Abstract
Background:
The digital world has undergone an essential metamorphosis in recent years, making the easy sharing of information possible, including those related to pharmacovigilance and the safety aspects of pharmaceutical and other healthcare products. These new interactive ways pose both opportunities and challenges to healthcare/pharmaceutical companies. The Pharmacovigilance Working Group “Ernesto Montagna” of the Italian Society of Pharmaceutical Medicine (SIMeF) decided to carry out a survey to gain a better understanding of the role of pharmacovigilance in digital activities.
Methods:
The Pharmacovigilance Working Group “Ernesto Montagna” sent a questionnaire via Computer-Assisted Web Interview (CAWI) technology to the members of the Pharmacovigilance Working Group (N = 257). The questionnaire was composed of 11 questions in four clusters exploring: (i) digital channels and projects implemented by the healthcare/pharmaceutical companies; (ii) governance tools in place for digital channels and projects; (iii) management of adverse events collected from digital channels and projects; (iv) impact of artificial intelligence on pharmacovigilance activities.
Results:
Ninety-three members of the Group “Ernesto Montagna” completed the questionnaire. The results show that, in the panorama of Italian healthcare/pharmaceutical companies, digital activities are ongoing, but there are still areas of uncertainty: on when a pharmacovigilance team should be involved, on the governance tools and on the guidance to be used to ensure effective governance of digital projects.
Conclusion:
In a scenario which is evolving very quickly, a critical factor is the availability of specific and updated regulations. Scientific societies, such as SIMeF and Farmindustria, the Italian national Pharma-Companies Association, could give a valuable contribution to the development of appropriate guidance together with the competent authorities.
Plain Language Summary
Background
In recent years, the digital world has undergone a revolutionary change, with the possibility for users to share contents with a virtually infinite audience easily.1,2 In the field of healthcare, the rapidly growing diffusion of electronic diagnostic and monitoring tools for health parameters as wearable devices has made it possible to share health information on medical conditions, medicines, adverse events (AEs) in a quantity and at speed unthinkable in the past.3–22 This evolution meets the desire of patients to seek and exchange health information, to treat themselves and their loved ones.4,5 This “digital revolution” soon involved pharmacovigilance (PV) and launched it into the world of Pharmacovigilance 2.0. The digital world is a potentially inexhaustible source of unsolicited safety information which is potentially of interest from a PV perspective in specific areas; 6 however, with some limitations, as it is unstructured, i.e., not subject to strict quality rules. The digital sources generate a massive amount of information overwhelming the operational capability of PV units, which are already in charge of the oversight of traditional sources. 7 The potential for analysing and managing this incessant flow of information lies in the development of artificial intelligence (AI) systems, which will help not only the identification of reports but also the control of their quality and their follow-up.8–10 Projects in this direction are being set up11,12,19,20 and are hampered by costs, by technical difficulties 21 and probably by the lack of clear regulatory guidance for the digital and social media area. Pharmacovigilance legislation, and regulations concerning other healthcare products, such as medical device, appears to be lagging behind the rapid development of the sector.13–17 Only in the Guideline on Good Pharmacovigilance Practices – Module VI (VI.B.1.1.4) 14 a mention of the reports of AEs coming from the internet and digital media and their collection and management by the Marketing Authorization Holder (MAH) could be found. The boundaries within which to move (company-sponsored versus non-company sponsored digital media) and the ways to demonstrate the actual active monitoring of activities are still not completely clear. As for Italy, there are no national guidelines providing information linked to the legislation on some areas of uncertainty, such as the limit of the responsibilities of the MAH during the collection of reports from social media, the emergence of new sources (e.g. apps able to collect AEs) and how to carry out follow-up activities. 11 Considering this background, the number and heterogeneity of projects related to the digital world and the considerable impact of digital activities on the collection of reports of AEs, the Working Group on Pharmacovigilance “Ernesto Montagna” of the Italian Society of Pharmaceutical Medicine (SIMeF) 23 considered the role of PV in the digital channels as a main emerging topic to address. The SIMeF gathers experts mainly from the pharmaceutical industry and contract research organizations (CROs) active in drug development and related disciplines. With about 1000 members, it is a significant cultural reality on a national level. The SIMeF working group, named after the late Ernesto Montagna, has been active since 1995 and is one of the most relevant PV working groups in Italy.
