Abstract
Medullary thyroid carcinoma (MTC) is a rare neuroendocrine tumor accounting for less than 5% of all thyroid cancers. An estimated 25% of cases are familial secondary to a germline mutation on the rearranged during transfection proto-oncogene (
Plain language summary
Medullary Thyroid Carcinoma (MTC) is a rare type of thyroid cancer, making up less than 5% of all cases. Some people inherit this condition due to a mutation in a specific gene called RET, while others develop it sporadically, with this gene being mutated in about 40-60% of these cases. The behavior of the disease can vary, and certain genetic changes, like the RET M918T variant, are linked to more aggressive forms of MTC. When MTC becomes advanced or spreads, treatment usually involves special drugs called multikinase inhibitors (MKI), like cabozantinib and vandetanib, which target the cancer’s growth pathways. There’s also a treatment specifically designed to block the RET gene’s activity. Deciding when to start these treatments isn’t straightforward. It requires a careful evaluation of each patient’s overall health, how much the cancer has spread, how quickly it’s progressing, other medical conditions, potential drug interactions, and the patient’s personal treatment goals. This decision-making process is best handled by a team of specialists who can tailor the approach to each individual’s needs. This summary provides an overview of the current treatments for MTC, focusing on their safety, effectiveness, and limitations, with the goal of helping doctors make informed decisions for managing patients with advanced stages of this disease.
Introduction
Medullary thyroid cancer (MTC) is a rare neuroendocrine tumor arising from the parafollicular cells of the thyroid gland.
1
MTC can occur in both sporadic and hereditary forms. Sporadic MTC accounts for approximately 75% of cases, while the remaining 25% are associated with inherited germline mutations in the rearranged during transfection (
The clinical presentation of MTC can vary widely; the most common initial symptom is the presence of a thyroid nodule. Other presenting features may include metastatic disease, symptoms related to hormonal hypersecretion (such as diarrhea and facial flushing), ectopic Cushing’s syndrome, or discovery during family screening. 5 One of the critical reasons for the poor prognosis associated with MTC is the late stage at which it is often diagnosed, with 70% of patients presenting with cervical lymph node metastases and 10% having distant metastases at the time of diagnosis. 2
The epidemiology of MTC reflects its rarity among thyroid malignancies. In the United States, it is estimated that approximately 1200 new cases of MTC are diagnosed each year and account for less than 5% of all thyroid cancers. 6 Despite its low prevalence, MTC is responsible for 13% of deaths attributed to thyroid cancer, highlighting its aggressive nature and significant impact on mortality within the thyroid cancer population.7,8 Furthermore, approximately 10.5% of patients with metastatic thyroid carcinoma have MTC.2,7–9 The prognosis for patients with MTC is generally poor, particularly for those with metastatic disease. The 10-year survival rate for patients with metastatic MTC is only 21%. 6
Over the past decade, the landscape of systemic treatment for MTC has evolved significantly with the introduction of several targeted tyrosine kinase inhibitors (TKIs). 10 These agents, which include the FDA-approved multikinase inhibitors (MKI) vandetanib, 11 cabozantinib, 12 and the highly selective RET inhibitor, selpercatinib, 13 have demonstrated improved progression-free survival (PFS) and, in some cases, overall survival compared to traditional cytotoxic chemotherapy. The approval of these TKIs has provided clinicians with more effective systemic treatment options for patients with advanced, progressive, or metastatic MTC. Despite the progress made with targeted therapies, the management of MTC remains challenging, and the prognosis for patients with advanced disease remains guarded. Ongoing research is focused on identifying additional therapeutic targets, exploring combination strategies, and optimizing the use of existing treatments to further improve outcomes for patients with this rare and aggressive form of thyroid cancer.2,14
This review will provide a comprehensive overview of the current systemic treatment options for MTC, focusing on the efficacy, safety, and practical considerations for utilizing these therapies in clinical practice. By summarizing the available evidence and highlighting the evolving treatment landscape, this review aims to assist clinicians in the optimal management of patients with advanced, progressive, or metastatic MTC.
