Abstract
Background:
An important factor hindering the growth of pharmacovigilance (PV) in resource-limited settings is the lack of adequate funds to establish a functional National Pharmacovigilance System. Consequently, the crucial function of monitoring and ensuring the availability of safe medicines in these settings cannot be guaranteed considering the peculiarities of diseases and medicines used.
Objectives:
The objective of this paper is to provide an overview as to the availability of potential sources of funds, which could be explored to ensure Medicine Safety and to proffer a potential framework likely to ensure sustainable funding of PV in Africa.
Methods/processes:
The process of developing this framework entailed a review of PV financing in some developed economies, a landscape study of funding of PV in some African countries, an in-depth understanding of the PV system and the organisational structure and nexus between the regulatory agencies and National Pharmacovigilance Centre. Critical points for consideration included the sources of funds, revenue pool, the disbursement of funds, budgeting and expenditure profile and the legal framework. Consultative meetings, webinars and interviews with experts were carried out.
Results:
The findings showed that most of the PV systems were mainly integrated into the regulatory agencies regarding operational and fiscal governance with few facilities being independent of the regulatory agencies. The main source of funding was from the government with significant donor funding which is ad hoc and non-sustainable. Several potential sources were identified but yet to be exploited. There were no legal provisions for PV financing. A framework likely to ensure sustainable PV financing is suggested to capture all available sources of funding, mine the potential sources providing a sizeable pool of revenue to address its activities and enabling legal framework which will engender autonomy. Furthermore, it will address the nexus between the regulatory agencies and the PV outfits, thus enabling appropriate share of resources and blockage of diversions.
Conclusion:
In all, addressing the various elements identified in this study and providing the legal provisions which guarantees some degree of autonomy will provide a sustainable mechanism for PV funding in the resource-limited setting of Africa.
Plain language summary
An important factor hindering the growth of pharmacovigilance (PV) in resource-limited settings following their entry into the WHO Programme of International Drug Monitoring is the lack of adequate funds to establish a functional National Pharmacovigilance System. This article provides an overview of various potential sources of funds in these settings and how they can be harnessed to fund PV.
We undertook a review of PV financing in developed settings and carried out a landscape study of funding of PV in some African countries, as well as having an in-depth understanding of the PV system and the organisational structure. The nexus between the regulatory agencies and National Pharmacovigilance Centre was noted.
We took into account the sources of funds, revenue pool, the disbursement of funds, budgeting and expenditure profile and the legal framework for the different African countries. We also identified the prevalent and potential sources of funds for PV. Consultative meetings, webinars and interviews with experts in PV were carried out as well.
We discovered that most of the PV facilities were mainly integrated into the regulatory agencies regarding operational and fiscal governance with few facilities being independent of the regulatory agencies. The main source of funding was from the government with significant donor funding which is ad hoc and non-sustainable. Several potential sources were identified but yet to be exploited. There were no legal provisions for PV financing.
We have now proposed funding models that may lead to increased revenue for PV in these countries as well as suggesting that a legal framework be provided to guarantee sustainability and address the nexus between the regulatory agencies and the PV outfits to ensure an appropriate share of resources and blocking diversions.
Introduction
The onerous responsibility of ensuring the safety of medicinal products and vaccines has become urgent and of utmost importance following the significant potential morbidity and mortality with the intake of these products. The duties are delegated to establishments with experts which operate within the context of the regulatory agencies or independently. Over the years, these set of activities have come under the umbrella of pharmacovigilance (PV) and the establishments addressing the activities and processes of this system have been designated as National Pharmacovigilance Centre (NPC) or units. These establishments are usually affiliated to the World Health Organization (WHO) Programme for International Drug Monitoring (PIDM) which was established in 1968 with 10 countries from Europe, America and Oceania pioneering the programme 1 as an international response to the thalidomide tragedy. 2
There was much political goodwill and determination from the governments of these countries to support the programme providing financial and logistic support. African countries were late entrants into the PIDM, starting in 1992 with South Africa and Morocco. The gradual entry of the African countries occurred over a period of three decades and now stand at 44. 3
The performance of African countries remains under par when assessed by various measures including the objective indices of number of reports forwarded to the Uppsala Monitoring Centre (UMC) Uppsala database – VigiBase©. Until recently, the number of reports from the entire African countries in the VigiBase averaged 1% with an unimpressive trajectory, though with some marginal increase following the monitoring of the COVID-19 vaccines. 4
The PV activities are hindered by a number of factors notably financial and logistics and thus unable to achieve the programmes objectives.5,6 Considering the competition by various sectors of the economy in the low-resource settings, it could be inferred that the low prioritisation of PV activities is an important factor. The resource challenges in most African countries with a low gross national income per capita 7 are beset by a huge debt profile, spiralling inflation and unfavourable foreign exchange status of local currency. The psyche of government usually relates to provision of medicines in the healthcare system which is considered beneficial with minimal consideration for potential harmful effects and the need to monitor them. In effect, the financing of PV is poorly prioritised. The various other factors that contribute to the poor funding of PV activities and measures to obviate them have not been studied in a focused manner. Again, how all these impact on the funding of PV in the very competitive scheme of national resource allocation and how to remedy them is still speculative.
