Abstract
Background:
Second malignant neoplasms (SMN) are among the most serious long-term adverse health conditions in cancer survivors. The aim of this study was to characterize clinical findings of patients who developed thyroid cancers as SMN, and to examine genomic alterations in thyroid cancer tissue.
Methods:
Retrospective analysis of medical records from patients seen for management of thyroid cancer over 10-year period was performed. Clinical and pathologic data were retrieved from their medical charts. Tumor DNA and RNA were extracted from formalin-fixed, paraffin-embedded tissue and subjected to next-generation sequencing (NGS) using Ion Torrent Oncomine Focus Assay. Microfluidic digital polymerase chain reactions (PCRs) were performed using QIAcuity Digital PCR System to identify BRAFV600E mutations and RET/PTC fusions.
Results:
Sixteen of 620 patients operated for thyroid cancer had history of previously diagnosed malignancy. Eight patients were male and eight patients were female, with a median age at diagnosis of 58.5 years (range, 4–78). Four patients had history of pediatric malignancy (PedCa), and 12 patients had a history of prior malignancy as an adult (AdCa). The latency periods for development of SMN in PedCa and AdCa patients were 10.8 (±5.2) years and 9.5 (±5.2) years, respectively. Histopathology revealed papillary thyroid cancers in 15 cases, and follicular thyroid cancer in 1 case. All tumors were classified as T1 or T2, and there were no patients presenting with metastases at the time of surgery. Genomic alterations were detected in 13/16 (81.2%) tumors including eight gene mutations (BRAFV600E (N = 4), RAS (N = 2), PI3CA (N = 2) and five gene fusions (RET/PTC1 (N = 4) and STRN/ALK (N = 1). In patients with PedCa and AdCa, mutations were detected in 1/4 (25%) and 7/12 (58.3%), respectively, p = 0.56; and fusions were detected in 3/4 (75%) and 2/12 (16.6%), respectively, p = 0.06. In patients with and without history of therapeutic irradiation, mutations were detected with the same frequencies (5/10 (50%), and 3/6 (50%), respectively, p = 1.0). Gene fusions were detected in patients with and without history of irradiation in 5/10 (55.5%) and 0/6 (0%), respectively, p = 0.09.
Conclusions:
Monitoring of cancer survivors for thyroid disorders allowed diagnosis of second thyroid cancers at early stages. Second thyroid cancers harbor genomic alterations that are typical for sporadic as well as for radio-induced thyroid cancers.
Background
Improvements in early detection, supportive care, and treatment have resulted in an increasing number of cancer survivors, with a current 5-year relative survival rate for all cancers combined of approximately 66.1%. 1 For some patients, these survival advances have been offset by the long-term late effects of cancer and its treatment, with second malignant neoplasms (SMNs) comprising one of the most potentially life-threatening sequelae. The number of patients with SMNs is growing, with new SMNs now representing about one in six of all reported cancers. Furthermore, among select subgroups of cancer survivors, the lifetime risk of developing second primary malignancies may be as high as 33%. 2 SMN also appear to be an important cause of death in cancer survivors. For patients with second incident cancers, 13% died of their initial cancer whereas greater than one-half (55%) died of their second malignancy. 2
Development of SMN is an important risk factor for survivors of pediatric malignancies. The survival rate for childhood cancer has increased from 1970 to the present, with 5-year survival rates now exceeding 80% overall and can reach up to 98–99% depending on specific diagnosis.3–7 The incidence of pediatric cancer is also increasing over time.4,5,8 As such, the population of pediatric cancer survivors will continue to expand with recent estimations of upward of 380,000 survivors of pediatric cancer alive today with most of these patients now as adolescents and young adults.6,9,10 Almost three quarters of pediatric cancer survivors will have at least one chronic health condition or complication by 30 years after diagnosis. The risk for development of SMN in adolescent and childhood cancer survivors ranges from 10% to 15% after 30 years of follow-up.7,11 The mortality attributable to second malignancies ranges from 12% to 19% in individuals who survived at least 5 years following a diagnosis of childhood cancer.6,10,12
Radiotherapy is an important risk factor for the development of SMN in cancer survivors. 6 The thyroid gland is uniquely sensitive to ionizing radiation, and radiotherapy was found to be associated with increased risk for development of thyroid cancer among pediatric as well as adult cancer survivors.3,13–15 Chemotherapy was also identified as factor contributing to development of thyroid cancer as SMN in childhood cancer survivors. 16 As with other SMN, thyroid cancer as a second distinct neoplasm has decreased survival in comparison with those who develop thyroid cancer as a primary neoplasm,2,17–19 and development of thyroid cancer as SMN was identified as an independent risk factor for decreased overall survival rate as compared with patients with primary thyroid cancers. 20
