Abstract
Landmark studies have convincingly demonstrated that atherosclerosis begins in youth. While generally asymptomatic, an increasing number of youth with disorders of lipid and lipoprotein metabolism, such as familial hypercholesterolemia, are being identified through selective and universal screening. While a heart healthy lifestyle is the foundation of treatment for all youth with dyslipidemia, lipid-lowering therapy may be required by some to prevent morbidity and premature mortality, especially when initiated at a young age. When appropriate, use of statins has become standard of care for reducing low-density lipoprotein cholesterol, while fibrates may be beneficial in helping to lower triglycerides. Many therapeutic options commonly used in adults are not yet approved for use in youth less than 18 years of age. Although currently available lipid-lowering therapy is well tolerated and safe when administered to youth, response to treatment may vary and some conditions lack an efficient therapeutic option. Thus, newer agents are needed to aid in management. Many are in development and clinical trials in youth are currently in progress but will require FDA approval before becoming commercially available. Many utilize novel approaches to favorably alter lipid and lipoprotein metabolism. In the absence of long-term outcome data of youth who were treated beginning at an early age, clinical registries may prove to be useful in monitoring safety and efficacy and help to inform clinical decision-making. In this manuscript, we review currently available and novel therapeutic agents in development for the treatment of elevated cholesterol and triglycerides.
Keywords
Introduction
In 1955, niacin was considered the initial therapeutic option for reducing elevated levels of blood cholesterol. 1 Since that time, the treatment of both elevated levels of cholesterol and triglycerides (TGs) has evolved greatly. With recognition of atherosclerotic cardiovascular disease (ASCVD) as a major public health challenge worldwide, over the past two decades, many newer lipid-lowering therapies (LLT) have been developed. Some have evolved from knowledge gained with the use of targeted treatment of individuals with rare disorders of lipid and lipoprotein metabolism, while others were informed by results of Mendelian randomization studies. While data of LLT initiated in youth and continued for up to 20 years have been promising for familial hypercholesterolemia (FH), treatment of other lipid disorders, such as those with elevated TG have been less successful. In addition, while most currently available LLT options are well tolerated and improve lipid levels, not all youth are able to reach a desirable treatment target. Thus, additional therapeutic options are needed.
In this manuscript, we review currently available and novel therapeutic agents in development for the treatment of elevated cholesterol and TG. We have categorized these novel agents into those that predominantly reduce (1) low-density lipoprotein cholesterol (LDL-C); (2) TG, or (3) both. Included in our discussion of the later are agents that reduce Lp(a). Since clinical trials using currently available and evolving novel LLT are limited in youth, much of the evidence supporting the mechanisms of action, safety and efficacy are summarized from data derived from adults, and are so noted. Data for youth have been included whenever available. It should be noted that many, but not all LLT have been FDA approved for youth less than 18 years-of-age.
Therapeutic agents that predominantly lower LDL-C
Traditionally, statins have been the predominate LDL-C lowering agent utilized in clinical practice. Table 1 illustrates the LDL-C lowering medications and their therapeutic targets. Several emerging non-statin therapies that target LDL-C are discussed below.
Novel therapeutic agents for lowering low-density lipoprotein cholesterol.
