Abstract
For paediatric patients with type 1 diabetes, intensified insulin therapy with either multiple daily injection or insulin pump therapy is currently the only method of treatment. To optimize this therapy, insulin analogues are fixed parts of all therapy regimens. New ultra-rapid insulins seem to be beneficial not only in adults but also in this age group. New developments in long-acting analogues have demonstrated safety and will be regular in paediatrics, we hope, soon. Furthermore, the psychosocial approach for consideration of real-life aspects becomes more the focus of therapeutic regimens and is implemented into international guidelines. Technical improvements, such as continuous glucose monitoring, particularly in combination with pump therapy, support the great success of rapid-acting analogues by reducing hypoglycaemias. Non-insulin agents such as SGLT2-inhibitors show beneficial aspects in people with type 1 diabetes. For outpatient care with these currently off-label-used drugs, special training for measurement of ketones should be imperative.
Introduction
Diabetes mellitus type 1 is the most common metabolic disease in childhood and adolescence in Germany, with approximately 30,000 children and adolescents affected up to age 19 years. The insulin regimen can be carried out as an intensified therapy using multiple daily injections (MDIs) according to the principle of basal and bolus doses with syringes or pen injections or alternatively, as continuous subcutaneous insulin infusion (CSII) insulin pump therapy.
The existing 2014 guideline for insulin therapy of diabetes in children and adolescents is currently being revised by the International Society for Paediatric and Adolescent Diabetes (www.ispad.org) and will be published as an updated version in 2018. The basic principles of intensified insulin therapy will likely remain unchanged. The new technological advances, such as continuous subcutaneous glucose measurement (CGM) and sensor-augmented pump therapy (SaP) are of particular importance in children and adolescents due to age-related metabolic fluctuations; they have also been included in the guideline and will be featured as a separate chapter. A further focus of the innovations lies in the area of psychosocial support, which takes into account the influences of diabetes on psychological development, as well as adjusting to the changed family schedules with long school hours and need for external support. 1
In this respect, clarification is needed if newly developed insulin analogues are to help improve therapy. Furthermore, adjunct therapy to insulin with non-insulin drugs also seems to be an interesting approach in paediatrics.
Insulin therapy in children and adolescents with type1 diabetes
While in our practice preschool children are treated for diabetes onset with pump therapy, the intensified insulin therapy with MDIs is the first-line choice of therapy for patients with onset at school age. No premixed or combination insulin preparations are used for insulin therapy in the paediatric age group due to the need for flexibility in therapy adjustment. In addition to conventional human insulins [regular and isophane (NPH) insulins], various long-acting and short-acting insulin analogues have become available that can be selected for therapy according to the patient’s individual daily schedules (Table 1). The ultra-rapid insulin Fiasp, which is approved for adults, has the same pharmacokinetic and pharmacodynamic profile in the paediatric group as in adults.
Insulins used in paediatrics (German insulin products).
