Abstract
Background:
Outpatient hysteroscopic surgery requires patients to be anaesthetised and recover quickly, and the drugs used must be safe and effective. Remifentanil is typically co-administered with propofol as total intravenous anaesthesia (TIVA) for hysteroscopy because of its favourable pharmacokinetic and pharmacodynamic properties. However, the optimal dose of remifentanil when co-administered with propofol without neuromuscular blocking agents (NMBAs) has not been established.
Objectives:
In this sequential dose-finding study, the 90% effective dose (ED90) of remifentanil effect-site concentration (Ce) combined with propofol without NMBAs during outpatient hysteroscopy was calculated to minimise the side effects of using higher doses.
Design:
This sequential dose-finding study was conducted in August 2022.
Methods:
Forty patients who underwent outpatient hysteroscopy under TIVA were included in the study. With a biased coin up-and-down design, the initial remifentanil Ce was established at 2 ng/mL, and the subsequent remifentanil dosage was determined based on the reaction of the previous patient. The primary outcome was a remifentanil Ce that resulted in successful TIVA by maintaining a Narcotrend index < 60, surgical pleth index (SPI) < 50, and without patient movement throughout hysteroscopy. Secondary outcomes included rates of hypotension-related symptoms and interventions, drug consumption, post-anaesthesia care unit (PACU)-estimated visual analogue scale (VAS) and Ramsay sedation scores, modified Aldrete scores, and other adverse effects of anaesthesia. The ED90 and 95% confidence intervals (CI) were estimated using isotonic regression methods and bootstrapping.
Results:
For TIVA without NMBAs during outpatient hysteroscopy, the ED90 Ce of remifentanil combined with propofol was determined to be 2.75 ng/mL (95% CI, 2.50–3.00 ng/mL). The incidence of peri-operative adverse effects of anaesthesia was relatively low. All the patients had satisfactory VAS, Ramsay sedation, and modified Aldrete scores in the PACU.
Conclusion:
Remifentanil at a Ce of 2.75 ng/mL is recommended for TIVA combined with propofol in outpatient hysteroscopic surgery.
Trial registration:
http://www.chictr.org.cn (ChiCTR2200062284; 31/7/2022).
Plain language summary
Why was the study done? Outpatient hysteroscopy under total intravenous anaesthesia (TIVA) requires the patient to be anaesthetised and recover quickly. Because outpatient surgery does not require hospital admission, the anaesthetic drugs used in the outpatient surgery must be safe and effective. This study explored the effective dose of effect-site concentration (Ce) of remifentanil combined with propofol for outpatient hysteroscopic surgery under TIVA to provide patients with satisfactory anaesthetic effects and minimise the side effects of using higher doses.
What did the researchers do? The research team recruited 40 patients scheduled to undergo selective outpatient hysteroscopy between August 2022 and February 2023. If a satisfactory anaesthetic effect was obtained, the next patient was 1/9-times as likely to receive a lower dose or 8/9-times as likely to receive the same dose of remifentanil as the preceding patient. When a satisfactory anaesthetic effect was not achieved, the remifentanil dosage was increased for the following patient. In addition, a satisfactory anaesthetic effect was determined by monitoring the depth of anaesthesia. Using this method, the optimal dose of remifentanil for TIVE in outpatient hysteroscopic surgery was determined.
What did the researchers find? The effective Ce of remifentanil combined with propofol for TIVA during outpatient hysteroscopy under anaesthesia depth monitoring was 2.75 ng/mL in 90% of the included patients. The incidence of perioperative adverse effects of anaesthesia was relatively low. Accordingly, a small Ce of remifentanil co-administered with propofol can be considered effective and feasible for minimising the adverse events associated with a higher Ce of remifentanil.
What do the findings mean? Based on these findings, a remifentanil Ce of 2.75–3.0 ng/mL with propofol is recommended for TIVA in hysteroscopic surgery.
