Abstract
Background:
Studies have suggested that glucagon-like peptide-1 receptor agonists (GLP-1 RAs) may modulate asthma-related immune pathways, but evidence directly linking the use of GLP-1 RAs to asthma onset remains limited.
Objectives:
We aimed to evaluate whether the use of GLP-1 RAs in patients with type 2 diabetes mellitus (T2DM) is associated with a reduced risk and severity of asthma development.
Design:
This retrospective cohort study was designed to evaluate asthma onset and severity in patients with T2DM treated with GLP-1 RAs, using data from the National Health Insurance Database (2011–2015).
Methods:
Asthma onset and severity were evaluated in 1345 patients with T2DM treated with GLP-1 RAs from a cohort of 1,936,512 individuals, excluding those with pre-existing asthma. Asthma risk was assessed across four severity levels.
Results:
The study demonstrated a mean follow-up duration of 2.92 ± 1.82 years. Notably, treatment with GLP-1 RAs significantly reduced asthma risk compared with the non-GLP-1 RA group, as indicated by an adjusted hazard ratio (HR) of 0.67 (95% confidence interval (CI), 0.45–0.76), suggesting a consistent class effect. A protective trend was observed across various severity levels of asthma. The HRs for the GLP-1 RA group compared with the non-GLP-1 RA group for cases with no acute exacerbations (No-AE), acute exacerbations (AE), and status asthmaticus (Status) were 0.55 (95% CI, 0.37–0.62), 0.59 (95% CI, 0.39–0.66), and 0.83 (95% CI, 0.56–0.93), respectively. However, in cases requiring endotracheal intubation, the HR was 0.96 (95% CI, 0.65–1.09).
Conclusion:
Our study highlights a consistent effect of GLP-1 RAs in reducing asthma risk and severity, except in cases requiring endotracheal intubation, suggesting that GLP-1 RAs may contribute to reducing asthma incidence and severity in patients with T2DM.
Plain language summary
Our study shows that medicines called GLP-1 receptor agonists, used to treat type 2 diabetes, reduce the chance of getting asthma in people with type 2 diabetes. These medicines also make asthma less severe for most patients, except in cases needing a breathing tube. This suggests GLP-1 receptor agonists may help prevent asthma and reduce its symptoms in people with type 2 diabetes.
Keywords
Introduction
The incidence of diabetes mellitus in Taiwan has progressively risen from 5.8% to 9.3% between 2005 and 2014, based on data from the Taiwan National Health Insurance Database (NHIRD). 1 Consistent with this trend, the World Health Organization Global Report on Diabetes reported that the global prevalence of diabetes among adults (aged ⩾18 years) increased from 4.7% to 8.5% between 1980 and 2014. 2 Asthma, a chronic inflammatory respiratory disease, increased in prevalence from approximately 7% in 2001 to 12.4% by 2017 in Taiwan.3–7 Its global prevalence varies from 0.9% to 21.3%, 8 affecting approximately 358 million individuals worldwide. 9 Furthermore, patients with diabetes have a heightened susceptibility to various pulmonary conditions, including asthma, pneumonia, pulmonary fibrosis, and chronic obstructive pulmonary disease (COPD). 10 Lee et al. 11 reported that patients diagnosed with diabetes had a 1.75-fold higher incidence of asthma compared with those without diabetes, with a 1.38-fold increased risk of asthma associated with elevated glycated hemoglobin (HbA1c) levels and a 1.02-fold increased risk in those with concurrently elevated insulin levels. A prospective cohort study also found a positive association between asthma and an increased risk of type 2 diabetes mellitus (T2DM) in individuals with asthma. 12
Metformin, commonly prescribed as a first-line treatment for diabetes mellitus, has been linked to a reduced frequency of asthma attacks, fewer asthma exacerbations, and reduced rates of asthma-related hospitalizations. 13 Glucagon-like peptide-1 receptor agonists (GLP-1 RAs), a class of second-line treatment agents, have been associated with reduced healthcare encounters for asthma symptoms, fewer asthma exacerbations, 14 and a reduced risk of asthma, 15 but the data are limited.