In realisation of this situation, PAVIA (PhArmacoVIgilance Africa), an European-Developing Countries Clinical Trials Platform (EDCTP), sponsored project intend to strengthen PV in the African setting. 8 In this regard, a sustainable funding mechanism is crucial. This paper evaluates the PV system, potential sources of funding and proposes a framework that is likely to guide the establishment of a sustainable financial system.
Conceptual framework
Definitionally, financial sustainability entails a long-term, continuous, adequate, stable, vested and interest-free funding. The funding mechanisms determining its sustainability should ensure the availability of funds for its stipulated functions.
To espouse the approach to developing a sustainable financing framework for PV in the healthcare system amidst competing demands and interests, a proper understanding of the milieu and overarching important considerations likely to impact on the process and eventual outcome is imperative. This also requires clarity regarding potential challenges, the essential elements of a financing model and the critical points to address, as highlighted below:
The notable challenges in PV financing include, though not limited to, the following: undermining of funding of PV activities in healthcare/pharmaceutical systems, low prioritisation of PV funding in the fiscal scheme of country’s pharmaceutical care, conflict of interest of major stakeholders, notably local manufacturers undermining the setting up of a robust mechanism to ensure appropriate funding as well as lack of budgetary provisions and legal structures to support funding.
It is imperative that the importance of PV be realised in the polity and placed on an appropriate pedestal so as to ensure adequate and sustainable funding. In view of the nature of PV, the ethics required in its operations and the set objectives to be realised, optimal considerations should be given to the funding mechanisms and the financial dynamics. The essential elements of any PV financing model should include, and not limited, the following:
Insulation of PV establishments from influences/interests likely to undermine its function.
Credibility of the source of funding.
Transparency and accountability in financial operations.
Availability of adequate funding devoid of bureaucratic hindrances.
Sustainability of funding to ensure continuity of PV programmes.
Funding should be holistic in scope so as to cover the PV outfits at all levels: facility, local, state or provincial, national, regional/continental as well as those at international/global levels.
Further strengthening of collaboration between Public Health Programmes (PHPs) and National Medicines Regulatory Agencies (NMRA)/NPC.
Critical points for consideration in PV financing
There are critical issues to consider in PV financing since they are determinants in the development of a functional financing model. These include the following (Figure 1):
PV system /NMRA structure
Sources of fund and financial pool
Budgeting and expenditure profile
Disbursement and allocation of funds – scope
NMRA and regulatory activities
PV facility and PV-specific activities
Enabling legal provisions

Critical points for consideration in PV financing.
PV system/NMRA structure
An in-depth understanding of the PV system, its operations and its nexus with the NMRA is of utmost importance for a proper characterisation of PV financing. The multiplicity of organisational structure of PV establishment vis a vis the NMRA implies the need for a financial model which will address the nexus between the two outfits. The prevalent organisational structures include the following:
The PV outfit fully integrated within the NMRA as a unit, department or a directorate. The operational name is the NPC.
The PV outfit is semi-autonomous (as a parastatal or directorate) with administrative issues handled independently but fiscal issues tied to the NMRA.
The PV outfit totally autonomous, independent regarding administrative and fiscal issues through interacting on matters of medicine safety.