Molecular basis of MTC
The discovery of
The RET protein has an extracellular ligand-binding domain composed of a cadherin-like region and a highly conserved cysteine-rich domain. The intracellular region consists of two tyrosine kinase domains. Ligand binding causes the cysteine-rich domain to facilitate receptor dimerization, producing autophosphorylation and activation of tyrosine kinase and signaling pathways including
Although it is not clear how
Multikinase inhibitors
Vandetanib
Vandetanib is an MKI approved for advanced MTC,10,31 targeting the
The ZETA study
This was a multicenter, randomized, double-blind phase III clinical trial that evaluated the efficacy and safety of vandetanib in patients with advanced MTC. The trial included 331 patients with locally advanced or metastatic MTC who were randomized to vandetanib 300 mg daily or placebo until disease progression. This study found that patients treated with vandetanib showed an improvement in PFS compared to placebo (hazard ratio (HR) 0.46; 95% confidence interval (CI) 0.31–0.69,
Furthermore, vandetanib demonstrated improvement in objective response rate (ORR; 20%, 95% CI 8%–39%), disease control rate (73%, 95% CI 54%–88%), and calcitonin and CEA levels.
11
An essential piece of information is that documentation of RECIST tumor progression was not mandatory to participate in this trial.
35
Post hoc analysis of the ZETA trial evaluating patients with symptomatic and progressive disease (
Response and side effects of systemic treatments for MTC.
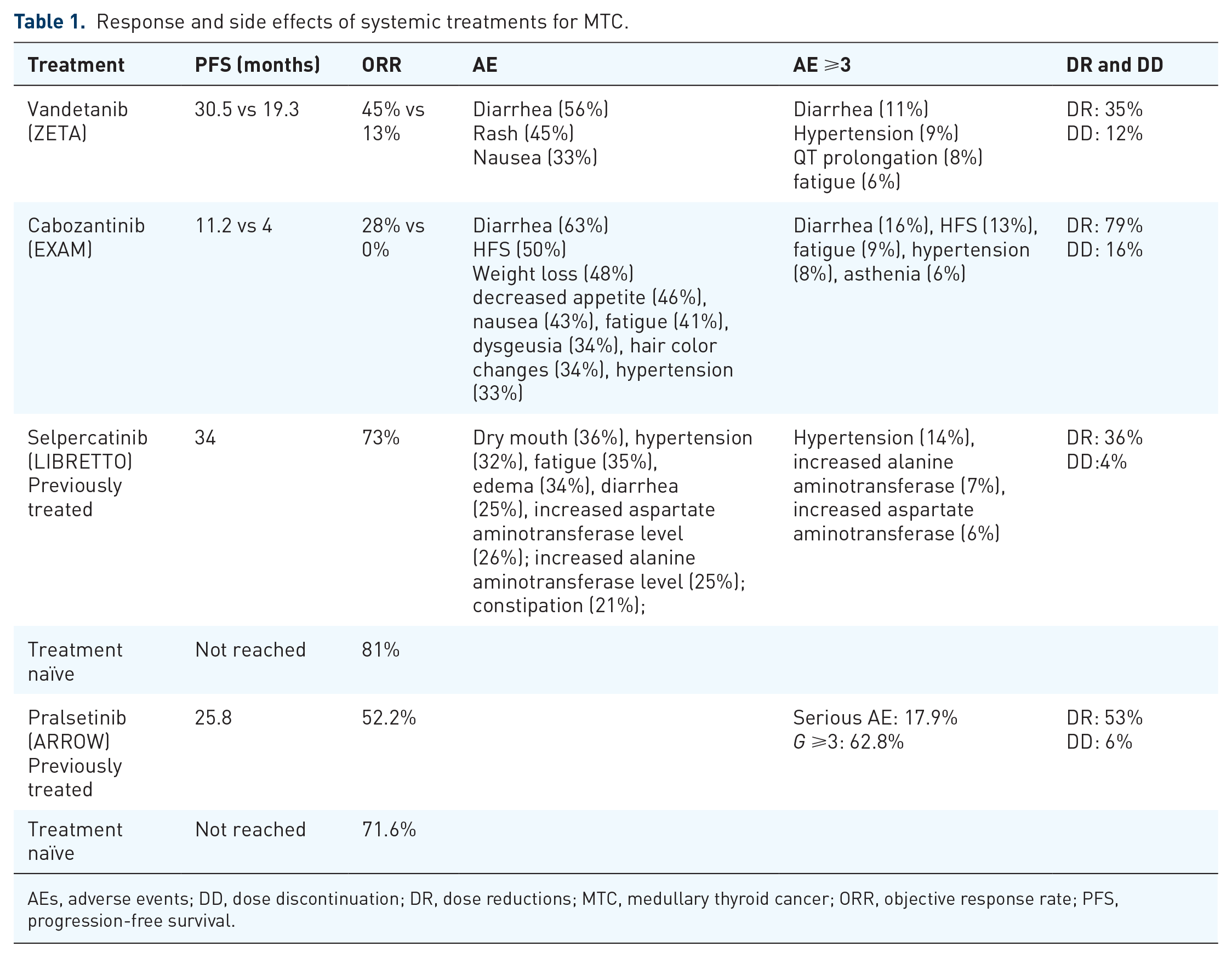
AEs, adverse events; DD, dose discontinuation; DR, dose reductions; MTC, medullary thyroid cancer; ORR, objective response rate; PFS, progression-free survival.
