Abstract
Hypertension is the leading cause of cardiovascular (CV) morbidity and mortality in adults over the age of 65. The first part of this paper is an overview, summarizing the current guidelines on the pharmacological management of hypertension in older adults in Europe and the USA, and evidence from key trials that contributed to the guidelines. In the second part of the paper, we will discuss the major challenges of managing hypertension in the context of multimorbidity, including frailty, orthostatic hypotension (OH), falls and cognitive impairment that are associated with ageing. A novel ‘BEGIN’ algorithm is proposed for use by prescribers prior to initiating antihypertensive therapy to guide safe medication use in older adults. Practical suggestions are highlighted to aid practitioners in making rational decisions to treat and monitor hypertension, and for considering withdrawal of antihypertensive drugs in the complex older person.
Background
Cardiovascular disease (CVD), primarily stroke and coronary heart disease, causes an estimated four million deaths per year in Europe of which over 80% are in older people aged over 65 years [Townsend et al. 2015]. It has been reported that the mortality risk from stroke and coronary heart disease doubles for each 20 mmHg systolic blood pressure (SBP) increase or 10 mmHg diastolic blood pressure (DBP) increase over 115/75 mmHg [Jones and Hall, 2004].
Despite the widespread use of antihypertensives in older adults, there are notable differences between international guidelines and expert opinion on whom to treat, at what stage, to what target and with which drug class. There have been several large placebo-controlled, randomized controlled trials to investigate optimal management of hypertension in older adults; however, there has only been one trial specifically investigating the very old (over 80 years of age) [Beckett et al. 2008]. Several existing trials have recruited participants that have fewer comorbidities than the general older population [Van Spall et al. 2007; Benetos et al. 2016; Barnett et al. 2012]. The optimal treatment of hypertension in the context of multimorbidity, frailty, orthostatic hypotension (OH), falls and cognitive impairment is particularly challenging and is commonly encountered in caring for the older patient.
What are the key guidelines from Europe and the USA to guide the pharmacological management of hypertension in older adults?
Key recommendations focusing on the older population in European and American guidelines are summarized in Table 1 [National Institute for Health and Clinical Excellence, 2011; Mancia et al. 2013; James et al. 2014; Weber et al. 2014]. For a comprehensive review of international hypertension guidelines, we direct the reader to a recent systematic review [Alhawassi et al. 2015]. For the very old (those over the age of 80), guidelines from Europe recommend that pharmacological treatment commences with an SBP ⩾ 160 mmHg, which is in contrast to American guidelines that recommend initiating pharmacological therapy at SBP ⩾ 150 mmHg.
International hypertension guidelines.
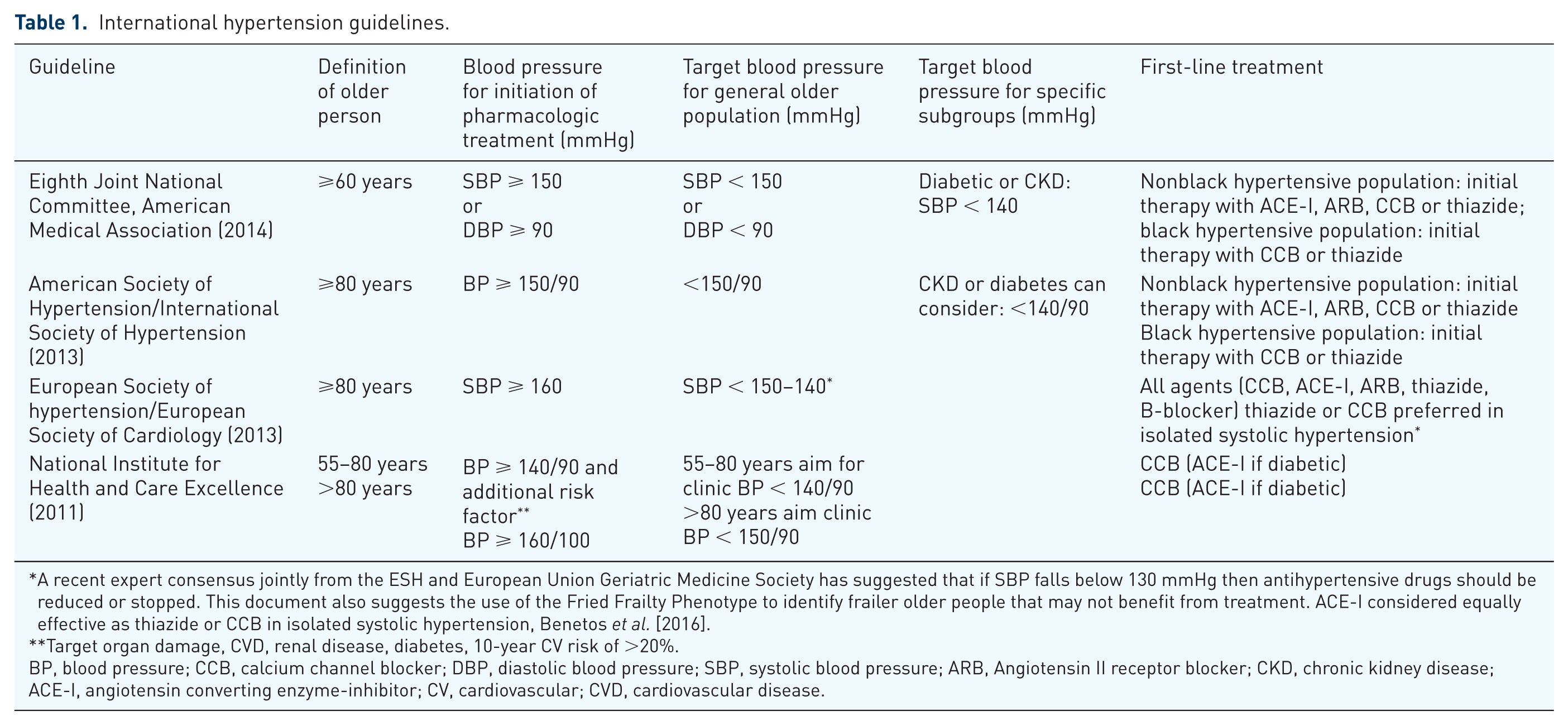
A recent expert consensus jointly from the ESH and European Union Geriatric Medicine Society has suggested that if SBP falls below 130 mmHg then antihypertensive drugs should be reduced or stopped. This document also suggests the use of the Fried Frailty Phenotype to identify frailer older people that may not benefit from treatment. ACE-I considered equally effective as thiazide or CCB in isolated systolic hypertension, Benetos et al. [2016].
