Abstract
Background:
As the number of medications increases, the appropriateness of polypharmacy may become questionable due to the heightened risk of medication-related harm.
Objectives:
(1) To investigate the relationship between the number of current medications used by older adults and three indicators of potentially inappropriate polypharmacy: (a) the mean number of potentially inappropriate medications (PIMs), (b) the average count of drug–drug interactions, and (c) the anticholinergic burden; (2) To characterize the population-based burden of potentially inappropriate polypharmacy by calculating the proportion of individuals with these indicators.
Design:
We conducted a population-based observational study using the Quebec Integrated Chronic Disease Surveillance System.
Methods:
We included all individuals over 65 years insured by the public drug plan on April 1st, 2022. For each individual, we calculated the number of current medications and the number of (a) PIMs (Beers 2019), (b) drug–drug interactions (Beers 2019), and (c) anticholinergic burden (Anticholinergic Cognitive Burden (ACB) scale). The association between the number of medications and these indicators was quantified using linear regression. Prevalence with 99% confidence intervals (CIs) was calculated.
Results:
A total of 1,437,558 individuals (mean age: 75; 55% female) were included, taking an average of 4.9 medications (±4.1). For each additional medication, the mean number of PIMs, drug–drug interactions, and anticholinergic burden increased by 0.11, 0.04, and 0.17, respectively (
Conclusion:
The strong association between the increasing number of medications and reduced polypharmacy quality underscores the importance of medication count beyond therapeutic indications. With widespread medication use, many older adults face quality issues.
Plain language summary
Introduction
With the aging population, the use of multiple medications, known as polypharmacy, has become increasingly prevalent. 1 In the province of Quebec, Canada, a third of community-dwelling older adults were using 10 or more prescribed medications in 2022. 1 The World Health Organization has outlined new goals aimed at ensuring optimal medication use by 2030. 2 However, even when polypharmacy is deemed appropriate for managing all existing conditions, it can be challenging to ensure that its benefits outweigh its risks. 3 As the number of medications increases, so does the risk of medication-related problems. Potential harm may arise from the use of medications whose associated risks outweigh the benefits. Such potentially inappropriate medications (PIMs) are populated in lists developed by recognized experts. The Beers criteria, for example, notably contains 30 medications or medication classes to be avoided in older adults. 4 It is very common for older adults to use PIMs; for instance, in 2022, half of community-dwelling older adults in Quebec had at least one of such PIMs. 1 In addition, drug–drug interactions can lead to adverse outcomes. Drug–drug interactions that should be avoided in older adults are similarly listed in the Beers criteria. 4 Harm can further result from the anticholinergic burden, which represents the cumulative anticholinergic effect of a medication regimen. Increased anticholinergic burden has been associated with a heightened risk of cognitive impairment, among other adverse effects. 5
Studies have demonstrated that polypharmacy is the strongest predictor of PIM use,6,7 and that it is also associated with drug–drug interactions, 8 and an increase in anticholinergic burden. 9 Therefore, the quality of a medication regimen may be compromised by the high number of medications, 10 even when theoretically required by an individual’s conditions. Although polypharmacy is associated with PIMs, drug–drug interactions, and anticholinergic burden, to our knowledge, no study has empirically quantified these associations on a population-wide scale. Establishing a connection between the number of medications and the quality of a medication regimen could assist healthcare providers and public health efforts in identifying and addressing potentially at-risk polypharmacy cases. This approach could aid in distinguishing appropriate from potentially inappropriate polypharmacy, a recognized need.3,11–13
The overarching aim of our study was to characterize the association between quality and quantity of medications, beyond clinical characteristics and therapeutic indications. Our specific objectives were (1) to investigate the relationship between the number of medications used by older adults and three indicators of potentially inappropriate polypharmacy: (a) the mean number of PIMs, (b) the average count of drug–drug interactions, and (c) the anticholinergic burden; (2) to characterize the population-based burden of potentially inappropriate polypharmacy by calculating the proportion of individuals with these indicators.
