Abstract
Purpose:
This study was designed to investigate the prophylactic effect of oral olanzapine in postoperative nausea and vomiting after gynecologic laparoscopic surgery.
Methods:
ASA I–II, aged 18–75 years, planned to undergo gynecologic laparoscopic surgery with general anesthesia in adult female patients. Using the randomized numbers table, the patients were placed in two groups. Oral olanzapine 5 mg or placebo was given 1 h before anesthesia. All patients received standard antiemetic prophylaxis with dexamethasone and granisetron. The primary outcome was nausea and/or vomiting in the 24 h after the postoperative.
Results:
A total of 250 patients were randomized, and 241 were analyzed. The primary outcome occurred in 10 of 120 patients (8.3%) in the olanzapine group and 23 of 121 patients (19.2%) in the placebo group (p = 0.014). According to Kaplan–Meier analysis, the probabilities of nausea and/or vomiting in the 24 h after the postoperative in the olanzapine group were lower than in the placebo group (log-rank p = 0.014). In a multivariate Cox analysis, the variables of use of olanzapine [hazard ratio (HR): 0.35, 95% confidence interval (CI): 0.16–0.79; p = 0.012] and use of vasoactive drugs (HR: 2.48, 95% CI: 1.07–5.75; p = 0.034) were independently associated with nausea and/or vomiting in the 24 h after the postoperative.
Conclusion:
Our data suggest that olanzapine relative to placebo decreased the risk of nausea and/or vomiting in the 24 h after gynecologic laparoscopic surgery.
Trial registration:
The trial was registered prior to patient enrollment at The Chinese Clinical Trial Registry (https://www.chictr.org.cn/showproj.html?proj=166900, link to registry page, Principal investigator: Nanjin Chen, Date of registration: 25 April 2022).
Plain language summary
Introduction
Postoperative nausea and vomiting (PONV) is a common and distressing complication that can occur after general anesthesia. 1 Even with the use of propofol and antiemetic drugs, up to 45% of patients may experience PONV within 24 h of surgery. 2 Laparoscopic techniques have become more popular due to their advantages such as reduced pain, faster recovery, and shorter hospital stays compared to laparotomy.3,4 However, studies have shown that patients who undergo laparoscopic surgery under general anesthesia are particularly prone to PONV. This is because the introduction of gas into the abdomen during laparoscopy can stimulate the intestines, leading to the release of serotonin and activation of 5-HT3 receptors.5,6 Gynecologic laparoscopic surgery has been identified as an independent risk factor for PONV.7,8 Patients who have multiple risk factors for PONV may have an increased likelihood of experiencing these symptoms, with studies reporting rates ranging from 10% to 80%. 6 These complications can greatly impact patient satisfaction, prolong hospital stays, and increase costs.7,8 Unfortunately, there is still a lack of magic bullets to alleviate this problem.
Today, the prevention of PONV involves the use of various antiemetic medications. These include sections, which are antagonists of the 5-HT3 receptor, NK1 antagonists, glucocorticoids like dexamethasone, and antidopaminergic agents such as droperidol, which are D2 antagonists. 7 Olanzapine, as a second-generation thiophene benzodiazepine, is one of the traditional treatment drugs for mental disorders.9,10 It acts as a multi-receptor antagonist, blocking receptors for dopamine, serotonin, epinephrine, histamine, muscarine, and more. 11 Due to its extensive pharmacodynamic characteristics, olanzapine has a wide range of indications and a better adverse reaction profile than traditional antipsychotics. Coincidentally, the above receptors seem to be closely related to the occurrence of nausea and vomiting, and multiple clinical studies have indeed confirmed that olanzapine can effectively relieve nausea and vomiting caused by advanced malignant tumors 12 and chemotherapy drugs.13,14 Olanzapine is therefore recommended as a first-line prophylactic agent for chemotherapy-induced nausea and vomiting.15,16 Nevertheless, it is still uncertain whether olanzapine is efficacious for sudden nausea and vomiting linked to gynecologic laparoscopic surgery. Hence, the main aim of this study was to assess the prophylactic impact of oral olanzapine on nausea and vomiting following gynecological laparoscopic surgery.
