Abstract
Purpose:
Weekly gemcitabine + paclitaxel (wGT) administration is widely applied in real-world clinical practice. The 28-day and 21-day regimens of wGT are the most widely accepted regimens. We evaluated the efficacy and safety of wGT administration in patients with metastatic breast cancer (MBC) and compared the two regimens.
Methods:
Patients with human epidermal growth factor receptor 2 (HER-2)-negative MBC who received wGT between October 2013 and October 2016 were identified using an electronic database. The outcome variables included progression-free survival (PFS), overall survival (OS), objective response rate (ORR), and safety profile. Propensity score matching was performed to minimize potential confounders.
Results:
A total of 140 patients were included. The median PFS and OS was 7.8 [95% confidence interval (CI) = 7.0–8.7] months and 22.5 (95% CI = 18.8–26.1) months, respectively. The toxicity of wGT was manageable. Among the patients, 90 (64.3%) received the 21-day regimen and 50 (35.7%) received the 28-day regimen. A higher number of younger patients and patients receiving later-line therapy received the 28-day regimen. There was no significant difference between the two groups in PFS after propensity score matching, though subgroup analysis showed that patients with early relapse benefited more from the 28-day regimen. The ORR was numerically higher in 28-day regimen (37.8% versus 28.0%, p = 0.310). However, the 21-day regimen was better tolerated than the 28-day regimen.
Conclusion:
wGT administration showed efficacy and safety in patients with MBC. The efficacy was comparable between the two regimens after adjustment for confounding factors while the 21-day regimen was better tolerated.
Plain Language Summary
Weekly gemcitabine + paclitaxel (wGT) administration is widely applied in real-world clinical practice. The 28-day and 21-day regimens of wGT are the most widely accepted regimens. We evaluated the efficacy and safety of wGT administration in patients with metastatic breast cancer (MBC) and compared the two regimens. Patients with human epidermal growth factor receptor 2 (HER-2)-negative MBC who received wGT between October 2013 and October 2016 were identified using an electronic database. The outcome variables included progression-free survival (PFS), overall survival (OS), objective response rate (ORR), and safety profile. Propensity score matching was performed to minimize potential confounders. We found that the efficacy was comparable between the two regimens after adjustment for confounding factors while the 21-day regimen was better tolerated.
Introduction
Breast cancer is the most common cancer and leading cause of cancer mortality in women worldwide and in China.1,2 Despite the advances in cancer diagnosis and treatment, the recurrence rate of breast cancer is still as high as 20–40%. Metastatic breast cancer (MBC) remains an incurable disease. The median survival from the diagnosis of MBC is 2–3 years. 3
Chemotherapy remains an important treatment for MBC. The recommended cytotoxic drugs in metastatic settings include taxanes [paclitaxel (PTX) and docetaxel], gemcitabine (GEM), vinorelbine, and capecitabine. The combination of GEM and PTX (GT) has been proved to be superior to PTX monotherapy in response rate and overall survival (OS), one of the few studies that showed survival benefit of combination therapy compared to monotherapy in MBC. Thus, the GT regimen is a standard option for patients with MBC who require combination chemotherapy, with the following recommended dosage regimen: GEM (1250 mg/m2, d1 and d8) and PTX (175 mg/m2, d1) every 3 weeks. 3
In addition, in the past decades, strong evidence has been reported on the superiority of the weekly paclitaxel regimen over the standard triweekly regimen for early-stage and metastatic cancers.4–6 Thus, it is reasonable to hypothesize that the weekly GT (wGT) regimen would also show a better performance. Several small-size studies have confirmed the tolerability and efficacy of wGT.7–9 Although similar efficacy was observed between wGT and triweekly GT in phase III trials, wGT is widely used in clinical practice due to its convenience in terms of dose adjustment and patient management.10–12
Research on optimization of the dosage regimen of wGT is ongoing. Two regimens of wGT have been widely accepted and used in the real world after years of practice: the 28-day regimen (GT on days 1, 8, and 15, every 28 days)7,12–15 and the 21-day regimen (GT on days 1 and 8, every 21 days).9–11,16–18 To the best of our knowledge, no study has investigated the difference between the two wGT regimens in terms of efficacy, safety, or patient preference.