Therefore, we conducted a study to investigate the association between the use of GLP-1 RAs and the onset and severity of asthma, using data from the Taiwan NHIRD.
Materials and methods
Data collection and participants
Data from the NHIRD, established by the Taiwanese government, were used, encompassing 1,936,512 patients evaluated from 2011 to 2015. The National Health Insurance program has consistently achieved an annual coverage rate of 96% to 99.6% and has covered more than 23 million Taiwanese residents since 1995. To analyze data from the NHIRD, medical conditions were classified according to the International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM). Diseases defined as T2DM and asthma were diagnosed by specialists or specialists in relevant medical fields.
Ethical approval
This retrospective cohort study was approved by the Institutional Review Board of Tri-Service General Hospital (IRB No. E202416042). Informed consent was waived because the study utilized a de-identified database.
Study design and classified patients
All patients were aged ⩾18 years and had a diagnosis of T2DM (ICD-9-CM: 250.XX). Eligible participants had at least three outpatient visits or inpatient admissions and documented use of GLP-1 RAs for a duration exceeding 90 days (Figure 1).
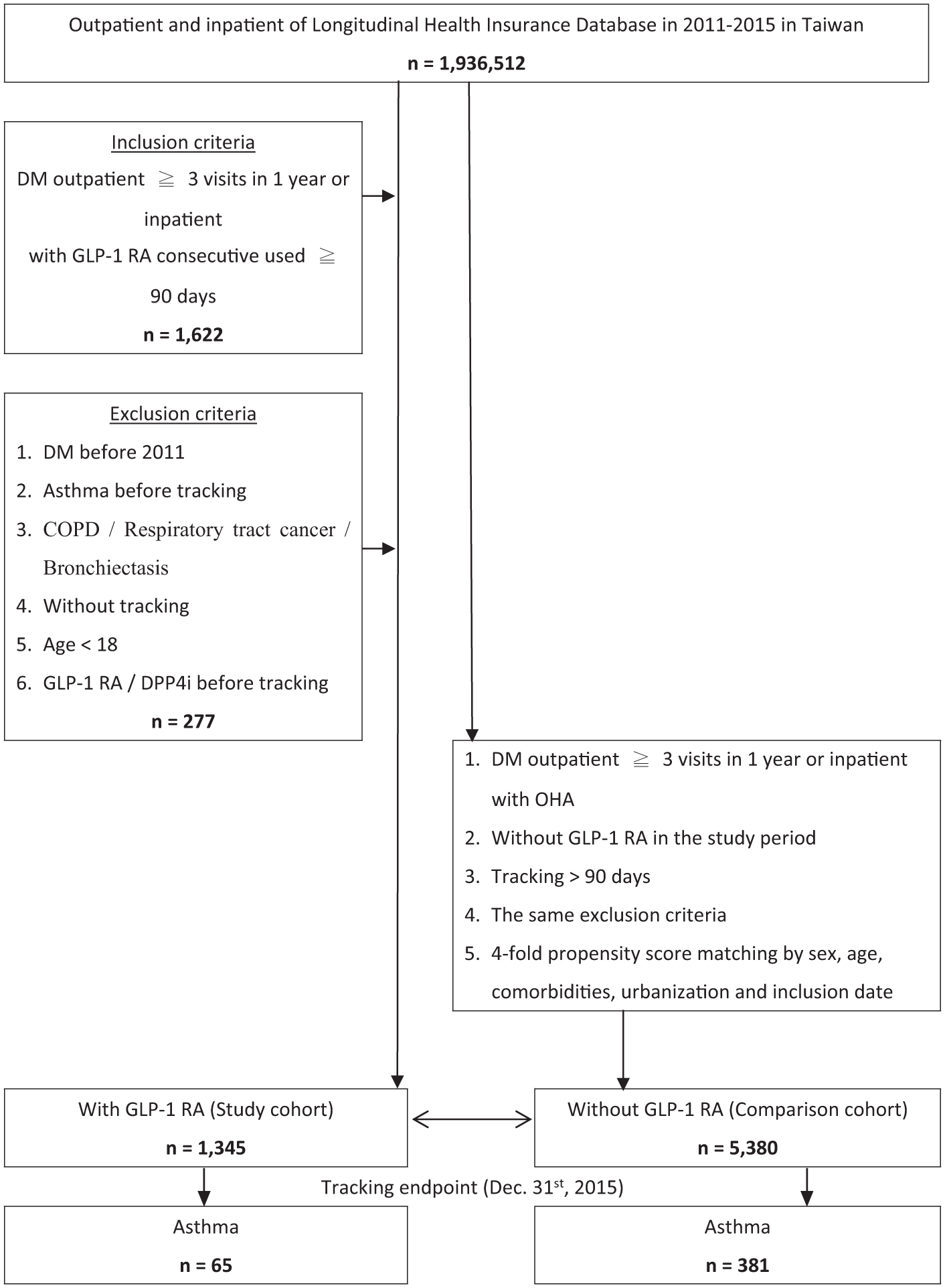
Flowchart of study sample selection.