Materials and methods
Study design
This study is a single-center, double-blind, randomized, parallel-group, placebo-controlled trial. The trial was registered prior to patient enrollment at The Chinese Clinical Trial Registry (https://www.chictr.org.cn/showproj.html?proj=166900, link to registry page, Principal investigator: Nanjin Chen, Date of registration: 25 April 2022). The research was carried out following the guidelines of the Helsinki Declaration (revised in 2013). Consecutive patients who had gynecologic laparoscopic surgery at the Taizhou Hospital of Zhejiang Province, from 1 June 2022 to 30 December 2022, were included in this study. Inclusion criteria: Patients undergoing gynecological laparoscopic surgery under general anesthesia in our hospital, ASA I–II grade, age 18–75 years old, with at least two risk factors for PONV were selected. The following exclusion criteria were applied: (i) patients with mental illness or history of mental illness; (ii) history of chemotherapy; (iii) a history of vestibular disorder or dizziness or nausea or vomiting within 24 h before surgery, or receiving conventional antiemetic therapy, including systemic corticosteroids; (iv) pregnant or breast-feeding patients; (v) allergic to olanzapine; (vi) myocardial infarction or unstable angina 6 months before planned surgery; (vii) history of severe arrhythmias (e.g. ventricular tachycardia or ventricular fibrillation); (viii) second New York Heart Association (NYHA) of heart failure grade II or higher; (ix) experiencing postural hypotension or vasovagal syncope half a year prior to the scheduled surgery; and (x) inability to swallow medications.
Randomization and blinding
In accordance with pharmaceutical manufacturing specifications and clinical practice codes, the Taizhou Hospital of Zhejiang Province’s pharmacy was in charge of the preparation, randomization, and blinding of the study drug. Using the randomized numbers table, the patients were placed in two groups. During the study, the pharmacy compounded olanzapine tablets and placebo tablets into blinded capsules that matched each other. During a preoperative office visit, a study investigator registered every participant, and an investigator received a single capsule from a pharmacy on the day of the surgery. Throughout the enrollment period, participants, investigators, anesthesiologists, and surgeons were blinded to the treatment group.
Study treatments
Within an hour of entering the operating room, medication for the study was given in the preoperative holding area. Upon entering the operating room, patients immediately underwent standard monitoring, which encompassed noninvasive measurement of arterial blood pressure, electrocardiography, pulse oximetry, and capnography. Before the administration of anesthesia, a total of 500 ml of a lactated Ringer’s solution was intravenously infused. General anesthesia was induced using propofol 1.5–2.5 mg/kg, sufentanil (0.3–0.4 μg/kg) (according to ideal body weight), and neuromuscular blockade was achieved with rocuronium 0.6 mg/kg. Endotracheal intubation or supraglottic airway is required at the discretion of the attending anesthesiologist. Regardless of the method of airway management, general anesthesia was maintained during the procedure with sevoflurane in oxygen and air (FiO2 0.6) to keep the State Entropy value between 40 and 60. Propofol and remifentanil were used to maintain anesthesia, and if needed, intermittent doses of muscle relaxants would be used to maintain adequate muscle relaxation throughout the surgery. Sufentanil analgesia was administered according to the anesthesiologist’s judgment of the operation and circulatory fluctuations. A dose of dexamethasone 5 mg was administered immediately after anesthetic induction, and a dose of granisetron 3 mg was administered approximately 30 min before emergence from anesthetic. The nasopharyngeal temperature was monitored throughout the surgery and maintained at 36°C–37°C using a warming pad. Patients who had undergone tracheal intubation received neostigmine 0.4–0.7 mg/kg before tracheal extubation to reverse the neuromuscular blockade caused by the intubation. Extubation was done after spontaneous ventilation of the patient became adequate. After surgery, PCA consisting of sufentanil 2 μg/kg and 0.9% saline was administered intravenously for 48 h (0.5 ml bolus dose, 2 ml basal rate, and 15-min lockout). 17
