Abstract
Background:
Type 2 diabetes (T2D) is associated with an increased risk of heart failure (HF) and cardiovascular mortality. A large-scale meta-analysis on HF found that diabetes was more frequent in women than men, and diabetes appeared to have attenuated the otherwise protective effect of female sex on progression of cardiomyopathy. The exact underlying mechanisms for this remain unclear. Here, we aimed to determine the effect of sex on the phenotypic expression of diabetic heart disease in patients with T2D.
Methods:
A total of 62 male [mean age 44 ± 8 years, body mass index (BMI) 33 ± 5 kg/m2, mean HBA1c of 7.8 ± 1.8%] and 67 female (44 ± 10 years, BMI 35 ± 6 kg/m2, HBA1c 7.6 ± 1.2%) T2D patients on oral glucose-lowering treatment, and 16 male (48 ± 17 years, BMI 25 ± 3 kg/m2) and 14 female (50 ± 10 years, BMI 25 ± 4 kg/m2) controls were recruited. Left ventricular (LV) volumes, mass, function and deformation, and left atrial (LA) volumes and function were assessed using cardiac magnetic resonance imaging (CMR).
Results:
Participants in all groups were of similar age, and there were no significant differences in blood pressure (BP), diabetes duration or metabolic profile between the two diabetes groups. Concentric remodeling was present in both sexes (p < 0.0001), with greater degree of concentric hypertrophy in males (12%, p = 0.0015). Biplane LA ejection fraction (LAEF) (p = 0.038), peak systolic circumferential strain (p < 0.0001) and diastolic strain rates (p = 0.001) were significantly reduced in men compared with women with T2D. There were no significant differences in biplane LAEF, peak systolic circumferential strain and diastolic strain rates in women with T2D compared with female controls. Whereas in women with T2D, glycaemic control was linked to LV contractile function, there was no such relationship in men with T2D.
Conclusion:
Male sex adversely affects the phenotypic expression of diabetic heart disease. The striking differences in the cardiac phenotype between male and female patients with T2D promote awareness of gender-specific risk factors in search of treatment and prevention of diabetes-associated HF.
Condensed Abstract:
We aimed to determine the effect of sex on the phenotypic expression of diabetic heart disease in patients with T2D. While our findings support the notion that in T2D, male sex adversely affects the phenotypic expression of diabetic heart disease, this is in apparent conflict with the previous large-scale study showing diabetes attenuates the otherwise protective effect of female sex on progression of cardiomyopathy. Further longitudinal studies looking at gender differences in clinical outcomes in T2D patients are needed. These sex-related differences promote awareness of sex-specific risk factors in search of treatment and prevention of diabetes-associated HF.
Keywords
Introduction
Type 2 diabetes (T2D) is associated with an increased risk of both heart failure (HF) and cardiovascular mortality.1,2 Diabetes has been consistently found to be a stronger risk factor for heart disease in women as compared with men. 2 Whereas in the last several decades, mortality from cardiovascular disease (CVD) has shown a marked decline, 3 this decline in all-cause mortality has been demonstrated only among men with diabetes, and not among women. HF is a significant cause of death in patients with diabetes. 4 A large-scale meta-analysis on HF found that the prevalence of T2D was higher in women than men, including patients with both reduced and preserved ejection fractions, 5 suggesting that diabetes attenuates the otherwise protective effect of female sex on the progression of cardiomyopathy, 5 although the exact underlying mechanisms remain unclear. This highlights the need to better understand the gender differences in heart disease related to diabetes.
Patients with diabetes have been phenotyped extensively, with a nuanced description of disease burden using non-invasive imaging technologies. 2 Several studies showed strong predictors of adverse cardiovascular events,6,7 such as adverse left ventricular (LV) structural remodeling, and subtle LV contractile dysfunction even in the absence of abnormal LV ejection fraction (EF) in patients with T2D.8–13 Advanced imaging techniques now also allow detailed phenotyping of atrial structure and function. Recently, left atrial (LA) function has emerged as an important prognostic factor in patients with dilated cardiomyopathy, 14 and in patients with HF with preserved EF. 15 Impaired LA function has been demonstrated in patients with T2D in a few recent studies.16,17
Cardiac magnetic resonance imaging (CMR) has now become the gold standard method for assessment of cardiac volumes and function. 18 As a result, using CMR and plasma markers of metabolic control, we aimed to assess sex differences in atrial and LV remodeling and function in patients with T2D.