We conducted a cumulative analysis from January 1, 2011, to December 31, 2015, to examine the incidence and exacerbation patterns of asthma. Therapeutic regimens in the non-GLP-1 RA group encompassed oral antidiabetic drugs, including metformin, sulfonylureas, thiazolidinediones, meglitinides, α-glucosidase inhibitors, sodium-glucose cotransporter 2 inhibitors, and insulin.
The exclusion criteria included: (1) T2DM before the index date; (2) patients with a documented history of asthma; (3) participants with concurrent comorbidities, such as COPD, respiratory tract cancer, or bronchiectasis; (4) participants without continuous follow-up; (5) individuals aged <18 years; (6) individuals who had received GLP-1 RAs or dipeptidyl peptidase-4 (DPP-4) inhibitors before the index date. 16
Propensity score in comparison cohort
A 1:4 propensity score matching approach was implemented to balance the baseline characteristics of the two groups, including age, sex, ethnicity, medical conditions, complications of T2DM, and residential status. Following the propensity score matching procedure, which was conducted using a pool of 1,936,512 patients who met the predefined criteria, the final cohort comprised 1345 patients in the GLP-1 RA group and 5380 patients in the non-GLP-1 RA group.
Outcome measurements
We collected all available data during the defined period. Age, sex, residential location, living environment (urbanization level), 17 season of disease onset, and health conditions (including hypertension, T2DM, hyperlipidemia, obesity, chronic kidney disease, macrovascular or microvascular disease, and obstructive sleep apnea) were all recorded using relevant ICD-9-CM codes in the NHIRD. Comorbid conditions and vascular complications associated with T2DM were determined using ICD-9-CM codes extracted from outpatient claims data before the index date. These encompassed hypertension (ICD-9-CM: 401.1, 401.9, 402.10, 402.90, 404.10, 404.90, 405.1, 405.9), dyslipidemia (ICD-9-CM: 272), obesity (ICD-9-CM: 278), and sleep apnea (ICD-9-CM: 780.51, 780.53, 780.57). In addition, T2DM-related microvascular complications included diabetic retinopathy (DR; ICD-9-CM: 250.52, 362.0), diabetic neuropathy (ICD-9-CM: 250.6, 357.2), and diabetic nephropathy (ICD-9-CM: 250.4, 250.42). Macrovascular complications comprised cerebrovascular events (ICD-9-CM: 430–438), and peripheral vascular disease (ICD-9-CM: 440.2–440.3, 440.8–440.9, 443, 444.22, 444.8, 447.8–447.9). The primary outcomes assessed in this study encompassed the development of asthma and distinct outcome events, including the following: no acute exacerbations (No-AE), acute exacerbations of asthma (AE), status asthmaticus (status), and the necessity for endotracheal intubation. No-AE was characterized by a confirmed asthma diagnosis with ongoing treatment but no exacerbation episodes (ICD-9-CM: 493.00, 493.10, 493.20, 493.90). AE was identified when patients required systemic corticosteroids and an emergency department visit during follow-up (ICD-9-CM: 493.02, 493.12, 493.22, 493.92). Status was defined as severe asthma that did not respond to multiple rounds of systemic corticosteroids or beta-agonists during an emergency department visit (ICD-9-CM: 493.01, 493.11, 493.21, 493.91). Endotracheal intubation referred to patients with AE or Status who required mechanical ventilation via endotracheal intubation (ICD-9-CM: 96.04 for AE or Status).
