Abstract
Introduction:
The decision to deprescribe medications used for both disease prevention and symptom control (dual-purpose medications or DPMs) is often challenging for clinicians. We aim to establish the impact of deprescribing DPMs on patient-related outcomes for older adults near end-of-life (EOL).
Methods:
This systematic review was conducted according to the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guideline. Literature was searched on PubMed, EMBASE, CINAHL, PsycINFO and Google Scholar until December 2019 for studies on deprescribing intervention with a control group (with or without randomisation); targeting ⩾65-year olds, at EOL, with at least one life-limiting illness and at least one potentially inappropriate DPM. We were interested in any patient-related outcomes. Studies with similar outcome assessment criteria were subjected to meta-analysis and narrative synthesis otherwise. The risk of bias was assessed using Cochrane Risk of Bias and ROBINS-I tools for randomised controlled trials (RCTs) and quasi-experimental non-randomised controlled studies, respectively.
Results:
Five studies covering 689 participants with mean age 81.6–85.7 years, the majority (74.6–100%) with dementia were included. The risk of bias was moderate to low. The deprescribing of DPMs lowered the risk of mortality (risk ratio (RR) = 0.59, 95% confidence interval (CI) = 0.44–0.79) and referral to acute care facilities (RR = 0.40, 95% CI = 0.22–0.73), but did not have a significant impact on the risk of falls, non-vertebral fracture, emergency presentation, unplanned hospital admission, or general practitioner visits. No significant difference was observed in the quality of life, physical and cognitive functions between the intervention and control groups.
Conclusion:
There is some evidence that deprescribing of DPMs for older adults near the EOL can lower the risk of mortality and referral to acute care facilities, but there are insufficient good-quality studies powered to confirm a benefit in terms of quality of life, physical or cognitive function, health service utilisation and adverse events.
Plain Language Summary
Keywords
Introduction
Older adults with life-limiting illnesses (LLIs) and limited life expectancy (LLE) frequently receive potentially inappropriate medications (PIMs),1,2 even in the last few months of their life.3,4 PIMs are often associated with poor health outcomes such as the increased risk of falls, rate of hospitalisation, the incidence of adverse drug events and risk of mortality while the decreasing quality of life (QOL).1,2,5,6 Older adults with LLIs are more vulnerable to poor outcomes, primarily due to alteration in pharmacology, as a result of a change in the body physiology 7 and frailty-associated cachexia.8,9 Medication that was once beneficial for disease prevention could be less relevant, and it may be more appropriate to taper or withdraw such PIMs under the supervision of healthcare professionals, referred to as deprescribing. 10
It is commonly acknowledged that the use of preventive medications for long-term benefits in older adults at the end-of-life (EOL) is inappropriate.1,2,11 For instance, statins are beneficial with at least two years of continuous use, but for short-term use, the risk of adverse drug events outweighs potential benefits.12,13 It is also known that people with LLIs and LLE often become clinically unstable at the terminal phase and symptoms could emerge at any time. 14 At this point, proactive pharmacological management using symptom-control medicines becomes indispensable. But some medications can have dual-purpose, that is, they could be used for both disease prevention and symptom control. These medications are referred to in this review as dual-purpose medications (DPMs). For example, hypoglycemic agents can be used for both short-term and long-term risks of hyperglycemia, 1 anti-psychotics can be used for the symptom management of delirium at the EOL15,16 as well as in the treatment of behavioural and psychiatric symptoms of dementia (BPSD), 17 diuretics are commonly used for the symptom management of dyspnoea due to pulmonary oedema at the EOL18,19 and also for the secondary prevention of chronic heart failure. 20 Deprescribing such medications in the clinical setting can be complex and challenging to consider due to the patient resistance to discontinuation combined with multiple specialists having oversight for the care of patients with multiple co-morbidities. 21 Moreover, clinician’s decisions on deprescribing can be complicated by uncertainty around disease prognosis with the changing goal of care towards the EOL.22,23 Evidence from a recent systematic review suggests that deprescribing preventive medications at the EOL improves medication appropriateness and has potential for improved health outcomes, 24 but whether deprescribing DPMs improve patient outcomes is unclear. This review aimed to fill that knowledge gap by exploring the impact of deprescribing potentially inappropriate DPMs on patient-related outcomes for older adults with LLIs and LLE to assist clinicians’ decisions.