Methods
Study design and data source
We conducted a population-based study using the Quebec Integrated Chronic Disease Surveillance System (QICDSS), managed by the National Public Health Institute of the province of Quebec, Canada (
With data available since 1996, the QICDSS combines, under a unique anonymized identification number, information from five databases: (1) the health insurance registry, which contains sociodemographic data; (2) the MED-ÉCHO database, which contains hospitalization data; (3) the vital statistics death database, which contains the date of death and associated causes; (4) the physician claims database, which contains data on all fee-for-service billings, including diagnostic codes; (5) the pharmaceutical services database, which contains data on prescription medication claims under the public drug plan. The public health plan, which encompasses hospitalization and medical care coverage, covers approximately 99% of the province’s population. 15 Furthermore, about 90% of individuals aged 65 years and older are covered by the public drug plan, which specifically addresses medication coverage. 15
Study population
We identified all community-dwelling individuals aged over 65 years who were covered by the public drug plan on April 1st, 2022. Individuals had to be covered by the public health plan throughout 2022 and by the public drug plan from October 1st, 2021, to April 1st, 2022. We chose a 6-month lookback period to consider medications with infrequent dispensing (e.g., denosumab). We excluded individuals residing in long-term care facilities because information on their medication use is not included in the QICDSS.
Current medications on April 1st, 2022
The common denomination code (equivalent to the fifth level of the Anatomical Therapeutic Chemical (ATC) classification) was used to ascertain medications in the pharmaceutical services database of the QICDSS. Medications with multiple active ingredients were classified under one code (e.g., a tablet with two active ingredients was counted as one medication). For a medication to be considered, its duration of treatment, as recorded by the pharmacist at the time of dispensing, had to include the date of April 1st, 2022 (Supplemental Figure S1). As-needed medications were also included under the same condition. All types of medications were considered (oral, topical, inhaled, etc.). Medications not covered by the public drug plan (e.g., over-the-counter medications, natural products) are not included in the database and therefore could not be assessed. The number of medications taken by each individual on April 1st, 2022, was calculated. We created categories for each number of medications (1, 2, . . ., ⩾20). For the analyses, we combined users of ⩾20 medications as they represented less than 0.5% of the population. The number of medications in 1 day represents the clinical situation where an individual presents themselves to a health professional with their medication list.
PIMs, drug–drug interactions, and anticholinergic burden
PIMs were identified using the 2019 update of the American Geriatrics Society Beers Criteria. 4 These criteria are widely used in studies conducted with administrative data because they require limited clinical information.16–18 The list was adapted to align with available medications in Canada and data in the QICDSS. 16 Any medication listed in Table 2 of the Beers criteria was considered a PIM. However, doxepin and digoxin were excluded as PIMs since dosage information was not considered in our study. Aspirin was classified as a PIM if it was used for primary prevention, according to the 2023 update of the Beers criteria. 19 Diagnostic codes related to coronary events, heart failure, and cerebral or vascular diseases were used to ascertain the indication of aspirin in secondary prevention. This approach was unique to aspirin because the change in recommendation for aspirin use was significant between 2019 and 2023.4,19 Aspirin’s role in primary prevention has been subject to extensive new research and guideline updates, 19 making it a critical exception. The total number of PIMs for each individual was calculated, with each medication counted once, even if it appeared multiple times in the Beers criteria. We further categorized individuals into two groups: no PIMs and ⩾1 PIMs.
Drug–drug interactions were identified using the 2019 update of the American Geriatrics Society Beers Criteria which lists “potentially clinically important drug–drug interactions that should be avoided in older adults.” 4 A combination of medications listed in Table 5 of the Beers criteria was considered as a drug–drug interaction. The total number of drug–drug interactions for each individual was calculated. We further categorized individuals into two groups: no drug–drug interactions and ⩾1 drug–drug interactions.