As cancer survivors are at high risk of developing thyroid cancer as SMN, development of appropriate strategy for patient’s evaluation, diagnosis, and optimal treatment for these patients is of the utmost importance. 21 Advances in our understanding of the pathogenesis of thyroid cancer were fostered principally through next-generation sequencing (NGS) technology. This technique allows for simultaneous high-throughput sequencing analysis of variable genetic alterations and provides a comprehensive understanding of tumor biology. The identification of genetic alterations associated with thyroid cancer has improved thyroid cancer diagnoses and treatment in patients with sporadic as well as radio-induced thyroid cancers.22–24 In contrast to the extensive set of data regarding clinical, pathological, and molecular features of sporadic and radio-induced thyroid cancers, there is limited information regarding the clinical and pathological features of thyroid cancers that have been diagnosed as a second malignancy in cancer survivors.2,20,25,26
In this study, we characterized clinical findings of patients who developed thyroid cancers as SMN and have examined thyroid oncogene mutations and fusions in thyroid cancer tissue samples.
Material and methods
Patients and tumor samples
The protocol for this study was approved by institutional review board (IRB) at Walter Reed National Military Medical Center (WRNMMC) and National Capital Region (NCR). Department of Pathology at WRNMMC reviewed medical records of patients diagnosed and treated within the National Capital Region (NCR) within the past 10 years to evaluate for cancer diagnosis prior to diagnosis of thyroid cancer. Patients were selected for potential inclusion in this study if they had a cancer diagnosis at least 36 months prior to the diagnosis of thyroid cancer to allow for treatment of the original cancer to be completed. Subjects meeting the following eligibility criteria will be included in the study: (1) Availability of specimens from thyroid tumor for DNA/RNA testing (cytological samples or frozen section or paraffin-embedded tumor tissue); (2) confirmed histological diagnosis of thyroid tumor after excision; (3) complete pathological report including histological type, level of encapsulation, presence of multifocal growth, extra-thyroidal extension, involvement of lymph nodes (number of positive and number of examined), presence of distant metastases on diagnosis; (4) follow-up data regarding treatment.
Tumor samples were obtained from the Department of Pathology at WRNMMC and histological diagnoses of thyroid cancers were confirmed by two pathologists.
Extraction of nucleic acids
DNA and RNA were extracted from formalin-fixed, paraffin-embedded (FFPE) tissue samples. The FFPE samples were de-paraffinized with xylene and rehydrated through ethanol. Samples were then treated with a Protease Digestion Buffer and then were digested with Proteinase K.
Extraction of DNA and RNA from FFPE cancer tissue samples was then performed using King Fisher Duo Thermo Fisher (Thermo Fisher Scientific Inc., Waltham, MA, USA) per the manufacturer’s instruction. Initial DNA and RNA quantification was performed by using NanoDrop spectrophotometer (Thermo Fisher Scientific). Quality control and evaluation of nucleic acid concentration was performed by RT-PCR via QuantStudioTM 6 Flex (Thermo Fisher Scientific).
Library preparation and next-generation sequencing
DNA samples were diluted to a concentration of 10 ng/15 μL in 15 μL total volume in nuclease-free water. Ten nanograms RNA was added with 3 μL SuperScript IV VILO Master MIX and additionally diluted to 15 μL total volume in nuclease-free water. Complementary DNA (cDNA) synthesis prior to library preparation for RNA panel was carried out using SuperScript VILO cDNA Synthesis Kit (Thermo Fisher Scientific) using ProFlex PCR System (Thermo Fisher Scientific).
Library preparation was performed using the Ion AmpliSeq Kit for Chef DL8 on the Ion Chef Instrument using the Oncomine Assay (comprising the DNA Oncomine Focus Assay and RNA Oncomine Fusions assay (Thermo Fisher Scientific)) following manufacturer’s instructions using a total of 10 ng input DNA and RNA per sample (minimum 0.83 ng/μL sample DNA concentration). A maximum of eight DNA/RNA samples were prepared, bar-coded per run/plate, and amplified, respectively, on a Ion 520 chips and then templated and sequenced using the using the Ion 510 & Ion 520 & Ion 530 Kit according to manufacturer’s instructions.