Methods
The collated studies were approved by the National Research Ethics Committees (REC Ref 13/SW/0257, REC reference 15/WM/0222, and 13/WM/0311) and informed written consent was obtained from each participant. Patients were recruited prospectively from general practice surgeries in Oxfordshire and Leicestershire, United Kingdom. General practitioners and diabetes specialist nurses were provided with detailed information about the aims and objectives of the study, and the eligibility criteria. Potentially suitable participants who were willing to consider participating in the study were then referred to study investigators for a screening visit to confirm their eligibility. A total of 62 male, 67 female patients with T2D, and 16 male and 14 female non-diabetic controls were recruited.
Exclusion criteria
Subjects were excluded if they had a previous diagnosis of CVD, contraindications to magnetic resonance (MR) imaging, ischemic changes on 12-lead electrocardiogram (ECG), severe renal impairment (estimated glomerular filtration rate below 30 ml/min/1.73 m 2 ), or if they were on treatment with insulin.
Controls had no history of heart disease, diabetes mellitus (fasting glucose level ⩾6.7 mmol) or hypertension, and were not taking any medications.
Anthropometric measurements
Height and weight were recorded and body mass index (BMI) calculated. Brachial blood pressure (BP) was recorded as an average of three supine measures taken over 10 min (DINAMAP-1846-SX, Critikon Corp., Tampa, FL, USA). Fasting venous blood was drawn for glucose, glycated haemoglobin (HBA1c), triglyceride and renal function. HbA1c and insulin were checked in patients with diabetes only, not in controls.
Cardiac magnetic resonance imaging
LV volumes, mass and function were assessed using CMR. Images for LV volumes and diastolic function were acquired using a steady state free precession (SSFP) sequence and analysed using cmr42© (Circle Cardiovascular Imaging Inc., Calgary, AB, Canada). 18 LV short-axis epicardial and endocardial borders were contoured manually at end-diastole and end-systole for determining end-diastolic volume (EDV), end-systolic volumes (ESV) and stroke volume (SV). Myocardial mass was calculated by subtracting the endocardial volume from the epicardial volume and excluding papillary muscles and trabeculations. LV mass was calculated based on prior knowledge of myocardial density (1.05 g/cm3) and indexed to body surface area (BSA). LV mass to volume ratio was calculated by dividing the LV mass by the LV EDV.
For the LA volumetric and functional analysis,19,20 the biplane area-length method (excluding the appendage and pulmonary veins) was employed. The LA endocardial border was contoured manually in both the 2- and 4-chamber views, with the mitral annulus serving as the anterior border. LA volumes were calculated using the area length method: volume = (0.85 × area 2 )/length. LAEF was derived as follows: LAEF = (LAVmax–LAVmin)/LAVmax. Surrogates of LA reservoir function that is, reservoir volume (LAVmax–LAVmin) and LA conduit function that is, conduit volume (LV stroke volume–LA reservoir volume) were also calculated. All volumetric and mass data were indexed to BSA. Global circumferential peak systolic strain (GCPSS) and global circumferential peak diastolic strain rate (GCPDSR) were determined using tissue tracking software.
Statistical analysis
All statistical analysis was performed with commercially available software package [Prism version 7.0a, 2016; and SPSS Statistics version 20 (IBM, Armonk, NY, USA) for multivariable linear regression]. All data were checked for normality using Kolmogorov-Smirnov test and presented as mean ± standard deviation (SD). Comparisons between the four groups were performed by 1-way ANOVA with post hoc Bonferroni corrections. Bivariable correlations were performed using the Pearson or Spearman method as appropriate. Student’s t test was used for comparison of normally distributed data sets where data were obtained for only two T2D groups (male and female). Significance was defined as p < 0.05. Multivariable linear regression was performed to identify independent determinants of LA EF, GCPSS and GCPDSR. The model contained the following covariables: systolic BP, diastolic BP, plasma triglyceride levels, BMI, and HbA1c.