There are variants of these main models with interaction of the Ministry of Health, NMRA and other support outfits with the NPC as exemplified by the Netherlands organisational structure. 9
The most common organisational structure in African countries is that in which the PV outfit is integrated into the NMRA’s administrative and fiscal operations (Figure 2). The PV outfits in Morocco and Tunisia are independent of their NMRAs regarding their administrative and fiscal operations (Figure 2). The relevance of these structures will be discussed below.
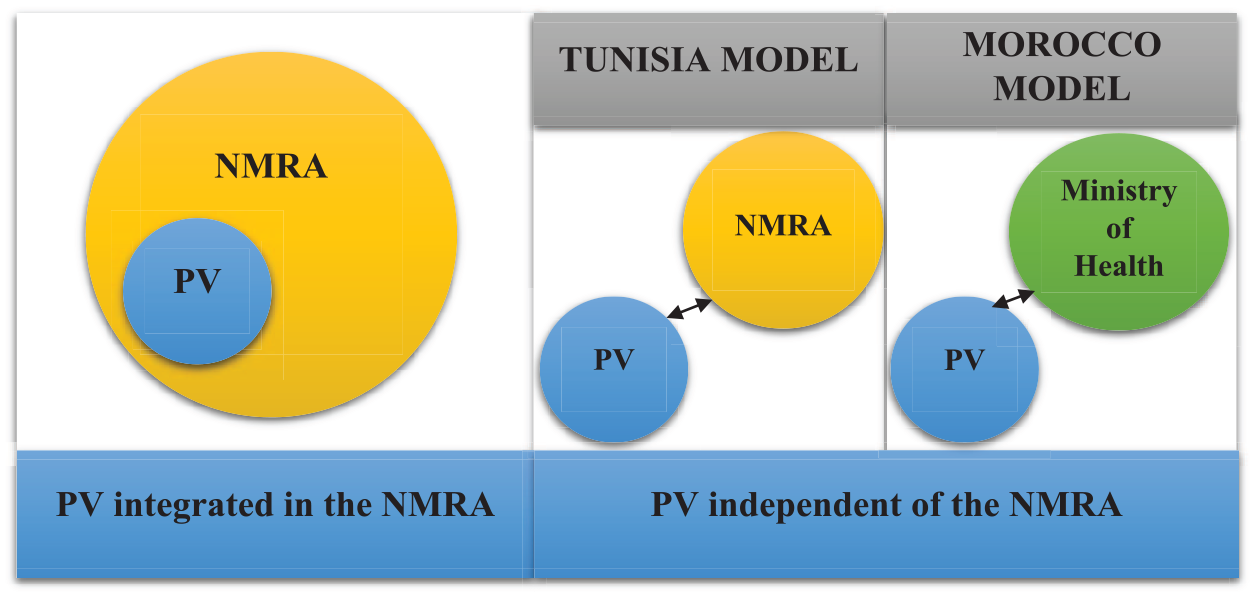
Organisational structure of regulatory agencies in relation to the PV outfit with variant organisational structures for PV outfits independent of the NMRA.
Sources of fund and financial pool
There are several potential sources of funds for PV. Indeed, financial sustainability may require focusing on several funding sources. To a large extent, the source(s) should provide an uninterrupted flow of funds to the revenue pool, and essentially, the pool should be maintained at a critical level, robust to absorb shocks and not easily decapitated by minor programmatic challenges. Some potential sources are mentioned below (Box 1). The extent to which the extant funding mechanisms are able to access these sources is of paramount importance. The revenue pool available for PV is further impacted by a number of other factors, such as local policies and laws that impact on the revenue pool. For instance, during appropriation, the PV-generated revenue can be allocated to non-PV projects. Again, arbitrary diversion of funds, outright fraud, non-compliance to established procedures, inappropriate implementation of post-authorisation safety studies, PV Inspection and other revenue generation activities. Again, the organisational structure which determines the principal recipient of the available fund is an additional factor.
Sources of funds.

The details of these sources and on how to access them vary from country to country. There are some local measures such as the revenue law and the treasury single account, which, in most settings, demands the deposition of monies accruing to any individual unit to be deposited into a central account. Most times, this generated revenue does not revert to the unit. Due to appropriation by authorities results in far less amounts shared to the PV unit and may impact negatively on the funds available for PV activities (Figure 3).

Factors impacting on the size of the revenue pool.
