Abstract
Objective:
Limited real-world efficacy and safety data exist regarding the use of trastuzumab deruxtecan (T-DXd) in the Chinese population with human epidermal growth factor receptor (HER2)-positive and HER2-low advanced breast cancer (BC). This multicenter, observational, real-world study aimed to evaluate the efficacy and safety of T-DXd for the treatment of Chinese patients with HER2-positive and HER2-low advanced BC.
Methods:
The medical records of 61 patients were collected from The Second Hospital of Dalian Medical University, Beijing Chaoyang District Sanhuan Cancer Hospital, Beijing Jingxin Hospital, and Cancer Hospital of the Chinese Academy of Medical Sciences. The primary endpoint of the study was progression-free survival (PFS), and the secondary endpoints were overall survival (OS), objective response rate (ORR), disease control rate (DCR), time to response (TTR), and safety. PFS and OS were analyzed using the Kaplan–Meier method and log-rank test.
Results:
The primary endpoint, PFS was 10.51 months (95% confidence interval (CI), 3.02–NE) in the HER2-low group and 10.18 months (95% CI, 3.88–NE) in the HER2-positive group. Regarding the secondary endpoints in the HER2-low and HER2-positive groups, OS data were immature, ORR rates were 37.93% and 62.50%, DCR rates were 79.31% and 87.50%, and the median TTR rates were 1.28 and 1.31 months, respectively. In the subgroup analysis, front-line treatment with T-DXd was associated with increased beneficial effects. The primary adverse events (AEs) related to T-DXd treatment were gastrointestinal reactions and bone marrow suppression, which were predominantly grades 1–2, with no severe grade 4/5 AEs reported, only one patient developed infectious pneumonia.
Conclusion:
This study was the first multicenter, real-world study of T-DXd for advanced BC in China. The findings demonstrated that T-DXd may be an effective antitumor treatment with controllable adverse reactions in patients with advanced BC irrespective of HER2 expression levels.
Introduction
Breast cancer (BC) is one of the most common malignancies affecting women worldwide.1,2 According to the 2020 World Health Organization report, approximately 2.26 million new cases and 680,000 deaths from BC are reported annually, and these numbers are continuously increasing.3,4 In China, BC accounts for 20% of all malignancies, establishing it as one of the most prevalent cancers in the nation.5,6 Approximately 3%–10% of cases show distant metastases at the time of diagnosis.7,8 Approximately 30% of patients progress to advanced stages of the disease, with a stark 20% 5-year survival rate and a median overall survival (OS) time of 2–3 years, significantly impacting women’s health and imposing a substantial economic burden. 9 Therefore, the identification of effective treatments for advanced BC remains of utmost importance.
Human epidermal growth factor receptor (HER2) plays a critical role in cell proliferation and invasion and is considered an important target for tumor treatment. 10 Approximately 20% of BC patients show HER2 overexpression and amplification (HER2-positive). 11 HER2-positive BC is often more aggressive than other BC subtypes, leading to an increased risk of recurrence and metastasis.12,13 Approximately 60% of HER2-negative metastatic BCs have low HER2 levels (Immunohistochemistry (IHC) 1+ or 2+ with negative In situ hybridization (ISH)). These “HER2-low” tumors, which include both hormone receptor-positive and -negative types, have varied prognoses and treatment responses. Previous common HER2-directed therapies had not improved outcomes in patients with this subtype; therefore, HER2-low BC was treated like HER2-negative cancers, with limited options after progression on primary therapy and poor prognosis.14,15
Numerous drugs targeting HER2 have been developed in recent years, including monoclonal antibodies, 16 small molecule tyrosine kinase inhibitors (TKIs), 17 and antibody–drug conjugates (ADC).18,19 ADC drugs deliver potent cytotoxic drugs to HER2-expressing cancer cells, utilizing the precision of targeted therapy to hinder tumor growth and mitigate adverse drug reactions. 20 Trastuzumab deruxtecan (T-DXd) is a novel topoisomerase I inhibitor ADC characterized by high membrane permeability and robust cytotoxicity. It aims to circumvent microtubule drug resistance and elicits a bystander effect, which makes it effective against heterogeneous tumors, particularly in BC. 21 Following the positive outcomes of the DESTINY-Breast01 trial, the FDA approved T-DXd for late-line treatment of HER2-positive advanced BC. 16 Subsequent trials, such as DESTINY-Breast02, 03, and 04, further validated its efficacy in the front-line treatment of HER2-positive and HER2-low advanced BC.21 –24 These studies are prospective registration studies with strict inclusion and exclusion criteria, and mainly involving foreign populations outside China. In February 2023, T-DXd received approval for commercialization in China, expanding the treatment options for BC patients. 25