Results
Participant characteristics
Demographic and clinical data are shown in Table 1: 62 male T2D patients (mean age 44 ± 8 years, BMI 33 ± 5 kg/m2, mean HBA1c of 7.8 ± 1.8%), 67 female T2D patients (mean age 44 ± 10 years, BMI 35 ± 6 kg/m2, mean HbA1c of 7.6 ± 1.2%), 16 male non-diabetic volunteers (mean age 48 ± 17 years, BMI 25 ± 3 kg/m2) and 14 female non-diabetic volunteers (mean age 50 ± 10 years, BMI 25 ± 4 kg/m2) were recruited. Participants in all groups were of similar age, and there were no significant differences in BP, diabetes duration, diabetes treatment or metabolic profile between the two diabetes groups. Whereas there was no significant difference in systolic BP between the patients with T2D, and gender-matched controls, diastolic BP and resting heart rates were higher in both the male and the female patients with T2D compared with gender-matched controls; although these remained within normal limits.
Clinical characteristics.
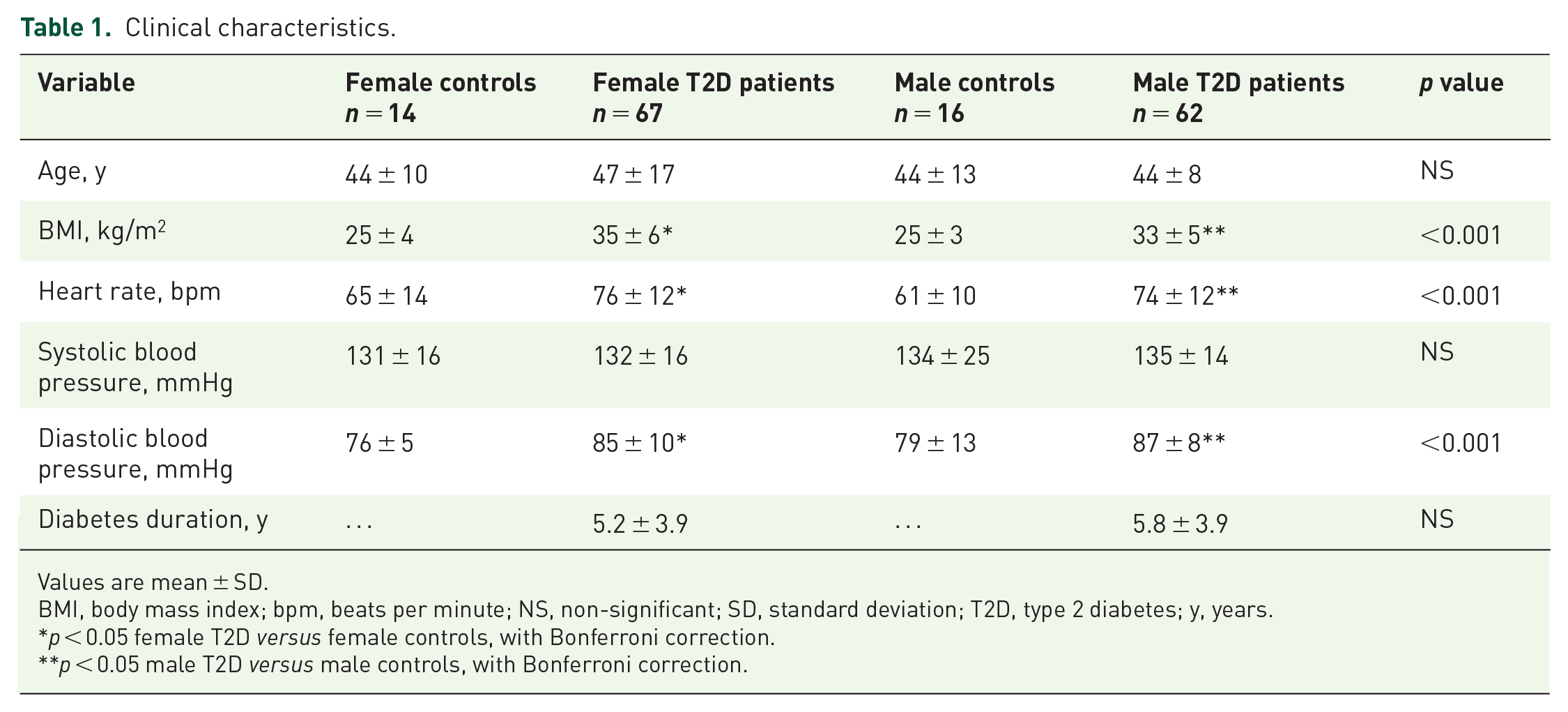
Values are mean ± SD.