Methods
The reporting of this study was per the Preferred Reporting Items for Systematic Reviews and Meta-Analyses statement. The protocol was registered in the Open Science Framework (https://osf.io/ur73q/). 25 The definitions of key terms used in this review are provided below.
Deprescribing: Tapering or withdrawal or discontinuation of an inappropriate medication, supervised by a healthcare professional, with the goal of reducing or managing polypharmacy and improving patient outcomes. 10
PIMs: Medicine(s) or medication class(es) that should generally be avoided in ⩾65 years either due to their ineffectiveness or unnecessarily high risk for an older person or when a safe alternative is available for a similar disease condition. PIM identified with any explicit and implicit criteria was included in this study.24,26
DPMs: Medications used for both prevention and symptom control identified in older adults with LLE. A list of DPMs generally known to be used in older adults with LLE has been obtained from previous studies.1,27–29
LLI: Terminal illness that limits life expectancy, such as the advanced or end-stage condition of cancer, dementia, heart failure, chronic obstructive pulmonary disease and kidney disease.
EOL: EOL is a period preceding an individual’s natural death from a process that is unlikely to be arrested by medical care, with LLE. A patient with at least one LLI with an irreversible decline and life expectancy of less than or equal to 12 months was considered to be at EOL in this review. However, studies using terms such as terminally ill, terminal illness, under palliative care or frail were also considered as being EOL-relevant when the life expectancy was not specified. EOL was considered based on Criteria for Screening and Triaging to Appropriate aLternative care (CriSTAL or CrisTAL criteria) 30 if the target population’s mean age was ⩾80 years and met at least two criteria, or if the target population’s mean age was ⩾65 years and met at least four criteria, these participants were deemed to be at the EOL.
Patient-related outcomes: Our inclusion criteria considered patient-related outcomes as any clinical or patient-reported measures, such as QOL, short-term mortality, falls, hospital referral or admission, emergency presentations, fractures, or medication-related adverse effects within 12 months.
Impact: Any change in the estimate of a primary outcome (in any unit) following the intervention was defined as impact.
Study selection
PubMed, Embase, Cumulative Index to Nursing and Allied Health Literature, PsycINFO and Google Scholar were searched for eligible articles from inception to December 2019 in English. Keywords and controlled vocabulary were used with appropriate Boolean logics, synonyms, and limiters (Supplementary Table S1 for literature search strategy for more details). The bibliographies of relevant reviews were manually assessed for additional references. Duplicate articles were located and removed in Endnote software, followed by Covidence software and then manually. The target population was patients in any setting with an average age of ⩾65 years, at the EOL due to any LLI, prescribed with at least one potentially inappropriate DPM (Supplementary Table S2 for list of DPMs) and receiving any type of strategy to deprescribe such medications. For articles potentially eligible on the title and abstract but not specifying EOL or equivalent keywords or life expectancy, the full texts were assessed for the EOL definition using a validated tool called Criteria for Screening and Triaging to Appropriate aLternative care (CriSTAL criteria) for high risk of short-term death. 30 Any controlled intervention (tool or guideline) targeted at deprescribing in older patients at home, pharmacy, clinics, nursing homes, residential aged care facilities (RACFs), and hospitals was included. Articles not reporting on any patient-related outcomes or where it was unclear whether patients were at the EOL were excluded. Conference proceedings, review articles, unpublished literature, before-after interventions and any ongoing studies were excluded. Two authors (SS and AP) independently performed the initial screening of the title and abstract followed by full-text assessment. Any disagreements were resolved with a consensus involving additional evaluation by the other authors.
Data extraction
A purpose-built data extraction form was used to collect data on design, demographics, life expectancy, LLIs, settings, sample size, follow-up period, PIM assessment tools, potentially inappropriate DPMs targeted/deprescribed and their therapeutic categories, deprescribing interventions and intervention providers, comparator and outcomes. Corresponding authors of eligible publications were approached for missing or ambiguous information, where necessary. Two authors (SS and AP) independently conducted data extraction and agreed on consensus basis.
Risk of bias assessment
Cochrane risk of bias tool and Risk Of Bias In Non-randomised Studies of Interventions (ROBINS-I) were used by two authors (SS and AP) independently to assess the risk of bias for randomised controlled trials (RCTs) and quasi-experimental non-RCTs, respectively.
Data synthesis and analysis
A narrative synthesis of data was conducted for all outcome measures with a calculated 95% confidence interval (CI) around the published estimates. Meta-analyses of selected homogeneous outcome measures used the random-effects model to pool the risk ratios (RRs) and 95% CIs using RStudio version 1.3.959 (PBC, Boston MA).
