Abstract
Introduction:
Anticholinergic drugs are associated with poor outcomes in older patients but no specific intervention strategies aimed at reducing anticholinergic drug exposure have been described.
Objectives:
To identify whether a consultant-led medication review targeting anticholinergics would reduce anticholinergic drug exposure [number of anticholinergic drugs and Anticholinergic Risk Scale (ARS) score].
Methods:
The first phase of the audit included 70 consecutive admissions (mean age 84 years, 53 women). ARS score was calculated on admission and after initial consultant review. Re-audit was undertaken on another 70 consecutive admissions (mean age 83 years, 43 women) after introducing a system of informing the responsible consultant of the ARS score at their first review.
Results:
Drugs with anticholinergic effects (n = 53) were prescribed preadmission to 45/140 (32%) of patients. Consultant geriatrician review reduced ARS scores (p = 0.01), especially following the introduction of the information system (p = 0.002). In the first arm of the audit, 51 (73%) patients had ARS of 0 after a consultant’s review compared with 47 (67%) patients on admission, whilst 67 (96%) patients had ARS of 2 or less after a consultant’s review compared with 63 (90%) patients on admission. In the second arm of the audit, 59 (84%) patients had ARS of 0 after a consultant’s review compared with 48 (69%) patients on admission, whilst 70 (100%) patients had ARS of 2 or less after a consultant’s review compared with 69 (99%) patients on admission. Anticholinergic drugs were either stopped, or their dose reduced, in 35% of patients in the first arm of the audit and in 73% of patients in the re-audit (odds ratio 5.0, 95% confidence interval 1.4–17.8).
Conclusion:
Consultant-led medication review (standard practice) was effective at reducing anticholinergic drug exposure in the acute setting. A system of alerting clinicians to patients prescribed anticholinergic medications further reduced anticholinergic drug exposure.
Introduction
Anticholinergic (antimuscarinics) drugs are prescribed in 33–47% of older patients in hospital for various conditions, including overactive bladder syndrome and extrapyramidal symptoms [Bostock et al. 2010, 2013; Kosheido et al. 2012; Lowry et al. 2011a]. Numerous drugs including tricyclic antidepressants, antipsychotics, antihistamines and antiemetics have well recognized anticholinergic effects, in addition to other pharmacodynamic effects. Adverse effects associated with anticholinergic drug prescribing include falls, delirium, dizziness, cognitive impairment, impulsive behaviour, constipation, dry mouth and dry eyes [Bostock et al. 2010; Rudolph et al. 2008]. In older people, there is an association between anticholinergic drugs and poorer cognitive [Fox et al. 2011; Han et al. 2008] and physical function [Landi et al. 2007; Lowry et al. 2011a] as well as increased morbidity and hospitalization [Fox et al. 2011; Lowry et al. 2012b]. However, evidence for the association between anticholinergic drug use and increased mortality is mixed. Walesby and colleagues found an increased risk of death in hospitalized older people with highest anticholinergic risk scores compared with lowest scores [hazard ratio (HR) 1.20, 95% confidence interval (CI) 1.02–1.41] and the association remained significant after adjustment for other baseline variables (HR 1.31, 95% CI: 1.09–1.58, p for trend < 0.001) [Walesby et al. 2013] but in a care-home population higher anticholinergic scores were not significantly associated with higher mortality [Kumpula et al. 2011]. Hilmer and colleagues found most of the adverse effects and poor outcomes of polypharmacy in older people were attributable to anticholinergic drugs [Hilmer et al. 2007]. The Anticholinergic Risk Scale (ARS) is a rank categorical list of commonly prescribed drugs with antimuscarinic effects [Rudolph et al. 2008]. The ARS ranks medications for anticholinergic potential on a three-point scale. The ARS score is the sum of points for all the anticholinergic medications taken by a patient. Higher ARS scores carry a greater risk of anticholinergic adverse effects, with scores of at least 3 carrying a particularly high risk. In acutely hospitalized frail older people, a group at high risk of serious adverse reactions, higher ARS scores are independently associated with poorer functional ability and institutionalization [Kosheido et al. 2012; Lowry et al. 2011a]. However, it is not known whether the total number of anticholinergic drugs and the ARS scores can be effectively or safely reduced in this population. Nevertheless, acute hospitalization for any event may provide an ideal opportunity to intervene to reduce anticholinergic burden while the patient is in a monitored environment.
