Abstract
Type 2 diabetes mellitus is a prevalent, progressive disease with a need for innovative therapeutic agents to continue to advance disease management. Dapagliflozin is the second agent in a new class of oral antihyperglycemic drugs: sodium-glucose cotransporter 2 (SGLT2) inhibitors. SGLT2 is responsible for the majority of renal glucose reuptake; inhibition of the cotransporter allows for increased renal glucose excretion that consequently leads to reduced plasma glucose levels. Because this mechanism does not require the action of insulin, dapagliflozin rarely causes hypoglycemia and is effective in patients both early and late in the course of their disease. Studies of dapagliflozin have demonstrated efficacy both as monotherapy and in combination with oral antihyperglycemic agents and insulin. Dapagliflozin has been shown to decrease hemoglobin A1c (HbA1c) values 6 mmol/mol (0.5%) to 8 mmol/mol (0.7%). The most common adverse reactions observed with dapagliflozin in clinical trials were female genital mycotic infections, urinary tract infections, and nasopharyngitis. Dapagliflozin is a new oral agent for type 2 diabetes with short-term efficacy similar to dipeptidyl peptidase 4 inhibitors; its long-term safety and efficacy are unknown.
Introduction
Type 2 diabetes mellitus (T2DM) is a worldwide problem that is growing in prevalence. An estimated 347 million people worldwide are diagnosed with diabetes, and 90–95% of those have T2DM [Danaei et al. 2011]. Chronic hyperglycemia due to diabetes is associated with both microvascular and macrovascular complications and can ultimately result in death [American Diabetes Association, 2014; Mathers and Loncar, 2006]. Despite these known consequences, many patients are unable to optimally regulate their blood glucose control. Only about half of patients with T2DM achieve their glycemic, blood pressure (BP), or lipid goals, and less than 20% meet all three [Stark Casagrande et al. 2013]. Medication adverse effects, drug–drug interactions, concurrent illnesses, and hospitalizations can all cause hyperglycemia and result in difficulty maintaining glycemic control [Fowler, 2011]. Current guidelines advocate for initiating lifestyle modifications and metformin as first-line therapy, but beyond that, antihyperglycemic management becomes very patient specific [American Diabetes Association, 2014; Inzucchi et al. 2012]. Even when patients are able to achieve a target hemoglobin A1c (HbA1c) goal of less than 53 mmol/mol (<7%), it is difficult to maintain this long term as their disease progresses [UK Prospective Diabetes Study 1995a, 1995b]. Antihyperglycemic agents with innovative mechanisms and synergistic effects when used in combination with other antihyperglycemic agents are necessary to extend the number of treatment options available to patients with T2DM.
Sodium-glucose cotransporter 2 (SGLT2) inhibitors are a new therapeutic class of oral agents for the treatment of T2DM. This therapeutic class currently includes three agents: canagliflozin, dapagliflozin, and empagliflozin. Dapagliflozin was approved in the US on 8 January 2014 and has previously been approved and is used in 38 other countries, including Europe, under the trade name Forxiga (Bristol-Myers Squibb Company, Middlesex, UK) [AstraZeneca, 2012; Bristol-Myers Squibb Company, 2014]. A fixed-dose combination of dapagliflozin and metformin (Xigduo [Bristol-Myers Squibb Company, Middlesex, UK]) was also recently approved in Europe [AstraZeneca, 2014a]. This review will focus on the efficacy and safety of dapagliflozin for the treatment of T2DM.
Pharmacology
Dapagliflozin competitively, reversibly, and highly selectively inhibits SGLT2. Type 2 SGLT2s are expressed in the kidney and on the epithelial lining of the S1 segment of the proximal convoluted tubule. Physiologically, these transporters are responsible for approximately 90% of renal glucose absorption [Wright, 2001; Wright and Turk, 2004]. By blocking SGLT2 with dapagliflozin, reabsorption of glucose into the bloodstream is diminished. Dapagliflozin promotes glucose filtration through the kidneys and into the urine to be eliminated from the body. To quantify the degree of glucose excretion that occurs with dapagliflozin, studies have examined 24 h glucose excretion amounts in healthy subjects as well as patients with T2DM given a range of dapagliflozin doses. Dapagliflozin doses of 20–100 mg have resulted in urinary glucose excretion of approximately 60 g over 24 h in healthy volunteers [Komoroski et al. 2009]. In subjects with T2DM who received dapagliflozin doses between 2.5 and 20 mg, the 24 h glucose excretion after 1 day ranged between 38 and 77 g and after 14 days ranged between 42 and 73 g [Kasichayanula et al. 2011a]. In comparison, patients who have a mutation of the SGLT2 gene SLC5A2 can excrete up to 125 g per day of glucose with no clinically relevant adverse outcomes [van den Heuvel et al. 2002]. Studies have demonstrated that the 24 h urine glucose excretion with dapagliflozin represents only about 40–50% of the human-filtered glucose load. One potential reason for this ceiling effect is that when SGLT2 is inhibited, SGLT1 may compensate by increasing reabsorption of glucose [Defronzo et al. 2013].
Dapagliflozin is dosed starting at 5 mg orally in the morning and can be titrated up to 10 mg orally in the morning if clinically indicated. Dapagliflozin is 78% bioavailable and rapidly absorbed. Its half life is 12.9 h, qualifying for once-daily dosing [Bristol-Myers Squibb Company, 2014]. Dapagliflozin is not known to have any meaningful drug–drug interactions. It is predominantly metabolized by UGT1A9 and has minor cytochrome 450-mediated metabolism. Dapagliflozin has been evaluated in combination with glimepiride, metformin, pioglitazone, and sitagliptin; it neither affects the metabolism of these antihyperglycemic agents nor is its metabolism affected by them and there are no known pharmacokinetic (PK) alterations [Bristol-Myers Squibb Company, 2014; Kasichayanula et al. 2011b]. Because dapagliflozin can cause decreases in systolic BP via its osmotic diuretic effect, patients receiving antihypertensive agents (especially loop diuretics) or those known to experience hypotension should be closely monitored when initiating or titrating dapagliflozin [Bristol-Myers Squibb Company, 2014].