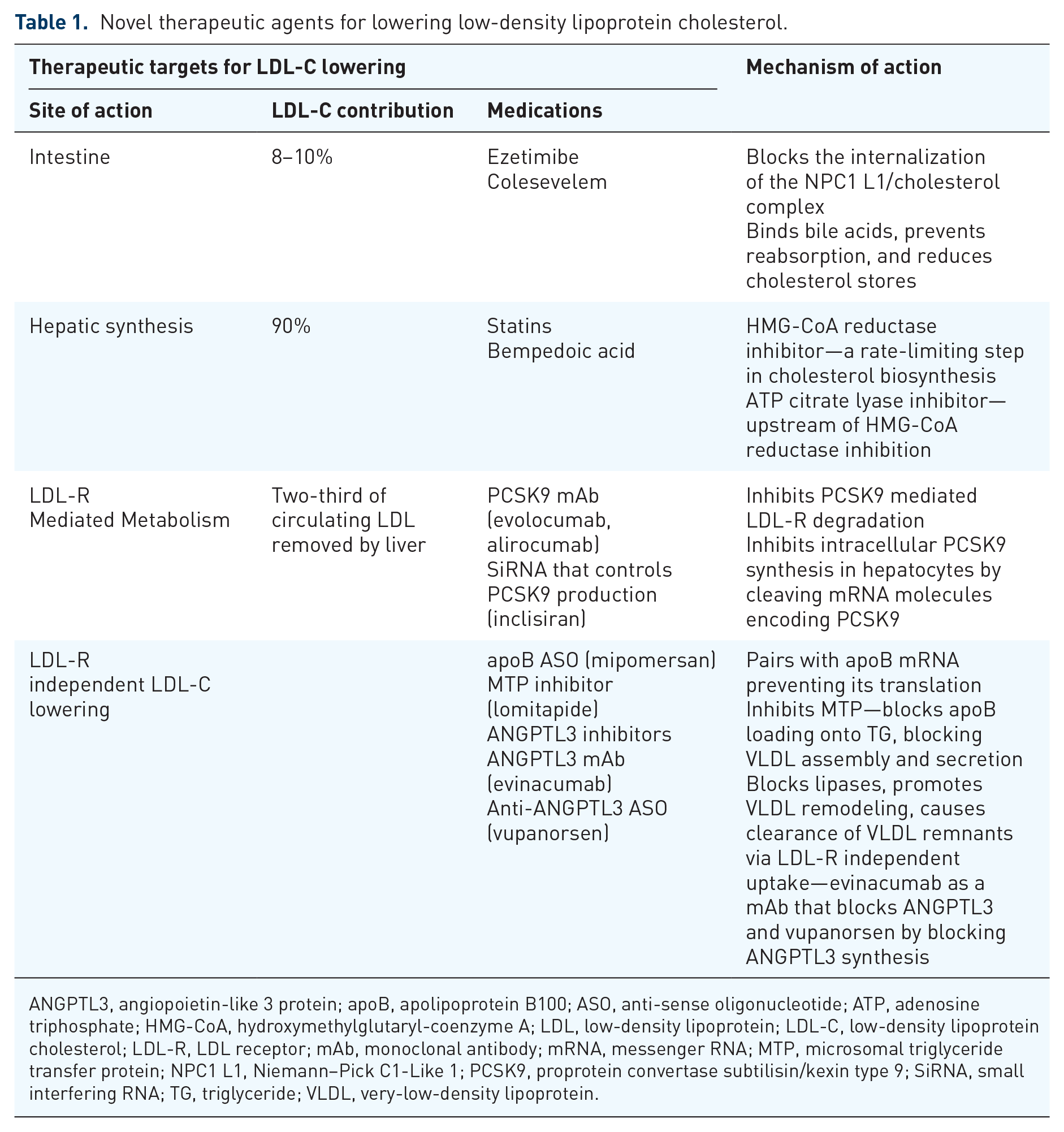
ANGPTL3, angiopoietin-like 3 protein; apoB, apolipoprotein B100; ASO, anti-sense oligonucleotide; ATP, adenosine triphosphate; HMG-CoA, hydroxymethylglutaryl-coenzyme A; LDL, low-density lipoprotein; LDL-C, low-density lipoprotein cholesterol; LDL-R, LDL receptor; mAb, monoclonal antibody; mRNA, messenger RNA; MTP, microsomal triglyceride transfer protein; NPC1 L1, Niemann–Pick C1-Like 1; PCSK9, proprotein convertase subtilisin/kexin type 9; SiRNA, small interfering RNA; TG, triglyceride; VLDL, very-low-density lipoprotein.
Therapeutic agents that interfere with dietary absorption of cholesterol
In most individuals, dietary cholesterol contributes about 8–10% of the circulating cholesterol. 2 Currently, there are two medications commonly used to reduce absorption of dietary cholesterol, bile acid sequestrants and ezetimibe. Even though ezetimibe is not a novel therapeutic agent, we have included it in this review to describe current evidence, given the increased use of this drug in youth.
