Abstract
Background and Aims:
Chemical impurities discovered in angiotensin receptor blocker (ARB) products in late 2018–2019 resulted in recalls of various products and has likely had downstream effects for patients and prescribers. The purpose of this study is to determine how the valsartan recall impacted clinical endpoints and prescribing of antihypertensives.
Methods:
This was a retrospective, single-center, cohort study including patients receiving recalled valsartan with essential hypertension who were mailed a recall letter on 12 March 2019. Mean blood pressure endpoints were collected 6 months before (pre-recall) and after the recall letter was mailed (post-recall). Antihypertensive medication changes and titrations were also characterized post-recall.
Results:
A total of 300 patients meeting eligibility criteria were included. There was no statistically significant difference in mean systolic blood pressure (SBP) or diastolic blood pressure (DBP) when pre- and post-recall blood pressures were compared (SBP: 137.2 mmHg versus 135.8 mmHg, p = 0.125; DBP: 78.6 mmHg versus 78.5 mmHg, p = 0.900). In addition, the percentage of patients with controlled blood pressure readings was similar in the pre- and post-recall timeframes (28% versus 27%, p = 0.72). A total of 33 medication changes involving valsartan occurred, with approximately one-third being changed to another ARB (n = 11) or drug class (n = 12). In total, 11 valsartan medication changes were specifically documented to be related to the valsartan recall.
Conclusion:
The results of this study indicate the valsartan recalls that occurred in 2019 did not significantly impact the clinical outcomes of the studied population.
Plain Language Summary
Impact of a medication recall on Veterans’ outcomes
Background:
Chemical impurities discovered in a class of blood pressure medications known as angiotensin receptor blockers (ARBs) occurred in late 2018–2019. This resulted in recalls of various products and has likely had downstream effects for patients and prescribers.
Objective:
The purpose of this study is to determine how the recall of valsartan, which is a medication in the ARB class, impacted clinical endpoints and prescribing of medications for blood pressure.
Methods:
This was a retrospective, single-center, cohort study including patients receiving recalled valsartan with high blood pressure who were mailed a recall letter on 12 March 2019. Blood pressure endpoints were collected 6 months before (pre-recall) and after the recall letter was mailed (post-recall). Medication changes and titrations were also characterized post-recall.
Results:
Three hundred patients meeting eligibility criteria were included. There was no difference found in systolic blood pressure (SBP) or diastolic blood pressure (DBP) when pre- and post-recall blood pressures were compared (SBP: 137.2 mmHg versus 135.8 mmHg; DBP: 78.6 mmHg versus 78.5 mmHg). In addition, the percent of patients with controlled blood pressure readings was similar in the pre- and post-recall timeframes (28% versus 27%). A total of 33 medication changes involving valsartan occurred, with approximately one-third being changed to another ARB (n = 11) or drug class (n = 12). Eleven valsartan medication changes were specifically documented to be related to the valsartan recall.
Conclusions:
The results of this study indicate the valsartan recalls that occurred in 2019 did not significantly impact the clinical outcomes of the studied population.
Background
An estimated 4500 drugs and devices are recalled per year in the United States (US) by the US Food and Drug Administration (FDA). The potential for injury from the recalled products can range from being unlikely to having the potential to cause harm or death. 1 In July 2018, chemical impurities discovered in valsartan products prompted the FDA to investigate many angiotensin receptor blockers (ARBs).2,3 This resulted in subsequent recalls of various manufacturer and lot numbers of valsartan and other ARBs including irbesartan and losartan. N-nitrosodimethylamine (NDMA), N-nitroso-N-diethylamine (NDEA), and N-nitroso-N-methyl-4-aminobutyric acid (NMBA) are three chemicals that have been identified and commonly attributed to the manufacturing process.2–4 These impurities have raised concern because certain concentrations of these chemicals are considered probable human carcinogens.2,4 The recall effectively disrupted the availability of valsartan, irbesartan, and losartan-containing products with estimated impacts on one-sixth of US ARB manufacturers. 2
ARBs are considered to be one of the first-line agents for the reduction of cardiovascular mortality in hypertension, acute myocardial infarction, and heart failure with reduced ejection fraction.5–8 In addition to the cardiovascular benefits, this drug class provides renal protective effects in patients with albuminuria and chronic kidney disease.9,10 As a class, ARBs exert their action via the renin-angiotensin system (RAS). This physiologic pathway is involved in the homeostasis of fluid management and blood pressure. 11 When overactivated, the RAS system can lead to the pathogenesis of hypertension, cardiac hypertrophy, and kidney dysfunction. The ARBs interfere with the RAS by blocking a key component in this pathway, angiotensin II, therefore decreasing vasoconstriction and preventing these downstream effects.11,12