Target organ damage, CVD, renal disease, diabetes, 10-year CV risk of >20%.
BP, blood pressure; CCB, calcium channel blocker; DBP, diastolic blood pressure; SBP, systolic blood pressure; ARB, Angiotensin II receptor blocker; CKD, chronic kidney disease; ACE-I, angiotensin converting enzyme-inhibitor; CV, cardiovascular; CVD, cardiovascular disease.
The key evidence that underpins the guidelines in Table 1 and shows cardiovascular (CV) risk reduction and mortality benefit of blood-pressure lowering in hypertensive older adults comes from randomized placebo-controlled trials conducted over the last 35 years (Table 2) [Amery et al. 1985; Dahlöf et al. 1991; Shep Cooperative Research Group, 1991; Medical Research Council Working Party, 1992; Staessen et al. 1997; Beckett et al. 2008]. These trials clearly demonstrate the benefit of lowering blood pressure in healthy older adults who have a systolic blood pressure (BP) ⩾ 160 mmHg.
Key Randomized trials comparing antihypertensive use to placebo in older adults.
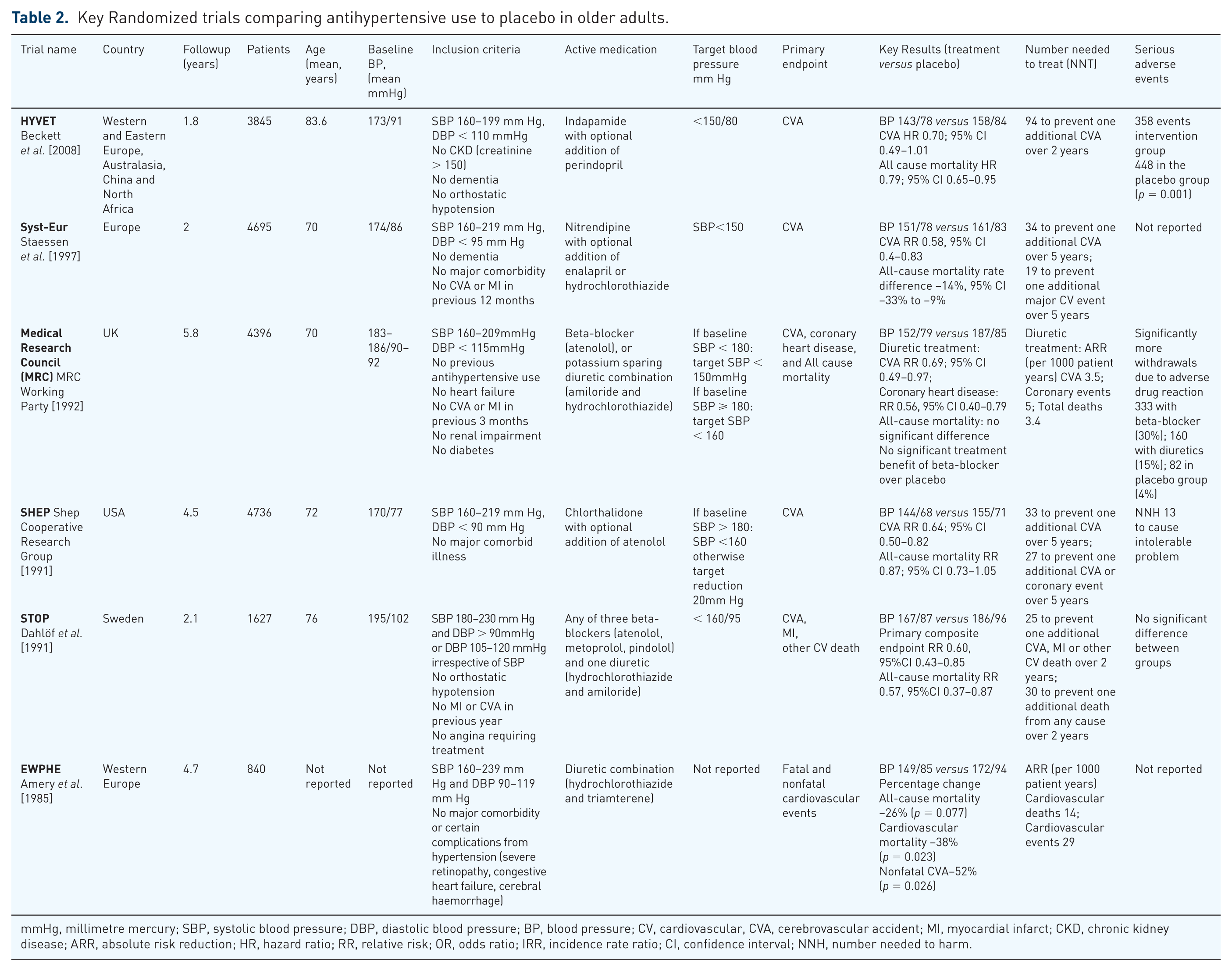
mmHg, millimetre mercury; SBP, systolic blood pressure; DBP, diastolic blood pressure; BP, blood pressure; CV, cardiovascular, CVA, cerebrovascular accident; MI, myocardial infarct; CKD, chronic kidney disease; ARR, absolute risk reduction; HR, hazard ratio; RR, relative risk; OR, odds ratio; IRR, incidence rate ratio; CI, confidence interval; NNH, number needed to harm.
