Abstract
Background:
Drug recalls safeguard patients from potentially harmful existing pharmaceutical products in the market due to safety concerns or manufacturing issues.
Objective:
To analyze the drug recall patterns, frequencies, and causes based on the publicly available data from the Nepalese Drug Regulatory Authority, Department of Drug Administration (DDA).
Design:
A 2-year retrospective secondary analysis was conducted based on drug recalls in Nepal, which is available from the official website of DDA, Nepal.
Methods:
The substandard drug recalls data from April 17th, 2023 to May 14th, 2025 were included in the study from which information were extracted across various domains, including the drug name, dosage forms, manufacturing and expiry date, reasons for the recall, and recall date whereas, inclusion of falsified medicines and information regarding the “special permission,” “news & updates,” and “notices” sections within the DDA website were excluded from the studies to ensure only drug recalls were incorporated.
Results:
The study showed that 50 recalls were made over the 2 year period. The majority of the recalled drugs were antibiotics (16%). The most common reasons for drug recalls were assay failure (34%) and non-compliance with standards set out by the Indian Pharmacopoeia-2022 (40%), followed by United States Pharmacopoeia-2022 (28%). Oral formulations (74%) were most commonly recalled, out of which tablets (32%), suspensions (16%), and syrups (12%) were recalled in greater frequencies among all the recall notices. Most recalls were made from domestic pharmaceutical companies (84%). The majority of the drugs (34%) were recalled after 15 months of the finished product being in the market, whereas only 26% were recalled within the first 5 months.
Conclusion:
A robust and continuous evaluation of drug recalls by regulatory authorities can help reduce their frequency, lessen their impact on the healthcare system, and improve overall drug safety.
Plain language summary
1. Drug recalls are crucial to protect the public by removing and recalling substandard drugs.
2. This unique study retrospectively analyzes the two-year publicly available drug recall data from the National Drug Regulatory Authority of Nepal.
3. Drug recalls in Nepal are increasing in order, with several quality control failures and non-compliance with good manufacturing practices.
4. The findings emphasize the effective development of testing sites and focus on quality control parameters of drugs to ensure drug safety.
Introduction
Drug recall occurs when a medication is withdrawn from the market due to safety concerns or manufacturing issues. 1 A drug recall is the most effective way to protect the public from a defective or potentially harmful pharmaceutical product 2 , and is essential for ensuring product quality and protecting public health. Generally, drug recalls are classified into Class I, II, or III based on the severity of potential health risks, where Class I poses serious health risks or death, Class II involves reversible side effects with low risk, and Class III concerns minor, non-harmful defects, such as packaging issues.3,4
Drug recalls also raise concerns about substandard and falsified medicines. Substandard medicines do not meet with the standards and requirements outlined in the Good Manufacturing Practices (GMP) regulations. 5 The World Health Organization (WHO) defines substandard medical products or out-of-specification products as those authorized products failing to meet the quality standards or their specification, or both. 6 WHO also defines “falsified medical products” as “Medical products that deliberately/fraudulently misrepresent their identity, composition or source.” 6
Literature highlights that the trend of drug recalls is increasing yearly. 7 As of 2025 AD, the United States Food and Drug Administration listed 17 drug recalls. 2 Similarly, a study on FDA drug recall characteristics revealed that the common reasons for drug recalls were contamination, mislabeling, adverse reactions, defective product, and incorrect potency. 8 Similarly, a long-term FDA drug recall study also found that the most common causes were non-compliance with Current Good Manufacturing Practices (cGMP), primarily due to impurities and contaminants, followed by control and labeling/packaging issues.1,7
Similarly, a study by the Saudi Food and Drug Authority reported that the most frequent causes of drug recalls were non-compliance with the manufacturer’s specifications (33.2%), contamination (23.7%), regulatory violations (20.5%), and the patterns of voluntary recall requests by pharmaceutical companies. 9 Identifying the reasons behind recalls helps to develop effective strategies to enhance drug safety, therapeutic effectiveness, regulatory adherence, and overall patient care. 10 A review revealed that the increasing rate of pharmaceutical product recalls, driven by enhanced inspections and industry modernization, has prompted regulatory agencies to tighten the standards to ensure quality, prevent future recalls, and safeguard public health. 11