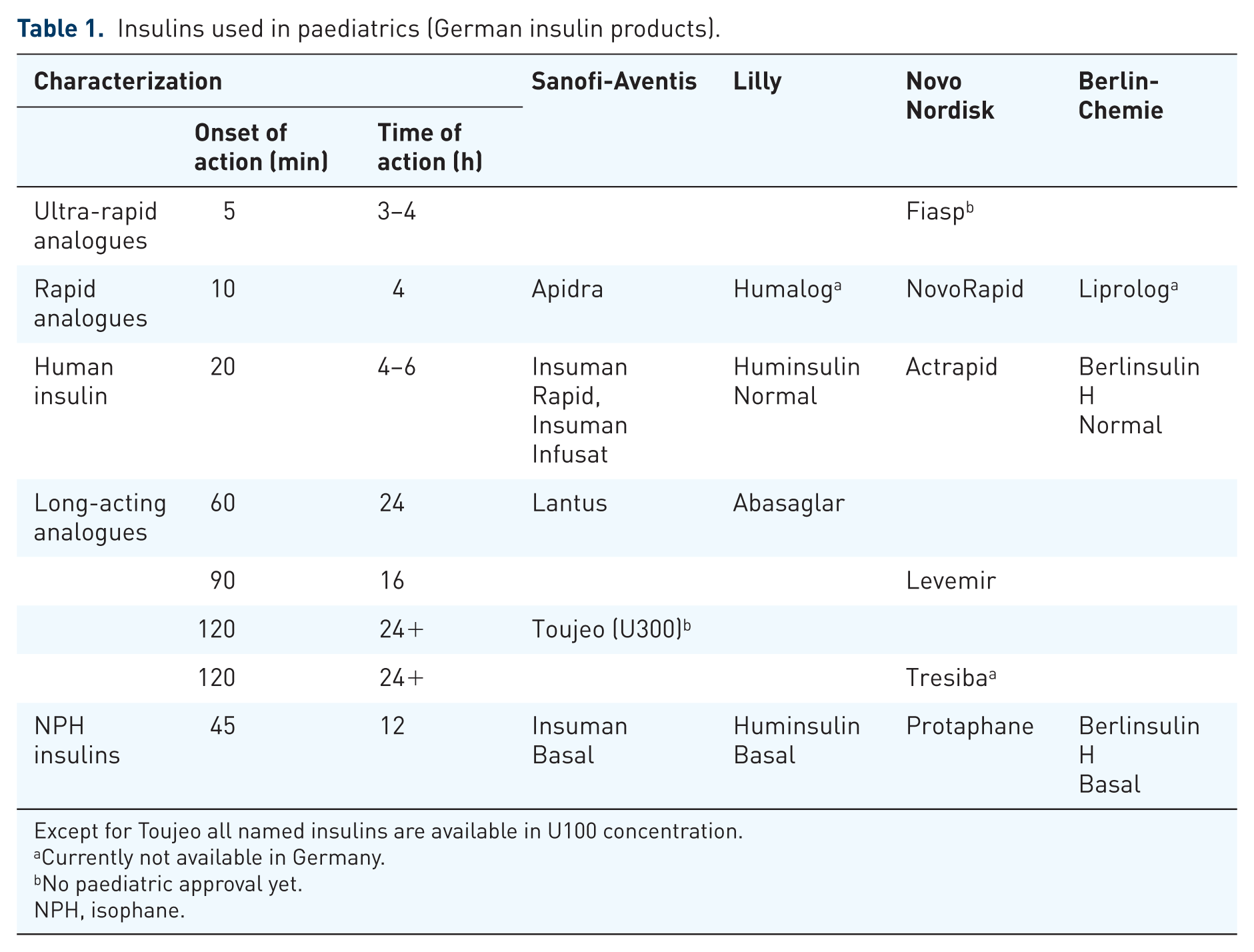
Except for Toujeo all named insulins are available in U100 concentration.
Currently not available in Germany.
No paediatric approval yet.
NPH, isophane.
In outpatient care, insulin is mostly used in the concentration U100 (100 IU insulin/ml). In contrast to this, there are also recently more highly concentrated insulins that are only available as disposable pens to prevent dosing errors, which could potentially happen when using cartridges for reusable pen injectors.
Lower concentrated U40-insulin is no longer used, even in neonatal diabetes or infants.
Principle of intensified insulin therapy: multiple daily injections
The basis for the intensified insulin therapy is the basal-bolus principle. In this regimen, a long-acting insulin is used to cover the glucose levels resulting from gluconeogenesis, which is administered primarily in a fixed dose. Separately, the prandial insulin is calculated according to carbohydrate counting being dosed flexibly according to the current glucose measurement and planned activity level.