To investigate the role of PV in the digital area, the Working Group “Ernesto Montagna” conducted a survey aimed to describe the current status of the activities of healthcare/pharmaceutical companies and the role of PV in the oversight of the digital channels. This survey provides an updated picture of the situation in one of the five largest European countries, with a population of 60 million and with a strong pharmaceutical sector: 65,000 employees, € 32.2bn of production, composed of national and international healthcare/pharmaceutical companies. 18
The analysis of the information collected reveals the different ways through which the participating companies manage social media and digital channels and thus identifies possible areas of in-depth study and interest for further examination of the issue and for the development of future guidelines helping to provide a more consistent and homogeneous methodological approach.
Materials and methods
The SIMeF Working Group “Ernesto Montagna” designed the survey and administered the questionnaire through a computer platform using the Computer-Assisted Web Interview (CAWI) technology.
To obtain a quick and straightforward questionnaire the terms were used in their common meaning; in particular, in this article the term “AI” 24 is to be understood in the definition provided by the Oxford Dictionary (“The theory and development of computer systems able to perform tasks normally requiring human intelligence, such as visual perception, speech recognition, decision-making, and translation between languages.”)
The invitation to this survey was distributed in July 2019 to the members of the Pharmacovigilance Working Group “Ernesto Montagna” of SIMeF (257 participants). The Working Group includes members belonging to pharmaceutical companies (both international and national), medical device manufacturers, consulting companies and CROs working in the PV area (offering services like Qualified Person of