The most common side effects associated with vandetanib are hypertension, diarrhea, dermatitis, fatigue, nausea, headache, hypocalcemia, decreased glucose, increased alanine aminotransferase, QT prolongation, and torsades de pointes. 38 After 48 months of vandetanib use, cases of renal failure, heart failure, cholecystitis, acute pancreatitis, posterior encephalopathy, and skin cancer have been reported 37 (Table 1).
The EMA has highlighted the importance of RET mutation status in guiding the use of vandetanib for advanced MTC. 39 While vandetanib is approved for treating symptomatic, aggressive, and unresectable locally advanced or metastatic MTC, its efficacy is reportedly lower in patients without RET mutations. Consequently, the EMA recommends determining RET mutation status when feasible to inform treatment decisions and specifically recommends that vandetanib should not be administered to patients in whom RET mutation status is not known or is negative (https://www.ema.europa.eu/en/documents/dhpc/direct-healthcare-professional-communication-dhpc-caprelsa-vandetanib-restriction-indication_en.pdf).
Additionally, the EMA has mandated further research to evaluate the benefit-risk profile of vandetanib in RET-negative patients, such as the international observational study (Caprelsa 104), which aims to compare PFS between RET-positive and RET-negative groups. These findings emphasize the need for personalized therapeutic strategies in MTC management.
Cabozantinib
Cabozantinib is an MKI targeting
The EXAM study
This phase III clinical trial included 330 patients with recurrent, metastatic, or locally advanced unresectable MTC. Patients were randomized to receive either cabozantinib at a dose of 140 mg or placebo. The study demonstrated a significant improvement in PFS for the cabozantinib group compared to the placebo group, with a median PFS of 11.2 versus 4 months, respectively (HR 0.28, 95% CI 0.19–0.40). A partial response (PR) was observed in 27% of the patients treated with cabozantinib.
