Abstract
Background:
Candesartan cilexetil is a widely used angiotensin II receptor blocker with minimal adverse effects and high tolerability for the treatment of hypertension. Candesartan is administered orally as the prodrug candesartan cilexetil, which is wholly and swiftly converted to the active metabolite candesartan by carboxylesterase during absorption in the intestinal tract. In populations with renal or hepatic impairment, candesartan’s pharmacokinetic (PK) behavior may be altered, necessitating dosage adjustments.
Objectives:
This study was conducted to examine how the physiologically based PK (PBPK) model characterizes the PKs of candesartan in adult and geriatric populations and to predict the PKs of candesartan in elderly populations with renal and hepatic impairment.
Design:
After developing PBPK models using the reported physicochemical properties of candesartan and clinical data, these models were validated using data from clinical investigations involving various dose ranges.
Methods:
Comparing predicted and observed blood concentration data and PK parameters was used to assess the fit performance of the models.
Results:
Doses should be reduced to approximately 94% of Chinese healthy adults for the Chinese healthy elderly population; approximately 92%, 68%, and 64% of that of the Chinese healthy adult dose in elderly populations with mild, moderate, and severe renal impairment, respectively; and approximately 72%, 71%, and 52% of that of the Chinese healthy adult dose in elderly populations with Child–Pugh-A, Child–Pugh-B, and Child–Pugh-C hepatic impairment, respectively.
Conclusion:
The results suggest that the PBPK model of candesartan can be utilized to optimize dosage regimens for special populations.
Plain language summary
Background:
Candesartan cilexetil is a widely used angiotensin II receptor blocker with minimal adverse effects and high tolerability for the treatment of hypertension. Candesartan cilexetil is wholly and swiftly converted to the active metabolite candesartan by carboxylesterase during absorption in the intestinal tract. Candesartan’s pharmacokinetic behavior may be altered in patients with renal or hepatic impairment.
Methods:
We developed PBPK models using the reported physicochemical properties of candesartan and clinical data. We validated the PBPK models.
Results:
We found that the elderly population needs dosage adjustments.
1. Doses should be reduced to approximately 94% of Chinese healthy adults for the Chinese healthy elderly population
2. Doses should be reduced to approximately 92%, 68%, and 64% of that of the Chinese healthy adult dose in elderly populations with mild, moderate, and severe renal impairment
3. Doses should be reduced to approximately 72%, 71%, and 52% of that of the Chinese healthy adult dose in elderly populations with Child Pugh-A, Child Pugh-B, and Child Pugh-C hepatic impairment.
Conclusion:
The PBPK model of candesartan can be utilized to optimize dosage regimens for special populations.
Keywords
Introduction
Hypertension is one of the most significant cardiovascular disease risk factors. Antihypertensive medications are required to manage hypertension to reduce blood pressure (BP) to target levels. According to their action mechanisms, antihypertensive medications are divided into five classes: beta-blockers, diuretics, calcium channel blockers, angiotensin-converting enzyme inhibitors, and angiotensin receptor blockers.1,2 Candesartan is administered orally as the prodrug candesartan cilexetil, rapidly and wholly hydrolyzed by carboxylesterase during its absorption in the intestinal tract to the active metabolite candesartan.3,4 Candesartan has minimal adverse effects and excellent tolerability. It can be used alone or with other antihypertensive medications. It is appropriate for all stages of hypertension, particularly in patients who cannot tolerate angiotensin-converting enzyme inhibitors, and has become one of the most preferred antihypertensive medications.5,6 The estimated bioavailability for candesartan is 15%. Because this metabolite is firmly bound to plasma proteins, its distribution volume (0.13 L/kg) is relatively low. 7 Candesartan is only partially inactivated by hepatic metabolism (CYP2C9) and is predominantly eliminated unchanged through the urinary and biliary systems. Several clinical pharmacokinetic (PK) studies imply that dose modification may be necessary for specific situations, such as renal/hepatic impairment and elderly populations. 8
Elderly populations are prone to high BP in general.9,10 Due to aging and complex underlying diseases treated by multi-drug therapy, the pathophysiological mechanisms and PKs of elderly populations are complex, and they face a substantially increased risk of high BP.3,11 The balance between the achievement of goal BP and the reduced risk of side effects has become a key concern for clinicians.12–14 Few randomized controlled studies have evaluated geriatric high BP patients of varying ages, disease stages, and degrees of susceptibility, particularly in the Chinese elderly population. It is essential to reduce the safety risk and maximize the efficacy-to-safety ratio of candesartan in geriatric patients. To maintain the same level of efficacy and safety in elderly adults as in non-elderly adults, it is necessary to forecast the PK profile in virtual Chinese elderly populations.