Bile acid sequestrants (BAS)
BAS bind to bile acids, removing these from enterohepatic circulation, resulting in up-regulation of LDL-R and increased LDL-C clearance. Colesevelam is the only BAS approved for the treatment of pediatric patients with HeFH, approved for use as an adjunct to diet and exercise in FH, alone or in combination with statin therapy in October 2008. Colesevelam can reduce LDL-C by 7–15%. 3 The side effect profile includes bloating, constipation, and malabsorption of fat-soluble vitamins, all of which interferes with adherence. 4 Both colestipol and cholestyramine have been studied in pediatric patients but are not FDA approved and can bring LDL-C levels down by 10–20%.5–7
Ezetimibe
With a median follow-up of 6 years, the study showed that compared to placebo, ezetimibe resulted in incremental lowering of LDL-C levels and improved cardiovascular outcomes. 9 A 2018 meta-analysis illustrated moderate to high-quality evidence that ezetimibe had modest beneficial effects on lowering risk of ASCVD endpoints, primarily driven by a reduction in non-fatal myocardial infarction (MI) and non-fatal stroke, but little or no effect on fatal endpoints. 10
Ezetimibe is also used in youth with homozygous familial hypercholesterolemia (HoFH) as an adjunct to statins and apheresis, in sitosterolemia as an adjunct to dietary therapy, in HeFH and mixed hyperlipidemia as an adjunct to dietary changes and statins, or as monotherapy in the rare event statins are not tolerated. While safe and effective, because of its modest ability to lower LDL-C when used as a monotherapy, ezetimibe tends to be a preferred second-line, add-on therapy.
Therapeutic agents that reduce hepatic synthesis of cholesterol
About 90% of cholesterol synthesis occurs in the liver.
Statins
Traditionally, statins have been the primary LDL-C lowering agents, recommended as first-line treatment in all lipid guidelines. Statins act by inhibiting HMG-CoA reductase. In response to reduced intrahepatic cholesterol, LDL receptor (LDL-R) activity is upregulated, enhancing cellular uptake of circulating LDL. They have a favorable safety profile and excellent short- and long-term data with benefits outweighing the risks. The FDA has approved lovastatin (1987), pravastatin (1991), simvastatin (1991), fluvastatin (1993), atorvastatin (1996), rosuvastatin (2003), and pitavastatin (2009) 15 for use in children. The use of these medications is supported by multiple clinical trials,13,16–25 with significant LDL-C reduction, favorable effect on ASCVD risk and overall low adverse effects. Recently, long-term follow-up data have showed that initiation of statin therapy during childhood in patients with FH slowed the atherosclerotic progression and reduced the risk of cardiovascular disease in adulthood.26,27
Bempedoic acid
Hyperuricemia has been reported with the use of bempedoic acid, secondary to inhibition of renal tubular secretion of uric acid. 37 Adverse events included nasopharyngitis, myalgia, upper respiratory tract infections, dizziness, and diarrhea. 38 In the CLEAR HARMONY trial, adverse events occurred with similar frequency in those who received bempedoic acid and placebo. In addition to uric acid, bempedoic acid was associated with mild increases in blood urea nitrogen and creatinine and decreases in hemoglobin. 39 The risk of myotoxicity and rhabdomyolysis is considered low since the drug is not activated in skeletal muscles.
Therapeutic agents that act through LDL-R-mediated metabolism
This class of medications include PCSK9 inhibitors (PCSK9i)–PCSK9 monoclonal antibodies (PCSK9 mAb) and small RNA molecules that interfering with PCSK9 production (inclisiran).
PCSK9i is an excellent example of groundbreaking research that has been successfully translated from bench-to-bedside. In 2003, a gain-of-function mutation of PCSK9 was described in individuals with autosomal dominant hypercholesterolemia. 40 In 2005, PCSK9 sequencing of 128 individuals of African descent with low LDL-C and a history of reduced risk of ASCVD showed two loss-of-function mutations. 41 These findings, corroborated in subsequent studies, were supportive of PCSK9 gain-of-function increasing risk of ASCVD.42,43
PCSK9 monoclonal antibodies
Following the completion of multiple compelling clinical trials, PCSK9 mAb were approved in 2015, leading to a paradigm shift in primary and secondary prevention.