Considering the short period since its market introduction and the high cost of T-DXd in China, there is currently insufficient real-world data on the efficacy and safety of T-DXd among late-stage BC patients with low HER2 expression or HER2 overexpression in the Chinese population. In this study, we collected data from 61 clinical patients across four centers between October 2020 and March 2024. This study aimed to evaluate the efficacy and safety of T-DXd in Chinese patients with HER2-low and HER2-positive BC and provide real-world evidence for clinical practice. Compared with prospective registration clinical trials, our unique features include the following: broader inclusion criteria for research subjects including advanced BC patients with ECOG scores 0–4, which can better reflect the application of T-DXd in an actual medical environment with a more diverse and broader population. And being the first multicenter study on Chinese BC patients, ours can provide more comprehensive and representative insights into the T-DXd for HER2 low and positive advanced BC.
Materials and methods
We used the STROBE cohort reporting guidelines. 26
Study design and participants
This is a multicenter, observational, real-world study. The enrolled patients were those who received T-DXd for the treatment of metastatic BC between October 2020 and March 2024, and the study was carried out at the following four centers: The Second Hospital of Dalian Medical University, Beijing Chaoyang District Sanhuan Cancer Hospital, Beijing Jingxin Hospital, and the Cancer Hospital of the Chinese Academy of Medical Sciences. Patients who met the following criteria were enrolled in the study: pathologically confirmed invasive BC with HER2-positive or HER2-low immunohistochemistry; at least one distal measurable lesion that met the definition of RECIST 1.1 solid tumor efficacy evaluation criteria; the test values for blood routine labs, liver and kidney function, and electrocardiogram examinations are in the normal range. All patients who met these criteria were included in the study to avoid possible selection bias; the exclusion criteria for this study are as follows: cognitive impairment and disorders of volition and behavior, presence of other tumors, severe comorbid conditions, and patients with incomplete clinical data.
Treatment protocol
The planned dose of T-DXd was 5.4 mg/kg diluted in 5% glucose to a volume of 100 ml for intravenous infusion every 3 weeks. Tumor site computed tomography or magnetic resonance imaging data were re-examined every two cycles; BC tumor markers were monitored during the treatment of T-DXd. Liver and kidney function and electrocardiogram were re-checked every cycle. Blood routine laboratories were monitored 1–2 times per week to evaluate adverse reactions related to T-DXd treatment; treatment was discontinued upon tumor progression or the occurrence of intolerable adverse reactions.
Follow-up and efficacy and safety assessment
Follow-up was completed through a review of medical records and telephone interviews; the follow-up deadline was July 30, 2024. The primary research endpoint was progression-free survival (PFS), which was defined as the time from the start of T-DXd treatment to disease progression or the occurrence of any event leading to death; progression events were assessed by researchers following RECIST 1.1 criteria. Secondary endpoints included OS, defined as the time from the start of treatment to any event leading to death, objective response rate (ORR), defined as the proportion of patients with the best tumor response assessment as complete response or partial response (PR), disease control rate (DCR), defined as the proportion of patients with the best tumor response assessment as complete response, PR, or stable disease (SD), and time to response (TTR), defined as the time from the start of treatment to response. Safety assessment included adverse events (AEs) assessed by researchers based on NCI CTCAE 5.0 standards.
Statistical analysis
Statistical analyses were performed using SAS (version 9.4). (SAS Institute Inc., North Carolina, USA). All analyses were descriptive in nature. PFS and OS were analyzed in all enrolled patients. ORR and DCR were analyzed in patients with any measurable baseline lesion. Safety analyses were performed in patients who had received at least one treatment dose.