A physiologically based PK (PBPK) model considers the physiological and biochemical properties of organisms and the physicochemical, anatomical, and thermodynamic properties of a drug.15–17 This model simulates drug distribution, transportation, and metabolism in various body regions by treating human tissues and organs as independent compartments linked by blood circulation. To predict the PK and efficacy of drugs in humans, the PBPK model combines the physical and chemical properties of drugs, the parameters of the human physiological system, and the mechanical PK data.18–21 As such, it can be used to process medical dynamics data based on the material balance principle. 22 Moreover, PBPK is commonly used to characterize PK changes in the body under various complex clinical conditions and, according to previous studies, is an effective method to study the distribution and metabolism of drugs in the human body. 23 In this study, a PBPK model was developed and validated for extrapolation to the healthy elderly Chinese population and the elderly Chinese population with hepatic and renal impairment to serve as a guide for devising individualized medication regimens for these populations.
Methods
Modeling platform and data collection
The whole-body PBPK (WB-PBPK) models of candesartan in European and Chinese healthy adults, the healthy elderly population, and the healthy elderly population with hepatic and renal impairment were developed using the PK-Sim® software (Open Systems Pharmacology Suite, version 11.1). The WebPlotDigitizer® (Ankit Rohatgi, version 4.2) software was used to derive the PK data points from the PK study reported in the literature. PK parameters were computed utilizing noncompartmental model analysis with Phoenix WinNonlin® software (version 8.3.5.340; Pharsight, Mountain View, CA, USA). The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 24
Generic workflow for model development
Figure 1 depicts the generic workflow for scaling drug PK from healthy subjects to patients with hepatic and renal impairment and from European adults to Chinese adults and elderly populations using the PBPK model.

Generic workflow for model development.
Adult PBPK model development
This study utilized a combined ‘bottom-up’ and ‘middle-out’ strategy to facilitate model development. Candesartan’s absorption, distribution, metabolism, and excretion mechanism were determined by accumulating data on its physicochemical properties, conducting
Using Monte Carlo simulations, system-specific parameters obtained from the PK-Sim built-in database (i.e. physiological and anatomical parameters of the virtual population) were fixed to suit 4 mg oral tablet clinical data in European healthy subjects. The model was validated using clinical data from other concentrations and healthy Chinese subjects. Table 1 summarizes the physicochemical, biopharmaceutical, and PK parameters of candesartan.
Summary of input compound parameters of candesartan and candesartan cilexetil in the PBPK model.

CES2: Carboxylesterases 2;
PBPK modeling in healthy subjects
Based on the average value obtained from the software, a European virtual adult was created to represent the mean adult of the created population. Based on mean population values, the age, weight, height, and body mass index (BMI) of the individual were 27.00 years, 77.00 kg, 174.20 cm, and 25.37 kg/m2, respectively. At the same time, a virtual population of 100 individuals (50% female subjects) aged 20–40 years with a BMI of 18–24 kg/m2 was constructed using the ‘population’ module of the software to characterize the PK behavior of candesartan in the European healthy population.
The virtual populations were created based on the dosing regimen, and the population prediction means and 5th–95th concentration range were obtained. The average folded error was used to compare predicted and measured concentrations of the maximum concentration (
Scaling to the European patients with renal impairment
The ‘Chronic Kidney Disease’ module built into PK-Sim was used to display changes in European patients with renal impairment, including GFR, kidney volume, kidney blood flow, plasma protein binding, hematocrit, gastric emptying time, and small intestinal transit time.
33
Renal clearance in European patients with renal impairment was predicted based on the


where CLR represents the value of observed renal clearance,
Scaling to the European patients with hepatic impairment
Child–Pugh classification is the most widely used method to categorize the hepatic function in populations with hepatic impairment. Scoring is based on clinical features and laboratory-based parameters, and patients are classified into Child–Pugh-A (CP-A), Child–Pugh-B (CP-B), and Child–Pugh-C (CP-C) groups based on the extent of hepatic impairment.