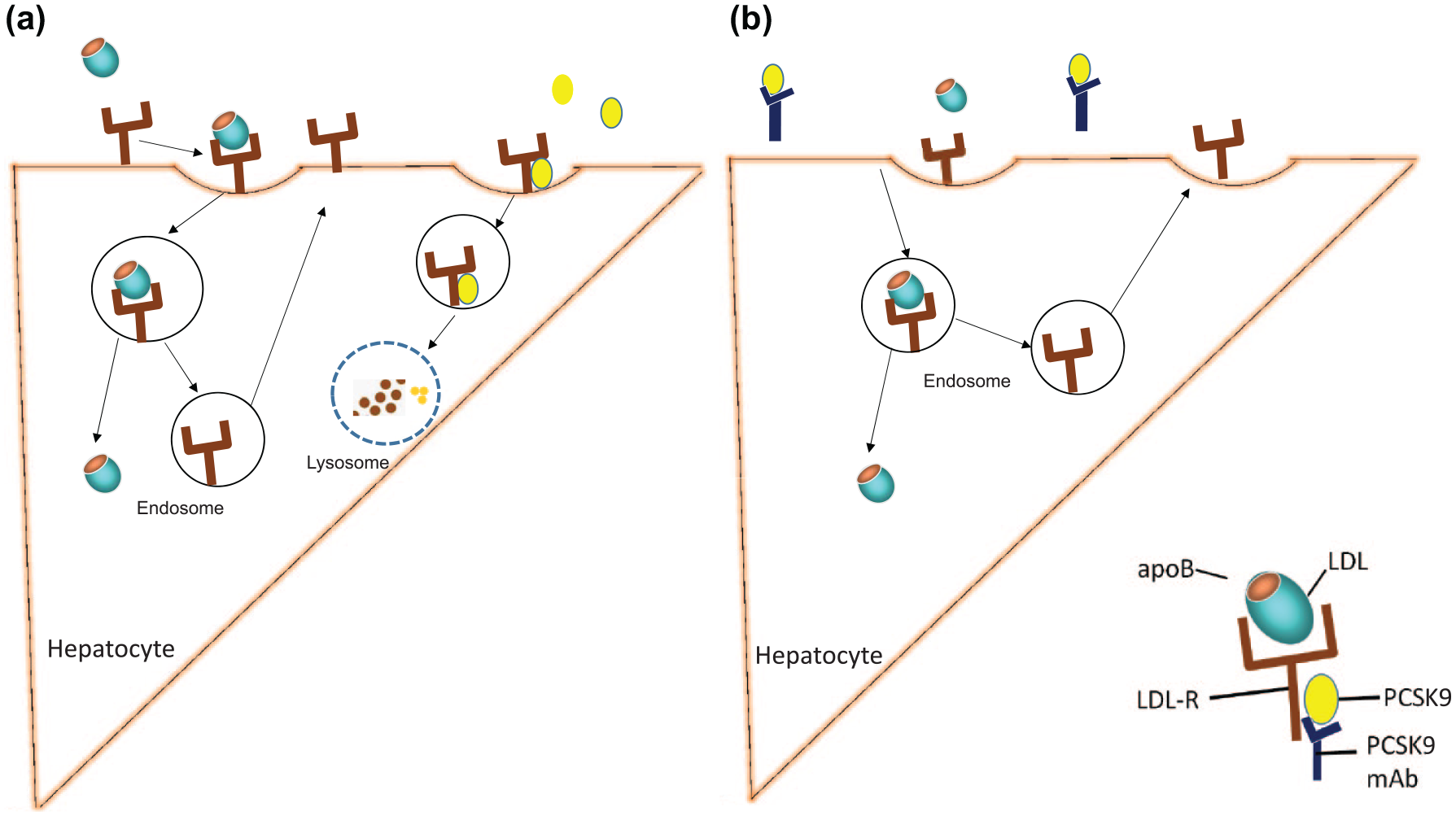
The mechanism of action of PCSK9 inhibitors. (a) Shows that LDL-C attaches to LDL-R to incorporate it into the hepatocyte. This can either be recycled when not attached to PCSK9. When the secreted PCSK9 attaches to the LDL-R, it eventually leads to lysosomal degradation of PCSK9. (b) Illustrates that in the presence of PCSK9 monoclonal antibody, the PCSK9 is inactivated, preventing the LDL-R from being degraded. This allows for the LDL-R to be recycled, prompting LDL-C uptake, and reducing LDL-C levels in the serum.
The ODYSSEY KIDS study, a phase-2 trial, assessed the efficacy, safety, and dose selection of alirocumab in HeFH. Youth 8–17 years of age were included who, despite the use of optimal LLT, had an LDL-C ⩾ 130 mg/dL. 64 Subjects were divided into four cohorts, utilizing multiple doses with a maximum dose of 300 mg every 2 weeks. At week 8, the cohort receiving the highest dose was found to have the greatest reduction in LDL-C. 64
More clinical trials are underway that are further supportive of the use of these medications in children with HoFH (NCT03510715) and HeFH (NCT03510884). Of these, NCT03510715 is an open-label trial with alirocumab causing significant LDL-C reductions and a favorable safety profile in children with HoFH.