The recall process typically involves identifying the defect, notifying regulatory authorities, issuing public alerts, and retrieving the affected products from distributors, retailers, and consumers.1,3,12,13 Manufacturers, regulatory agencies, or customer complaints can initiate recalls, which are classified by the severity of potential health risks, ranging from minor to life-threatening conditions.11,12,14,15 The recall process involves a planned specific course of action, which addresses the depth of recall, the need for public warning, and the extent of effectiveness checks for the recall.3,16,17
Nepalese perspective
Nepal, a low-middle-income country, aims to ensure access to safe, efficacious, and quality standard of drugs. At the same time, the focus remains on regulating and controlling unsafe drug production, sales, export, and import through the National Drug Regulatory body, the Department of Drug Administration (DDA). 18 The DDA operates under the legal framework of the Drugs Act 1978, promulgated by the government of Nepal. 18 The chapter 5, section 14 of the Drugs Act 1978, mandates the return of a drug that is not safe, efficacious, and of quality standards for public consumption. 18 The provision also explains that the manufacturer or its authorized agent is responsible for withdrawing such products from the seller or distributor. DDA is the regulatory authority focused on inspecting and recalling pharmaceutical products with quality issues. 14 A study conducted on the drug recalls from Nepal revealed that 346 pharmaceutical products were recalled during the 2010–2020 period, emphasizing the question on the quality of drugs and their safety. 16 Coming to recent times, this study aims to gather the current trends on the drug recalls within the country and what changes have occurred after 5 years of the prior study. 16 The Ministry of Health and Population, Nepal, also recently issued a notice regarding providing opinions and suggestions on the draft bill to make provisions regarding medicine and health supplies. 19 Accordingly, this study examines publicly available data from the DDA, Nepal, which covers the last 2 years. The focus is limited to recalls of substandard drugs, providing evidence-based recommendations and insights for health authorities, policymakers, and other stakeholders on the current trend of drug recalls. This study provides the latest updates in drug recalls and captures the current state of regulatory oversight, manufacturing practices, and market quality in Nepal, including the outdated data, which might dilute the contemporary relevance. The findings will help to support regulatory authorities in strengthening enforcement measures during production, thereby ensuring the safety and efficacy of medicines before they are introduced to the market. Moreover, regular and systematic evaluations of drug recalls will improve patient safety, reduce the frequency of recalls, and minimize their impact on the healthcare system.
Method
This study follows a 2-year retrospective secondary analysis conducted based on the drug recall data extracted from the publicly available official website of the DDA 20 from April 17th, 2023, to May 14th, 2025. This study aims to address the research gap by examining the frequency and causes of recalls over the recent 2-year period. While substandard drug recall alerts of 2-year spans were included, the data relating to falsified medicines, along with information regarding the “special permission,” “news & updates,” and “notices” sections within the DDA website, were excluded. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (Supplemental File 1). 21
Data collection process
After reviewing the DDA website, the section titled “updates” was selected, and within it, the “Recalls & Drug Alert” option was chosen. 20 Following the aim of the study, 2-year retrospective data were collected by downloading each recall and drug alert notice. The notices were saved according to the drug recall date in ascending order. Each notice was reviewed, and information was extracted across various domains, including the drug name, dosage form, manufacturing date, expiry date, industry type (domestic or multinational), reason for recall, and recall date.
Drug recalls were classified into chemical, microbiological, physical, and regulatory quality failures, expressed as percentages (%), and evaluated for non-compliance based on respective pharmacopoeial standards and regulatory guidelines. The recalled drugs were further organized according to pharmaceutical dosage form and route of administration. The remaining shelf life was calculated by determining the number of days and months between the recall and expiry dates. To ensure completeness, the 2-year dataset from the DDA website was cross-checked to confirm that no information was missed. The Nepalese National Formulary (NNF) 2018 and National List of Essential Medicine (NLEM) 2021 were used to classify the recalled drugs according to their therapeutic categories.22,23 For privacy and confidentiality, the proprietary (brand) names, batch numbers, and company names of recalled drugs were excluded.