For the time-to-event endpoints, the Kaplan–Meier method was used to estimate medians, and Kaplan–Meier curves were plotted to present the results intuitively. The survival rates at different landmarks were estimated using the Kaplan–Meier method. For ORR and DCR analyses, the Clopper–Pearson method was used to calculate the 95% confidence interval (CI). Safety was analyzed descriptively.
Results
Study population
The first patient was enrolled in October 2020, and the last patient completed the study visit in March 2024. A total of 61 eligible participants were included from 9 provinces across the country, and their sources covered 4 different types of hospitals: the Chinese Academy of Medical Sciences, a Class-A tertiary specialized cancer hospital with authority in cancer diagnosis and treatment; The Second Hospital of Dalian Medical University, a Class-A tertiary general hospital that provides comprehensive medical services; The Beijing Chaoyang District Sanhuan Cancer Hospital, a medium-sized specialized tumor hospital focusing on the diagnosis and treatment of tumor diseases; and the Beijing Jingxin Hospital, a medium-sized general hospital capable of serving various patients. There were four cases of study termination because they used only one cycle of treatment (Figure 1). The demographic and baseline characteristics of the patients are summarized in Table 1. The 61 patients were all female and the median age was 55 years. Twenty-nine exhibited low HER2 expression (47.50%), while 32 were HER2 positive (52.50%). More than 30% of patients had an ECOG score of 2–4. T-DXd was administered as first- or second-line therapy in 14 cases (22.95%), as a third line or fourth line in 28 cases (45.90%), and beyond the fourth line in 19 cases (31.15%). Among patients with metastasis, there were previously been treated with anti-HER2 targeted drugs such as pertuzumab (31.15%) or trastuzumab (49.18%), 21 had received CDK4/6 inhibitors (34.43%), and 15 had been treated with ADC. Visceral metastasis was observed in 52 cases (85.25%).

Flow diagram.
Baseline demographic and clinical characteristics.

CDK4/6, cyclin-dependent kinase 4/6; HER2, human epidermal growth factor receptor. Fish, fluorescence in situ hybridization.
Effectiveness
Follow-up data were collected from the 61 patients included in the study up to July 30, 2024. In the HER2-low expression and HER2-positive expression groups, PFS was 10.51 months (95% CI, 3.02–NE) and 10.18 months (95% CI, 3.88–NE), respectively (Figure 2(a) and (b)). The secondary endpoint OS was not mature at the end of the study. Table 2 shows that the ORR of 29 patients with HER2-low expression was 11 (37.93%) and that of 32 patients with HER2-positive tumors was 20 (62.50%); the DCR was 23 and 28 (79.31% and 87.50%), respectively. The median TTR for the HER2-low expression population was 1.28 months (95% CI, 1.25–1.31), and the median TTR for the HER2-positive population was 1.31 months (95% CI, 1.28–2.33). In the HER2-low and HER2-positive groups, complete response was achieved in 0 and 2 (6.25%) patients, respectively, PR in 11 (37.93%) and 18 (56.25%), and SD in 12 (41.38%) and 8 (25.00%), respectively. Progressive disease occurred in 6 (20.69%) and 4 (12.50%) patients in the HER2-low and -positive groups, respectively (Table 2). A waterfall plot is shown in Figure 2(c) and (d).

Efficacy of T-DXd and median PFS and overall survival of patients with low HER2 expression and those with HER2 overexpression. (a) Kaplan–Meier curve for PFS in low-HER2 patients. (b) Kaplan–Meier curve for PFS in HER2-positive patients. (c) Waterfall plot showing the effectiveness of T-DXd in low-HER2 patients as determined by PD, SD, and PR. (d) Waterfall plot showing the effectiveness of T-DXd in HER2-positive patients as determined by PD, SD, and PR.
Best overall response (BOR).