The anticholinergic burden was measured using the Anticholinergic Cognitive Burden (ACB) scale. This validated scale has been used to measure risk of cognitive decline in older adults.20,21 The anticholinergic burden score was assigned to each medication (with ACB levels ranging from 0 to 3), 5 and the scores were subsequently summed for each individual according to the medications they were taking. We further categorized ACB levels into three dichotomous groups: ACB level ⩾1 (including ACB levels ⩾2 and ⩾3, compared to ACB level 0), ACB level ⩾2 (including ACB level ⩾3, compared to ACB levels 0 and 1), and ACB level ⩾3 (compared to ACB levels 0, 1, and 2). Higher ACB levels indicate a greater anticholinergic burden and an increased risk of cognitive impairment, among other negative consequences. 5
Other variables
Age, sex, social and material deprivation index, living zone (urban vs rural), and the presence of chronic diseases were used exclusively to characterize the studied population and were not incorporated into subsequent analyses, aligning with the objective to examine pharmacotherapy independently of the patient’s characteristics. Deprivation indexes are validated proxies of socioeconomic status based on postal code. 22 The quality of social networks can be expressed by the social deprivation index, whereas the quality of material goods owned by the population studied can be gauged by the material deprivation index. The first quintile of both indexes includes the least deprived individuals while the fifth quintile contains the most deprived. 22 We used validated definitions, that is standardized, evidence-based set of criteria based notably on diagnosis codes and procedures, to accurately identify individuals with the following chronic diseases: asthma, chronic obstructive pulmonary disease, dementia, diabetes, hypertension, mood disorders, osteoporosis, schizophrenia, and stroke. 15 In addition, we calculated the comorbidity score, considering the 31 diseases included in the combined comorbidity score, which has been validated for the Quebec population. 23 The comorbidity score predicts the impact of chronic diseases on mortality, with an increase in risk associated with a higher score. 23
Statistical analyses
All individuals of the population, and all individuals with at least one medication on April 1st, 2022, were characterized with descriptive statistics. We studied the entire population, not just those with medications, to assess the public health impact of potentially inappropriate polypharmacy. While focusing on individuals with medications is clinically valid and relevant, examining the broader population highlights the potential for substantial public health benefits from population-wide interventions.
The mean number of PIMs, of drug–drug interactions, and the mean anticholinergic burden score, with their 99% confidence intervals (CIs), were calculated for each number of current medications. We used 99% CI, which is a stricter threshold than the conventional 95%, because our large population provides very high statistical power, allowing even small differences to be statistically significant. The association between the number of current medications and (1) mean number of PIMs, (2) mean number of drug–drug interactions, and (3) mean anticholinergic burden score was observed graphically to facilitate the visualization and interpretation of the relationship between the number of current medications and those continuous variables.24,25 Because the relationship was mostly linear, we conducted three separate univariate linear regressions to assess the association (trend) between the number of current medications (treated as a continuous variable) and each of the following: the mean number of PIMs, drug–drug interactions, and the ACB score.
For each group of individuals with the same number of current medications, we calculated the proportion of individuals with (1) at least one PIM, (2) at least one drug–drug interaction, (3) an anticholinergic burden of ⩾1, ⩾2 and ⩾3. In addition, we determined the proportion of individuals with potentially inappropriate polypharmacy, defined as having at least one of the three indicators (PIM, drug–drug interaction, or anticholinergic burden) for each group with the same number of current medications. From a public health perspective, to estimate the burden of potentially inappropriate polypharmacy in the population, we calculated the proportion of individuals exposed to one of the three indicators (PIMs, drug–drug interactions, anticholinergic burden) using the entire population as the denominator. We also calculated those proportions using the number of individuals with at least X number of current medications as the denominator.
Sensitivity analyses were conducted to validate the robustness of the association between the number of current medications and the three indicators. We used 2019 data for temporal validation, as it is the most recent year before the pandemic and the year of the last update of the Beers criteria before our study period. 4
All analyses were conducted using SAS version 9.4 (SAS Institute, Cary, NC, USA).