Outcomes
Ramsay Sedation Score and Visual Analogue Scale (VAS) scores were used to measure postoperative pain and sedation levels. One hour (at the post-anesthesia care unit), 2 h, and 24 h (on the floor) after surgery, we assessed the incidence of PONV, use of rescue antiemetic, Ramsay Sedation Score, and VAS pain scale. From 0 to 10, nausea severity was evaluated verbally using a numerical rating scale. During the questionnaire, patients verbally rated their perceived nausea, with 0 representing no nausea and 10 representing the worst possible nausea. All vomiting events were recorded. Vomiting or retching is considered vomiting. If two or more vomiting episodes or nausea persist for more than 10 min, 10 mg of metoclopramide will be administered as a rescue antiemetic. Complete response is defined as the absence of PONV within the first 24 h after anesthesia and the absence of rescue antiemetic medication. Record any side effects reported by the patient during the study period.
Nausea and/or vomiting within 24 h after undergoing laparoscopic surgery for gynecological purposes is the main result. Additional outcomes comprised the prevalence of intense postoperative queasiness (defined as any numerical assessment scale score exceeding 3 for queasiness within a 24-h period following laparoscopic surgery for gynecological purposes), emesis, and adverse reactions.
Statistical analysis
We aimed to detect an absolute between-group difference of 8.6 percentage points in the incidence of PONV. We assumed that the rate of PONV would occur in 17% of the patients in the placebo group and 8.4% of those in the olanzapine group. Therefore, we estimated that a total sample of 248 patients would need to be enrolled for the trial to have 95% power at an overall two-sided α level of 0.05. Sample size calculations were performed using G*power 3.1.9.7 (Germany). An analysis of descriptive statistics was conducted to summarize the baseline demographic and clinical characteristics of patients. The data are expressed as mean, standard deviation, median, and interquartile range. The Shapiro–Wilk test was used to assess normality (p value >0.05). Student’s t-test or the corrected t-test was used to compare groups of continuous variables that followed a normal distribution. The Mann–Whitney U test was utilized to conduct comparisons of different variables. The χ2 test or Fisher’s test was utilized to perform comparisons of categorical variables. Kaplan–Meier curves and the log-rank test were performed to estimate the incidence of PONV. The value of the variables was assessed using a univariate Cox proportional hazards regression adjusted model. p Values <0.05 were considered significant. All statistical analyses were performed with SPSS 26.0 (USA) and Graphpad Prism 8.0 (USA).
Results
Participant and baseline characteristics
Figure 1 shows the distribution and randomization of the study patients. Of 271 patients, 250 patients who underwent gynecologic laparoscopic surgery were included based on the inclusion and exclusion criteria. Nine patients, including four in the Olanzapine group and five in the Placebo group, were excluded after the study drug administration due to a transition from laparoscopic surgery to open surgery. Finally, 121 patients in the olanzapine group and 120 patients in the placebo group completed the study and were evaluated for all study outcomes.

Patients who underwent randomization started the study and were included in the primary analysis.
Demographic and baseline characteristics of 241 patients are presented in Table 1. There were no significant differences in age, Body Mass Index (BMI), American Society of Anesthesiologists (ASA) grading, and other basic information between the olanzapine group and the placebo group. The prevalence of underlying diseases (such as hypertension, diabetes, and chronic liver disease) was basically the same between the two groups. In addition, there was no significant difference in the Apfel risk scores between the two groups, which considered postoperative use of opioid analgesics, non-smoking, and history of PONV or motion sickness.
Baseline characteristics of 241 patients.

