Abstract
Stress fractures have traditionally been classified into three categories: fatigue fractures due to overuse of bone with normal elastic resistance; insufficiency fractures due to everyday physiological stress on fragile bone with poor elastic resistance; and pathologic fractures due to bone weakness involving tumors. The concept of atypical fractures has emerged and is considered a type of stress fracture. However, there has been some inconsistency in interpretation when using the traditional classification of stress fractures, and atypical femoral fractures (AFFs) can potentially be classified into subtypes: “typical” AFFs involving bone turnover suppression due to specific drugs (e.g. bisphosphonates) and fragility fractures of the bowed femoral shaft. In this article, the classification of stress fractures is redefined with the addition of atypical fractures as a fourth category, in which biological activity for fracture healing is absent, to promote consistent understanding and interpretation of clinical conditions involving stress fractures.
Keywords
Introduction
Stress fractures have traditionally been classified into three types: fatigue fractures due to overuse; insufficiency fractures due to bone fragility; and pathologic fractures due to bone weakness involving tumors. 1 Fatigue fractures occur in normal bone that has been overused (e.g. in athletes and military personnel), whereas insufficiency fractures develop in fragile bone that is repeatedly subjected to low levels of stress during everyday physical activity.
In recent years, there has been increasing awareness of the concept of atypical fractures.2–4 While it is widely known that suppression of bone turnover by specific drugs (e.g. bisphosphonates) increases the risk of atypical femoral fracture (AFF), 5 this type of fracture occurs where there is focal thickening of the lateral cortex and is considered to be caused by mechanical stress.6–10 Therefore, atypical fracture can be regarded as a type of stress fracture. However, it has been suggested that AFF can potentially be classified further into bone turnover suppression-related “typical” AFF, which is more common in the subtrochanteric region, and mid-shaft stress fracture of the bowed femoral shaft (SBF).11,12 Although the pathology of SBF fits with the concept of an insufficiency fracture, bone in patients with AFF is not necessarily fragile or weak.12–14 In this article, we propose addition of a fourth category in the classification of stress fractures to cover this particular type of fracture.
Fatigue fractures
Fatigue fractures due to overuse were first described by a Prussian military surgeon in 1855. 15 The fractures occurred in soldiers who were engaged in long marches and had persistent pain in the feet in the absence of trauma. With the advent of radiography, this type of fatigue fracture was identified as a metatarsal shaft fracture called “march fracture.” Overuse bone injuries in young people (e.g. athletes and military personnel) who engage in vigorous physical activity and are subjected to high levels of stress are now widely recognized.16–18 A fatigue fracture that develops in any bone with normal elastic resistance and adequate strength is considered a stress fracture. Although the treatment of fatigue fractures is based on removal of excessive and repeated stress, top athletes require individualized treatment that often includes internal fixation.
Insufficiency fractures
Insufficiency fractures develop in fragile bone with less than normal elastic resistance to mechanical force and may be caused by normal or physiological levels of stress. 1 Insufficiency fracture is an important category of stress fracture that distinguishes stress fractures in abnormal bone except those that are tumor related. Certain diseases (e.g. osteogenesis imperfecta, Paget’s disease of bone, osteomalacia, rickets, hypophosphatasia, osteoporosis, rheumatoid arthritis, hyperthyroidism, osteomyelitis, and poliomyelitis) can reduce the elastic resistance of bone and cause insufficiency fractures.1,19 Patients who develop this type of fracture tend to be older than those who develop fatigue fractures because of the increasing incidence of underlying disease with advancing age. 1 Treatment of this type of stress fracture ideally includes not only the fracture itself but also the underlying disease in order to improve the elastic resistance and strength of abnormal bone.
Pathologic fractures
A pathologic fracture is a stress fracture that occurs in bone at the site of a tumor. 1 Tumors associated with this type of stress fracture include benign bone cysts, malignant bone tumors, and bone metastases from various types of cancer.
Atypical fractures
AFFs are atypical fractures that affect the femoral shaft with or without minor trauma and are reported to be due to suppression of bone turnover by specific drugs (e.g. bisphosphonates, denosumab, glucocorticoids, or proton pump inhibitors).2–6,20–22 Figure 1 shows a representative X-ray image of subtrochanteric AFF in a 66-year-old woman who had been taking alendronate for 14 years. Healing of these fractures tends to be delayed because of widespread suppression of bone turnover (Figure 2).4–6,12,23–27 Other factors potentially involved in the pathogenesis of AFF include ethnicity,
4
body mass index,
20
loading stress in daily life,7–10 femoral morphology,6–8,11,12,14,28,29 and bone density, quality, and strength.12–14 Mild mechanical stress during daily activities is undoubtedly involved in the onset of AFF.4,6–10 Mechanical studies using finite-element methods by our research group and Haider

X-ray image for a 66-year-old woman with a subtrochanteric atypical femoral fracture. She complained of persistent pain in the left thigh and had been taking alendronate for 14 years. The left femur shows significant focal thickening of the lateral cortex with an apparent incomplete fracture line in the subtrochanteric region (arrowhead). The contralateral (right) femur shows a diffusely thickened lateral cortex but no fracture line in the subtrochanteric region (arrows).

