Abstract
Objective:
The objective of this study was to raise awareness of hypersensitivity reactions to human insulin analogs (HIAs) in insulin-naïve patients and encourage consistent and detailed reporting of HIA reactions.
Data sources:
A search of PubMed, MEDLINE and International Pharmaceutical Abstracts using the terms ‘insulin’ and ‘hypersensitivity’ was completed with English language, humans, and publication date after 1 January 1990 as limits.
Study selection and data extraction:
The initial search identified 598 articles. These titles and abstracts were reviewed for relevance (e.g. mention of HIA) resulting in the exclusion of 477 articles. The full texts of the remaining titles were evaluated in addition to each article’s references to identify additional reports meeting criteria (
Data synthesis:
Evidence supporting hypersensitivity reactions to HIAs was variable, potentially due to a variety of causes (e.g. difficulty identifying a true case of hypersensitivity reaction to HIAs). Inconsistencies were noted for the identification, confirmatory testing, management, and reporting of these reactions. Management strategies included use of insulin desensitization protocols, antihistamines, steroids, immunosuppressant/immunomodulator therapies, conversion to noninsulin therapies, and pancreas transplantation.
Conclusions:
Complete and consistent identification, evaluation, management, and reporting of these reactions is essential. Specific aspects of the patient’s history should be reported, including previous insulin exposure, the specific HIAs used, duration of use prior to the reaction, a clear timeline of the reaction, and discussion of precipitating events or confounding factors.
Background
Prior to the emergence of recombinant human insulin (regular) and human insulin analogs (HIAs), hypersensitivity reactions to insulin were commonly seen in clinical practice. Reactions occurred in as many as 50% of patients treated with insulin.1,2 The higher percentage of reactions was predominantly due to the purification process initially utilized during the manufacturing of bovine and porcine insulins. 1 Insulin manufacturing processes evolved over time, resulting in more purified animal insulins, but reactions persisted and the desire for an insulin that mimicked natural physiologic insulin release remained.1,3 Eventually, technological advancements led to the development of regular insulin, and ultimately, HIAs.1,4 These innovations in insulin therapy decreased the incidence of hypersensitivity reactions, with the current reported incidence as low as under 0.1% with HIAs, which closely resembles what was reported in initial clinical trials.2,5–7
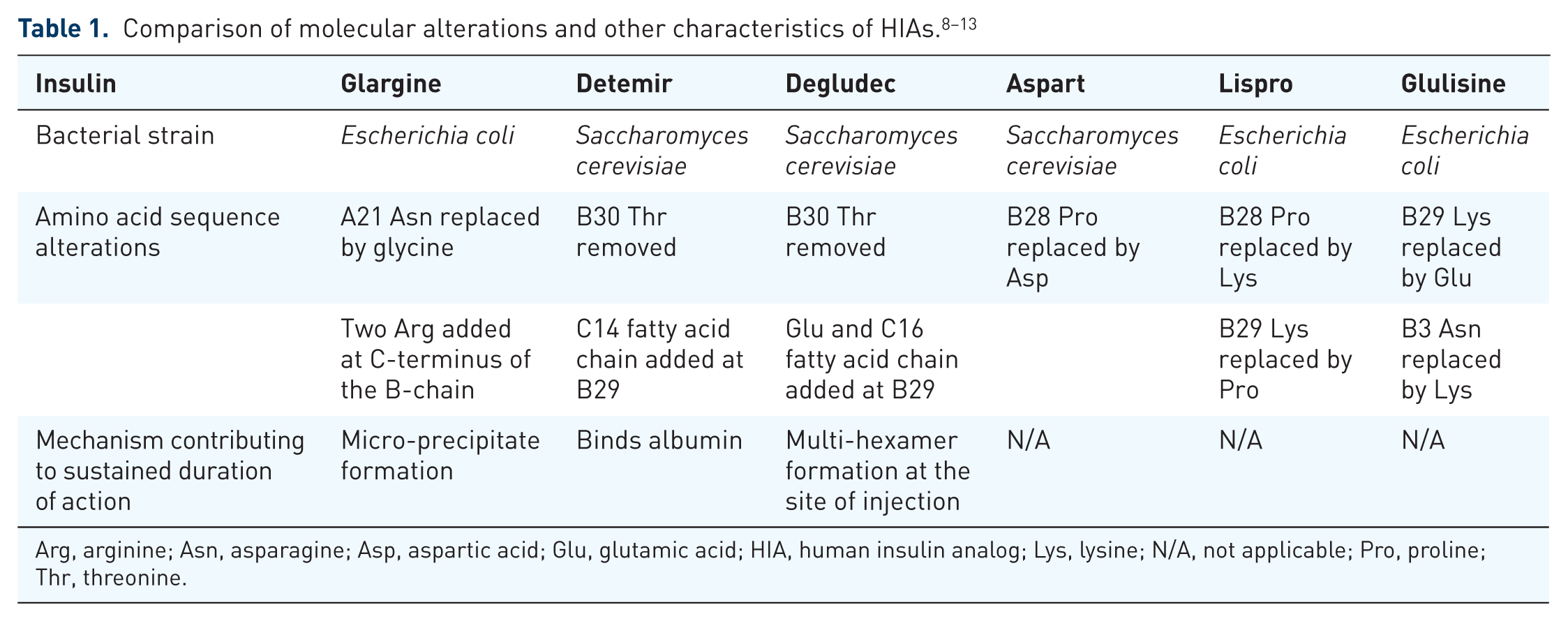
In addition, HIAs closely mimic the structure of endogenous insulin with only slight modifications that alter the pharmacokinetic properties (Table 1). In current practice, HIAs are available as basal insulin (i.e. glargine, detemir, degludec) and mealtime insulin (i.e. aspart, lispro, glulisine). Due to these pharmacokinetic properties, HIAs are generally preferred over neutral protamine-Hagedorn (NPH) or regular insulin, and therefore are the focus of this review.
Although changes have been made to the manufacturing process of insulin, several of the preservatives and other excipients used to manufacture insulin have remained the same (e.g. meta-cresol, zinc, glycerin; Table 2). There are various reports in the literature describing reactions that have been attributed to excipients within insulin (i.e. protamine,14–19 meta-cresol1,14,15,17,20–23 and zinc14–19,23,24). In addition, the bacteria utilized during the manufacturing process has been speculated to act as a potential antigen. 25 Due to the presence of case reports describing hypersensitivity reactions to specific excipients, clinicians may feel inclined to attribute hypersensitivity reactions from HIAs to these components, rather than considering the possibility that it may be the actual HIA itself.17,25