Overall, PCSK9 mAb therapy has demonstrated an ability to substantially lower LDL-C with minimal adverse events. Even when used as monotherapy, PCSK9 mAb has demonstrated robust activity, and there is a monotonic relationship between achieved LDL-C and major cardiovascular outcomes. 67 Evolocumab and alirocumab are fourth-generation IgG monoclonal antibodies that are fully humanized, thus greatly reducing the potential for therapy-associated production of anti-drug or neutralizing antibodies. Current evidence supports their use in individuals with HoFH or HeFH who require additional lowering of LDL-C, despite maximum statin therapy. Response to treatment in those HoFH is variable, depending on residual LDL-R activity (LDL-R defective vs LDL-R null).
Both drugs have a favorable safety profile. There are no risks of rhabdomyolysis, myopathy, neutralizing antibodies, or incident risk of type-2 diabetes. Evolocumab administered 420 mg once monthly is approved for
Small interfering RNA (SiRNA) that control PCSK9 production (inclisiran)
In 2006, the Nobel Prize in Physiology or Medicine was jointly awarded to Andrew Fire and Craig Mello for their discovery of RNA interference – gene silencing by double-stranded RNA, a discovery eventually responsible for the development of inclisiran. 68 It is an incremental therapeutic advancement from PCSK9 mAb.

The mechanism of action of Inclisiran within the hepatocyte. Incisiran is an siRNA Blocking PCSK9 Transcription. Inclisiran is conjugated to GalNac for liver specific entry. The PCSK9 gene transcription results in PCSK9 mRNA. Inclisiran binds to PCSK9 mRNA, forms RISC complexes, and thereby promotes PCSK9 mRNA degradation and prevents synthesis of PCSK9.
Clinical data: The ORION trials studied the utility of inclisiran in clinical practice. ORION-9 included 482 adults with HeFH randomized to either 300 mg inclisiran or placebo at baseline, 3 months and then every 6 months for a total of four doses. The data showed an LDL-C reduction of 47.9% at day 510 and a time-averaged LDL-C reduction of 44.3% over the 18-month trial. 71 Using a similar design, 1,561 patients with ASCVD were assessed in ORION-10 and 1,617 patients in ORION-11. 72 Inclisiran lowered TC, non-HDL-C, apoB and TG as well as reducing Lp(a) by 18.6–25.6%, the latter a known independent, causative CVD risk factor. Though not yet approved for clinical use in the USA, based upon data from these trials, it is anticipated that the subcutaneous dose will likely be 300 mg twice a year. Twice a year dosing will likely enhance adherence to inclisiran. 73 The European Commission granted marketing authorization for inclisiran in Europe in December 2020. 74
The most frequently reported adverse events include a self-limiting rash, hyperpigmentation, cough, musculoskeletal and back pain, and acute nasopharyngitis.70,75,76 In one study, serious adverse events occurred in 11% of patients who received inclisiran compared to 8% of those who received placebo. Injection site reactions occurred in 5% of the subjects who received inclisiran. 76
LDL-R-independent reduction of LDL-C
The main therapeutic agents in this category include mipomersan, lomitapide, and evinacumab.
Mipomersen
The first antisense oligonucleotide (ASO) to be used in dyslipidemia management, mipomersen, was approved in 2013. It is a second-generation ASO directed toward the coding region of apoB RNA. 77
Use of mipomersen in youth has primarily been in those with HoFH with the goal of achieving additional LDL-C lowering in those unable to reach their LDL-C target. 83 In January 2013, the FDA approved mipomersen in the United States as an orphan drug for the management of HoFH, provided physicians registered in a Risk Evaluation and Mitigation Strategy (REMS). In Europe, the Committee for Medicinal Products for Human declined to approve mipomersen for clinical use, citing the potential risks outweighed the benefit of the drug. The product was withdrawn from the market in 2019 secondary to hepatotoxicity. 37
Microsomal triglyceride transfer protein (MTP) inhibitor: lomitapide
In 2012 lomitapide became the first MTP inhibitor approved by the FDA for HoFH as an adjunct to diet and other lipid-lowering therapies.