The prevalence of hypertension is approximately 45% in the Veteran population. 13 Valsartan and losartan are the two preferred ARBs on the Veterans Affairs (VA) Pharmacy Benefits Management (PBM) formulary. 14 Supplies within VA were not affected by valsartan recalls until March 2019, at which time the recall included valsartan strengths ranging from 40 mg to 320 mg tablets from various manufacturers. In response to the recalls, VA PBM published guidance that recommended contacting patients through a mailed letter with instructions to continue taking affected product until replaced with unaffected supply on the next fill, as uncontrolled hypertension resulting from abruptly discontinuing the medication carries more risk than short-term exposure to small amounts of a possible carcinogen. As the unaffected supply was provided on the next refill, patients did not accrue additional cost. The letter also encouraged the recipient to communicate with their provider or pharmacist to obtain new medication, if needed. 15
The downstream effects of the ARB recalls have not been fully elucidated, but have possibly impacted patient trust in prescription drugs and medication adherence. In addition, prescribing difficulties arise with challenges in changing to alternatives within the same or different drug class. The outcomes associated with the limited accessibility of ARB products increases the concern for inadequate control of chronic diseases. Increased risk of cardiovascular morbidity and mortality is the major consequence associated with uncontrolled hypertension and other diseases. 16 An analysis of insurance claims and Canadian health information databases including 55,461 patients performed by Jackevicius et al. demonstrated an increase in emergency room visits for hypertension in patients that received recalled valsartan product. 17 In this analysis, 73.8% of patients were changed to another ARB and 8.8% were changed to a non-recalled valsartan product. The purpose of this study is to determine how the valsartan recall impacted clinical endpoints and subsequent prescribing of antihypertensives.
Methods
Study design
This retrospective, single-center, cohort study was conducted within a single, tertiary care VA health care system that serves as a referral, teaching, and research facility. Electronic health records were utilized to conduct manual data extraction during the study timeframe defined between September 2018 to September 2019. The study was approved by the Durham VA Medical Center Research Institutional Review Board (identifier: 02258).
Subjects
A convenience sample was chosen based on the first 300 patients from a randomized, computer-generated list of recall letter recipients that met the pre-specified inclusion and exclusion criteria. The study included patients with essential hypertension, prescribed valsartan from a lot number that was recalled as of 1 March 2019, and mailed a recall letter on 12 March 2019. Patients were excluded if they were prescribed valsartan for less than 6 months, received valsartan from a non-VA provider, or were without blood pressure values within the study timeframe.
Endpoints
The primary endpoint was the difference in mean blood pressure within 6 months before (pre-recall) and 6 months after the recall letter was mailed (post-recall). The mean of two blood pressure readings were collected from distinct outpatient visits during the study time frame, if two readings were available. If a visit was associated with multiple readings, the last blood pressure was utilized for data collection. If more than two blood pressure readings were available pre-recall, the two readings collected closest to the recall letter being mailed (12 March 2019) were selected. Post-recall blood pressure values were collected within 6 months after 26 March 2019, accounting for an additional 2 weeks for letters to be received by Veterans. Secondary endpoints included the change in the percent of patients with controlled blood pressure pre-recall versus post-recall and the number of patients experiencing acute hypertensive crisis within 3 months. As patient-specific blood pressure goals may vary based on comorbidities, controlled blood pressure was analyzed using less than 140/90 mmHg and less than 130/80 mmHg as goals. 8 The percentage of patients experiencing acute hypertensive crisis was defined as a systolic blood pressure (SBP) ⩾180 mmHg and/or diastolic blood pressure (DBP) ⩾120 mmHg within 3 months of the recall letter. 8 A post hoc analysis was conducted in these patients to determine if it was associated with an emergency room visit encounter. Additional secondary endpoints included the number of patients with medications changed to other antihypertensives, as well as the number of antihypertensive medication changes or titrations in the post-recall period. The medication changes did not evaluate manufacturer changes of the same medication as all patients were switched from affected supply of valsartan to unaffected supply.