Prior insulin exposure warrants consideration when discussing insulin hypersensitivity reactions as this may increase a patient’s risk for developing a hypersensitivity reaction to HIAs.14,28 The higher immunogenicity of older insulins and the potential for the development of higher antibody levels with long-term insulin use may affect tolerability of HIAs. 29 Therefore, to fully appreciate the true incidence of HIA hypersensitivity reactions, a focus on evaluating documented reactions in insulin-naïve patients is necessary.
Hypersensitivity reactions are classified into four different types. Anaphylaxis is a type one reaction that is immunoglobulin (Ig) E mediated and is associated with a rapid onset. Type two reactions are cytotoxic in nature, occur minutes to hours after exposure and are IgG and IgM mediated. Immune complex reactions (type three) are also IgG and IgM mediated, but occur after several hours. The fourth type of reaction is a delayed reaction, occurring days after exposure, and is T-cell mediated. 30 Reactions attributed to insulin vary, but tend to be classified as type one (anaphylaxis, wheal and flare), type three (erythema, edema), or type four (erythema, induration), with type one being the most common.4,17,25,30,31 Though these reactions are rare as previously stated, the implications are significant for the subset of patients requiring insulin for survival.
To assist in the diagnosis of a hypersensitivity reaction, confirmatory testing may be undertaken. Skin prick testing (SPT) may be the preferred method of assessing any IgE-mediated reaction as it is quick (results within 15–20 min), cost effective and better tolerated by patients, yet may be insufficient to correctly diagnose an insulin allergy.32,33 In addition, SPT may not be accurate in other situations, such as patients taking medications that may affect SPT results (e.g. antihistamines), negative SPT results despite a clear clinical history regarding the allergy, or patients with significant dermatologic issues that affect the ability to perform the test. 33 Intradermal testing (IDT) has been proposed as a more accurate method for correctly identifying an insulin hypersensitivity reaction, 32 with the drawbacks of being more invasive, and carrying a higher risk of an anaphylactic reaction if a severe allergy is present. 33 Allergen-specific IgE testing is also a potential option, but again, is more invasive and time consuming. 33 Delayed, cutaneous reactions may be best evaluated using a patch test34,35 and a skin biopsy may assist in determining the type of cutaneous reaction. 35 If possible, rechallenging with the offending agent may also add in confirming a hypersensitivity. 36
The objective of this systematic review is to raise awareness of hypersensitivity reactions to HIAs in insulin-naïve patients, by summarizing the available literature regarding HIA-induced hypersensitivity in insulin-naïve patients due to the increased utilization of HIAs as initial insulin therapy. A secondary objective is to encourage reporting of these events and provide an overview of key information that should be collected regarding a potential or confirmed HIA hypersensitivity reaction.