Statistical analysis
SPSS software, version 22 (IBM Corp., Armonk, NY, USA), was used for statistical analysis. The chi-square test was used for categorical variables, and Student’s t-test was applied for continuous variables to compare demographic characteristics. Hazard ratios (HRs) and 95% confidence intervals (CIs) were calculated using multivariate Cox proportional hazards models to evaluate asthma risk. Kaplan–Meier curves were generated, and the log-rank test was applied to assess differences in asthma risk between the GLP-1 RA and non-GLP-1 RA groups. Statistical significance was determined using a two-sided p value <0.05.
Results
Study population and methods
This retrospective cohort study identified 1,936,512 patients from the Taiwan NHIRD, of whom 1622 were assessed for eligibility after applying inclusion criteria (age ⩾18 years, T2DM diagnosis, no prior asthma). We excluded 277 patients based on exclusion criteria (age <18 years, T2DM diagnosed before 2011, prior asthma or pulmonary disease, loss to follow-up, or unknown sex). After 1:4 propensity score matching (based on age, sex, comorbidities, and diabetes duration), a final cohort of 6725 patients was formed, comprising 1345 in the GLP-1 RA group and 5380 in the non-GLP-1 RA group. Baseline characteristics, including demographics (age, sex, and ethnicity), social factors (residence and urbanization level), and comorbidities (including hypertension, dyslipidemia, obesity, cardiovascular disease, chronic kidney disease, and Charlson Comorbidity Index), were balanced between groups post-matching (all p-values >0.05). The mean age was 65.26 ± 18.62 years (51.9% male), with mean follow-up durations of 2.97 ± 1.86 years (GLP-1 RA) and 2.91 ± 1.81 years (non-GLP-1 RA; p = 0.28). These findings are summarized in Table 1.