Ethics statement
Our study did not require an ethical board approval because we reviewed previously published studies that are publicly accessible, and each of the included studies stated that an ethics approval was obtained.
Results
A total of 10,826 studies (PubMed: 4381, Embase: 2143, CINHAL: 1140, PsycINFO: 3055, Google Scholar: 105, Other sources: 2) were identified and after removal of duplicates, 9247 studies were screened for the title and abstract, leading to 56 for full-text eligibility; 5 met the criteria for inclusion (Figure 1).

PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) flow diagram of the screening process.
Five studies31–35 conducted in the United Kingdom,
31
Belgium,
32
Israel,
33
Australia,
34
and Norway
35
were included in this systematic review. There were four RCTs31,32,34,35 and one quasi-experimental study
33
involving 689 participants (mean age: 81.6–85.7 years) with 3–12 months follow-up and conducted in a hospital setting (
Deprescribing interventions and intervention providers
The deprescribing interventions were either active deprescribing33–35 or indirect deprescribing (recommendation),31,32 but there was either withdrawal, cessation, tapering or discontinuation of medication after the intervention (Table 1). The intervention providers were either physician/geriatrician31–33 or multidisciplinary team. 34
Characteristics of study participants and deprescribing interventions.
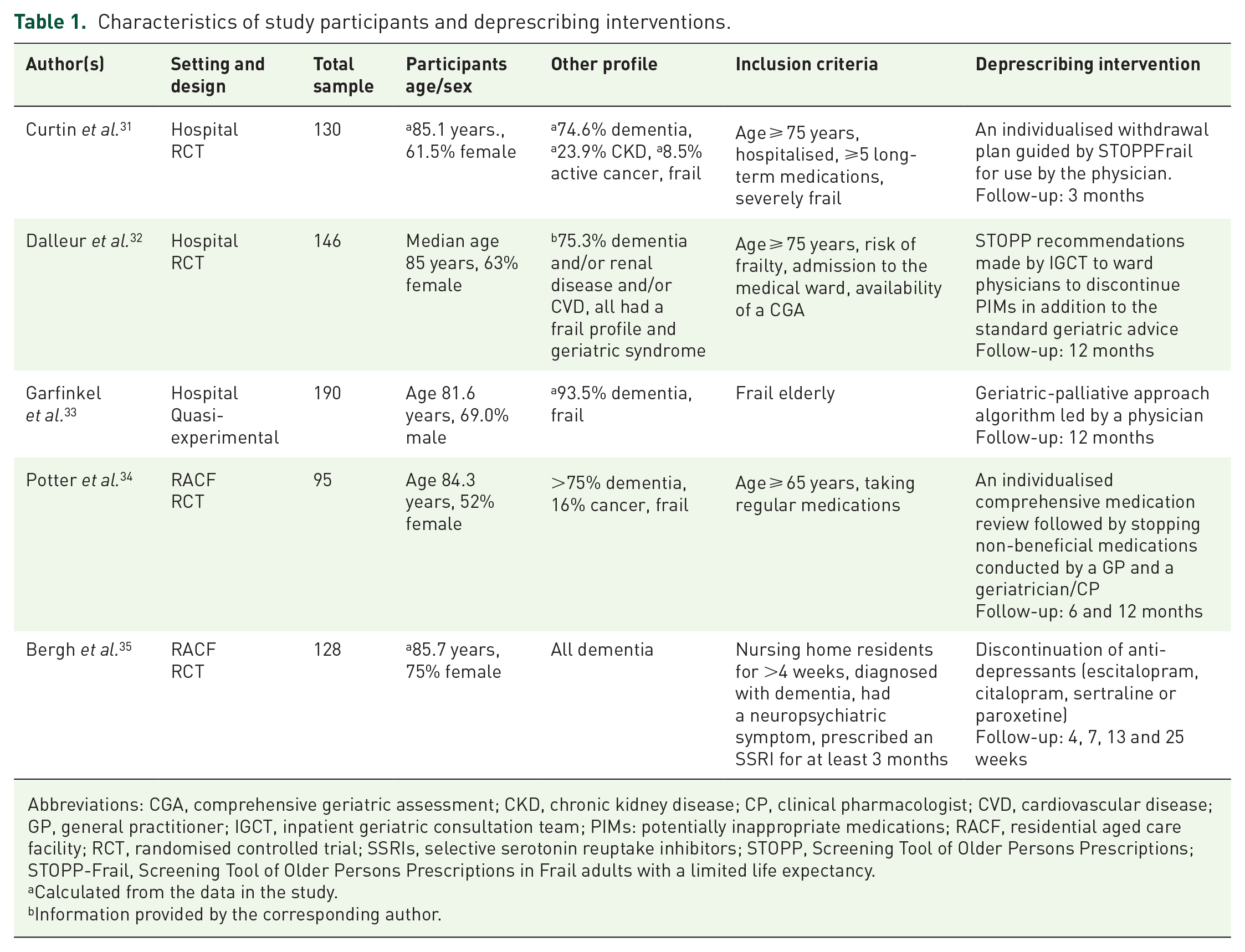
Abbreviations: CGA, comprehensive geriatric assessment; CKD, chronic kidney disease; CP, clinical pharmacologist; CVD, cardiovascular disease; GP, general practitioner; IGCT, inpatient geriatric consultation team; PIMs: potentially inappropriate medications; RACF, residential aged care facility; RCT, randomised controlled trial; SSRIs, selective serotonin reuptake inhibitors; STOPP, Screening Tool of Older Persons Prescriptions; STOPP-Frail, Screening Tool of Older Persons Prescriptions in Frail adults with a limited life expectancy.
Calculated from the data in the study.
Information provided by the corresponding author.
The three studies which involved an active deprescribing intervention are described below.
Identification of medications suitable for dose reduction or discontinuation using a geriatric-palliative criterion (algorithm), led by a physician. 33 The algorithm considered risk-benefit evaluation based on indication, dosing rate, age, frailty and adverse effects on an evidence-based consensus to either stop, change, reduce the dose or continue the medication.
An individualised medication review followed by a planned cessation of non-beneficial medications by a general practitioner (GP) and a geriatrician/clinical pharmacologist. 34 The medication review identified target medications for withdrawal that included inappropriate medications, medications with no symptom benefits but with possible or potential adverse drug withdrawal event (ADWE) and medications with symptomatic benefit (symptom being stable). The target medications were tested against deprescribing criteria (algorithm) using baseline data.