Clinical efficacy
Use as monotherapy
Dapagliflozin has been studied as monotherapy in five clinical trials. In two of the trials, dapagliflozin monotherapy was added onto a background of diet and exercise, in two more, dapagliflozin was compared with metformin monotherapy, and in the fifth study dapagliflozin was used as monotherapy in both treatment-naïve and treatment-experienced patients.
Ferrannini and colleagues conducted a 24-week, parallel-group, double-blind, placebo-controlled trial in which 485 drug-naïve patients with a baseline HbA1c from 53 mmol/mol (7.0%) to 86 mmol/mol (10%) were randomized to receive placebo or dapagliflozin 2.5 mg, 5 mg, or 10 mg each morning (main cohort) or each evening (exploratory cohort). There were an additional 73 patients included in a high-HbA1c cohort who had a baseline HbA1c from 87 mmol/mol (10.1%) to 110 mmol/mol (12%); these patients were randomized to dapagliflozin 5 mg or 10 mg each morning. Patients who received placebo had a mean change in HbA1c of −3 mmol/mol (−0.23%) ± 1 mmol/mol (0.10%) and patients in the main cohort had changes in HbA1c that ranged from −6 mmol/mol (−0.58%) ± 1 mmol/mol (0.11%) to −10 mmol/mol (−0.89%) ± 1 mmol/mol (0.11%), of which the dapagliflozin 5 mg and 10 mg doses produced an HbA1c reduction that was statistically better than placebo (p < 0.001 for 5 mg and p < 0.0001 for 10 mg). Reductions in HbA1c for the evening dose cohort were similar [−9 mmol/mol (−0.79%) ± 1 mmol/mol (0.10%) to −9 mmol/mol (−0.83%) ± 1 mmol/mol (0.11%)]. The high-HbA1c cohort experienced greater reductions in HbA1c [−29 mmol/mol (−2.66%) ± 14 mmol/mol (1.26%) to −32 mmol/mol (−2.88%) ± 15 mmol/mol (1.41%)]. Fasting plasma glucose (FPG) was decreased by −15.2 ± 4.2 mg/dl to −28.8 ± 4.0 mg/dl in the main cohort and by −77.1 ± 53.4 mg/dl to −84.3 ± 61.0 mg/dl in the high-HbA1c group [Ferrannini et al. 2010].
Similarly, Bailey and colleagues evaluated 282 drug-naïve patients with inadequately controlled T2DM [baseline HbA1c ≥ 53 mmol/mol (≥ 7%) and ≤ 86 mmol/mol (≤ 10%)] who received either placebo or dapagliflozin 1 mg, 2.5 mg, or 5 mg/day for 24 weeks. Patients who received dapagliflozin compared with those who received placebo had greater reductions in HbA1c [−8 mmol/mol (−0.68%) for 1 mg, −8 mmol/mol (−0.72%) for 2.5 mg, −9 mmol/mol (−0.82%) for 5 mg, and +0.2 mmol/mol (+0.02%) for placebo, p < 0.001], FPG (−11 mg/dl for 1 mg, −21.6 mg/dl for 2.5 mg, −28.5 mg/dl for 5 mg, and +0.23 mg/dl for placebo, p < 0.05), and weight (−2.69 kg for 1 mg, −2.64 kg for 2.5 mg, −2.69 kg for 5 mg, and −0.96 kg for placebo, p < 0.01) between baseline and week 24. All p values represent dapagliflozin compared with placebo [Bailey et al. 2012].
A randomized, double-blind, parallel-group, placebo-controlled dose-ranging study by List and colleagues evaluated 389 treatment-naïve patients with T2DM assigned to receive placebo, dapagliflozin 2.5 mg, 5 mg, 10 mg, 20 mg, 50 mg, or metformin XR 750 mg, force titrated to 1500 mg at week 2 for 12 weeks. At study end, all dapagliflozin groups and the metformin group achieved significant reductions in HbA1c compared with placebo. Adjusted mean reductions in HbA1c were −6 mmol/mol (−0.55%) to −10 mmol/mol (−0.9%) for dapagliflozin, −8 mmol/mol (−0.73%) for metformin, and −2 mmol/mol (−0.18%) for placebo. Similar reductions were seen in FPG: −16 to −30 mg/dl for dapagliflozin, −18 mg/dl for metformin, and −6 mg/dl for placebo. Patients in all groups lost weight; mean percent weight reductions were −2.5% to 3.4% for dapagliflozin (−1.3 to −2.0 kg), −1.7% for metformin, and −1.2% for placebo [List et al. 2009].
Henry and colleagues conducted two randomized, double-blind, double-dummy, multinational studies in treatment-naïve patients with T2DM randomized to receive dapagliflozin plus placebo, metformin XR plus placebo, or dapagliflozin plus metformin XR for a total of six study groups. In study 1, patients randomized to receive dapagliflozin received a 5 mg dose and in study 2, patients receiving dapagliflozin received a 10 mg dose. Patients receiving metformin received a dose titrated to 2000 mg. At the end of 24 weeks, patients receiving combination therapy of dapagliflozin and metformin in both trials had a greater HbA1c reduction and greater reductions in mean FPG values than patients receiving monotherapy. However, dapagliflozin monotherapy with daily doses of 5 and 10 mg produced changes in HbA1c of −13 mmol/mol (−1.19%) [−15 mmol/mol (−1.36%), −11 mmol/mol (−1.02%)] and −16 mmol/mol (−1.45%) [−17 mmol/mol (−1.59%), −14 mmol/mol (−1.31%)], respectively and changes in FPG of −42.0 mg/dl (−43.4 mg/dl, −36.8 mg/dl) and −46.5 mg/dl (−51.4 mg/dl, −41.4 mg/dl), respectively. Interestingly, dapagliflozin 10 mg monotherapy was as effective as metformin XR monotherapy in reducing HbA1c [−16 mmol/mol (−1.45%) with dapagliflozin 10 mg versus −15 mmol/mol (−1.35%) to −16 mmol/mol (−1.44%) with metformin XR] and produced greater reductions in FPG (−46.5 mg/dl with dapagliflozin 10 mg versus −33.5 mg/dl to −34.8 mg/dl with metformin XR) and weight (−3.33 kg with dapagliflozin 10 mg versus −1.29 to −1.36 kg with metformin XR) than metformin XR [Henry et al. 2012].