The mechanism of action of lomitapide. MTP is required for assembly and secretion of apoB-containing lipoproteins in the liver (apoB100) and intestine (apoB48). After production in the liver, VLDL is released into the plasma, where the TG content of the VLDL is hydrolyzed into free fatty acids, eventually forming LDL. By inhibiting MTP, lomitapide reduces the production and release of VLDL and LDL levels in plasma while at the same time reducing TG levels by reducing intestinal chylomicron formation.
Lomitapide was approved by the FDA in 2012. 79 The drug is given orally, once a day, initial as a 5 mg dose. If tolerated, the drug is titrated to a maximum dose of 60 mg a day. 37 The chief adverse events of lomitapide include diarrhea and hepatic steatosis—both likely linked to the intracellular increase in TG associated with impaired assembly and secretion of apoB-containing lipoproteins. Close monitoring of dietary fat intake is required with use of lomitapide.
Adverse events include gastrointestinal symptoms, liver dysfunction, and hepatic steatosis.85,91,92 Lomitapide impair the absorption of fat-soluble vitamins as well. 93 Because a high-fat meal can potentiate GI side effects, dietary fat should be limited in individual receiving this drug. In pediatric case series, GI side effects included nausea, vomiting, and reduced appetite. Two youth had thickened cardiac valves. 94
Lomitapide is approved for treatment of adults with HoFH through REMS. Used with caution, the drug has the potential of mitigating ASCVD risk, especially in those who do not meet their LDL-C targets with statins, ezetimibe, resins, and PCSK9i. In addition, it may improve quality of life by reducing the need for or frequency of lipid apheresis, and provide an alternative to those who do not have access to or decline apheresis. At this time, lomitapide it is not approved for pediatric use.
Inhibition of angiopoietin like 3 (ANGPTL3)
The role of ANGPTL3 in lipoprotein metabolism was initially defined in obese KK mice. This mouse model exhibits a mutant phenotype characterized by abnormally high levels of plasma insulin (hyperinsulinemia), glucose (hyperglycemia), and lipids (hyperlipidemia), although one strain (KK/San) was found to have abnormally low plasma lipid levels (hypolipidemia). 95 When the region including ANGPTL3 loss-of-function variant was introduced to atherogenic apoE-knock out mice, the prevalence of baseline atherosclerotic lesions declined. 96 In human studies, genetic variants in ANGPTL3 showed a strong association between plasma levels of ANGPTL3 and TG. 97 Addition publications support the association of ANGPTL3 loss-of-function and low cholesterol levels.98,99 These findings support development of drugs targeting ANGPTL3 inhibition as a therapeutic strategy.
Monoclonal antibodies, which target ANGPTL3, inhibit both lipases and promotes VLDL remodeling, causing preferential removal of TRLPs. As a consequence, reduced levels of VLDL limit LDL production, and lower circulating LDL-C.100,101
Evinacumab: This fully humanized ANGPTL3-blocking monoclonal antibody works by binding and reducing the activity of ANGPTL3 (Figure 4).

Mechanism of action of ANGPTL3 inhibition. ANGPTL3 is synthesized and secreted by hepatocytes. It inhibits LPL and endothelial lipase and thereby regulates the concentrations of apoB-containing lipoprotein turnover – namely VLDL, IDL and LDL. Since it causes clearance of triglyceride-rich lipoproteins upstream of LDL production, it can cause an LDL-R independent reduction in apoB-containing lipoproteins.
Reported adverse events with use of evinacumab included headache and upper respiratory infections. Urinary tract infection, arthralgia, and myalgia also occurred. Elevated liver enzymes have reported in some individuals treated with evinacumab.103,107,108
Evinacumab has been shown to be effective as an adjunctive therapy in HoFH and HeFH patients receiving maximally tolerated doses of statin; and approved for youth ⩾12 years with HoFH in February 2011. 109 Efficacy is dependent upon residual LDLR activity (defective > null). However, some lipid-lowering effect may be seen in those with complete absence of the LDL-R (null-null variants), a group relatively unresponsive to PCSK9 inhibition. For youth 12 years of age and older, evinacumab is administered intravenously at a dose of 15 mg/kg/dose every 4 weeks.