Effect of basal insulins: long, but as flexible as possible
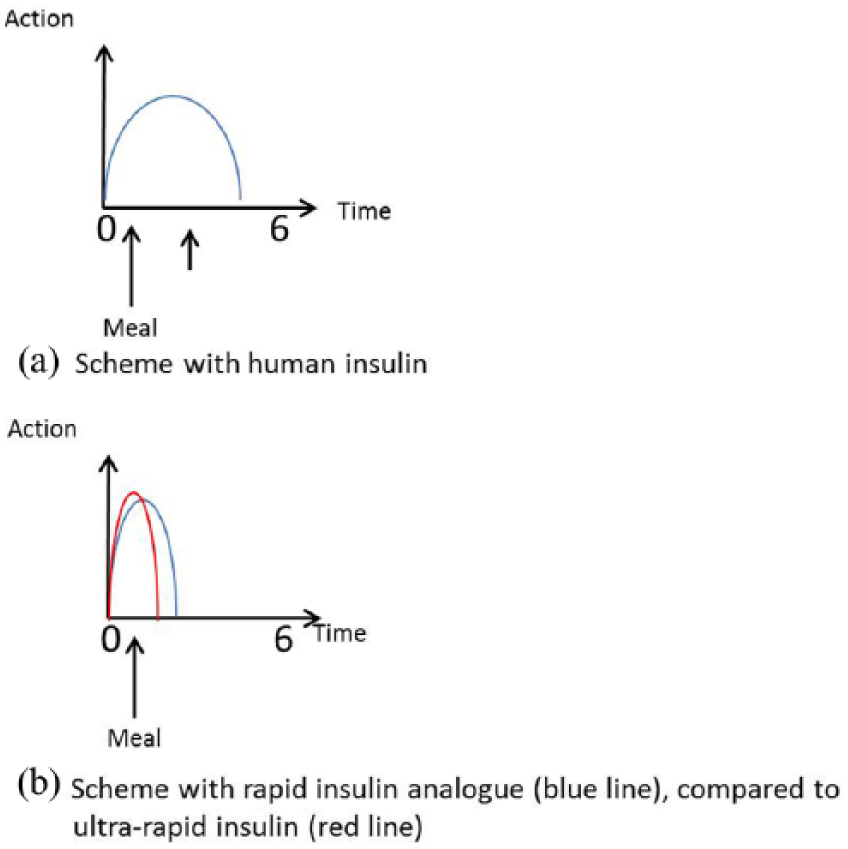
A normal-growing child with type 1 diabetes needs about 1 IU of insulin per kg of body weight per day after the end of the remission phase. Approximately one third of this is to be administered as basal insulin. This dose is then divided again according to the requirements so that two thirds of the basal amount is administered at night and one third during the daytime. It should be noted that the insulin action is at its maximum at midday due to the circadian variation of insulin sensitivity and as more physical activity takes place during the day (Figure 1a), thus, the basal action should be the least at this time. Furthermore, the nightly excess of growth and sex hormones, particularly in the second half of the night, triggers an increase in insulin requirements. Especially during puberty, this can be observed as a ‘dawn phenomenon’.

Various regimens of basal insulin therapy.
When initiating multiple daily injections, the use of NPH insulin is recommended to tailor the dose to the circadian variation of insulin requirement. With the intermediate-action NPH insulin, the basal dose is often distributed over three separate injection times to match the basal insulin need at different times (Figure 1b), as in children and adolescents, NPH is seldom active as long as 12 h. Thus, one more injection of NPH is needed at lunch time. The negative perception of this additional needle prick is often highlighted as an argument against NPH schemes. However, in context, it more precisely mimics physiologic insulin secretion, compared with long-acting analogues. This approach is particularly recommended in primary school children, although it often requires the help of parents or other caregivers to ensure proper insulin injections.
With increasing age and the importance of school lunch without supervision, experience shows the midday NPH dose is frequently omitted by older children and adolescents. In such a situation, a regimen using a long-acting insulin analogue twice daily (e.g. Detemir) is an alternative (Figure 1c). Especially for older adolescents, a further simplification of the treatment regimen with a basal insulin administration once daily can be a choice for improving basic therapeutic adherence (Figure 1d).
New basal insulins
Innovations in the basal insulin analogues in the past few years have been achieved with different approaches. Insulin Degludec forms subcutaneous multihexamers after injection which lead to a more stable and persistent insulin effect compared with, for example, glargine. 2 Degludec has also shown itself to be safe and effective in different therapeutic challenges. 3 Degludec is approved for children and young people but is currently not available on the German market.
