Abstract
Background:
As prostaglandin medications, crucial in glaucoma treatment, become more widely used, their local adverse events are increasingly observed.
Objectives:
To evaluate the common adverse events of four clinically commonly used prostaglandin F (FP) receptor agonists in the treatment of glaucoma in the Food and Drug Administration Adverse Event Reporting System (FAERS) database.
Design:
We screened and analyzed the generic and brand names of latanoprost, bimatoprost, travoprost, and tafluprost in the FAERS database and summarized and cleaned the baseline information of subjects receiving the above-mentioned drugs.
Methods:
Perform descriptive statistical analysis on the baseline information of subjects using the drugs. Conduct disproportionality analysis of drug-related adverse events. The criteria for positive signals of adverse events are established by simultaneously meeting the thresholds set by four methods: the ratio of reported odds, proportional reporting ratio, Bayesian confidence propagation neural network, and multi-item gamma Poisson shrinker. Additionally, assess the cumulative risk curves for drug-induced time of the aforementioned drugs and use one-way ANOVA to compare differences in drug-induced time across different groups.
Results:
The study included 1567 latanoprost, 1517 bimatoprost, 696 travoprost, and 82 tafluprost subjects. Adverse events mainly affected eye disorders, with significant issues in iris hyperpigmentation, ocular pemphigoid, corneal endothelial cell loss, periorbital fat atrophy, corneal irritation, eyelash growth, and ocular hyperemia. The time to onset varied among drugs, with latanoprost showing the longest (mean days = 344.37) and bimatoprost the shortest duration (mean days = 155.65; p < 0.001).
Conclusion:
Although signal detection analysis based on the FAERS database cannot establish a definitive causal relationship, our study found that FP receptor agonists used in glaucoma can cause various adverse events. Assessing their clinical suitability and potential side effects is crucial for providing personalized treatment and ensuring medication safety.
Plain language summary
Why was the study done? Prostaglandin medications are crucial in treating glaucoma but can cause local adverse events. As the use of these medications increases, it’s important to understand their common side effects. The Food and Drug Administration Adverse Event Reporting System (FAERS) is a database that contains adverse event reports, medication error reports and product quality complaints resulting in adverse events that were submitted to the Food and Drug Administration.
What did the researchers do? We analyzed the FAERS database to evaluate the common adverse events of four prostaglandin medications commonly used to treat glaucoma: latanoprost, bimatoprost, travoprost, and tafluprost.
What did the researchers find? The study included 1567 latanoprost users, 1517 bimatoprost users, 696 travoprost users, and 82 tafluprost users. The main adverse events affected eye disorders, with significant issues including iris hyperpigmentation, ocular pemphigoid, corneal endothelial cell loss, periorbital fat atrophy, corneal irritation, eyelash growth, and ocular hyperemia. The time to onset varied among drugs, with latanoprost showing the longest and bimatoprost the shortest duration.
What do the findings mean? Although signal detection analysis from the FAERS database cannot establish a definitive causal relationship, prostaglandin medications used in glaucoma treatment can cause various ocular adverse events during long-term use. Understanding these side effects is crucial for providing personalized treatment and ensuring medication safety.
Introduction
Glaucoma is the second leading irreversible cause of blindness globally, characterized by optic nerve atrophy and visual field defects. 1 It is primarily an ocular disease characterized by pathologically elevated intraocular pressure (IOP), and it can be classified into open-angle glaucoma, angle-closure glaucoma, secondary glaucoma, and progressive glaucoma. 2 The treatment measures for glaucoma mainly include medication, laser therapy, and surgical intervention, with drug therapy being the primary means to lower IOP. Therefore, glaucoma patients typically require long-term use of eye drops to control IOP, and some patients may even need lifelong medication. 3 Prostaglandin drugs are first-line clinical medications for treating glaucoma and are widely used in clinical practice.4,5
Naturally occurring prostaglandins, such as PGF2α, exhibit low receptor affinity and nonspecific binding; for instance, PGF2α can bind to prostaglandin F (FP) as well as EP1, EP2, and EP3 receptors. However, prostaglandin derivatives (prostanoids) present in prostaglandin-class antiglaucoma eye drops have a strong affinity and readily bind to their respective receptors. 6 These prostanoids mainly include derivatives of PGD2, PGE2, and PGF2α.7,8 Among them, derivatives of PGF2α are considered the most effective local ocular hypotensive agents, primarily reducing IOP by increasing aqueous humor outflow through the uveoscleral pathway.2,8 Approximately 20%–40% of aqueous humor outflow occurs through the uveoscleral pathway, not influenced by the mechanism of IOP reduction by prostaglandin drugs that impact the trabecular meshwork outflow pathway. 9 Prostaglandin antiglaucoma eye drops commonly employ derivatives of PGF2α, owing to their relatively fewer adverse effects and relatively low frequency of drug usage (once daily), with the most widely used in clinical practice being FP receptor agonists, 10 such as latanoprost, 11 bimatoprost, 12 travoprost, 13 and tafluprost. 14 However, adverse events induced by long-term use of prostaglandin drugs for glaucoma treatment have been increasingly reported. These drugs may impact the digestive system, respiratory system, cardiovascular system, skin, and hair. Adverse effects on the digestive system manifest as gastrointestinal reactions and abnormal liver function, 15 while respiratory system effects may include symptoms resembling colds and upper respiratory tract infections. 16 Recent literature reports suggest potential cardiovascular effects, including localized myocardial ischemia, angina, or reduced heart rate. 17 Effects on the skin and hair may include the occurrence of white hair, increased hair growth, and the development of rashes, blisters, and papules. 18 Ocular application of prostaglandin drugs can lead to local adverse events such as conjunctival hyperemia, thickening and elongation of eyelashes, darkening of periocular skin and iris pigmentation, eyelid inflammation, and macular edema. 19 Therefore, evaluating the clinical adverse events of prostaglandin drugs is of crucial significance in optimizing the clinical medication for glaucoma patients.