Ethics
The provincial Public Health Research Ethics Board and the Quebec Commission protecting access to information have approved the use of the QICDSS for surveillance purposes. No written informed consent was required. The ethics board of the
Results
There were 1,665,850 community-dwelling individuals aged over 65 years on April 1st, 2022. We excluded 228,292 individuals who were not covered by the public health plan throughout 2022 or by the public drug plan from October 1st, 2021, to April 1st, 2022. Thus, our study included 1,437,558 individuals (mean age 75.5 years, 54.8% female, mean number of medications 4.9 ± 4.1; Table 1). From them, 1,214,195 (84.5%) had at least one medication on April 1st, 2022 (mean: 5.8 ± 3.8 medications; Table 2). Most medication users (55.9%) had at least five current medications. More than 4 in 10 individuals in the population (43.9%) had at least one PIM, 6.0% had at least one drug–drug interaction, and 29.5% had an ACB level of one or higher.
Characteristics of community-dwelling adults aged over 65 years insured by the public drug plan in Quebec, Canada, on April 1st, 2022.

Presence of individual comorbidities using the validated case definitions of the Quebec Integrated Chronic Disease Surveillance System.
Number of current medications, potentially inappropriate medications, drug–drug interactions, and anticholinergic burden level in community-dwelling adults aged over 65 years insured by the public drug plan in Quebec, Canada, on April 1st, 2022.

Since our study focused on a single day, some individuals may not have been recorded as using medications on that specific day, despite actually being on treatment. This could include individuals who were late in renewing their prescriptions or those who were previously hospitalized, where medications might have been provided by the hospital while they still had prescriptions at home. In addition, some individuals with chronic conditions may be managed through non-pharmacological treatments, particularly among the youngest segment of our older population. Finally, those who are at the end of life may not be using medications.
ACB, Anticholinergic Cognitive Burden.
The relationship between the number of current medications used by older adults and the mean number of PIMs, the average count of drug–drug interactions, and the anticholinergic burden is presented graphically in Figure 1. The presence of PIMs, drug–drug interactions, and anticholinergic burden increased mostly linearly with the number of current medications (Figure 1; Supplemental Table S1). Linear regression indicated that with each additional medication, the mean number of PIMs, drug–drug interactions, and the anticholinergic burden increased by 0.11, 0.04, and 0.17, respectively (

Association between potentially inappropriate medications, drug–drug interactions, anticholinergic burden, and the number of medications in community-dwelling medication users aged over 65 years in Quebec, Canada, on April 1st, 2022.
To address objective 2 from a clinical perspective, Figure 2 shows the proportions of individuals with PIMs, drug–drug interactions, and anticholinergic burden, according to the number of current medications they were taking. For example, among individuals with five current medications, more than half (53.8%) had a least one PIM, 2.9% had at least one drug–drug interaction, and 31.0% had an ACB level ⩾1. Among individuals with eight current medications, the proportions increased: 76.1% had a least one PIM, 9.6% had at least one drug–drug interaction, and 51.8% had an ACB level of one or more. Among individuals with 10 current medications, all proportions further increased: 83.5% had a least one PIM, 17.2% had at least one drug–drug interaction, and 63.5% had an ACB level of one or more.