Tapering off and replacement of escitalopram, citalopram, sertraline and paroxetine (selective serotonin reuptake inhibitors or SSRIs) by a placebo over the first week after baseline assessment. 35 Information on the intervention provider was not provided but the intervention was assigned randomly.
The two studies that followed an indirect deprescribing intervention are discussed below.
An individualised withdrawal plan for a pre-determined medication with possible ADWE and unlikely ADWE, guided by STOPPFrail criteria was formulated by a research physician to make a recommendation to the attending physician. Also, a restarting plan was provided. 31 The recommendation was documented in the patient medical record and communicated to the attending physician, who decided whether or not to implement the recommended plan and changes.
Oral and written recommendations made to ward physicians by two geriatricians to discontinue PIMs identified by systematically screening the list of medications taken by patients on admission through the use of 64 STOPP criteria. 32
Comparator
The comparators in all the studies were the control groups, which received one of the following:
Usual pharmaceutical care, that is, hospital physician and pharmacist care. 31 This study used an individualised withdrawal plan guided by the STOPPFrail criteria in the intervention group.
Usual care but were visited as frequently as the intervention group. On each visit, their blood pressure was measured and significant concerns were reported to nursing staff for appropriate follow-up. 34 This study used an individualised medication review followed by a planned cessation of non-beneficial medications in the intervention arm.
No placebo but continued with the same anti-depressant. 35 The group that received placebo (intervention) had their SSRIs tapered off and replaced by a placebo over the first week after baseline assessment.
Standard care from inpatient geriatric consultation team involving two geriatricians without previous experience with STOPP criteria. 32 The intervention group had two geriatricians who used 64 STOPP criteria to deprescribe PIMs.
No changes in medication but hospitalised in the same departments and treated by the same team. 33 The study used a geriatric-palliative algorithm to deprescribe in the intervention group.
The components of “usual care” or “standard care” were not generally described in the studies.
Targeted and deprescribed DPMs
Overall, the included studies targeted 404 DPMs for deprescribing (Table 2). These were primarily central nervous system medications (anti-depressants,
Targeted and successfully deprescribed dual-purpose medications.