Ion TorrentTM 2X Oncomine Focus Assay was used for NGS to determine the mutation status of the tumor. The Oncomine Focus Assay allows concurrent analysis of DNA and RNA, enabling sequencing of 35 hotspot genes, 19 genes associated with copy number gain, and 23 fusion genes, all in a single workflow using the Ion PGM System. The DNA panel can identify hotspot mutations in the following genes: AKT1, ALK, AR, BRAF, CDK4, CTNNB1, DDR2, EGFR, ERBB2, ERBB3, ERBB4, ESR1, FGFR2, FGFR3, GNA11, GNAQ, HRAS, IDH1, IDH2, JAK1, JAK2, JAK3, KIT, KRAS, MAP2 K1, MAP2 K2, MET, MTOR, NRAS, PDGFRA, PIK3CA, RAF1, RET, ROS1, and SMO. The RNA panel can identify rearrangements in ALK, RET, ROS1, NTRK1, NTRK2, NTRK3, FGFR1, FGFR2, FGFR3, MET, BRAF, RAF1, ERG, ETV1, ETV4, ETV5, ABL1, AKT3, AXL, EGFR, ERBB2, PDGFRA, and PPARG, and all fusions were assessed for this validation. Nineteen copy number variant (CNV) targets are also included in the Oncomine Focus Panel.
NGS data analysis
Data analysis was carried out using Ion Torrent Suite Browser version 5.0 and Ion Reporter version 5.0. The Torrent Suite Browser was used to perform initial quality control including chip loading density, median read length, and number of mapped reads. The Coverage Analysis plugin was applied to all data and used to assess amplicon coverage for regions of interest. Variants were identified by Ion Reporter filter chain 5% Oncomine Variants (5.0).
Primary analysis was performed by Torrent Server (v 5.0) and further by Ion Reporter Server hosting informatics tools (Ion Reporter Software v5.0) for variant analysis, filtering, and annotations. Automatic workflow (Oncomine Focus v2.0, DNA and fusions/DNA/fusions, Single Sample) with preconfigured parameter settings (Oncomine Variants 5% Comfidence Interval (CI) somatic copy number variation (SCNV) ploidy ⩾ gain of 2 over normal) was utilized. Actionable variants with respect to labels, guidelines, and current global clinical trials were selected by Oncomine Knowledgebase, genomic analysis software.
Microfluidic digital PCR
Microfluidic digital PCR procedure was performed using the QIAcuity 8, 5-plex, the QIAcuity PCR Kit, and the 24-well 26 K Nanoplates (Qiagen, Hilden, Germany) per manufacturer’s instructions. The dPCR reaction mixture was assembled as follows: QIAcuity 4X Probe PCR Master Mix 10 μL, 10X Primer/Probe mix (0.8um Primer/0.4um probe) 4 μL, RNase-free water and DNA/cDNA template in a final volume of 40 μL. The primer/probes for detection of BRAFV600E, RET/PTC1, and RET/PTC3 were made using locked nucleic acid (LNA) technology (Qiagen). LNA bases are placed within PCR amplification primers and target-specific probes, ideally positioned at or adjacent to the mutation sites for detecting either the mutant or wild type sequence. The mixture was prepared in a pre-plate and then transferred into the 24-well 26 K Nanoplate and sealed with the Nanoplate seal. The later was then loaded to the QIAcuity 8 instrument, which is a fully automated system.
The workflow included (1) priming and rolling step to generate and isolate the chamber partitions, (2) the amplification step under the following cycling protocol: 95°C for 2 min for enzyme activation, 95°C for 15 s for denaturation, and 60°C for 30 s for annealing/extension for 40 cycles, and (3) the imaging acquisition step of all wells.
The partitions that have a target molecule inside emit fluorescence light and are brighter than those without target and are detected by selecting and reading the fluorophores in the following channels FAM, HEX, and CY5. The full workflow time was around 2 hours for the three steps. The experiments were performed using a negative control (no template control, NTC) and a positive control (DNA or RNA samples from thyroid cancer cell lines with established mutation status). All reactions had at least 25,400 partitions. Data were analyzed using the QIAcuity Suite Software V1.1.3 (Qiagen).
Statistical analysis
Statistical analysis was performed with Fisher’s exact test to compare categorical data and t test to compare continuous variables, p value < 0.05 was considered as statistically significant.