BMI, body mass index; bpm, beats per minute; NS, non-significant; SD, standard deviation; T2D, type 2 diabetes; y, years.
p < 0.05 female T2D versus female controls, with Bonferroni correction.
p < 0.05 male T2D versus male controls, with Bonferroni correction.
Plasma metabolic phenotype
Plasma biochemistry results are summarized in Table 2. There were no significant differences in plasma triglyceride content, lipid profile and glycaemic control between female and male T2D patients.
Biochemistry.

Values are mean ± standard deviations.
HDL, high density lipoprotein; LDL, low density lipoprotein; NS, non-significant; T2D, type 2 diabetes.
Cardiac geometry and function
CMR results are summarized in Table 3. LV volumes and ejection fraction (EF) were similar across the groups. However, whereas LVEF was not significantly different across the groups, subtler functional differences in strain parameters were evident in peak systolic circumferential strain (p < 0.0001) and diastolic strain rates (p = 0.001) in males with T2D compared with females. Strain parameters were not significantly different between the male controls and male T2D patients, or between female controls and female T2D patients.
CMR findings.
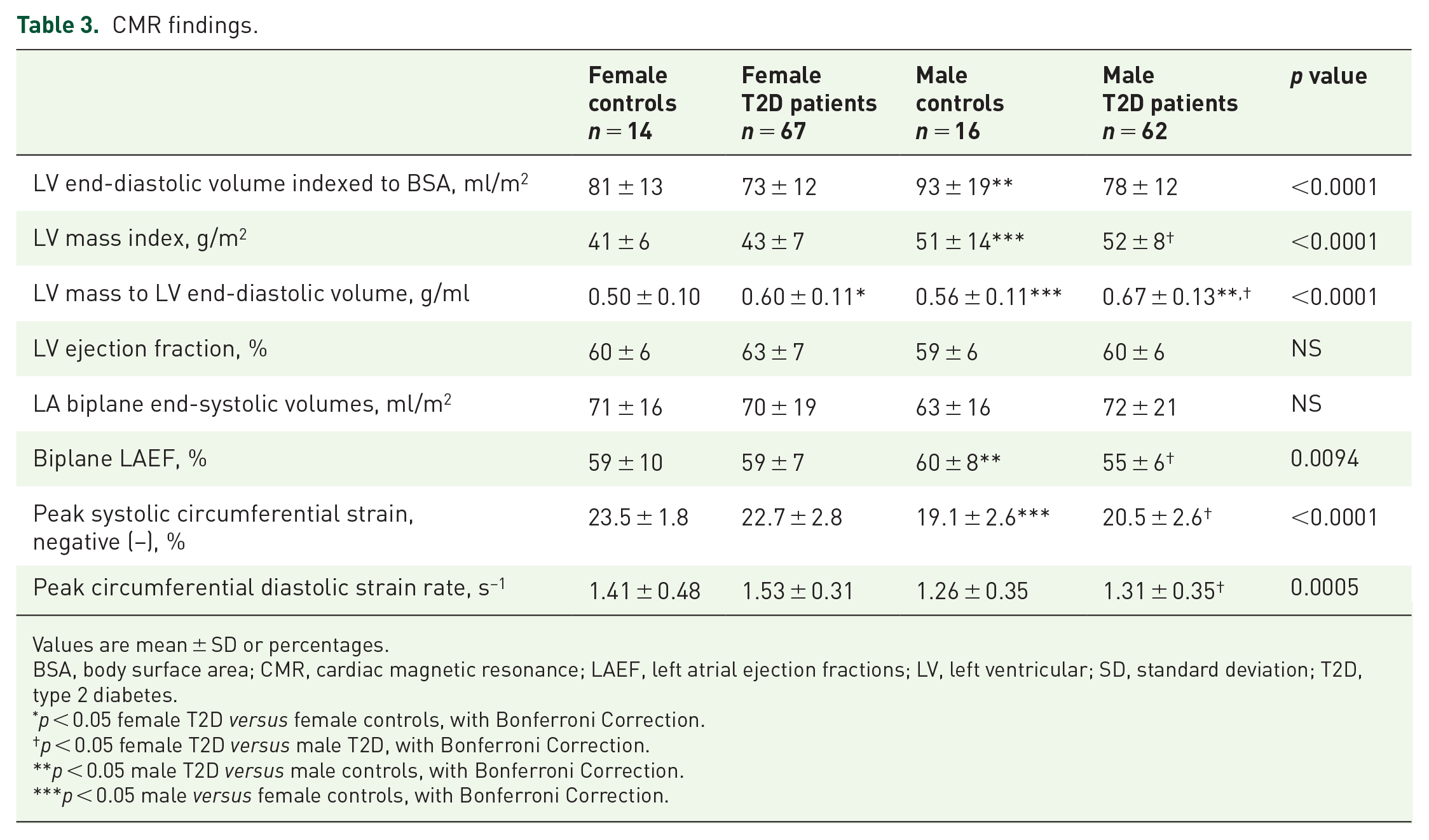
Values are mean ± SD or percentages.