12
Final survival analysis showed a 5.5-month increase in overall survival. However, this was not statistically significant (26.6 vs 21.1 months, HR 0.85, CI 0.64–1.12;
Cabozantinib is associated with a range of adverse events (AEs) that physicians should recognize timely for appropriate management. The most common AEs reported include diarrhea, fatigue, nausea, decreased appetite, palmar-plantar erythrodysesthesia syndrome (hand-foot syndrome), weight loss, and dysgeusia (altered taste; Table 1). These events are typically mild to moderate in severity but can significantly impact a patient’s quality of life if not properly managed. Diarrhea is one of the most frequently reported AEs, occurring in up to 69% of patients. This can lead to dehydration, electrolyte imbalances, and impaired nutritional status if not promptly addressed. Prompt intervention with anti-diarrheal medications, dose modifications, and supportive care is essential to manage this side effect. Palmar-plantar erythrodysesthesia syndrome, or hand-foot syndrome, is another common AE, affecting up to 45% of patients. This condition is characterized by redness, swelling, and pain in the palms of the hands and soles of the feet, which can be debilitating and lead to difficulty with daily activities. Proper skin care, dose modifications, and the use of topical treatments are important in managing this side effect. Fatigue is also a frequently reported AE, occurring in up to 45% of patients. This can significantly impact a patient’s quality of life and ability to perform daily activities. Careful monitoring and the use of supportive measures, such as energy conservation techniques and physical therapy, are important in managing cabozantinib-related fatigue. In addition to these common AEs, cabozantinib has also been associated with more serious, potentially life-threatening side effects, including hemorrhage, gastrointestinal perforations and fistulas, thrombotic events, impaired wound healing, hypertension, proteinuria, hypocalcemia, increased liver enzymes, and osteonecrosis of the jaw (Table 1).
Selective RET inhibitors
The advantages of selective
Half maximal inhibitory concentration (nM) for multikinase inhibitors and selective RET inhibitors.

Source: Adapted from Subbiah et al. 45
RET, rearranged during transfection; VEGFR, vascular endothelial growth factor receptor; WT, wild type.
Selpercatinib
Selpercatinib (formerly known as LOXO-292) is a highly selective ATP-competitive small molecule
The LIBRETTO-001 study
This was an open-label phase I–II clinical trial that evaluated the efficacy and safety of selpercatinib in patients with
Long-term data from the LIBRETTO-001 trial confirm the safety and efficacy of selpercatinib in patients with RET-mutant MTC and RET-fusion-positive thyroid cancer. 56 Among cabozantinib/vandetanib-naïve MTC patients, the ORR was 82.5%, with a median PFS not reached at a median follow-up of 42.4 months. For pretreated MTC patients, the median PFS was 41.4 months. In treatment-naïve TC patients, the ORR was 95.8%, and the median PFS was not reached at a median follow-up of 24.9 months. Selpercatinib continues to demonstrate durable and robust responses, with a favorable safety profile in both treatment-naïve and previously treated patients. 56 These findings are highly encouraging and warrant further exploration.
The LIBRETTO-531 study
This global, phase III, open-label trial compared the efficacy and safety of selpercatinib versus vandetanib/cabozantinib (control group) as first-line therapy in patients with progressive, locally advanced or metastatic
Pralsetinib
Pralsetinib is an inhibitor of wild-type
The ARROW study
The ARROW phase I/II study evaluated the safety and antitumor activity of pralsetinib, a selective RET inhibitor, in patients with RET-altered thyroid cancers.
58
Among 122 patients with RET-mutant MTC and 20 with RET-fusion-positive thyroid cancers, pralsetinib demonstrated high efficacy. Treatment-naïve RET-mutant MTC patients achieved a 71% overall response rate (ORR), while those previously treated with cabozantinib, vandetanib, or both had a 60% ORR. RET-fusion-positive thyroid cancer patients showed an ORR of 89%. Common grade ⩾3 AEs included hypertension (17%), neutropenia (13%), lymphopenia (12%), and anemia (10%), with serious AEs reported in 15% of patients. Despite its strong antitumor activity, pralsetinib’s safety profile necessitates careful management, as 4% of patients discontinued treatment due to AEs, and 1% experienced a fatal treatment-related event. These findings support pralsetinib as a promising therapy for RET-altered thyroid cancers.