35
Parameter information is shown in Table 2.
36
The model was validated based on the real-world PK study reported by Liu
Changes in PBPK parameters that are altered in hepatic impaired individuals

CP-A, Child–Pugh-A; CP-B, Child–Pugh-B; CP-C, Child-Pugh-C; PBPK, physiologically based pharmacokinetic.
Chinese elderly population PBPK model development
The scaling of elderly Chinese populations was performed in two steps. In the first step, PK-Sim was used to automatically scale the parameter changes of anatomic, anthropometric, and physiological in the Chinese elderly population based on the final PBPK model of Chinese healthy subjects, keeping drug-specific parameters constant. In the second step, the exposure of candesartan was separately simulated within the subpopulations of the elderly with hepatic and renal impairment based on the prevalence of hepatic and renal dysfunction in this population.
Model predictability and dose optimization
The prediction accuracy was evaluated graphically by comparing the
Result
PK profiles in healthy subjects
The PBPK model was first applied to simulate the concentration–time profiles in healthy European subjects after single doses of candesartan at different dose levels. For single-dose simulation, European healthy subjects in each dose group were given candesartan at 4, 8, and 16 mg. Then, the PBPK model was applied to simulate the concentration–time profiles in healthy Chinese subjects after single doses of candesartan at 16 mg. As shown in Figure 2, the model accurately described the observed PK profile across the regimens investigated. These results indicated a reasonable assumption of candesartan’s absorption and elimination mechanism.