Thus, we described and compared the efficacy and safety of different regimens of wGT in patients with MBC for the first time. In order to minimize treatment selection bias and provide evidence that is comparable to that from a randomized controlled trial, propensity score analysis was applied in the analysis of a real-world dataset from China. 19
Methods
Patients and treatment
Patients with human epidermal growth factor receptor (HER2)-negative MBC who received wGT at Fudan University Shanghai Cancer Center between October 2013 and October 2016 were retrospectively identified using the center’s electronic database. Demographic and clinicopathological data were extracted from the patients’ electronic medical records. Patients without complete treatment history or detailed dosing records were excluded. All the procedures performed in the study were in accordance with the ethical standards of the institutional research committee (approval number, 1812195-6) and the 1964 Helsinki declaration and its later amendments. The need for written informed consent was waived due to the study’s retrospective nature. The following dosage regimen is used for wGT in our center: PTX 80 mg/m2 plus GEM 800 mg/m2 administered on days 1, 8, and 15 every 28 days (28-day regimen) and PTX 80 mg/m2 plus GEM 1000 mg/m2 administered on days 1 and 8 every 21 days (21-day regimen). In both regimens, PTX was administered intravenously over a period of 1 h before GEM administration (usually administered over 30 min). All patients were pretreated with dexamethasone, cimetidine, and promethazine. Treatment was stopped in case of disease progression, intolerable toxicity, or patient request.
Dose-related parameters
Dose intensity (mg/m2/w) was defined as the cumulative dose divided by the body surface area and duration of chemotherapy exposure. The relative dose intensity (RDI) was calculated as the ratio of the actual dose intensity to the planned dose intensity, expressed as a percentage value. The RDI of the wGT regimen was determined by averaging RDIs of the individual agents.
In the 28-day regimen, patients are treated for three consecutive weeks out of four, at a dose intensity of 60 mg/m2/w for PTX and 600 mg/m2/w for GEM. A relatively lower dose of PTX (53.3 mg/m2/w) and higher dose of GEM (666.7 mg/m2/w) are administered in the 21-day regimen.
Efficacy and safety
The efficacy endpoints included progression-free survival (PFS), OS, and objective response rate (ORR). PFS was defined from the date of wGT initiation until documented disease progression or death. OS was calculated from the date of treatment initiation to death from any cause or the last follow-up. ORR was defined as the percentage of patients showing a complete response and partial response (PR). Tumor responses were re-confirmed by the authors according to the Response Evaluation Criteria in Solid Tumors 1.1
Adverse events (AEs) were determined retrospectively based on the patients’ medical records and laboratory test results. Patients who did not undergo a post-treatment safety evaluation were excluded from the safety analysis population. AEs were assessed according to the National Cancer Institute Common Terminology Criteria for Adverse Events version 4.0.
Statistical analysis
Descriptive analysis was performed to summarize the patients’ demographic and clinicopathological characteristics. Quantitative data are presented as the median (range) or number of patients (percentage). The Kaplan–Meier method was used to estimate the median PFS and OS, and the log-rank test was used to compare them. Predictive factors for PFS were determined using univariate and multivariate Cox regression analyses. Fisher’s exact test or the chi-square test was used to compare independent categorical variables; the t test was used for comparing continuous variables.
To create a randomized clinical trial-like setting, propensity score matching (PSM) was performed using the one-to-one nearest neighbor matching method without replacement. Variables that were independent predictors of the treatment outcomes in multivariate Cox regression analyses or were significantly different between the two groups were used for matching. Variables were dichotomized when possible to optimize the matching algorithm.
Tumor response analyses included all patients with evaluable disease. Safety analysis included patients who underwent a post-treatment safety evaluation after receiving wGT. AEs were summarized using percentages and frequency counts. Statistical analyses were conducted using SPSS IBM® version 22 (SPSS Inc., Chicago, IL) and Software R. All p values were two sided, and the significance level was set at p < 0.05.