Analysis
A paired t test was used to analyze continuous data for the primary endpoint. Nominal data for the controlled blood pressure endpoint was analyzed using McNemar’s test. The level of significance was set a priori at 0.05. Descriptive statistics were utilized for baseline characteristics and the additional secondary endpoints. These analyses were conducted using Microsoft Excel Version 2016 (Microsoft Corp., Redmond, WA, USA).
Results
A total of 563 patients were mailed a valsartan recall letter from the health care system. Screening was stopped after the pre-specified sample size of 300 patients meeting inclusion and exclusion criteria was reached (Figure 1). Baseline patient demographics and prescribed antihypertensives are shown in Table 1. Of all the patients prescribed recalled valsartan product, 38% were prescribed 320 mg tablets. The remainder of patients were prescribed 40 mg tablets (13.3%), 80 mg tablets (19.3%), and 160 mg tablets (29.3%). The second most commonly prescribed antihypertensive included a beta-blocker, which was prescribed in almost half of patients, with metoprolol (n = 73) and carvedilol (n = 70) accounting for most of the beta-blockers prescribed.

Flow of participants included in the final sample.
Demographic information and baseline antihypertensives.
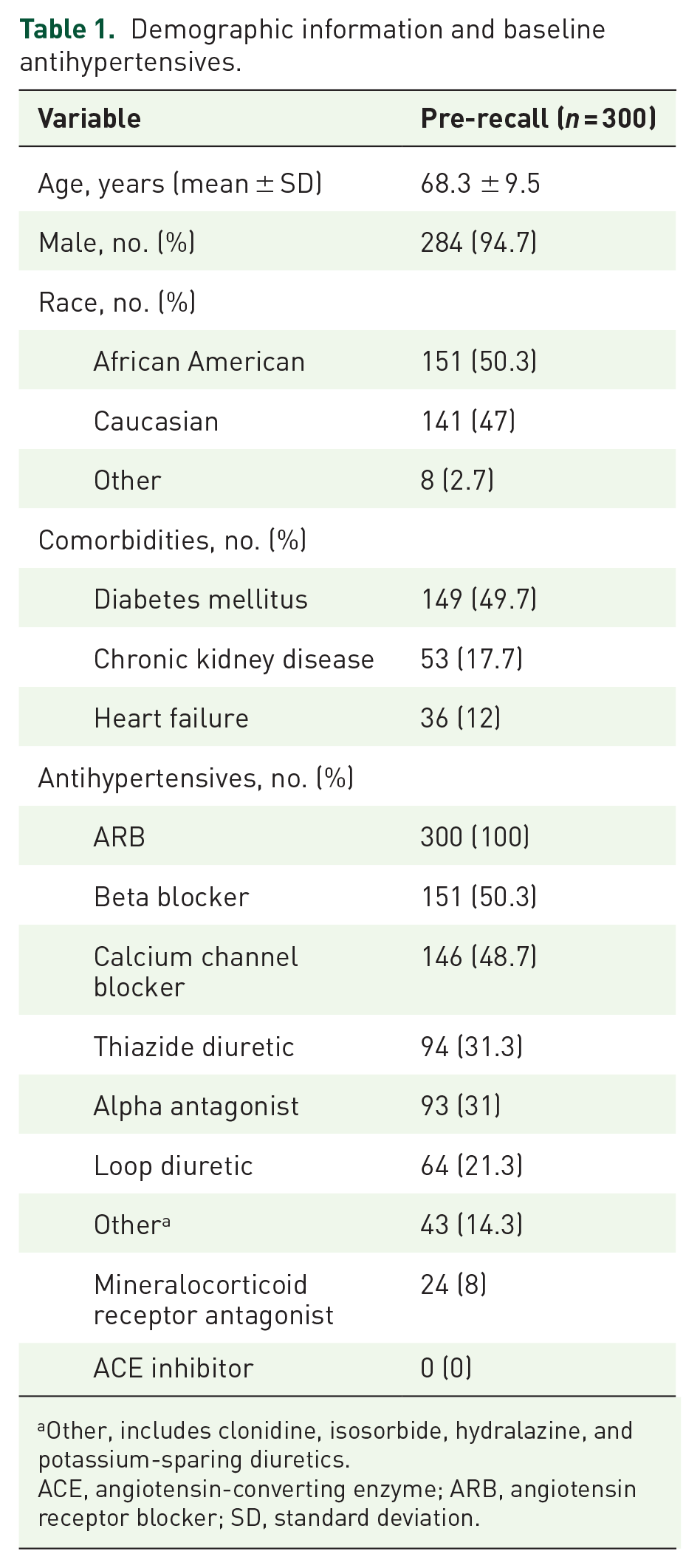
Other, includes clonidine, isosorbide, hydralazine, and potassium-sparing diuretics.