Table 2 is not a systematic overview of all international trials that have investigated the effect of blood-pressure lowering on CV outcomes in hypertension. For a more detailed review of the international trials, we direct the reader to a recent systematic review and meta-analysis by Ettehad and colleagues [Ettehad et al. 2016]. This meta-analysis found relative risk reductions from lowering SBP by 10 mmHg, including baseline BP < 160 mmHg, of major CVD events by 20%, stroke by 27%, coronary heart disease by 17%, heart failure by 28% and all-cause mortality by 13%. These results compare with a previous meta-analysis of BP-lowering randomized controlled trials involving hypertensive patients up to the year 2014, which found relative risk reductions from lowering blood pressure by 10/5 mmHg (SBP/DBP) of 36%, 16% and 11% for stroke, coronary heart disease and all-cause mortality, respectively [Thomopoulos et al. 2014].
It should be borne in mind that the absolute risk reduction is highly dependent on the baseline CV risk of the hypertensive patient [Sundstrom et al. 2014]. The blood-pressure-lowering-treatment trialists’ collaboration found that the number needed to treat (NNT) for 5 years to prevent one CV event ranged from 26 in those with highest risk (>21% 5-year CVD risk), up to 71 in those with lowest CVD risk (<11% 5-year risk of CVD).
What target blood pressure should we aim for in people aged 65 years and over?
The evidence is mixed on the optimal blood pressure for hypertensive older adults and is a patient-specific decision to be made based on expected therapeutic benefit (if CV risk is relatively low then the benefits of intensive lowering are substantially lower), and patient tolerance of antihypertensive therapy. A recent meta-analysis showed that more
Nonetheless, the convincing evidence for benefit for intensive lowering of blood pressure does not answer the widely debated question of: to what target should blood pressure be lowered in older adults? The SPRINT trial has demonstrated CV and all-cause mortality benefit in lowering SBP to <130 mmHg in adults over the age of 75 with high CV risk [Wright et al. 2015]. The investigators of the SPRINT trial recommend lowering BP to SBP < 120 mmHg in older adults, although the average SBP achieved in those over the age of 75 that were intensively treated was 123 mmHg. Therefore, ‘how low to go?’ remains unclear, and whilst it is reasonable to expect a cut-off below which blood-pressure lowering with antihypertensives increases the risk of mortality [Mancia and Grassi, 2014; Verdecchia et al. 2014], no RCTs have shown this to date.
The complexity of managing hypertension in older adults
Hypertension management in the older population is often complicated by the multiple pathologies associated with ageing. Although there are numerous clinical challenges in this population, five major challenges in the context of the older hypertensive patient are multimorbidity, frailty, OH, falls, and cognitive impairment. These conditions commonly overlap with one another. This following section will discuss these challenges and offer some practical suggestions to rationalize hypertension management in the older population.
Hypertension and multimorbidity
Multimorbidity is defined as the presence of two or more long-term conditions [Fortin et al. 2005]. The risk of multimorbidity increases with age, and its prevalence is estimated at 65% in those aged 65–84 years and increases to 82% in those aged over 84 years [Barnett et al. 2012].
Hypertension features very commonly in a multimorbidity setting, as both conditions are common among older adults. Indeed, at least two thirds of hypertensive patients have another long-term condition [Barnett et al. 2012].
It has become increasingly clear that managing hypertension in isolation is not conducive to achieving a patient-centred approach for older adults [Benetos et al. 2015b]. However, there is limited available evidence to support hypertension management within a multimorbidity framework [Onder et al. 2014].
The Criteria to Assess Appropriate Medication Use Among Elderly Complex Patients (CRIME) project highlighted the lack of guidelines for the treatment of multimorbidity associated with ageing, and the close association with polypharmacy through a prescribing cascade [Onder et al. 2014].
Polypharmacy is independently associated with poor health outcomes including falls, electrolyte disturbance, heart failure, raised blood pressure, hospitalization and premature mortality [Gnjidic et al. 2012; Mukete and Ferdinand, 2016]. Polypharmacy is often associated with suboptimal prescribing regimens, as the affected population is more likely to both take at least one potentially inappropriate medication and to not take an indicated medication [Kuijpers et al. 2008; Belfrage et al. 2015]. For instance, antihypertensives commonly contribute to inappropriate polypharmacy in older people with life-limiting illness [Todd et al. 2016]. It is important, therefore, to consider the role and the place of each medication, its risk-to-benefit profile and whether it is consistent with the individual’s health care goals. Polypharmacy is not a reason to avoid prescribing an antihypertensive but it is an indicator to consider a medication review and closely monitor the effects of treatment.
Expert consensus from the CRIME project made the following key recommendations for the management of hypertension in the older person with multiple comorbidities:
In patients with dementia, cognitive impairment or functional limitation: Tight blood pressure control (<140/90 mmHg) is not recommended Use of more than three antihypertensives should be avoided.