We describe an audit of the total number of anticholinergic drugs and the ARS scores in hospitalized elderly individuals with the principal aim of verifying whether these parameters on admission differed before and after consultant geriatrician review, and to evaluate the effect of regular calculation and consideration of ARS scores on reducing anticholinergic drug exposure.
Methods
The audit was conducted in two acute geriatric assessment wards in Aberdeen Royal Infirmary (Aberdeen, UK). The wards admit frail older patients with multiple comorbidities rather than single organ pathology, and specialize in comprehensive geriatric assessment of acutely unwell individuals. Demographic data including age, sex, place of residence, history of dementia or delirium and total number of drugs were collected prospectively into an audit database. In the absence of agreed gold standards, two audit standards were arbitrarily set, as follows: at least 90% of patients should have an ARS score of up to 2 after initial consultant review; and at least 80% of patients should have an ARS score of 0.
The first phase of the audit assessed standard practice and included 70 consecutive patients aged at least 65 years admitted to the wards between 25 April 2011 and 9 May 2011. Standard practice involved a review of all new admissions to the ward by one of the four consultant geriatricians, as well as a multidisciplinary team of therapists, including a ward pharmacist. They would undertake comprehensive geriatric assessment, which included a review of the appropriateness of prescribed medications. The assessment would culminate in a detailed plan for the ongoing care of each patient and an estimated date of discharge. The unit had a median length of stay of 5 days at the time of the audit.
To ensure prescription records were as accurate as possible, medication reconciliation was obtained by collecting information from patients, carers, electronic general practitioner medical records and medical notes. ARS score was then calculated on admission and after initial consultant geriatrician review. Briefly, the ARS score was developed by Rudolph and colleagues [Rudolph et al. 2008] following review of the 500 most frequently prescribed drugs within the Veterans Affairs Boston Healthcare System. For each drug the following information was retrieved from various databases: dissociation constant for the muscarinic receptor, rates of anticholinergic effects compared with placebo in experimental studies, and medical literature on anticholinergic adverse effects. Each drug was then ranked on a scale of 0–3 according to its anticholinergic potential [0: limited or none (e.g. atorvastatin); 1: moderate (e.g. ranitidine); 2: strong (e.g. tolterodine); and 3: very strong (e.g. amitriptyline)]. Finally, an individual's ARS score can then be calculated as the sum of the ARS rankings assigned for each of the drugs taken by the patient. Although ipratropium and tiotropium were not ranked in the original ARS score, both have significant anticholinergic effects [Casarosa et al. 2010]. An adjudicating team [one clinical pharmacologist (author AAM), and one geriatrician (author RLS)] allocated ipratropium a ranking of 3 as it is associated with a 15% incidence of dry mouth and tiotropium a ranking of 2 as it has less activity on the muscarinic receptor [Casarosa et al. 2010]. Topical medications or medications taken only on an ‘as required’ basis were excluded from ARS score calculations.
Re-audit was undertaken from 5 June 2011 to 20 June 2011. The same four consultants were told about the result of the first audit at an informal unit presentation. A list of drugs with ARS scores was then pasted on the ward round trolley and the doctors’ room wall, as a reminder of the anticholinergic drugs (Figure 1). For every new admission, the ARS score was calculated by one of the authors (HST), who was independent of the responsible clinical team. The consultant in charge of the patient was informed either verbally or in writing of their patient’s ARS score by HST in advance of their ward round and they were specifically asked to review anticholinergic medications in patients with ARS scores greater than 0. The ward pharmacist was not involved in this audit.