Some of the potential sources for PV funding are yet to be explored, and where and when they are explored are suboptimal in their reach. These include the various revenue-generating commercial services. The various health insurance schemes are yet to be implemented, and early attempts remain infantile with poor coverage of the population. The Ghanaian experience is a case in point, where the capitation payment model used in a pilot implementation of the national health insurance scheme failed due in part to political reasons and the perception of the population. 10 The promotion of health insurance schemes to increase its penetration in the country polity with due recognition of the need to contribute to PV activities will definitely be a veritable source. 11
There is also a need for the government to put in place earmarking policies as one of the mechanisms for generating revenue for PV. Earmarking is a promising source of revenue mobilisation if properly applied, for instance, to the consumption tax of alcohol, tobacco and other food products such as sugary beverages that impact negatively on health (‘sin’ tax). 12 The earmarking measures have been used for other purposes in several countries including South America, Philippines, Egypt, Ghana and South Africa. This can be applied to PV with appropriate advocacy to the government to enable the development of politically acceptable policies. Other consumption tax for example, value-added tax (VAT) has been a useful source. The Task Force on Fiscal Policy on Health 13 highlighted that raising the price of tobacco, alcohol and sugary beverages by increasing excise taxes not only reduces morbidity and mortality, but also generates additional tax revenues. Lotteries have also been a useful source. It is envisaged that for potential sources of funding, sensitisation of the public with focused advocacy and legal provisions could be put in place to ensure that laws are enacted to enable funds to be used to ensure the safety of medicines and vaccines. Incorporation of the intended source and modus in the PV policies is a useful first step in resource-limited settings. This is followed by champions in the legislature proposing a bill for the enactment of the intended law. A small percentage (even 1%) of VAT, health insurance, earmarking (sin tax) would be of immense value for PV. In essence, the process will entail the creation of awareness/sensitisation of the public, focused advocacy, engagement of the legislature followed by executive endorsement.
The role of different categories of donors in the development and sustenance of PV has been substantial over the years. The ad hoc and perennial nature of this source do not guarantee financial sustainability and do not allow for proper budgeting. However, sizeable donations may increase the revenue pool, and considering the focus of these donations, they may be useful in achieving the objectives of many public health situations. Multi-donor sources should be sought after, and dependency on sole donors discouraged.
Pharma consortium and PV tax on pharmaceutical sales
A pharmaceutical consortium dedicated to PV activities could be a potential source of financing for PV activities. However, this is usually difficult to achieve at national levels considering the chain of command. The Head Office of the Pharma is located in foreign countries and are unlikely to adopt such policies. Of interest is the support from big pharma during the COVID pandemic. The most likely approach to achieve success at the national level is the PV tax on pharmaceutical sales. This could be established after due consultation in accordance with statutes. However, the possible backlash of an increase in drug prices (mark-up) must be dealt with.
Budgeting and expenditure profile
Realistic budgeting is an important element in ensuring judicious fiscal planning. One of the factors highlighted in the African PV financial landscape survey was the poor budgeting. 14 There was a poor understanding and arbitrariness of the budgeting process. There is a need to relate PV activities 15 (Box 2) to financial expenditure so as to develop a realistic budget which can be reflected in a dedicated budget line in the national fiscal plan. The expenditure profile in PV result from wages, operational costs and development of infrastructure which vary to a large extent depending on the programmes and growth of the outfit. The expenditure including purchasing arrangements must be well planned having in view available resources (Figure 1).
Some functions of the PV system. 15

DD, drug dependence; DSMB, data and safety monitoring board; ICSRs, individual case safety reports; PBRER, periodic benefit–risk evaluation reporting; PSURs, periodic safety update report; SFs, substandard and falsified medicines; SOP, standard operating procedures.
The WHO PV Tool Kit 16 is a useful resource to achieve this. Again, there should be compliance with extant provisions of the national laws and provisions to ensure transparency and accountability. Attention to detail must be applied in the development of budgets by attributing realistic cost to the intended activities and items for purchase to achieve its objectives.
Allocation and disbursement of funds – scope
The mechanisms for allocation and distribution of funds depend on the levels in view. This paper deals with national and subnational levels which include Zonal and/or Regional Centres, Primary Care and Hospitals – Cottage and Tertiary and PHP.