Statistical analysis
Once all the notices were downloaded from the official website and saved, the data were entered into MS Excel 2013. The descriptive statistics, such as frequency and percentages, were used to show the data in tabular format.
Result
A total of 50 pharmaceutical product recalls were made during the 2-year period. Following the classification of drugs based on NNF 2018 and NLEM 2021, antibiotics were the most frequently recalled drug class (n = 8; 16%), followed by non-steroidal anti-inflammatory drugs (NSAIDs) (n = 7; 14%). The data show recall across 20 different drug classes (Table 1).
Drug recall frequency distribution based on classification.
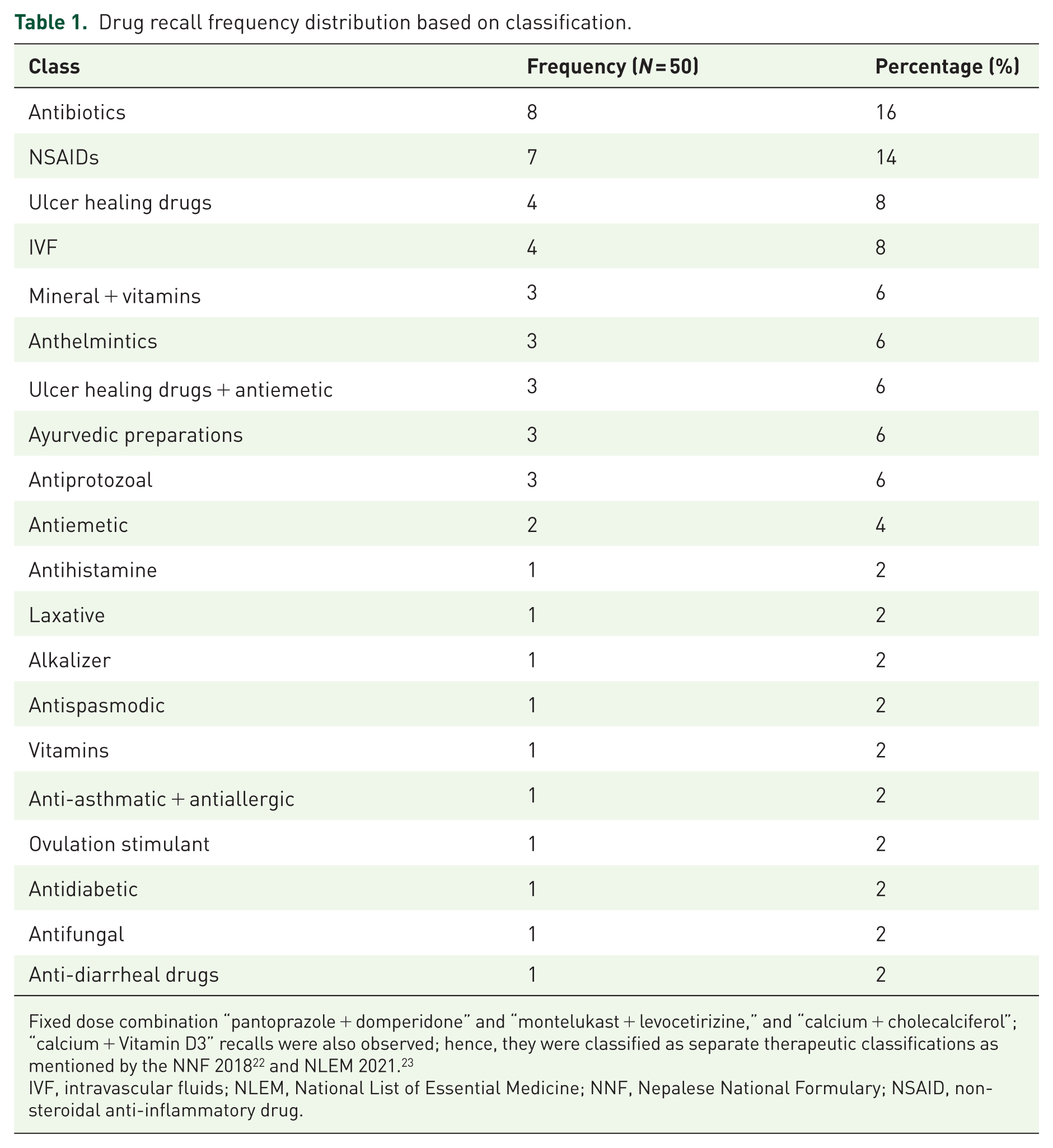
Fixed dose combination “pantoprazole + domperidone” and “montelukast + levocetirizine,” and “calcium + cholecalciferol”; “calcium + Vitamin D3” recalls were also observed; hence, they were classified as separate therapeutic classifications as mentioned by the NNF 2018 22 and NLEM 2021. 23
IVF, intravascular fluids; NLEM, National List of Essential Medicine; NNF, Nepalese National Formulary; NSAID, non-steroidal anti-inflammatory drug.
Drug recall reasons
Among the 50 pharmaceutical products recalled, chemical quality failures were most common (60%), with assay failures (34%) being the predominant type. Microbiological quality issues accounted for 22%, mainly due to sterility failures (14%). Physical quality defects accounted for 16% of total recalls, most of which were related to physical condition (8%) and particulate matter (6%). Regulatory/other violations were minimal (2%). Overall, chemical quality issues were the major contributor to drug product recalls (Table 2). Most recalls were due to failure to comply with standards set out by the Indian Pharmacopoeia (IP)—2022 (40%), followed by the United States Pharmacopoeia (USP)—2022 (28%) and the British Pharmacopoeia (BP)—2023 (12%; Table 3).
Reasons for drug recalls by Quality Domain.