Results
Study cohort
Medical records of patients diagnosed and treated within the Walter Reed National Military Medical Center (WRNMMC) and National Capital Region (NCR) for thyroid cancer between 2010 and 2020 were reviewed for history of prior malignant neoplasm diagnosed at least 36 months prior to thyroid cancer diagnosis. This revealed 18 patients with a history of prior malignant neoplasm. Of these 18 patients, 16 had confirmed histologic diagnosis of thyroid cancer and tissue samples available for analysis and were subsequently included into the study.
Patient’s characteristics at the primary cancer diagnoses
The information on demographic and clinical data at the time of diagnoses of the first malignancies are summarized in Table 1.
Demographic and clinical data at the time of diagnoses of the first malignancies.
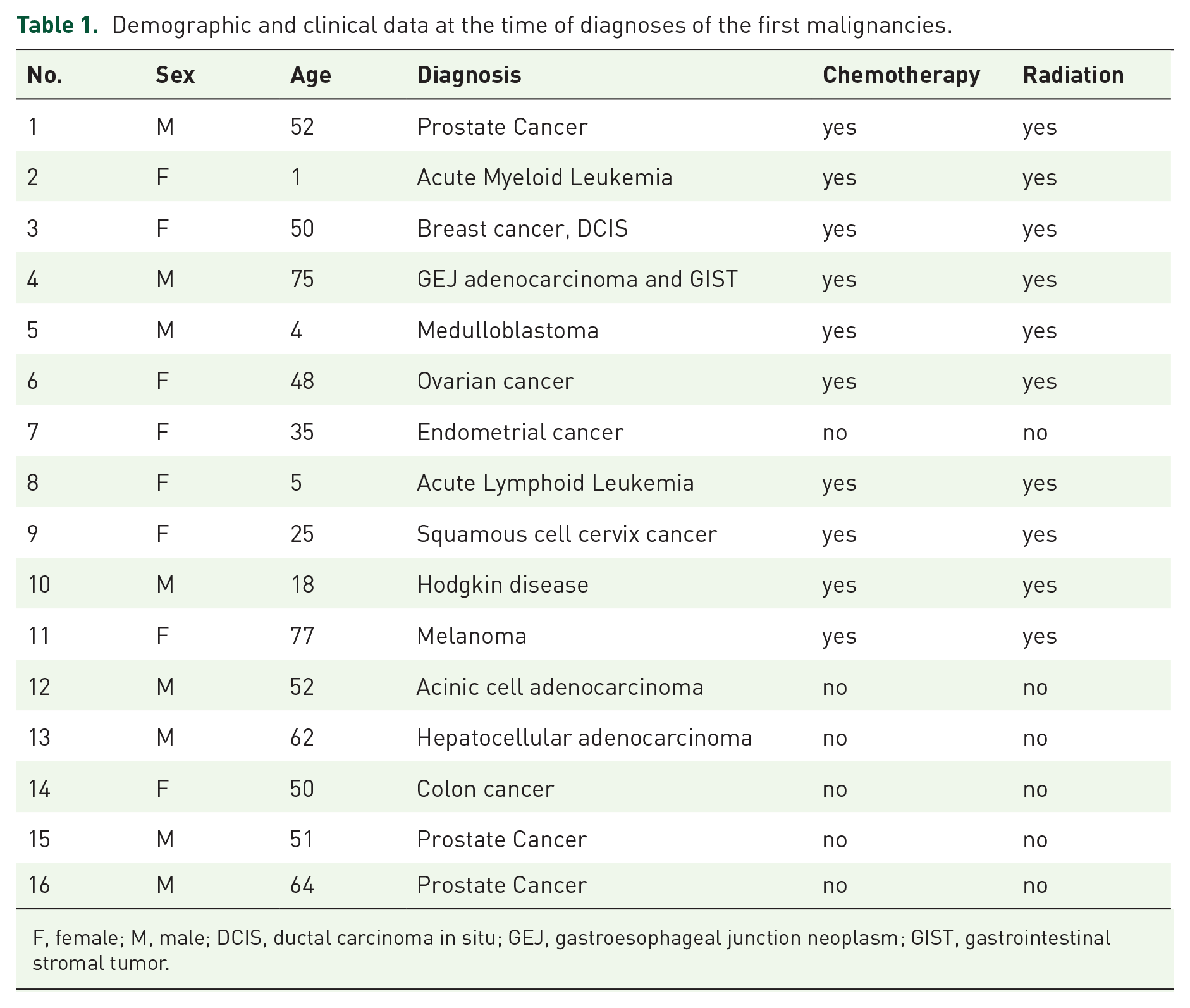
F, female; M, male; DCIS, ductal carcinoma in situ; GEJ, gastroesophageal junction neoplasm; GIST, gastrointestinal stromal tumor.