Representative histology for an incomplete subtrochanteric atypical femoral fracture (Figure 1) shows suppression of bone remodeling (a, b, hematoxylin-eosin staining; c, safranin O staining). (a) Lower magnification of the bone sample harvested from the cortex on one side of the fracture line during prophylactic internal fixation (20×). The upper side is the surface of the focally thickened lateral cortex and the left side is the fractured surface. (b, c) Higher magnification of the region indicated by a square in Figure 2a (100×). The fractured surface is covered with fragile degenerated bone with no osteoclasts, osteoblasts, or cartilage cells. Endochondral ossification (arrowheads) is found in only a small part of the area adjacent to the degenerated tissue.
Classification of AFF subtypes
To our knowledge, the first description of a stress fracture of the femoral shaft associated with bowing deformity dates back to 1975. 30 After encountering an elderly woman with a similar stress fracture of the femur in 1998, 11 well before the first report of an association between suppression of bone turnover and AFF, we focused on this type of stress fracture in elderly Japanese women. This type of fracture was recently defined as an SBF.7,8,11–13 Figure 3 shows a representative X-ray image of SBF in a 73-year-old woman with no history of exposure to specific drugs (e.g. bisphosphonates, denosumab, glucocorticoids, or proton pump inhibitors). Although all patients with SBF fulfill the case definition of AFF put forward by the American Society for Bone and Mineral Research Task Force in 2014, 6 a number of SBF cases did not have exposure to drugs such as bisphosphonates, and the fracture basically developed in the mid-shaft at the apex of curvature.11,12 A multifactorial investigation of Japanese patients with AFF showed that mid-shaft AFF tends to develop in elderly women with bowing deformity of the femoral shaft and low bone strength8,13 but normal biological activity without suppression of bone turnover. 12

X-ray images for a 73-year-old woman with a mid-shaft stress fracture of the bowed femoral shaft. She had no history of exposure to specific drugs (e.g. bisphosphonates, denosumab, glucocorticoids, or proton pump inhibitors). (a) A displaced fracture of the left femoral shaft (AO/OTA classification, 32A3). The fracture line is transverse with a small medial spike. (b) The contralateral right femur shows bowing deformity in the lateral direction. A slight transverse fracture line (arrowhead) is identified in one of the lateral cortices with slight focal thickening (arrows).
In contrast, biological activity tends to be suppressed due to long-term use of particular drugs in “typical” subtrochanteric AFF, 12 and bone strength in patients with this type of fracture is higher than the average for those of the same age and sex. 13 Based on these site-related characteristics, we have proposed a theoretical classification of AFF subtypes, called Oh’s subtype theory, which can be applied to most patients with AFF, especially in East Asian countries. 12 Although some cases may not be classifiable, most cases of AFF in the East Asian population can be classified as SBF (insufficiency fracture with adequate biological activity that frequently develops in the mid-shaft due to bone fragility and bowing deformity) or “typical” AFF (intractable fracture with inadequate biological activity that often develops in the proximal diaphysis due to long-term pharmacologic suppression of bone turnover).
New classification of stress fractures
From the perspective of stress fractures, it should be noted that not only SBF (AFF associated with bowing of the femur and bone fragility) but also some femoral diaphysis fractures in fragile bone due to certain diseases (e.g. osteomalacia, rickets, and hypophosphatasia) would essentially be classified as insufficiency fractures, which generally have sufficient biological activity for fracture healing despite fulfilling the current diagnostic criteria for AFF.6,13 Figure 4 shows representative imaging findings for a 70-year-old woman with an incomplete subtrochanteric AFF who had vitamin D deficiency osteomalacia but adequate biological activity for bone healing. However, there is no bone fragility and no biological activity in cases of “typical” AFF, which frequently develops in the subtrochanteric region.12–14 Therefore, given the difficulties regarding the establishment of diagnostic criteria and treatment guidelines for AFF with different pathologies, we have suggested a classification of AFF subtypes, 12 and we concurrently redefine the traditional classification of stress fractures. “Atypical fracture” would be included as a fourth subtype of stress fracture in this new classification (Table 1).