Data sources
A comprehensive literature search was conducted using PubMed, MEDLINE and International Pharmaceutical Abstracts using the terms ‘insulin’ and ‘hypersensitivity’. The search was limited to the English language, humans, and publication date after 1 January 1990. Because the first HIA, lispro, was not commercially available in the United States and Europe until 1996, the year 1990 was selected to allow for available preclinical trials to be included. The literature search and evaluation were performed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) standards.
Study selection and data extraction
Literature was included for consideration if an HIA (i.e. lispro, aspart, glulisine, inhaled insulin, glargine, detemir, degludec) was contained within the title, abstract or manuscript. Failure to mention an HIA within the aforementioned areas resulted in exclusion. Using the search terms and exclusion criteria noted above, 598 articles were initially identified for consideration (after removal of duplicates). Both authors independently reviewed each title and abstract for relevance (e.g. HIA identified as offending agent) resulting in the exclusion of 447 and 30 articles, respectively. In the case of a discrepancy regarding relevance, the article was automatically included for consideration. The full texts of the remaining 121 titles were evaluated to determine final eligibility. Each of these article’s references were also reviewed to identify any additional sources not included in the original search. This resulted in identification of an additional 14 articles for consideration and subsequent title and abstract review. In circumstances where an article featured multiple case reports of insulin hypersensitivity, only the cases that involved administration of an HIA prior to the allergic reaction were included in the analysis. Due to the potential for prior insulin exposure to contribute to HIA hypersensitivity reactions,7,14 only cases where the authors explicitly stated or otherwise suggested the patient was insulin naïve were included. Cases in which an HIA was initiated at the same time or in combination with another insulin were included since the patient was still considered insulin naïve. If the specific insulin administered was not clearly stated, the article was excluded. Upon extensive review of these 135 full texts, 118 articles were excluded for not meeting the aforementioned inclusion criteria. In total, 17 articles met the prespecified inclusion criteria as shown in Figure 1.

Study selection. HIA, human insulin analog.
Literature review
Basal insulin analogs
Detemir
Published literature related to detemir hypersensitivity include both localized injection site31,37,38 and systemic 15 reactions. Of the four cases describing localized hypersensitivity reactions, all noted the presence of rash-like reactions with nonpruritic nodules several centimeters in diameter that occurred 2–12 h after injection and disappeared immediately 37 or up to 72 h later. 31 Only one article utilized confirmatory testing. 31 In this article, the two insulin-naïve patients (patients 1 and 2) did not experience immediate reactions after IDT, but did ultimately test positive for delayed hypersensitivity reactions to detemir. 31 Of note, patient 1 also reacted to multiple other types of insulin, including additional HIAs (lispro and glargine). 31 All of these cases were resolved with the discontinuation of detemir.15,31,37,38
The authors of the case describing a systemic reaction to detemir suggested the presence of a serum sickness type III reaction, 15 which consisted initially of an urticarial rash. The patient was continued on detemir and started on a 5-day prednisone taper, which resulted in temporary resolution of symptoms. However, 3 days after completing the taper, the hypersensitivity reaction returned and symptoms worsened (e.g. angioedema, fever, arthralgias). After completing a second course of prednisone with subsequent resolution of symptoms, the reaction reoccurred resulting in the discontinuation of detemir. No confirmatory testing was completed to rule out other potential causes of the hypersensitivity reaction and details regarding further management of the patient’s diabetes were not included.
Several of the authors discussed that the additive, myristic acid, may have had a potential role in the development of hypersensitivity reactions to detemir, 39 but the theory remains unproven.15,31
Glargine and degludec
The articles describing hypersensitivity reactions to glargine did not meet the criteria for inclusion in this analysis, primarily due to previous exposure to other insulins prior to glargine initiation. To date, no cases have been reported in the literature describing hypersensitivity reactions to degludec, possibly due to its relatively short existence on the market.
Bolus insulin analogs
Aspart
Case reports describing hypersensitivity reactions to aspart included localized reactions,40–42 with two cases progressing to systemic reactions.40,42 The presence of pruritus was consistent in all three cases with wheal-and-flare reactions noted in two.40,42 The reported onset of these reactions ranged from 5 to 10 min and lasted anywhere from several hours to a full day. Confirmatory testing for insulin hypersensitivity included SPT, IDT and laboratory evaluation for elevated antibody levels. The management of each case varied based on the results of these tests.