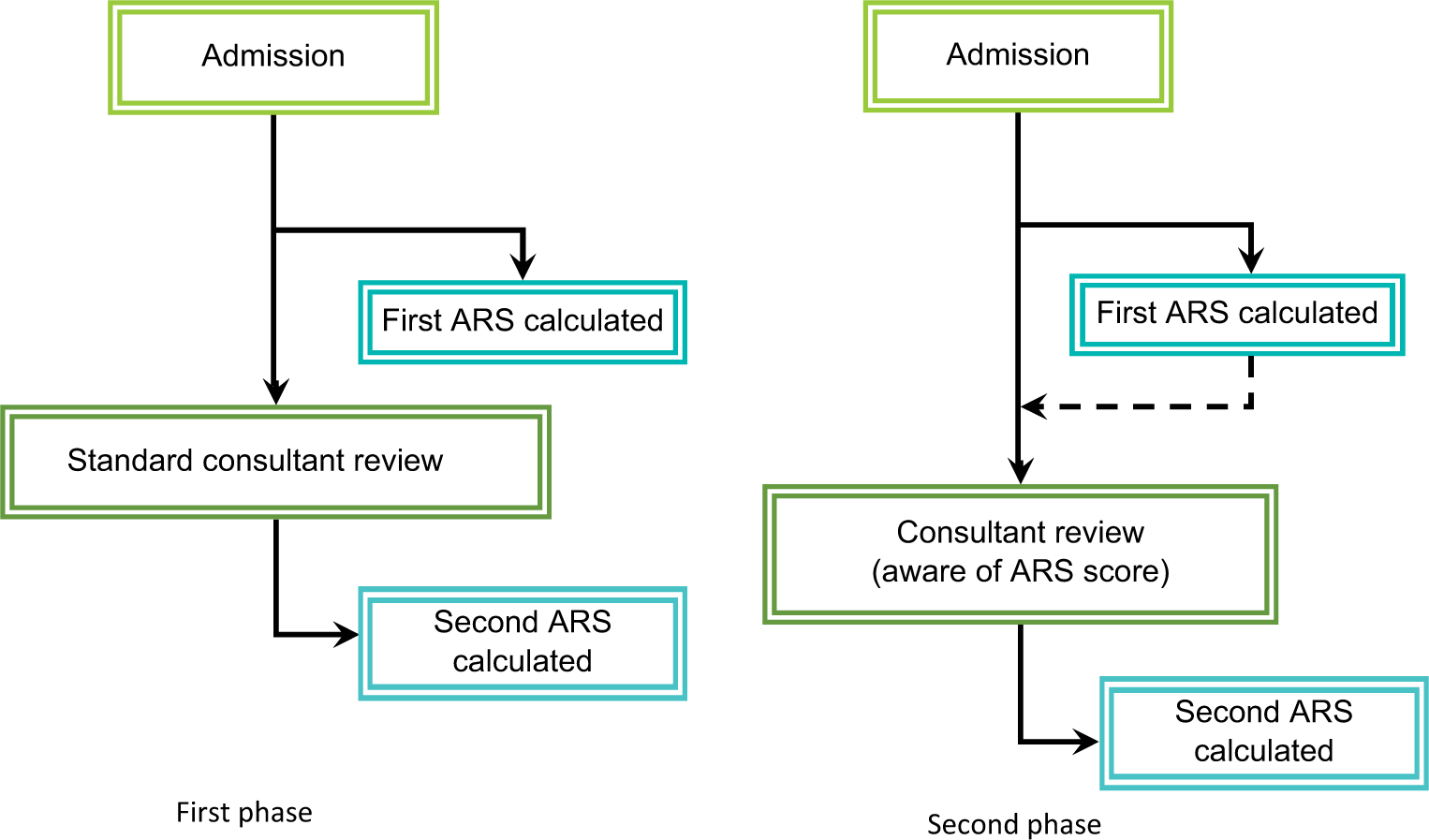
Audit design. ARS, Anticholinergic Risk Scale.
Formal ethical approval was not sought since this is not required for audit under national or local institutional guidelines.
Statistical analyses
In both phases of the audit, ARS scores before and after consultant review were compared using Wilcoxon signed rank test. Since ARS scores are insensitive to dose reductions and some anticholinergic medications should not be withdrawn suddenly, the difference in proportions of patients with medications stopped or dose reduced between the two audit arms was compared using Fisher’s exact test. Significance was assumed when p was less than 0.05. The odds ratio (OR) and 95% CI was calculated for the proportions of patients with anticholinergic medications reduced or withdrawn in the second phase relative to the first. Binary logistic backward stepwise regression analysis was carried out to identify factors independently associated with a reduction in either ARS score or dose reduction in at least one anticholinergic drug. Factors entered into the regression model were age, sex, residence (care home or own home), total number of prescribed drugs, preadmission ARS score, delirium, dementia and whether the clinician was aware of the ARS score. IBM SPSS v19.0 was used for all analyses.
