Abstract
Background:
Masculinising hormone therapy with testosterone is used to align an individual’s physical characteristics with their gender identity. Standard testosterone doses and formulations recommended for hypogonadal cisgender men are typically administered, although there are currently limited data evaluating the use of 1% testosterone gel in gender-affirming hormone therapy regimens.
Objectives:
The objective of the study was to assess the prescription patterns and serum total testosterone concentrations achieved with 1% testosterone gel in trans and gender diverse individuals.
Materials and Methods:
A retrospective cross-sectional analysis was undertaken of trans individuals at a primary and secondary care clinic in Melbourne, Australia. Sixty-seven individuals treated with 1% testosterone gel were included. Primary outcomes were testosterone dose and serum total testosterone concentration achieved.
Results:
Median age was 25 (22–30) years and median duration of testosterone therapy was 12 (7–40) months. Thirty-five (52%) individuals had a nonbinary gender identity. Initial median testosterone dose was 25 mg (12.5–31.3) daily. Fifty-two (78%) individuals commenced doses <50 mg daily, the recommended starting dose for hypogonadal cisgender men. Median total testosterone concentration achieved was 11.9 nmol/l (7.3–18.6). Polycythaemia (haematocrit >0.5) was documented in eight of 138 (6%) laboratory results in six individuals.
Discussion and Conclusions:
One percent testosterone gel achieves serum total testosterone concentrations in the cisgender male reference range. A high proportion of individuals had a nonbinary gender identity and most individuals commenced a lower dose than that typically administered to hypogonadal cisgender men, potentially related to slow or ‘partial’ masculinisation goals.
Introduction
Testosterone therapy is a necessary component of management for some transgender (trans) individuals (including those with a binary and/or nonbinary gender identity) to permit development of physical characteristics to align with their gender identity. Testosterone therapy is associated with reductions in gender dysphoria and improvement in mental health.1–3 Standard doses of parenteral or transdermal testosterone used to treat hypogonadal cisgender men are recommended in consensus guidelines for the care of trans adults.4,5
One percent testosterone gel is a transdermal formulation recommended in the position statement on the hormonal management of adult trans and gender diverse individuals. 6 In Australia, 1% testosterone gel is available in a metered-dose pump in which each pump actuation delivers 12.5 mg testosterone, or as individual sachets each containing 50 mg testosterone. The recommended starting dose in hypogonadal cisgender men is 50 mg once daily. 7 There are, however, currently limited published data evaluating the use of 1% testosterone gel in trans and gender diverse individuals.
As such, in this retrospective audit of adult trans and gender diverse individuals prescribed 1% testosterone gel, we aimed to evaluate the serum testosterone concentrations achieved, the testosterone dose prescribed, the prescription patterns of 1% testosterone gel by gender identity and the prevalence of polycythaemia.
Materials and methods
A retrospective audit of electronic medical records was performed of consultations for trans and gender diverse individuals at a primary care clinic and an endocrine clinic in Melbourne, Victoria, Australia. Data were collected from new consultations between 22 February 2016 and 12 September 2021. The study was approved by the Austin Health Human Research Ethics Committee (Audit/21/Austin/69) and Thorne Harbour Health (THH/CREP 19/015) that waived the need for informed consent.
This retrospective cross-sectional analysis included trans and gender diverse individuals treated with 1% testosterone gel (Testogel; Besins Healthcare, Sydney, Australia) who had at least one serum total testosterone concentration available while on testosterone therapy. If an individual changed testosterone formulation, their 1% testosterone gel dose and serum total testosterone concentration were included in this analysis if there was clear documentation of the timing of previous formulation and there was an adequate time between cessation of the previous regimen and subsequent laboratory results on 1% testosterone gel. Individuals were excluded if laboratory results were not available in our clinic databases.