ACE, angiotensin-converting enzyme; ARB, angiotensin receptor blocker; SD, standard deviation.
There was no statistically significant difference in mean blood pressure pre- and post-recall as seen in Table 2. However, 120 patients (40%) were missing one blood pressure reading within the pre- and/or post-recall period (n = 42 pre-recall, n = 40 post-recall, n = 38 missing one reading pre- and post-recall). There was also no difference in the number of patients with controlled blood pressure readings. Using a blood pressure goal of <130/80 mmHg, 28% were considered controlled pre-recall versus 27% post-recall (p = 0.720). More patients had mean blood pressure that met a goal of <140/90 mmHg, 57% were considered controlled pre-recall versus 61% post-recall (p = 0.218). Approximately 9% (n = 27) of patients experienced acute hypertensive crisis based on recorded blood pressure in the 3 months after the recall letter was mailed; 2% of patients (n = 6) were associated with an emergency room visit encounter.
Blood pressure outcomes pre- and post-recall letter.

DBP, diastolic blood pressure; SBP, systolic blood pressure; SD, standard deviation.
The mean number of antihypertensives prescribed was similar between timeframes (3.05 in the pre-recall versus 3.08 in the post-recall period). There were 177 blood pressure medication changes or titrations that occurred within the post-recall period. Of these, 132 were medication changes and 55 were medication titrations. A mean of 0.59 [standard deviation(SD) 0.95] changes or titrations occurred per patient, with the majority of patients (n = 198) having no medication changes or titrations. Numerical increases in prescribing of other antihypertensives were noted aside from ARBs in the post-recall period as shown in Figure 2; 11% of changes were specifically related to valsartan (n = 33). The outcomes of valsartan changes included drug discontinuation (n = 10), changing to another ARB product (n = 11), or changing to another antihypertensive agent (n = 12). Documented reasoning for these outcomes included adverse drug reactions such as hypotension or acute kidney injury, worsening of end-stage renal disease, an outside provider decision, or a non-documented reason. Of these changes, 11 (approximately one-third) were documented to be related to the national valsartan recall, which led to changes to another ARB or other antihypertensive class such as angiotensin-converting enzyme inhibitors or calcium-channel blockers.

Prescribed antihypertensives by drug class compared in the pre- versus post-recall group.
Discussion
Recalls associated with manufacturing impurities continue to plague the healthcare system. 18 In late 2019 and early 2020, the high-volume medications ranitidine and extended-release metformin were added to the growing list of medications recalled for NDMA contamination.19,20 As recalls are becoming more common, studies of the impact on clinical outcomes for chronic diseases and how recalls are handled will continue to be important.
The severity of conditions being treated and risk of drug discontinuation on clinical outcomes are important factors for the management of future medication recalls. The risk of uncontrolled blood pressure due to abrupt drug discontinuation in the case of valsartan increases the concern for potentially negative health outcomes. The present study found no difference in blood pressure outcomes related to the national valsartan recall, as there was no statistically significant difference in the change in mean blood pressure readings or the proportion of patients who met controlled blood pressure goals pre- versus post-recall. These results suggest that patients were more or less comfortable with continuing valsartan after the recall, with only 3.6% of patients opting to change to a different medication specifically because of the recall. The average number of antihypertensives prescribed pre- and post-recall were very similar, which also could account for no difference observed. While reassuring, these results may not be applicable to all medication recalls. The VA healthcare system was able to effectively source non-impacted supplies of valsartan quickly in this instance. Complete removal of products, such as the recall of all ranitidine products in 2019/2020, or more extensive recalls that cause significant shortages may prove more impactful on these outcomes. 20 The availability of appropriate alternatives within the same or different drug class are also considerations in drug shortage management. In this case, unaffected valsartan, other ARBs, and other drug classes are widely used for the treatment of hypertension and easily accessible, which may not be the case for other recalled medications.