In patients with a life expectancy of fewer than 2 years: Tight blood pressure control (<140/90 mmHg) is not recommended.
In patients with symptomatic OH or falls with OH: Number of antihypertensives should be reduced Multiple antihypertensives should be avoided.
Clinical expertise and judgement are needed to prevent a fragmented care plan. It is likely that a geriatrician or general practitioner (GP) with special interest in older adults is best placed to deliver a holistic and individually tailored care plan. Performing a comprehensive geriatric assessment (CGA) is paramount to a patient-centred approach. The CGA is an evidence-based multidimensional and multidisciplinary approach for assessing the complexities of care required by an older person [Benetos et al. 2015b]. This approach combines a medical, psychological, social, environmental and functional assessment to deliver a holistic care plan, and ideally involves a team of specialists including a geriatrician (or GP with a special interest), a specialist nurse, an occupational therapist, a physiotherapist, a social worker and a pharmacist. It has been shown that a CGA can improve the appropriateness of prescriptions and reduce the risk of adverse drug reactions in older adults with multimorbidity and polypharmacy [Schmader et al. 2004].
Case study
Mr B is a 76-year-old retired builder. Mr B was diagnosed with hypertension 12 years ago, and has the following conditions: chronic obstructive pulmonary disease (COPD), heart failure, mild cognitive impairment and osteoarthritis. Mr B is currently prescribed a calcium channel blocker (CCB) and a loop diuretic, both to be taken daily in the morning. He has been provided with a week’s supply of corticosteroid for exacerbations of his COPD to be taken when needed. He took a 5-day supply of 40 mg prednisolone daily for a week. He started taking nonsteroidal anti-inflammatory drugs (NSAIDs) bought from his local pharmacy following a recent flare of his knee arthritis. His wife was concerned at his high BP readings at home over the last 4 days (SBP 170–195 mmHg). Mr B was unable to visit his GP due to the pain in his knees. Mr B’s wife has noticed that her husband is increasingly forgetful of taking his morning medications, and often resorts to taking them in the evening.
Mr B exemplifies the commonly encountered complexities of managing the older person with hypertension and multimorbidity. Mr B’s recent high BP readings are multifactorial. A recent course of corticosteroids and NSAIDs have contributed to an increased BP secondary to fluid retention [Mukete and Ferdinand, 2016]. He was only intermittently taking his antihypertensives, as he attributed worsening ankle swelling to them. He was missing doses of his morning diuretic and occasionally making this up in the evening when he remembered, and subsequently making frequent trips to the bathroom at night.
A practical management approach
A CGA approach is recommended. Depending on the local healthcare system, either a GP with special interest in older people or a hospital geriatrician should take overall responsibility for planning, instigating and evaluating the management plan for Mr B. The following actions might be considered:
Actions to optimize the use of medicines
Exercise caution in the use of NSAIDs due to the potential for drug–drug and drug–disease interactions. Encourage use of paracetamol and refer for physiotherapy.
Consider switching the CCB to an angiotensin converting enzyme-inhibitor (ACE-I) given the beneficial effect of ACE-I on secondary prevention in heart failure, and the common adverse drug reaction to the calcium channel blocker (ankle swelling).
If the patient’s heart failure is stable, consider commencing a beta-blocker (e.g. bisoprolol, carvedilol or sustained-release metoprolol).
Follow up Mr B within 14 days to review his tolerance to the medication changes, to check his blood pressure and renal function. This is feasible in a nurse-led hypertension clinic in some settings [Ali et al. 2011; Clark et al. 2010].
Actions to educate Mr B and his wife about safe medicine use
Discussion with Mr B and his wife about overall management plan through shared decision-making process including blood pressure targets, goals of treatment, and other relevant healthcare professionals involved in care.
Refer Mr B and his wife to their community pharmacist for a discussion about whether a medication-compliance aid (MCA) could be helpful for them.
Educate Mr B and his wife that it is best to take the diuretic before 2 pm to avoid sleep interference.
Educate Mr B and his wife about the side effects of corticosteroids, particularly in relation to his hypertension.
Encourage Mr B to talk to his health professional (e.g. GP, geriatrician, pharmacist) prior to using any over-the-counter (OTC) medications.
Hypertension and frailty
Frailty is a common condition of increased vulnerability to physical stressors as a result of reduced physiological reserve and is closely related to the ageing process [Poudel et al. 2013]. An estimated 25–50% of people aged 85 years and older are frail [Buckinx et al. 2015].
Whilst it remains unclear if blood-pressure lowering is beneficial to all older people, it is the population of frail older people in whom there appears to be potential risks of causing harm by lowering high blood pressure [Odden et al. 2015].
