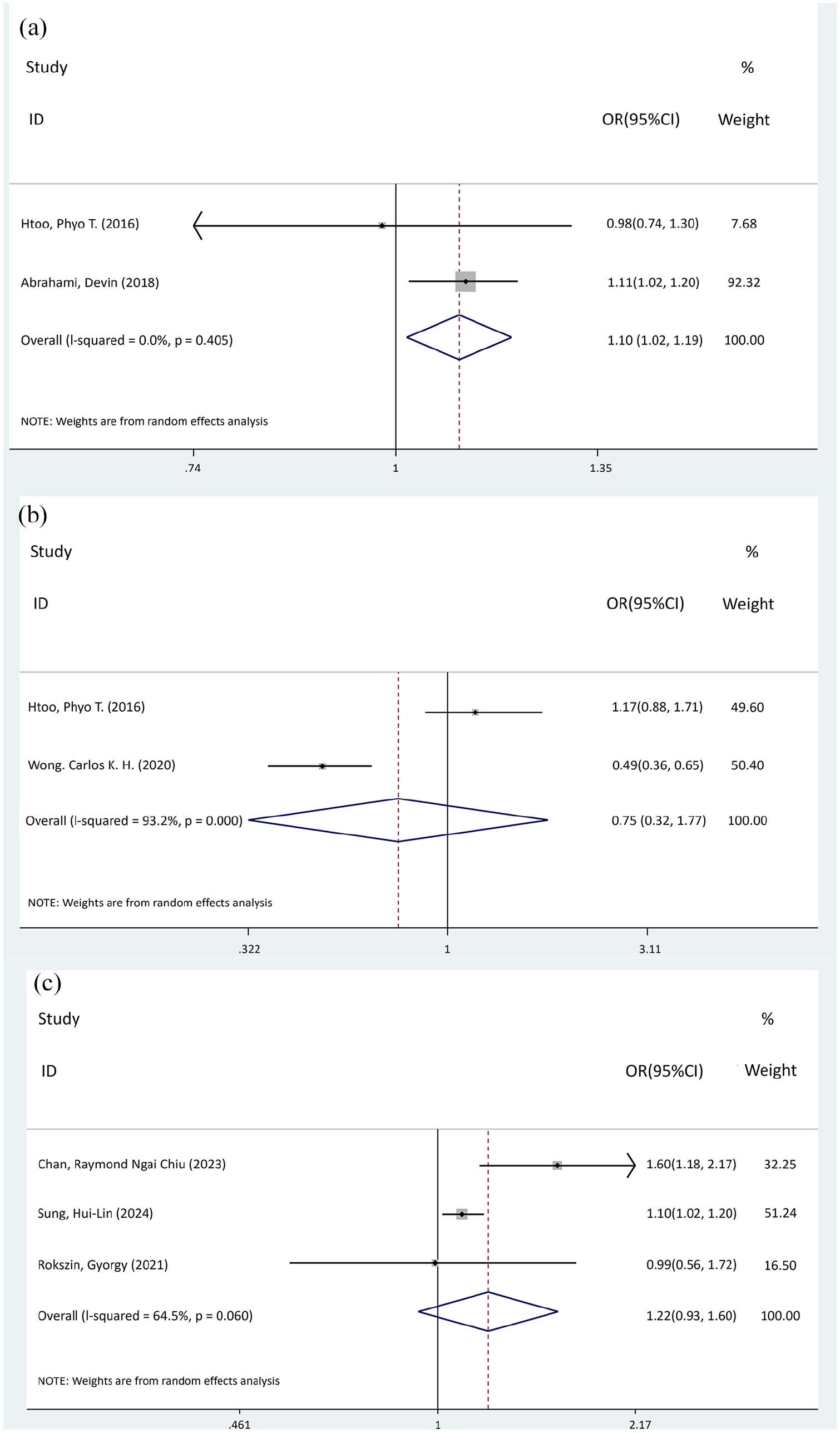
Forest plot of the subgroups between DPP-4i and the incidence of CRC across drugs in the control group. (a) SU, p = 0.017; (b) TZDs, p = 0.518; (c) SGLT-2i, p = 0.155.
Publication bias
To assess publication bias in the studies examining the relationship between exposure to DPP-4i and the incidence of CRC, Begg’s test was employed. The analysis revealed no significant publication bias (p = 0.386), as depicted in Supplemental Figure 1.
Discussion
This study investigates the association between exposure to DPP-4i and the incidence of CRC in patients with T2DM. Our comprehensive analysis of eight studies in this meta-analysis indicates a potential correlation between DPP-4i exposure and an increased incidence of CRC.