Pharmacovigilance - QPPV support, case processing, PV consultants). The survey consisted of 11 questions grouped in four clusters exploring specific thematic areas: (i) digital channel and project implemented by the healthcare/pharmaceutical companies; (ii) governance tools in place for these channels and project; (iii) management of AE reports collected through these channels and project; (iv) impact of AI on PV activities. Participants had three months to answer: the last answers were returned in October 2019. This survey did not include patients and did not require any informed consent or Ethics Committee approval. Table 1 shows questions and clusters.
Clusters and questions.
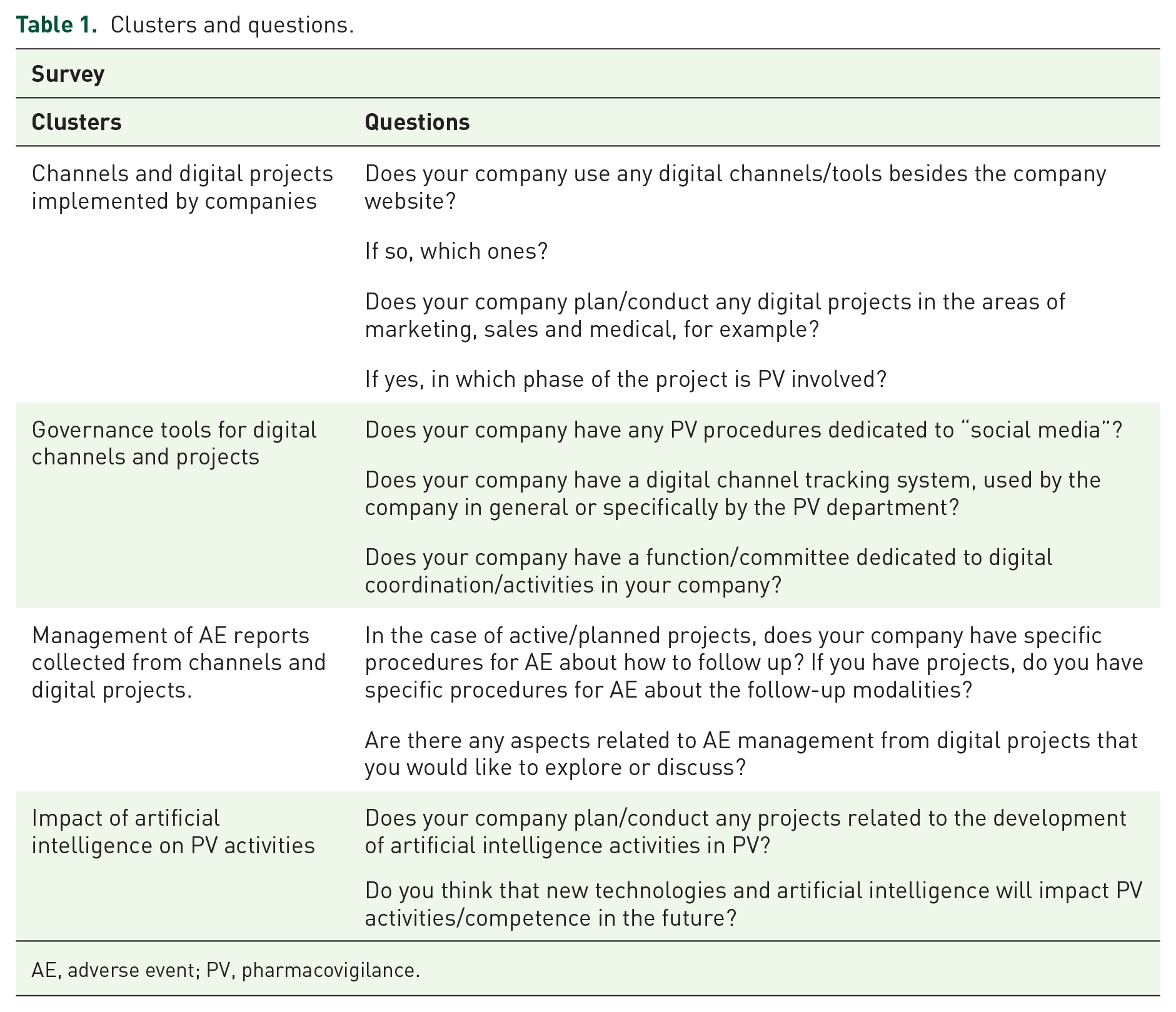
AE, adverse event; PV, pharmacovigilance.
Results
Ninety-three members answered the questionnaire (40% of members to which the questionnaire was distributed). The response rate highlights that the topic is interesting both for pharmaceutical companies (Italian headquarters – 21/93, equal to 23% of respondents – and affiliates of multinationals companies – 36/93, equal to 39%) and for those companies providing services to pharmaceutical companies (29/93, equal to 31%), as presented in Figure 1.

Companies responding to the survey (total n = 93).
The following paragraphs report the results of each cluster of questions.
Channels and digital projects
The responses to questions of the first cluster are presented in Table 2.
Channels and digital projects implemented by companies.
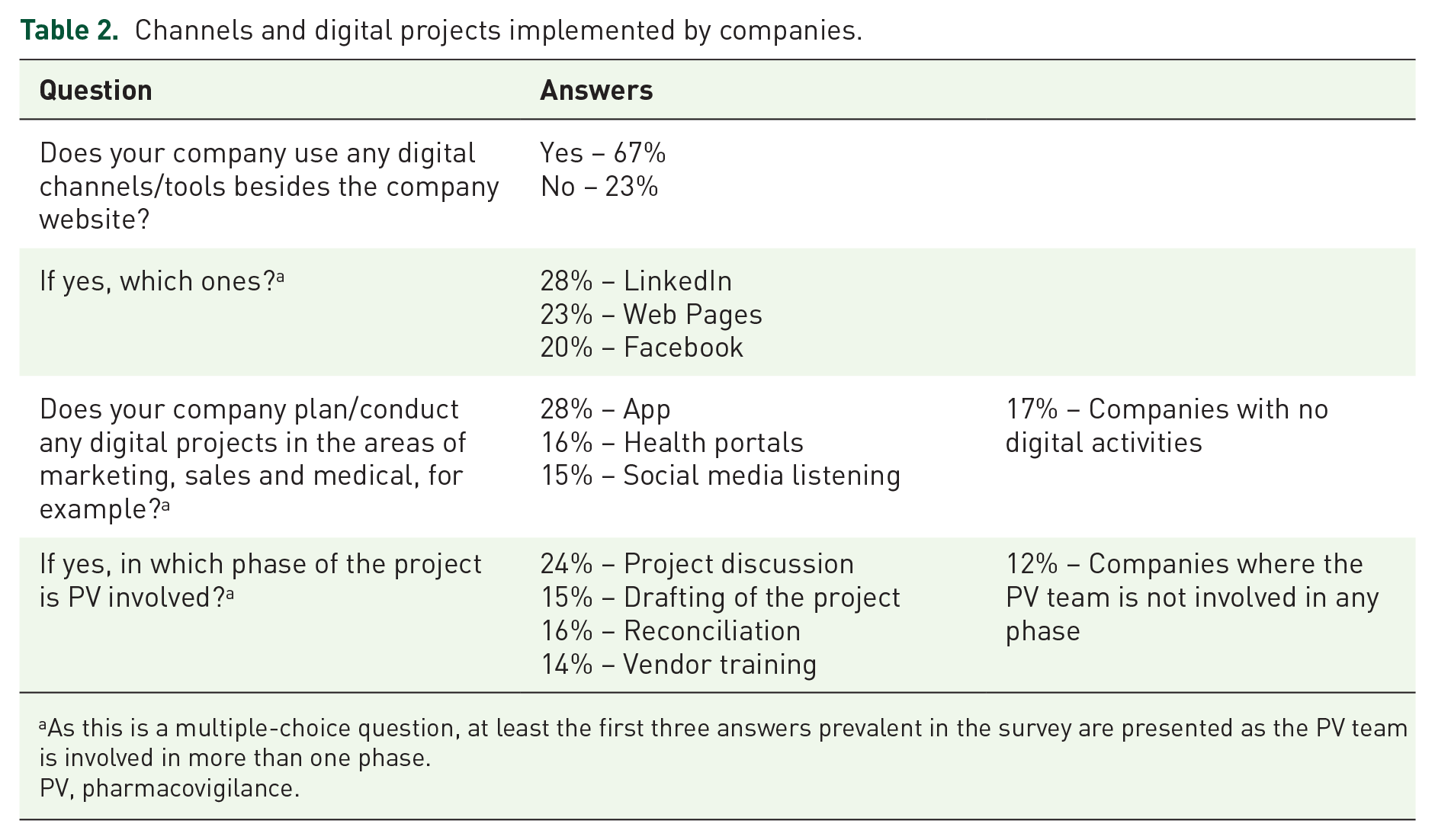
As this is a multiple-choice question, at least the first three answers prevalent in the survey are presented as the PV team is involved in more than one phase.
PV, pharmacovigilance.
On the whole, healthcare/pharmaceutical companies are very active both in institutional channels (LinkedIn, and company websites) and in specific initiatives on therapeutic areas (such as apps, health portals, social media listening) where there is a high probability of collecting AEs.