The primary outcomes of interest were serum total testosterone concentration and 1% testosterone gel dose. The initial 1% testosterone gel dose was stratified into groups of <50 mg or ⩾50 mg, the recommended starting dose for hypogonadal cisgender men. We also analysed the prescription patterns by gender identity and prevalence of polycythaemia. Polycythaemia was defined as haematocrit >0.5, as described in the Endocrine Society clinical practice guidelines of gender-dysphoric/gender-incongruent persons. 4
Gender identity was obtained from intake forms and medical records. Classification of binary and/or nonbinary gender identity was as previously described. 8 In short, binary gender identity was defined as identification as exclusively male (including trans male, trans man and transgender male) and nonbinary gender identity encompassed all other identities (including nonbinary, transmasculine, genderqueer and agender). If an individual reported more than one gender identity, of which one was categorised as nonbinary, they were categorised as having a nonbinary gender identity for the purpose of data analysis.
As data were obtained retrospectively, total testosterone concentrations were measured using immunoassays available as standard care in Australia. All laboratories were accredited by National Association of Testing Authorities (NATA, the national accreditation body for Australia). Serum total testosterone concentrations are typically measured at baseline, every 3 months during the first year of testosterone treatment, and then every 6–12 months once stable. 6
Statistical analyses were performed using STATA version 17.0 software.
9
Data were not normally distributed so median [interquartile range (IQR)] is reported. Differences in age, duration of follow-up, testosterone dose and serum testosterone concentration stratified by gender identity were compared using the Mann–Whitney
Results
Data were collected from 89 individuals prescribed 1% testosterone gel, of whom 67 had documentation of serum total testosterone concentration and testosterone dose (Figure 1). Fifteen individuals did not have follow-up laboratory studies available in our database, six individuals had recently commenced testosterone therapy and did not have follow-up laboratory results and one individual changed to intramuscular testosterone undecanoate prior to follow-up laboratory results were undertaken.

Cohort selection strategy.
The median age was 25 (22–30) years and median duration of testosterone therapy was 12 (7–40) months. Baseline serum total testosterone concentration prior to commencement of testosterone therapy was 1.1 nmol/l (0.9–1.5). Thirty-five (52%) individuals had a nonbinary gender identity (Table 1). Individuals with a nonbinary gender identity were older than those with a binary gender identity at baseline consultation [28 years (23–40)
Gender identity (

Gender identity was not available for three individuals.
Individuals could indicate more than one gender identity, so total number exceeds number of individuals included in this analysis. Gender identities for those indicating more than one gender identity: transmasculine/nonbinary (