Patients with T2DM exhibit a higher incidence of CRC compared to non-T2DM individuals, which may be attributed to metabolic dysregulation and elevated levels of free insulin-like growth factor-1 (IGF-1). 17 Research has demonstrated that IGF-1 mRNA is highly expressed in CRC tissues relative to adjacent non-tumor tissues. 18 In addition, both colorectal and CRC cells express IGF-1 receptors, which, when activated by IGF-1 and insulin, can accelerate cellular proliferation and inhibit apoptosis. Given that T2DM patients typically have higher levels of IGF-1 and insulin, this condition may facilitate the rapid proliferation of CRC cells, ultimately leading to CRC. 19
DPP-4 is expressed by epithelial cells in the colon, rectum, liver, and kidney, and plays a role in inactivating cytokines associated with CRC progression, including IGF-1. 20 However, DPP-4i may impede this inactivation process by blocking DPP-4 activity, potentially increasing IGF-1 levels in T2DM patients, thereby promoting CRC development.
In addition, CRC cells express glucagon-like peptide-2 receptors (GLP-2R), and their migration activity and proliferation are significantly enhanced upon stimulation by GLP-2.21,22 Animal experiments have demonstrated that treating mice with GLP-2 analogs significantly accelerates CRC progression. 23 Normally, DPP-4 rapidly degrades GLP-2 in CRC patients, but exposure to DPP-4i prolongs the half-life of GLP-2, thereby potentially exacerbating CRC through enhanced GLP-2 activity.
Conversely, some studies suggest that DPP-4 facilitates the interaction between the interleukin-6 (IL-6) promoter and signal transduction proteins, increasing IL-6 gene activity. 24 IL-6 can amplify CRC gene expression through activation of cGMP, 25 indicating a positive correlation between DPP-4 activity and CRC gene expression. Therefore, DPP-4i might reduce CRC incidence by inhibiting IL-6 production. However, the relationship between DPP-4i exposure and CRC incidence in T2DM patients remains complex, as evidenced by the potential correlation identified in this study between DPP-4i exposure and increased CRC incidence.
Current data show that CRC incidence in men is 1.5 times higher than in women, 5 potentially due to higher rates of smoking and alcohol consumption among men. In addition, emerging research suggests the involvement of a “sex hormone-intestinal microbial axis” in CRC pathogenesis. Estrogen is known to promote the growth of bacteria that produce short-chain fatty acids (SCFAs), which regulate intestinal microbiota composition. SCFAs can activate the G protein-coupled receptor 109A (GPR109A) on human colon cancer cells, a receptor associated with apoptosis, thereby directly inhibiting colon cancer growth. By contrast, elevated levels of free testosterone increase the expression of inflammatory mediators such as cyclooxygenase-2 (COX-2) and inducible nitric oxide synthase (iNOS), exacerbating colon inflammation and raising colon cancer incidence. 26 This dual influence of male gender and DPP-4i suggests that DPP-4i may more significantly promote cancer in males than in females, a finding supported by our study’s observation of a higher incidence of CRC in male participants exposed to DPP-4i compared to controls.
From a geographical perspective, the incidence of CRC in Western countries and high-income Asia-Pacific regions was relatively high in 1990 but declined by 2019. By contrast, CRC incidence has increased in East Asia and Southeast Asia during the same period. 5 This trend may be attributed to the adoption of high-fat diets and Westernized lifestyles in Eastern countries. 27 However, the results of this meta-analysis indicate no significant correlation between exposure to DPP-4i and CRC incidence, regardless of whether patients are from Eastern or Western countries. This could be due to the limited sample size in the subgroup analyses, which may reduce statistical power. Thus, future studies should include a larger cohort to enhance the robustness of these findings.
Risk factors for T2DM include smoking, obesity, poor diet, and environmental influences. 28 Similarly, obesity, smoking, and excessive consumption of processed and red meats are primary risk factors for CRC. 29 Studies of early-onset T2DM (diagnosed before age 40) highlight obesity, high-sugar beverages, and smoking as prevalent characteristics contributing to the high incidence of T2DM among younger individuals.30,31 By contrast, disturbances in the intestinal flora are a principal risk factor in elderly T2DM patients. 32 Consequently, younger T2DM patients, who exhibit a broader range of CRC risk factors compared to their older counterparts, may experience an increased incidence of CRC under DPP-4i exposure. Fortunately, our statistical results reveal that while the incidence of CRC is higher in the DPP-4i-exposed group among patients under 65 years old, there is no significant difference between the exposed and control groups in patients over 65 years old. This suggests that the cancer-promoting effects of DPP-4i are more pronounced in younger individuals than in the elderly.
Previous studies have indicated that SU can stimulate the production of reactive oxygen species (ROS) in the pancreatic β cell line MIN6. ROS may induce the opening of mitochondrial permeability transition pores, consequently disrupting mitochondrial membrane potential and leading to adenosine triphosphate (ATP) depletion, which results in the release of mitochondrial Ca2+ and an increase in intracellular Ca2+. Elevated intracellular Ca2+ levels can induce apoptosis in CRC cells, thereby reducing the incidence of CRC. 33 Thus, DPP-4i may increase the incidence of CRC more than SU. Furthermore, no significant differences in CRC incidence were observed when the control drugs were TZDs and SGLT-2i, which could be attributed to variations in participant characteristics across studies, contributing to high heterogeneity in the statistical results. The underlying mechanisms require further investigation.