The Milan 75+ study of more than 1500 community-dwelling participants attending geriatric outpatient clinics found that mortality risk had an inverse linear relationship to blood pressure below SBP 180 mmHg frailer patients (impaired functional and cognitive capacity) [Ogliari et al. 2015]. Lowest mortality was reported at BP 165/85 mmHg, and a U-shaped curve was demonstrated around this nadir. Indeed, participants with an SBP < 120 mmHg had a 64% greater relative risk of mortality compared with those with an SBP in the range 160–179 mmHg [Ogliari et al. 2015]. The Predictive Values of Blood Pressure and Arterial Stiffness in Institutionalized Very Aged Population (PARTAGE) study of nursing home residents older than 80 years (1127 participants) found strong evidence to show the harms of lowering SBP < 130 mmHg using two or more antihypertensives [Benetos et al. 2015a]. Compared with other participants in the 2 years follow up, those that were being treated with multiple antihypertensives lowering BP to <130 mmHg were estimated to be 78% more likely to die [hazard ratio (HR) 1.78, 95% confidence interval (CI), 1.34–2.37
Benetos and colleagues suggest a straight-forward approach to the management of the frail older person given the paucity of evidence [Benetos et al. 2015b]. They recommend a CGA in any older patient recognized as frail, followed by a cautious approach of low-dose monotherapy for hypertension > 160 mmHg SBP with a goal of reducing this to SBP < 150 mmHG. If SBP drops below 130 mmHg, then consider reducing antihypertensive treatment using a safe approach to ‘deprescribing’ [Scott et al. 2015; Page et al. 2016a]. Indeed, a meta-analysis of RCT data for hypertension treatment in the over 80 s showed that lowest-intensity antihypertensive therapy with least reduction in BP resulted in a significant total mortality reduction [Bejan-Angoulvant et al. 2010]
Hypertension and OH
OH, defined as drop in SBP ⩾ 20 mmHg or drop in DBP ⩾ 10 mmHg within three minutes of standing from sitting or supine position, is commonly seen in older people with hypertension. The prevalence of OH in hypertensive older adults differs substantially between observational studies. The EPICARDIAN population-based Spanish study reported the prevalence of OH as 8.1% amongst hypertensive older adults [Saez et al. 2000], whilst a study of community-dwelling British women reported a prevalence of OH of 79% in hypertensive women aged 60–80 years [Kamaruzzaman et al. 2010]. The use of antihypertensives is commonly implicated in OH [Kamaruzzaman et al. 2010]. However, knowing that hypertension is of itself an important risk factor for OH, the coexistence of these conditions can pose a dilemma for clinicians. Symptomatic OH may manifest as dizziness, light-headedness and syncope and thus is a major cause of falls in the older person [Rubenstein et al. 1990]. In the MOBILIZE Boston study of more than 700 community-dwelling older people, OH in the context of uncontrolled hypertension (BP > 140/90) was associated with a 2.5 times greater risk of falls than in those with uncontrolled hypertension but without OH [Gangavati et al. 2011]. Nonetheless, a relaxation in BP goals, combined with a stepwise approach to discontinuing antihypertensives if three or more are prescribed is a sensible approach [Onder et al. 2014]. Renin–angiotensin system blockers and selected CCBs are less likely to cause OH in comparison to other antihypertensive classes and therefore may be considered an appropriate switch. Splitting doses and night-time dosing of one antihypertensive, if more than one required, may help to alleviate symptomatic OH. Other medication classes can precipitate OH, for example, tricyclic antidepressants, therefore discontinuing or reducing antihypertensive drugs should be considered within a wider medication assessment.
Hypertension and falls
Falls are a major cause of harm to older people and usually take place in the context of multiple risk factors, including impaired balance, OH and polypharmacy [Rubenstein, 2006]. Evidence for a direct association between the use of antihypertensives and falls in older adults has been conflicting [Leipzig et al. 1999; Woolcott et al. 2009; Zang, 2013]. A 2009 meta-analysis investigated the main drug classes associated with falls in older adults [Woolcott et al. 2009]. It found that the use of antihypertensives in those older than 75 years was not associated with falls, whilst there was an association in those included studies with an average age less than 75 [Woolcott et al. 2009]. A nationally representative sample of older US citizens (mean age 80 years) found that antihypertensive medication use is not associated with risk for a first fall [Tinetti et al. 2014]. However, for older adults with a history of falls, the use of antihypertensives more than doubles the risk of a serious fall-related injury. It also appears that the risk of falls and serious injury, for example, hip fracture with use of antihypertensives, is partly dependent on the duration for which patients have been using the medication [Tinetti et al. 2014]. Older patients recently started (last 8 weeks) on antihypertensives or had their treatment intensified seem to be at the highest risk of falls and hip fracture due to hypotension [Butt et al. 2012; Shimbo et al. 2016]. In contrast, over the longer term, there is evidence to suggest that some classes of antihypertensive drugs (calcium-channel blockers, thiazide-like diuretics and beta-blockers) can be protective against the risk of hip fracture, and this was also indicated in the HYVET trial [Peters et al. 2010; Ruths et al. 2015]. These complex associations require further epidemiological and biomedical investigation.
Researchers investigating the preferences for using antihypertensives in older people with hypertension and falls risk, found that approximately half the older people surveyed preferred to take antihypertensive medications to reduce CV risk at the expense of increasing their falls risk, whilst the other half would avoid the medicines [Tinetti et al. 2008].
Hypertension and cognitive impairment
The risk of both hypertension and dementia increases with advancing age [Kennelly et al. 2009]. The prevalence of dementia in the over-65 population is estimated at 6.5% in the UK, and the prevalence of milder cognitive impairment is likely to be higher [Banerjee, 2013].
Systematic review of observational studies and expert consensus has shown that hypertension through midlife is an important modifiable risk factor for the development of dementia [Deckers et al. 2015], and pooled analysis found that midlife hypertension increases the risk of dementia by 61% [Barnes and Yaffe, 2011]. Uncertainty remains, however, as to whether blood-pressure lowering with antihypertensives can modify the risk of developing dementia in hypertensive patients with no prior cerebrovascular disease [McGuinness et al. 2009; Hughes and Sink, 2016]. The Syst-Eur placebo-controlled trial indicated that long-acting CCBs could reduce the risk of developing dementia by 55% in older hypertensive patients [McGuinness et al. 2009]. Meanwhile, the HYVET and MRC trials, in which the primary active treatment was a diuretic, demonstrated no significant risk reduction in dementia incidence [McGuinness et al. 2009].