Introduction
Hysteroscopy has gained popularity in gynaecology with the development of assisted reproductive technology and minimally invasive techniques as an outpatient procedure. 1 Various anaesthesia and pain-relief methods have been described for outpatient hysteroscopy procedures, including paracervical block, sedation, and oral analgesics. However, in mainland China, outpatient hysteroscopic surgery is mainly performed under total intravenous anaesthesia (TIVA). 2 As the number of patients undergoing outpatient surgery increases, several initiatives are being developed and validated to shorten the post-operative recovery time and length of hospital stay. Some side effects of neuromuscular blocking agents (NMBAs), including allergic reactions and residual muscle relaxants, may delay the discharge of patients undergoing outpatient procedures and are associated with patient morbidity. Therefore, various studies have examined the induction and maintenance of anaesthesia without using NMBAs for outpatient surgery to avoid the residual effects of NMBAs. 3 To date, many studies have reported the use of propofol in combination with opioids in clinical anaesthesia without NMBAs.4,5
Remifentanil stands out among the various available opioids because of its quick onset and predictable offset, even after a lengthy infusion duration without accumulation. 6 Therefore, remifentanil is the preferred opioid for TIVA, especially in outpatient surgery. 7 However, if used in high dosage intraoperatively, it increases the risk of post-operative nausea and vomiting, post-operative hyperalgesia, and cardiovascular depression. 8 Therefore, determining the optimal dose of remifentanil in combination with propofol for TIVA without NMBAs during outpatient hysteroscopy is important to minimise post-operative recovery time in a manner that is compatible with the concept of enhanced recovery after surgery (ERAS).
Outpatient surgery is increasingly adopting ERAS protocols and anaesthesia depth monitoring is recommended for patients during surgery. 9 Increasing evidence suggests that inadequate or excessive anaesthesia depth may affect patient outcomes due to improper delivery of anaesthetic drugs. In recent years, various devices for non-invasive continuous intraoperative monitoring of anaesthesia depth and nociception-anti-nociception balance have become available. Narcotrend, an electroencephalogram (EEG) monitor used to assess anaesthetic depth, is now commercially available in Europe and has received Food and Drug Administration clearance in the United States. 10 According to the results of several studies, compared to a standard practice protocol, Narcotrend may help evaluate the hypnotic component of anaesthesia, decrease medication consumption, and shorten recovery periods.11,12 The surgical pleth index (SPI) is a non-invasive tool that estimates intraoperative nociception. 13 The SPI values range from 0 to 100. It can also easily monitor the nociception–anti-nociception balance during general anaesthesia by using pulse oximetry. 14 Photo-plethysmography from pulse oximetry data and pulse plethysmography, which correlates with the balance of the autonomic nervous system, are used to calculate the SPI. 15 A previous study has reported that the SPI is correlated with the effect-site concentration of remifentanil and surgical stimulation. 14 Furthermore, the requirement for opioids was dramatically decreased in a prior trial involving 170 outpatients when the remifentanil dosage was adjusted according to the SPI. 16
Therefore, this study aimed to determine the optimal dosage of remifentanil in combination with propofol for TIVA without NMBAs during outpatient hysteroscopic surgery using Narcotrend and SPI monitoring.
Materials and Methods
Patient Recruitment
This study was conducted in accordance with the principles of the Declaration of Helsinki. On 18 September 2021, the China Ethics Committee of Registering Clinical Trials at the West Hospital affiliated with Sichuan University approved this prospective, biased coin up-and-down (BCUD)-based, sequential, dose-finding study (Chairperson Taixiang Wu; Ethical No. ChiECRCT20210385). This study was registered at http://www.chictr.org.cn (ChiCTR2200062284; 31/7/2022). The reporting of this study conforms to the CONSORT statement. All methods were performed in accordance with relevant guidelines and regulations. 17 In this study, 48 women aged 18–50 years with an American Society of Anesthesiologists (ASA) physical status of I or II who were scheduled for selective outpatient hysteroscopy at International Peace Maternity and Child Health Hospital between August 2022 and February 2023 were included. All the participants provided informed consent. The exclusion criteria were as follows: (1) inability to provide written informed consent, (2) allergy to the intended medication, (3) history of cardiovascular, lung, renal, or brain illness, (4) history of chronic pain, (5) history of alcohol or drug abuse, and (6) body mass index <18 or >30 kg/m2.