The “violation of the departmental decision,” here refers to the drug “Clomifene,” as mentioned in Supplemental File 2, where the company producing the drug violated the rules regarding product license, sales and distribution registration certificate. Assay failure represents the condition where the product failed to meet the labeled amount of Active Pharmaceutical Ingredient as per the official monograph. 24 In the paper, microbial contamination refers to the presence of microorganisms or toxins in the product, which is also a quantitative term denoting microbial overgrowth that goes beyond the limit test set by the official monograph. 25 Sterility failure describes a condition in which the product is contaminated with viable microorganisms. Sterility is sufficiently high probability that a product processed in an aseptic system is devoid of viable microorganisms. 26
Pharmacopoeial standards not met.

The reasons for drug recalls were also classified according to which pharmacopoeia literature was not complied with. For example, IP-2022 means the drug did not comply with the standards outlined in the 2022 version of the Indian Pharmacopoeia. The pharmacopoeia is the primary source of official quality standards and specifications for medicine and its excipients. 27 The number on the suffix refers to the revision date/years.
AMV, Analytical Method Validation; API, Ayurvedic Pharmacopoeia of India; BP, British Pharmacopoeia; IP, Indian Pharmacopoeia; USP, United States Pharmacopoeia.
Dosage forms recalled
The oral route of administration was the most commonly recalled (n = 37; 74%) product, while the parenteral route accounted for 26% of the total recalled drugs (Table 4). Among drugs administered orally, tablets were the most commonly recalled products (n = 16; 32%), followed by suspension (n = 8; 16%). Among drugs given via parenteral route, the commonly used dosage form recalled included intravascular fluids (n = 5; 10%) followed by injectable drugs (n = 4; 8%).
Drug recalls based on route of administration and dosage forms.

Expiry days and months remaining after the recall period
Following the data analysis, 34% of drugs were recalled when the expiry date remaining was over 15 months (n = 17). However, 52% of drugs were recalled when the remaining expiry dates were less than 10 months (Table 5).