58
Updated analysis of the thyroid cancer population included 61 patients with
Additional MKI for MTC
Lenvatinib
Lenvatinib is an MKI primarily targeting VEGFR 1–3, fibroblast growth factor receptors 1–4, RET, c-KIT, and platelet-derived growth factor receptor alpha (PDGFRα). By inhibiting these pathways, lenvatinib exerts potent anti-angiogenic and anti-proliferative effects. 60 Clinical trials and guidelines underscore its efficacy in improving PFS and overall response rates in radioactive iodine-refractory thyroid carcinoma, 61 although its use is associated with notable adverse effects such as hypertension, fatigue, and proteinuria. 61 This drug is not approved for MTC. In a phase II trial involving 59 patients with unresectable progressive MTC, the ORR by RECIST v1.0 was 36% (95% CI 24%–49%), with all responses being partial. 62 Importantly, this response was consistent regardless of prior anti-VEGFR therapy. The disease control rate was notably high at 80% (95% CI 67%–89%), with stable disease (SD) observed in 44% of patients. The median time to respond was 3.5 months (95% CI 1.9–3.7), and the median PFS reached 9.0 months (95% CI 7.0–not evaluable). Common grade 3/4 AEs included manageable toxicities such as diarrhea (14%), hypertension (7%), decreased appetite (7%), fatigue, dysphagia, and elevated liver enzymes (5% each). 62
Lenvatinib has shown promise as a salvage therapy for advanced metastatic MTC in patients who have lost clinical benefit from previous TKIs. In a real-world study involving 10 patients, lenvatinib demonstrated disease control, with SD observed in all patients at the first evaluation, and SD or PR maintained in nine patients at the last evaluation. 63 Bone metastases showed SD control in approximately 60% of cases. While tumor markers (calcitonin and CEA) initially decreased, they increased at later evaluations. AEs necessitated dose reductions in seven patients. This study highlights lenvatinib as a potential option for patients with progressive MTC, particularly those with negative RET status or limited access to newer targeted therapies. 63
Sorafenib
This MKI is used in the treatment of advanced renal cell carcinoma, hepatocellular carcinoma, and thyroid carcinoma.64,65 Sorafenib inhibits kinases such as VEGFR, RET, RAS, BRAF, and PDGFR. 65 Common side effects of sorafenib include hand-foot skin reaction, diarrhea, hypertension, and fatigue. More severe side effects can include cardiovascular events, hemorrhage, and hepatotoxicity, necessitating close monitoring during treatment. 66
In the context of MTC, sorafenib has shown efficacy in managing disease progression. A meta-analysis of 8 trials involving 101 patients with metastatic MTC reported that the overall PR rate was 21% (95% CI 9–33), and the SD rate was 58% (95% CI 41–75). 67 This suggests that sorafenib can achieve disease control in a significant proportion of patients. The most observed adverse effects (AEs) included hand-foot syndrome, diarrhea, alopecia, mucositis, skin rash, fatigue, and hypertension. 67
Real-life studies
Real-life studies are crucial in medicine, especially for drugs treating advanced thyroid carcinoma, as they provide insights into the drug’s effectiveness and safety outside the controlled environment of clinical trials. These studies capture a broader range of patient responses and side effects, reflecting diverse populations and real-world conditions. Consequently, they help refine treatment protocols, guide clinical decision-making, and improve patient outcomes by bridging the gap between clinical research and everyday practice. A work published in 2022 evaluated real-life studies of MKI in thyroid carcinoma. 68 It included four studies in MTC with vandetanib, cabozantinib, and lenvatinib.63,69–71 These real-life studies reported comparable PFS to pivotal clinical trials and similar toxicity profiles. Other studies with smaller samples have also found similar results.70,72