Results
Patient and treatment characteristics
A total of 140 patients were included in this analysis. Patient and treatment characteristics are presented in Table 1. The median age was 55 (28–72) years. Half of the patients had ⩾3 metastatic sites (53.6%), and 78.6% of patients showed visceral involvement.
Patient and treatment characteristics.
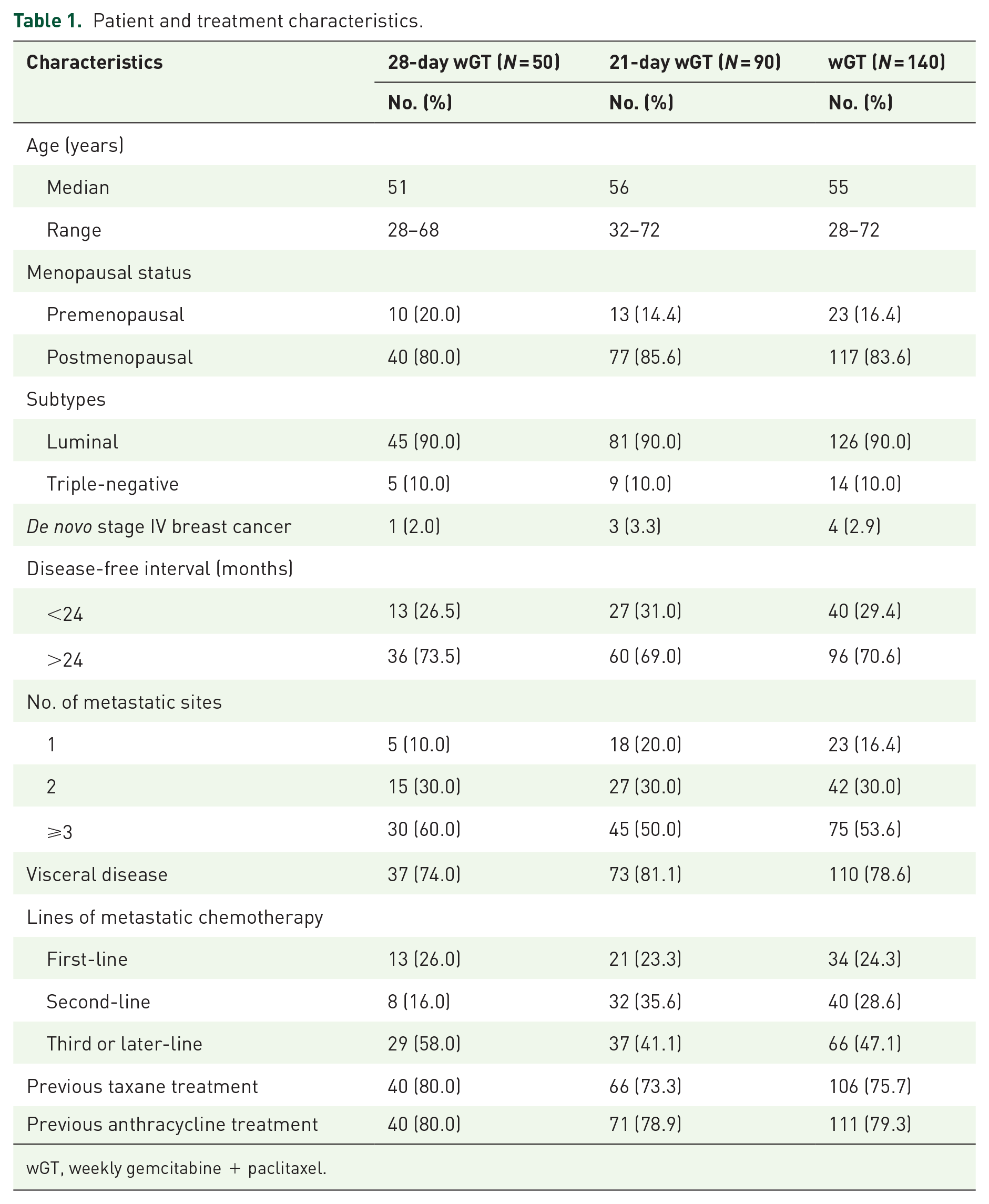
wGT, weekly gemcitabine + paclitaxel.