TIVA protocol
All patients followed the standard protocol and fasted for at least 8 h before surgery. After entering the operating room, left upper limb venous access was obtained and lactated Ringer’s solution was infused at a rate of 10 mL/min to avoid the bias of intravenous drugs. Non-invasive blood pressure, arterial oxygen saturation, electrocardiogram, and Narcotrend monitoring were all performed as standard practices once the patients were in the operating room. An index finger pulse oximeter sensor on the operating side was used to track SPI (S5 monitor; GE Healthcare, Helsinki, Finland). A MAP of <65 mmHg was considered hypotension. A 50 mg rescue dose of phenylephrine was administered as the first sign of hypotension. Bradycardia was defined as a heart rate (HR) of <50 beats per minute. A rescue dose of atropine (0.5 mg) was administered whenever bradycardia was observed. Patients were placed in the lithotomy position for hysteroscopic surgery before anaesthesia. Remifentanil and 1% propofol were used for the induction and maintenance of anaesthesia; however, no NMBAs were used. Remifentanil and propofol were not pre-mixed. Remifentanil was diluted in 50 mL 0.9% saline to a concentration of 20 μg/mL. Remifentanil diluted in 0.9% saline was used as an isotonic solution. Remifentanil and propofol were administered via a two-channel target-controlled infusion (TCI) pump for intravenous injections (CP-730TCI; Inc., Beijing SLGO, China). Under the Marsh pharmacokinetic model, the recommended induction effect-site concentration (Ce) of propofol is 6.0 μg/mL (initial infusion rate = 600 mL/h). Remifentanil was administered intravenously at a predetermined Ce in the Minto pharmacokinetic model 5 min after propofol administration (initial infusion rate = 600 mL/h). Manually assisted respiration was used to maintain the end-tidal carbon dioxide (EtCO2) between 35 and 45 mmHg. When the patient lost consciousness and the Narcotrend index decreased below 40, a laryngeal mask airway (LMA) was placed. The patient underwent mechanical ventilation with a tidal volume of 8 mL/kg, 60% oxygen concentration, a respiration rate (RR) of 12 times/min, and an adjusted RR to maintain an EtCO2 between 35 and 45 mmHg. After laryngeal mask airway (LMA) intubation, the propofol maintenance target concentration was set to 4 µg/L according to the guidelines for the safe practice of TIVA. 18 After surgery, patients were administered 50 mg of flurbiprofen axetil for analgesia. After removing the LMA, the patients were transferred to the post-anaesthesia care unit (PACU) and monitored for 60 min. The patients who met the discharge criteria were discharged from the PACU. Anaesthesia was induced by a specialist anaesthesiologist who was aware of the exact drug dosages. The patients and the remaining anaesthesiologists were blinded to the administered dosage.
Visual analogue scale (VAS) scores were used to evaluate pain intensity, with zero indicating no pain and 10 representing the worst pain imaginable. The Ramsay sedation score was used to evaluate the quality of recovery from anaesthesia as follows: restless and agitated = 1 point, cooperative and tranquil = 2 points, sleepy, can follow instructions = 3 points, brisk response to optical and tactile stimuli = 4 points, sleep state, response to strong stimuli but slow reaction = 5 points, and no response from deep sleep = 6 points. Discharge readiness was assessed using the modified Aldrete score (Table 1).
Modified Aldrete score.

SpO2, Peripheral oxygen saturation.
Biased coin up-and-down (BCUD) design of the study
Using a BCUD method, the Ce of remifentanil required for outpatient hysteroscopic surgery in 90% of patients (90% effective dose [ED90]) was estimated. In this study, the concentrations and step size of the dosage of remifentanil were selected according to the guidelines for the safe practice of TIVA. 18 A remifentanil Ce of 2.0 ng/mL was administered to the first patient. An unsatisfactory anaesthetic effect was characterised by the presence of any kind of bodily movement, a frown, failed LMA insertion, a Narcotrend index >60, an SPI > 50, or a sudden increase in the SPI of >10 points. If a satisfactory anaesthetic effect was observed, the Ce of remifentanil was considered suitable. Therefore, the next patient was 1/9 times as likely to receive a lower dose (decreased by 0.5 ng/mL) or 8/9 times as likely to receive the same dose as the preceding patient. The following patient was administered a dosage increased by 0.5 ng/mL when a satisfactory anaesthetic effect was not observed. The Ce levels of remifentanil ranged from 2.0 ng/mL to 6.0 ng/mL. Dose assignment was implemented using the BCUD scheme prepared by a statistician in Microsoft Excel 2016; only the specialist anaesthesiologist had access to this information, ensuring the double-blind nature of this study.