Therapeutic agents to lower TG
Historically, effective therapies that target TG lowering have been challenging. Fibrates, though not approved for use in youth less than 18 years-of-age, have been extensively used for treatment of hypertriglyceridemia (HTG) in adults. There is no evidence in youth that omega-3-fatty acids (O3FAs) have been effective in the treatment of mild-to-moderate HTG. Several promising investigational therapeutic agents, which act through the lipoprotein lipase (LPL) complex, are currently in development: (1) antisense oligonucleotides (Volanesorsen® and AKCEA-APO-CIII-LRx) which reduce apoC3 and (2) Monoclonal antibodies (evinacumab) and GalNac conjugated ASO which targets ANGPTL3 mRNA in the liver (Vupanorsen/IONIS-ANGPTL3-LRX). Lomitapide, which inhibits MTP and is currently approved for treatment of HoFH as an adjunct to diet and other lipid-lowering therapies, can also lower TG. While O3FAs are generally of limited benefit in youth, iIcosapent ethyl (VASCEPA®), an ethyl ester of eicosapentaenoic acid (EPA), has been used by some for management of HTG. Safety and effectiveness of icosapent ethyl in youth have not been established. Table 2 illustrates novel medications lowering TG levels.
Medications lowering triglyceride levels.

ANGPTL3, angiopoietin-like 3 protein; ApoC3, apolipoprotein C3; ASO, antisense oligonucleotide; DGA, diacylglycerol acyltransferase; DHA, docosahexanoic acid; EPA, eicosapentanoic acid; GalNac, triantennary N-acetyl galactosamine; LDL, low-density lipoprotein cholesterol; LPL, lipoprotein lipase; mAb, monoconal antibody; mRNA, messenger RNA; MTP, microsomal triglyceride transfer protein; TG, triglyceride; VLDL, very-low-density lipoprotein.
None of these medications shown are FDA approved for children <18 years-of-age.
Inhibition of apoC3
Antisense oligonucleotide (ASO) inhibiting apoC3 (Volanesorsen)
Mechanism: Volanesorsen is a second-generation antisense oligonucleotide. It selectively binds the apoC3 messenger ribonucleic acid (mRNA), preventing translation and allowing mRNA degradation, thereby promoting TG clearance and the lowering plasma TG levels through LPL-independent pathways (Figure 5). The results of early clinical trial data were quite promising.111–113

The mechanism of action of an ASO targeting apoC3. Volanesorsen is an ASO that binds to apoC3 mRNA, leading to its degradation, and preventing translation of apoC3 protein. This allows ribonuclease H1-mediated mRNA degradation, thereby promoting TG clearance through LPL-independent mechanisms.
ApoC3 is an apolipoprotein synthesized in the liver, and a component of atherogenic TG rich lipoproteins (TRLPs) such as VLDL, chylomicrons and remnant lipoproteins. ApoC3 affects TG levels by inhibiting of LPL dependent and independent pathways. It may also directly regulate enterocyte metabolism of TGs. It reduces receptor-mediated clearance of TRLPs by the liver, inhibits hepatic lipase, and increases intrahepatic assembly and secretion of TG rich VLDL. In humans, loss-of-function mutations of the APOC3 gene results in lower levels of plasma TG and LDL-C, increased HDL-C levels and reduced ASCVD risk. 114
Despite the impressive ability of volanesorsen to lower TGs, the occurrence of thrombocytopenia and injection-site reactions have raised concerned about its use. Up to 30% of subjects discontinued use of the drug in phase-3 studies due to unpredictable thrombocytopenia, which required the drug administration schedule to be changed, interrupted or discontinued. The European Medicines Agency gave conditional marketing authorization of the drug for patients with confirmed FCS provided that extra data were collected in a registry study. 118 In 2018, the US FDA refused to approve volanesorsen for the treatment of FCS based on safety issues of thrombocytopenia and risks of bleeding. No pediatric data are available.
Antisense oligonucleotide inhibiting apoC3 with GalNac adult (AKCEA-APOC3-LRx)
Mechanism: AKCEA-APO-CIII-LRx is a third-generation ligand-conjugated antisense (LICA) drug with an N-acetylgalactosamine-containing additive (GalNac). The drug targets APOC3. It is anticipated to increase first pass clearance, and lower the risk of thrombocytopenia due to higher tissue selectivity. Because of its longer half-life, lower dosing preparations may be required for effective TG lowering, a potential advantage over Volanesorsen. 119
Inhibition of angiopoietin like 3 (ANGPTL3)
This class of therapeutic agents is capable of lowering both LDL-C and TG.