Another approach to altering insulin activity is changing the insulin concentration. The long-established basal insulin analogue glargine was formulated in a higher concentration (U300) and marketed as Toujeo from the company Sanofi. This concentration change resulted in a flatter profile and a longer duration of action. 4 Toujeo is presently approved for adults; regulatory trials in paediatrics are not completed yet.
With Abasaglar, Lilly is the first company to offer a biosimilar insulin, and since it is glargine, it has also received direct approval for children and adolescents. In contrast to generics in single-molecule drugs, biosimilars require a significantly more complex authorization process, since insulin, for example, as a peptide hormone, also has a secondary and tertiary structure that must be identical to that of the original preparation. For Abasaglar, it shows no different conformation compared with the ‘original’ glargine. 5
Prandial bolus: rapid insulins for quick action
For the insulin doses covering meals or glucose corrections, human and short-acting insulin analogues are available. With the help of an individualized insulin plan, insulin doses can be calculated exactly for each meal and circadian insulin sensitivity, so there is a high degree of flexibility in meal and activity schedules for patients and families.
For calculation of the insulin dose, the respective mealtime insulin sensitivity factor (insulin units per unit of carbohydrate) is multiplied by the amount of carbohydrates of the meal. The difference between the current glucose level and the target value is divided by the correction factor. These two dose portions are combined and then administered together. The factors can differ in the course of the day, according to the circadian variation of insulin sensitivity.
Today, human insulin and fast-acting insulin analogues are alternative choices as mealtime insulin (Table 1). If a large main meal is to be followed by snack later on (for example, breakfast and a school snack) regular human insulin can be used to cover both (Figure 2a). In contrast, if only one meal immediately following the injection is to be covered, a fast analogue can be used, as described above (Figure 2b). This regimen implies a certain loss of flexibility since the timing and the amount of the intermediate meal time are already more or less fixed at the time of the insulin injection. The clear advantage is, however, that a child, for example, in the primary school, can take his fixed breakfast meal without having to take an insulin injection during the school break.
Insulin administration examples in different meal settings.
The use of regular human insulin as the prandial dose is often referred to as ‘old fashioned’ due to the need for delaying the meal after the insulin injection because of the slower onset of insulin action. However, the ‘meal without injection’ strategy, as described above, is highly appreciated by children and families. Maximum flexibility is achieved by educating and training the families in all of the different options for mealtime insulin dosing. The use of fast-acting insulin analogues is nowadays an established component of therapeutic regimens in Germany (Figure 3).

Evaluation of the Diabetes-Patienten-Verlaufsregister (DPV) database for the diabetes centre Auf der Bult.
New development: ultra-rapid insulins
For subcutaneous insulin therapy there is the striking postulate, ‘Wrong insulin at the wrong time in the wrong amount in the wrong place.’ It should be noted that each subcutaneous insulin is fixed in its amount and is subject to a certain delay in action; in contrast to this, endogenous insulin from the pancreas is immediately released into the portal circulation when the blood glucose increases, where it can take a direct start in the liver. 6
In order to further accelerate the clinical action of the insulin, faster insulins are desired for prandial use. In spring of 2017, the insulin ‘Fiasp’ (faster insulin aspart) from Novo Nordisk was approved for the European market. Faster aspart is a new formulation of insulin aspart, adding the excipients niacinamide and L-arginine to the insulin preparation. Both additives are so-called substances ‘generally regarded as safe’ according to the classification of the US Food and Drug Administration (FDA), for which no health hazard is to be expected. Niacinamide causes a faster absorption, speeding up the monomer formation, and L-arginine serves to stabilize the solution. In clinical studies in adults it has been shown that Fiasp appears approximately 5 minutes faster in the blood. Pharmacodynamically, there is approximately 70% higher reduction of the blood glucose level in the first 30 minutes. 7 In clinical studies, significant glycated haemoglobin (HbA1c) improvements were seen with the use of Fiasp. 8

Example of sensor-assisted pump therapy in case of imminent hypoglycaemia.
In early studies in children and adolescents, these pharmacokinetic and pharmacodynamic effects are shown to have been preserved, 9 while the regulatory studies in this age group have not yet been completed.