Outcomes
The primary outcome was the satisfactory anaesthetic effects of outpatient hysteroscopic surgery. An unsatisfactory anaesthetic effect has been defined in the previous text. Therefore, a satisfactory anaesthetic effect was characterised as no body movement, successful LMA insertion, Narcotrend index between 20 and 60, and SPI between 20 and 50. Secondary outcomes included the incidence of bradycardia and hypotension, chest wall rigidity, duration of anaesthesia and surgery, induction dose of propofol and remifentanil, total infusion dose of propofol and remifentanil, recovery time, extubation time, level of sedation after surgery (Ramsay sedation scores), PACU-estimated VAS scores, PACU-related modified Aldrete score, time to discharge, and adverse effects such as intraoperative awareness, emergence agitation, delayed awakening, shivering, post-operative nausea, and vomiting. The baseline characteristics of the patients, such as height, weight, age, baseline MAP, and baseline HR, were recorded. All primary and secondary outcomes were observed by anaesthesiologists who were blinded to the dose assignments and stored in a secure electronic database using Excel 2010 (Microsoft Corp.).
Statistical analysis
According to simulation studies and the BCUD design, a sample size of 20–40 patients may offer stable estimates of the target dose for the most realistic cases, which was the basis for this dose-finding investigation. 19 Forty-eight patients were included in this analysis.
The ED90 was defined as the Ce of remifentanil required for outpatient hysteroscopic surgery associated with a 90% anaesthetic success rate and was estimated using the isotonic regression method. The 95% confidence interval (CI) was calculated using the bias-corrected percentile method with 2000 bootstrap replicates. Isotonic regression and bootstrapping analyses were performed using R statistical software (version 3.4.4, Vienna, Austria). The Shapiro-Wilk test was used to check whether continuous variables were normally distributed. Normally distributed continuous variables are presented as mean (standard deviation), and if the distribution is not normal, variables are presented as median (interquartile range) or numbers (percentages). Statistical analyses were performed using GraphPad Prism 8 (GraphPad Software, San Diego, California, USA) and SPSS version 18.0 (SPSS Inc., Chicago, IL, USA) for Windows. Statistical significance was set at p < 0.05.
Results
The patient recruitment data are shown in Figure 1. Forty-eight patients who underwent outpatient hysteroscopic surgery with TIVA were enrolled. Four patients were excluded because of a high body mass index (n = 2), history of chronic pain (n = 1), or refusal to participate (n = 1). Two patients were given neuromuscular blocking medications, one patient had difficult intubation with an LMA, and one patient got the erroneous dosage of remifentanil since the research assistant prepared a remifentanil dose of 50 μg/mL instead of 20 μg/mL, all dropped out of the study. After applying the inclusion and exclusion criteria, 40 patients were included in this study. Table 2 shows patient characteristics. The duration of surgery and anaesthesia were 25.55 ± 4.96 min and 15.80 ± 5.00 min, respectively.

Diagram of the study. A total of 48 patients who underwent outpatient hysteroscopic surgery were included in the study. Three patients did not meet the inclusion criteria and one patient declined to participate and was excluded from the study. LMA intubation failed in one patient, one patient received the wrong dose of remifentanil, and two patients received neuromuscular blocking agents; 40 patients completed the protocol.
Demographic and surgical characteristics of the patients.

Data are presented as mean (SD) unless otherwise indicated.
HR, heart rate; MAP, mean arterial pressure; PACU, post-anaesthesia care unit.
ED90 of remifentanil
Figure 2 shows the sequence of effective and ineffective responses to remifentanil dose during TIVA administration in 40 consecutive patients. Remifentanil was administered at Ce levels of 2.0, 2.5, 3.0, and 3.5 ng/mL, but never at Ce levels of 4.0–6.0 ng/mL. The observed and pooled adjacent-violators algorithm (PAVA)-adjusted response rates for each remifentanil dose are presented in Table 3. The remifentanil Ce ED90 value in outpatient hysteroscopic surgery under TIVA was 2.75 ng/mL (95% CI, 2.5–3.37 ng/mL) as estimated by the isotonic regression approaches. The primary outcome was reached in the majority of the included women at a Ce of remifentanil of 3.0 ng/mL; anaesthesia failed in two of the 20 patients.