Many medication changes and titrations occurred during the post-recall period, which is reasonable given these occurred over a 6-month period. In addition, less than 30% of patients had controlled blood pressure based on the goal of less than 130/80 mmHg. Valsartan-specific medication changes were overall limited, indicating that instructions from the mailed patient letter were assumed to be followed. Valsartan medication changes were evenly split between a change to another ARB and a change to another drug class. This contrasts with the findings of Jackevicius et al., which showed the majority changed to another ARB. 17 The results of Jackevicius et al. were similar to an additional Canadian analysis conducted in a younger patient population by McAlister et al.17,21 Another analysis by Desai et al., 22 which utilized prescription records in the US, demonstrated a shift in prescribing to other ARBs while the total number of ARBs prescribed did not decrease after FDA recalls. Similarly, an analysis conducted in Germany by Rudolph et al. found an increase in alternative ARBs after the valsartan recall, but no difference in utilization of other antihypertensive classes occurred. 23 The difference in medication changes seen between the present study and other studies could be attributed to the fact that a large proportion of the patients in the present study were on valsartan 320 mg tablets. Valsartan 320 mg tablets do not have equivalent dosing when converting to losartan, which is the other preferred formulary alternative by the VA PBM.14,24 This likely led to more medication changes to non-ARB drug classes.
A small proportion of patients experienced acute hypertensive crisis 3 months after the recall letter was mailed, with an even more limited group being associated with an emergency room visit encounter. The Canadian analysis by Jackevicius et al. did find a statistically significant increase in emergency department (ED) visits for hypertension immediately after the recall (0.11% versus 0.17%, p = 0.02). 17 However, these results are challenging to compare with the present study given differences in study design. Jackevicius et al. were also able to determine hypertension-related ED visits out of the total ED visits for a larger population, 17 whereas the present study identified ED visits based on those that met the definition of acute hypertensive crisis. In addition, the timeframe for the hypertension-related ED visits was over 3 months for the present study, versus the previously mentioned rates for the study by Jackevicius et al., 17 which were over 1 month immediately following the recall. The present study is also limited given there is no comparator for acute hypertensive crisis or related ED encounters pre-recall. Clinically, however, the results of Jackevicius et al. represent an overall small proportion of the total ED visits. 17 Jackevicius et al. also noted that the hypertension-related ED rates could encompass both uncontrolled blood pressure as well as patients presenting to acquire another antihypertensive. 17 Based on the results of the present study, we believe the national recall did not significantly impact clinical outcomes in the studied population.
Several limitations should be noted. As this was a retrospective study, the concern for information and confounding bias is possible. In addition, the present study included a small sample size limiting the statistical power of the results. Blood pressure is a variable endpoint and 40% of patients did not have two blood pressure readings available in the pre- and post- settings. This could make the results more challenging to interpret as blood pressure can be impacted by a variety of factors. The studied population may not be comparable with the civilian population for multiple reasons. The patients included were older Veterans, mostly male, and about half of them had comorbid diabetes. A smaller percent of patients had comorbid heart failure (12%). Moreover, other comorbid conditions such as coronary artery disease or atrial fibrillation were likely common in this patient population as beta-blockers were the second most commonly prescribed antihypertensive. With an average of three antihypertensives per patient, this could also be reflective of patients with long-standing hypertension. These patients can be more challenging to get to a targeted blood pressure goal. Patients included were also more likely to be more clinically complex based on the study design, which required two blood pressure readings from separate visits within approximately 1 year. Almost half of the patients excluded were due to not having blood pressure readings within the study timeframe, predisposing the included patients to have more frequent interactions with the healthcare system. In addition, the general differences between VA and non-VA medical care, including the use of a closely monitored formulary system, limits generalizability outside of VA settings. Medication adherence was also not evaluated. The present study evaluated antihypertensives that were prescribed, but did not review in-depth refill history. Therefore, patient compliance could have been impacted by the recalls and would have not been accounted for. Mailed patient letters were utilized to disseminate recall information; therefore, the present study did not evaluate other possible means of communication (newspaper, television, etc.). Lastly, although the VA health care system was not impacted by recalled valsartan product until March 2019, media attention of the recalls started in July of 2018. 2 This could have caused more patients to have requested to be switched from valsartan to another medication prior to the study timeframe, thus excluding these patients from the present study.
The present study showed the national valsartan recall did not significantly impact clinical outcomes in the studied population and a limited number of medication changes that were attributed to the recall occurred.