Governance of digital activities
Responses to questions of the second cluster are presented in Table 3.
Governance tools for digital channels and projects.

PV, pharmacovigilance.
More than 40% of healthcare/pharmaceutical companies do not have PV procedures focused on digital activities as well as a structured governance system and organized system for tracking/recording of digital channels; an additional 16.1% have at least some description of PV activities in different procedures.
The presented data reflect a fluid and progressively evolving situation among the companies responding to the survey. Less than 70% of the healthcare/pharmaceutical companies have (or are implementing – 11.8%) a function/committee dedicated to digital activities and that might be global (26.7%) or local (28.2%).
Management of adverse event reports collected from channels and digital projects
The responses to questions of the third cluster are presented in Table 4.
Management of adverse event reports collected from channels and digital projects.

AE, adverse event.
In total, 52% of the Companies, once the AE reports were collected, did not define through a procedure how and whether to carry out follow-up activity for the cases coming from these sources or declare to have difficulties in managing specific aspects related to this topic.
A total of 47% of the companies expressed the need to investigate aspects related to the management of the AE reports obtained from the digital channels.
Figure 2 shows the situation of the participating companies in terms of PV procedures dedicated to the follow-up of AEs from digital channels. Only 35% of the healthcare/pharmaceutical companies have in place a procedure on how to conduct follow-up for AEs received from digital activities and 13% of companies are preparing a procedure on this topic.
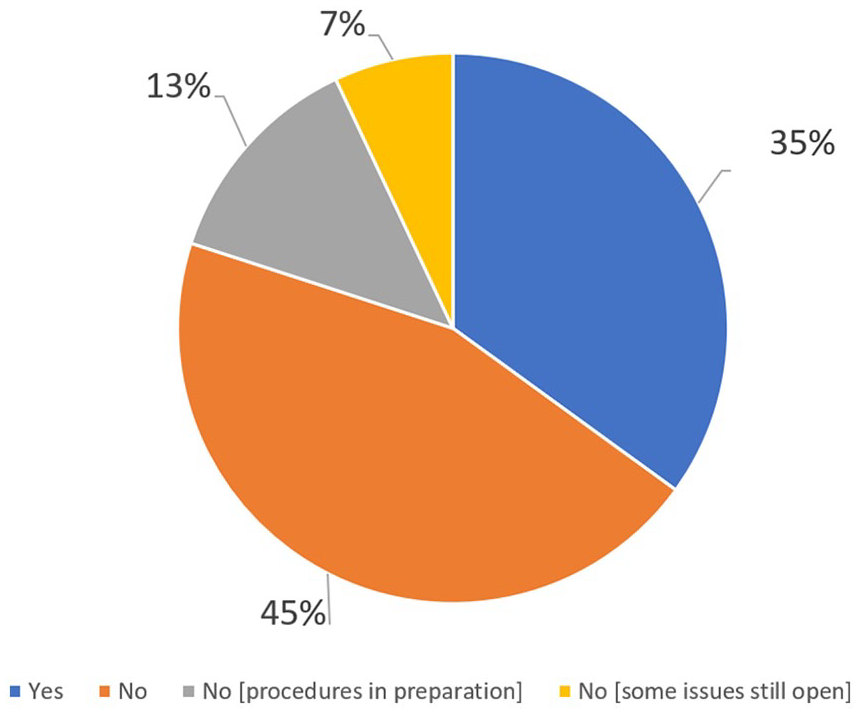
Procedures for the oversight of digital activities.