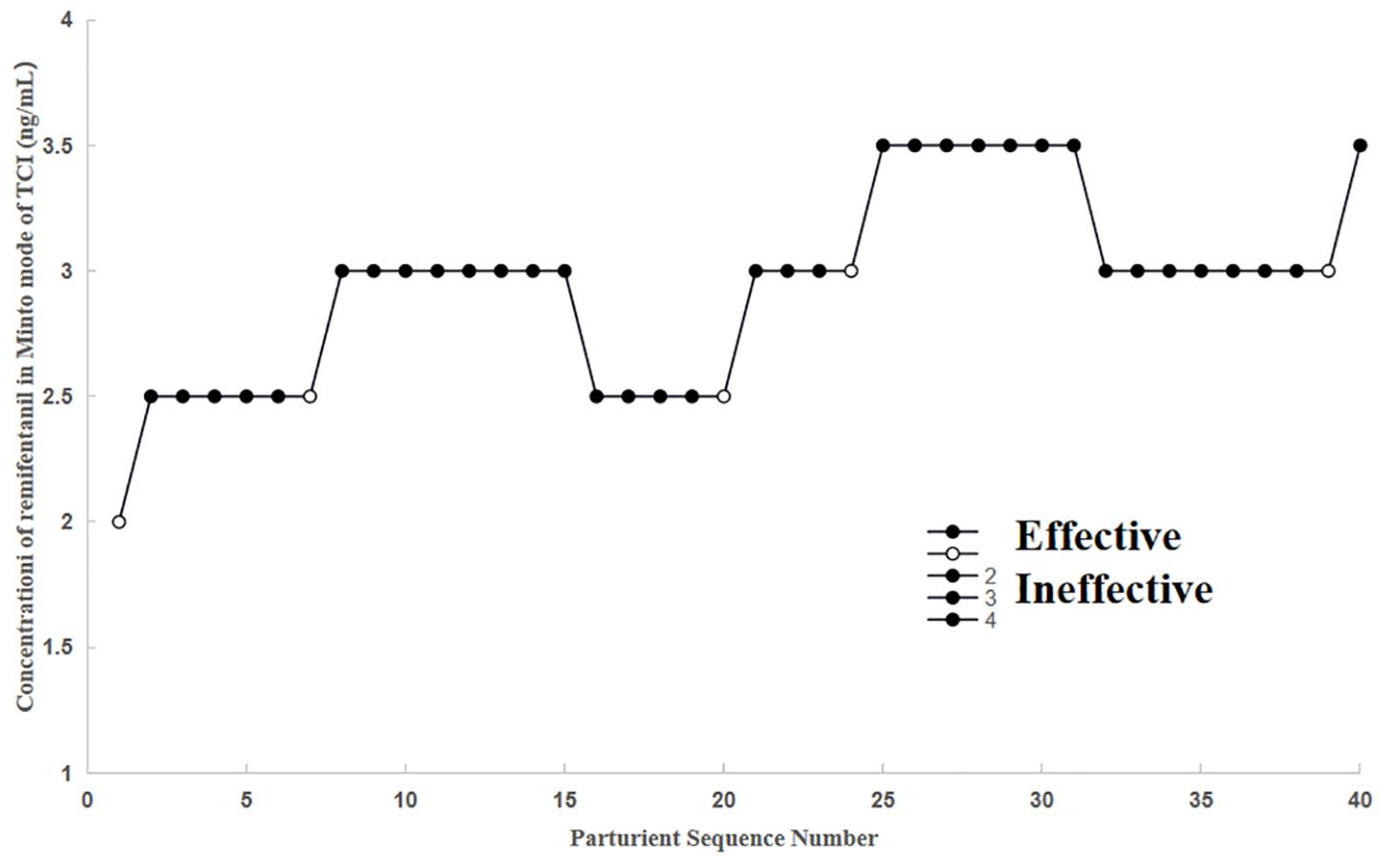
The patient allocation sequence and response to the assigned dose of the study. The patient sequence number (x-axis) is the order of patient exposures using the biased coin up-and-down design. The assigned dose levels are presented on the y-axis.
Observed and PAVA-adjusted response rates.

PAVA-adjusted response rates were estimated using the weighted isotonic regression method.
PAVA, pooled-adjacent-violators algorithm.