T0: before induction of anesthesia. Apfel score was based on four risk factors for nausea and vomiting: being female, postoperative opioid use, non-smoking status, a history of PONV, and/or motion sickness. The incidence of PONV was 10%, 20%, 40%, 60%, and 80%, respectively, when 0, 1, 2, 3, and 4 risk factors were present.
Student’s t-test, Mann–Whitney U test, χ2 test, and Fisher’s exact test.
DAP, Diastolic arterial pressure; HR, hazard ratio; IQR, interquartile range; SAP, Systolic arterial pressure.
There was a significant difference between the two groups in intraoperative blood loss, with a median blood loss of 30 ml (interquartile range, 20–70) in the olanzapine group and 20 ml (interquartile range, 20–50) in the placebo group, p = 0.018. In other intraoperative management characteristics, such as anesthetic use, intraoperative infusion, and operation duration, there was no difference between the two groups (Table 2).
Intraoperative management characteristics.

Mann–Whitney U test, χ2 test, and Fisher’s exact test.
IQR, interquartile range; PACU, post-anesthesia care unit.
The primary outcome, nausea, and/or vomiting in the 24 h after postoperative occurred in 10 of 120 patients (8.3%) in the olanzapine group and 23 of 121 patients (19.2%) in the placebo group (p = 0.014) (Table 3). There was no difference between the two groups in the incidence of vomiting. However, the incidence of nausea and severe nausea in the olanzapine group was significantly lower than that in the placebo group. There was a difference in received rescue antiemetic between the two groups (3.3% versus 11.7%, p = 0.014).
Clinical outcome.

χ2 test and Fisher’s exact test.
According to the result of the Kaplan–Meier analysis, the probabilities of nausea and/or vomiting in the 24 h after the postoperative in the olanzapine group were lower than in the placebo group (log-rank p = 0.014) (Figure 2). In a multivariate Cox analysis, the variables of use of olanzapine [hazard ratio (HR): 0.35, 95% confidence interval (CI): 0.16–0.79; p = 0.012] and use of vasoactive drugs (HR: 2.48, 95% CI: 1.07–5.75; p = 0.034) were independently associated with nausea and/or vomiting in the 24 h after the postoperative (Table 4).
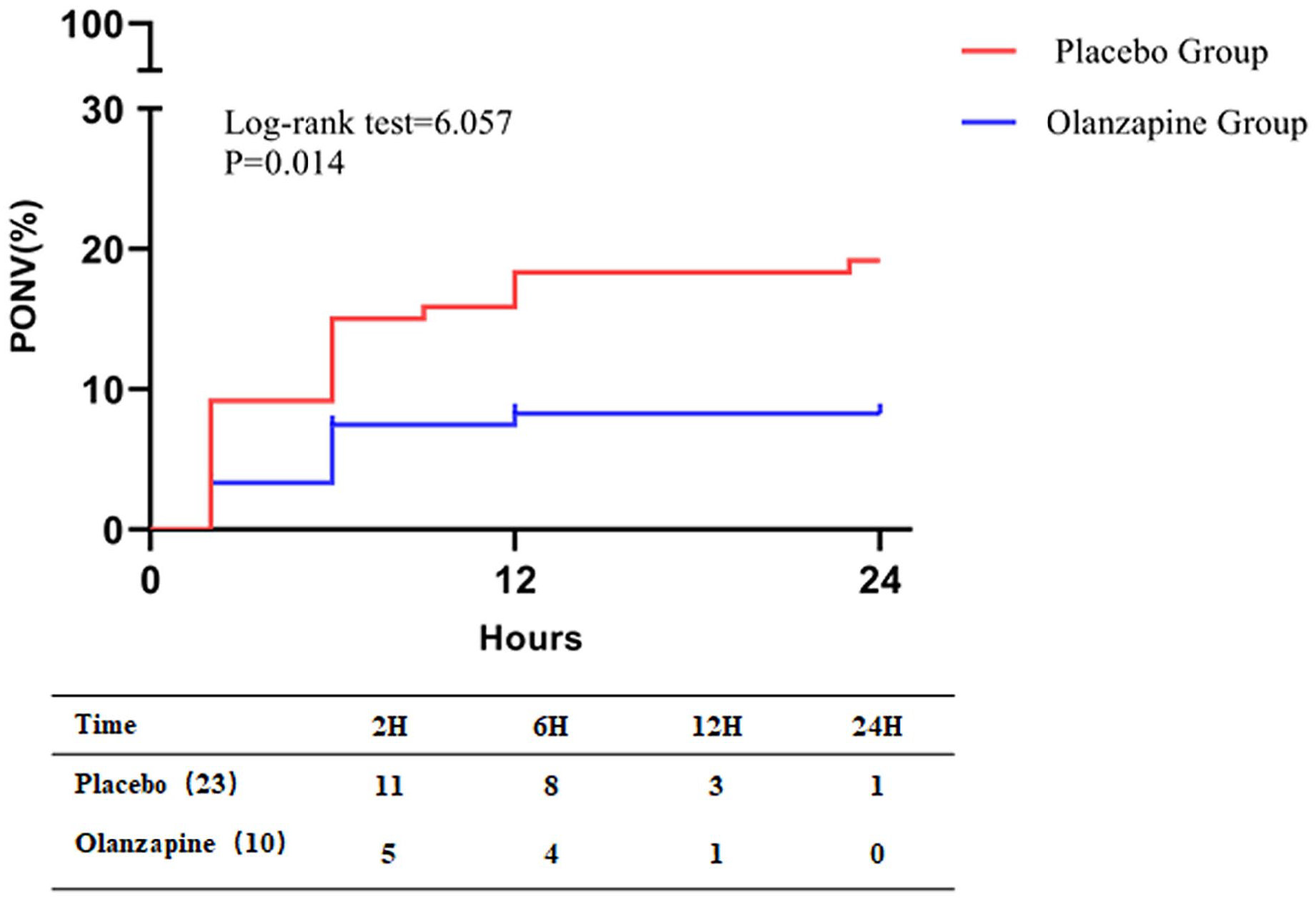