The study by Kaku and colleagues evaluated the safety and efficacy of dapagliflozin monotherapy in Japanese patients with T2DM who were either treatment naïve or had limited treatment experience. This was a randomized, double-blind, placebo-controlled clinical trial that randomized patients to placebo or dapagliflozin 1 mg, 2.5 mg, 5 mg, or 10 mg daily. Patients who were receiving treatment for T2DM within 6 weeks of enrollment underwent a 6-week washout period prior to a 4-week placebo run-in period. All other patients were excluded from the washout but also underwent the 4-week placebo run-in period. At the end of 12 weeks, all doses of dapagliflozin produced significant HbA1c reductions compared with placebo; the change in HbA1c compared with placebo ranged from −5 mmol/mol (−0.49%) to −9 mmol/mol (−0.80%), p < 0.0001. Similar changes were seen in FPG reductions; FPG in the placebo group increased by 11.17 ± 3.43 mg/dl compared with reductions in the dapagliflozin groups ranging from −15.61 ± 3.43 mg/dl in the 1 mg group to −31.94 ± 3.57 mg/dl in the 10 mg group (all p < 0.0001 compared with placebo) [Kaku et al. 2013].
Across these five dapagliflozin monotherapy studies in patients with T2DM, dapagliflozin was demonstrated to be efficacious. The patient characteristics and HbA1c outcomes are summarized in Table 1, but in general the monotherapy trials of strictly drug-naïve patients included equal representation of male and female patients who were 44–55 years old, had T2DM duration of 0.25–2 years, and baseline HbA1c values of approximately 64 mmol/mol (8%) to 75 mmol/mol (9%) [with the exception of the high HbA1c cohort in the Ferrannini study that had a baseline HbA1c of approximately 97 mmol/mol (11%)]. Doses of dapagliflozin monotherapy ranged from 1 mg to 50 mg daily, and all groups showed an HbA1c improvement over baseline. The range of HbA1c improvement was −6 mmol/mol (−0.55%) to −16 mmol/mol (−1.45%), with a larger HbA1c improvement of −29 mmol/mol (−2.66%) to −32 mmol/mol (−2.88%) achieved in patients with a higher baseline HbA1c [Ferrannini et al. 2010; Bailey et al. 2012; List et al. 2009; Henry et al. 2012]. Patient demographics and efficacy outcomes were slightly different in the study by Kaku and colleagues; this study included both treatment-naïve and treatment-experienced patients who were predominantly male and had T2DM for 4–5 years prior to study enrollment. The HbA1c improvements with dapagliflozin were less dramatic [−1 mmol/mol (−0.12%) to −5 mmol/mol (−0.44%)] in this study population compared with the other four monotherapy studies [Kaku et al. 2013].
Efficacy of dapagliflozin monotherapy in patients with T2DM.
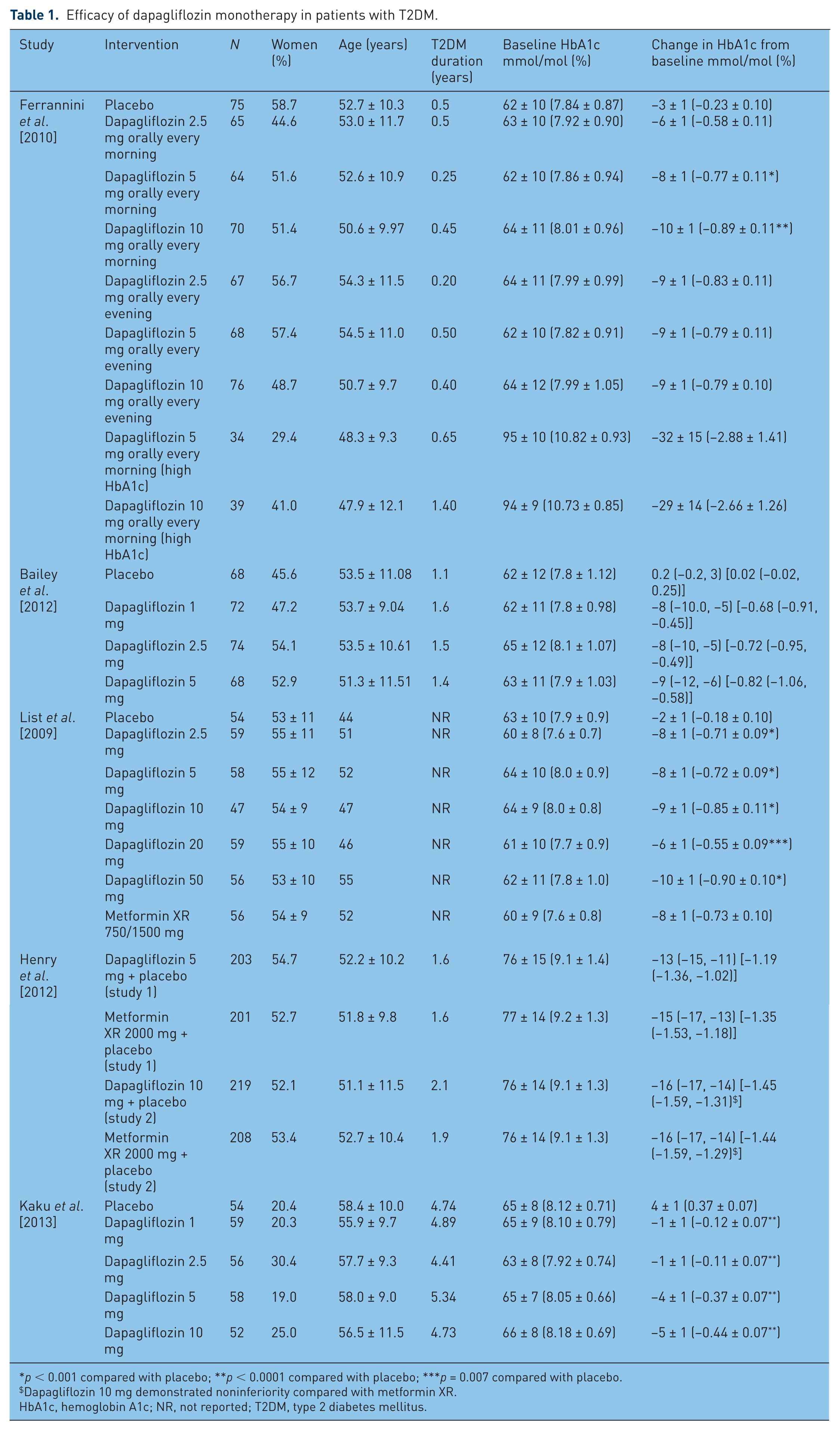
p < 0.001 compared with placebo; **p < 0.0001 compared with placebo; ***p = 0.007 compared with placebo.