Intraoperative outcomes
Table 4 presents the intraoperative patient outcomes. Rates of 7.5% for hypotension, 7.5% for bradycardia, 12.5% for body movement, and 2.5% for chest wall rigidity were recorded, whereas hypertension and tachycardia were not observed. The incidence of ephedrine use after anaesthesia was 7.5%. The initial doses of propofol and remifentanil were 85.96 ± 9.32 mg and 44.18 ± 6.13 μg, respectively. The total infusion doses of propofol and remifentanil were 286.15 ± 21.88 mg and 239.25 ± 18.86 μg, respectively. The recovery time and extubation times were 7.53 ± 1.01 min and 7.70 ± 1.02 min, respectively. The mean (standard deviations [SDs]) MAP, HR, SPI, and Narcotrend index at baseline (T1); 10 s before intubation (T2); after intubation (T3); cervical dilatation (T4); operation start (T5); 5 (T6), 10 (T7), 15 (T8), and 20 (T9) min after surgery; and at extubation (T10) are presented in Figure 3.
Peri-operative outcomes.
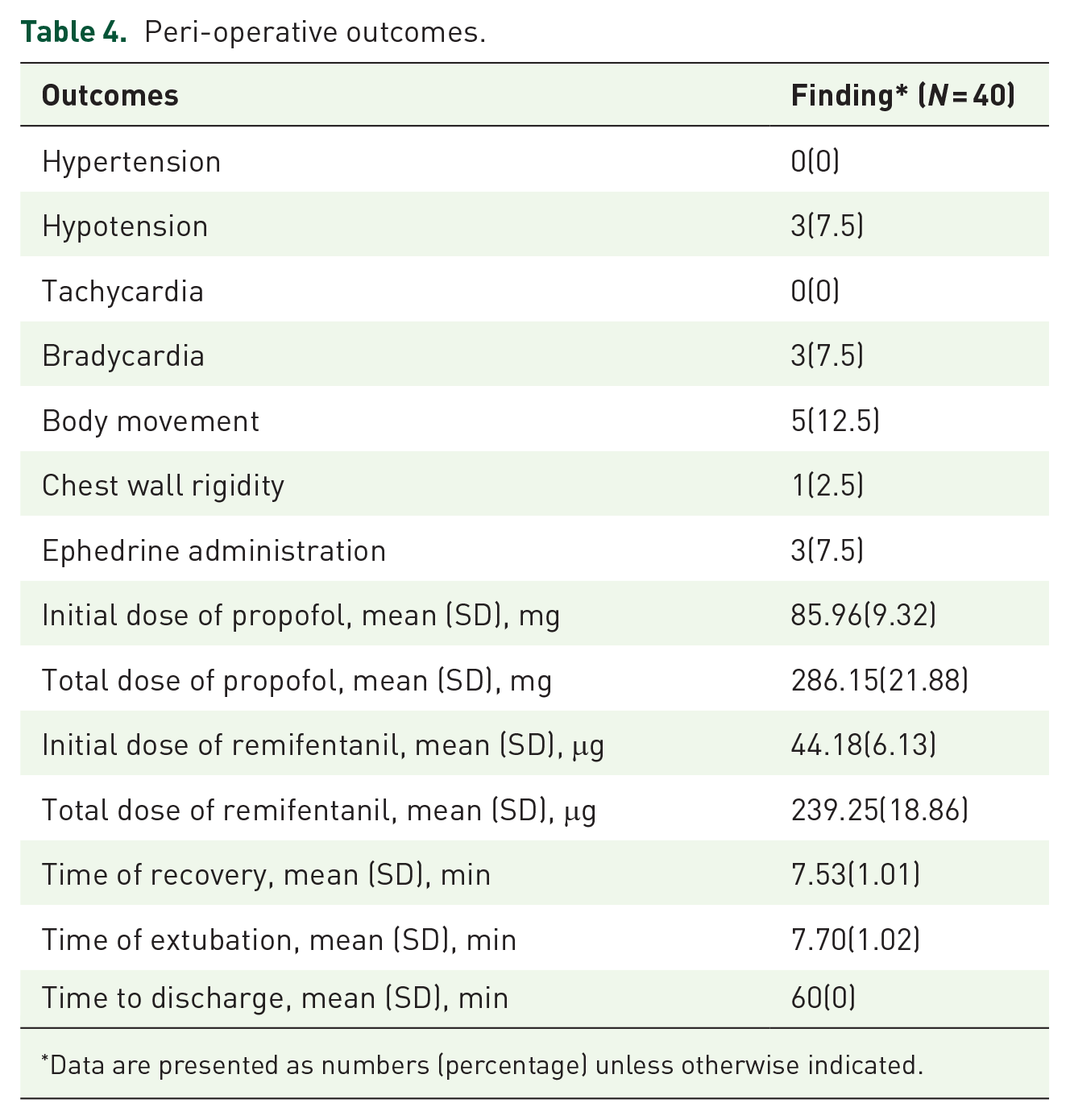
Data are presented as numbers (percentage) unless otherwise indicated.