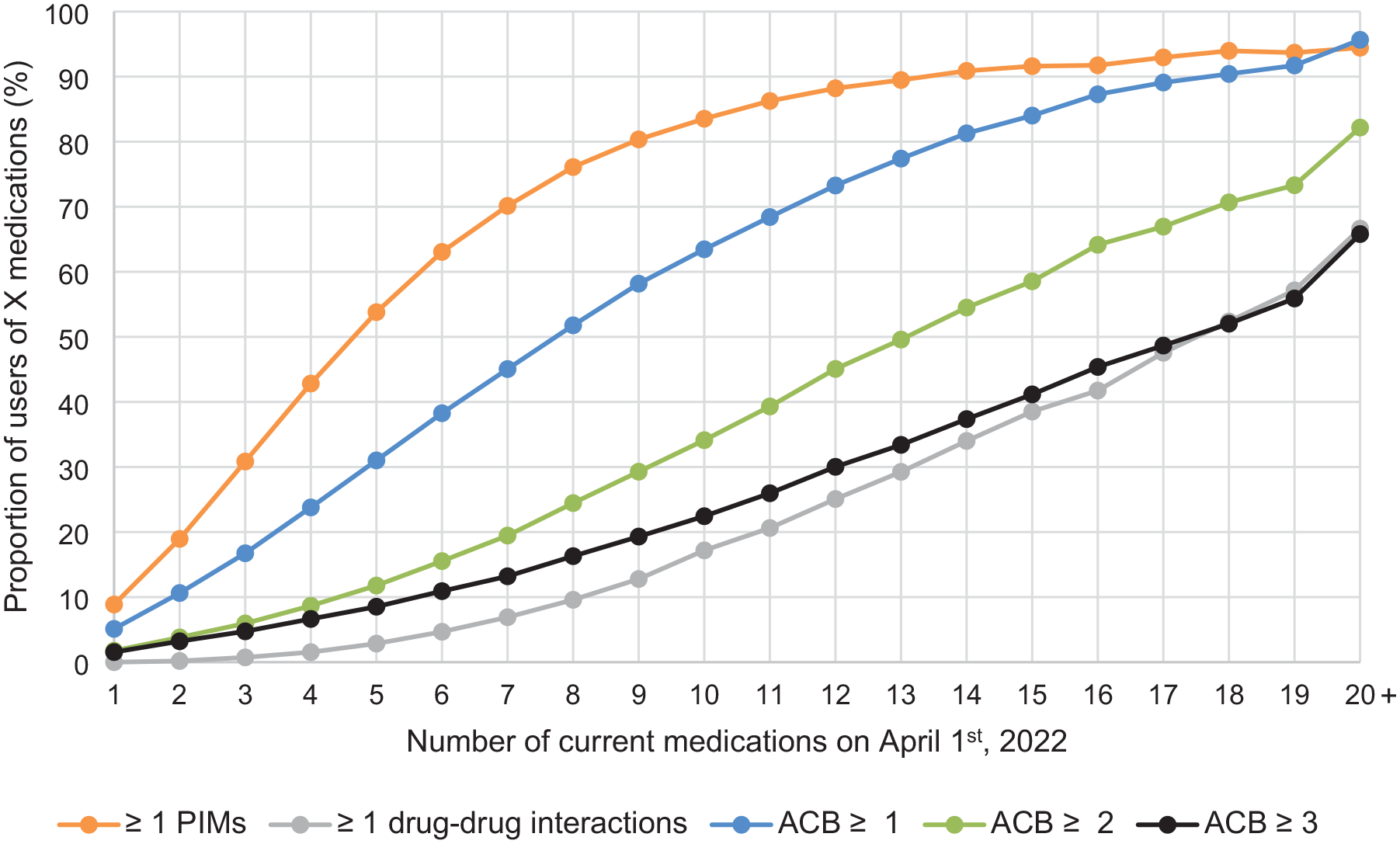
Proportions of community-dwelling medication users aged over 65 years in Quebec, Canada, with indicators of potentially inappropriate polypharmacy according to the number of current medications on April 1st, 2022.
Table 3 exposes the population-based burden of potentially inappropriate polypharmacy. On April 1st, 2022, 52.0% of the population had at least one PIM, one drug–drug interaction, or an ACB level ⩾1, and 45.5%, with an ACB ⩾3 (Table 3). Nearly 40% of the population had at least five current medications including at least one indicator of potentially inappropriate polypharmacy (Table 3). Those using at least 10 current medications represented 13.1% of the population, and those combining 10 current medications with at least one of the three indicators comprised 12.6% of the population (Table 3). The burden of potentially inappropriate polypharmacy when restricted to medication users is included in Supplemental Files (Supplemental Tables S2 and S3). For example, among individuals using at least five current medications, 83.4% had therapy with at least one indicator (Supplemental Table S3).
Proportion of community-dwelling individuals aged >65 years in Quebec, Canada, with at least one PIM, one drug–drug interaction, or an anticholinergic burden according to the number of current medications on April 1st, 2022.

The burden of potentially inappropriate polypharmacy when restricted to medication users is presented in Supplemental Table S2.