The Food and Drug Administration Adverse Event Reporting System (FAERS) database is widely utilized for assessing adverse events occurring during clinical and patient medication processes by collecting real-world sample data, and it has been extensively employed to evaluate risk signals for drug adverse events. 20 Building upon this, the present study utilizes the FAERS database to assess the adverse events of four of the most commonly used prostaglandin drugs for glaucoma treatment in clinical practice. Leveraging large-sample real-world data, this study holds crucial significance in guiding the clinical optimization of medication decisions for glaucoma patients and offering personalized treatment plans.
Methods
Study design and data source
We conducted a retrospective pharmacovigilance study based on the FAERS database, which is globally recognized as an openly accessible repository for adverse event reports (https://fis.fda.gov/extensions/FPD-QDE-FAERS/FPD-QDE-FAERS.html). The FAERS database encompasses voluntary reports from various sources, including healthcare professionals, patients, pharmacists, and pharmaceutical companies, supporting the FDA’s post-marketing surveillance initiatives for drugs and therapeutic biologics. 21 It includes patient information, adverse event data, drug usage details, report sources (RPSR), treatment duration, drug indications, and patient outcomes (OUTC). The database adheres to national safety reporting guidelines, encoding all adverse events using the preferred terms (PTs) from the Medical Dictionary of Regulatory Activities. Moreover, PTs can be further categorized into high-level group terms (HLGTs) and system organ classes (SOCs), or clustered using standardized MedDRA queries (SMQs) for specific medical conditions. The database comprises seven categories of data: demographic and administrative information (DEMO), drug information (DRUG), indications for use (INDI), adverse events (REAC), OUTC, RPSR, and drug therapy start and end dates (THER). Given the public accessibility of the FAERS database and the anonymous and de-identified nature of patient records, the study does not involve informed consent or ethical approval. To ensure the inclusion of the most recent and comprehensive reports, we extracted all FAERS reports recorded from the first quarter of 2004 to the third quarter of 2023. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (Supplemental Material). 22
Procedures
Between the first quarter of 2004 and the third quarter of 2023, the FAERS database accumulated a total of 20,214,432 original reports, reduced to 16,964,230 unique reports after eliminating duplicates, 23 encompassing 804,070 different drugs. To mitigate the influence of combination therapy and drug coadministration on the relationship between drugs and adverse events, we specifically selected the “Primary Suspect Drug” (PS) code for drug’s reported role in the event,24–26 while excluding “Secondary Suspect Drug,” “Concomitant,” and “Interacting.” Among these, we focused on the top four FP receptor agonists for glaucoma treatment: latanoprost (62,799 reports), bimatoprost (25,518 reports), travoprost (13,092 reports), and tafluprost (2569 reports). The corresponding reported subjects for these drugs were 1,567, 1,517, 696, and 82, respectively. To analyze drug information and adverse event reports from subjects, disproportionality analysis methods were employed for signal detection, as outlined in the process flow in Figure 1.