Pump therapy is increasing
An insulin pump is a technical aid that uses a motor to deliver a defined amount of insulin via a subcutaneous catheter. The fundamental principle of the differentiated substitution of basal and bolus insulin also applies to pump therapy. However, the reservoir of an insulin pump is filled with one insulin only, usually a fast-acting analogue. To cover the basal insulin requirement, the insulin is delivered via the pump as a continuous basal-rate infusion, followed by manual bolus delivery at meals, or for correction.
The basal rate can be dosed in the smallest increments (depending on the pump model, the smallest dosing of up to 0.01 IU/h is possible). This flexibility, as well as the option to deliver prandial insulin several times a day without injections, has led to CSII being established as the initial therapy for type 1 diabetes in the preschool age.
Furthermore, the mealtime insulin can also be calculated exactly in decimal ranges of an insulin unit, which cannot be achieved with a pen or a syringe. This determination of the correct prandial insulin dose can be made by a bolus calculator integrated into the pump, individually programmed according to the patient’s dosing regimen. These bolus computers have been shown to be advantageous in the day-to-day routines and correspondingly, in improving glycaemic control. 10
In everyday life, for example, of children that are not under immediate parental supervision, the barrier to dosing insulin with an insulin pump for other caregivers such as kindergarten personal or teachers is significantly lower than for injections.
The current data from the Diabetes-Patienten-Verlaufsregister (diabetes registry in Germany and Austria) registry group, published in the German Diabetes Report, show that insulin pump therapy is used in 90% of patients under 5 years of age, and approximately 50% of paediatric patients, overall. 11
In adolescence, the primary indication for pump therapy is the dawn phenomenon, characterized by hyperglycaemia in the early-morning period caused by anti-insulin hormones such as growth and sex hormones.
Fluctuation in the blood glucose values despite using analogue insulins in MDI is an additional indication to switch to pump therapy, particularly when a high evening basal dose comes with a threat of nocturnal hypoglycaemia. The indication for insulin pump therapy differs a little internationally in the various scientific societies. 12
After an individual education on the principles of the pump therapy, including discussing all the advantages and disadvantages of the respective systems, the choice of pump model should be made by patients and families themselves. Of course, the reason for choosing pump therapy in the first place should be considered, since not all insulin pumps are suitable for some principles (i.e. the need for automatic suspension of the insulin delivery in patients exhibiting repeated severe hypoglycaemia).
In Germany, the insured person or the parents must apply for insulin pump therapy directly to their health insurance. If necessary, a medical opinion must be submitted from the prescribing physician on request of the medical service of the health insurance company when the latter examines the application.
Following the already mentioned circadian rhythm, the basal insulin is dosed according to the different insulin sensitivities during the day, in which case, a different amount can be administered every hour. The basal insulin is approximately one third of the daily total dose. The pattern of the basal rate differs between different age groups.
In order to find an estimate for the age-appropriate dosage, there are tables showing the average distribution depending on age and weight. 13 The carbohydrate and correction factors also follow the circadian variation of insulin sensitivity. The target glucose value for setting the dose calculation is 100 mg/dl per 5.6 mmol/l (120 mg/dl per 6.7 mmol/l for small children), and 120 mg/dl per 6.7 mmol/l (140 mg/dl per 7.8 mmol/l for small children) at night.
In the adult population, the use of new ultra-rapid analogues in pump therapy showed even more beneficial results than in MDI. 14
Sensor therapy: the way to a new gold standard
As a further technological tool, SaP therapy has been ‘on the advance’ in recent years.
The glucose content of the interstitial fluid is measured by means of an enzyme-coated probe, which is placed into the subcutaneous tissue by injection and can remain there for about 1 week depending on the model, transmits glucose wirelessly to a receiving device. Compared with a static image produced by a blood glucose measurement, this CGM provides dynamic data. For comparison: while a photo shows a football in the goal, the game history can be described with CGM before and on the way to eventual score. With CGM, there are several ways of viewing the data:
Realtime: In addition to the current value, the receiving device of the sensor also displays trend arrows that indicate the predicted development of the glucose, so that direct consequences for the therapeutic adjustment are possible (e.g. ingestion of glucose at an already low value with an additional downward arrow). Furthermore, the devices can emit different alarms, which warn the user of high or low values.