ACB, Anticholinergic Cognitive Burden; PIM, potentially inappropriate medication.
99% confidence intervals are identical to the proportions presented in two decimal places. They are not shown to simplify reading.
Example of interpretation: 39.3% of individuals in the whole population have at least five current medications and at least one indicator.
Sensitivity analysis
Our analyses of the 2019 data showed that the association between the number of medications and the mean number of PIMs, drug–drug interactions, and ACB level is stable over time. The proportion of individuals using PIMs, drug–drug interactions, or an ACB level ⩾1, ⩾2, and ⩾3 was also stable over time (Supplemental Tables S4–S9; Supplemental Figures S2 and S3).
Discussion
We empirically described the strong association between the increasing number of medications and the increasing presence of PIMs, drug–drug interactions, and anticholinergic burden in community-dwelling medication users aged over 65 years.
Our study is among the few that simultaneously examines the association between the number of current medications and PIMs, drug–drug interactions, and ACB burden.10,17,26 Moreover, it is one of the few studies to empirically quantify these associations. Several studies have highlighted the association between polypharmacy and the use of PIMs. A systematic review focusing on community-dwelling older adults in the United States showed a consistent association between polypharmacy and the use of PIMs from the Beers criteria. 7 Similar results were also reported in Europe 27 and China. 28 However, these studies typically measured the risk of using at least one PIM, rather than the number of PIMs. For instance, an analysis of administrative data from 1996 in older American adults revealed that those taking more than the median annual number of medications (14) faced nearly triple the risk of having a medication from the Beers list (odds ratio = 2.9; 95% CI: 2.3–3.6) compared to those taking 14 and fewer medications. 29 More recent data from the US Veterans database for 2015–2016 indicated that the prevalence of PIM users increased with the number of medications taken: 14.3%, 59.9%, and 83.3% among individuals using 0–4, 5–9, and ⩾10 chronic medications, respectively. 30 Our study contributes to the literature by precisely quantifying the linear association between the number of medications used and the number of PIMs. Regarding drug–drug interactions, a similar association was exposed between simultaneous polypharmacy and drug–drug interactions in a 2005 observational study of Swedish adults aged 75 years and over (mean age of 82 (±5.3) years old). 8 In a small Turkish study involving 420 outpatients aged 60 years and over, individuals using ⩾5 medications received a mean number of 8.1 ± 2.9 medications and had a mean ACB score of 0.99 ± 1.13. 9 This aligns with our study showing a mean ACB score of 1.0 at a threshold of eight medications.
While the presence of PIMs, drug–drug interactions, and anticholinergic burden monotonically increase with the increasing number of current medications, some thresholds are interesting to discuss. From the threshold of five medications, more than 50% of individuals within each category of medication users were exposed to at least one PIM. The presence of PIMs was the indicator that most contributed to the prevalence of potentially inappropriate polypharmacy in our study. Anticholinergic burden also contributed to potentially inappropriate polypharmacy, with 50% of the population being exposed to an ACB level ⩾1 at a threshold of eight medications. Drug–drug interaction was the indicator that contributed less to the prevalence of potentially inappropriate polypharmacy. This may be because prescribers and pharmacists have been aware of the risks of drug–drug interactions for a long time and that several software programs offer assistance for their management.31,32 This could indicate that efforts to improve medication use that are only focused on reducing drug–drug interactions would miss more pressing opportunities to improve medication regimens.
The threshold of five medications is commonly used to define polypharmacy.11,13 Our results show that 47.1% of the population had at least five medications, and 83.4% of them had a regimen that could potentially increase medication-related harm. Thus, from a public health perspective, our results show that the traditional threshold of five medications is a reasonable cut-point for reviewing medication regimens to ensure they are appropriate. However, given the time and resource constraints associated with clinical activities in pharmacy, it may be reasonable to set a threshold higher than five medications for focusing medication review efforts on a targeted number of older adults.33–35 For example, individuals with at least eight medications represent a quarter of our population, among whom nearly everyone (93.2%) was at increased risk of medication-related harm.