Incidence of PONV during 24 h after surgery in both groups.
Univariate and multivariate analyses for 24-h PONV in the overall patient population.

HR, hazard ratio; PONV, postoperative nausea and vomiting.
Discussion
As far as we know, this clinical study is the initial attempt to prove the preventive impact of olanzapine on acute nausea and vomiting following gynecological laparoscopic surgery. The study design involves randomization, double-blinding, and placebo control. During this experiment, it was discovered that the inclusion of olanzapine, as opposed to a placebo, in patients undergoing gynecological laparoscopic surgery with general anesthesia and receiving a two-drug combination for PONV prevention, resulted in a significant reduction in the occurrence of nausea and/or vomiting within 24 h after the surgery, decreasing from 19.2% to 8.3%. This result is consistent with a previous study which implies that olanzapine can alleviate the occurrence of nausea and vomiting after outpatient surgery. 18
The pathophysiological mechanism of nausea and vomiting is itself very complex and is not fully understood. 19 Mechanical and drug effects can stimulate the release of neurotransmitters in the central nervous system, which then act on the receptors in the vomiting center, posterior brain region, and isolated fascicular nucleus, and eventually cause various physiological reactions to vomiting in patients. 20 Hypotension is one of the causes of nausea and vomiting, 20 which explains the association between intraoperative vasoactive drug use and PONV in our results. The influencing factors of operation-related nausea and vomiting are more complex, and the risk and degree of PONV can be affected by patient, surgical, and anesthetic factors.21–23 The most predictive characteristics associated with increased risk of PONV have been evaluated and found to be female, young (<50 years old), non-smoking status, postoperative opioid use, history of motion sickness, and/or PONV. 20 This also indicates that the occurrence of PONV in gynecological laparoscopic surgery patients is more difficult to intervene than in other surgical patients. The findings of this study offer a new solution to this conundrum.