Abbreviations: PPIs, Proton Pump Inhibitors; S, successful deprescribing percentage; SSRIs, selective serotonin reuptake inhibitors; T, targeted number of dual-purpose medications.
Average successful deprescribing of all the targeted medications that included medications other than dual-purpose medications.
Only targeted SSRIs (escitalopram, citalopram, sertraline and paroxetine).
The following sections detail the impacts of the deprescribing intervention on QOL, mortality, adverse events and health service utilisation.
Patient-related outcome measures
QOL: Three RCTs reported on QOL (Table 3). One study
31
assessed QOL through scores derived from the QUALIDEM (QOL instrument for proxy completion) instrument at 3 months. This study showed that the baseline score (Intervention group (I): 6.96, Control group (C): 7.58) declined at 3 months (I: –2.43, C: –2.85) but there was no significant difference between the two groups (
Physical and cognitive functioning: Two studies34,35 reported on the functional status and found no significant differences in either the physical or cognitive functions between the intervention and control groups at 6.25
35
and 12 months
34
(Table 3). For assessing physical function, the Lawton and Body’s Physical Self-Maintenance Scale score (I: 18.5
Mortality: The four studies reporting on mortality31–34 showed that it varied greatly (absolute risk difference of 0.5
32
–24.0%
33
at 12 months) through 9.2% at 3 months between intervention and control groups. However, only the largest study with a 12-month follow-up reported that the difference was statistically significant (
Falls: Three studies31,34,35 reported the adverse outcome of falls. A meta-analysis of the overall impact of deprescribing on falls based on two studies (Figure 2 and Table 3) showed no significant reduction in the risk of falls at 3 or 12 months after the intervention (RR, 0.87; 95% CI, 0.65–1.17). The third study
35
reported that the mean ± SD change in the number of falls per day in 21 days was similar (I: 0.01 ± 0.02
Non-vertebral fractures: This adverse event was reported by two studies31,34. The overall impact of deprescribing on non-vertebral fractures (Figure 2 and Table 3) showed that the risk of this adverse event was not reduced significantly after the intervention either at 3 or 12 months (RR: 0.67; 95% CI: 0.10–4.56).
Unplanned hospital admission: Two studies reported on this outcome at 3 months 31 and 12 months 34 and found that the overall impact of deprescribing (Figure 2 and Table 3) showed the risk of unplanned hospital admission was not different between the intervention group compared to the control group (RR: 1.12; 95% CI: 0.74–1.69).
Emergency presentation: One study that reported on emergency presentations at 3 months 31 found that the risk of this event in the intervention group was not significantly different from that of the control group (RR: 0.60; 95% CI: 0.15–2.41; Table 3).
GP visits: This outcome was reported by one study 34 and the analysis of data showed that the risk at either 6 or 12 months was not different between the intervention and the control group (Table 3).
Referral to acute care facilities: This event was reported by one study,
33
and it found that the deprescribing intervention group using the geriatric-palliative algorithm had a significantly lower 12-month referral rate to acute care facilities than the control group that had no change in their medications (11.8%
Other patient-related outcomes: Other patient-related outcomes were reported by two studies34,35 that included 12 months follow-up of sleep quality
34
; and three months follow-up of depression,
35
neuropsychiatric symptoms,
35
extrapyramidal side effects
35
and body weight.
35
No significant difference was reported in these outcomes except for depression in the 3-month follow-up study. This study involved tapering off and replacing escitalopram, citalopram, sertraline and paroxetine with placebo over the first week and reported significantly higher Cornell scores for depression in the intervention group than the placebo control group (6.03 ± 4.76
Impact of deprescribing of dual-purpose medications on patient-related outcomes according to follow-up duration.

QOL was measured by QUALIDEM 31 and QOLAD (total score: 52)34,35; Physical function was measured by MBI (total score: 100) 34 or Lawton & Body’s Physical Self-Maintenance Scale 35 ; Cognitive function was measured by MMSE (total score: 30) 34 or Severe impairment battery scale. 35
Abbreviations: C, control group; d, Day; GP, general practitioner; I, intervention group; m, Month; MBI, Modified Barthel Index; MMSE, Mini–Mental State Examination; NR, not reported; QOL, quality of life: QOLAD, Quality of Life in Alzheimer’s Dementia; QUALIDEM, quality of life instrument for proxy completion; wk, Week.
Calculated from the data given in the study.
Statistically significant.