Expiry days and months remaining after the recall date.

Discussion
The study explores and characterizes Nepal’s 2-year drug recall data under different domains (Supplemental File 2). Neupane et al.’s study on drug recalls also shows an increasing trend of recalls of substandard drugs. 16 Following the gap in time after the prior study and to provide insights into the current recall trend, we conducted this study from a pharmaceutical point of view.
This study revealed that antibiotics were the most frequently recalled drug class (16%), followed by NSAIDs (14%) and proton pump inhibitors (8%). These findings were consistent with a recent study in Nepal, which showed antimicrobial agents as the most frequently recalled drug class. 16 This highlights the lack of change and acceptance of previous literature by policymakers and the national regulatory authority in prioritizing the safety of essential drugs.
The increasing number of recalled antibiotics as substandard drugs raises concerns about antimicrobial resistance (AMR) status. Literature also points out how substandard and falsified drugs are related to the rise of AMR.28–30 However, the literature points out the need to establish proper causal association and increase the interventional roles from governmental and industrial levels in mitigating the issues of substandard and falsified drugs.28,29 Such recalls often lead to changes in medication regimens. One study found that several drug recalls required patients to actively change their medications. Most patients switched to a different brand of the same drug, while some had to transition to an entirely different drug class. 31 As a result, cases like this could lead to an increased rate of potential drug–drug interactions, too. 32 Similarly, several studies showed that the major consequences of drug recall were medication switching, increased drug interaction, short-term adverse events, choosing an inappropriate inventory policy, and recent shortages owing to the widespread availability of alternative treatments in the same or similar drug class, subsequently leading to economic burden to the patients.31,33–36
The study showed that assay failures were the most common reason for recalls (34%), followed by sterility issues (14%), product specification issues (10%), and physical condition problems (8%). This is due to non-compliance with cGMP and product specifications as mentioned in the pharmacopoeial standard. These findings also comply with a long-term FDA drug recall study highlighting the most common causes of sterility and non-compliance with cGMP. 1 Similarly, another study also showed impurities/contaminants (37%), control (28%), and labeling/packaging (19%) issues as the major causes of recalls. 37 A previously conducted study on Saudi Arabia also showed that non-compliance with the manufacturer’s specifications and contamination were the most common reasons for recall. 9
The recall of pharmaceutical products due to assay failure and non-compliance has significant impact on both patients and healthcare providers. 38 So the effective implementation of cGMP by pharmaceutical manufacturing companies is a key strategy to mitigate drug recalls. Additionally a robust and continuous evaluation of drug recalls by regulatory authorities can help reduce their frequency, mitigate their impact on the healthcare system, and enhance overall drug safety.
This study underscores the need for robust regulatory oversight, improved manufacturing practices and quality attributes, and transparent communication to minimize risks and maintain patient safety. Previous studies also highlighted that increasing physician awareness, strengthening pre-approval testing, and implementing post-marketing surveillance strategies are crucial in mitigating risks to patient safety and public health.34,35
This study revealed that tablets, suspensions, and intravenous fluids were the most commonly recalled, with assay failure being the primary reason. Such pharmaceutical quality failures and subsequent recall of these drugs pose a significant challenge for pharmaceutical companies, affecting not only the companies themselves but also market supply, patient safety, the environment, and the overall growth of the pharmaceutical industry. 37 A study conducted in China between 2007 and 2012 of 41 food and drug recall events showed a sharp and significant drop in the company’s stock prices. The same study highlighted stakeholders who took necessary measures to combat the company’s reputation after a drug recall bounced back. 39
Most of the drug recalls made in the 2-year period were found to be of domestic manufacturers (84%). This raises the critical question on the quality of medicines produced by such domestic manufacturers in the Nepalese pharmaceutical market.