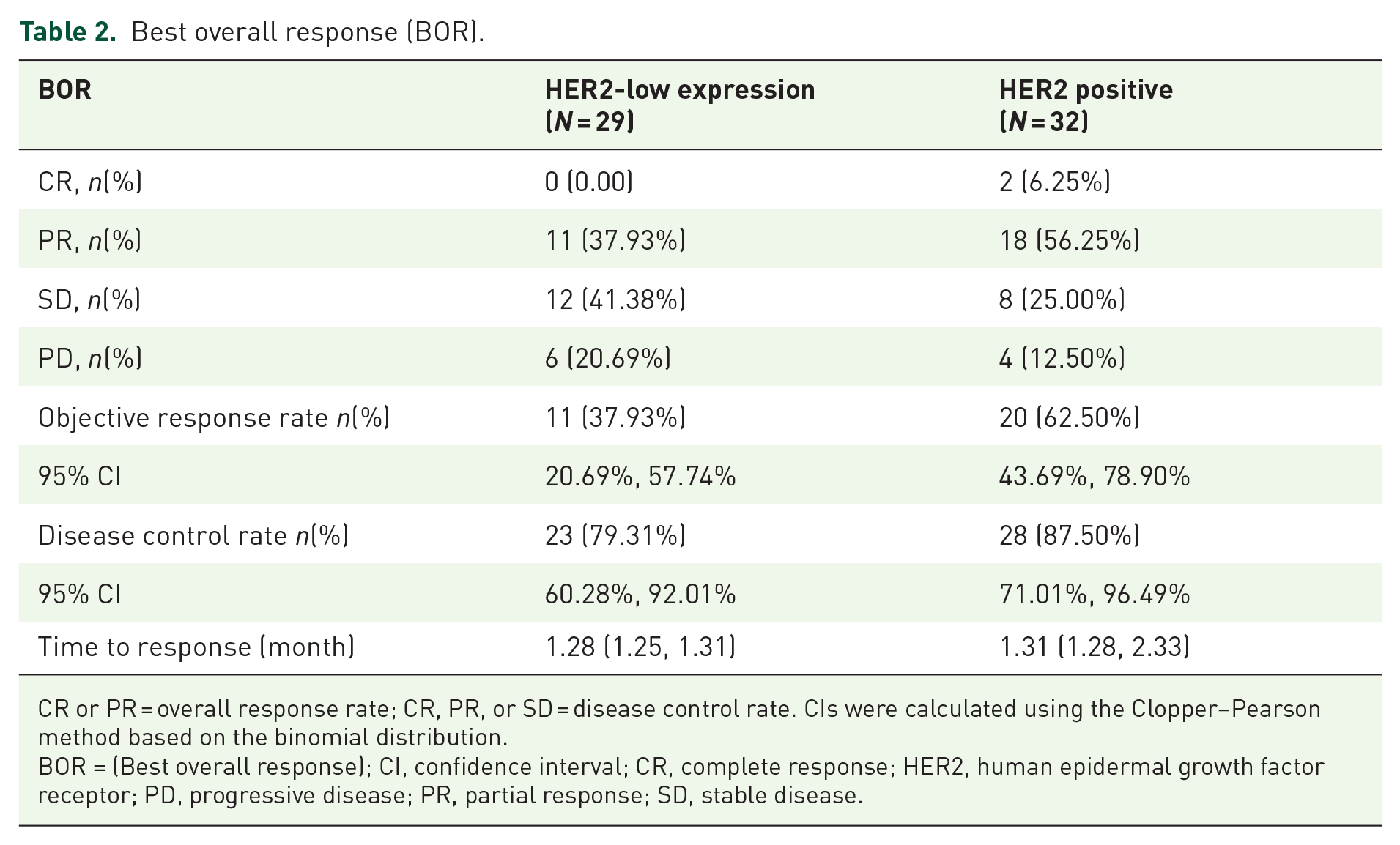
CR or PR = overall response rate; CR, PR, or SD = disease control rate. CIs were calculated using the Clopper–Pearson method based on the binomial distribution.
BOR = (Best overall response); CI, confidence interval; CR, complete response; HER2, human epidermal growth factor receptor; PD, progressive disease; PR, partial response; SD, stable disease.
Safety
According to ECOG scores, weight, and economic status, 54 patients (88.52%) received a reduced initial dose of T-DXd, whereas 5 patients (8.20%) had their dose adjusted during treatment due to gastrointestinal and bone marrow suppression toxic side effects (Table 3). Adverse reactions occurred in 59 patients, and the most frequent AEs were nausea in 48 (78.69%), anorexia in 45 (73.77%), leukopenia in 21 (34.43%), anemia in 18 (29.51%), alopecia in 14 (22.95%), vomiting in 12 (19.67%), diarrhea in 9 (14.75%), thrombopenia in 9 (14.75%), and constipation in 6 (9.84%) patients. Among the total 61 patients, 17 cases (27.87%) adopted the NK1 + Granisetron + Dexamethasone regimen for preventive antiemesis, and 29 (47.54%) adopted Granisetron + Dexamethasone regimen (Table 4). Among them, there were five cases of Grade 3 or higher leukopenia AEs (8.20%), four cases of anemia (6.56%), and one case of thrombopenia (1.64%; Table 5).