Older adults with dementia are at higher risk of OH, polypharmacy, including adverse drug reactions, and frailty [Welsh et al. 2014]. This increased vulnerability, along with an impaired capacity to share in the decision-making process, means that prescribing antihypertensives in this population must be done very cautiously. In the absence of guidance, as mentioned above, the CRIME panel recommended avoidance of stringent BP targets and polypharmacy in older adults with cognitive impairment [Onder et al. 2014].
The CV benefit of treating hypertension in older adults shown in Table 2 cannot be extrapolated to people with dementia, given their under-representation in trials. A systematic review of observational studies of hypertension treatment in people with dementia reported hypertension as a comorbidity in 45% of people with dementia, and 73% were prescribed treatment [Welsh et al. 2014]. Diuretics were the most commonly prescribed treatment [Welsh et al. 2014], despite evidence suggesting that these are amongst the least well tolerated antihypertensive class given their side effect of excessive urination [Tedla and Bautista, 2016]. Calcium channel blockers and ACE-Is appear to offer some protection against cognitive decline in older hypertensive patients [Tedla and Bautista, 2016], and given the relative intolerance of patients to diuretics, it may be sensible to avoid diuretics as a first-line antihypertensive for people with coexisting hypertension and dementia.
Suggestions that can support safe medication use more broadly in patients with cognitive impairment are made in a later section of this paper.
Which antihypertensive class should be used in older adults?
Calcium-channel blockers and ACE-I are the most commonly prescribed antihypertensive classes in the older population [Jarari et al. 2015]. Nevertheless, meta-analyses have shown that there is not one antihypertensive class that is superior to others for all patients, and the benefit of antihypertensive therapy in reducing the risk of stroke and major CV events is largely driven by the lowering of blood pressure
Comorbidities that influence choice of antihypertensive. *
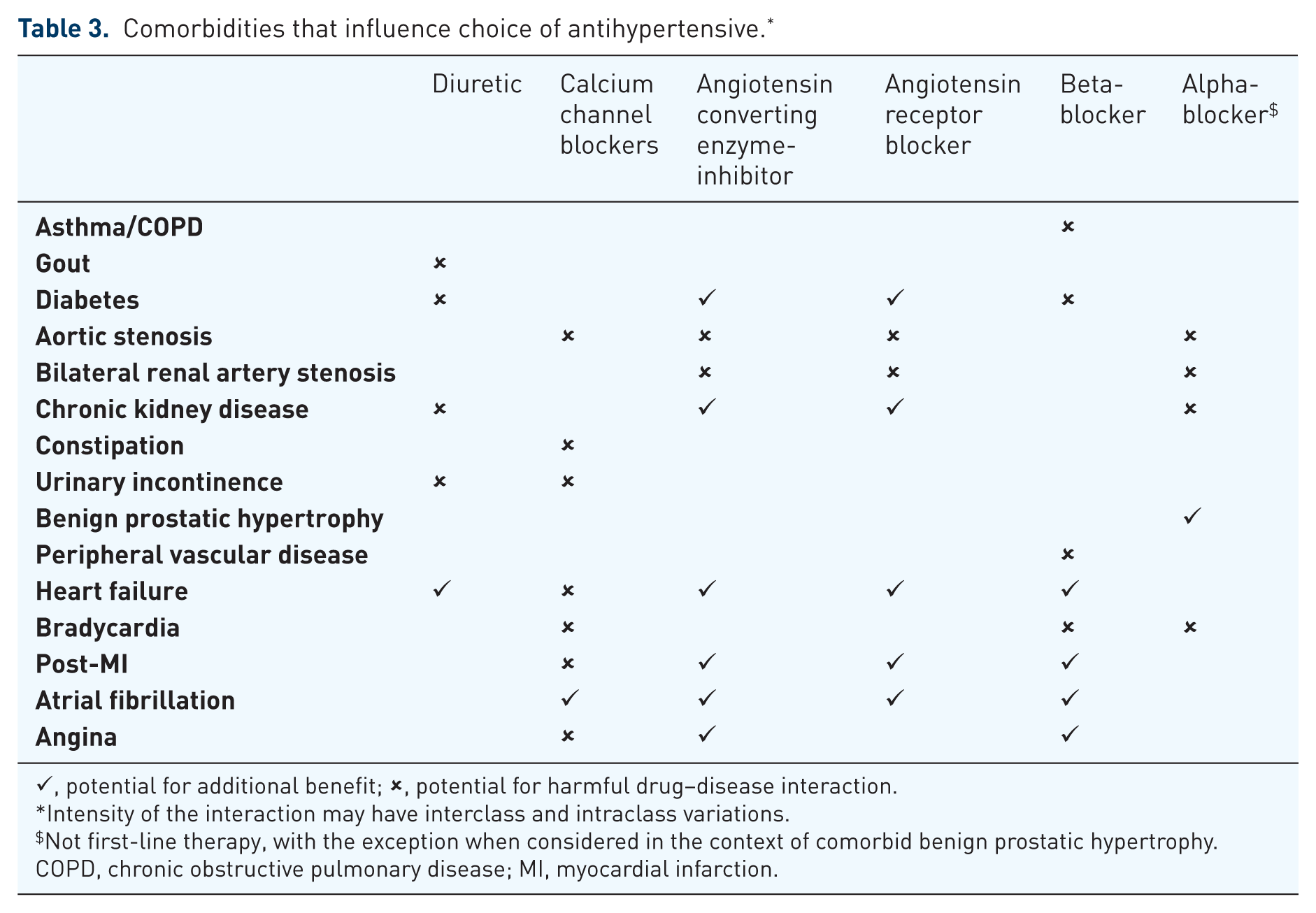
✓, potential for additional benefit; ✗, potential for harmful drug–disease interaction.