The government should ensure a sustainable funding by expenditure earmarking to PV as a subsector of the health system guaranteeing allocation of funds from the Ministry of Health or relevant agencies. This will increase the financial pool available for PV activities.
The allocation of resources is to a large extent, determined by the organisational structure. As mentioned above, when the primary recipient is the NMRA with the integrated PV outfit, the allocation of funds to the latter has been arbitrary. Most times, the priorities of the agencies are set far and above those of PV. It may not relate to budgetary provisions. It is therefore necessary to define a clear formula for the funds to be allotted predicated on the PV budget. The absence of a clearly defined formula is a frequent cause of friction and underfunding of PV.
In the instance where the PV outfit is the primary recipient of the pooled revenue, the main concern is the judicious use of funds vis a vis a realistic budget. The funds allocated to other sublevels should depend on the responsibilities they bear and the expected deliverables.
Legal framework
One of the main findings in the African PV funding landscape study was the absence of a legal framework in most of the countries. 14 One exception was Zimbabwe, which has a legal provision for the NMRA which also oversees PV in the integrated system operating in the country. However, there is no formula to ensure clarity in the allocation/distribution of resources from the NMRA to the PV outfit.
The legal framework should address all steps identified in the chain of critical points likely to influence PV finances. The fiscal policies of the government should be inclusive of issues related to Medicine Safety. The USA Prescription Drug Users Fee Act (PDUFA) is an example of a source of funding backed by legislation. 17
A WHO study reported a decrease from 100% to 60% in donor funding between 1995 and 1997 in Uganda as a result of changes in the NMRA funding policy, with increase in the proportion from government and industry. 18 A study by Ndomondo-Sigonda et al. 19 in 2020 confirmed this earlier finding of the WHO study, stating that the Uganda National Drug Authority (NDA) is currently 98.25% funded through fees for service with minimal contribution from donors.
Areas to be addressed include statutory allocation of funds from the government and a provision granting financial autonomy to the PV outfits, thus enabling retention of their fees for service provided, for example, exemption from revenue law/single treasury account. Legal provisions to enable earmarking in identified areas including health insurance should also be considered. In effect, definite policies, laws and regulations must be put in place to ensure a favourable environment for PV, using provisions of the legislative and executive arms of government.
Methods/process
The process of developing a model for sustainable financing of PV activities in resource-limited African countries required extensive consultation and an understanding of the prevailing PV system and the financial dynamics. This series of evaluations was carried out during the period 2019–2022 in the context of the overall PAVIA project by Work Package 2, with some contribution from Work Package 1.
The various steps entailed the following:
A review of funding of PV in some well-developed economies.A purposed literature search was conducted mainly targeting PV financing models from developed and well-resourced countries, with a view to understanding the PV financing models and systems in these countries. Information about these models was collected from journals and other online information through basic Google searches. The search was further extended to include multilateral/bilateral funding agencies such as the World Bank, Global Fund, EDCTP and Bill and Melinda Gates Foundation (among others).
A landscape questionnaire survey 14 of the funding modalities for PV in African countries provided a situational analysis and notably provided information regarding the organisational structure of the regulatory agencies and the nexus with the National PV outfits, sources of revenue and the constraints in ensuring sustainable funding.
Consultative meetings, webinars and interviews with experts on sustainable funding for PV were solicited, as reflected in the Acknowledgement section.
The information thus obtained enabled the development of potential frameworks for PV financing models.