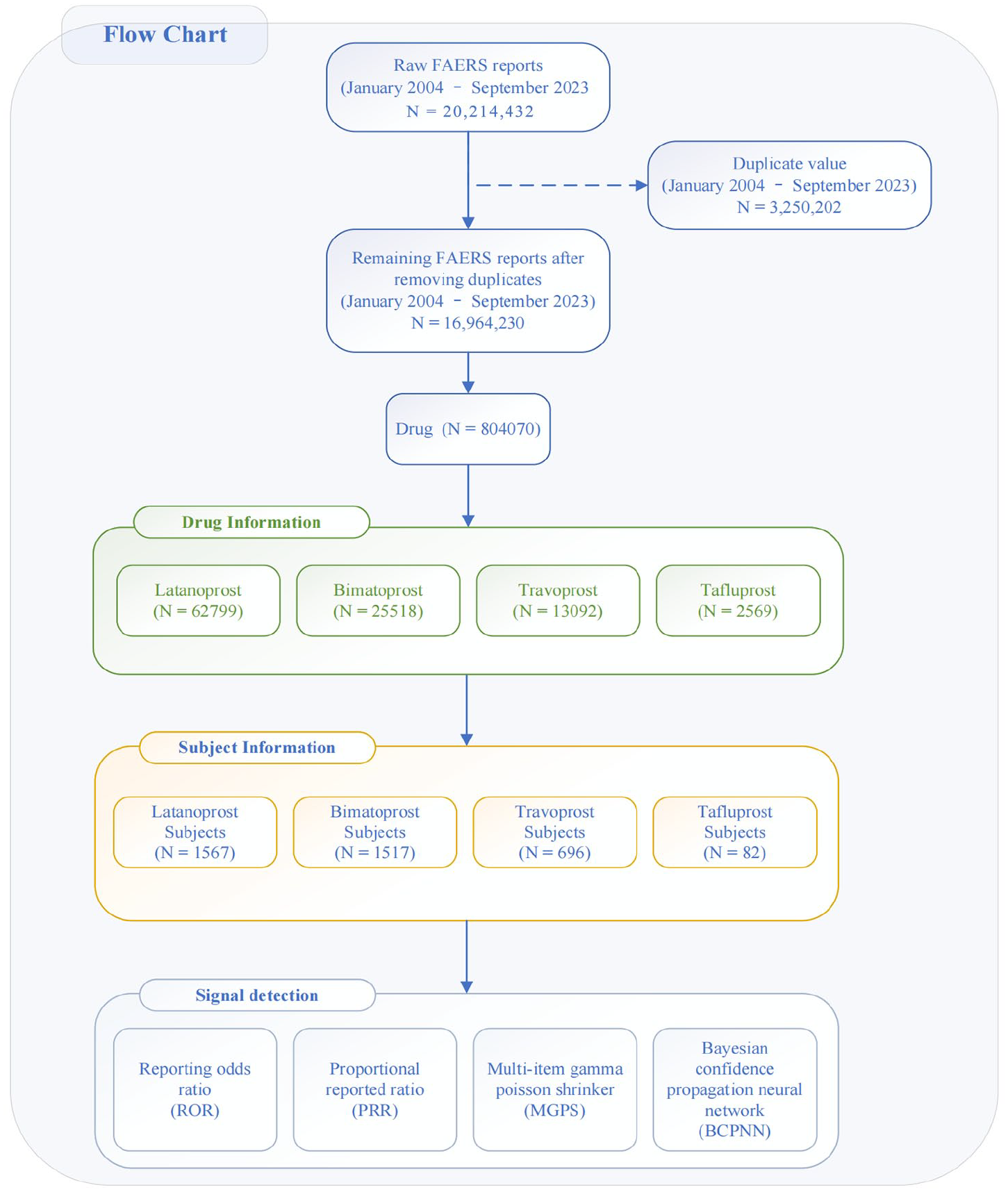
Flowchart of data cleaning process for adverse event data of prostaglandin analog drugs for glaucoma based on the FAERS database.
Statistical analysis
Signal detection utilized the ratio of reported odds (ROR), 27 proportional reported ratio (PRR), 28 Bayesian confidence propagation neural network (BCPNN), 29 and multi-item gamma Poisson shrinker (MGPS) 30 of the disproportionality method. The four methods mentioned above are based on mining potential positive signals through the comparison of target events and target drugs with all other events and drugs using a fourfold table calculation method (Tables 1 and 2). The criteria for positive signals are as follows: (1) for ROR, the standard is a ⩾3 and 95% CI >1; (2) for PRR, the standard is a ⩾3 and 95% CI >1; (3) for BCPNN, the standard is E(IC) > 0; and (4) for MGPS, the standard is empirical Bayesian geometric mean lower 95% CI for the posterior distribution (EBGM05) > 2 and a > 0. In our study, the adverse events selected as positive signals needed to meet the criteria of the above four methods, indicating a potential correlation between drugs and events. And further analyze the drug usage time of positive signal drugs causing drug-related adverse events, comparing the differences in time of onset for different drugs. The calculation of drug-induced time originates from the initial drug intake time of the same subject and the time of reporting adverse drug reactions other than drug product-related adverse events. Therefore, the drug-induced time is the difference between the two aforementioned time points. Statistical analysis was conducted using SPSS (version 26.0; IBM, Armonk, NY, USA), GraphPad Prism (version 10.1.2; GraphPad Software, San Diego, CA, USA), Microsoft Excel 2019 software (Microsoft, Redmond, WA, USA), and R (version 4.2.2; R Foundation for Statistical Computing, Vienna, Austria), where p < 0.05 was considered statistically significant. In the R data analysis process, we utilized major packages including ggplot2 (version 3.4.4), ggrepel (version 0.9.4), dplyr (version 1.1.4), and DescTools (version 0.99.52).