There were eight female and eight male patients. Four patients were diagnosed with pediatric or adolescent cancer (PedCa), and 12 patients were adults (AdCa) at the time of their primary cancer diagnosis. Average age at primary cancer diagnosis was 7 (±7.5) years for pediatric patients, 53.9 (±14.8) years for adult patients (p < 0.0001).
Most of pediatric/adolescent patients had hematological malignancies as their primary diagnoses. Among the adult patients, the primary diagnoses were solid tumors in all cases. One patient experienced treatment for three different malignancies including leiomyosarcoma (age = 43), DCIS (age = 49), and melanoma (age 77) prior to diagnosis of thyroid cancer at the age 85 years.
All pediatric patients were exposed to chemotherapy, and 6/12 adults were exposed to chemotherapy as part of the treatment of their initial malignancy, p = 0.23. All pediatric patients were exposed to therapeutic radiation; and 6/12 adults were exposed to therapeutic radiation for treatment of their initial malignancy, p = 0.23.
Clinico-pathological characteristics of patients with thyroid cancer
In all cases, the diagnoses of thyroid cancer as SMNs were established during the follow medical management for the first malignancy. The demographic, clinical, and pathological data are summarized in Table 2.
Clinico-pathological characteristics of patients with thyroid cancer.

AUS, atypia of undetermined significance; CPTC, classical papillary thyroid cancer; F, female; CPTC, classical papillary thyroid cancer; FTC, follicular thyroid cancer; FVPTC follicular variant of papillary thyroid cancer; M, male; PTC, papillary thyroid cancer; RAI, radio-active iodine; FLUS, follicular lesion of undetermined significance; TNM, tumor, node, metastasis.
Analysis of the patient’s age at diagnosis of first malignancy and second thyroid cancer showed that two of four PedCa patients developed thyroid cancer during pediatric/adolescence and two PedCa patients developed thyroid cancer in adulthood.
The mean latency period for development of second thyroid cancer was 9 year (range, 3–22). In PedCa and AdCa patients, the latency periods were 10.8 (range, 3–16) years, and 9.5 (range, 3–22) years, respectively, p = 0.64.
Fine needle aspiration biopsy was performed in 15/16 patients. Benign cytology was reported in 2 cases, and 12 cases cytological diagnoses evoked surgery being either malignant, suspicious for malignancy, follicular neoplasms or atypia of undetermined significance.
The final pathology revealed papillary thyroid cancers (PTCs) in 15 cases, and minimally invasive follicular thyroid cancer (FTC) in one case. Among PTCs, there were 10 classical PTCs, 4 FVPTCs, and 1 solid variant PTC. One tumor showed invasive features to include extra-thyroidal invasion into strap muscle. Loco-reginal metastases to lymph nodes or distant metastases were not reported. There were seven lesions that were graded as T1a, seven lesions that were T1b, and two lesions that were graded as T2.
Surgical treatment consisted of total thyroidectomy (11 cases) or hemi-thyroidectomy (5 cases). Treatment with radio-active iodine (RAI) was performed in four cases. Tumor recurrences or metastatic progression were not reported in any of examined patients during the follow-up periods.
Next generation sequencing
The Oncomine Focus Assay was used to analyze nucleic acids extracted from the FFPE tissue specimens from 16 patients with second thyroid cancer. A cutoff of 500X coverage was applied to all analyses. The run was considered successful and the sequencing quality adequate when the following quality metrics were met: (1) mapped reads ⩾ 300,000; (2) average base coverage depth ⩾ 1000; (3) amplicons having at least 500 reads: ⩾ 90%; (4) no strand bias: ⩾ 90%; (5) amplicons read end-to-end: ⩾ 85%. The limit for a single RNA library was set at 5000 mapped reads. Based on these criteria, DNA was of sufficient quality for analysis in all 16 cases, whereas RNA were evaluable in 10 cases. The Oncomine Focus Assay revealed DNA alterations in 8/16 examined cases and results are summarized in Table 3.