New classification for stress fractures and characteristics in each category.

Not only stress fractures of the bowed femur associated with bone fragility in the elderly7,8,11–13 but also some femoral diaphysis fractures in fragile bone due to certain diseases (e.g. osteomalacia: Figure 4) would essentially be classified as an insufficiency fracture, which generally has sufficient biological activity for fracture healing, despite fulfilling the current diagnostic criteria 6 for atypical femoral fracture.
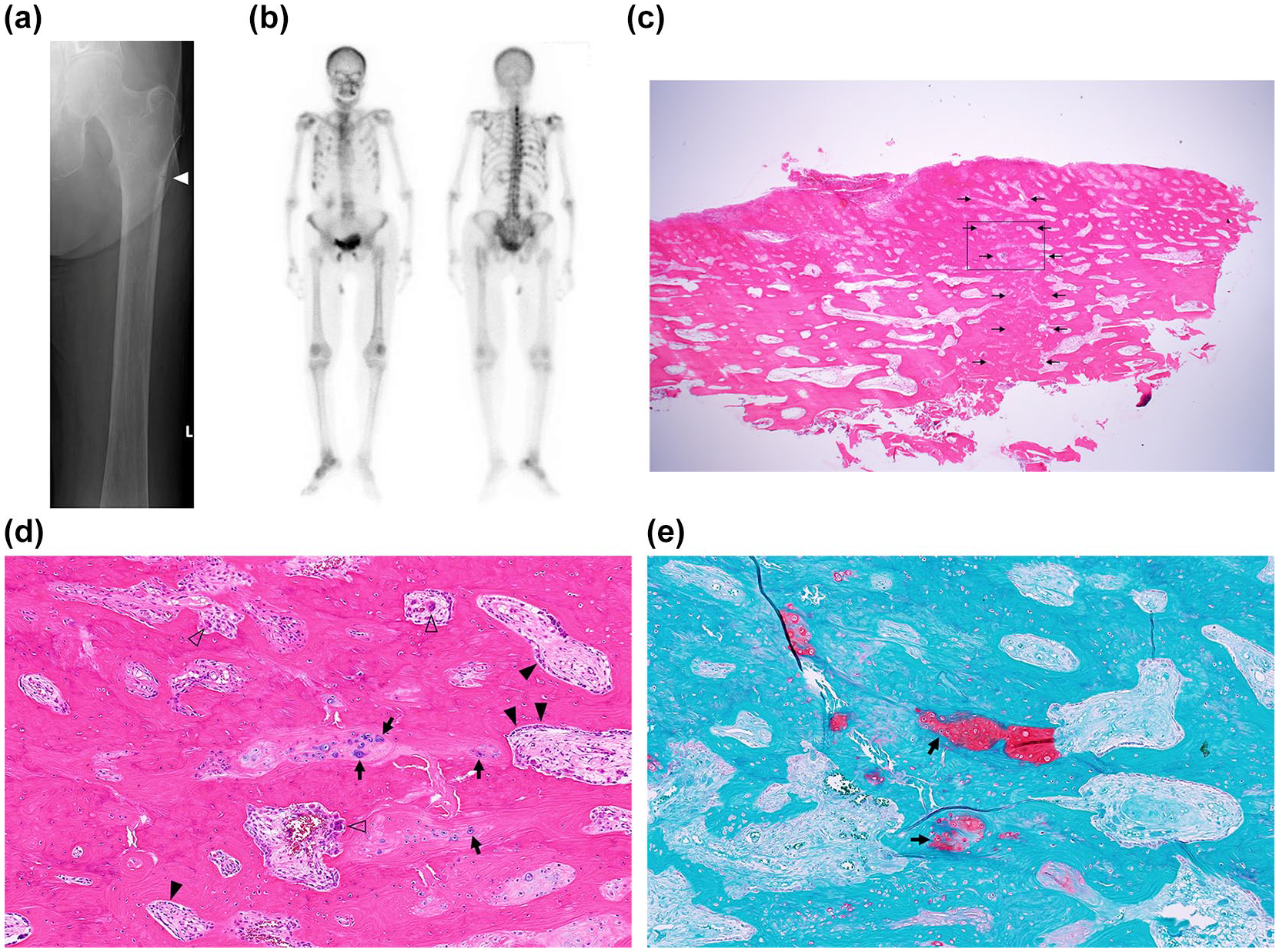
An exceptional case of incomplete subtrochanteric atypical femoral fracture in a 70-year old woman. She complained of severe pain in the left thigh. Although she had been taking a proton pump inhibitor for 6 years, she had never been exposed to bisphosphonates. Dual-energy X-ray absorptiometry of the left femoral neck revealed severely decreased bone mineral density of 0.212 g/cm2, and blood tests (bone-specific alkaline phosphatase, 156 μg/L; tartrate-resistant acid phosphatase-5b, 1710 mU/dL; phosphorus, 2.2 mg/dL; calcium, 7.4 mg/dL; 25-hydroxyvitamin D, 4.3 ng/mL; fibroblast growth factor 23, 5.9 pg/mL) confirmed the diagnosis of vitamin D deficiency osteomalacia. (a) X-ray image of the left femur shows slight focal thickening of the lateral cortex with an incomplete fracture line in the subtrochanteric region (arrowhead). (b) Bone scintigraphy shows uptake in multiple ribs and vertebral bodies. (c) Lower magnification of the cortical bone sample including the fracture line which was harvested during prophylactic internal fixation (hematoxylin-eosin stain, 12.5×). The surface of the focally thickened lateral cortex is on the upper side and the incomplete fracture site, which is evident on the X-ray image, is located in the center of the sample (small arrows). (d, e) Higher magnification of the region indicated by a square in Figure 4c (100×; d, hematoxylin-eosin stain; e, safranin O stain). Active bone remodeling is demonstrated by abundant infiltration of both osteoclasts (white arrowheads) and osteoblasts (black arrowheads). Interspersed chondrocytes (black arrows) with deposition of cartilage matrix, which is stained red by safranin O, suggest endochondral ossification.
Recent studies have raised the possibility of vitamin D deficiency in patients with stress fractures,31–33 which may also include those with fatigue or insufficiency fractures. Low vitamin D levels would be consistent with underlying bone fragility in patients with insufficiency fractures including SBF (insufficiency fracture in the femoral diaphysis associated with bowing deformity)12,13 but possibly not in those with “typical” AFFs.