Although 34% of drugs were recalled more than 15 months before their expiry date, 26% were recalled with only 0–5 months or more than 5–10 months remaining until expiry, highlighting a greater concern. This reveals that before the drugs were recalled, they were sold and consumed by several patients, highlighting a major ethical issue. It can be observed that a few of the drugs were recalled when the expiry dates were less than 100 days. This highlights the incompetence of the existing national regulatory body in effectively testing the drugs and post-marketing surveillance. Despite the inclusion of a drug research laboratory as mentioned in section 6 within the Drugs Act 1978 18 of Nepal, the testing of drugs is still low in number. This huge conundrum should prompt the policymakers, the National Drug Regulatory body, and the Ministry of Health and Population to recognize the essence of testing labs by increasing their quantity and resources at the provincial levels. With the increase in the testing sites, such substandard and low-quality medicines can be removed from the market, thereby helping protect public health. 40
Recommendations
The findings of this study can be helpful for policymakers in planning strategies regarding the drug recall pattern and trend in Nepal. Focusing on the utmost quality of antimicrobials, intravenous fluids are crucial to mitigate the rising issues of AMR and toxicity. To increase the testing sites, the government of Nepal, DDA, Ministry of Health and Population, could enter into a memorandum of understanding (MoU) with the universities’ pharmaceutical laboratories, helping to test the drugs, and increasing the scope of post-marketing surveillance. However, it is also crucial to note that these laboratories follow good laboratory practices and are accredited by the DDA as per the Drugs Act 2035.
Limitations
This study characterized the retrospective data into several domains from the pharmaceutical point of view, providing information on the therapeutic category, dosage form recalled, reason for failure, and days remaining until expiration. Along with the strengths, the study also had some limitations. The inclusion of only 2 years of data constrains our findings and might affect the generalizability. The study also does not explain the inclusion of batch size or the frequencies of samples tested, as they were not included in the DDA notices. The study also did not classify the recall severity into classes I, II, and III.
Conclusion
A 2-year retrospective analysis of drug recalls shows that antibiotics are the most frequently recalled drug class, followed by NSAIDs. By contrast, the primary reasons for drug recalls included failure to meet pharmacopoeial standards, such as assay, sterility, production specifications, physical conditions, and the presence of particulate matter in the product. Tablets, suspensions, and intravenous fluids were the most common recalled drugs. More than 50% of the products recalled had expiry dates of less than 10 months, emphasizing the critical issue and underscoring the need for increasing the drug testing sites around the country.
This study also identifies the primary causes and patterns of recalls, offering information to enhance quality assurance systems, influence regulatory frameworks, and ultimately protect patient safety. Similarly, the study also emphasizes the urgent need for intervention and continuous surveillance from regulatory bodies to draft necessary bills regarding the safety and efficacy of medicines during their production, which ultimately contribute to scientific understanding and practical improvements in the drug quality system. According to the study, the trend of drug recalls is increasing, and it is recommended that regulatory agencies take the appropriate steps to stop and manage excessive drug recalls. The findings from this study are both descriptive and significant for public health and regulatory research.
Supplemental Material
sj-docx-1-taw-10.1177_20420986251398725 – Supplemental material for Drug recall, its frequencies and conclusion: a retrospective secondary analysis involving 2-year publicly available data from Nepal
Supplemental material, sj-docx-1-taw-10.1177_20420986251398725 for Drug recall, its frequencies and conclusion: a retrospective secondary analysis involving 2-year publicly available data from Nepal by Sachita Barma, Nabin Pathak, Shreya Dhungana, Prabhat Kumar Jha and Sunil Shrestha in Therapeutic Advances in Drug Safety
Supplemental Material
sj-xlsx-2-taw-10.1177_20420986251398725 – Supplemental material for Drug recall, its frequencies and conclusion: a retrospective secondary analysis involving 2-year publicly available data from Nepal
Supplemental material, sj-xlsx-2-taw-10.1177_20420986251398725 for Drug recall, its frequencies and conclusion: a retrospective secondary analysis involving 2-year publicly available data from Nepal by Sachita Barma, Nabin Pathak, Shreya Dhungana, Prabhat Kumar Jha and Sunil Shrestha in Therapeutic Advances in Drug Safety
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