Two real-life studies with vandetanib are important to mention. The first one is a retrospective analysis from Italy with a sample of 79 MTC patients treated with vandetanib which identified predictors of prolonged response and better outcomes. 34 Patients treated for ⩾12 months demonstrated significantly longer PFS compared to the ZETA trial, with younger age at diagnosis and treatment initiation being notable predictors in long- and very long-term responders. In addition, early treatment initiation in symptomatic patients, even without RECIST progression, and the development of AEs were associated with improved outcomes. These findings highlight the importance of timely intervention with vandetanib in younger, symptomatic patients with good ECOG performance status. 34 The second one is a long-term follow-up study from the Institut Gustave Roussy of 76 patients with advanced MTC treated with vandetanib over a maximum follow-up of 11 years. 37 The median PFS was 22.7 months, and 27.6% of patients were classified as long-term users (treatment >48 months), with a median treatment duration of 68.1 months and a median PFS of 73.2 months. Long-term users showed an 85.7% ORR and a median response duration of 70.4 months. Younger age at diagnosis and the absence of tumor progression before treatment initiation were significant predictors of durable response. However, long-term use was associated with late-onset AEs, including renal failure. 37
Ectopic Cushing’s and MTC
Paraneoplastic syndrome of ectopic adrenocorticotropic hormone (ACTH) secretion inducing severe Cushing syndrome in MTC occurs in approximately 0.6% of cases. The presence of hypercortisolism resulting from ectopic Cushing’s is associated with a dismal prognosis resulting in fatal complications in approximately 50% of affected cases. 73 Therefore, given the poor outcomes of this associated manifestation, the management of hypercortisolism should be treated urgently as an oncologic emergency. Medical management to attempt rapid stabilization may include cortisol-lowering therapies, such as metyrapone or ketoconazole, mifepristone to block the glucocorticoid receptor in hyperglycemic patients, and bilateral adrenalectomy in specific scenarios, in reality, management of the underlying malignancy is essential to provide a long-term resolution of the hypercortisolism. 74 MKIs have demonstrated beneficial effects in the management of ectopic Cushing’s syndrome from MTC-related ACTH secretion.75–78 Recently, emerging reports have also supported a beneficial effect in hypercortisolism control from high-specific RET inhibitor selpercatinib in RET-altered MTC.79,80 Therefore, based on supporting evidence of oral antineoplastic agents for MTC in both tumor responses and rapid hypercortisolism control, ectopic Cushing’s in MTC should be considered as an urgent indication to start systemic therapy. Aggressive supportive measures to monitor electrolytes and resulting complications from cortisol excess are essential to facilitate the tolerability of systemic therapy.
Resistance to MKI and specific RET inhibitors and future options
Patients treated with MKI and RET-specific inhibitors may present with acquired resistance, defined as an initial clinical response to therapy and subsequent resistance to therapy despite continuous treatment with the MKI, 2 or primary resistance, where there is no clinical response. This last rare situation may present in around 2% of clinical trials.57,59,81 Several mechanisms may explain resistance to these treatments.82,83 Resistance may occur due to the selection of clones with new on-target somatic mutation or the development of alterations off-target, such as MET or KRAS amplifications.15,81,83–86 Second-generation selective RET inhibitors such as TPX-0046 inhibit RET solvent from mutations but lack activity against gatekeeper mutations. 81 However, the clinical trial of TPX-0046 was terminated (NCT04161391). Several second-generation RET inhibitors are under scrutiny (NCT04161391, NCT04683250, NCT05241834).
