Abstract
Background
Owing to its rapid bactericidal activity and relatively low levels of resistance in most pathogens, gentamicin remains a popular drug in our hospital for suspected serious gram-negative infections, particularly as empirical therapy in combination with a broad-spectrum beta-lactam antibiotic. There are a number of different methods available for gentamicin therapeutic drug monitoring (TDM), including population-based, linear regression and Bayesian methods. Our current hospital guidelines recommend a population-based nomogram with a single gentamicin concentration required to be taken between 6 and 14 hours postdose [Begg and Barclay, 1995]. This method is only applicable to patients <65 years with an estimated glomerular filtration rate (eGFR) >60 ml/min and, therefore, is not ideal for many hospital patients. TDM is performed notoriously poorly in many clinical settings [Leong et al. 2006], but it remains uncertain whether suboptimal monitoring in adults adversely impacts on hard clinical outcomes. Indeed, it has been proposed that lower doses may be effective, making toxicity less likely with less need for monitoring [Begg et al. 2001]. The need for TDM, particularly in patients with uncomplicated infections and normal renal function, has been questioned [Christensen et al. 1997; Logsdon and Phelps, 1997; Winston and Benowitz, 2003]. This is reflected in recent changes to Australian Therapeutic Guidelines [Therapeutic Guidelines Limited, 2010] that no longer advocate TDM for empiric gentamicin therapy of 3 days or less. In the new guidelines, there is a distinction between empiric and directed therapy. For empirical treatment the recommended duration is 48 h (maximum three doses) and monitoring is not advocated. For directed therapy >48 h, aminoglycosides are now reserved for a few specific indications and monitoring is recommended. Two plasma concentrations are required to estimate the 24-hour area under the curve (AUC) using computerised methods. This appears to be a cost-efficient approach to TDM, where resources are focused on providing service to high-risk patient groups [Slaughter and Cappelletty, 1998].
The aims of this study were to determine the outcomes of treatment in patients with gram-negative infections treated with once-daily gentamicin in our institution and the adequacy of gentamicin TDM. The study was carried out before the implementation of the new Australian guidelines.
Methods
Consecutive microbiology results of patients with confirmed gram-negative cultures were obtained retrospectively. Only patients treated with at least one dose of gentamicin were included. Patients were excluded if age was less than 18 years, they received care in the intensive care unit, or were on immunosuppressant medicines or undergoing chemotherapy. Medical records were reviewed for patient demographics, diagnosis, culture source, renal function, comorbidities, gentamicin therapy and monitoring, concomitant antibiotics, antimicrobial sensitivity, clinical and microbiological outcomes. A favourable clinical outcome was defined as clinical improvement (clinical resolution with normalisation of temperature and white cell count) as documented by the patient’s treating physician with survival to discharge and no gentamicin-associated toxicity. A microbiological cure was based on repeat cultures demonstrating clearance of the organism. Nephrotoxicity was defined as a rise in serum creatinine of ≥40 μmol/l [Nicolau et al. 1995]. Gentamicin TDM was assessed to determine whether results were within the target range, if the timing of the sample was correct, if appropriate adjustments were made where indicated and for patients >65 years or with eGFR <65 ml/min, whether two blood levels were obtained [Begg and Barclay, 1995]. Data were entered into Microsoft Access Database with descriptive statistics obtained. The audit was approved by the hospital Drugs and Therapeutics Committee.
Results
Four hundred and sixty eight subjects were identified from culture results. Three hundred and sixty eight either did not receive gentamicin or met one of the exclusion criteria, leaving 100 patients for inclusion in the analysis: 52% were male, median age 64 years (range 17–97 years). Serial assessment of renal function for the duration of gentamicin therapy occurred in 97% of patients. Total body weight was recorded in 56% (median 74.5 kg, range 35–134 kg). Height was recorded in only 13%; 68% had two or more significant comorbidities (median 3, range 0–10). Baseline kidney function was normal in 62%, eGFR was 30–59 ml/min in 23%, 15–29 ml/min in 11% and <15 ml/min in 4%. Empirical therapy accounted for 72% and the remaining 28% was directed treatment. Concomitant antibiotics were given to 97 out of the 100 subjects, with the majority receiving ampicillin or ticarcillin/clavulanic acid (see Table 1). The organism was identified on blood culture in 45%, urine culture in 43% and aspiration of liver abscesses in 12%. Organisms identified included Escherichia coli (47%), Klebsiella species (23%), Pseudomonas species (11%), Proteus species (7%), Enterobacter species (2%), Acinetobacter species (2%) and single isolates of Morganella morganii, Serratia marcescens and Citrobacter freundii. A total of 95% of isolates were sensitive to gentamicin (67% to both gentamicin and the concomitant antibiotic, and 28% to gentamicin alone). In two patients, the organism was resistant to gentamicin but sensitive to the concomitant antibiotic; in two it was resistant to gentamicin and concomitant antibiotic but sensitive to the subsequent antibiotic therapy; and in one was resistant to all antibiotics prescribed.