Baseline characteristics of the study.
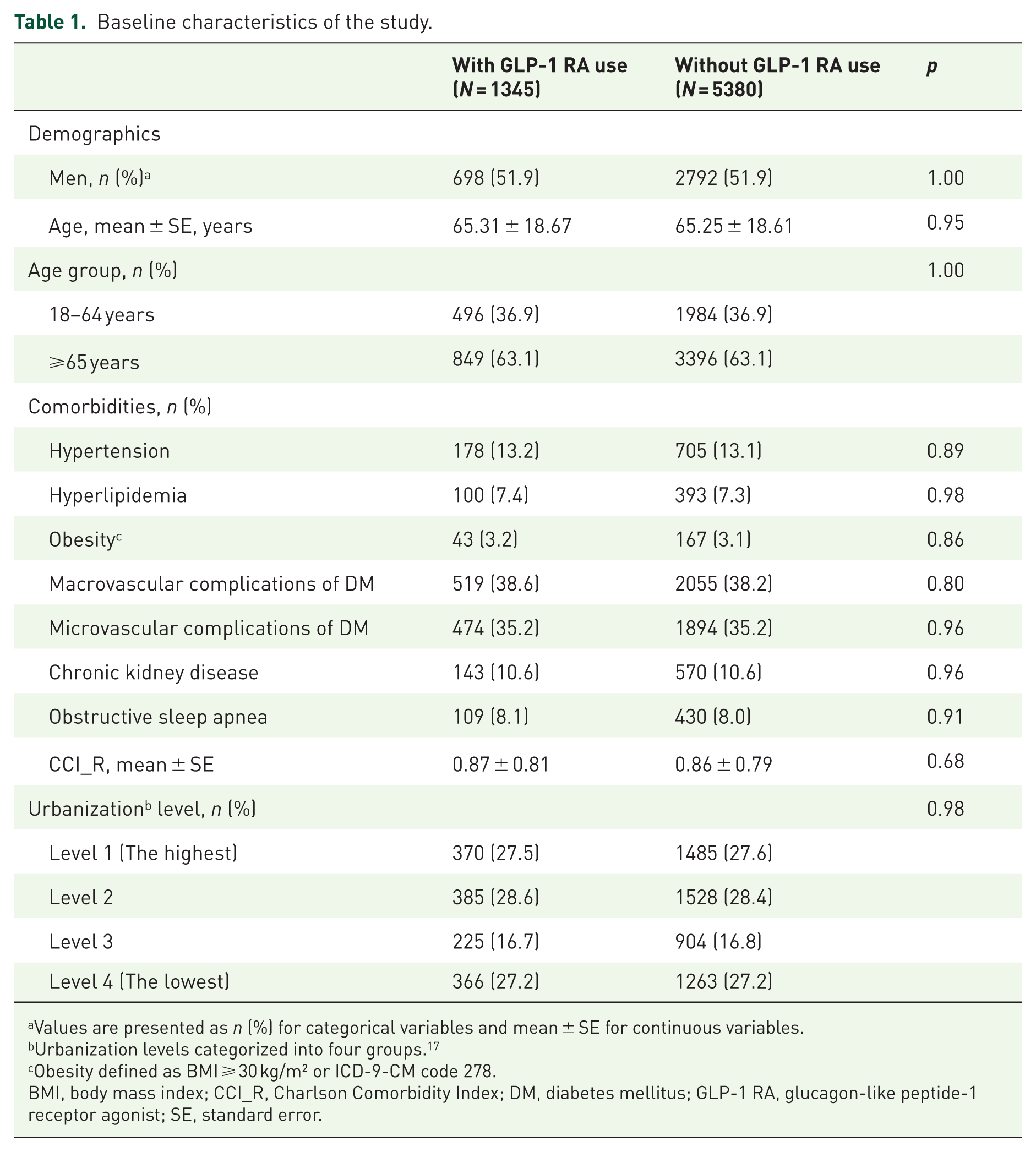
Values are presented as n (%) for categorical variables and mean ± SE for continuous variables.
Urbanization levels categorized into four groups. 17
Obesity defined as BMI ⩾ 30 kg/m² or ICD-9-CM code 278.
BMI, body mass index; CCI_R, Charlson Comorbidity Index; DM, diabetes mellitus; GLP-1 RA, glucagon-like peptide-1 receptor agonist; SE, standard error.
Asthma incidence and severity outcomes
At the study endpoint, asthma developed in 65 patients (4.8%) in the GLP-1 RA group compared with 381 (7.1%) in the non-GLP-1 RA group (Table 2). The mean time to asthma exacerbation was significantly longer in the GLP-1 RA group (2.75 ± 1.70 years) than in the non-GLP-1 RA group (2.47 ± 1.61 years; p < 0.05). Table 3 presents the results of a Cox regression analysis assessing the association between risk factors and asthma incidence. In the GLP-1 RA group, asthma severity outcomes included 19 No-AE cases, 18 AE cases, 16 Status cases, and 12 endotracheal intubation cases, compared with 135, 121, 76, and 49 cases, respectively, in the non-GLP-1 RA group. The crude HR for asthma incidence in the GLP-1 RA group was 0.66 (95% CI, 0.45–0.77; p < 0.001), with an adjusted HR of 0.67 (95% CI, 0.45–0.76; p < 0.001) after controlling for confounders (age, sex, obesity, and comorbidities). Adjusted HRs for asthma severity in the GLP-1 RA group were 0.55 (95% CI, 0.45–0.76; p < 0.001) for No-AE, 0.59 (95% CI, 0.39–0.66; p < 0.001) for AE, 0.83 (95% CI, 0.56–0.93; p = 0.02) for Status, and 0.96 (95% CI, 0.65–1.09; p = 0.12) for endotracheal intubation (Table 4).