Random effect models and forest plots showing the impact of deprescribing of dual-purpose medications on mortality, adverse events and healthcare utilization: (a) impact on mortality. (b) Impact on falls. (c) Impact on non-vertebral fractures. (d) Impact on unplanned hospital admission.
Risk of bias
The risk of bias of all four RCTs31,32,34,35 was assessed using the Cochrane Risk of Bias Tool. The overall risk of bias of included studies was moderate to low (Supplementary Table S3 and S4). All the studies had a low risk of bias only on blinding of outcome assessors and half of the studies had not blinded participants and administering personnel as well as had incomplete outcome data. Three of the four studies had ensured randomisation, concealment allocation, and avoided selective reporting of the outcome. But all studies had one or more other sources of bias, including inclusion bias, 35 bias due to sample size,31,34 respondent bias, 34 contamination bias31,32 and bias on the readiness of implementing the deprescribing recommendation. 31 The remaining one quasi-experimental study 33 was assessed using the ROBINS-I tool and the overall risk of bias for this study was serious. Although most of the items had a low risk of bias in this study, serious concerns arose for confounding and selection.
Discussion
This review on deprescribing interventions involving DPMs for older people with LLI near the EOL in hospitals and RACFs found a substantial reduction in 12-month mortality (RR = 0.59), 12-month referral to acute services (60% decrease), and worsening of depression scores (rise by 1.61,
The approach to deprescribing intervention in each study varied but in general was either active deprescribing or recommendation by a variety of health professional teams – physician/geriatrician, or multidisciplinary team of GP and geriatrician or clinical pharmacologist. While this may reflect locally relevant practice, it limits the comparability of effectiveness estimates.
This review suggested a general decline in the QOL irrespective of its state (good or poor), types of deprescribing and whether deprescribing was performed. The QOL did not differ significantly between intervention and control groups for up to 12 months. A systematic review on a similar population also concluded that the QOL may not improve significantly, 36 but the data were heterogeneous and medications were not categorised into preventive, symptom control or dual-purpose, whereas our review excluded studies exclusively on preventive and symptom-control medications. Another systematic review on deprescribing of anti-psychotics in older adults with dementia found low or no clear evidence on QOL. 37 In our review, data on QOL could not be pooled because the included studies had heterogeneity in terms of the assessment tool, DPMs deprescribed and follow-up periods. Particularly concerning was the overall impact of high sample attrition (38.9 34 –57.7%) 31 on the reliability of the QOL measure.
Deprescribing interventions appear to generate no significant alteration to physical and cognitive function at 6.25 and 12 months. The tool used for the assessment of both the functions varied in the included studies, therefore, data were not pooled. Most participants included in the studies of our review had dementia. People with dementia are known to have a gradual and prolonged decline in their functional capacity in long-term care and palliative care. 38 The ability of current treatments to slow down the functional deterioration in LLIs and EOL is rather limited, but a systematic review has indicated accelerated cognitive decline in dementia patients on benzodiazepines 39 ; by implication, deprescribing could be hypothesised to improve cognitive and functional capacity. In our review, however, the findings showed that functional capacity did not differ significantly after deprescribing.
In this review, three31,33,34 of the four31–34 studies reported on mortality showing a non-significant effect of deprescribing. However, the overall risk of mortality in the intervention group was lower compared to the control group in hospitals and RACFs when the pooled data derived from the meta-analysis of the four studies was considered. Meta-analysis utilises the data pooled from the individual studies, which can increase the statistical power and has been proven useful when individual studies are not sufficiently powered due to a small sample size.40,41 Previous systematic reviews of deprescribing of PIMs in older adults, not necessarily at the EOL, had shown similar results irrespective of LLIs,24,42,43 but the focus of those reviews were not on DPMs.
The outcomes on the risk of falls were reported to be non-significant by the two RCTs,31,34 but the authors reported the possibility that they were underpowered to detect significant changes in these outcomes. While recent studies have found that DPMs, such as anti-psychotics, digoxin and diuretics,44–46 may increase the risk of falls, our study could not confirm this. Referral to acute care facilities reported by the quasi-experimental study, 33 showed significantly lower referrals in the intervention group than the control group.