Findings
Literature search on the funding of PV in some well-developed economies
The establishment of the PIDM by the WHO required a substantial grant from the US government, facilitated by a Presidential executive order, and later the Swedish government following its transfer to Uppsala, Sweden in 1978. 1 The UMC, as it is called, eventually became self-funded as a Foundation and financially independent, relying on internally generated revenue mainly from sales of its dictionaries. 20
Funding of activities to ensure the safety of medicinal products has been supported by the governments of the various countries, usually through their agencies and competent authorities. In the United States, the FDA benefitted from the PDUFA17,21 which was an executive-legislative overture to financially empower the FDA to enhance its efficiency in the handling of processes for licensure since 1992. This was specifically extended to enhance activities relating to ensuring the safety of medicines in a subsequent legislative provision in USA Prescription Drug Users Fee Act IV (PDUFA IV)Act 2007. 21
In the European Union, the European Medicines Agency (EMA) coordinates the PV system and operates processes and services to support PV. The EMA charges and collects fees from pharmaceutical companies for carrying out PV activities. The fee regulation: [Council Regulation (EC) No. 297/95] and the PV fee regulation [Regulation (EU) No. 658/2014] determines EMA fees. The PV fee regulation covers two types of fees: procedure-based fees and annual fees relating to PV activities of the EMA, and it is applicable to nationally authorised medicines. Annual fees relating to centrally authorised medicines are covered by Regulation (EC) No. 297/95.22,23
In the United Kingdom, the Medicines and Healthcare products Regulatory Agency (MHRA) is the national competent authority and main regulatory body regarding medicines and PV and is an executive agency of the Department of Health, which is responsible for matters of legislation and finance. PV funding by the MHRA is met through fees by the pharmaceutical industry, mainly for marketing authorisation of their products24,25 and complex licensing decisions are referred to the agency’s independent advisory committees to address conflicts of interests. 25 Further notable funding sources include the Bill and Melinda Gates foundation and the WHO mainly to address safety monitoring for new medicines in low- and middle-income countries (LMICs). 26
In Germany, PV is based on a highly complex and centralised system. Whereas Adverse Drug Reactions (ADRs) related to synthetic medicines are centrally collected by the Federal Institute for Drugs and Medical Devices (Bundesinstitut für Arzneimittel und Medizinprodukte, BfArM) , ADRs resulting from biologicals must be reported to the Federal Institute for Vaccines Paul-Ehrlich-Institut (PEI). Even though both agencies are independent and act as centralised agencies, they have a nearly identical legal basis and have similar instruments at their disposal when it comes to PV and ADR reporting. BfArM and PEI are both under the supervision of the Federal Ministry of Health. Both national competent authorities are financially dependent on the Federal Ministry of Health. 27
The French national competent authority, Agence Nationale de Sécurité du Médicament et des Produits de Santé (ANSM), is a public institution that is financed by the Social security budget and other resources of the Agency. It is responsible for the legal framework, finances and overall supervision of the French PV system. The regional centres rely on financing from the state budget and political priorities set by the Health Ministry. 28
In the Netherlands, the Medicines Evaluation Board is the drug regulatory authority. The Netherlands Pharmacovigilance Centre (Lareb) carries out PV tasks such as spontaneous reporting system and its signal detection for registered and unregistered drugs and health products falling under the food legislation. The Medicines Evaluation Board, which is the drug regulatory authority funds the former while the latter is funded by the Inspectorate of Health for non-registered medicinal products and the Food and Consumer Product Safety authority for products falling under their responsibility). Lareb also receives funding from the Ministry of Health to provide information about Adverse Events Following Immunusations (AEFIs) and for having a teratology information service.9,29
In Japan, the substantial source of PV funding is from user fees and other contributions, with much less from the government. 30
Findings from the landscape study of PV funding in African countries
In a preliminary PV landscape study of 24 African countries, 14 the findings were remarkable and notably included the following:
The funding from the government for PV though significant is amorphous with budgetary allocations only partially, if released.
The funding mechanisms of PV are inextricably linked to the NMRAs.
Limited statutory provisions (legal framework) for PV financing.
Significant donor funding (multilateral and bilateral agencies).
Prominence of PHPs budgetary items regarding PV activities is not clear.
No clear formula for release of PV related funds by NMRAs to NPCs.
Absence of permission to accrue, retain and use revenue for PV activities.
Involvement of PV personnel in other activities outside PV.
Unclear disclosure of funds received from donor bodies.
The various factors hindering and those likely to improve and ensure sustainable PV funding are stated in an earlier publication. 14
Considerations for proposed model(s)
In essence, considering the factors outlined above, the framework for any model for PV financing in Africa will be based on the prevalent organisational structure for PV establishment and a likely futuristic option as highlighted above, thus:
NMRA with an integrated PV outfit
○ NMRA – principal recipient of funds
Independent PV outfit operating outside the administrative ambits of the NMRA, though interacting on issues of Medicine Safety (and variants thereof)
○ PV outfit – principal recipient of funds
NMRA with an integrated PV outfit
This is the operating framework for Medicine regulation and PV in most African countries. The degree of integration may vary from country to country, but for a majority of countries the governance is intertwined. Figure 4 illustrates this integrated NMRA-PV model. The recipient of the revenue from the sources shown is usually the NMRA. The allocation from the pool to the PV establishment or for PV activities is determined by the regulatory agencies according to their set priorities.