JiXiong and colleagues 41 performed SPT using aspart, regular, NPH and protaminated regular (Novolin 30R), with the only positive reaction occurring to aspart. Additionally, the patient had elevated total IgE and human insulin specific IgE. The patient’s aspart was ultimately switched to regular with complete resolution of symptoms noted a few days later.
Fujikawa and colleagues 42 utilized IDT to evaluate reactivity to various insulins. The patient had a positive reaction to aspart, lispro, glargine, detemir, regular, NPH and biphasic insulins. This patient also had elevated human insulin specific IgE in addition to insulin-binding antibodies. The reaction was not alleviated by antihistamine therapy or utilization of a subcutaneous insulin desensitization protocol, so continuous subcutaneous insulin infusion (CSII) with lispro was initiated. The reaction resolved and blood glucose control was achieved. After 1 year of CSII therapy, IgE levels had declined, and IDT demonstrated improvement in tolerance to lispro and glargine, so the patient was converted to this regimen. No further reactions occurred at 3 years of follow up and blood glucose control was maintained.
Zhang and colleagues
40
also performed IDT using lispro, regular (Humulin R; Lilly USA, Indianapolis, IN, and Novolin R; Novo Nordisk, Bagsvaerd, Denmark), aspart and glargine, resulting in positive reactions for each, in addition to pruritus with lispro. Antibody testing for serum IgE and IgG was performed, but results were negative. Therefore, the patient was desensitized with regular (Novolin R)
Two additional case reports met inclusion criteria; however, patient-specific characteristics may have contributed as confounders to the observed reactions. Mizuhashi and colleagues 43 presented a case of urticariforme erythema appearing immediately after injection of aspart and protaminated aspart, persisting for approximately 20 min. An induration of the skin at the injection site also appeared and persisted for 3–4 days. In close time proximity to this reaction, the patient was diagnosed with adenocarcinoma of the lung with elevated levels of interleukin 6 (IL-6) noted. Human insulin specific IgE and IgG anti-insulin antibodies were also elevated. The patient’s insulins were replaced with mitiglinide and voglibose without improvement in glycemic control. The patient received chemotherapy to reduce the tumor size, which subsequently resulted in decreased antibody levels. The authors postulate that the increase in serum IL-6 levels related to the adenocarcinoma resulted in a paraneoplastic syndrome causing the insulin resistance and hypersensitivity reaction in this patient, since the patient was able to maintain glycemic control on only oral antidiabetic therapy after receiving chemotherapy. Kawanami and colleagues 44 also described a unique case of insulin hypersensitivity with localized pruritic wheal-and-flare reactions occurring within 1 min of injecting aspart and NPH and lasting for hours. This patient’s case was complicated by a type B insulin resistance syndrome, resulting in the presence of anti-insulin receptor antibodies. Additionally, a skin biopsy was performed which revealed changes indicative of a severe insulin allergy. Glycemic control was unsuccessful with a combination of multiple oral-based regimens. The patient was ultimately treated with a 3-day burst of intravenous methylprednisolone followed by oral prednisolone for 18 months. This helped suppress the insulin resistance syndrome enough for glycemic control to be achieved solely using a combination of gliclazide and liraglutide.
Lispro
A total of six cases of hypersensitivity reactions to lispro met the inclusion criteria.14,16,23,24,28,45 All six cases resulted in localized reactions, with three cases progressing to systemic symptoms.14,23,28 Two of the cases described unique patient case scenarios24,45 that will be reviewed separately due to alternative hypotheses regarding the potential causes of reaction. Localized reactions in the remaining four cases varied, but included typical localized reaction symptoms consistent with previously described cases (e.g. urticaria, wheal and flare, pruritus).14,16,23,28 The reported systemic reactions were generalized uritcaria, 14 pruritus on the palms and soles progressing to dyspnea, 23 and generalized uritcaria with severe bronchospasms requiring emergent treatment. 28 The reported onset for each case was immediately after or within minutes of an injection. The duration of symptoms, when reported, varied from minutes to hours. Similar to the cases with aspart, confirmatory testing included SPT, IDT, laboratory evaluation for elevated antibody levels, and patch testing. 23 The findings and treatment strategies significantly differed and will be described separately.