Endpoint characteristics of the study.

Values are presented as n (%) for categorical variables and mean ± SE for continuous variables.
Urbanization levels categorized into four groups. 17
Obesity defined as BMI ⩾ 30 kg/m² or ICD-9-CM code 278.
CCI_R, Charlson Comorbidity Index; DM, diabetes mellitus; GLP-1 RA, glucagon-like peptide-1 receptor agonist; SE, standard error.
Cox proportional hazards regression analysis of risk factors associated with asthma incidence.

HRs were estimated using Cox proportional hazards regression models.
Multivariable analysis was adjusted for the following covariates: age, hypertension, hyperlipidemia, obesity, macrovascular complications of DM, microvascular complications of DM, chronic kidney disease, obstructive sleep apnea, Charlson Comorbidity Index score, urbanization level, and season.
CCI_R, Charlson Comorbidity Index; DM, diabetes mellitus; GLP-1 RA, glucagon-like peptide-1 receptor agonist; HR, hazard ratio.
Adjusted HRs for asthma severity outcomes in patients with and without GLP-1 RA use.

The reference group comprised patients without GLP-1 RA use.
PYs were calculated from study entry to asthma outcome, censoring, or the end of follow-up.
Asthma severity categories: No-AE, AE; Status, defined as severe asthma not responding to standard treatments with bronchodilators and corticosteroids; ET-tube intubation, requiring endotracheal intubation.
Adjusted HR was adjusted for the variables listed in the baseline characteristics.
AE, acute exacerbations; CI, confidence interval; HR, hazard ratio; GLP-1 RA, glucagon-like peptide-1 receptor agonist; No-AE, no acute exacerbation events; PYs, person-years; Status, status asthmaticus.
Subgroup and sensitivity analyses
Subgroup analyses by GLP-1 RA type revealed that all types used for more than 1 year (except exenatide) were associated with a reduced risk of asthma (p < 0.001; Table 5). Interaction analyses indicated no significant effect modification by age or sex (p > 0.05). Kaplan–Meier analysis confirmed a significantly lower cumulative asthma risk in the GLP-1 RA group after 2 years (log-rank p < 0.001), as presented in Figure 2.
Adjusted HRs for asthma risk associated with GLP-1 RA use and duration.

The reference group comprised patients without GLP-1 RA use.
Adjusted HRs were estimated using Cox proportional hazards regression models, adjusted for age, hypertension, hyperlipidemia, obesity, macrovascular and microvascular complications of DM, chronic kidney disease, obstructive sleep apnea, Charlson Comorbidity Index score, urbanization level, and season.
Duration of GLP-1 RA exposure was categorized as short-term (90–364 days) or long-term (⩾365 days).
Subgroup analyses were conducted by individual GLP-1 RA type (exenatide, lixisenatide, liraglutide, dulaglutide, and semaglutide).
CI, confidence interval; DM, diabetes mellitus; GLP-1 RA, glucagon-like peptide-1 receptor agonist; HR, hazard ratio.

Kaplan–Meier analysis of cumulative asthma risk among patients with DM aged 18 years and older, stratified by GLP-1 RA use, with log-rank test.
Discussion
Our study demonstrated that individuals in the GLP-1 RA group exhibited a 33% reduction in the risk of developing asthma compared with those in the non-GLP-1 RA group. Furthermore, the GLP-1 RA group had a significantly lower risk of developing all asthma subtypes (No-AE, AE, and Status) compared with the non-GLP-1 RA group. However, no significant difference was observed in the rate of endotracheal intubation. Several factors may have influenced these results, including conservative physician preferences and the use of bilevel positive airway pressure ventilation for patients with respiratory failure instead of endotracheal intubation. This study further demonstrated that each class of GLP-1 RAs significantly reduces the incidence of asthma in patients with T2DM when the duration of therapy exceeds 1 year. These findings suggest that prolonged use of GLP-1 RAs is associated with a cumulative protective effect against asthma development. This effect may be attributed to multiple factors that potentially modulate pathways underlying asthma pathogenesis.