The proposed model for financing PV: integrated NMRA-PV and independent PV organisational models.
To ensure PV is not marginalised with this prevalent framework, the following considerations should be addressed:
A clear proportion of the revenue should be allotted to the PV establishment for PV activities.
Some formula for sharing of revenue should be clearly stated and backed by statute.
Funds, including donations primarily dedicated for PV activities, should not be diverted by the NMRA.
PV outfit independent of NMRA
PV outfits with an administrative structure independent of the NMRA are not common. In Africa, the PV outfits in Morocco and Tunisia are independent of the NMRA.
In Morocco, the Centre Anti Poison et de Pharmacovigilance du Maroc (CAPM) is completely independent of the drug regulatory body but works closely with them to achieve PV objectives. As a result, it reports directly to the Ministry of Health and thus benefits from independent, regular funding enshrined in the finance law.
The CAPM director is in charge of managing the finances allocated for covering investment and operating expenses, according to a predetermined budget. The salaries are excluded from this budget and are paid directly by the Ministry of Health. The CAPM also sources funds from other international organisations (WHO, USAID, Centre for Disease Control – CDC).
Of interest, in Tunisia, the Chalbi Belkahia National Centre of Pharmacovigilance predated its Regulatory agency, it still maintains its independent status with its direct funding from government. With this framework, the PV outfits are the principal recipient of resources. Internal mechanisms have to be put in place to manage the resources in a prudent and transparent manner. There may be variants of these two organisational structures (Figure 3) which form the framework for the model options as with Lareb. 29
The main focus for either framework is revenue pooling mechanisms which determine the available funds for PV activities. As mentioned above, there are several potential sources of funds.
These may fall into two main categories: statutory funding backed by a legal framework and the perennial ad hoc funding. While the former is likely to be sustainable, the latter is not. The various measures including intense advocacy to position PV in the National polity so as to secure this funding should not be underestimated. The LMICs priorities set by governments are at times uninformed, misplaced and definite revenue generating measures are absent and when present infantile and tied to political squabbles. Areas such as health insurance are futuristic due to the poor coverage, reluctance of stakeholders to provide support and unwillingness of the legislature to put in place the legal framework. 10
It is essential that the main objective is the sustainability of financing for whichever framework is operational. It is pertinent to ensure an adequate revenue flow and maintain an appropriate revenue–expenditure balance. The robust nature of this model is such that it should be able to absorb shocks from the peculiar demands of the PV system such as seen in crisis situations. This is more so for the LMICs with erratic economies where perturbations in the overall economy can crash the PV establishment.
Conclusion
In essence, the availability and sustainability of funding for PV activities is paramount if the requisite monitoring to ensure the safety of medicines is to be done. The advocacy to the stakeholders on the need to adequately fund the PV activities bearing in mind the sacrosanct nature of its operation and deliverables should not be undermined. There exist multiple sources of potential revenue which should be fully explored, and due regard should be given to the organisational structure, notably the nexus between the regulatory agencies and the PV outfits. The sustainability of the model hinges to a large extent on the legal framework; statutory provision of funds, unbundling of the system from bureaucratic and fiscal hindrances so as to provide a seeming autonomy of a robust financial operation. Again, the sustainability of any financial model will depend on fiscal discipline with prudent expenditures subject to a transparent accounting procedure.
Footnotes
Acknowledgements
This paper was developed within the framework of an EDCTP2-funded project PAVIA. The funding of PAVIA by the EDCTP is gratefully acknowledged. The participation of all stakeholders and their contribution is noted with due gratitude. The participation and the contribution made by the following experts during the various webinars and other personal correspondence is noted with thanks: Members of PAVIA Advisory Board, Members of PAVIA Steering Committee, Members of PAVIA Executive Board, Dr. Sten Olsson (now Late), formerly of the WHO-UMC Uppsala, Dr. Helen Byomire Ndagije (President African Chapter ISoP), Professor Parthasarathi Gurumurthy, (Botswana Medicines Regulatory Agency), Ms. Helena Sköld WHO Uppsala Monitoring Centre (WHO-UMC), Uppsala, Sweden.