A summary of the statistics of patient exposure in T-DXd.

HER2, human epidermal growth factor receptor; T-DXd, trastuzumab deruxtecan.
A summary of AEs in T-DXd.

AE, adverse event; HER2, human epidermal growth factor receptor; T-DXd, trastuzumab deruxtecan.
AEs in DS-8201 with grade ⩾3.

AE, adverse event; HER2, human epidermal growth factor receptor; T-DXd, trastuzumab deruxtecan.
Discussion
Summary of results
T-DXd has been approved in China for the treatment of advanced HER2-positive and HER2-low BC.21,27,28 However, there is a lack of large-sample real-world study data from China. This study investigated the efficacy and safety of T-DXd using real-world research data. The findings indicate that T-DXd remains effective in heavily pre-treated advanced BC patients with positive or low HER2 expression, brain metastases, and multiple-line treatment failures. T-DXd treatment shows marked antitumor activity, manageable adverse reactions, and clinical utility.
Effectiveness
Previous studies mostly focused on Western populations, Asian regions, and so on. In the prospective, open-label phase II DAISY trial evaluating the efficacy of T-DXd in 177 French patients with metastatic BC with HER2 overexpression (IHC3+ or IHC2+/ISH+, n = 72, cohort 1) and HER2-low expression (IHC2+/ISH− or IHC1+, n = 74, cohort 2), the median PFS was 11.1 and 6.7 months, respectively. 27 Similarly, the global phase III DESTINY-Breast03 study (NCT03529110) demonstrated that, from North America, Asia, Europe, Australia, and South America among patients with HER2-positive metastatic BC who had previously received trastuzumab and paclitaxel treatment, T-DXd showed statistically and clinically significant improvements in PFS and OS compared with trastuzumab emtansine (T-DM1). Data from a subgroup analysis of the Asian population in the DESTINY-Breast03 study showed that among the patients who received T-DXd, 149 were from relevant Asian countries and some regions of China. Specifically, 37 were from the Republic of Korea, 37 were from Japan, 35 were from the Chinese mainland, 31 were from the Taiwan region of China, and 9 were from the Hong Kong Special Administrative Region of China indicating that T-DXd is a suitable treatment option for Asian patients with HER2-positive metastatic BC. 29 At the ESMO Congress in September 2024, a clinical study was released, which compared the efficacy and safety of T-DXd or standardized chemotherapy regimens in treating patients with hormone receptor-positive, HER2-low-expressing or HER2-ultra-low-expressing advanced BC who were resistant to endocrine therapy but had not yet received chemotherapy for the first time. The results suggested that T-DXd demonstrated a significant PFS benefit in patients with HER2-low-expressing and HER2-ultra-low-expressing advanced BC, and its safety was consistent with that of previous studies. 30 However, there are very few real-world studies on the Chinese population. Clinical trials usually have strict inclusion and exclusion criteria and are conducted for specific indications of drugs, which leads to a relatively narrow study population. In our real-world study of the Chinese population, the median PFS was 10.51 months in patients with low HER2 expression (n = 29), which was comparable with the median PFS in DESTINY-Breast 04 trial (mPFS in ITT HER2 low population, 9.9 months). The median PFS with 10.18 months was observed in those with HER2-positive expression (n = 32), although numerically lower than the DESTINY-Breast 02 and DESTINY-Breast 01 studies,31,32 it is important to note that in this real-world study, nearly 40% of HER2-positive patients had an ECOG score of 2 or more and had poor baseline status. Besides, we adjusted the treatment dosage and cycle regimen according to the specific circumstances of patients (such as their financial situation, physical tolerance, etc.), thus affecting the effectiveness of T-DXd treatment in real clinical practice. Overall, this was the first real-world study on advanced BC patients in the Chinese population and our research aligns with the outcomes of previous clinical trials. T-DXd has exhibited favorable efficacy in patients with advanced BC irrespective of HER2 expression levels. Notably, the efficacy and survival rate in front-line treatment may possess greater advantages compared to later-line treatments. Moreover, our results, which are derived from the real medical environment, offer significant references for clinicians in making decisions regarding drug therapies in subsequent clinical practice.