Dapagliflozin 10 mg demonstrated noninferiority compared with metformin XR.
HbA1c, hemoglobin A1c; NR, not reported; T2DM, type 2 diabetes mellitus.
Use in patients receiving oral antihyperglycemic agents
In addition to being studied as monotherapy, dapagliflozin has been studied as adjunctive therapy to several oral agents. Bailey and colleagues performed a randomized, double-blind, placebo-controlled trial in 546 patients with T2DM who were receiving metformin (>1500 mg daily) with inadequate glycemic control [baseline HbA1c from 63 mmol/mol (7.92%) to 66 mmol/mol (8.17%)]. These patients were randomized to receive placebo or dapagliflozin 2.5 mg, 5 mg, or 10 mg daily. At 24 weeks, all three dapagliflozin groups demonstrated improvements in HbA1c superior to that of placebo. Therapy with dapagliflozin added to metformin resulted in an additional HbA1c lowering of 4–6 mmol/mol (0.4–0.55%) (p < 0.001 compared with metformin alone). Additionally, patients receiving dapagliflozin in addition to metformin experienced larger reductions in FPG (−17.8 to −23.4 mg/dl) and weight (−2.2 to −3.0 kg) compared with metformin alone (−5.9 mg/dl and −0.9 kg) that were statistically significant (p < 0.002 for FPG and p < 0.0001 for weight) [Bailey et al. 2010]. In an extension of the same study, 71.2% of the original patient population was followed up for 102 weeks. At 102 weeks, patients in the dapagliflozin plus metformin groups continued to have more substantial reductions in HbA1c [−5 mmol/mol (−0.48%) to −9 mmol/mol (−0.78%)], FPG (−19.3 to −24.5 mg/dl), and weight (−2.2 to −3.4 kg) compared with metformin alone [HbA1c +0.2 mmol/mol (+0.02%), FPG −10.4 mg/dl, and weight −0.7 kg; p-values not reported] [Bailey et al. 2011].
Nauck and colleagues conducted a randomized, double-blind, active-controlled study that compared the efficacy and safety of dapagliflozin added to metformin with that of glipizide added to metformin. Patients receiving metformin monotherapy with inadequately controlled glycemia received either dapagliflozin or glipizide, titrated over 18 weeks either up to 10 mg or up to 20 mg, respectively. At 52 weeks, patients in both the dapagliflozin and glipizide groups experienced a decrease in HbA1c −6 mmol/mol (−0.52%) [95% CI −7 mmol/mol (−0.6%) to +5 mmol/mol (+0.44%) for each group]. Based on predefined noninferiority criteria, dapagliflozin was shown to be noninferior to glipizide. Body weight was significantly reduced from baseline to week 52 in the dapagliflozin group; patients receiving dapagliflozin lost 3.22 kg while patients in the glipizide group had gained 1.44 kg at 52 weeks (p < 0.0001) [Nauck et al. 2011]. A 2-year extension of this same study demonstrated more sustained HbA1c reduction with dapagliflozin versus glipizide [−4 mmol/mol (−0.32%) versus −2 mmol/mol (−0.14%)] and continued weight reduction in the dapagliflozin group (−3.7 kg versus +1.36 kg for glipizide). The authors concluded that sustained therapy with dapagliflozin demonstrated continued reductions in HbA1c and weight [Del Prato et al. 2011].
Dapagliflozin has also been studied as an add-on therapy to glimepiride. Strojek and colleagues conducted a 24-week randomized, double-blind, placebo-controlled trial in which patients were randomly assigned to either placebo or dapagliflozin 2.5 mg, 5 mg, or 10 mg added onto open-label glimepiride 4 mg. After 24 weeks, all groups receiving dapagliflozin demonstrated substantial reductions in HbA1c [−6 mmol/mol (−0.58%) to −9 mmol/mol (−0.82%) for dapagliflozin plus glimepiride versus −1 mmol/mol (−0.13) for glimepiride alone, p < 0.0001] and FPG (−16.8 to −28.5 mg/dl for dapagliflozin plus glimepiride versus −2.0 mg/dl for glimepiride alone). The reductions in FPG by the dapagliflozin 5 mg and 10 mg doses added to glimepiride were statistically significant compared with glimepiride alone. Patients in the dapagliflozin 5 mg and 10 mg groups also had sustained reductions in weight [Strojek et al. 2011].
Dapagliflozin was studied by Rosenstock and colleagues as an add on to pioglitazone therapy. Patients with T2DM who were either treatment naïve or were taking metformin, a sulfonylurea, or pioglitazone were maximized on pioglitazone therapy over the course of 10 weeks and then randomized to receive either placebo or dapagliflozin 5 mg or 10 mg in addition to pioglitazone at a dose of at least 30 mg. Patients in the groups that had dapagliflozin added to pioglitazone experienced an additional 5–6 mmol/mol (0.4–0.55%) greater HbA1c reduction compared with pioglitazone alone at 24 weeks [−9 mmol/mol (−0.82%) to −11 mmol/mol (−0.97%) for dapagliflozin plus pioglitazone versus −5 mmol/mol (−0.42%) for pioglitazone] and this effect was sustained at 48 weeks [−10 mmol/mol (−0.95%) to −13 mmol/mol (−1.21%) for dapagliflozin plus pioglitazone versus −6 mmol/mol (−0.54%) for pioglitazone]. Treatment with dapagliflozin added to pioglitazone also resulted in an additional 10–20 mg/dl reduction in FPG (−22.8 to −33.1 mg/dl versus −13.1 mg/dl) and less weight gain (+0.69 to +1.35 kg versus +2.99 kg) than pioglitazone alone at week 48 [Rosenstock et al. 2012].