In addition, in two cases, the amplification of Androgen Receptor were identified with copy number gain of 6.1 in one BRAF positive tumor, and 5.8 in one tumor with HRAS mutation. The Oncomine Focus Assay analysis of RNA samples revealed gene translocations in 2 cases, including one case with CCDC6 – RET (RET/PTC1) fusion, and one case with STRN – ALK fusion.
Microfluidic digital PCR
The coexisting BRAFV600E and TERT promoter mutations contribute to development of aggressive PTC and PTC-specific mortality.22,27,28 Therefore, we analyzed BRAFV600E and TERT228 using microfluidic digital PCR. In PTCs tissue samples, BRAFV600E was detected in four cases (all cases were found to be BRAFV600E positive by NGS), and the ratio of mutant/total BRAF alleles ranged from 9.5% to 47.5%.
Taking into consideration high sensitivity of dPCR for detection of genomic alterations in FFPE samples, we used this technique for analysis of thyroid cancer–associated gene fusions to include (RET/PTC1, RET/PTC3, and ETV6/NTRK3). RET/PTC3 and ETV6/NTRK1 were not detected in examined RNA samples. In contrast, microfluidic dPCR allowed detection of RET/PTC1 in four cases, including three cases that were negative for fusions by NGS.
Analysis of genomic alterations and clinico-pathological data
Together, the use of the Oncomine Focus Assay and microfluidic dPCR allowed detection of genomic alterations in 13/16 examined samples, including 8 mutations and 5 fusions. Results of mutations/fusions analysis in function of clinico-pathological characteristics are summarized in Table 4.

FTC, follicular thyroid cancer; PTC, papillary thyroid cancer.
In patients with PedCa and AdCa, mutations were detected in 1/4 (25%) and 7/12 (58.3%), respectively, p = 0.56; and fusions were detected in 3/4 (75%) and 2/12 (16.6%), respectively, p = 0.06. In patients with and without history of therapeutic irradiation, mutations were detected with the same frequencies (5/10 (50%), and 3/6 (50%), respectively, p = 1.0). Gene fusions were detected in patients with and without history of irradiation in 5/10 (50%) and 0/6 (0%), respectively, p = 0.09. Most of classical PTCs harbored BRAFV600E mutations or RET/PTC1 fusions, and follicular variant PTCs had RAS mutations (Figure 1).

Microscopic images showing histological details two PTCs. (a) C-PTC harboring BRAFV600E demonstrates papillary patterns of growth (hematoxylin and eosin staining, magnification 40×); (b) detail of follicular growth areas of a NRAS-positive PTC (hematoxylin and eosin staining, magnification 40×).
Discussion
In this study, we characterized clinical and pathological characteristics of patients with thyroid cancers that developed as SMN, and examined genomic alterations in thyroid cancer tissue samples. This project was designed as a single institution, retrospective study and included data from patients operated for thyroid cancer at National Capital Consortium (NCC) over a 10-year period.
Of 620 patients treated for thyroid cancer within the NCC, within the past 10 years, 18 (2.9%) were identified as having at least one prior malignancy. Analysis of clinical and pathological findings has shown that most of examined tumors were PTCs. In all cases, surgeries were performed on early stage tumors (all tumors were classified as T1and T2, and there were no cases with metastases at the time of surgery). These results are in line with previously reported data in cancer survivors who developed second thyroid cancer after hematopoietic cell transplantation. 29 However, our results are different from the previously reported population-based studies 6 demonstrating that thyroid cancer as SMN accounts for approximately 0.59% of total cases of thyroid cancers, and up to 35% of patients with second cancers have metastases at the time of surgery.
The explanations for these discrepant results could rely on differences in studies design as well as on differences in patient’s populations and medical settings. One of the inclusion criteria’s for this study was the 36 months as the time interval to identify cancer as a second malignancy. This time frame was chosen to exclude cases that were likely to represent synchronous cancers. In addition, radio-induced thyroid cancers have been found to develop after at least 4 years post-exposure to ionizing radiation.30,31 Whereas our study have included patients with at least 36 months between the diagnoses of second thyroid cancer and primary malignancy, the population-based studies have included patients with at least 12 months between diagnoses. The fact that secondary thyroid cancers were detected at early stages in our study could be explained by specific medical setting at National Capital Consortium. All patients were treated within the military health system where follow-up and access to care is readily available. In all patients, tumors were found during routine examinations (physical and/or radiographic follow-up). It has been shown previously that surveillance with ultrasound improve the early diagnoses of clinically relevant thyroid tumors, 14 and it is likely that surveillance and access to radiographic evaluation contributed to early discovery of second thyroid cancers in this study.