Intensity of the interaction may have interclass and intraclass variations.
Not first-line therapy, with the exception when considered in the context of comorbid benign prostatic hypertrophy.
COPD, chronic obstructive pulmonary disease; MI, myocardial infarction.
Practical approach: some key considerations when prescribing antihypertensives to the older person
Before prescribing: is there a strong indication to start pharmacological management?
Antihypertensives are prescribed to manage the risk of hypertension with the view to a long-term benefit. The numbers NNT, shown in Table 2, are following multiple years of treatment, and therefore it must be considered whether your patient is likely to benefit from this medication during their remaining lifetime [Holmes et al. 2013]. The BEGIN algorithm is a novel tool developed by the authors of this paper, combining geriatric and pharmacy expertise, to support prescribers when considering whether pharmacological therapy should begin (Figure 1). Remember that if you would not start a therapy in your patient then you probably should not continue it either. The ‘start low, go slow’ approach to medication prescribing in older adults is an important approach to the use of antihypertensives [Pretorius et al. 2013].
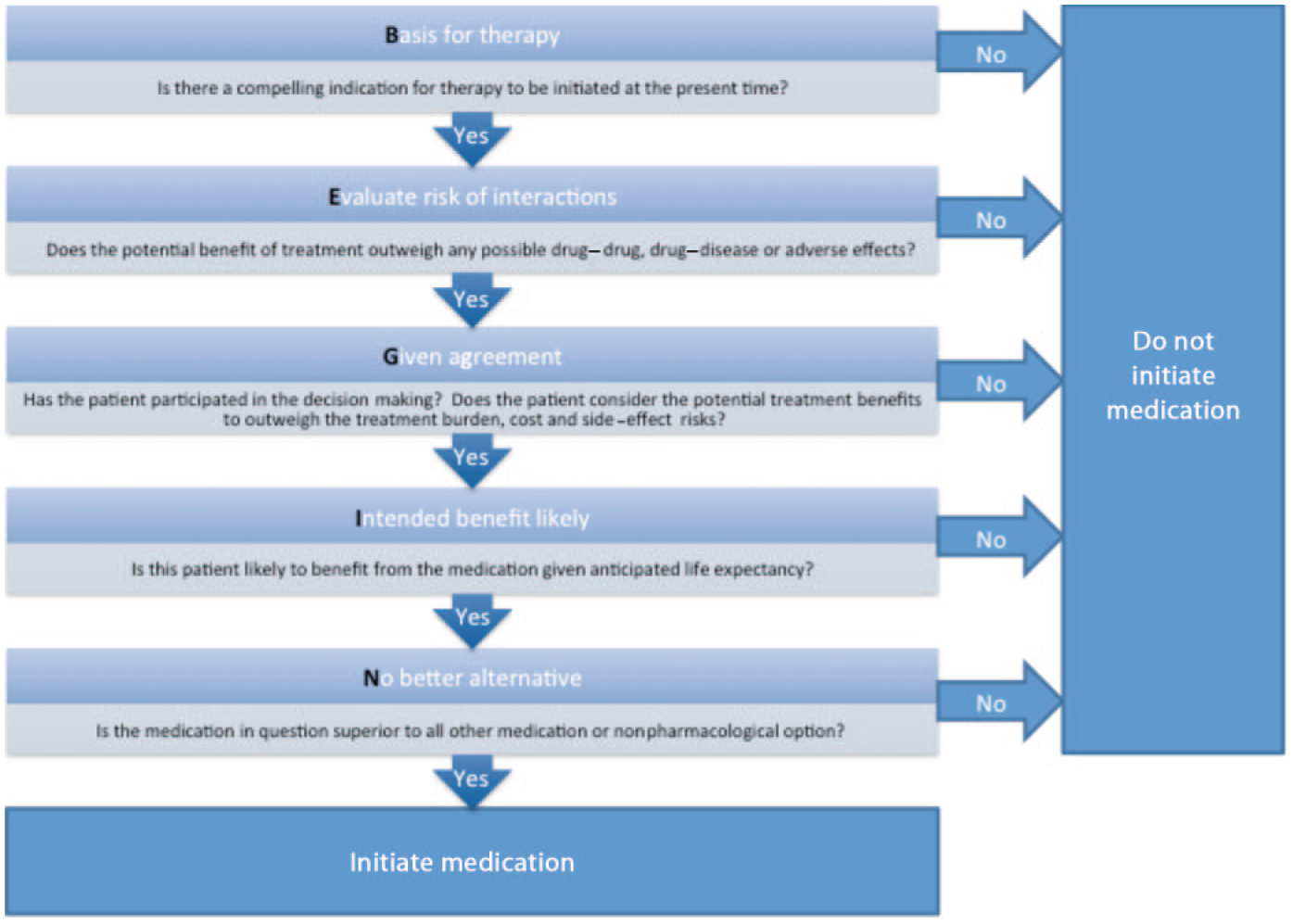
The BEGIN algorithm developed by the authors of this paper to help guide appropriate pharmacological intervention.
Prescribing: does the patient understand the benefits of using this medication within their individual context?
Older patients commonly feel that they have not been provided adequate information about their medications by the prescribing practitioner [Paulino et al. 2004]. Poor health literacy is associated with worse health outcomes, including premature mortality [Berkman et al. 2011]. The health system and health professionals have a duty to support the health literacy of patients to reduce the risk of adverse outcomes. ‘Teach-back’ is a method which is evidence driven and can help to ensure that a patient has sufficiently understood the healthcare information provided to them [Sudore and Schillinger, 2009]. Teach-back involves asking the patient to repeat back in their own words the key information that the practitioner has provided and, if necessary, reteaching the information. This is simple and effective to increase a patient’s understanding of information, but requires patience and potentially more consultation time [Griffey et al. 2015].