Treatment selection and timing of systemic therapy initiation
The optimal timing for starting systemic treatment in patients with advanced MTC is less clear compared to radioiodine-refractory differentiated thyroid cancer (DTC).6,87–89 In radioiodine-refractory DTC, data support that progressing target lesions larger than 1 cm in diameter, according to RECIST 1.1 criteria (showing more than a 20% increase), constitute a clear criterion for considering the initiation of systemic treatment.71,90–92
As previously mentioned, the ZETA trial, although demonstrating an interesting ORR, was unable to accurately determine the real PFS in the cohort of patients treated with vandetanib. 35 This was primarily because the criteria for initiating systemic treatment relied mainly on the presence of advanced disease with calcitonin levels exceeding 500 pg/mL. Relying solely on calcitonin levels without documented disease progression through imaging might have resulted in the treatment of patients with SD or indolent tumors that did not require immediate intervention. 35
As the first study with systemic therapy performed in thyroid cancer, the ZETA trial underscored the importance of documented disease progression as a critical inclusion criterion, a principle subsequently adopted in the EXAM trial of cabozantinib. 12 This highlighted the need for a more comprehensive approach to patient selection for systemic therapy, combining biomarkers with imaging studies to ensure appropriate and timely intervention.
The systemic therapies currently approved for MTC have not demonstrated an improvement in OS. Consequently, there is a lack of evidence-based guidelines on when to initiate these treatments and how to monitor patients with indolent disease. Treatment decisions are primarily based on the clinical judgment and experience of healthcare providers. The ESMO guidelines for the management of thyroid cancer strongly recommended to involve a multidisciplinary team—including surgeons, endocrinologists, nuclear medicine physicians, medical and radiation oncologists, pain management specialists, and palliative care experts—to ensure the best possible care for these patients. 88 Active treatment, whether locoregional interventions or systemic TKIs, should be considered in cases with symptoms, lesions near vital structures, high tumor burden, or disease progression. 88
On the other side, the first American Thyroid Association guidelines for MTC considered that asymptomatic patients with small-volume metastatic disease that remains stable or progresses slowly, as determined by anatomical imaging, typically do not require immediate systemic therapy. For these patients, whose calcitonin and carcinoembryonic antigen doubling times exceed 2 years, the initiation of systemic treatment may not be necessary. Instead, careful monitoring and regular follow-up should be prioritized. 89
When considering systemic therapy for such patients, it is crucial to engage in a comprehensive discussion. This conversation should cover the potential benefits and risks associated with treatment, the patient’s quality of life, and the natural history of their disease. The decision to start systemic therapy should be a shared one, taking into account the patient’s preferences, overall health status, and the likelihood of treatment-related side effects.
89
The updated American Thyroid Association guidelines for MTC provided a clear and consistent recommendation: patients with significant tumor burden and symptomatic or progressive metastatic disease, as defined by RECIST criteria, should be considered for systemic therapy with MKIs targeting both
These guidelines have traditionally applied to all patients, regardless of the presence of germline or somatic
One significant limitation of this approach is the limited and uneven availability of selective
The identification of RET mutations plays a pivotal role in the management of MTC.39,94 Germline RET mutation testing is essential for all patients diagnosed with MTC, either before or after surgery, as it informs hereditary risk assessment and screening for multiple endocrine neoplasia type 2 (MEN2) syndromes. Furthermore, testing for somatic RET mutations is critical in patients being considered for systemic therapy, particularly with highly selective RET inhibitors such as selpercatinib and maybe in cases with structural disease, biochemical persistence of the disease, and risk factors for structural disease development. 95 The availability of RET mutation status allows for personalized treatment strategies, maximizing therapeutic efficacy and minimizing unnecessary interventions. 39 This approach aligns with current guidelines and recent evidence highlighting the transformative impact of RET-targeted therapies on disease outcomes.