Impact of AI on pharmacovigilance activities
The responses to questions of the fourth cluster are presented in Table 5. The last cluster of questions aims to analyse the perception of healthcare/pharmaceutical companies’ workers regarding the possible impacts of AI 24 in PV activities: a significant minority (25.8%) of healthcare/pharmaceutical companies are actively working on projects with AI, while the majority of healthcare/pharmaceutical companies judge that AI will impact PV activities in the future.
Impact of artificial intelligence on pharmacovigilance activities.

PV, pharmacovigilance.
Discussion and conclusion
To our knowledge, this is the first systematic survey of the activity of Italian healthcare/pharmaceutical companies in the field of PV and digital channels and activities. One of the strengths of our research is that SIMeF is one of the leading Italian scientific societies for professionals in the field of pharmaceutical medicine. SIMeF includes companies, mainly from the pharmaceutical industry, and CROs operating in Italy; therefore, these results can be reasonably considered representative of the situation in Italy in terms of numbers, distribution over the territory and relevance of the companies involved. Another strength of our research is the CAWI methodology, which allowed a straightforward collection of data, minimizing the mistakes and maximizing the participation of the companies in the survey. The simplicity of the tool used somehow limited the detail of the data; this was necessary to obtain a high response rate to the questionnaire. In this way, we think we achieved the main objective of the survey, which is to get a first-hand overview of the situation from personnel directly involved in PV activities. The above mentioned strength is also a limitation of our research: on one hand this may provide responses which accurately reflect the situation; however, this may not provide information on the opinion of the higher management, which might provide insight into the steps that the companies are planning. The overall result of our survey returns contrasting evidence. There is a broad consensus of the involvement of PV in digital channels and on the relevance of digital activities as a source of valuable information and the future impact of the AI. Nevertheless, if we consider the results presented in Table 2, despite many companies being active with digital channels and conducting digital projects, early PV involvement (project discussion or drafting of the project) occurs in less than 40% of the healthcare/pharmaceutical companies and even not at all in 12% of them.
Focusing on governance aspects, about a half of the participants have already in place or under preparation procedures dedicated to digital activities and more than a half have function/committee (global or local) dedicated to ensuring the oversight of digital projects/channels (as shown in Table 3). Nevertheless, for about 40% of the healthcare/pharmaceutical companies pooled, the approach to digital aspects remains theoretical, without procedures or a tracking system dedicated to the oversight of digital activities (as presented in Table 3).
The main reason for the incomplete development of the abovementioned governance systems might be the rapid growth of the digital area in the absence of specific guidance from the competent authorities.
As a consequence, the companies still have doubts also on specific technical aspects, such as follow-up of AEs from digital channels; in fact 47% of the respondents declared they would like to explore more in depth the aspects of AE management (see Table 4).
As often happens, problems generate opportunities, and we think that, in this context, pharma-companies associations (such as Farmindustria) and scientific associations (such as SIMeF) should cooperate in the preparation of structured guidelines. This approach could help the healthcare/pharmaceutical companies to effectively manage PV processes related to digital activities. In addition, these guidelines could be discussed with the competent authorities to shape future PV in the digital area. This contribution can be even more valuable, now that the COVID-19 pandemic has accelerated digitalization in health care activities.
Footnotes
Acknowledgements
Mrs Sabrina Lucioni of SIMeF secretariat supported the authors in the organization and conduct of the survey.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