Is the patient likely to adhere to the treatment?
The benefit of lowering blood pressure with antihypertensives is highly dependent on patient adherence [Mazzaglia et al. 2009]. After 10 days starting a new medication, one in three patients is nonadherent [Barber et al. 2004]. Nonadherence in older adults is associated with a multitude of complex factors that require exploration in an individual context (Table 4) [Steinman and Hanlon, 2010; Notenboom et al. 2014; Ryan and Hill, 2016]. Where nonadherence to antihypertensives is suspected, it is important that the consultation covers three areas [Hameed et al. 2016]: (1) patient’s risk associated with raised blood pressure through using absolute numbers and visual aids, and contextualizing the risk by explaining the number of similar patients out of 100 or 1000 that would be expected to suffer a CV event over ‘x’ years; (2) patient’s lifestyle, as this may not be conducive to adherence to regular medication dosages and this is an opportunity to revisit nonpharmacological approaches and potentially reduce pill burden with an agreed goal of higher BP target; (3) patient’s experience of medication use, particularly side effects, with consideration given to alternative pharmacological agents, simplifying dosing regimens such as single-pill-combination drugs, and using more favourable preparations. Nurse prescribers and nurse-led clinics can be helpful interventions to improve blood pressure control in hypertensive patients [Clark et al. 2010; Ali et al. 2011]; however, there is a need for robust exploration of the cost effectiveness of implementing such an intervention across primary care.
Common barriers to adherence and potential solutions.

MCA, medication-compliance aid.
Has the patient been involved in the decision-making process?
Shared decision making is ‘a process in which clinicians and patients work together to select tests, treatments, management or support packages, based on clinical evidence and the patient’s informed preferences [Coulter and Collins, 2011]. Where the benefits and harms of treatment at an individual level are unknown in many cases of hypertension, a shared process of decision making is an ethical imperative and thus, an endpoint in itself. Nonetheless, it has been shown that patients attending consultations with shared decision making, and undergoing motivational interviewing approaches are more likely to adhere to treatment, better self-manage their long-term condition and show higher levels of satisfaction [Coulter and Collins, 2011; Elwyn et al. 2014]. Core components of a consultation embracing these patient-centred methods for the management of hypertension include: (1) the patient narrative and professional expertise inputting to a joint information gathering and sharing, (2) goal setting and action planning (3) agreeing a shared care plan where responsibilities are outlined both for the patient and professional [Coulter et al. 2013].
After prescribing: Who will monitor the treatment?
Monitoring of therapy often falls short of recommended good practice and can cause significant harm [Steinman and Hanlon, 2010]. Each additional prescriber increases adverse drug events in older patients by 30% [Pretorius et al. 2013]. Furthermore, patients over the age of 80 are less likely to achieve target BP with multiple doctors’ involvement in their care [Chowdhury et al. 2013]. Therefore, having one prescriber (e.g. GP) to take overall responsibility for all medication prescribing and review prevents silo prescribing for individual conditions and encourages a holistic approach. It is important that upon starting a new antihypertensive there is a planned review date in the near future [Pretorius et al. 2013]. The patient can be advised that if they have problems with the therapy prior to this review then they should be in contact with you as the prescriber. A good communication channel and reassurance of support from a single port of call is empowering for the older person and is likely to encourage the patient to report any adverse events early and maintain compliance.
When should antihypertensive medications be reduced or stopped?
The cessation of a medication with the intent to manage polypharmacy, pill burden and improve health outcomes is known as ‘deprescribing’ [Page et al. 2016b]. The rationale for deprescribing antihypertensives needs to be considered in the individual context. It needs to consider the actual adverse effects experienced and the blood pressure target recommended for that individual [Page et al. 2016b]. Where practical, the antihypertensive medication should to be tapered to avoid rebound hypertension. The DANTE study in the Netherlands found that deprescribing antihypertensives in older adults with mild cognitive deficit resulted in modest increases in SBP 7.4 mmHg (95% CI, 3.0–11.7) and DBP 2.6 mmHg (95% CI, 0.3–4.9) [Moonen et al. 2015].
A recent randomized controlled trial of deprescribing antihypertensives in older people having frequent falls or OH found no increase in mortality amongst those patients that antihypertensives were deprescribed for [Potter et al. 2016]. Indeed, observational data suggests that approximately one in four people who are normotensive while taking antihypertensive medications will remain normotensive after ceasing it [Iyer et al. 2008]. If they return to a hypertensive state, this often happens within the first month and usually within the first year. Lower blood pressure while on treatment and monotherapy are correlated to greater likelihood of remaining normotensive after withdrawing treatment [Iyer et al. 2008].
Conclusion
There is a fine line between providing benefit and harm with the pharmacological management of hypertension in older adults, however it is currently unclear where this line sits. A target blood pressure of <150/90 mmHg appears beneficial to reduce CV morbidity and mortality in otherwise well individuals. The over-65 age group is the most medically heterogeneous population, and a ‘one size fits all’ approach is not in their best interests. Therefore, individualized decisions in the context of commonly encountered conditions (multimorbidity, frailty, falls and cognitive impairment) are crucial to a patient-centred and effective management plan. Functional and cognitive impairment, patient preference and shared decision making are key elements to safe prescribing.
Further research is needed to determine the optimal target to which BP should be lowered, and whether there is benefit from using antihypertensives in frail older adults and those over 90 years of age.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