Although hereditary MTC is primarily driven by germline RET mutations, additional somatic events can significantly influence disease behavior and therapeutic choices. Recent findings indicate that a noteworthy subset of MTC tumors harbor in-frame RET insertions/deletions, which can be associated with more aggressive disease yet remain highly responsive to selective RET inhibitors such as selpercatinib. 96 Somatic retroposed copies of the RET gene have also been detected in both sporadic and hereditary MTC, underscoring the complexity of tumor evolution and the potential for additional mutational “hits” beyond the germline event. 97 Furthermore, heterogeneity in RET mutations—where primary tumors and their metastases display distinct somatic variants—has been documented in advanced MTC, highlighting the importance of a comprehensive molecular assessment that extends beyond germline testing alone. 98 Together, these studies support the inclusion of somatic genetic testing in hereditary MTC to refine prognostication, guide targeted therapy, and ensure that all actionable RET alterations are identified. However, while this approach is seldom required in routine clinical practice, it can be valuable for clinical research and for guiding future research directions.
Neoadjuvant therapy for locally advanced MTC
There is limited published data on the use of MKIs as neoadjuvant therapy in patients with MTC. The earliest report, dating back to 2010, described a patient initially misdiagnosed with anaplastic thyroid carcinoma and treated with sunitinib, resulting in tumor reduction. The correct diagnosis of MTC was confirmed through pathological examination after surgical intervention. 99 A second case report was published with successful results after the off-label use of lenvatinib. 100
Another case reported the use of vandetanib in a pediatric patient, where surgical treatment became feasible with an R1 resection after 66 months of therapy, including a dose escalation from 100 to 200 mg of vandetanib at 43 months. 101 In addition, Russel et al. 102 showed an R1 resection after crizotinib/alectinib in one patient with MTC presenting an ALK fusion.
To our knowledge, to date, there have been no published reports on the use of cabozantinib as neoadjuvant therapy for MTC.
In a published series, none of the six MTC patients treated with vandetanib as neoadjuvant therapy achieved sufficient tumor reduction to allow for surgical resection. 103 In contrast, selpercatinib has demonstrated a higher ORR in patients with advanced MTC. 57 A recent case series of neoadjuvant therapy with selpercatinib reported tumor reductions ranging from 24% to 55% in four patients, enabling R1 resection after selpercatinib treatment. 104 However, in the recently published neoadjuvant real-world experience in Latin America, the only patient who received selpercatinib experienced only a 25% reduction in tumor size, with the final surgical outcome classified as R2 due to an extensive inaccessible mediastinal mass. 103
Another crucial point is that the necessary time for MKI or highly selective RET inhibitors to ensure a safe surgical procedure is at least five half-lives of the administered drug. 105 In addition, the decision to resume these drugs after a major surgical procedure should be based on clinical judgment regarding adequate wound healing. Theoretically, after five half-lives, the body has almost completely eliminated these drugs. However, their half-lives vary significantly, with lenvatinib having an estimated half-life of 28 h, and vandetanib 19 days. Therefore, five half-lives for lenvatinib are 6 days, but for vandetanib, it is more than 3 months. Most authors recommend waiting 1 week for surgery for most MKIs, except for vandetanib. However, in the published successful neoadjuvant case with vandetanib, it was discontinued 1 week before surgery and resumed 1 week after with no complications. 101
In conclusion, the timing and choice of systemic therapy in advanced MTC should be personalized, considering the patient’s clinical status, disease progression, and availability of targeted therapies. A multidisciplinary approach and thorough patient engagement are crucial for optimal treatment planning and execution.
Figure 1 presents a streamlined algorithm designed to assist in identifying the most appropriate moment to initiate systemic therapy for patients with advanced MTC.

Proposed algorithm for the treatment of advanced MTC.
Conclusion
Patients with metastatic and progressive MTC are at risk of local complications from tumor growth as well as systemic complications due to hormonal production, such as Cushing’s syndrome and severe diarrhea. Moreover, these patients have significantly reduced survival rates. The advent of TKIs for MTC represented a paradigm shift, offering a viable clinical option for patients previously considered untreatable. Currently, the development of newer therapies, particularly selective RET inhibitors, marks a significant milestone in the treatment of MTC. Clearly, the future for patients with metastatic, progressive, and symptomatic MTC is becoming increasingly hopeful.