BSA, body surface area; CMR, cardiac magnetic resonance; LAEF, left atrial ejection fractions; LV, left ventricular; SD, standard deviation; T2D, type 2 diabetes.
p < 0.05 female T2D versus female controls, with Bonferroni Correction.
p < 0.05 female T2D versus male T2D, with Bonferroni Correction.
p < 0.05 male T2D versus male controls, with Bonferroni Correction.
p < 0.05 male versus female controls, with Bonferroni Correction.
There was a stepwise decline in LV end-diastolic volumes indexed to BSA in the order of greatest volumes to the smallest: non-diabetic male controls, female controls, male T2D patients and female T2D patients. Concentric remodeling was present in both sexes of diabetes groups compared with controls (p < 0.0001), as defined by increased LV mass-to-volume ratio (LVMVR). However, the degree of concentric hypertrophy was greater in male patients compared with female patients with T2D (12%, p = 0.002). LV mass index was significantly increased in T2D patients of both genders compared with controls. Finally, there were no significant differences in biplane LA volumes across the groups. Biplane LAEF was significantly reduced in male patients compared with female patients with T2D (p = 0.032) and compared with male controls (p = 0.038). There were no significant differences in biplane LAEF, global peak systolic circumferential strain and diastolic strain rates in females with T2D compared with female controls (Figure 1).