Retrospective analysis: In addition to the real-time display of the data, each sensor system offers software with which the data can be analysed retrospectively. The glucose curve of the last hours can be considered, in order to derive direct consequences for future action. With this form of analysis, many conclusions can also be drawn about the overall control of diabetes. Regular patterns in the glucose curves, for example, give information about the effect of certain meals and may indicate necessary changes in dosing or behaviour.
In addition to the above, the ‘FreeStyle Libre’ device, the so-called flash glucose measurement, is available. The main difference to the CGM devices is that the values are not sent continuously to the receiver but are only displayed when actively retrieved by scanning through the user. Thus, this device has no alarm function.
Glucose sensor systems provide users and treatment team with a significant amount of information. In addition to the direct therapeutic aspects, there is also the possibility for diagnostic use of CGM for the therapy evaluation or in the case of therapeutic changes, for example, in mature-onset diabetes of the young forms.
Furthermore, the additional comfort of this measurement tool should not to be underestimated from the patient’s point of view, since the number of blood glucose measurements and the related aspects of lancing pain and measurement time can be significantly reduced. The German S3 guideline of the AGPD (paediatric section of German Diabetes Society), updated in 2015, takes into account that the blood glucose measurement is no longer regarded solely as a control, but the type of glucose self measurement is an integral part of the education program. 15
Since September 2016, real-time CGM devices are part of the approved reimbursement of the statutory health insurance funds. 16 If patients are treated with their intensive insulin therapy by a professional diabetes team experienced in dealing with such devices, and training of the patients and evaluation of the therapy is ensured, these devices can be prescribed. Indications are, above all, not achieving the therapeutic goal after taking advantage of all other possibilities. So far, the ‘Libre’ system mentioned above is not part of the regular CGM reimbursement scheme.
According to the present guidelines, an experience from practice diabetology, the indications for using CGM are mainly the following:
Reducing hypoglycaemia: sensor-augmented pump therapy as the method of choice
SaP describes the support of insulin pump therapy by a continuous glucose measurement. Here, you have the option to choose between different devices from different manufacturers, and to combine any insulin pump with a CGM system in order to take advantage of the CGM (alarm messages, trend displays) for therapy. It should be remembered that not every combination is useful here, so an accurate planning of CGM therapy by patients and the training team has to be done together.
In order to be able to use the data of the sensor measurement directly for therapy adjustment, the insulin pumps of the Medtronic company have incorporated safety algorithms to prevent hypoglycaemia. The first of these devices became available 7 years ago and was able to suspend the insulin delivery for 2 h when reaching a threshold value.
The newest model, MiniMed 640G (Medtronic, Inc., Northridge, CA, US), can interpret the sensor values with a calculation algorithm and predicts the glucose curve for the subsequent 30 min. If a defined threshold value is predicted to be surpassed, the insulin delivery is suspended for a period of 30–120 min. If the calculation results are predicted to be in the safe range above this limit again, the insulin infusion is resumed. 19
This form of therapy requires trust in the system, 20 since use is associated with a transfer of tasks and responsibility to the device’s algorithm. Since the patients and their parents have to modify their attitudes regarding ‘treatment of hypoglycaemia’ established by conventional education, a good, detailed and repetitive structured training of the patients, for example, according to the training programme SPECTRUM, 21 is necessary.
Non-insulin adjunctive therapy: chance or threat
Metformin was often considered as adjunct therapy to insulin in type 1 diabetes as it is an old and well-known drug that reduced insulin need, and with benefits to obesity in patients with type 2 diabetes, and particularly for obese type 1 patients. However, recent publications show no benefit to this approach, and an increase of side effects. 22
SGLT2-inhibitors, which showed large benefits in cardiovascular outcomes in type 2 patients 23 are under investigation in people with type 1 diabetes, as well. In type 1, agents from these group showed benefits in HbA1c reduction, 24 as well as improved weight, blood pressure and hypoglycaemia rates. In the younger population, dapagliflozin, for example, is one substance from this group that appears to be well tolerated, 25 with the same pharmacokinetic and pharmacodynamic profile.