The antiemetic effect of olanzapine was first discovered in the palliative care of cancer patients. 24 Many clinical trials have also confirmed the benefits of olanzapine in controlling nausea and vomiting.11,25,26 The mechanism of olanzapine’s antiemetic effect is related to its antagonism against various neurotransmitter receptors, such as serotonergic (5-HT) 2a, 2c (5-HT2c), 3 (5-HT3), and 6 (5-HT6) receptors; dopamine D1, D2, D3, and D4 receptors; catecholamine alpha1 adrenergic receptors, histamine H1 receptors, and acetylcholine muscarinic receptors.25,27 The above receptors related to specific neurotransmitters may be involved in the occurrence of nausea and vomiting, and can play an effective antiemetic role when blocked by olanzapine, especially 5-HT2c, 5-HT3, and dopaminergic D2. The blocking effect of olanzapine on many of these receptors also makes it a single broad-spectrum antiemetic agent for a variety of nonspecific causes of nausea and vomiting.
Existing studies have shown that the most common side effects of olanzapine are drowsiness and dizziness but these adverse reactions are not serious. 28 These adverse reactions were not observed in this study, which may also related to the low dose of olanzapine we used. Clinical studies by Hashimoto et al. have confirmed that 5 mg olanzapine combined with aprepitant, palonosetron, and dexamethasone can not only effectively treat nausea and vomiting in patients with cisplatin chemotherapy but also have higher safety than 10 mg olanzapine in drowsiness.11,29 In olanzapine-related studies, no significant extrapyramidal reactions have been reported, 15 which also confirmed that olanzapine has a high enough safety in preventing and treating refractory nausea and vomiting. More importantly, the occurrence of nausea and vomiting after gynecological laparoscopic surgery is usually caused by multiple factors and may require multi-drug combination therapy, which increases the risk of drug interaction, which is perfectly addressed by the fact that olanzapine can act on a large number of receptors.
Several limitations should be noted in this study. One limitation of this study is the small sample size of the single-center study, which included only a small number of female patients undergoing gynecological laparoscopic surgery, which may affect the accuracy of the results. Therefore, we will study the effect of olanzapine on perioperative nausea and vomiting in a multicenter with more subjects undergoing other procedures. Second, the use of only a single dose of 5 mg is also one of the limitations of this study. We will refer to the pharmacokinetic characteristics of olanzapine and further explore the optimal dosage and duration of olanzapine for the prevention of PONV in subsequent studies.
Conclusion
The results of this study support that oral administration of 5 mg olanzapine before surgery could significantly reduce the risk of nausea and/or vomiting after gynecological laparoscopic surgery, and no obvious adverse reactions were observed. These results suggest that olanzapine may be a potentially useful option for preventing postoperative nausea and vomiting, particularly in individuals at high risk for these complications.
Supplemental Material
sj-xlsx-1-taw-10.1177_20420986241244593 – Supplemental material for Olanzapine for the prevention of postoperative nausea and vomiting after gynecologic laparoscopic surgery: a randomized controlled trial
Supplemental material, sj-xlsx-1-taw-10.1177_20420986241244593 for Olanzapine for the prevention of postoperative nausea and vomiting after gynecologic laparoscopic surgery: a randomized controlled trial by Nanjin Chen, Shuman Ji, Junfei Liu, Liping Wang, Fenglin Chen, Yanwu Zhu, Jiao Li, Minjuan Chen, Lingyang Chen, Mingcang Wang, Ruyi He, Xiaopeng Mei, Zhanqin Zhang, Shengwei Jin, Jingming Zheng and Yongpo Jiang in Therapeutic Advances in Drug Safety
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