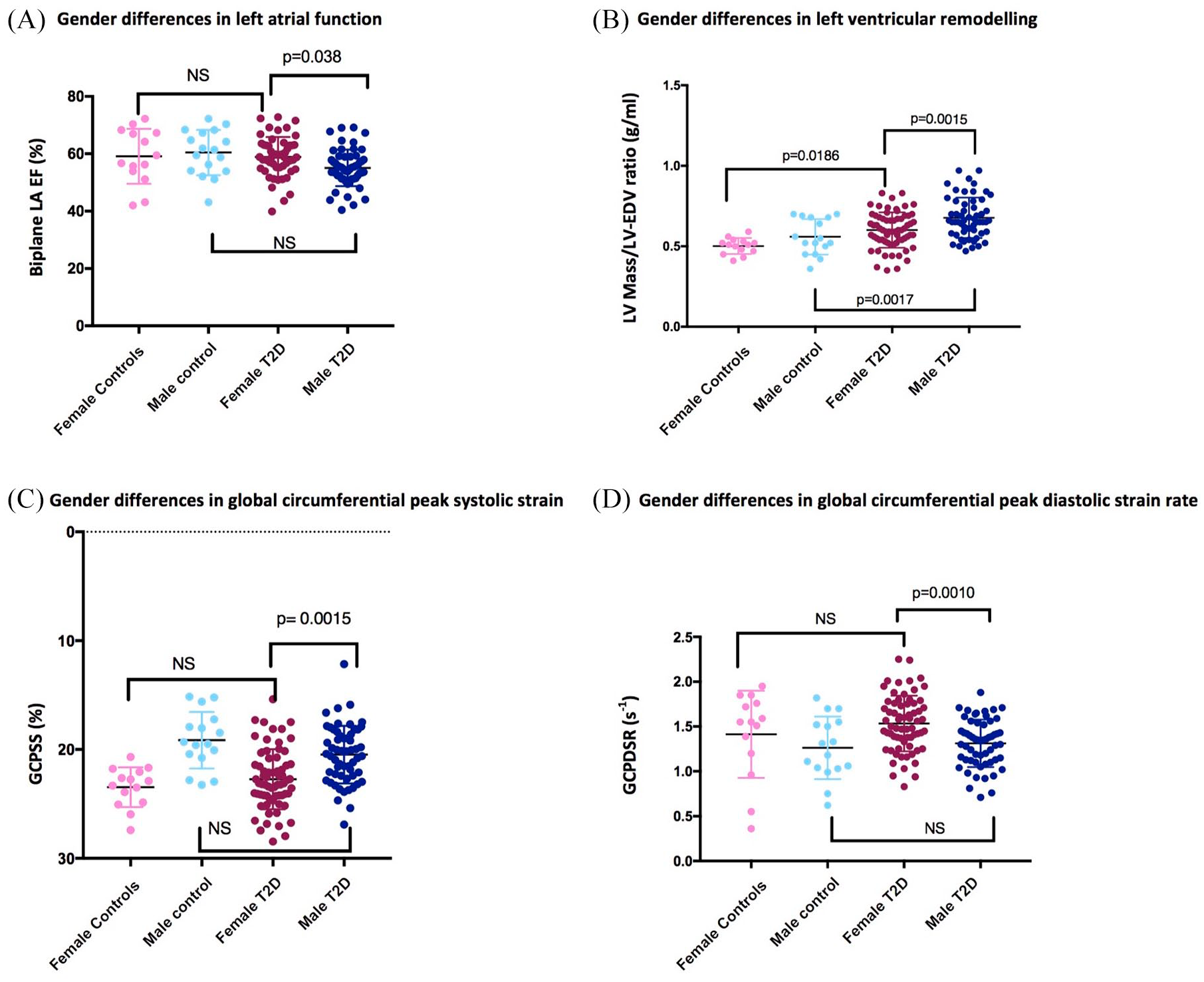
Differences in cardiac geometry and function between female and male patients with T2D, and female and male controls: (A) biplane LAEF; (B) LVMVR (g/ml); (C) GCPSS (%); (D) GCPDSR (s–1).
Sex-specific associations of glycemic control and cardiac parameters
Figure 2 represents the associations of HbA1c and CMR-assessed cardiac parameters.

Associations between glycaemic control as measured by HbA1c and GCPSS, LVMVR, GCPDSR in female and male T2D patients, biplane LAEF. Associations between: (A) GCPSS and HbA1c in female T2D patients; (B) GCPSS and HbA1c in male T2D patients; (C) LVMVR in female T2D patients; (D) LVMVR in male T2D patients; (E) GCPDSR and HbA1c in female patients; (F) GCPDSR and HbA1c in male patients; (G) biplane LAEF and HbA1c in female patients; and (H) biplane LAEF and HbA1c in male patients.