The heart rate (HR), mean arterial pressure (MAP), surgical pleth index (SPI) index and Narcotrend index at baseline (T1); 10 s before intubation (T2); after intubation (T3); cervical dilatation (T4); operation start (T5); 5 (T6), 10 (T7), 15 (T8), and 20 (T9) min after operation; and extubation (T10). The solid diagrams represent the mean number of each observation, and the bars represent their standard deviation (SD).
Post-operative outcomes
Table 5 presents all postoperative study outcomes. All patients had satisfactory VAS and Ramsay sedation scores in the PACU. Only two patients required rescue analgesics in the PACU. Other adverse symptoms such as intraoperative awareness, emergence agitation, delayed awakening, shivering, postoperative nausea, and vomiting were not observed. The modified Aldrete score was used to determine whether a patient could be discharged from the PACU. Furthermore, a modified Aldrete score of ⩾9 suggested that the patient could be discharged from the hospital. In this study, all the patients had modified Aldrete scores⩾9 at 10, 20, 30, and 40 min in the PACU. After 60 min of monitoring, all patients were discharged directly from the PACU.
Post-operative outcomes.

Data are presented as number (percentage) unless otherwise indicated.
IQR, interquartile range.
Discussion
In this BCUD dose-finding study, the ED90 Ce of remifentanil in combination with propofol for TIVA without NMBAs during outpatient hysteroscopy under Narcotrend and SPI monitoring was 2.75 ng/mL (95% CI, 2.5–3.37 ng/mL) in 90% of the included patients. Accordingly, such a small Ce of remifentanil co-administered with propofol can be considered effective and feasible for minimising the adverse events associated with a higher Ce of remifentanil. Based on these findings, a remifentanil Ce of 2.75–3.0 ng/mL with propofol is recommended for TIVA in hysteroscopic surgery.
Clinical anaesthesia requires precise dosing; therefore, identifying the ED90 prior to determining the dosage of drugs to be administered is crucial. One of the most popular approaches to determining the effective dose/concentration of drugs is the BCUD sequential method. 20 The BCUD sequential method has several benefits, including minimized sample size, use of concentrated test doses, and efficiency of implementation. 21 It is a trustworthy strategy for determining optimal dosages in clinical studies. 22
Hysteroscopy, an endoscopic procedure, is widely used to diagnose and treat intrauterine diseases. 23 Furthermore, with the development of the ERAS concept, outpatient surgery is becoming increasingly popular. Therefore, outpatient hysteroscopic surgery requires patients to be anaesthetised and recover rapidly, and the anaesthetic drugs used must be safe and effective. Although spinal anaesthesia is linked to a lower thoracic fluid load and better haemodynamic control than general anaesthesia in hysteroscopic procedures, 24 TIVA still has the advantage of rapid recovery in outpatient hysteroscopic surgery because spinal anaesthesia requires a longer recovery time and has associated side effects due to its invasive nature.24,25 Various anaesthesia and pain-relief methods have been described in outpatient hysteroscopic surgery, including paracervical block, sedation, and oral analgesics. 2 However, in mainland China, outpatient hysteroscopic surgery is mainly performed under TIVA because most of them are operative hysteroscopies.
Maintaining an appropriate depth of anaesthesia and analgesia is important in hysteroscopic surgery. Longer hospital stays, higher healthcare expenses and worse patient outcomes are associated with excessive nociceptive stimulation during surgery. 26 Although EEG-based hypnotic monitors such as Narcotrend may help minimize the dosage of hypnotic medications and maintain haemodynamic stability in patients with altered states of consciousness during surgery, these may not be suitable tools for assessing the response to noxious stimuli.27,28 During general anaesthesia, SPI is a reliable objective method for tracking the nociception-anti-nociception balance. 14 When compared to heart rate, response entropy, and state entropy, SPI had the strongest correlation with effect-site concentration of remifentanil in previous studies.29,30 During an outpatient hysteroscopic surgery, women can experience severe pain from the introduction and withdrawal of the hysteroscope, as well as, during distension of the uterine cavity and intrauterine procedures. The VAS pain score during hysteroscopic procedures is approximately 6–9. Where TCI remifentanil is administered with propofol, target remifentanil concentrations of 2–6 ng/mL are commonly used as previously recommended. 18 The interaction between remifentanil and propofol in TCI-based TIVA may induce deep or shallow anaesthesia. As an adjuvant drug, remifentanil can reduce adverse consequences and lower sedation requirements, while maintaining haemodynamic stability and satisfying anaesthesia depth. However, when the blood concentration of remifentanil is high, the synergistic effect of propofol and remifentanil leads to more adverse effects, such as muscle rigidity. In this study, the ED90 Ce of remifentanil was maintained sufficiently low for hysteroscopic surgery by combining SPI-guided analgesia with Narcotrend-guided sedation.