Concomitant antibiotics used in addition to gentamicin.
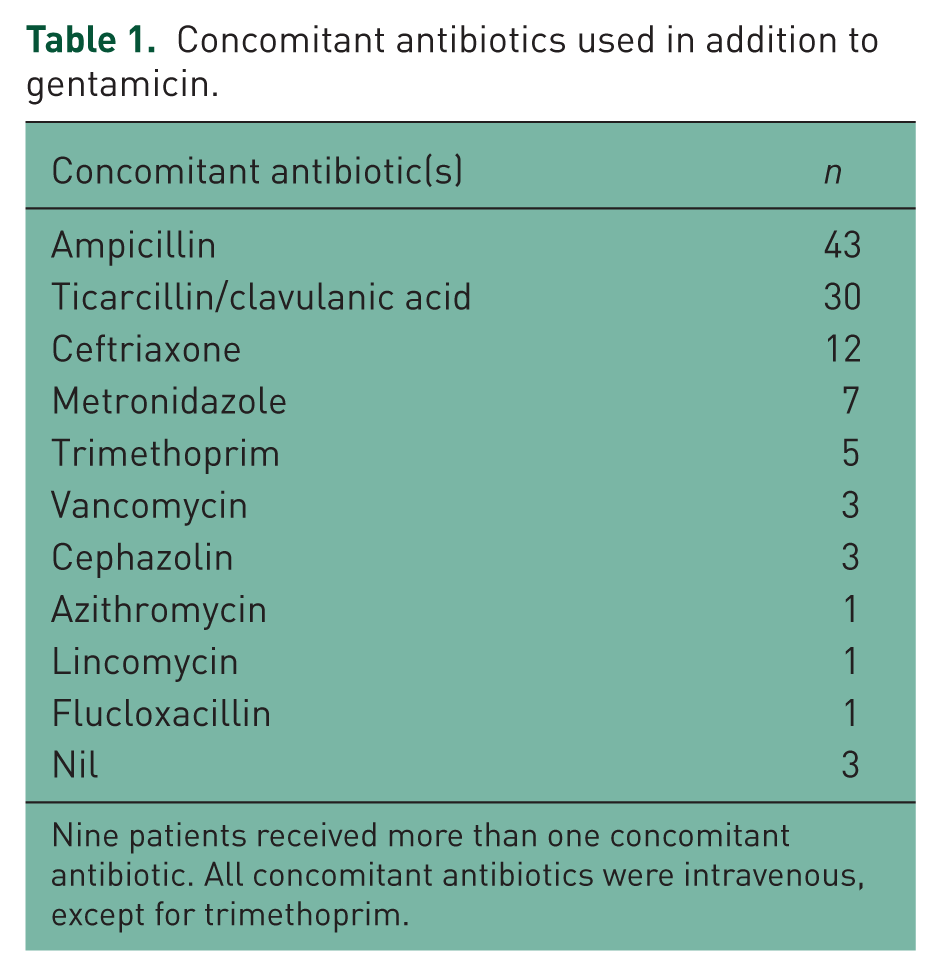
Nine patients received more than one concomitant antibiotic. All concomitant antibiotics were intravenous, except for trimethoprim.
Mean gentamicin dose was 289 ± 75 mg (3.9 ± 0.9 mg/kg, where weight was known) and mean duration 2.9 ± 2.5 days. Most patients received ≤3 days gentamicin (72%), 20% received 4–6 days therapy and 8% received therapy for 7 or more days. Gentamicin treatment for more than 3 days was considered inappropriate or of uncertain benefit in 43% (12/28). TDM was performed in 46% and only in 21% of the total population was it optimal. When performed, gentamicin concentrations were in the target range in 20%; were taken >14 hours postdose in 26% of cases, were potentially toxic in 17% and gentamicin ceased, were potentially toxic in 9%, but gentamicin was continued, potentially toxic in 9% and the dose reduced, low in 15% and the dose increased and were low in 4% and no action taken. Of the 72 patients that had empiric gentamicin therapy, only 26 (36%) had TDM performed and in only 8 (11%) was it performed appropriately.
A favourable clinical outcome was documented in 90% of patients and microbiological cure in 26%. There were nine fatalities. Two were related to treatment failure, one as a result of multiresistant Acinetobacter sepsis, with both occurring in elderly patients with significant comorbidities that developed progressive multi-organ failure. It is unlikely that gentamicin TDM would have prevented either fatality. Other deaths were either not directly related to gram-negative infection or expected, with none resulting from suboptimal TDM; see Table 2 for brief death narratives. All deaths occurred in patients receiving empiric gentamicin therapy.