Disparity between the sexes in incidence, disease aggressiveness, and prognosis has been observed in thyroid cancer. Female patients are 3–4 times more likely to develop thyroid cancer, and there is evidence that differences in the levels of estrogen and testosterone impact the severity and progression of thyroid cancer. A previously reported study demonstrated that men with thyroid cancer are more likely to present with more advanced disease, aggressive histological subtypes, and older age. 32 In the recent detailed review of 40 studies encompassing 1,613,945 patients, females were more likely to develop a second primary thyroid cancer (RR = 1.58, 95% CI, 1.2–2.01). 21
In our study, the female:male ratio was derived from among patients with thyroid cancer as a second malignancy. This cohort of patients included individuals with history of exposure to therapeutic radiation. Therefore, we have examined previously published data on sex disparity among patients with radio-induced thyroid cancer. Recent analysis of patients with sporadic and radio-induced PTC revealed significant differences in gender distribution. Among patients with sporadic and radio-induced PTCs, the female:male ratio were 3.45 to 1 and 1.8 to 1, respectively. 29 Interestingly, among children (less than 14 years old) the ratio of female:male patients in sporadic and radio-induced groups were 4.45 to 1, and 1.4 to 1, respectfully.
The origins and mechanisms of these sex differences remain a major unresolved question in cancer biology. Ongoing studies suggest a complex relationship between sex and the cancer genome landscape. The potential consequences of sex-biased gene mutations or copy number alterations include gender specific perturbations of signaling pathways, metabolic processes, changes in mRNA abundance, differential chromatin architecture, and DNA repair efficacy. 33 Significant insight into these questions on mechanism will arise from tumor whole genome sequencing and chromatin profiling efforts.
In this study, fine needle aspiration biopsies (FNAB) of thyroid nodules were performed in 15/16 patients. The cytological diagnoses of malignancy were established in seven cases, suspicious or atypia of undetermined significance in six cases, and benign cytology in two cases.
Previous study demonstrated utility of molecular testing for diagnosis of malignancy in thyroid nodules, especially in cytologically indeterminate thyroid nodules. 6 Therefore, we extended this study and examined genomic alterations in second thyroid cancers.
Next-generation sequencing (NGS) allows for simultaneous high-throughput sequencing analysis of variable genetic alterations and provides a comprehensive understanding of tumor biology. We used an Oncomine Focus Assay to determine actionable mutations/fusions in second thyroid cancers. This panel covers hotspot mutations/fusions of 52 genes with the potential for near-term clinical relevance. This approach allowed detection of mutations in 8/16 examined tumors, and gene translocations in 2/16 examined cases. Since analysis was performed using FFPE archival tissue samples, the extracted RNA in certain samples was not sufficient quality for detection of gene fusions. The high level of RNA fragmentation in FFPE samples was reported as one of the limiting factors in sequencing using Oncomine Focus Assay. 34
We recently demonstrated that microfluidic digital PCR is a highly sensitive technique for detection of gene mutations in FFPE thyroid tissue samples. 27 It has been also shown that digital PCR technique has higher sensitivity for detection of mutant alleles as compared with NGS. 35 Therefore, we have performed microfluidic digital PCR to examine gene fusions in RNA extracted from second thyroid cancers. Using this approach, we detected RET/PTC1 in three samples that were fusions-negative by NGS. In total, genomic alterations were found in 13/16 (81.25%) examined cases, and included 8 cases with gene mutations and 5 cases with gene fusions.