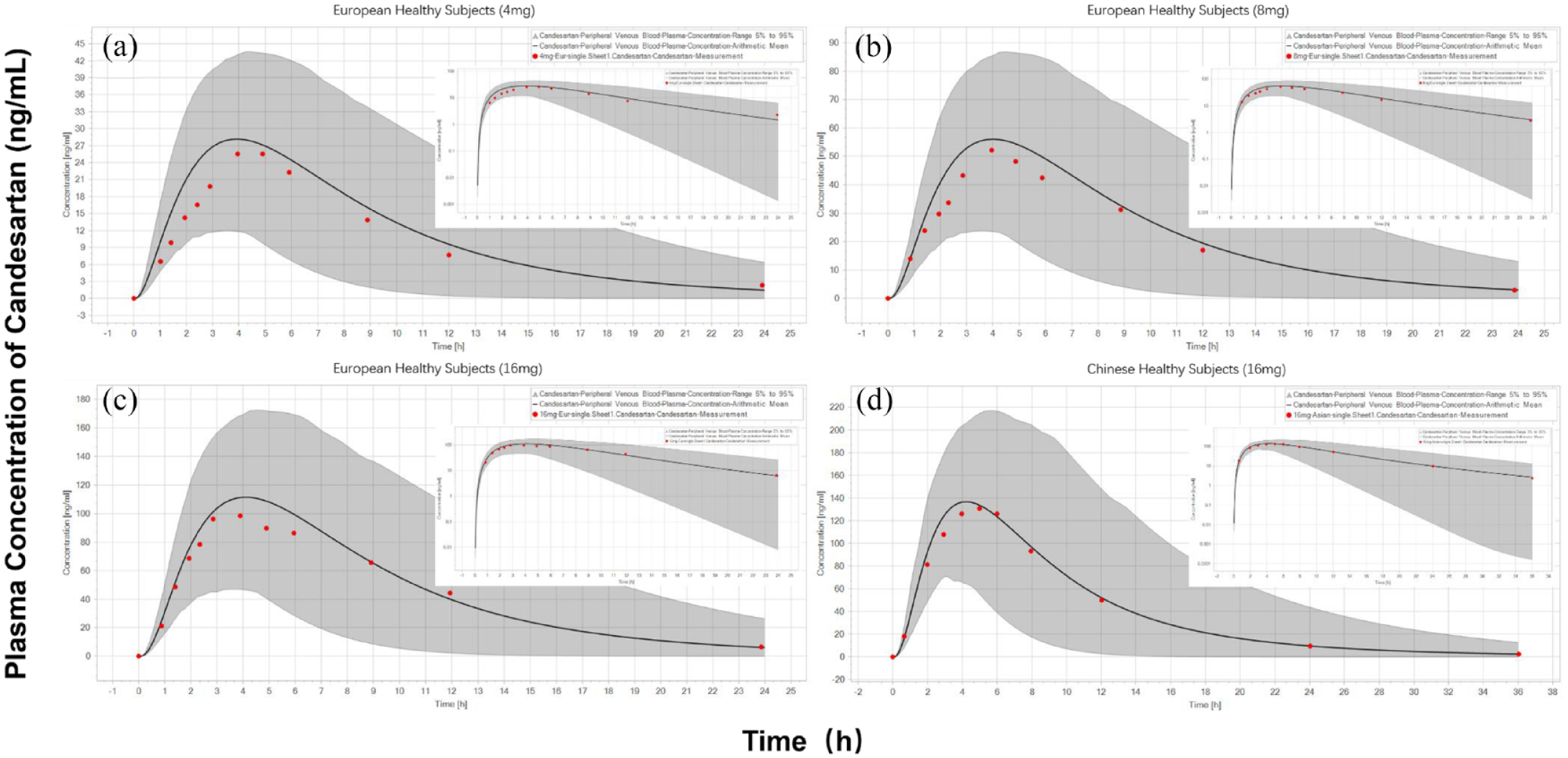
Prediction of the PK profiles for candesartan at a series of doses using the PBPK model. Simulation (mean predictions in black lines and 5th–95th percentiles of predictions in gray shade) of PK profiles for a single oral dose of 4, 8, and 16 mg of candesartan (log scale was on the right top in each dose panel) in European healthy subjects (a–c) and Chinese healthy subjects (d).
PK profiles in European elderly patients with renal impairment
Based on the data from the PK study of candesartan in European elderly patients with varying degrees of renal impairment, the PK characteristics of candesartan in the renal-impairment population were explored. As shown in Figure 3, the extrapolation model results showed that the predicted and observed values of plasma drug concentration–time profiles in the normal European adults, and European elderly patients with moderate, and severe renal-impairment populations fit well, with most observations falling within the 5th–95th range. The effect of renal impairment on fold changes of

Prediction of the pharmacokinetic profiles for candesartan in European patients with normal/mild (a), moderate (b), and severe (c) renal impairments using the PBPK model. Simulations were compared with the corresponding observed clinical data (red dot).
Fold changes of candesartan exposure in patients with renal impairment compared with normal function.
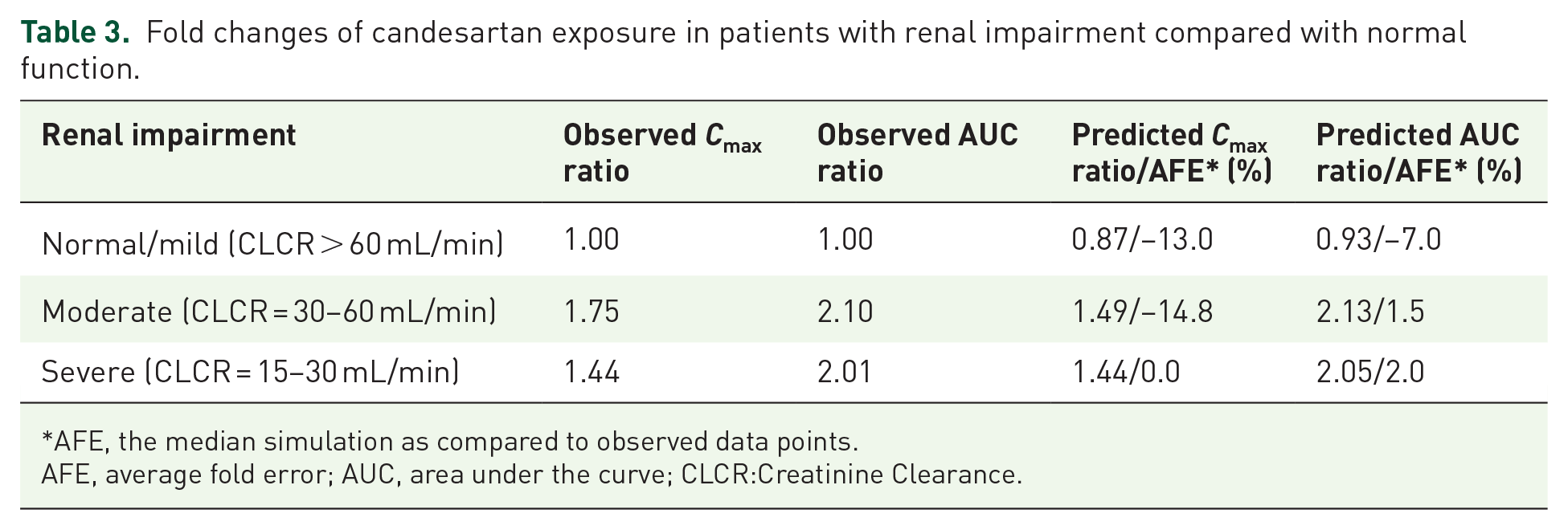
AFE, the median simulation as compared to observed data points.