Thirty-three (26.2%) of 126 patients with hormone receptor–positive breast cancer showed progression during the first 2 years of adjuvant endocrine therapy and were considered to be primary resistant to endocrine therapy. Eighty patients had progressed on endocrine therapy in metastatic setting, and 62 showed treatment failure after receiving at least two regimens of endocrine therapy before the current regimen.
A total of 761 cycles of wGT were delivered, with a median follow-up of 19.0 (1.6–77.0) months; each patient received a median of 6 (1–16) cycles. The median RDI was 88.7% (45.8–107.6%). Reasons for treatment discontinuation included treatment completion (60, 42.9%), disease progression (51, 36.4%), intolerable toxicity (19, 13.6%), patient request (7, 5.0%), and loss to follow-up (3, 2.1%). The proportion of patients who discontinued treatment due to intolerable toxicity was greater in the 28-day regimen group than in the 21-day regimen group (18.0% versus 11.1%).
Among 89 patients who discontinued treatment before disease progression, 70 patients (78.7%) continued receiving maintenance therapy. Maintenance therapies included endocrine therapy (55.7%), oral chemotherapies (30.0%; e.g. capecitabine chemotherapy), and GEM or PTX monotherapy (14.3%).
Treatment outcomes in all eligible patients
At the time of analysis, 128 patients (91.4%) showed disease progression, and 81 patients had died (57.9%). The median PFS was 7.8 [95% confidence interval (CI) = 7.0–8.7] months. The median OS was 22.5 (95% CI = 18.8–26.2) months.
Among 131 patients with evaluable disease, 48 patients achieved PR (36.6%) and 66 patients (50.4.0%) had stable disease. This resulted in an ORR of 36.6%.
The predictive factors for PFS are shown in Table 2. Visceral disease [hazard ratio (HR) = 2.37, 95% CI = 1.47–3.81, p < 0.001], later-line of chemotherapy (HR = 1.22; 95% CI = 1.05–1.40, p = 0.007), and previous exposure to taxanes (HR = 1.74; 95% CI = 1.15–2.65, p = 0.009) were independent predictive factors of worse treatment outcomes.
Predictive factors for PFS on univariable and multivariable analyses.

p< 0.05.
CI, confidence interval; HR, hazard ratio; PFS, progression-free survival.
Toxicity profiles of the patients
A total of 135 patients were evaluated for toxicity (Table 3). The hematological side effects included leukopenia (64.4%), neutropenia (57.0%), anemia (32.6%), and thrombocytopenia (17.8%). Most of the treatment-related toxicities were mild. Neutropenia was the most frequent grade 3/4 AE (26.6%), followed by leukopenia (16.3%). Four patients had febrile neutropenia (3.0%). Peripheral neuropathy was the most common non-hematological AE (29.6%). 4.4% of patients had grade 3/4 neuropathy. Hypersensitivity reactions were observed in five patients (3.7%) with standard premedication. No treatment-related death was documented during drug administration.
Adverse events in patients based on safety analysis (N = 135).

The incidence of all-grade AEs, except rash, was higher in the 28-day regimen group than in the 21-day regimen group. Grade 3/4 leukopenia, neutropenia, thrombocytopenia, peripheral neuropathy, and hypersensitivity reactions occurred at a greater frequency in the 28-day regimen group, while grade 3/4 anemia and fatigue were more frequent in the 21-day regimen group.