In the female T2D cohort there was a weak correlation between LV GCPSS and HbA1c (r = 0.2995, p = 0.018); but the correlation of HbA1c with LV GPDSR (r = –0.2183, p = 0.088), LVMVR (r = 0.2324, p = 0.069), and biplane LAEF (r = –0.1765, p = 0.174) did not reach statistical significance. In the male T2D cohort, HbA1c did not correlate with LVMVR, peak circumferential diastolic strain rate, and circumferential peak systolic strain; but there was a trend for significant correlation with LAEF (r = 0.2329, p = 0.087).
Gender-specific associations of BP and cardiac parameters
There was no significant correlation of systolic BP with biplane LAEF in either the female (p = 0.640) or the male (p = 0.584) cohorts with T2D. Similarly, diastolic BP was also not linked to biplane LAEF in either of the T2D cohorts (female T2D p = 0.416; male T2D, p = 0.297). Neither systolic nor diastolic BP were linked to LVMVR, LV global peak circumferential strain or LV global circumferential diastolic strain rates in either of the sexes with T2D.
Gender-specific associations of BMI and cardiac parameters
There was no significant correlation of BMI with biplane LAEF (p = 0.468), with the peak global circumferential strain (p = 0.182), with GCPDSRs (p = 0.224), or with LVMVR (p = 0.902) in female patients with T2D. Similarly, there was no significant correlation of BMI with biplane LAEF (p = 0.723), with the peak global circumferential strain (p = 0.853), or with LVMVR (p = 0.243) in men with T2D in this study. However, there was a trend in clinical significance in the correlation between BMI and GCPDRSs (r = 0.465, p = 0.069) in men with T2D only.
Multivariable predictors of LA function, and strain parameters
In a model including the systolic BP, diastolic BP, plasma triglyceride levels, BMI and HbA1c as covariables, HbA1c (β = 0.315, p = 0.019) was the only predictor of peak global circumferential strain in the female T2D cohort, whereas diastolic BP (β = –0.277, p = 0.015) and BMI predicted (β = –0.277, p = 0.025) GCPDSRs. None of these covariates were predictors of biplane LAEF in the female T2D cohort. The same model for the male T2D cohort showed plasma triglyceride levels to be the only predictor of both peak global circumferential strain (β = 0.322; p = 0.034) and GCPDSR (β = –0.390, p = 0.008). None of the other covariates in the model were associated with the biplane LAEF, or strain parameters in the male T2D cohort.
Late gadolinium imaging
Late gadolinium imaging confirmed the presence of enhancement in a non-ischemic pattern (affecting the mid-wall) in three female patients, four male patients with T2D, and one female control in the Leicester cohort. None of the Oxford cohort participants had significant enhancement. No patients included in this study had myocardial infarction.
Discussion
By examining ventricular remodeling, deformation and atrial function, our findings characterize for the first time that in patients with T2D, male sex adversely affects the phenotypic expression of heart disease in diabetes, with greater LV concentric hypertrophy, worse LV and LA function, compared with female gender. However, when compared with male and female age-matched controls, concentric remodeling is present in both sexes of patients with T2D, but with a greater degree of concentric hypertrophy in males. LA and LV function and deformation were preserved in women with T2D. Whereas in women with T2D glycaemic control is linked to LV contractile function, there was no such relationship in men with T2D.
LV remodeling
We have shown here concentric remodeling was present in both sexes with T2D; however, the degree of concentric hypertrophy was greater in males. Consistent with our findings, the recently published prospective UK Biobank study has also shown that diabetes is associated with a reduction in LVEDV, 21 although the differences in LVEDV between the female patients with T2D and the female controls did not reach significance in the UK Biobank Study.