Initial median 1% testosterone gel dose was 25 mg (12.5–31.3) daily. Fifty-two (78%) individuals commenced doses <50 mg daily. Individuals with a nonbinary gender identity had a lower initial dose compared to individuals with a binary gender identity (12.5
From 67 individuals, 152 individual serum total testosterone concentration results were available following initiation of testosterone therapy. Median serum total testosterone concentration was 11.9 nmol/l (7.3–18.6) (Figure 2). There was no between-group difference in serum total testosterone concentration for individuals with a nonbinary or binary gender identity across the follow-up period [10.9 nmol/l (6.9–18.6)
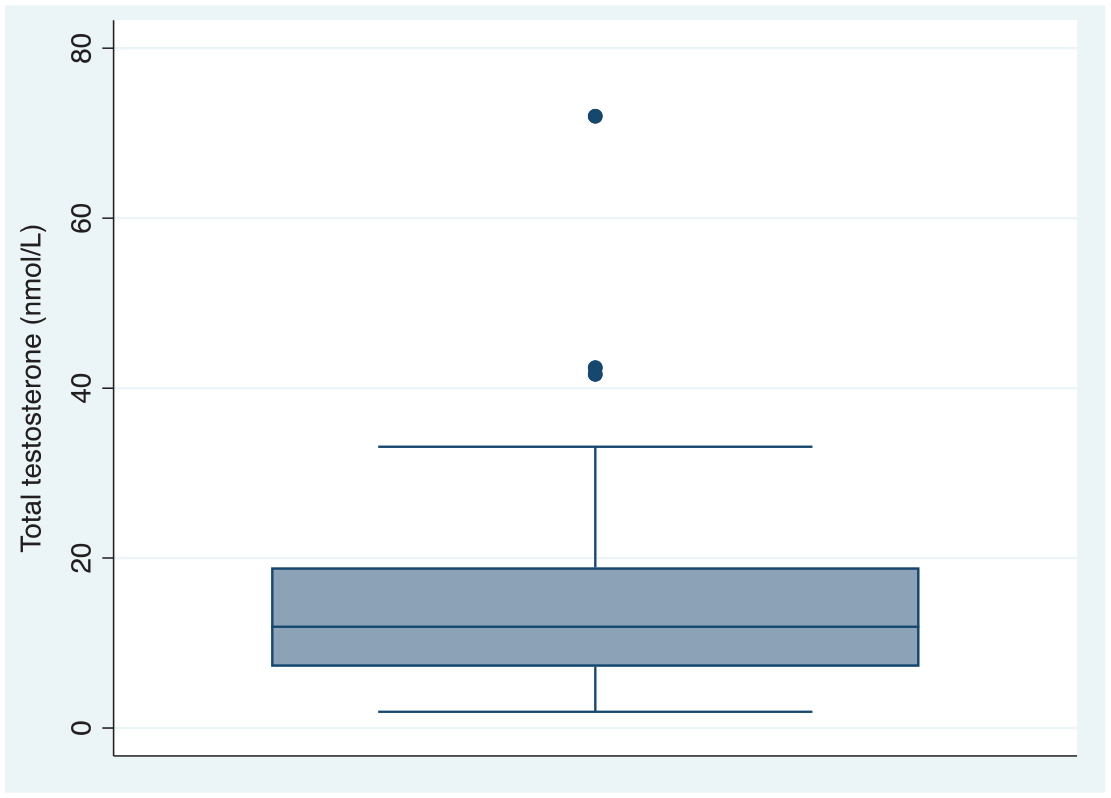
Serum total testosterone concentration (nmol/l).
Serum total testosterone concentration by 1% testosterone gel dose.

IQR, interquartile range.
Median (IQR) is presented. If an individual had more than one serum total testosterone concentration available on a particular dose, the average of these readings was taken.
Of 138 full blood examination results, polycythaemia (haematocrit > 0.5) was documented in eight (6%), with three ⩾0.52 (2%). The maximum haematocrit was 0.54. Three of these readings were in two individuals whose therapy was changed from intramuscular testosterone undecanoate and had improved following the previous results. In all, three total testosterone concentrations were in range 10–30 nmol/l, with three < 10 nmol/l and two > 30 nmol/l. Six individuals had haematocrit >0.5. No individual had haemoglobin >180 g/l.
Discussion
In this retrospective analysis of trans and gender diverse individuals, 1% testosterone gel achieved serum testosterone concentrations within the male reference range, though was frequently initiated at a lower dose than that recommended for cisgender hypogonadal men. A nonbinary gender identity was reported in 52% of individuals in this analysis and was associated with a lower 1% testosterone gel dose at initiation. Polycythaemia was recorded in 6% of laboratory results.
Transdermal testosterone gel and serum testosterone concentrations in trans individuals
Testosterone is commonly administered
Thirteen percent of our cohort were treated with 1% testosterone gel, with a further 13% treated with 5% testosterone cream, and the remaining 74% treated with intramuscular testosterone. Transdermal testosterone gel was prescribed in 22% of individuals in an early report from the European Network for the Investigation of Gender Incongruence (ENIGI). 12 Similarly, 24% of trans individuals were treated with transdermal testosterone gel in a cohort from Germany, with a median testosterone dose 50 mg (25–75) daily. 13
Consensus guidelines recommend targeting serum testosterone concentrations in the cisgender male reference range for trans individuals desiring full masculinisation [typically 320–1000 ng/dl (11.1–34.7 nmol/l)].
4
It should be noted that there are limited supportive data in the trans population and suggested sex steroid concentration targets are based on expert opinion. In a previous cross-sectional analysis of our databases, median serum total testosterone concentration measured 5.6 nmol/l (1.1–17.1) with transdermal testosterone, compared to 11.5 nmol/l (1.5–21.1) with intramuscular testosterone undecanoate and 12.1 nmol/l (1.35–20.45) with intramuscular testosterone enanthate.