Patient characteristics before and after PSM
Ninety (64.3%) of 140 patients received the 21-day regimen, while 50 patients (35.7%) received the 28-day regimen. A comparison of patient characteristics between the two groups is shown in Table 4. Patients who were younger or were receiving later-line chemotherapy in metastatic settings received the 28-day regimen more commonly than the 21-day regimen. Age, visceral involvement, previous taxane treatment, and lines of chemotherapy were selected for propensity score estimation. Fifty matched pairs were generated by PSM. There was no significant difference in the variables between the groups after PSM.
Comparison of patient characteristics between the two regimen groups.
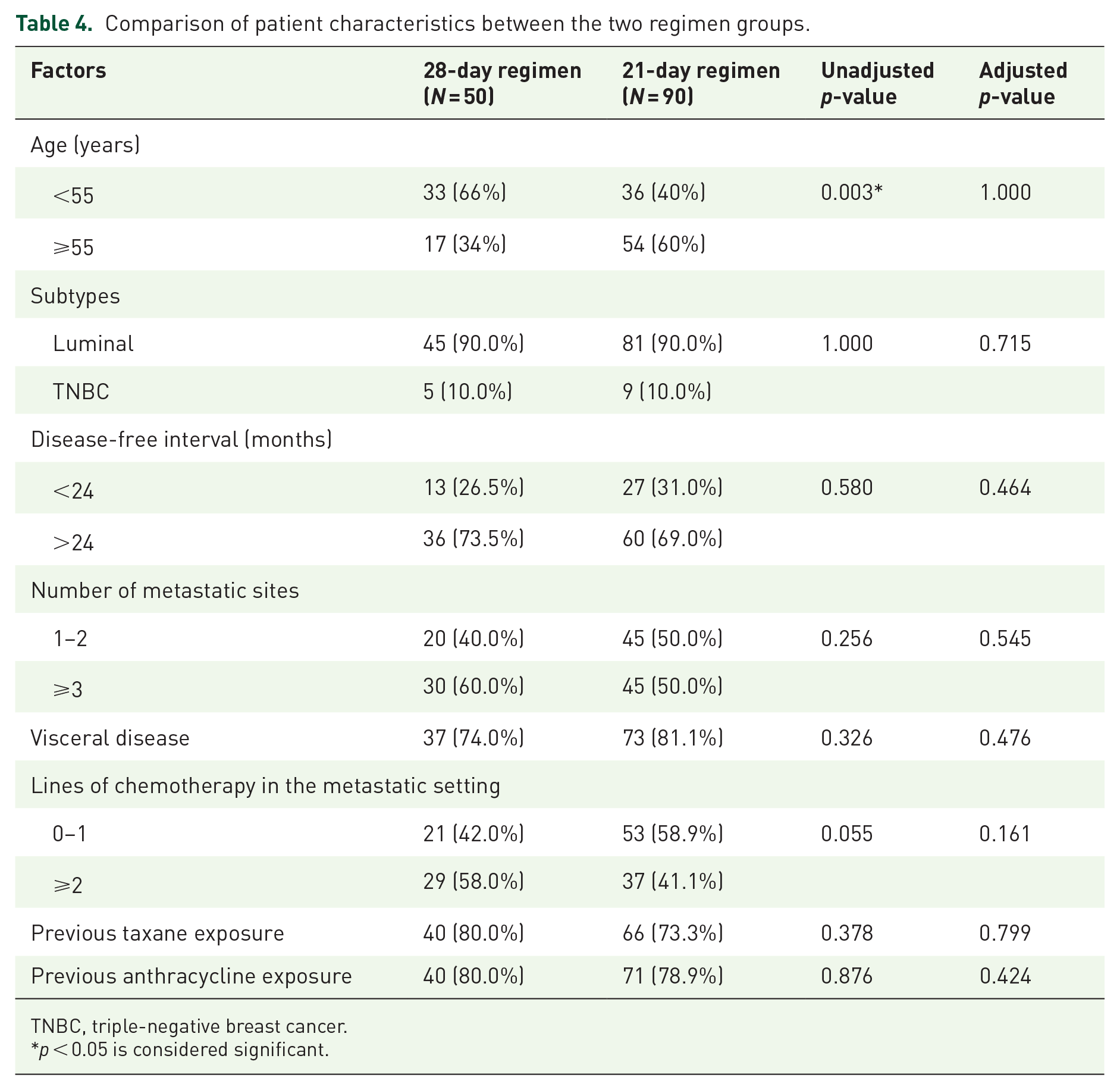
TNBC, triple-negative breast cancer.
p < 0.05 is considered significant.