Four-grid table of disproportionality analysis method.

Principle of disproportionality measure and standard of signal detection.

BCPNN, Bayesian confidence propagation neural network; CI, confidence interval; IC, information component; MGPS, multi-item gamma Poisson shrinker; PRR, proportional reported ratio; ROR, reporting odds ratio.
Results
Subject information
The subjects using the four FP receptor agonists for glaucoma treatment are predominantly female, constituting 59.2% in latanoprost, 75.2% in bimatoprost, 60.3% in travoprost, and 61% in tafluprost. The age distribution of users for all four drugs is concentrated between 65 and 85 years. With the exception of tafluprost, reports for the other three drugs are predominantly sourced from physicians. The countries contributing the most reports for all four drugs are the United States and Japan. Regarding subject outcomes, the information is primarily concentrated on “Other serious (important medical event)” and “Required intervention to prevent permanent impairment/damage.” For further details, refer to Table 3.
Demographic information of prostaglandin medications for treating glaucoma.
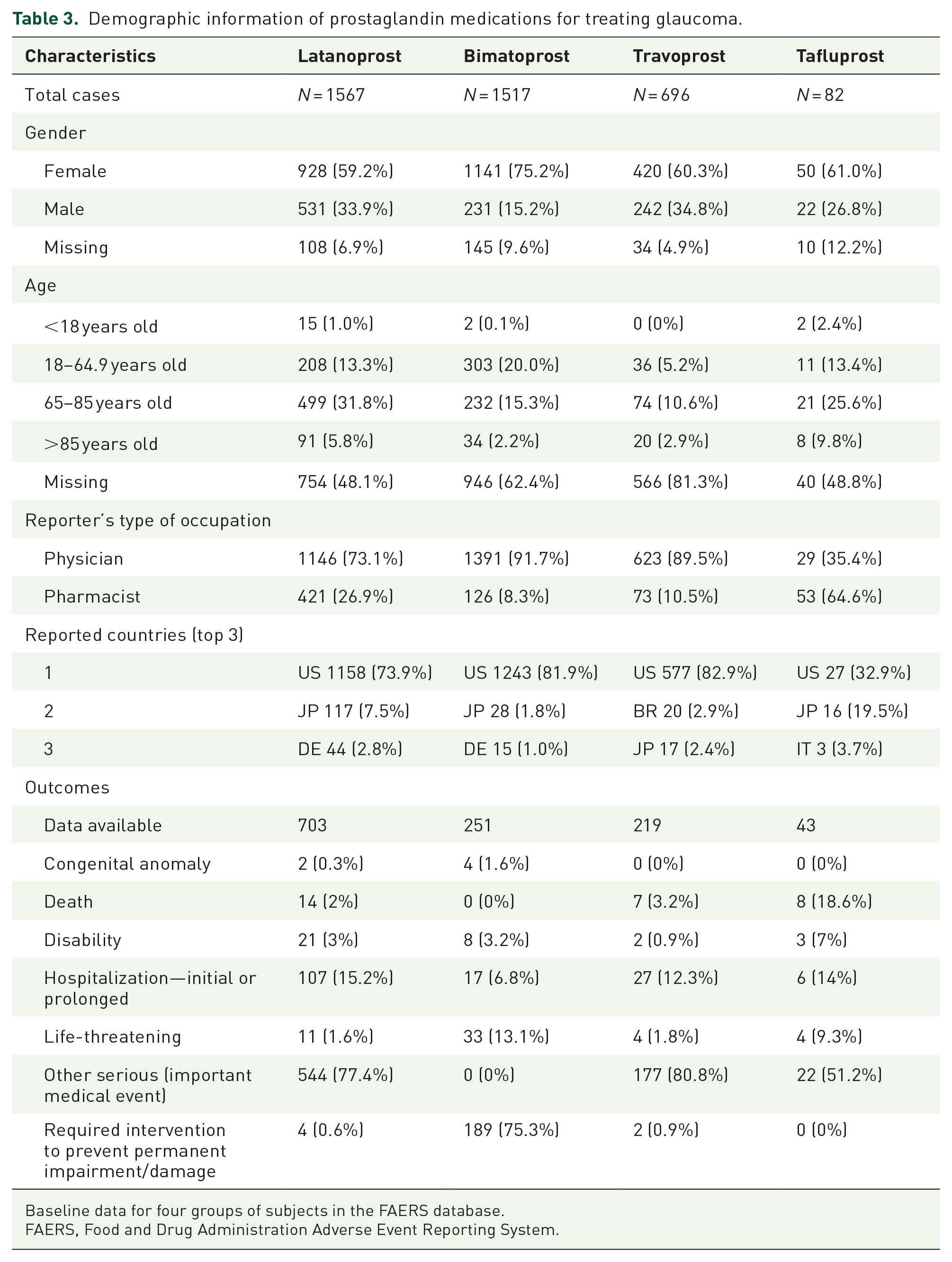
Baseline data for four groups of subjects in the FAERS database.