Multiple studies have recently shown LV concentric remodeling to represent the main structural characteristic of diabetic heart disease, indicated by increased LVMVR. 22 Importantly, LV concentric remodeling is known to be more strongly predictive of cardiovascular mortality than eccentric remodelling.6,23
There is now a wide range of literature that supports the idea that accumulation of myocardial lipids plays a role in concentric remodeling in diseases of upregulated fatty acid metabolism such as diabetes and obesity.8,24 Supporting this concept, we have previously shown myocardial triglyceride accumulation to be predictive of concentric LV remodeling in patients with T2D. 8 Similar to our findings in T2D, Rider et al. have previously shown greater LVMVR in both normal weight and obese males than normal weight and obese females. 25
LV deformation
Myocardial strain and strain rate allow more sensitive characterization of subtle myocardial performance. 26 Using CMR, multiple studies have shown subtle contractile abnormalities even in the absence of abnormal LVEF represented by impaired LV systolic strain,8,9 and diastolic strain rate in patients with T2D. 10 Here, we have determined male sex was associated with worse systolic strain and diastolic strain rate in patients with T2D compared with female sex. We are not aware of any previous study investigating gender differences in myocardial strain parameters of the LV in patients with T2D. This finding is also befitting of the idea that men with T2D show worse cardiac phenotype than women. The cause of sex-related differences in myocardial deformation in T2D is unclear, and unlikely to be accounted for by differences in body size given the lack of association between BMI and strain parameters in either sex.
LA function
In this study we have shown that LA function was reduced only in men with T2D, whereas women with T2D had preserved LA function. Furthermore, we showed that LA function was not linked to BMI, systolic or diastolic BP, LV remodelling or contractile function in either the male or the female T2D cohorts.
The LA has important functions, such as storing pulmonary venous return during LV contraction, acting as a conduit when the blood is passively transferred into the LV, and contributing significantly to LV filling with the active LA contraction during LV end-diastole. 27 It is well established that enlargement of the LA is a marker of mortality in the general population. 28
Based on our results, it is tempting to speculate that the incidence of atrial arrhythmias, including atrial fibrillation (AF), may be higher among men with T2D than women with T2D. Supporting this theory, there is 60% higher age-adjusted prevalence of AF in men compared with that in women in the general population. 29 Further, based on 38-year follow-up data from the Framingham Study, men had a 1.5-fold greater risk of developing AF than women after adjustment for age and predisposing conditions. 30 Importantly in the Framingham cohort, of the cardiovascular risk factors, only hypertension and diabetes were significant independent predictors of AF. Further studies are needed to assess sex differences in atrial arrhythmias in patients with T2D.
Limitations
Given the cross-sectional nature of the data, we cannot infer causality in the relation to the structural and functional sex differences observed. Furthermore, longitudinal studies are needed that include detailed phenotyping of patients to determine whether the observed sex-related differences are important in the development of future HF. Such questions should be answerable from population studies. Only in the Oxford cohort was CT coronary angiography performed to exclude significant obstructive coronary artery disease (>50% luminal narrowing) in T2D patients; as such, it is possible that occult coronary artery disease could be present in some of the patients.
HbA1c measurements were not taken in the control group of Leicester studies. Although diabetes was excluded on fasting glucose measurements, this does preclude investigation of the role of insulin resistance in the pathogenesis of cardiac remodelling.
Our sample size was small. We have not obtained additional information about the glycaemic variability in this study.
Conclusion
In T2D, male sex adversely affects the phenotypic expression of diabetic heart disease, with greater LV concentric hypertrophy, worse LV and LA function, compared with female gender. These striking differences in the cardiac phenotype promote awareness of gender-specific risk factors in search of treatment and prevention of diabetes-associated HF. Further large-scale longitudinal studies looking at sex differences in HF-related mortality in T2D are needed.
Footnotes
Author Contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The studies were supported by the Oxford Partnership Comprehensive Biomedical Research Centre and University of Leicester National Institute for Health Research Biomedical Research Centre. E.L. has received support from Wellcome Trust and National Institute for Health Research (NIHR). S.N. has received support from the Oxford British Heart Foundation (BHF) Centre of Research Excellence. G.P.M. has received support from NIHR. L.A. has received support from the BHF. A.S. has received support from NIHR. G.S.G. has received support from BHF. M.D. has received support from NIHR.
Conflict of interest statement
The authors declare that there is no conflict of interest.