Treatment outcomes of matched patients
After matching, the median number of treatment cycles was similar in the two groups (6 versus 6 cycles). However, the total number of treatment cycles and median RDI of regimen patients received were higher in the 21-day regimen group than in the 28-day regimen group (308 versus 224 cycles, p = 0.001; RDI 94.3% versus 83.4%, p = 0.005). Besides, fewer patients discontinued treatment due to intolerable toxicity in the 21-day group than in the 28-day group (8% versus 18%). The median RDI values of GEM and PTX in the 21-day group were 91.3% and 94.1%, respectively. The median RDI values of GEM and PTX in the 28-day regimen group were 83.1% and 86.3%, respectively.
There was no significant difference in treatment outcomes between the groups. The median PFS in the 21-day and 28-day regimen groups were 7.4 and 8.3 months, respectively (p = 0.437; Figure 1). The results remained consistent in Cox analysis (HR = 0.85; 95% CI = 0.564–1.281, p = 0.438) and in subgroup analysis; however, patients with early relapse seemed to benefit more from the 28-day regimen (Figure 2). The median OS durations in the two groups were 23.5 and 23.1 months, respectively (p = 0.848). In patients with evaluable disease, the ORRs in the 21-day and 28-day regimen groups were 28.0% and 37.8%, respectively (p = 0.310).
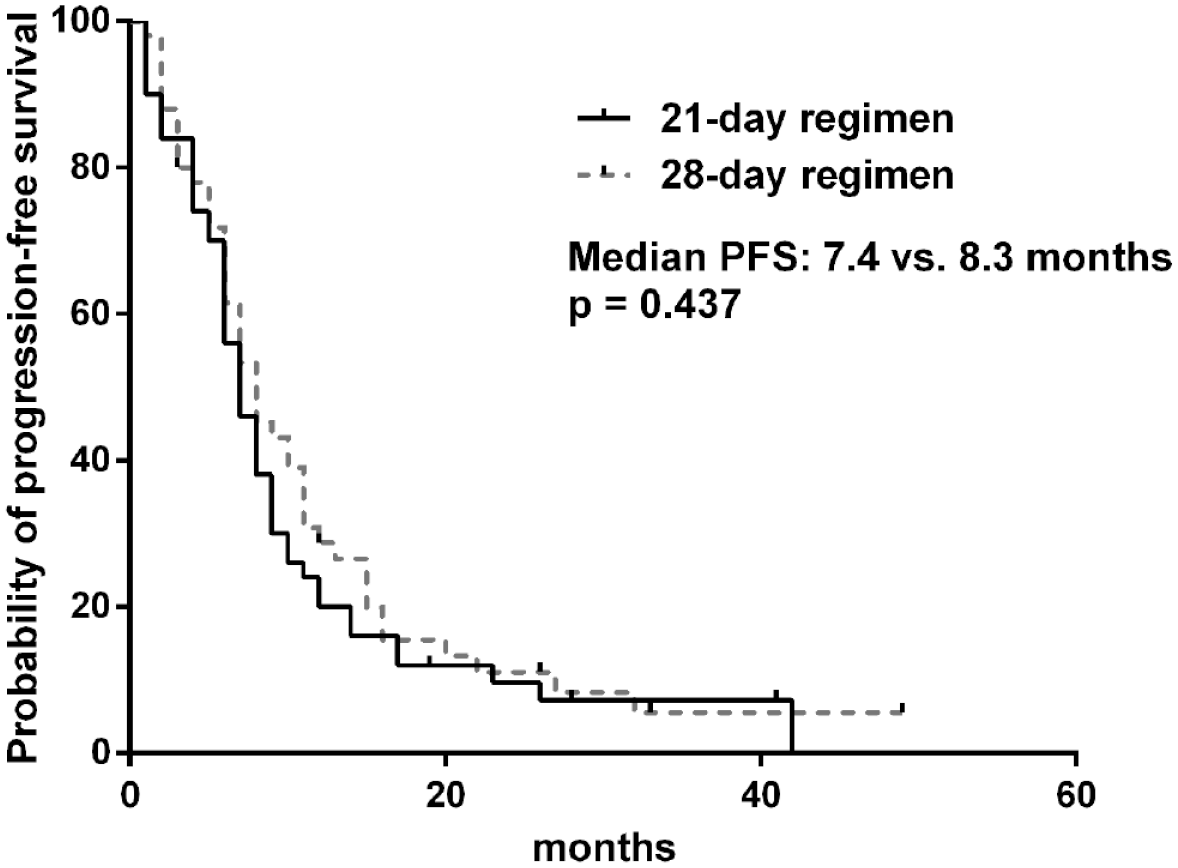
Kaplan–Meier survival curves for PFS by regimen group in propensity score–matched dataset (N = 100).

Forest plot of PFS in propensity score–matched dataset (N = 140).
Discussion
The aim of this study was to describe the efficacy and safety of wGT in patients with MBC and to compare two regimens of wGT via propensity score analysis using a real-world dataset from China. In this study, a median PFS of 7.8 months, median OS of 22.5 months, and acceptable toxicity confirmed the efficacy and safety of wGT in Chinese patients with MBC. Treatment outcomes (PFS, OS, ORR) were comparable between the two regimens of wGT after adjustment for confounding factors. In terms of safety, the 21-day regimen was better tolerated.
In a phase III study on triweekly GT administration in the first-line treatment, the median time to progression and OS were 6.14 and 18.6 months, respectively. 