Regarding the duration of drug exposure, the incidence of CRC was higher in the exposure group than in the control group for periods less than 1 year, but no significant differences were found when the exposure exceeded 1 year. This finding contradicts the hypothesis that longer drug exposure increases risk factors. It suggests that in the short term (<1 year), the cancer-promoting effects of DPP-4i may be more pronounced. Over time, these effects may diminish and the anticancer properties may prevail, leading to a non-significant impact of DPP-4i on CRC in the long term. In addition, the long-term use of DPP-4i for blood glucose control may also contribute to a decreased incidence of CRC.
Limitations
This study represents a comprehensive systematic meta-analysis of the association between DPP-4i exposure and CRC incidence in T2DM patients. However, it is not without limitations. First, the inclusion of case–control and cohort studies without RCTs might introduce publication bias and reduce the reliability of the results. Second, the etiology of CRC involves complex interactions among genetic, environmental, and lifestyle factors, where the influence of DPP-4i may be limited. Due to the lack of individual data, it is often infeasible to control potential covariates within a meta-analysis, which makes the reliability of the results decline. Therefore, more clinical research is necessary to elucidate the specific role of DPP-4i in CRC development. Furthermore, the study is grounded in a substantial sample derived from both cohort and case–control studies, albeit necessitating acknowledgment that the inferred causal association may be tentative and warrants additional substantiation.
Conclusion
In summary, the findings of this study suggest a potential correlation between exposure to DPP-4i and an increased risk of CRC in patients with T2DM. This association appears to be influenced by factors such as gender, age, duration of drug exposure, and the type of control drugs used. Consequently, during the clinical administration of DPP-4i for glycemic control in patients with T2DM, meticulous attention must be afforded to the presence of colorectal disorders among this patient cohort. However, given the complex and multifactorial pathogenesis of CRC, the precise role of DPP-4i requires further investigation.
Supplemental Material
sj-doc-1-taw-10.1177_20420986251318842 – Supplemental material for Impact of dipeptidyl peptidase-4 inhibitors on incidence of colorectal cancer in patients with type 2 diabetes mellitus: a systematic review and meta-analysis
Supplemental material, sj-doc-1-taw-10.1177_20420986251318842 for Impact of dipeptidyl peptidase-4 inhibitors on incidence of colorectal cancer in patients with type 2 diabetes mellitus: a systematic review and meta-analysis by Rongrong Fu, Jingqi Chen, Yingying Fang, Qingping Wu, Xiaoming Zhang and Zhiyan Wang in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-2-taw-10.1177_20420986251318842 – Supplemental material for Impact of dipeptidyl peptidase-4 inhibitors on incidence of colorectal cancer in patients with type 2 diabetes mellitus: a systematic review and meta-analysis
Supplemental material, sj-docx-2-taw-10.1177_20420986251318842 for Impact of dipeptidyl peptidase-4 inhibitors on incidence of colorectal cancer in patients with type 2 diabetes mellitus: a systematic review and meta-analysis by Rongrong Fu, Jingqi Chen, Yingying Fang, Qingping Wu, Xiaoming Zhang and Zhiyan Wang in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-3-taw-10.1177_20420986251318842 – Supplemental material for Impact of dipeptidyl peptidase-4 inhibitors on incidence of colorectal cancer in patients with type 2 diabetes mellitus: a systematic review and meta-analysis
Supplemental material, sj-docx-3-taw-10.1177_20420986251318842 for Impact of dipeptidyl peptidase-4 inhibitors on incidence of colorectal cancer in patients with type 2 diabetes mellitus: a systematic review and meta-analysis by Rongrong Fu, Jingqi Chen, Yingying Fang, Qingping Wu, Xiaoming Zhang and Zhiyan Wang in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-4-taw-10.1177_20420986251318842 – Supplemental material for Impact of dipeptidyl peptidase-4 inhibitors on incidence of colorectal cancer in patients with type 2 diabetes mellitus: a systematic review and meta-analysis
Supplemental material, sj-docx-4-taw-10.1177_20420986251318842 for Impact of dipeptidyl peptidase-4 inhibitors on incidence of colorectal cancer in patients with type 2 diabetes mellitus: a systematic review and meta-analysis by Rongrong Fu, Jingqi Chen, Yingying Fang, Qingping Wu, Xiaoming Zhang and Zhiyan Wang in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-5-taw-10.1177_20420986251318842 – Supplemental material for Impact of dipeptidyl peptidase-4 inhibitors on incidence of colorectal cancer in patients with type 2 diabetes mellitus: a systematic review and meta-analysis
Supplemental material, sj-docx-5-taw-10.1177_20420986251318842 for Impact of dipeptidyl peptidase-4 inhibitors on incidence of colorectal cancer in patients with type 2 diabetes mellitus: a systematic review and meta-analysis by Rongrong Fu, Jingqi Chen, Yingying Fang, Qingping Wu, Xiaoming Zhang and Zhiyan Wang in Therapeutic Advances in Drug Safety
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