The risk for euglycaemic ketoacidosis in this substance group, also highlighted by the FDA, 26 is often discussed. However, this is a rare event and a potentially preventable and manageable side effect but should always be considered when choosing patients for such treatment, if it becomes approved. Certainly, testing of blood ketones, whenever fasting or clinical signs of diabetic ketoacidosis such as nausea are present, and immediate interruption of treatment in such situations or when procedures may become necessary, as well as following common ‘sick day rules’ is recommended. Furthermore, one must remember that use in type 1 diabetes is still off label. Further studies on the underlying mechanism need to be performed.
Psychosocial aspects in training and long-term care, a key focus of the new guideline
The former standard that a child is in kindergarten or school in the morning and is supervised by the mother at noon is long overdue in our modern society. The schedules of school, work and family days are extremely different from one day to the another.
In kindergarten, elementary and boarding schools, children depend on the reliable help of trained adults, since they cannot take care of their diabetes alone. The mostly very cooperative care and teaching staff can take over many tasks after training. If this is not possible, an outpatient nursing service can be prescribed. The first aim should always be a trustful cooperation between parents and the care-giving institution. Information material for caregivers is available from AGPD in German language. 27
The above-described dose calculation of the insulin is usually taken over by the children in the preschool age by using dose calculation aids built into insulin pumps; nevertheless, there are many other aspects of the therapy that need to be considered when managing diabetes. Apart from the correct estimation of the carbohydrate content and the appetite of the child, other influencing factors such as exercise, intercurrent illness or stress may influence metabolic regulation. In the case of planned physical exercise, an adequate reduction of the previous insulin amount must be used because exercise leads to improved insulin sensitivity, potentially causing postexercise hypoglycaemia.
Using supplementary carbohydrates as routine prior to physical exercise should only be avoided when a calorie-conscious diet is in place. All these factors should also be considered when planning out-of-family care for children with diabetes.
The new paediatric guideline takes particular account of mental health. There should be a low-threshold access for patients to make contact with psychological specialists who are knowledgeable about diabetes. In addition, all members of the team should be trained not only on glycaemic control, but also on all aspects of the intellectual, school, emotional and social development of patients.
New family constellations and migrants with linguistic or cultural barriers can be everyday obstacles to care that require increased attention.
Age-appropriate education
Evaluated training programs for children and young people who address the needs of the respective age groups are established.28,29 There is also a medical and psychological guide for parents. 30
Conclusion
Flexible intensified insulin therapy with syringes or pen (MDI) or with an insulin pump (CSII) is regarded as the gold standard of therapy for type 1 diabetes in childhood and adolescence. The basis for the education is differential substitution of basal and bolus insulin and flexible meals according to carbohydrate counting. The general recommendations for healthy nutrition are an essential part of the training. The overreaching goal is a life that should differ as little as possible from that of children without diabetes.
In addition to social assistance for kindergartens and school days, technological advances are also available for therapeutic support. Subcutaneous glucose measurement procedures provide comprehensive information, which, together with integration into insulin pumps, also provide a protective function against hypoglycaemia, in addition to alarms concerning out-of-target glucose values. New insulins can help to further improve achieving the therapeutic goals.
For treatment of children and adolescents in diabetes centres, in addition to paediatric expertise, clear concepts for education, individualized therapeutic regimens and diabetes management plans for acute situations should be available. These plans need to consider the needs of the family, as well as the social and cultural context of the individual child.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
TB received honoraria and research support from Medtronic, Ypsomed, DexCom travel support Novo Nordisk.
OK received honoraria and research support from Abbott, Eli Lilly, Medtronic, Novo Nordisk, and Sanofi, and is a shareholder of DreaMed Ltd.
TD received honoraria and research support from Abbott, Bayer, BMS/ AstraZeneca, Boehringer Ingelheim, DexCom, Eli Lilly, Medtronic, Novo Nordisk, Sanofi and Roche, and is a shareholder of DreaMed Ltd.