3 Regardless of the differences between studies and worse patient characteristics in this study, wGT has a much better efficacy than the standard triweekly regimen. Despite administration regimens, another noted discrepancy between this randomized trial and our study was in terms of the use of maintenance therapy. The role of maintenance therapy in MBC patients is still controversial: the PFS and response duration were prolonged; however, there were conflicting results regarding OS; the quality of life was found to have reduced.20–23 Outside clinical trials, the longer the intravenous chemotherapy courses, the worse the patients’ compliance. Thus, maintenance therapy has been widely administered in daily clinical practice in China to achieve longer disease control. Oral chemotherapy (e.g. capecitabine) and endocrine therapy are common options for maintenance treatments. The frequent use of maintenance therapy in real-world settings may explain the longer PFS in this study.
Previous taxane exposure was an independent predictive factor for worse treatment outcomes after adjustment for other prognostic factors in the multivariate Cox model. This indicated that despite the synergistic effect of GEM and PTX, prior taxane exposure can still compromise the efficacy of PTX, which could be partially explained by the incomplete cross-resistance between different taxane regimens.24,25
In terms of safety, the wGT regimen was well tolerated; the toxicity profile was consistent with those in previous studies. Most of the treatment-related toxicities were mild. Neutropenia was the most frequent grade 3/4 AE (26.6%); the frequency was lower than that in a phase II study of triweekly GT in Chinese patients (45%) and in the AERO trial (67%), a phase II study of weekly GT in first-line setting.8,26 Peripheral neuropathy was the most common non-hematological AE (29.6%); the frequency was lower than that in a study involving consecutive weekly PTX administration plus a 21-day GEM regimen (38%). 27 One reason for the satisfactory safety profile in this study was that lower doses of GEM and PTX were administered.
Two different regimens of wGT were compared in this study. We found that the 21-day regimen has been used more commonly than the 28-day regimen in the last 3 years in our institution (64.2% versus 35.7%). However, patients with potentially unfavorable characteristics (such as younger age and later-lines of chemotherapy in metastatic settings) received the 28-day regimen more frequently than the 21-day regimen. Taxanes are known to be the most effective agents against MBC. 28 Since the dose intensity of PTX was relatively higher in the 28-day regimen, it is possible that oncologists chose this regimen for patients with a higher tumor burden to achieve better disease control.