Brief death narratives.

Only two subjects had a rise in serum creatinine >40 μmol/l, both in the empiric therapy group. However, in both cases, the rise in serum creatinine was preceded by acute renal impairment unrelated to gentamicin therapy. One patient had a fatal outcome and in the other, serum creatinine returned to baseline. Although formal testing was not performed, there was no documented ototoxicity.
Discussion
Gentamicin remains a popular bactericidal antibiotic because of its efficacy in treating gram-negative infections [Hatala et al. 1996] and its low cost. The fact that only one in five patients with positive cultures for gram-negative organisms were treated with gentamicin in the present study is perhaps surprising, and may suggest underutilisation in our hospital.
Despite the modest gentamicin doses used in our cohort of patients with multiple medical comorbidities, where the majority were over 60 years, these results suggest that current treatment of gram-negative sepsis is associated with a favourable clinical outcome and that the recent recommendations for no TDM in empiric gentamicin therapy are supported.
There may be a number of reasons for our findings. Gentamicin remains a highly efficacious, bactericidal drug, even when appropriate doses and monitoring are not routinely used. The minimum inhibitory concentration (MIC) of sensitive organisms in our hospital is <4 mg/l, such that a dose ≥3 mg/kg exceeds the MIC of most gram-negative organisms. Although combination with a broad-spectrum beta-lactam antibiotic would have contributed to the good clinical outcomes, gentamicin appears to have been the critical drug, with 95% of organisms sensitive, whereas 27% were resistant to the concomitant antibiotic. In only four patients was the organism resistant to gentamicin, but sensitive to the concomitant or subsequent antibiotic. With regard to toxicity, short courses and lower doses reduce the risk and few patients were treated with moderate or severe kidney disease. This study serves as a reminder that gentamicin remains a mainstay in suspected gram-negative infections and concerns about toxicity should not preclude judicious use.
Intensive-care patients were excluded in the present study because the volume of distribution of aminoglycosides is increased in critically ill patients with severe sepsis/septic shock, and individualised dosing is warranted [Buijk et al. 2002; Rea et al. 2008]. Similarly, patients on immunosuppressants or receiving cancer chemotherapy were excluded because of the potentially altered microbiological and sensitivity profiles in immunocompromised patients.
Appropriate dosing of gentamicin is important because of concentration-dependent killing, while a narrow therapeutic index has led to recommendations for therapeutic monitoring [Begg et al. 2001]. However, while individualised pharmacokinetic monitoring has been demonstrated to reduce aminoglycoside-associated nephrotoxicity [Streetman et al. 2001; Bartal et al. 2003], there are limited data that TDM improves clinical outcome [Crist et al. 1987; Destache et al. 1990; Van Lent-Evers et al. 1999].
Poor TDM of aminoglycosides is a common problem and methods used in dosage adjustment differ widely [Paterson et al. 1998; Leong et al. 2006; Avent et al. 2011]. The nomogram used in our hospital, applicable to patients <65 years with an eGFR >60 ml/min was not appropriate for many of our patients and in those in whom it was, monitoring was frequently suboptimal. Since aminoglycosides distribute only minimally into adipose tissue, the dosage should be based on the patient’s lean body weight, or total body weight if this is lower. However, ideal or lean weight was not calculated in most patients in the present study and weight must have been estimated in the 44% (or not considered at all) where it was not measured.
In this cohort, more than one quarter of empirically treated patients had TDM that would no longer be recommended under the new guidelines. Although all fatalities and nephrotoxicity occurred in the empiric treatment group, it is unlikely that TDM would have prevented these events.
The weaknesses of this study are that it was retrospective, uncontrolled and numbers were relatively small. Furthermore, there was no comparison with optimal TDM, which precludes an answer as to whether optimal TDM of gentamicin would make a difference to clinical outcomes. Nevertheless, given the very favourable outcome, there would not appear to be much margin for improvement. The study strengths are that hard endpoints were used and only patients with confirmed gram-negative cultures were included, where over half had positive blood cultures or liver abscess indicating severe infections.
Conclusions
The results of the present study suggest that empiric use of short-term gentamicin combined with a broad-spectrum beta-lactam antibacterial drug, is safe and effective in the treatment of most gram-negative infections in immunocompetent adults, even in the absence of appropriate TDM. The findings support the recent changes to the antibiotic guidelines where monitoring is not recommended for empirical short courses of gentamicin [Therapeutic Guidelines Limited, 2010].
Footnotes
Acknowledgements
We thank the following medical residents for painstakingly reviewing medical charts to collect data: Stephanie Sook-Kuan Lim, Stephanie Tan, Barbara van der Werf, Christopher Cooper.
The authors declare no conflicts of interest in preparing this article.
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