FAERS, Food and Drug Administration Adverse Event Reporting System.
System organ class report analysis
Adverse events associated with the four FP receptor agonists for glaucoma treatment primarily focus on SOC categories, namely eye disorders, general disorders and administration site conditions, and injury, poisoning, and procedural complications (Figure 2). Notably, tafluprost exhibits a relatively higher frequency of reports of nervous system disorders (Figure 3). The ROR analysis of latanoprost reveals a higher likelihood of inducing eye disorders (ROR, 95% CI = 28.53 (26.69–30.49)), product issues (ROR, 95% CI = 7.01 (6.26–7.85)), immune system disorders (ROR, 95% CI = 2.01 (1.65–2.46)), ear and labyrinth disorders, and injury (ROR, 95% CI = 1.78 (1.2–2.64)), poisoning, and procedural complications (ROR, 95% CI = 1.27 (1.15–1.4)). Bimatoprost may lead to eye disorders (ROR, 95% CI = 43.65 (40.84–46.67)), product issues (ROR, 95%CI = 2.82 (2.36–3.37)), skin and subcutaneous tissue disorders (ROR, 95% CI = 1.91 (1.7–2.14)), and immune system disorders (ROR, 95% CI = 1.87 (1.51–2.32)). Travoprost is associated with eye disorders (ROR, 95% CI = 31.37 (28.44–34.6)), product issues (ROR, 95% CI = 8.09 (6.89–9.5)), ear and labyrinth disorders (ROR, 95% CI = 5.33 (3.77–7.52)), and injury, poisoning, and procedural complications (ROR, 95% CI = 1.17 (1–1.36)). Tafluprost may cause eye disorders (ROR, 95% CI = 59.42 (43.81–80.6)) and surgical and medical procedures (ROR, 95% CI = 2.57 (1.05–6.25); Figures 4 and 5).

Distribution of system organ class induced by latanoprost and bimatoprost for glaucoma treatment.

Distribution of system organ class induced by travoprost and tafluprost for glaucoma treatment.

Forest plot of system organ class induced by latanoprost and bimatoprost for glaucoma.

Forest plot of system organ class induced by travoprost and tafluprost for glaucoma.
Preferred term reports analysis
For the four different FP receptor agonists used in glaucoma treatment, we conducted a PRR analysis for PTs. The top three PTs associated with latanoprost are iris hyperpigmentation (ROR, 95% CI = 535.05 (209.15–1368.77)), ocular pemphigoid (ROR, 95% CI = 436.37 (131.7–1445.91)), and conjunctival erosion (ROR, 95% CI = 213.91 (66.73–685.66)). Bimatoprost is linked to corneal endothelial cell loss (ROR, 95% CI = 5264.51 (2744.86–10097.09)), periorbital fat atrophy (ROR, 95% CI = 2992.8 (669.56–13,377.18)), and blepharal pigmentation (ROR, 95% CI = 1642.09 (1064.76–2532.47)). Travoprost is associated with corneal irritation (ROR, 95% CI = 2222.66 (619.53–7974.17)), growth of eyelashes (ROR, 95% CI = 593.59 (301.24–1169.67)), and eye allergy (ROR, 95% CI = 270.2 (133.38–547.34)). Tafluprost leads to the growth of eyelashes (ROR, 95% CI = 1974.37 (622.14–6265.69)), ocular hyperemia (ROR, 95% CI = 204.08 (125.06–333.04)), and eye irritation (ROR, 95% CI = 150.71 (85.51–265.6)) as the top three PTs. For further details, refer to Figures 6 and 7, as well as Table 4.