PSM was performed to balance patient characteristics between the groups. In the matched dataset with well-balanced patient and treatment characteristics, no significant difference was observed in treatment outcomes between the two groups. Exploratory analysis showed that patients with early relapse (a disease-free interval of <24 months) may benefit more from the 28-day regimen.
In terms of safety, the 21-day regimen was better tolerated than the 28-day regimen. The number of total treatment cycles and median RDI were higher in the 21-day regimen group than in the 28-day regimen group. After calculation, results showed that the median actual dose intensities of GEM were 608.7 and 498.6 mg/m2/w in the 21-day and 28-day regimens, respectively, while the median dose intensities of PTX were 50.2 and 51.8 mg/m2/w, respectively. Although there is no available head-to head comparison between the two groups, in a clinical trial, a 28-day regimen of wGT (PTX 100 mg/m2 + GEM 1000 mg/m2, days 1, 8, and 15; q28d) had to be terminated and changed into a 21-day regimen due to the intolerable toxicity in a clinical trial. 29
Although sequential monotherapy has been recommended by the National Comprehensive Cancer Network and Advanced Breast Cancer (ABC) 5 guidelines over combination chemotherapy for MBC in the last 2 years, our study findings are still valuable. First of all, combination chemotherapy is still important in the treatment of patients with visceral crisis or high tumor burden. Besides, doublet chemotherapy with confirmed efficacy and manageable toxicity is recommended by the Chinese Anti-Cancer Association Committee of the Breast Cancer Society (CBCS). A recently published article compared the ABC 4 and CBCS guidelines and analyzed the reasons underlying their differences, including differences in the preference for combination chemotherapy. 30 One of the main reasons is that Chinese patients are willing to bear additional AEs in pursuit of better efficacy. Rapid tumor shrinkage or symptom relief with combination chemotherapy will improve patient compliance. Due to these cultural reasons, wGT is widely used in real-world practice in China. The results of this study will be helpful to Chinese oncologists in clinical practice.
Our study provides the largest amount of data on the efficacy and safety of wGT in patients with MBC and is the first real-world study of wGT in a Chinese population. The identified predictors of worse outcomes could provide more clues for patient selection. To the best of our knowledge, this is also the first study to focus on different regimens of wGT. Another strength of this study was the application of PSM to minimize confounding and indication bias. Furthermore, to provide additional information, the RDI of each agent was calculated and compared in our study.
As a retrospective study, our study was subject to limitations including missing data and potential bias. Even though PSM simulates randomization, it can only correct for known confounders and cannot be a substitute for a randomized clinical trial. Besides, since non-hematological AEs were retrospectively extracted from previous medical records, their incidences were likely to be underestimated. Additionally, most patients in this study were evaluated every two cycles (6 and 8 weeks); this difference in evaluation frequency may also influence PFS time. Furthermore, quality of life, which could differ according to the regimens, was not routinely evaluated or recorded in our database. A prospective study conducted in a larger population focusing on patient’s quality of life could provide more comprehensive information in the future.
Conclusion
This study involved a comprehensive analysis of real-world data of patients with MBC treated with wGT. The wGT regimen was highly effective and tolerable in Chinese patients, even in heavily pretreated patients. After adjustment for confounding factors, the efficacy of the 21-day and 28-day regimens was found to be comparable, though the ORR was numerically higher in 28-day regimen. The 21-day regimen was better tolerated. This study provided valuable information on patient characteristics and treatment outcomes outside of clinical trials and offered useful clues for oncologist in making clinical treatment decisions: in patients with early relapse, higher tumor burden, and better tolerance, 28-day regimen might be a better option; however, in patients who could not bear or have concerns over toxicities, 21-day regimen is a more appropriate recommendation.