Some reports have described AFFs associated with inherited skeletal dysplasia,34–37 and there might be quite a few cases of AFFs with various latent inherited diseases (e.g. hypophosphatasia, X-linked hypophosphatemia, pycnodysostosis, and osteopetrosis). If bone fragility associated with inherited skeletal dysplasia triggers the onset of AFF, it could fall into the category of insufficiency fractures. However, latent diseases may enhance bone turnover suppression by specific drugs (e.g. bisphosphonates) and cause “typical” AFFs, and this remains a subject for future research.
Other fractures with atypical fracture characteristics
There have been several reports of periprosthetic or forearm fractures after prolonged treatment with bone resorption inhibitors that exhibited characteristics of stress/atypical fractures.38–41 More research is needed to define the diagnostic criteria and treatment for these types of fracture.
Conclusion
Stress fractures can currently be divided into four categories: fatigue fractures, insufficiency fractures, atypical fractures, and pathologic fractures. It should be noted that SBF (AFF associated with bowing of the femur and bone fragility) and some femoral diaphysis fractures in fragile bone due to certain diseases (e.g. osteomalacia) would essentially be classified as insufficiency fractures despite fulfilling the current diagnostic criteria for AFF.
Footnotes
Author contributions
YO: conceptualization; data curation; funding acquisition; investigation; methodology; project administration; validation; visualization; writing—original draft; writing—review and editing. KY: conceptualization; data curation; investigation; validation; visualization; writing—original draft; writing—review and editing. TY: conceptualization; methodology; writing—review and editing. MK: conceptualization; resources; supervision; writing—review and editing. AO: conceptualization; methodology; project administration; resources; supervision; and writing—review and editing.
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The department to which the first author (YO) belongs received funding for operating costs from Saku Central Hospital of the Nagano Prefectural Federation of Agricultural Cooperatives for Health and Welfare, Suwa Central Hospital, Doujin Hospital, Medtronic Sofamor Danek Co, Ltd, Stryker Japan K.K., HOYA Technosurgical Co, Ltd, and NuVasive Japan K.K. The other authors declare that they have no conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by JOA-Subsidized Science Project Research (grant no. 2021-2) and JSPS KAKENHI (grant no. JP19K18451). The sponsors had no involvement in the drafting of the manuscript or the decision to submit the manuscript for publication.
Ethical approval
This work was approved by the Tokyo Medical and Dental University Faculty of Medicine IRB (approval ID: M2000-2229). All procedures involving human participants were performed in accordance with the ethical standards of the relevant institutional and/or national research committees and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