Clinical relevance
The brain is a common site of metastasis in HER2-positive BC patients, and brain metastasis often occurs in late-stage disease.16,33 It was previously believed that only small molecule TKIs could effectively penetrate the blood–brain barrier. In recent years, new ADC drugs, represented by T-DXd, have shown efficacy against brain metastasis from HER2-positive BC. 28 Combined analysis of the DB01, DB02, and DB03 studies24,31,32 indicates that T-DXd shows beneficial effects in refractory/stable and untreated/active HER2-positive brain metastasis patients. The ORR for T-DXd in refractory/stable and untreated/active brain metastasis patients was reported as 45.1% and 45.4%, respectively. In this study, the ORR for heavily treated brain metastasis patients was 28.6%, showing considerable effectiveness, and >50% of these patients had low HER2 expression. Among patients diagnosed with HER2-positive BC, it has been evidently observed that in the cohort of those who have undergone no more than 3 lines of previous treatment, their median PFS is capable of reaching up to 10.55 months (95% CI, 6.37–NE). By contrast, for the group of patients who have received more than 3 lines of previous treatment, their median PES is significantly shortened to 3.88 months (95% CI, 1.81–10.18). The same trend has also been shown among BC patients with low HER2 expression. It has been observed that for those patients who have received no more than one line of chemotherapy, their median PFS can reach NR months (95% CI, 1.58–NE), while for those who have received more than one line of chemotherapy, their median PFS is shortened to 10.51 months (95% CI, 1.64–NE). The results suggest that the earlier patients commence treatment with T-DXd, the more likely it is for them to possess a longer median PFS period (Tables S1 and S2). These findings indicate that using T-DXd as the front-line treatment may provide patients with enhanced treatment benefits. This is consistent with the results of the DB01, DB02, and DB03 studies on T-DXd for HER2-positive patients.34 –36 In the real world, first-line treatment and subsequent-line treatments may have different impacts on drug efficacy and patients’ quality of life. First, regarding treatment efficacy, in cancer’s early stage, tumor cells may not be drug resistant. Effective first-line drugs can target tumor cells precisely. But as treatment lines rise, cells may resist prior drugs or patients’ health may decline, reducing drug efficacy. Second, in terms of patients’ survival, effective first-line drugs enhance quality of life. Later-line treatments bring more discomforts like nausea, vomiting, fatigue, Multiorgan Metastasis, and Visceral Crisis due to disease progress and side effect accumulation.
An analysis of previous drug usage showed no cross-resistance between T-DXd and anti-HER2-targeted drugs or ADC drugs. 34 In patients who had progressed after using multiple anti-HER2-targeted drugs or ado-trastuzumab emtansine or disitamab vedotin, T-DXd still demonstrated promising antitumor activity. However, in real-world clinical practice, many patients do not have the opportunity to receive later-line treatments after progression. The use of T-DXd as the front-line treatment may enable patients to achieve optimal benefits.
Safety
Gastrointestinal and hematologic AEs are common side effects of T-DXd. 37 Consistent with the results of previous studies, 38 in the DESTINY-Breast 01 trial, 31 the most common AEs were nausea (63%), neutropenia (63%), decreased appetite (60%), and anemia (58%); interstitial lung disease (ILD) and pneumonitis were observed in 10% of the patients. In DESTINY-Breast 03, 22 drug-related AEs occurred in 98.1% of the 257 patients (252 of 257), and nausea (72.8%), fatigue (44.7%), vomiting (44.0%), and neutropenia (42.8%) were the most common AEs. ILD and pneumonitis were observed in 10.5% of the patients (27 of 257; all <grade 3). The safety analysis in this study showed that the major adverse reactions to T-DXd were nausea in 48 (78.69%), leukopenia in 21 (34.43%), and anorexia in 45 patients (73.77%), and fatigue was also reported. Most of these AEs were grade 1 or 2, whereas severe grade 4/5 AEs were not observed. There was one case of pneumonia (1.64%). For grades 1–2, AEs such as nausea, fatigue, and diarrhea, which are usually transient, only simple symptomatic treatment or observation is required clinically, and they can return to normal without the need for special intervention measures. Generally, they will not lead to the interruption of treatment. However, for grades 4–5, AEs like thrombocytopenia and interstitial pneumonia severely affect the gas exchange function of the lungs, it is necessary to reduce the dosage or adjust the treatment plan and adopt better preventive and therapeutic strategies.