Results
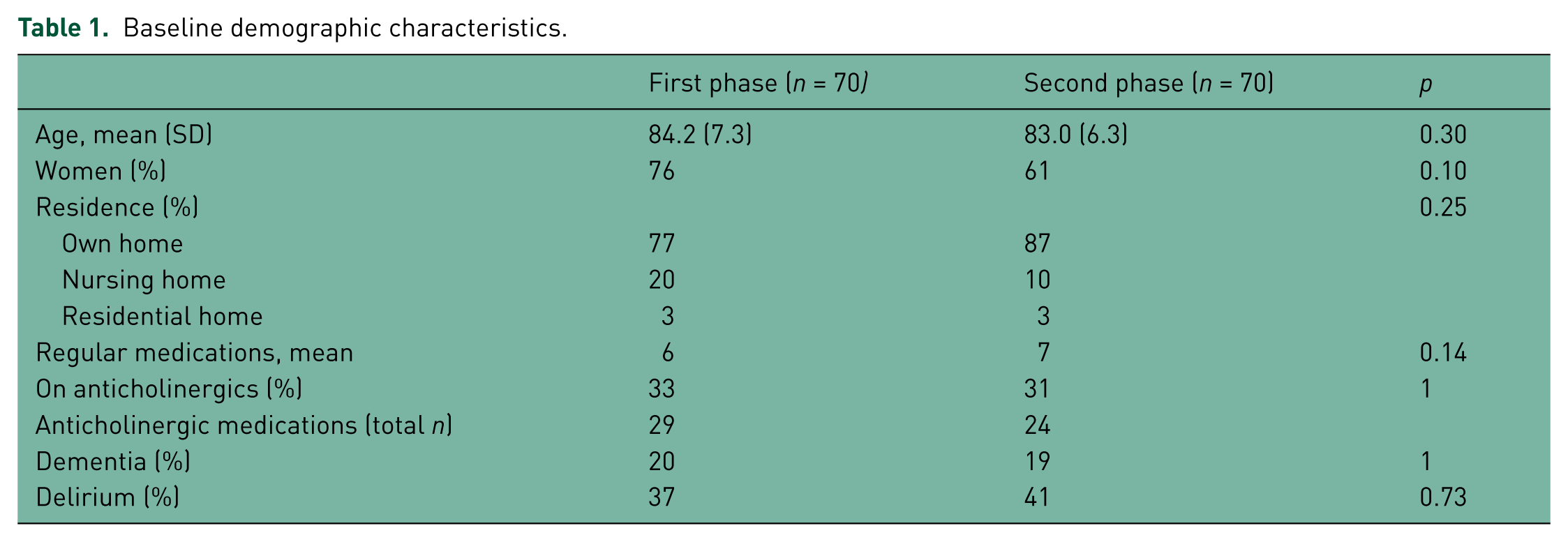
The demographic characteristics of the patients sampled are described in Table 1. There were no statistically significant differences in baseline demographics between the groups in the two phases of this audit.
Baseline demographic characteristics.
Fifty-three anticholinergic drugs were prescribed at baseline (preadmission) to 45/140 (32%) patients included throughout both phases of the audit. The most commonly prescribed anticholinergics were mirtazapine (n = 12), ranitidine (n = 7), madopar (n = 4), prochlorperazine (n = 4), olanzapine (n = 4), tiotropium (n = 3), amitryptyline (n = 2), oxybutynin (n = 2), quetiapine (n = 2), trazadone (n = 2) and tolterodine (n = 2). ARS scores fell significantly in both arms of the audit (see Table 2 and Figure 2), more so in the second arm. The proportion of patients on anticholinergics who had their medications either stopped or reduced rose significantly from 8 out of 23 (35%) in the first arm to 16 out of 22 (72%) in the second arm (OR 5.0, 95% CI 1.4–17.8). The total number of anticholinergic drugs prescribed fell from 29 to 20 in the first phase, and from 24 to 11 in the second. Both audit standards were met only during the second phase of the audit. Regression analysis showed the only independent predictors for reducing ARS score or reducing the dose of anticholinergic drugs were younger age, higher preadmission ARS score and clinician awareness of ARS score (see Table 3).
Main results.