As expected, BRAFV600E mutations were the most common genomic abnormalities in examined thyroid cancer samples. Consistent with previous reports, BRAFV600E mutation and RET/PTC fusions were detected in classical PTCs, whereas RAS mutations were detected in tumors with follicular patterns of growth (FVPTC and FTC). The total detection rate of genes fusions was 31%, and these chromosomal rearrangements were found only in patients with history of irradiation. Among patients with history of pediatric malignancy, 3/4 (75%) of second thyroid cancers harbored gene fusions and only one case had BRAFV600E mutation. In contrast, most of patients with history of malignancy in adulthood harbored gene mutations. These data are consistent with previous reports demonstrating higher frequency of genes fusions in pediatric PTC, as compared with adult patients with PTCs.30–32
Our results also suggest a role of irradiation during childhood for development of second thyroid cancer. In our study, the average age of pediatric patients at the time of diagnoses of primary neoplasm was 7 years, and all these patients received therapeutic radiation. The pediatric thyroid is known to be sensitive to ionizing radiation as evidenced by the increased incidence of thyroid carcinoma after the Chernobyl accident in 1986.36–40 Given that there was a high frequency of fusions in a group of patients with history of pediatric malignancy, their exposure to therapeutic radiation likely contributed to development of second thyroid cancer. These observations are in line with previous report indicating that gene fusions are a major driver of radiation induced thyroid cancers. 41
In our study, the 16 patients with a second malignancy as thyroid cancer had 14 different primary malignancies. Previously published epidemiological studies demonstrated associations between the histological types of first malignancies and any subsequent thyroid cancer. Specifically, a greater risk of developing a second malignancy as thyroid cancer was found following primary breast (56.6%, 95% CI, 44.3–68.9, p < 0.001), renal cell (12.2%, 95% CI, 7.68–16.8, p < 0.001), basal cell (7.79%, 95% CI, 1.79–13.7, p = 0.011), and ovarian cancer (11.4%, 95% CI, 3.4–19.5, p = 0.005). 21
The breast and the thyroid are hormone-responsive organs that are closely related to changes in endocrine function, and the coincidence of thyroid disorder and breast cancer has long been a subject of investigation. Epidemiological studies did not establish an association between therapeutic irradiation and development of second malignancy as thyroid cancer in breast cancer survivors. Sun et al. 15 studied 55,318 women with breast cancer and found that the risk of subsequently developing thyroid cancer among women who received radiotherapy was not significantly higher than that of women who received no radiotherapy. The biological factors underlying development of a secondary thyroid cancer in breast cancer survivors could include genetic susceptibility, hormonal signaling, and pathways controlling cancer stem cell (CSC) self-renewal. Specifically, activation of the sonic hedgehog (Shh) pathway, due to overexpression of the genes encoding the components of this pathway, stimulates the growth and spread of a wide range of cancer types, including breast cancer and thyroid cancer.42,43 Studies clarifying mechanisms of CSC self-renewal in different type of cancers are of great interest; however, further studies on the relevant risk factors are needed to better understand associations between the primary cancers and second malignancies.
This study has several limitations. First, this was a retrospective design for analysis of small number of patients in the same institution. Second, we observed a trend in increase of frequencies of gene fusions in patients with history of therapeutic irradiation, but number of examined patients did not allow to perform meaningful statistical analysis. Third, focused NGS did not reveal genomic alterations in almost 20% of examined second thyroid cancer. Therefore, a multicenter, prospective large cohort study could be conducted to establish an optimal surveillance protocol for second thyroid cancer in patients with history of malignancy.
In conclusion, in this pilot study we demonstrated that assessment of thyroid gland during routine follow-up in cancer survivors allowed early detection of second thyroid cancer. This study also showed that secondary thyroid cancers harbor genomic alterations that are typical for sporadic as well as for radio-induced thyroid cancers.
Disclaimer
The views expressed in this article are those of the authors and do not reflect the official policy or position of the US Air Force, the Department of Defense or the US government. Title 17 U.S.C. 105 provides that ‘copyright protection under this title is not available for any work of the United States Government’. Title 17 U.S.C. 101 defines a United States government work as ‘a work prepared by a military service member or employee of the United States government as part of that person’s official duties’. This work was prepared as part of the official duties of KR, JW, AP, MT, JC, KJ, and VV.
Footnotes
Author contributions
Conceptualization: V.V., K.J. and K.R.; Resurces: K.R., J.W., Methodology: K.R., A.P., M.T, J.C., Data analysis K.R, V.V., K.J.; Writing and Review: K.R.; K.J., J.W., V.V.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Henry M. Jackson Foundation grant ‘Research Accelerating Military Pediatrics’; award No.: 310534-4.00-76582.
Ethical approval
The protocol for this study was approved by the IRB at Walter Reed National Military Medical Center (WRNMMC) and National Capital Region (NCR). The approval ID: WRNMMC-EDO-2020-0603, 930615. The IRB determined that this project was ‘research not involving human subjects’, and therefore granted a waiver of informed consent.