Forest plot and heatmap (top 30) of preferred terms induced by latanoprost and bimatoprost for glaucoma.
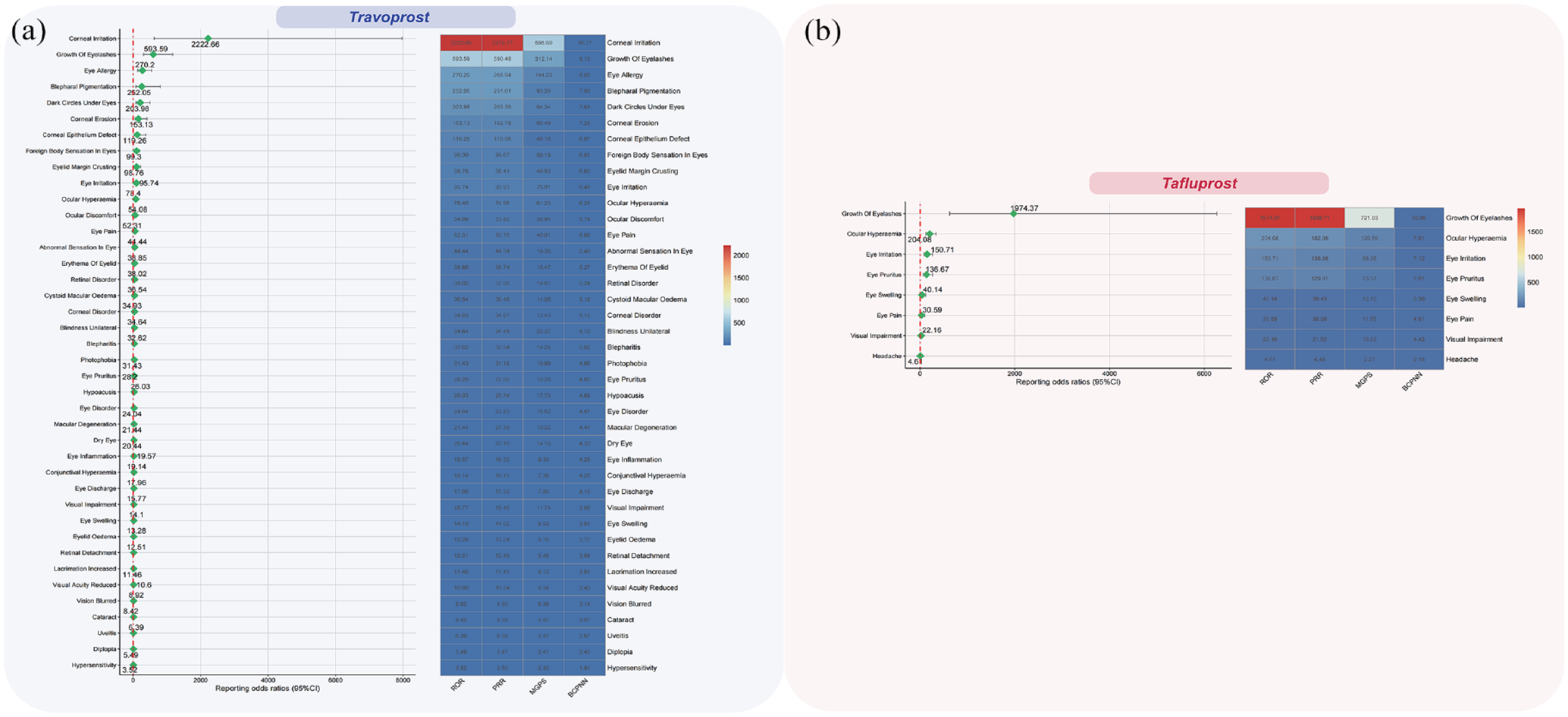
Forest plot and heatmap of preferred terms induced by travoprost and tafluprost for glaucoma.
Statistical values of adverse event reports for prostaglandin medications in the treatment of glaucoma.

The p value is obtained through a chi-square test.
BCPNN, Bayesian confidence propagation neural network; CI, confidence interval; MGPS, multi-item gamma Poisson shrinker; PT, preferred term; PRR, proportional reported ratio; ROR, reporting odds ratio; SmPC, summary of product characteristic.
Drug-induced time analysis
Due to the limited number of subjects (n = 17) with complete records of drug usage and reporting of adverse events for tafluprost, there is a significant difference in sample size compared to the other three groups. To mitigate the bias caused by this significant difference in sample size in the comparison of drug-induced time, 31 we primarily analyzed the comparison of drug-induced time among the other three groups. The mean drug-induced time in the latanoprost group was 344.37 days, while in the bimatoprost group, it was 155.65 days, showing a significant difference between the two groups (p < 0.001). Cumulative risk curve results among the three groups also indicated that the bimatoprost group had a higher risk of drug adverse events compared to the other groups under the same drug usage time (p < 0.001). Further details can be seen in Figure 8.