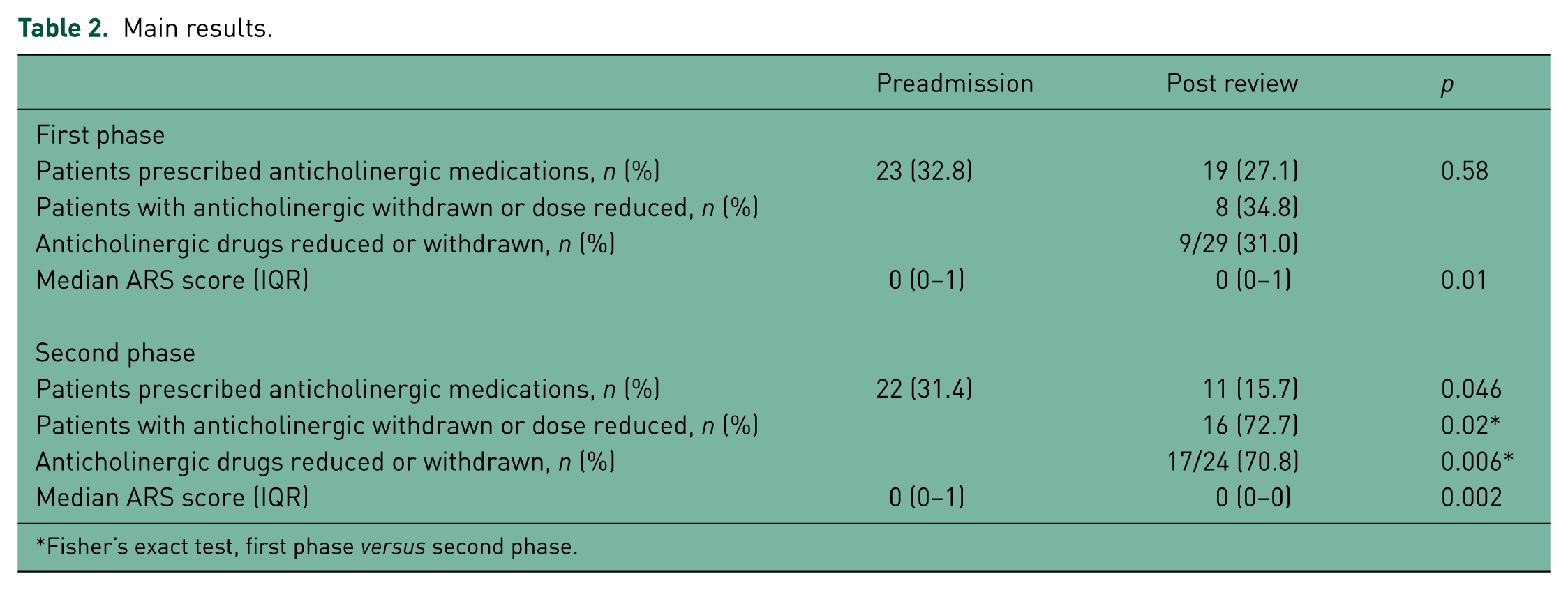
Fisher’s exact test, first phase versus second phase.
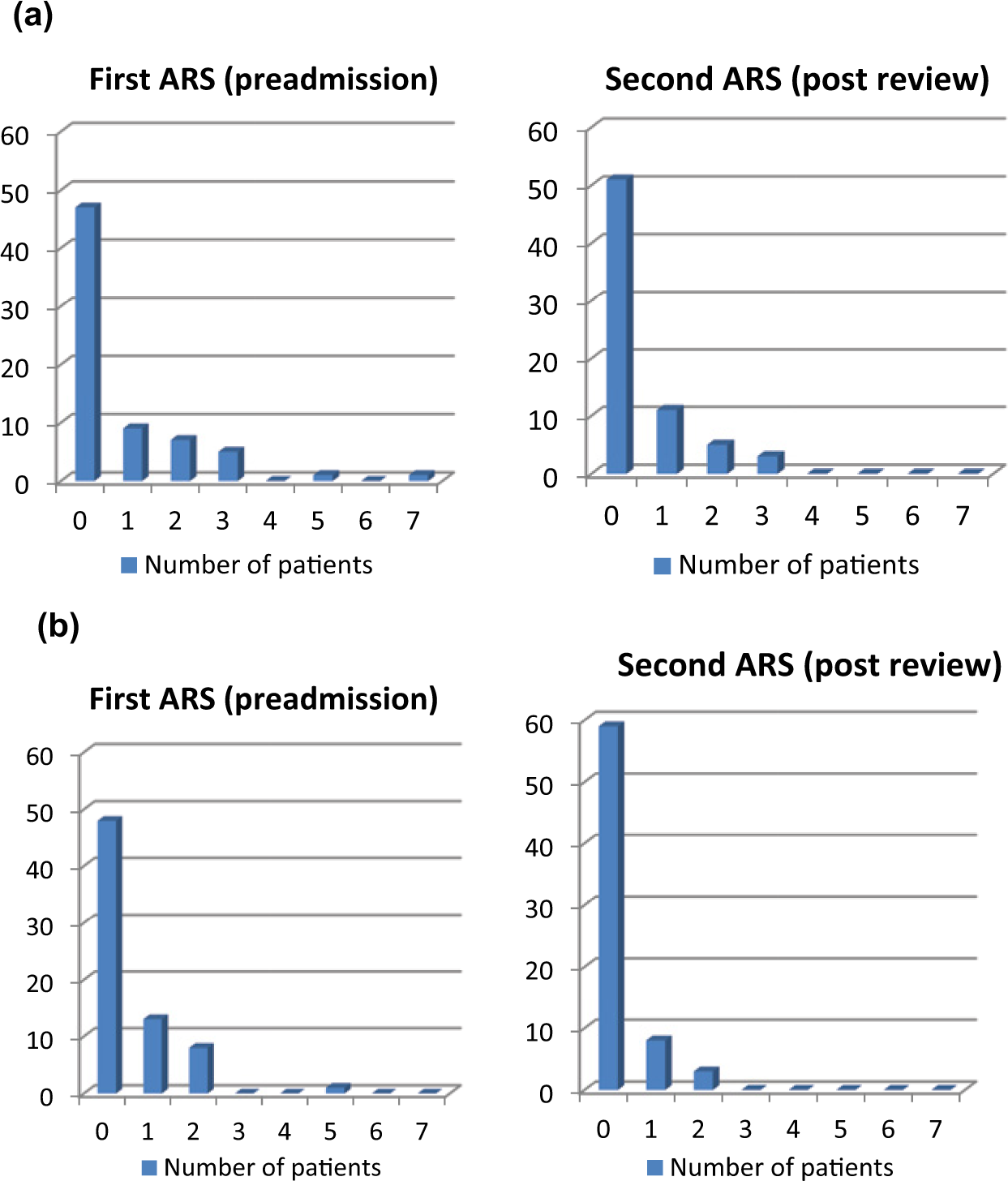
Difference in distribution of Anticholinergic Risk Scale (ARS) scores on admission and after review in (a) the first arm of the audit (p = 0.01) and (b) the second arm after institution of action plan (p = 0.002).
Logistic regression modelling of predictors of successful reduction in Anticholinergic Risk Scale (ARS) score or reduction in dose of anticholinergic drugs.

Variables removed from analysis: sex, total number of prescribed drugs, residence, delirium, dementia.
Reasons for neither stopping nor reducing anticholinergic medications in the second phase of the audit included psychiatric liaison team advice to deal with behavioural symptoms (n = 2); patient not wanting to change their medications (n = 2); fear of upsetting a planned discharge (n = 1) and consultant geriatrician thought medication still indicated (n = 1). No new anticholinergic medications were prescribed after initial review in either phase, and none of the medications stopped were restarted during the admission. Although not a prespecified endpoint, no significant safety issues were reported during the hospital stay in patients in whom anticholinergic medications were withdrawn or reduced.
Discussion
To our knowledge, this is the first study to show that consultant geriatrician review (standard practice) was effective at significantly reducing anticholinergic drug prescribing in a sample of frail, acutely ill, older patients. Moreover, anticholinergic drug exposure can be reduced further simply by alerting the clinical team to ARS scores on admission. Anticholinergic medications were commonly prescribed, even in this frail older population with a high prevalence of dementia and delirium. This is consistent with other studies in hospitalized individuals [Kosheido et al. 2012; Lowry et al. 2011a], though estimates of anticholinergic exposure in the literature vary greatly depending on the mechanism used to quantify it [Bostock et al. 2010]. The effect on ‘hard’ endpoints such as length of stay and mortality was not investigated. However, it was reassuring that no withdrawal symptoms were detected and none of the stopped anticholinergic medications were represcribed during the admission. Moreover, the effects of this simple intervention are underestimated by the ARS score because it does not take dose into account and ARS scores were only measured immediately after review. A number of drugs were signalled for gradual withdrawal and this should theoretically result in decreased ARS scores in time. The proportion of patients on anticholinergic medications who had at least one anticholinergic drug stopped or the dose reduced more than doubled after the intervention was introduced.