GLP-1 RAs, including liraglutide and exenatide, represent a class of therapeutic agents primarily used for the management of T2DM. GLP-1 RAs exert their effects by binding to GLP-1 receptors on pancreatic β-cells, activating the cyclic adenosine monophosphate pathway, and thereby enhancing insulin secretion.18,19 Oztay et al. 20 reported that GLP-1 RAs exhibit anti-inflammatory properties through multiple mechanisms. First, GLP-1 RAs suppress the expression of inflammatory genes, including nuclear factor kappa B subunit (NFKB) 1, NFKB2, and tumor necrosis factor (TNF). Second, serine proteinase inhibitor-9, the expression of which is induced by GLP-1 RAs, promotes cellular survival against attack by natural killer cells and cytotoxic T lymphocytes. Third, by downregulating inflammatory mediators (TNF-α, interleukin-6 (IL-6), and monocyte chemoattractant protein-1) and suppressing autoimmune responses, GLP-1 RAs enhance insulin sensitivity.
Moreover, empagliflozin and dulaglutide, originally developed for diabetes management, demonstrated significant reductions in lung fibrosis and obesity-induced airway hyperresponsiveness in a murine model. 21 Both agents significantly reduced weight gain, improved glucose homeostasis, and decreased inflammatory markers, suggesting their potential as effective treatments for asthma complicated by obesity. Khan et al. 22 reported that patients with overweight or obesity and concomitant asthma and T2DM experienced clinically significant improvements in asthma symptoms and fewer exacerbations when treated with liraglutide. Notably, this effect was observed independently of significant weight loss (defined as 2.9 kg over 52 weeks). In addition, serum periostin levels, a biomarker of airway eosinophilia in patients with asthma, were significantly reduced in individuals with asthma and diabetes treated with GLP-1 RAs. 23 Although clinical studies have not yet fully validated these findings, our results align with prior research and suggest that GLP-1 RAs may reduce the future incidence of asthma and mitigate severe exacerbations in patients with T2DM.
Previous studies have addressed the link between comorbidities and asthma and their influence on asthma outcomes. 24 Furthermore, our results also demonstrate improved outcomes for microvascular and macrovascular complications, as well as chronic kidney disease (CKD), in patients with T2DM treated with GLP-1 RAs, along with a lower Charlson Comorbidity Index score (Table 3). A prior study demonstrated that semaglutide significantly reduced the risk of major renal outcomes and cardiovascular mortality in patients with T2DM and CKD. 25 In the AWARD-7 trial, dulaglutide, a long-acting GLP-1 RA, achieved comparable blood glucose control to insulin glargine in patients with T2DM and moderate-to-severe CKD, while significantly attenuating the decline in estimated glomerular filtration rate. 26
Besides, cardiovascular outcome trials have demonstrated that GLP-1 RAs significantly reduce the incidence of major adverse cardiovascular events in patients with T2DM, 27 indicating improved cardiovascular safety. Populations with macrovascular or microvascular comorbidities warrant particular attention. Multivariable analysis revealed that, even after adjusting for other confounding factors, the risk of asthma remains significantly increased by over 50%, irrespective of the use of GLP-1RAs. Therefore, in patients with cardiovascular disease, the potential impact of underlying cardiovascular conditions should not be overlooked when using these medications, as this may contribute to suboptimal treatment outcomes.