Finally, dapagliflozin added onto sitagliptin with or without concurrent metformin use has been studied. Jabbour and colleagues randomized patients with T2DM to receive dapagliflozin 10 mg or placebo added onto sitaglipitin 100 mg ± metformin at a dose of at least 1500 mg daily. After 24 weeks, the group treated with dapagliflozin had an additional mean HbA1c reduction of 6 mmol/mol (0.5%) compared with the group that received placebo added to sitagliptin ± metformin. In patients with a baseline HbA1c of at least 64 mmol/mol (≥ 8%), the HbA1c lowering attributed to dapagliflozin was even more remarkable at −9 mmol/mol (−0.8%). The addition of dapagliflozin also resulted in more total body weight loss (−2.1 kg versus −0.3 kg) and greater reductions in FPG (−24.1 mg/dl versus 3.8 mg/dl). These glycemic and weight benefits were maintained up to 48 weeks in patients who underwent the 24-week study extension [Jabbour et al. 2014].
The addition of dapagliflozin to oral therapies of metformin, glimepiride, pioglitazone, and sitagliptin demonstrated significant reductions in HbA1c, FPG, and body weight compared with placebo. The patient demographics and HbA1c outcomes are presented in Table 2. In the two studies that were continued for 2 years, dapagliflozin demonstrated sustained improvements in efficacy endpoints, which is an important finding given the chronic nature of T2DM.
Efficacy of dapagliflozin added onto oral therapy in patients with T2DM.
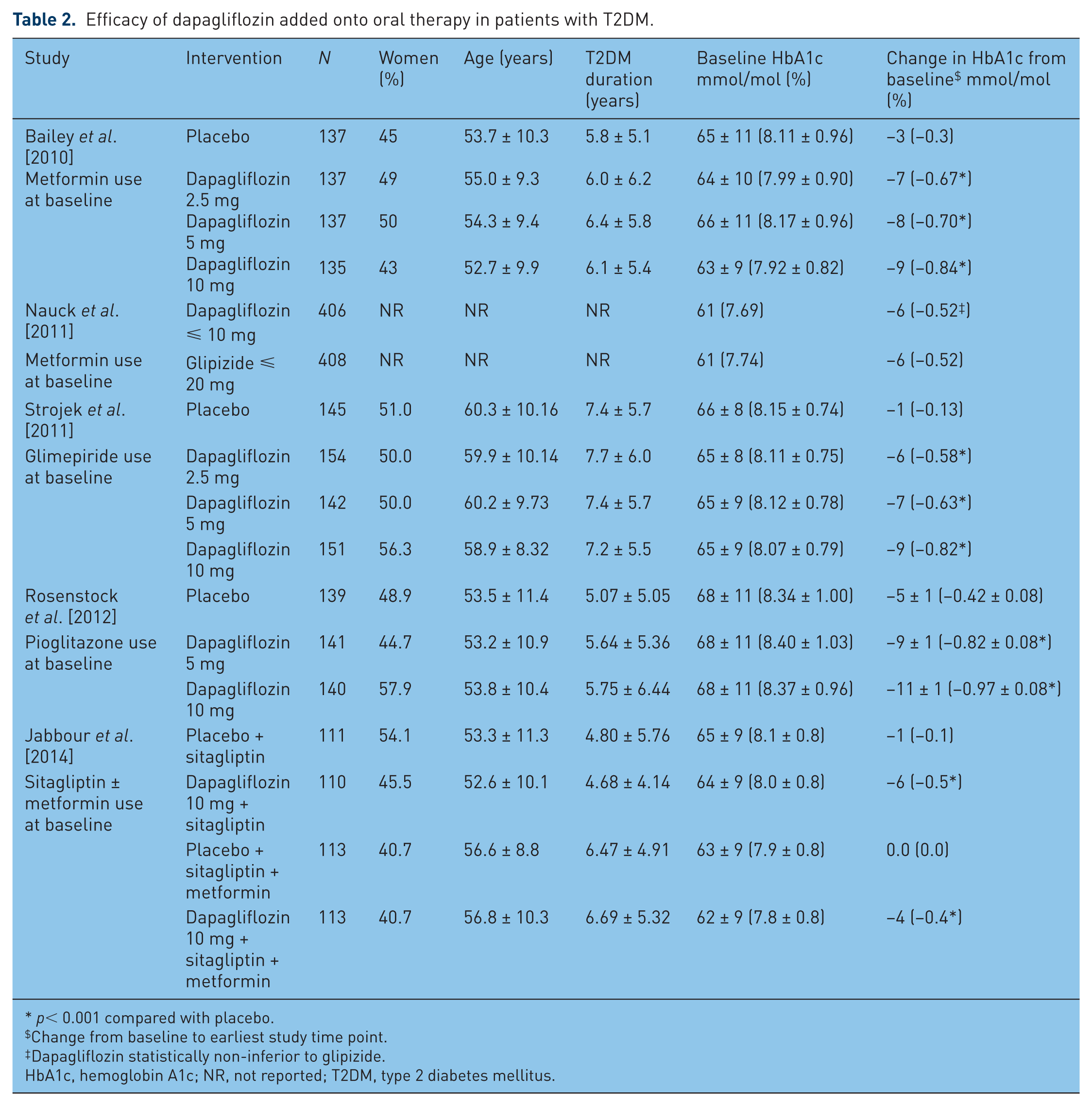
p< 0.001 compared with placebo.
Change from baseline to earliest study time point.
Dapagliflozin statistically non-inferior to glipizide.
HbA1c, hemoglobin A1c; NR, not reported; T2DM, type 2 diabetes mellitus.
Use in patients receiving insulin
Dapagliflozin has also been evaluated in patients receiving concurrent insulin. A study by Zhang and colleagues examined 58 patients with late-stage T2DM [defined as inadequate glycemic control, HbA1c ≥ 59 mmol/mol (≥ 7.5%) and ≤ 86 mmol/mol (≤ 10%), despite aggressive treatment with insulin and insulin-sensitizing agents for at least 6 weeks] who were randomized to placebo, dapagliflozin 10 mg, or dapagliflozin 20 mg for 12 weeks. Insulin sensitizers included stable doses of metformin greater than 1000 mg daily or pioglitazone greater than 30 mg daily or rosiglitazone 4 mg daily. Patients in this group had a preemptive 50% of insulin dose to reduce the risk of hypoglycemia, but doses of other medications remained constant. At baseline, patients in the late-stage group had a duration of T2DM between 11.1 and 19.3 years and an HbA1c from 64 mmol/mol (8.00%) to 68 mmol/mol (8.35%). At 12 weeks, patients in the late-stage dapagliflozin group had an HbA1c reduction of −7 mmol/mol (−0.6%) to −9 mmol/mol (−0.8%) compared with those receiving no dapagliflozin (no change in HbA1c). Patients in the late-stage dapagliflozin group also experienced larger reductions in weight (−4.3 to −5.05 kg) compared with those receiving no dapagliflozin (−1.55 kg) [Zhang et al. 2010].