Our finding that it is possible to reduce anticholinergic drug exposure during admission to hospital should now clear the way for studies into its effect on important outcomes, such as length of stay in hospital, mortality, institutionalization and quality of life. A reduction in anticholinergic drug exposure should also result in lower drug prescribing costs and a reduction in healthcare costs attributable to adverse drug reactions. General practitioner-led reviews in community settings have been less successful in reducing drug burden due to clinicians’ unease with stopping medications recommended by specialists, among other barriers [Gnjidic et al. 2010]. Nishtala and colleagues demonstrated that pharmacist-conducted medication reviews can reduce prescribing of sedative and anticholinergic drugs in older people outwith hospital, resulting in a significant decrease in the Drug Burden Index [Nishtala et al. 2009]. Although it requires confirmation in a larger study, our results suggest an admission to hospital under a geriatrician might provide an ideal opportunity to deprescribe anticholinergic drugs, particularly when they are considered inappropriate or suitable alternatives exist. In this setting, a specialist multidisciplinary team could re-evaluate the risks and benefits of continuing these medications whilst the patient is in an environment where the effects of deprescribing can be monitored and assessed. Calculation of the ARS score by any member of the clinical team (e.g. ward pharmacist) and a system of alerting the supervising clinician should help maximize this opportunity. The time taken to calculate the ARS for each patient was minimal (usually <1 min). Our results complement the emerging evidence base of the feasibility, safety and efficacy of deprescribing strategies in frail older populations in a number of settings [Beer et al. 2011; Garfinkel et al. 2007; Garfinkel and Mangin, 2010; Iyer et al. 2008; Jackson et al. 2004].
Regression analysis identified three main determinants of successful deprescribing. The correlation with higher ARS score was unsurprising since such patients are most likely to experience adverse effects and have the greatest potential for medication withdrawal. The relationship between awareness of the ARS score and anticholinergic medication withdrawal probably reflects a lack of clinician awareness of the number or potency of anticholinergic medications, or may lead to an altered risk/benefit assessment away from continued prescribing of anticholinergic therapy. More surprising was the association between lower age and medication withdrawal. This might be explained by a greater prevalence of ‘genuine’ indications for anticholinergics in older patients or a fear of deprescribing in the oldest individuals, either on the part of the clinician or the patients themselves.
Our study has important limitations. It was conducted in a single centre in the context of an audit with only four consultants, and it is possible results will not be sustained or reproducible if incorporated into routine practice. This merits further study. Although we did not measure inter-individual variability either at baseline or in response to the audit intervention, anecdotal evidence from our own audit suggests there may be variation amongst clinicians in willingness to react to a higher ARS score and attempt to reduce it (one of the consultants was less likely to reduce or stop anticholinergics than the other three). Further research into the effects of anticholinergic medication deprescribing is required to assess any health benefits or detriments and the economic impact. Decisions on deprescribing in patients with multiple comorbidities require complex decisions based on perceived risks, benefits and patient needs and preferences. It is therefore unclear whether the intervention would be successful or safe in nonspecialist settings or in different populations. Nevertheless, the poor outcomes for patients on anticholinergic medications reported in multiple observational studies [Fox et al. 2011; Han et al. 2008; Landi et al. 2007; Lowry et al. 2011a] justify further research into ways of minimizing the anticholinergic drug exposure in older patients.
Conclusion
Current practice in an acute geriatric medicine ward is effective at reducing anticholinergic drug exposure. Presenting medical teams with a simple calculation of patient ARS with prompts to consider reducing this resulted in further reduction of anticholinergic drug exposure in hospitalized patients. This novel intervention could potentially form part of existing pharmacist-led reviews of prescribed medications in frail, older people. This has potential to significantly improve wellbeing and outcomes in this patient group, as well as reducing prescribing costs and other costs associated with adverse drug reactions.
Key points
(1) Anticholinergic (antimuscarinic) medications are commonly prescribed in older people despite being associated with multiple adverse effects and poor outcomes.
(2) Routine consultant geriatrician review of acute elderly admissions decreased anticholinergic drug exposure.
(3) A simple system of alerting clinicians to anticholinergic drug exposure using the ARS further decreased anticholinergic drug prescribing in this vulnerable population.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare no conflict of interest in preparing this article.