Among the total 61 patients, 17 cases (27.87%) adopted the NK1 + granisetron + dexamethasone regimen for preventive antiemesis, and 29 (47.54%) adopted granisetron + dexamethasone regimen (Table 3). In actual clinical practice, it is essential to observe and follow up on patients, recording their symptoms, and the frequency and severity of vomiting. The combined use of an NK1 receptor antagonist, granisetron, and dexamethasone can effectively prevent nausea and vomiting caused by T-DXd and enhance patients’ tolerance. Antiemetic prophylaxis should be administered by referring to high to moderately emetogenic chemotherapy drugs, and routine laboratory work as well as liver and kidney function assessments need to be made regularly. It is worth mentioning that in this study, only one patient (1.64%) developed pneumonia, which was mild and far lower than the data reported in previous clinical studies. This may be related to the fact that many patients did not receive adequate doses due to poor baseline conditions or economic reasons. Nevertheless, in clinical practice, regular follow-up examinations, early detection, early diagnosis, and early intervention are particularly necessary for the management of ILD.
Limitations
This study had several limitations. First, due to the relatively short duration of the study, the long-term efficacy, safety, and survival outcomes of T-DXd could not be evaluated. In addition, the limited sample size and reliance on descriptive statistical methods may have introduced biases into the results. Moreover, the small sample size might make it impossible to conduct a comprehensive assessment of adverse reactions, though it can still represent the majority of adverse reactions to some extent. During the data collection process, it is necessary to conduct follow-up on patients. Depending on the different conditions of patients, the treatment methods adopted by doctors may vary. A larger sample size and extended follow-up periods are necessary to confirm the research results and provide supporting evidence.
Conclusion
This study was the first multicenter, real-world study of T-DXd for advanced BC in China, which evaluated the efficacy and safety of T-DXd in a more diverse and broader population. The findings demonstrated that T-DXd may be an effective antitumor treatment with controllable adverse reactions in patients with advanced BC irrespective of HER2 expression levels. The frontline treatment of T-DXd is expected to bring more clinical benefits to patients. Preventive triple antiemetics and AE monitoring are particularly necessary to improve patient tolerance and early intervention of severe AEs. The findings support the clinical use of the medication. Multi-center, large-sample clinical application data are necessary to further investigate this issue and provide high-level evidence support.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251318853 – Supplemental material for Efficacy and safety of trastuzumab deruxtecan in Chinese patients with HER2-positive and HER2-low advanced breast cancer: a multicenter, observational, real-world study
Supplemental material, sj-docx-1-tam-10.1177_17588359251318853 for Efficacy and safety of trastuzumab deruxtecan in Chinese patients with HER2-positive and HER2-low advanced breast cancer: a multicenter, observational, real-world study by Die Sang, Yanfang Su, Yurong Zhang, Yanfeng Guan, Shanmin Fan, Jintao Zhang, Lijun Zheng, Yanling Wang, Ying Guo, Zixuan Lei, Man Li and Peng Yuan in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251318853 – Supplemental material for Efficacy and safety of trastuzumab deruxtecan in Chinese patients with HER2-positive and HER2-low advanced breast cancer: a multicenter, observational, real-world study
Supplemental material, sj-docx-2-tam-10.1177_17588359251318853 for Efficacy and safety of trastuzumab deruxtecan in Chinese patients with HER2-positive and HER2-low advanced breast cancer: a multicenter, observational, real-world study by Die Sang, Yanfang Su, Yurong Zhang, Yanfeng Guan, Shanmin Fan, Jintao Zhang, Lijun Zheng, Yanling Wang, Ying Guo, Zixuan Lei, Man Li and Peng Yuan in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