Remifentanil is considered to be associated with cardiovascular depression during general anaesthesia. 31 The incidence of hypotension during TIVA with high-dose remifentanil has been reported to be as high as 30%. 32 The current study mainly recruited cases with ASA class I or II, and only 7.5% of patients experienced hypotension, which is significantly lower than the previous study. This may be because the blood concentration of remifentanil used in this study was low. The frequency of hypotension is higher in vulnerable individuals, although low-Ce remifentanil is less hypotensive. In this study, no isolated bradycardia was observed, which occurred in three (7.5%) hypotensive patients. The incidence of bradycardia while using remifentanil for TIVA induction or maintenance ranged from 7% to 19%. 32 Remifentanil may induce chest wall rigidity when administered rapidly and in high doses. 31 This can make manual face-mask ventilation difficult during anaesthesia induction. 33 In this study, remifentanil was administered using a TCI device, and only two (5%) patients developed chest wall rigidity during anaesthesia induction. Although face-mask ventilation was difficult in these patients, no changes in oxygen saturation were observed.
Intraoperative remifentanil does not provide effective postoperative analgesia. Furthermore, remifentanil-induced hyperalgesia or acute opioid tolerance may be triggered by a higher remifentanil dose (Ce > 5.0 ng/mL). 34 In our study, all patients had satisfactory VAS, Ramsay sedation, and modified Aldrete scores in the PACU, and none had intraoperative awareness, emergence agitation, delayed awakening, shivering, postoperative nausea, or vomiting. These results suggest that a combination of low-Ce remifentanil and propofol for TIVA without NMBAs is associated with fewer adverse events and can be safely administered during outpatient hysteroscopy.
Limitations
Our study had several limitations. First, in this trial, remifentanil was only administered at four different concentrations (2.5, 3, 3.5, and 2 ng/mL) with a 0.5 ng/mL increment or decrement between each dosage. For remifentanil, a more accurate ED90 dose and narrower 95% CI may be achieved using an even smaller increment or decrement, such as 0.25 ng/mL. Second, this study only considered data from women of reproductive age (defined as those aged 18–50 years). Therefore, these findings may not be applicable to other populations. Finally, this study was unable to assess the extent to which the ED90 Ce of propofol decreased when combined with remifentanil because neither propofol nor remifentanil alone met the clinical needs of hysteroscopic procedures. Further studies using other opioids at different dosages are required for comparison and clarification.
Conclusion
In this dose-finding sequential study, the ED90 Ce of remifentanil in combination with propofol for TIVA without NMBAs during outpatient hysteroscopy was determined to be 2.75 ng/mL (95% CI, 2.50–3.37 ng/mL). Therefore, clinicians should consider a Ce of remifentanil between 2.75 and 3.0 ng/mL co-administered with propofol to decrease the adverse events associated with high opioid doses. The efficacy of the reduced Ce of remifentanil should be confirmed by conducting additional studies with larger sample sizes.
Supplemental Material
sj-docx-1-taw-10.1177_20420986241289204 – Supplemental material for Determining the optimum dose of remifentanil in combination with propofol for total intravenous anaesthesia in hysteroscopy under Narcotrend and SPI monitoring
Supplemental material, sj-docx-1-taw-10.1177_20420986241289204 for Determining the optimum dose of remifentanil in combination with propofol for total intravenous anaesthesia in hysteroscopy under Narcotrend and SPI monitoring by Xiaoyu Zhang, Tao Xu, Xiaohu An, Jianwei Wang, Qiong Meng and Zifeng Xu in Therapeutic Advances in Drug Safety
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