AFE, average fold error; AUC, area under the curve; CLCR:Creatinine Clearance.
PK profiles in European elderly patients with hepatic impairment
Based on data from a PK study of candesartan in European elderly patients with varying degrees of hepatic impairment, the PK characteristics of candesartan in the hepatic impairment population were further explored. As shown in Figure 4, the extrapolation model results showed that the predicted and observed values of plasma drug concentration–time profiles in European adults, and European elderly patients with CP-A and CP-B hepatic impairment populations fitted well, with most observations falling within the 5th–95th range. On this basis, we predicted the plasma drug concentration–time curve profiles in CP-C of hepatic impairment populations. The effect of hepatic impairment on fold changes of

Prediction of the pharmacokinetic profiles for candesartan in European patients with normal (a), CP-A (b), CP-B (c), and CP-C (d) hepatic impairments using the PBPK model. Simulations were compared with the corresponding observed clinical data (red dot).
Fold changes of candesartan exposure in patients with hepatic impairment compared with normal function.

AFE, the median simulation as compared to observed data points.
AFE, average fold error, AUC, area under the curve; CP-A, Child–Pugh-A; CP-B, Child–Pugh-B; CP-C, Child-Pugh-C.
PK profiles in Chinese elderly patients
Scaling the age-dependent parameters according to the software’s built-in algorithm, a cohort of Chinese elderly population (50% women) aged 40–80 years. Exposure to 8 mg candesartan administered orally

Comparison of oral 8 mg candesartan plasma drug concentration–time profiles in Chinese healthy subjects and elderly patients at different renal/hepatic disease stages.
Fold changes of predicted candesartan exposure in Chinese elderly patients with renal or hepatic impairment compared with healthy subjects.

AUC, area under the curve; CP-A, Child–Pugh-A; CP-B, Child–Pugh-B; CP-C, Child-Pugh-C.
Discussion
In recent years, there has been an increase in the clinical demand for antihypertensive medications among special patients. An angiotensin II receptor blocker, candesartan cilexetil, is extensively used to treat hypertension and has shown promise in exceptional circumstances such as renal/hepatic impairment and elderly patients. Candesartan has the prospective advantages of being easy to administer, requiring less frequent drug monitoring, and having fewer drug–drug interactions. Due to a lack of clinical data in this patient population, however, off-label candesartan use in these patients must be extrapolated from the dosage regimen in healthy adults.
Traditional compartmental modeling approaches have limited predictive ability because they do not account for all of the physiological, anatomical, and biochemical changes associated with drug exposure, nor all the changes associated with drug absorption, distribution, metabolism, and excretion. By contrast, PBPK modeling utilizes existing drug disposition and physiology knowledge and enables extrapolation across various life stages. In this study, PBPK-based scaling from healthy populations to populations with renal impairment or hepatic impairment and from adults to elderly patients was used to predict the PK profiles of candesartan in populations with renal impairment or hepatic impairment and in elderly populations with or without renal or hepatic impairment. Utilizing age-specific physiology parameters, such as organ volume, blood flow, and hepatic and renal function, the model was then scaled to geriatric populations using adult-specific data.