In an adaptive trial by Wilding and colleagues, patients with T2DM inadequately controlled on insulin combined with oral antihyperglycemic therapies were randomized into two cohorts. In the first cohort, patients were randomized to receive single-blind dapagliflozin 20 mg after their insulin dose had been reduced by 50% in order to evaluate the addition of dapagliflozin to an insulin dose unlikely to cause hypoglycemia. Because the 50% insulin dose reduction was determined to be safe in combination with the addition of dapagliflozin, the study moved forward and additional patients were randomized into the second cohort to receive placebo, dapagliflozin 10 mg or 20 mg in addition to open-label therapy with their oral antihyperglycemic agents and 50% of their usual daily insulin dose. At 12 weeks, patients receiving dapagliflozin had mean changes in HbA1c versus those not receiving dapagliflozin of −8 mmol/mol (−0.7%) to −9 mmol/mol (−0.78%). Dapagliflozin-treated patients also experienced larger reductions in total body weight (−4.3 to −4.5 kg versus −1.9 kg). Equal numbers of patients treated with dapagliflozin and those not treated with dapagliflozin required uptitration of their insulin over the study period [Wilding et al. 2009].
In a subsequent study, Wilding and colleagues studied patients with T2DM who were receiving at least 30 units daily of insulin, with or without up to two oral antihyperglycemic agents and had inadequately controlled glycemia. These patients were randomized to receive placebo, dapagliflozin 2.5 mg, 5 mg, or 10 mg. No pre-emptive adjustments were made to patients’ insulin dosages. After 48 weeks, doses of 5 mg were increased to 10 mg. Insulin doses were maintained unless there was a clinically indicated reason to uptitrate a dose. If there were concerns of hypoglycemia and insulin therapy had already been discontinued, doses of oral antihyperglycemic agents could be adjusted. At 104 weeks, patients in the dapagliflozin groups had an HbA1c change of −7 mmol/mol (−0.6%) to −9 mmol/mol (−0.8%) while the group not receiving dapagliflozin had an HbA1c change of −4 mmol/mol (−0.4%). The mean insulin dose in the group that did not receive dapagliflozin increased by 18.3 units per day and patients gained 1.8 kg, while the mean insulin dose in the dapagliflozin groups remained constant and patients lost between 0.9 and 1.4 kg at 104 weeks [Wilding et al. 2014].
In each study, patients receiving insulin therapy with or without concurrent oral antihyperglycemic agents received additive glycemic benefit when dapagliflozin was added to their regimen. The study authors hypothesize that because dapagliflozin works independently of the effects of insulin and of β-cell function, it creates synergistic glucose-lowering effects in patients inadequately controlled on insulin therapy. Patients in these trials had longer durations of diabetes compared with other trials of dapagliflozin, demonstrating its efficacy across a range of patients with T2DM (Table 3).
Efficacy of dapagliflozin added onto insulin ± oral therapy in patients with T2DM.
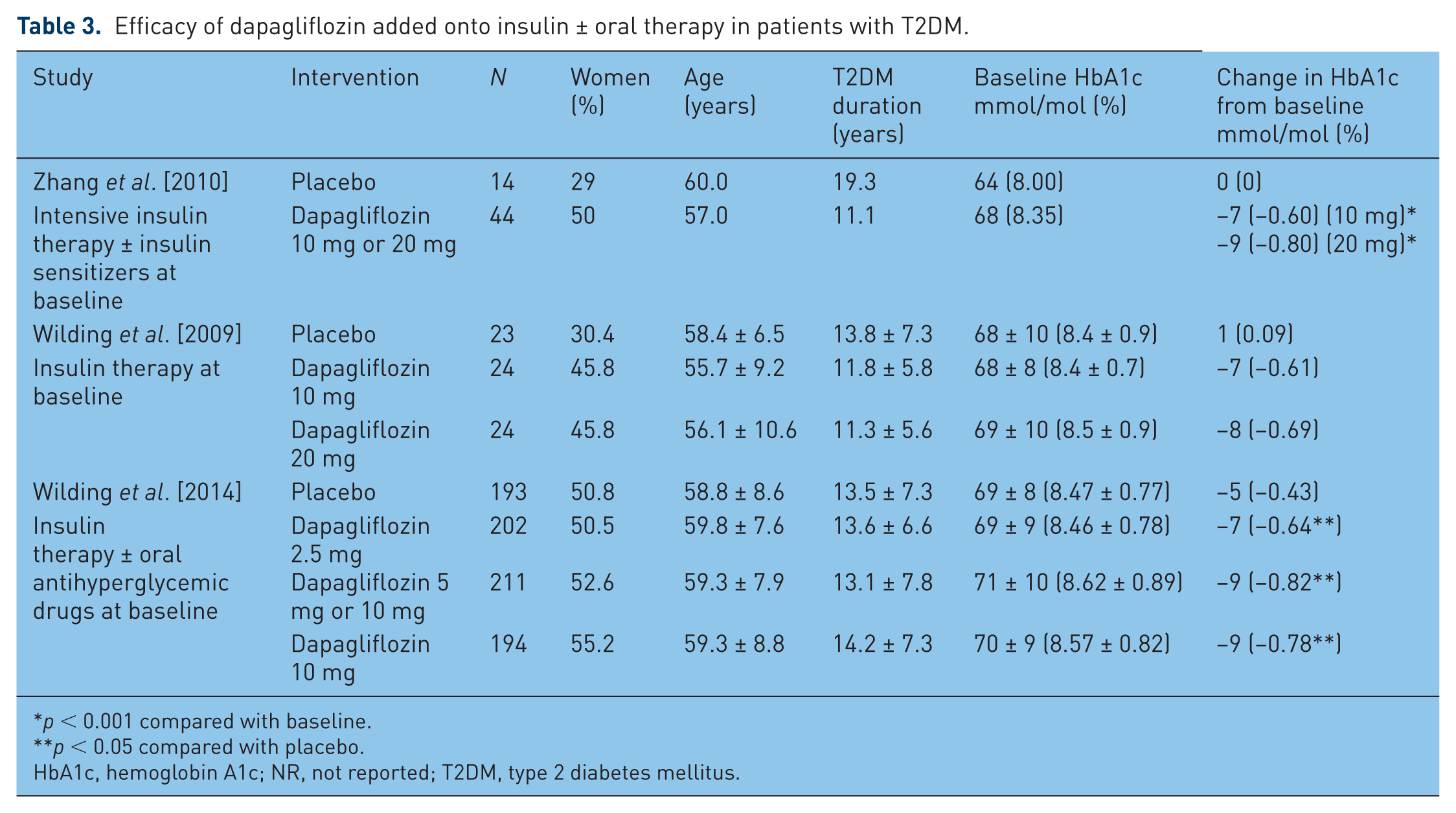
p < 0.001 compared with baseline.