Comparison of time to induce adverse events for latanoprost, bimatoprost, and travoprost.
Discussion
Prostaglandin drugs, as frontline medications for treating glaucoma, have raised significant concerns regarding their safety. In our current study, we conducted pharmacovigilance analysis using data reported in the FAERS database from the first quarter of 2004 to the third quarter of 2023. Our study is the first to use real-world data from the FAERS database to investigate adverse events associated with four commonly used ophthalmic prostaglandin analogs, observing the relationship between these ophthalmic medications and both ocular and systemic adverse events.
As our understanding of medications deepens, the use of prostaglandin drugs in ophthalmology continues to expand, necessitating recognition of both systemic and local side effects associated with these drugs. It is imperative to acknowledge that after instillation into the conjunctival sac, approximately 80% of eye drops flow into the nasolacrimal duct. Therefore, patients should be reminded to apply digital pressure to the inner canthus after installation to reduce systemic absorption of the eye medication. 32 Categorizing systemic adverse events induced by the four prostaglandin drugs, it is observed that latanoprost can cause labyrinth-related adverse events and immune system diseases, extending beyond ocular adverse effects. Local administration of latanoprost to the round window membrane in the ear can decrease local blood flow to the cochlea and result in hearing impairment. 33 Our team has previously conducted research on the mechanism of dry eye induced by preservative-free latanoprost in mice. The research findings indicate that latanoprost itself triggers inflammatory responses and oxidative stress damage. 34 In addition to ocular adverse events, bimatoprost is shown to induce skin and subcutaneous tissue disorders and immune system disorders. Previous research has indicated that both latanoprost and bimatoprost can induce increased melanin production, leading to periocular skin pigmentation.35,36 However, it is crucial to recognize that the inflammatory reactions induced by some prostaglandin-class drugs may be closely related to the preservative benzalkonium chloride in the medication, rather than the inherent side effects of the drugs themselves. 37 Therefore, the relationship between these adverse events and the prostaglandin analog drugs still requires further analysis through randomized controlled trials to mitigate the limitations inherent in the database due to its spontaneous reporting and the observational nature of the study.
Therefore, based on the potential to induce adverse events in different systems, we conducted a detailed analysis of adverse events using PTs for the four prostaglandin drugs. In the case of latanoprost, there is a risk of adverse events such as iris hyperpigmentation, ocular pemphigoid, and conjunctival erosion. Latanoprost, while treating glaucoma, tends to induce iris pigmentation, especially in eyes with green-brown, yellow-brown, and blue-gray-brown colors, with a decreasing incidence of adverse events in that order. 38 Patients receiving 3–6 months of latanoprost treatment had increased iris pigmentation, as confirmed for the first time in Phase III clinical trials. Moreover, iris color changes were found to be permanent in nearly two-thirds of patients, with no reversal of color observed after treatment cessation. Some patients may also use latanoprost as a medication to promote eyelash growth. 39 This aligns with the results of our real-world prostaglandin drug vigilance study. Furthermore, the relationship between glaucoma medications and the development of pseudo-pemphigoid in the eye has been described. Drug vigilance study results based on the FAERS database show a close association between the use of local ocular beta-blockers and pseudo-pemphigoid development. 40 In addition, the prostaglandin analog drugs used in the treatment of glaucoma in this study also pose a higher risk of inducing pseudo-pemphigoid, a rare adverse event in the eyes. Regarding the adverse effects of bimatoprost, we have identified a higher risk of corneal endothelial cell loss. Glaucoma patients are predisposed to corneal endothelial damage due to various medical and surgical interventions, as well as the condition itself. 41 At the molecular level, prostaglandin analogs are implicated in the activation of matrix metalloproteinases (MMPs), leading to the degradation of extracellular matrix components in the trabecular meshwork and Schlemm’s canal, primarily collagen. 42 Besides activating MMPs in targeted eye tissues, nontarget tissues such as the cornea also exhibit upregulation of MMPs. Studies have confirmed that the use of prostaglandin analogs results in a decrease in central corneal thickness.43,44 Furthermore, the bimatoprost drug, examined in our study for glaucoma treatment, carries a significant risk of inducing loss of corneal epithelial cells. Additionally, prolonged use of bimatoprost can lead to atrophy of the periorbital fat tissue, a notable adverse event characterized by periorbital yellowing, deepening of the upper eyelid sulcus, retraction of the eyelids, and enophthalmos. 45 Compared to other prostaglandin analogs such as latanoprost, tafluprost, and unoprostone, bimatoprost has a higher frequency of reported adverse effects, 46 consistent with the results of our study involving these four prostaglandin analog drugs. Similar adverse events related to ocular pigmentation, including the risk of iris and eyelid pigmentation, are consistent across the other three prostaglandin analog drugs. Additionally, we observed that the drug travoprost also carries risks of corneal irritation, eyelash growth, and eye allergy. However, the risk of corneal irritation with travoprost is higher compared to other prostaglandin analog drugs, possibly due to its relatively higher concentration of the preservative benzalkonium chloride (0.015%). 47 The induction of eyelash growth by prostaglandin analog drugs has been confirmed in numerous studies. 48 Adverse events associated with tafluprost include eyelash growth, ocular congestion, and ocular irritation.