14
It should be noted that a small proportion of individuals (
Transdermal testosterone in individuals with a nonbinary gender identity
Fifty-two percent of individuals in our analysis reported a nonbinary gender identity, compared with a 14% prevalence in a previous retrospective evaluation of our cohort.
8
There are currently limited data reporting testosterone doses or formulations in individuals with a nonbinary gender identity. A recent analysis noted a higher proportion of ‘partial’ hormone treatment among all individuals with a nonbinary compared with binary gender identity (11%
Clinical implications
One percent testosterone gel achieved serum testosterone concentrations in the cisgender male reference range and represents an alternative testosterone formulation for trans and gender diverse individuals seeking masculinisation. Most individuals commenced low-dose testosterone therapy in our analysis and this could be a consideration for individuals seeking slow or ‘partial’ masculinisation.
Intramuscular testosterone undecanoate is the preferred first-line treatment option for masculinising hormone therapy among trans people 19 and practitioners experienced in trans and gender diverse health in Australia. 20 Intramuscular testosterone formulations, however, have been associated with a higher prevalence of polycythaemia. 14 One percent testosterone gel therefore represents an alternate formulation for individuals with a contraindication to intramuscular testosterone, or in those who develop polycythaemia on intramuscular testosterone.
Several studies have evaluated clinical outcomes with different testosterone formulations. A randomised trial comparing transdermal testosterone, intramuscular testosterone undecanoate, and testosterone propionate/testosterone enanthate found no between-group difference in body composition or time to amenorrhoea. 15 A longitudinal analysis from ENIGI, however, reported more masculine body composition changes over 1 year with testosterone esters compared with transdermal testosterone or testosterone undecanoate. 21 In another prospective evaluation, transdermal testosterone gel was associated with higher rates of persistent vaginal bleeding and spotting compared with intramuscular testosterone formulations. 16 This is in contrast to a large retrospective cohort, in which there was no difference in time to amenorrhoea (3 months) with transdermal testosterone gel or intramuscular testosterone undecanoate. 13
Potential risks of testosterone must also be considered, in context of the lack of evidence regarding optimal serum total testosterone concentrations in trans individuals. Exogenous testosterone administration in older cisgender men has been associated with a greater increase in coronary artery noncalcified plaque volume 22 but this is not consistent among all studies. 23 This is an important consideration given higher rates of myocardial infarction in observational studies of trans individuals treated with testosterone compared with the general female population, 24 although the influence of serum testosterone concentration has not been evaluated.
Limitations
Limitations to this analysis are related to its retrospective design, including missing data as these were not collected in a standardised manner. Serum total testosterone concentration results were collected
Conclusions
One percent testosterone gel achieves serum total testosterone concentrations within the male reference range in trans and gender diverse individuals seeking varying degrees of masculinisation. Over 50% of individuals had a nonbinary gender identity and over 75% commenced a lower dose than that administered to hypogonadal cisgender men, potentially related to slow or ‘partial’ masculinisation goals.
Supplemental Material
sj-docx-1-tae-10.1177_20420188221083512 – Supplemental material for Testosterone concentrations and prescription patterns of 1% testosterone gel in transgender and gender diverse individuals
Supplemental material, sj-docx-1-tae-10.1177_20420188221083512 for Testosterone concentrations and prescription patterns of 1% testosterone gel in transgender and gender diverse individuals by Brendan J. Nolan, Sav Zwickl, Alex F. Q. Wong, Peter Locke, Satu Simpson, Ling Li, Jeffrey D. Zajac and Ada S. Cheung in Therapeutic Advances in Endocrinology and Metabolism
Footnotes
Author contributions
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: BJN and ASC have received product from Besins Healthcare for investigator-initiated clinical studies using estradiol and progesterone. No monetary support from Besins Healthcare was received for these studies and Besins Healthcare has had no input into the design, analysis or writing of any manuscripts.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: BJN is a recipient of an Australian Government National Health and Medical Research Council Postgraduate Scholarship (#2003939). ASC is supported by a National Health and Medical Research Council Early Career Fellowship (#1143333) and Investigator Grant (#2008956).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