The list of DPMs considered by our review was generated from the literature; diuretics and anti-psychotics are particularly worthy of discussion here. Diuretics, particularly furosemide are commonly prescribed until the last month of life in patients with terminal illnesses,18,28 most likely to manage symptoms such as dyspnoea secondary to pulmonary oedema.18,19 This is particularly important in patients with advanced heart failure, even towards the EOL.18,47 Diuretics are also used for the secondary prevention of disease but when the time-until-benefit is less than the estimated life expectancy, it could be reasonable to deprescribe diuretics used for the preventive measure. 28 Two studies included in our review reported 76% overall successful deprescribing of diuretics; one study stated recurrence of symptoms/signs as the cause of failure and the drug was re-administered, 33 and the other study stated that while not all targeted diuretics were deprescribed those that were attempted had 100% success. 34 However, neither study identified whether the diuretics were indicated for oedema, advanced heart failure or hypertension, or if they were used for the secondary prevention of disease. Anti-psychotics were deprescribed with just over 70% overall success.31,33,34 Anti-psychotics are commonly used in older adults with LLE47–49 and have played a significant role in the symptom management of delirium.15,16,28 Haloperidol has also been used for management of psychosis, terminal agitation/restlessness, nausea and vomiting at the EOL but requires close monitoring of extrapyramidal side effects. 16 For the management of BPSD, the STOPPFrail 13 and its update STOPPFrail version 2 50 recommend deprescribing neuroleptic anti-psychotics in those taking these medications for more than 3 months if there are no symptoms of BPSD. One of the three studies included in this review used STOPPFrail criteria 13 for deprescribing of anti-psychotics. The other two studies considered a risk/benefit assessment and where necessary tapering dose and/or monitoring for adverse withdrawal effects and/or considering alternative medication, although neither clarified whether the anti-psychotic was used for BPSD.33,34
There are some limitations to this review. We searched several databases to identify articles, but it is possible that the search was not exhaustive. We included Google Scholar, as although it lacks the ability to reproduce searches, we considered it may assist in locating non-indexed articles which could otherwise be missed by other databases. The tools used to measure QOL and functionality varied; hence meta-analysis was not feasible for these outcomes. The approach to identify PIMs was not uniform across the studies either. For instance, studies used explicit criteria such as STOPPFrail and STOPP, or a list of pre-determined medications or an algorithm to identify PIMs. The review contained only five studies, and they mostly had high attrition rates leading to loss of statistical power. This suggests that multi-centre studies with a larger sample size are necessary to build a valid estimate of the impact on health outcomes. It is also important to note that the outcomes were not the consequence of deprescribing of a single medication or exclusively DPMs. It also suggests that deprescribing studies focusing only on DPMs are rare in the literature. It would be worth exploring this area further, as deprescribing DPMs are often avoided by clinicians and patients due to uncertainty around the likely adverse events.
Conclusion
This review suggests that deprescribing interventions involving DPMs for older people with LLIs and LLE are rare in the literature, but there is some evidence that they can lower the risk of mortality and referral to acute care facilities. However, there is insufficient evidence to establish its impact on other outcomes, such as health service utilisation and adverse events. Reliable conclusions on whether deprescribing significantly improves or worsens QOL, physical or cognitive functions were precluded by the heterogeneity in their assessment tool, medications deprescribed and high sample attrition. More studies with a larger sample size, a focus on DPMs, and clear identification of LLE and EOL profiles of older participants are needed to confirm these findings further.
Supplemental Material
sj-docx-1-taw-10.1177_20420986211052343 – Supplemental material for Impact of deprescribing dual-purpose medications on patient-related outcomes for older adults near end-of-life: a systematic review and meta-analysis
Supplemental material, sj-docx-1-taw-10.1177_20420986211052343 for Impact of deprescribing dual-purpose medications on patient-related outcomes for older adults near end-of-life: a systematic review and meta-analysis by Shakti Shrestha, Arjun Poudel, Magnolia Cardona, Kathryn J. Steadman and Lisa M. Nissen in Therapeutic Advances in Drug Safety
Footnotes
Author contributions
SS and LMN conceived the study. All authors designed the study. SS and AP completed the screening process and quality assessment with input from MC, KJS and LMN. SS extracted data, and all authors provided input on synthesis. MC provided input on analysis. SS drafted the manuscript. All authors approved the final version for submission.
Availability of data and material
All data generated or analysed during this study are included in this published article and its supplementary information files.
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MC was the original designer of the CriSTAL tool used in this review to assist in the identification of articles meeting the end-of-life criteria when eligibility was ambiguous. She derives no financial or other benefits whatsoever from its use as it is open access. No other disclosures to report.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: SS was supported by the University of Queensland Research Training Programme (UQRTP) Scholarship from The University of Queensland.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