Finally, we also assessed the temporal differences in the induction of ocular adverse events among the four prostaglandin analog drugs. Latanoprost has a longer time to induce adverse events compared to bimatoprost and tafluprost. Therefore, in clinical practice, closer observation of adverse events should be maintained for drugs with shorter induction times. Timely optimization and adjustment of prostaglandin analog drug treatment for glaucoma should prioritize a careful consideration of the risks and benefits associated with the observed adverse events. However, it is also important to recognize that there are many missing values in the FAERS database concerning the time from drug use to the reporting of adverse events, and these reporting times are all spontaneous. As a result, it is impossible to capture the drug-induced time for all adverse events, which may lead to an overestimation or underestimation of the drug-induced time for these medications. Nonetheless, the current data results can provide potential preliminary data support for future clinical research.
Limitations
In this study, there are still certain limitations. Firstly, disproportionality analysis, a statistical method for identifying correlations between targeted drugs and adverse drug events, fails to establish a clear causal link between them. It also does not account for confounding factors such as age, gender, nationality, race, underlying diseases, and concurrent medications. Secondly, the FAERS database relies on spontaneous and voluntary reporting. This may lead to biases influenced by recent studies or media coverage. 49 Thirdly, the FAERS database cannot specifically quantify the risk of each adverse event; it can only use disproportionality analysis to identify “potential complications of adverse events.” Fourthly, despite the study’s relatively large population, external validation using data from other databases is recommended. Finally, the clinical application of FP receptor agonists in combination with other drugs has a broad population base. In the further studies, we will compare the changes in adverse drug reactions between combined therapy and the use of FP receptor agonists alone.
Conclusion
This study represents the first attempt to evaluate the adverse events induced by prostaglandin analog drugs for glaucoma based on real-world data, thereby contributing to the establishment of pharmacovigilance for the four prostaglandin analog drugs. Additionally, we confirmed the variability in the time to induce adverse events for each drug. Finally, this study provides additional reports on some rare and significant adverse events of the aforementioned drug post-marketing. Due to the limitations of the FAERS spontaneous reporting system, we recommend that further clinical studies be conducted in future drug safety evaluations to establish the mechanism and correlation between prostaglandin analog drugs and adverse reactions.
Supplemental Material
sj-docx-1-taw-10.1177_20420986241285929 – Supplemental material for Adverse events of topical ocular prostaglandin medications for glaucoma treatment: a pharmacovigilance study based on the FAERS database
Supplemental material, sj-docx-1-taw-10.1177_20420986241285929 for Adverse events of topical ocular prostaglandin medications for glaucoma treatment: a pharmacovigilance study based on the FAERS database by Shi-Nan Wu, Caihong Huang, Yu-Qian Wang, Xiang Li, Si-Qi Zhang, Xiao-Dong Chen, Dan-Yi Qin, Linfangzi Zhu, Jia-Yi Wen, Na-Chuan Luo, Jiaoyue Hu and Zuguo Liu in Therapeutic Advances in Drug Safety
Footnotes
Acknowledgements
None.
Declarations
Supplemental material
Supplemental material for this article is available online.
Abbreviations
FAERS Food and Drug Administration Adverse Event Reporting System
IOP Intraocular pressure
ROR Reporting odds ratio
PRR Proportional reported ratio
BCPNN Bayesian confidence propagation neural network
MGPS Multi-item gamma Poisson shrinker
PT Preferred term
HLGT High-level group term
SOC System organ class
SMQ Standardized MedDRA queries
DEMO Demographic and administrative information
DRUG Drug information
INDI Indications for use
REAC Adverse events
OUTC Patient outcomes
RPSR Report sources
THER Start and end dates of reported drug use
MMP Matrix metalloproteinase
FP Prostaglandin F
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