p < 0.05 compared with placebo.
HbA1c, hemoglobin A1c; NR, not reported; T2DM, type 2 diabetes mellitus.
Clinical safety
Adverse effects of dapagliflozin have been evaluated in clinical trials with special attention paid to hypoglycemia, hypotension, renal function, urinary tract infections (UTIs), and genital infections. These adverse effects have been highlighted as being probable consequences due to the mechanism of dapagliflozin and its subsequent PK and pharmacodynamic (PD) properties. Additionally, dapagliflozin has been evaluated for a potential association with bladder cancer.
Hypoglycemia
Across phase III studies, dapagliflozin infrequently resulted in hypoglycemia, and when it did occur, most episodes were minor in nature. In none of the studies did hypoglycemia in the dapagliflozin treatment groups lead to study discontinuation. The prescribing information recommends that caution be exercised when dapagliflozin is used in combination with insulin secretagogues (e.g. sulfonylureas) and insulin as combined use can result in hypoglycemia [Bristol-Myers Squibb Company, 2014]. This recommendation is derived from the studies by Strojek and Wilding in which dapagliflozin was added to glimepiride and insulin therapy, respectively. When dapagliflozin was added to glimepiride therapy, there was a higher incidence of hypoglycemia than in the placebo group (6.9–7.9% versus 4.8%), but none of the events led to study discontinuation [Strojek et al. 2011]. In the 2009 study by Wilding and colleagues in which dapagliflozin was added to patients on 50% of their total daily insulin dose, more patients in the dapagliflozin groups (seven in the 10 mg group and six in the 20 mg group) compared with placebo (three patients) experienced hypoglycemia. However, there were no major episodes of and no deaths due to hypoglycemia [Wilding et al. 2009]. In the 2014 study by Wilding and colleagues in which dapagliflozin was added onto a stable dose of insulin, hypoglycemic events were balanced across treatment groups and there were only three episodes deemed to be serious (two in dapagliflozin-treated patients and one in a placebo-treated patient) [Wilding et al. 2014]. Much like the 2009 Wilding study, the study by Zhang and colleagues preemptively reduced each patient’s daily insulin dose by 50% prior to the addition of dapagliflozin, but the impact of this on hypoglycemia is unknown as the study did not report adverse events [Zhang et al. 2010].
Hypotension
Dapagliflozin causes glucosuria and a subsequent mild osmotic diuretic effect, which can lead to hypotension in some patients. Patients who have limited renal function [estimated glomerular filtration rate (eGFR) < 60 ml/min/1.73 m2], older patients and those taking loop diuretics are advised to use dapagliflozin cautiously in order to avoid symptomatic hypotension [Bristol-Myers Squibb Company, 2014].
However, not all reductions in BP caused by dapagliflozin are negative. Several studies have evaluated the BP-lowering effects of dapagliflozin and have demonstrated significant reductions in seated BP compared with placebo. In the first study, patients with inadequately controlled T2DM and hypertension (HTN) despite treatment with an angiotensin-converting enzyme inhibitor (ACEi) or angiotensin receptor blocker (ARB), or other antihypertensive medication were randomized to receive dapagliflozin 10 mg or placebo. Those in the dapagliflozin-treated group experienced a reduced systolic BP of −4.3 mmHg (95% CI −6.5 to −2.0) compared with placebo (p < 0.0002). In a similar study that evaluated patients with inadequately controlled T2DM and HTN despite treatment with an ACEi or ARB, dapagliflozin 10 mg compared with placebo decreased systolic BP by −3.1 mmHg (95% CI −4.9 to −1.2, p = 0.001). While this BP-lowering effect was modest, it could be beneficial in patients with both uncontrolled T2DM and HTN who are close to, but not yet at, their goal BP [Weber et al. 2013a, 2013b].
In general, patients initiating dapagliflozin should have close follow up of both their blood glucose as well as their BP values. Patients should be routinely assessed for volume status and signs and symptoms of hypotension to avoid any unnecessary hypotension.
Renal function
Clinical trials of dapagliflozin have failed to demonstrate deleterious effects on renal function. However, because the site of action of dapagliflozin is in the kidneys, its effect on renal function and efficacy in the setting of renal impairment are important. Two studies of dapagliflozin PK and PD have demonstrated that the PK of dapagliflozin are directly influenced by renal function. Patients with impaired renal function are exposed to larger amounts of drug compared with those with normal function; however, larger systemic exposure is not linked to a more beneficial pharmacologic effect. In fact, as renal function declines, so does the effect of the drug. As the eGFR approaches 45 ml/min/1.73 m2, dapagliflozin becomes largely ineffective. Because of this, dapagliflozin prescribing information advises not initiating it in patients with an eGFR less than 60 ml/min/1.73 m2 and discontinuing the drug if patients develop a sustained eGFR of the same magnitude as it is ineffective and causes more adverse reactions [Kasichayanula et al. 2013; Kohan et al. 2014; Bristol-Myers Squibb Company, 2014].