There is a pressing need to better characterize inappropriate polypharmacy from appropriate polypharmacy.3,11,13,36 At a population level, the clinical definition of appropriate polypharmacy is challenging to apply, as the number and types of medications needed may vary based on an individual’s diseases, health goals, frailty status, and life expectancy perspectives. 37 However, our results reveal a strong association between the increasing number of medications and the appropriateness of polypharmacy. Based on the three indicators, polypharmacy could be deemed potentially inappropriate once the number of current medications exceeds a specific threshold. This assessment can be conducted independently of clinical conditions or treatment goals. Such an approach offers significant advantages from a public health standpoint, especially considering the limited availability of clinical data and the life goals of patients.
Strengths and limits
Our study has many strengths. Our population-based study gives a detailed and recent portrait of medication use in community-dwelling older adults. The analyses and variables are easy to interpret and implement in clinical contexts and for public health surveillance.
The model is not intended to explain the relationship between the variables, identify strategies to address the issue, or pinpoint factors for intervention to optimize medication use. Rather, it serves as a reminder that the quality of pharmacotherapy tends to be compromised when the number of medications is high, regardless of the underlying reasons for this increase. Our study shares the limitations of administrative databases. While we measured the dispensing of medications, we cannot guarantee their actual use. This limitation can impact levels of drug–drug interactions and anticholinergic burden, as both depend on the simultaneous use of medications. This assumption may not always hold, particularly if medications are taken only as needed. Therefore, the burden may have been overestimated in some cases. On the other hand, medication use can be underestimated by the exclusion of over-the-counter medications and medications excluded from the public insurance plan. Accordingly, the measure of all three indicators may also be underestimated. Also, PIM use was likely underestimated since we excluded medications that are PIMs according to their dosage (e.g., digoxin, doxepin). We believe this underestimation has little impact on our results because a previous study of a representative sample of Quebec’s 2017 population of individuals aged over 65 years revealed that only 0.1% and 0.2% were users of digoxin ⩾0.125 mg/day and doxepin ⩾6 mg/day, respectively. 16 To make sure our results remain relevant to clinical practices and trends, such as updates of the Beers criteria, our analysis should be repeated periodically. Despite those limits, our sensitivity analysis indicates that the strong association between the number of medications and the studied indicators is likely to persist. Future research should investigate the risks of adverse outcomes associated with different polypharmacy thresholds.
Conclusion
Our study confirms that the increased simultaneous medication use can lead to potential inappropriate polypharmacy. The numbers of PIMs, drug–drug interactions, and the anticholinergic burden continuously increase with the number of medications dispensed. Thus, there is a strong association between the increasing number of medications and the increasing potential for medication harm, with quality potentially compromised at traditional thresholds of polypharmacy. Our study reveals that even if all the medications are indicated, healthcare providers should carefully consider the number of medications because of the relationship between the number of medications and the potential for poor-quality outcomes.
Supplemental Material
sj-docx-1-taw-10.1177_20420986241309882 – Supplemental material for Association between number of medications and indicators of potentially inappropriate polypharmacy: a population-based cohort of older adults in Quebec, Canada
Supplemental material, sj-docx-1-taw-10.1177_20420986241309882 for Association between number of medications and indicators of potentially inappropriate polypharmacy: a population-based cohort of older adults in Quebec, Canada by Alexandre Campeau Calfat, Justin P. Turner, Marc Simard, Véronique Boiteau and Caroline Sirois in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-2-taw-10.1177_20420986241309882 – Supplemental material for Association between number of medications and indicators of potentially inappropriate polypharmacy: a population-based cohort of older adults in Quebec, Canada
Supplemental material, sj-docx-2-taw-10.1177_20420986241309882 for Association between number of medications and indicators of potentially inappropriate polypharmacy: a population-based cohort of older adults in Quebec, Canada by Alexandre Campeau Calfat, Justin P. Turner, Marc Simard, Véronique Boiteau and Caroline Sirois in Therapeutic Advances in Drug Safety
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