From a neuropathological perspective, semaglutide and dulaglutide have been shown to reduce the incidence of non-fatal stroke in patients with T2DM. Exenatide, dulaglutide, and liraglutide have demonstrated improved overall cognitive function in this population. Furthermore, semaglutide and dulaglutide have also demonstrated structural and electrophysiological benefits in patients with diabetic peripheral neuropathy. 28 Collectively, GLP-1 RAs appear to effectively mitigate multiple neurological complications associated with T2DM. 29
In the SUSTAIN-6 trial, semaglutide was associated with an increased incidence of new-onset or worsening DR compared with the placebo group (3% vs 1.8%). 30 Notably, the trial showed that semaglutide resulted in a more rapid reduction in HbA1c compared with placebo, which may have contributed to the early exacerbation of DR observed within the first 16 weeks. These findings are consistent with previous studies, such as the DCCT 31 and UKPDS, 32 which demonstrated that rapid HbA1c reduction was associated with transient exacerbation of DR. 33 In contrast, the LEADER trial reported no statistically significant difference between liraglutide and placebo in the incidence of DR. 34 Moreover, a large nationwide cohort study confirmed that GLP-1 RAs are associated with a reduced risk of DR. 35
Furthermore, the present study revealed that the use of semaglutide was not linked to an increased risk of DR progression, vision impairment, or an increased frequency of intravitreal injections over a 3-year follow-up period. 36
Regarding other influencing factors, such as the degree of urbanization, univariate analysis revealed statistically significant differences in highly urbanized areas. However, these differences were not observed in multivariable analysis, suggesting that while patients in highly urbanized areas may have a higher prevalence of asthma, urbanization alone does not contribute significantly to asthma risk after adjusting for other confounding factors. This finding suggests that the degree of urbanization is merely a superficial manifestation, with its underlying implication being that patients in highly urbanized areas may concurrently have multiple comorbidities. However, after adjusting for other confounding factors (comorbidities), its significance diminishes and does not reach statistical significance.
Traditionally, seasonal variations have been considered a significant factor influencing asthma exacerbations. However, current data demonstrate no statistically significant differences in either univariate or multivariable analyses, indicating that asthma occurrence in these patients is primarily driven by their underlying comorbidities rather than seasonal factors, which appear to have a minimal impact.
In contrast, Wang et al. 37 reported no statistically significant reduction in risk associated with the use of GLP-1 RAs or DPP-4 inhibitors in relation to asthma control in their meta-analysis. The limited number of studies and low incidence of asthma likely influenced these observed asthma-related outcomes. However, the authors also noted that, in previous studies,38,39 GLP-1 RAs have been shown to significantly inhibit allergic- and viral-induced airway inflammation, reduce airway eosinophilia, decrease mucus production, and mitigate airway hyperresponsiveness, which may confer a protective effect against asthma in animal models. Notably, our study examined individuals with T2DM who did not have preexisting asthma, assessing the incidence of asthma-related events following their inclusion in the study.
We highlight the study’s large sample size and the rigorous confirmation of T2DM and asthma diagnoses by trained physicians. Furthermore, the study design was strengthened by including participants diagnosed with T2DM but without asthma at the time of recruitment, enabling a longitudinal analysis of asthma incidence. This approach effectively minimized potential confounding effects from pre-existing, poorly controlled asthma on the study outcomes.
Limitations
We acknowledge several limitations of this study. First, patients with asthma or T2DM were identified based on insurance claims data, which lack details on the severity and chronicity of T2DM, as well as body mass index. Furthermore, in our database, the endotypes and phenotypes of asthma, as well as detailed concurrent health conditions and clinical symptoms, such as the presence of COPD and pulmonary function test results, were not available. In addition, none of the patients in this study received biological therapies, including anti-IL-4R agents, anti-immunoglobulin E agents, and anti-IL-5 agents. Moreover, treatment durations for each GLP-1 RA could only be approximated by calculating cumulative doses and dividing them by daily doses. Finally, the population-based design of this study is limited in its ability to elucidate the underlying mechanisms linking the use of GLP-1 RAs with asthma in patients with T2DM, highlighting the need for further studies in the future.
Conclusion
In conclusion, our study underscores a significant association between the use of GLP-1 RAs and a 33% reduction in the risk of asthma development in a Taiwanese cohort diagnosed with T2DM, compared with individuals not using GLP-1 RAs. Furthermore, the use of GLP-1 RAs is associated with a consistent pattern of protective effects against the onset of asthma across different levels of severity, suggesting a class-wide effect. However, this protective effect does not extend to events requiring endotracheal intubation. Our findings suggest that GLP-1 RAs may serve as protective agents against both the incidence and exacerbation of asthma in patients with concomitant T2DM.