Urinary tract and genital infections
UTIs are common in patients with T2DM and there are multiple proposed mechanisms, including hyperglycemia, glucosuria, and neurogenic bladder [Geerlings, 2008]. Because dapagliflozin induces glucosuria as its therapeutic mechanism, the incidence of UTIs associated with the drug has been of particular interest.
A recent review analyzed the safety data of 12 dapagliflozin clinical trials to evaluate the pooled risk of UTI in patients with T2DM taking dapagliflozin. The analysis included 4545 patients who received dapagliflozin 2.5 mg, 5 mg, or 10 mg daily and 1393 patients who received placebo. More patients in the dapagliflozin 5 mg and 10 mg groups (7.3% and 6.5%, respectively) compared with dapagliflozin 2.5 mg and placebo (4.2% and 4.5%, respectively) experienced signs and symptoms suggestive of a UTI. UTIs were more common in women than men, but in both sexes the infections were typically mild and easily treated with standard antimicrobial therapy. Subgroup analyses demonstrated that patients with a history of recurrent UTIs were more likely to experience a UTI while taking either placebo or dapagliflozin. In general, patients were treated for their UTI and did not discontinue study participation or study medication due to the infection [Johnsson et al. 2013a].
A similar review by the same authors analyzed the safety data for patients treated with dapagliflozin who experienced mycotic genital infections (vulvovaginitis or balanitis). The same 12 studies and 4545 patients who received dapagliflozin (N = 3152) or placebo (N = 1393) were included. There was a much stronger association between dapagliflozin treatment and mycotic genital infections: 4.1–5.7% of patients treated with dapagliflozin 2.5 mg, 5 mg, or 10 mg experienced a genital infection versus 0.9% of placebo-treated patients. As with the patients who developed a UTI, the genital infections were typically mild in nature and responded to standard antifungal therapy. Also similar to the UTI data, patients who had a history of recurrent mycotic genital infections were more likely to experience an infection while treated with either dapagliflozin or placebo [Johnsson et al. 2013b].
In both reviews of dapagliflozin-treated patients and the incidence of either UTI or mycotic genital infection, there did not appear to be a dose–response relationship. In both cases the groups receiving dapagliflozin 5 mg had a higher incidence of infection compared with either the 2.5 mg or 10 mg groups. There is, however, a dose–response relationship with glucosuria, suggesting that degree of glucosuria does not correlate with incidence of urinary tract or mycotic genital infection. As previously stated, these infections were mild in nature, easily treated, and rarely led to study discontinuation [Johnsson et al. 2013a, 2013b]. History of a UTI or genital infection does not preclude use of dapagliflozin, but patients should be advised of the incidence of infection and given instructions for seeking medical care if infection is suspected [Bristol-Myers Squibb Company, 2014]
Bladder cancer
In dapagliflozin clinical trials, bladder cancer was reported in 0.17% of patients treated with dapagliflozin versus 0.03% of those receiving placebo. Many of the cases occurred after less than a year on dapagliflozin therapy, which decreases the probability that the cancer was related to drug exposure. When study data are adjusted for patients receiving drug therapy for over a year, there have been four cases of bladder cancer in patients receiving dapagliflozin and none in patients receiving either placebo or a comparator agent. The US Food and Drug Administration has determined that these are too few cases to support a causal relationship; however, dapagliflozin should not be used in patients with active bladder cancer and should be prescribed with caution in those with a history of bladder cancer [Bristol-Myers Squibb Company, 2014].
Laboratory test alterations
Dapagliflozin has been shown to increase low-density lipoprotein (LDL) cholesterol, hematocrit, and serum phosphorus. In a pooled analysis of 13 clinical trials, patients treated with dapagliflozin 10 mg daily had increased LDL (2.9% versus −1.0%), hematocrit (2.3% versus −0.33%) and serum phosphorus (0.13 mg/dl versus −0.04 mg/dl) at 24 weeks versus placebo [Bristol-Myers Squibb Company, 2014]. Nauck and colleagues observed slight increases in blood urea nitrogen in dapagliflozin-treated patients; those receiving dapagliflozin had an increase of 0.5 ± 0.08 mmol/liter compared with an increase of 0.1 ± 0.07 mmol/liter in those not on dapagliflozin [Nauck et al. 2011]. Because elevations in these laboratory parameters can have negative consequences, patients should be monitored for these values when initiating or titrating doses of dapagliflozin.
Other safety concerns
Studies of dapagliflozin to date have only evaluated short-term effects; the long-term safety of dapagliflozin is unknown. Studies to evaluate cardiovascular morbidity and mortality in patients taking dapagliflozin are underway, including the DECLARE-TIMI 58 study [AstraZeneca, 2014b]. Additionally, dapagliflozin is being evaluated long term for malignancies (including bladder cancer) and potential hepatic adverse effects. Because dapagliflozin has not been studied in pregnant women, postmarketing surveillance will track outcomes for pregnant women who take the drug. Dapagliflozin may exert effects on other SGLT cotransporters and the consequences of this remain to be seen. Ongoing clinical trials and postmarketing surveillance data will be key for determining if there are any additional safety concerns to be noted with dapagliflozin.
Place in therapy
Dapagliflozin is orally available, causes little to no hypoglycemia, can be used as monotherapy or as an add on to insulin or other oral agents, and can reduce both weight and BP. However, there are no robust data or years of experience with dapagliflozin to know whether it is truly safe and effective. Based on these characteristics, dapagliflozin may be considered a second-line agent to treat patients with T2DM. Dapagliflozin may serve as a first-line option for patients in whom a traditional first-line agent (e.g. metformin) is contraindicated, but has several limitations that may limit its use first line in the general T2DM population. Due to the frequency of urinary tract and genital infections, providers should carefully consider the use of dapagliflozin in patients who are prone to these types of infections. Dapagliflozin does not have long-term cardiovascular or mortality data, so practitioners should consider these uncertainties when recommending or prescribing dapagliflozin.
Conclusion
Dapagliflozin, one of a group SGLT2 inhibitors, is effective as monotherapy and in combination with other antihyperglycemic agents in reducing HbA1C in T2DM. Its use is associated with small reductions in weight and BP. Dapagliflozin is well tolerated, with genitourinary infections the most significant adverse effect to date. Dapagliflozin and other SLGT2 inhibitors are an effective new oral treatment option for patients with T2DM but the long-term outcomes are unknown.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The author declares no conflict of interest in preparing this article.