Evinacumab
This fully humanized ANGPTL3-blocking monoclonal antibody can reduce both LDL-C and TG (please refer to the section on LDL-C lowering). In 2002, ANGPTL3 knock out mice were shown to have abnormally low lipid levels. By 2010, ANGPTL3 loss-of-function carriers were shown to have extremely low levels of LDL-C, VLDL-C, HDL-C, and TGs. These findings lend support to drug development targeting inhibition of ANGPTL3 as a therapeutic strategy (See 1.a.3. Inhibition of Angiopoietin Like 3 (ANGPTL3) for additional details.)
Antisense oligonucleotide (ASO) targeting ANGPTL3 mRNA in the liver
With TRLPs increasing recognized as being associated with increased ASCVD risk, 121 potent TG, and non-HDL-C lowering therapy such as vupanorsen could provide additional cardiovascular benefits. No pediatric data or ongoing trials in youth are available at the time.
Lomitapide
Lomitapide can reduce levels of both LDL-C and TG (See d.2. Microsomal TG transfer protein (MTP) inhibitor: lomitapide for more details)
Icosapent ethyl: Vascepa®
In a Commentary by the ESPGHAN Committee on Nutrition, addressing the various health claims made to support the use of O3FA in children, insufficient evidence was found toward supplementation of long-chain polyunsaturated fatty acids on cognitive function, attention-deficit hyperactivity disorder (ADHD), visual function in phenylketonuria, major clinical outcomes in cystic fibrosis or in asthma. 128
Vascepa has been approved as an adjunct to diet to reduce TG levels in adults with severe HTG (⩾500 mg/dL). It is not approved for pediatric use at this time.
Novel agents that target elevated Lp(a)
The National Lipid Association recommended Lp(a) be selectively measured in youth <20 years of age: (1) in clinically suspected or genetically confirmed FH; (2) if there is a significant family history of ASCVD; (3) a history of ischemic stroke of unknown etiology; or (4) if there is a family history of a parent or sibling with elevated Lp(a).129,130 At present, screening for Lp(a) is limited, which may be due a lack of uniformity in Lp(a) screening guidelines among various professional organizations, an incomplete understanding of age-based normative values and treatment goals, and lack of commercially available targeted Lp(a) therapeutic agents. However, pending the result of ongoing clinical trials, this may change.
Of the agents mentioned above, PCSK9i, inclisiran, mipomersan, and APO(a)-LRx antisense therapy can all potentially reduce Lp(a) levels. 131 On an average, the PCSK9 inhibitors can reduce Lp(a) by 20–25%132–134 and inclisran by 17%. 72 The ASOs targeting hepatic LPA messenger RNA by conjugation with GalNAc3, named APO(a)-LRx specifically reduced plasma levels of Lp(a) by 66–92% in a dose-dependent in participants with established cardiovascular disease and elevated Lp(a) levels. 135
While statins significantly reduce LDL-C levels, in some, they may modestly increase Lp(a). The overall cardiovascular impacts of these effects are incompletely understood. Although niacin and estrogen can both reduce Lp(a) levels, neither is recommended for this indication. 131
Conclusion
Over the past two decades, improvements in our knowledge of genetic disorders and advances in biomedical technology have prompted the discovery of novel targets for management of acquired and genetic lipid and lipoprotein disorders. This collaboration of basic science, biotechnology, and robust clinical research has facilitated the development of an increasing number of new therapeutic options to aid in ASCVD prevention. While several safe and effective therapeutic options are currently available for use in youth, more are needed, especially in those with TG elevations. Development of novel therapeutic agents and their subsequent approval for clinical use should include youth less than 18 years of age. While data from adult clinical trials are informative, more studies are needed in the pediatric population. If proven safe and effective, early intervention with newer therapeutic alternatives and additives have the potential of significantly reducing CVD risk and prevention of future ASCVD-related events in this unique population.
Footnotes
Author contributions
B.S. drafted the initial manuscript, and reviewed and revised the manuscript. A.P.A. conceptualized and designed the manuscript, drafted some sections of the manuscript, and critically reviewed the manuscript for important intellectual content. C.F. and D.P.W critically reviewed the manuscript for important intellectual content and revised the manuscript.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