In comparison to traditional PK methods, PBPK models have a significant impact on the formulation of clinical medication regimens for special populations, primarily by predicting plasma concentrations of drugs and providing an accurate method for assessing efficacy and risk.38,39 PBPK models have been used to predict exposure profiles in special populations
Candesartan cilexetil is an angiotensin II receptor blocker and it is widely used to treat hypertension and heart failure (AstraZeneca 2005). It is a prodrug that is completely converted to the active metabolite candesartan by the CES2 enzyme in the intestinal wall during absorption. During the PBPK modeling, we found that the lipophilicity of candesartan provided by the DrugBank did not truly describe the absorption process of candesartan in the body. Therefore, we optimized the parameters affecting the absorption process of candesartan through published PK literature43–46 and evaluated the accuracy of the PBPK model by comparing the Bias% between the predicted and the measured values of key PK parameters (such as
In terms of substance PKs, both the liver and kidneys play a crucial role. The bioavailability of a drug is significantly influenced by the quantity of drug absorbed and the liver’s first-pass metabolism. As a prodrug, candesartan cilexetil is rapidly and completely converted to candesartan by the CES2 enzyme in the intestinal wall during absorption. For candesartan, it undergoes hepatic metabolism through the CYP2C9 enzyme. Plasma protein binding in humans is >99%. By contrast, the GFR is primarily responsible for the elimination of the drug. Determining drug dosages for populations with hepatic and renal insufficiency has been a significant clinical challenge.
47
Because candesartan is partially excreted through the kidneys, patients with renal impairment may lead to decreases in CYP2C9 activities, which, in turn, affect the metabolism of candesartan.48,49 Candesartan is metabolized by CYP2C9 liver enzymes in the hepatically impaired population, necessitating dose modification. In the current study, all changes
Due to the fragility and susceptibility of the geriatric, clinical drug development has historically favored younger and middle-aged adults. Although those aged 65 and older receive the majority of drug prescriptions, they continue to be underrepresented in clinical trials. 50 Consequently, there is a lack of knowledge regarding the PK and PD (Pharmacodynamics) responses of the elderly, rendering the safety and efficacy of drugs in this population uncertain. 51 To contribute to its elucidation, this study extrapolated the reaction of healthy adults to candesartan exposure to describe the reaction of healthy elderly individuals and elderly individuals with hepatic or renal impairment. The results indicate that elderly populations with hepatic and renal impairment should have their dosages adjusted on a similar scale to the healthy elderly population.
Although the PBPK model developed in this study has a stable structure and excellent predictive ability, several limitations must be considered when evaluating the results. One limitation is that all the data used were extracted using software from published literature. Even though these data were not obtained directly from the researchers, the PK parameters calculated from these extracted data points were comparable to previously reported PK parameters, for which minor errors were inevitable but within acceptable limits. As only the immediate-release formulation of candesartan was considered during the development of the PBPK model, the model only provides a broad explanation of the PKs of candesartan at various oral dosages. Using the findings of this study, however, the model can be further evaluated and optimized in future studies.
Conclusion
The PBPK model developed in this study contributes to a more accurate description of candesartan’s PKs in elderly populations with renal and hepatic impairment. By incorporating changes in pathophysiological factors into the model and accurately extrapolating the model to these populations, this study demonstrated a method for enhancing the predictive capacity of the drug-disease model, thereby making it a valuable resource for future clinical individualized drug administration and evaluation.
Supplemental Material
sj-docx-2-taw-10.1177_20420986231220222 – Supplemental material for Physiologically based pharmacokinetic modeling of candesartan to predict the exposure in hepatic and renal impairment and elderly populations
Supplemental material, sj-docx-2-taw-10.1177_20420986231220222 for Physiologically based pharmacokinetic modeling of candesartan to predict the exposure in hepatic and renal impairment and elderly populations by Lingfeng Guo, Xinyu Zhu, Lei Zhang and Yichao Xu in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-3-taw-10.1177_20420986231220222 – Supplemental material for Physiologically based pharmacokinetic modeling of candesartan to predict the exposure in hepatic and renal impairment and elderly populations
Supplemental material, sj-docx-3-taw-10.1177_20420986231220222 for Physiologically based pharmacokinetic modeling of candesartan to predict the exposure in hepatic and renal impairment and elderly populations by Lingfeng Guo, Xinyu Zhu, Lei Zhang and Yichao Xu in Therapeutic Advances in Drug Safety
Supplemental Material
sj-xlsx-1-taw-10.1177_20420986231220222 – Supplemental material for Physiologically based pharmacokinetic modeling of candesartan to predict the exposure in hepatic and renal impairment and elderly populations
Supplemental material, sj-xlsx-1-taw-10.1177_20420986231220222 for Physiologically based pharmacokinetic modeling of candesartan to predict the exposure in hepatic and renal impairment and elderly populations by Lingfeng Guo, Xinyu Zhu, Lei Zhang and Yichao Xu in Therapeutic Advances in Drug Safety
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