Barranco and colleagues 28 performed SPT, which was positive for lispro and regular. IDT was also performed for protamine, which was negative. IgE testing for regular, lispro and protaminated lispro were also positive. A desensitization protocol utilizing regular was implemented over the span of 3 days without any reactions. The patient was successfully reinitiated on lispro. However, as his lispro dose increased, he developed occasional urticaria at the injection sites. The authors ultimately added daily cetirizine to the lispro regimen, which was successful.
Andrade and colleagues 17 attempted to rechallenge with lispro prior to completion of confirmatory testing, but plaque formation occurred. SPT was performed which was positive for protaminated lispro/lispro (Humalog Mix 25; Lilly USA, Indianapolis, IN), NPH, glargine, detemir, protaminated regular/regular Humulin M3), and regular (Humulin R, Actrapid; Novo Nordisk, Bagsvaerd, Denmark). Additional immunologic testing revealed elevated IgE for regular, bovine and porcine insulins. The patient’s insulin regimen was discontinued and oral therapy was resumed with no further reactions. Due to poor glycemic control, the authors anticipated needing to perform insulin desensitization for this patient in the future.
Matheu and colleagues
23
performed SPT and IDT, which were both positive for protamine but not regular insulin. Therefore, the patient was started on regular
Castéra and colleagues
14
performed IDT with positive reactions for protamine, and porcine, regular, aspart, lispro and glargine insulins. This resulted in localized reactions for all insulins tested and laryngeal tickling for all except lispro. Insulin-specific IgE was positive, but the protamine-specific IgE was negative. A successful insulin desensitization protocol
Case reports involving lispro that introduced confounding factors beyond the insulin analog itself included a delayed reaction that the authors attributed to the preservative meta-cresol and a report of cutaneous allergy to lispro precipitated by concomitant medication use.24,45
Kim and Baraniuk
24
presented a case of a patient on lispro
Pitrola and colleagues 45 described a reaction to protaminated lispro/lispro (Humalog Mix 25) that they suggest may be precipitated by the coadministration of a 3-hydroxyl-3-methylglutaryl co-enzyme A (HMG-CoA) reductase inhibitor (statin) and an angiotensin-converting enzyme inhibitor (ACE-I). The patient began developing painful, red lesions at the site of injection 3 months after insulin initiation. Insulin therapy was changed to protaminated aspart/aspart (Novomix 30; Novo Nordisk, Bagsvaerd, Denmark), but the reactions persisted, so insulin therapy was stopped and oral antidiabetic agents were reinitiated. SPT was completed, resulting in 3–4 mm wheal formation from aspart and regular and negative reactions to glargine and detemir. Anti-insulin antibodies were low at that time. Two years later, the patient was placed on glargine after failing to achieve blood glucose control on oral therapies. One week after initiation, urticaria developed at the injection site. Total serum IgE was elevated, but allergen-specific IgE for regular, porcine and bovine insulins was low. Repeat SPT resulted in 1–2 mm wheal formations from regular, porcine and bovine insulins. The authors hypothesized that a local reaction exaggerated by mast cell instability was experienced based on the low allergen-specific IgE levels and minimal reaction on SPT. To address the issue, the patient was taken off the statin, ACE-I and insulin therapy, with the intent that insulin may be retrialed in 9–12 months if deemed appropriate. The patient received liraglutide, glimepiride and metformin for diabetes management until glycemic control was lost, at which time glimepiride was discontinued and glargine was initiated. Six weeks after glargine initiation, the patient remained reaction free without coadministration of a statin or ACE-I.
Glulisine
Few case reports have been published regarding hypersensitivity reactions with glulisine. One article met the inclusion criteria of this systematic review. Heinzerling and colleagues 16 described a case series of four patients experiencing hypersensitivity reactions associated with insulin administration. One of the four cases involved a patient who had been on glulisine for 5 months before developing urticaria, erythema, flushing and pruritis. Of note, the patient was also taking NPH, though it appears to have been started 1 month after glulisine initiation. Due to the reaction, the patient was switched from glulisine to aspart with no resolution of symptoms. Oral antihistamines were not beneficial in resolving the reaction. SPT was performed and the patient experienced positive reactions to 0.9% sodium chloride, regular insulin and meta-cresol. Insulin-specific IgE, penicilloyl V antibodies, ampicilloyl antibodies and latex-specific antibodies were all elevated, whereas insulin-specific IgG was normal. A 2-day specific immunotherapy with regular insulin was performed resulting in resolution of symptoms. The patient was ultimately able to achieve glycemic control using only oral antidiabetic medications. Of note, the patient had a variety of pre-existing hypersensitivities, including environmental factors, specific foods and penicillin.
Inhaled insulin
No cases describing hypersensitivity reactions to inhaled insulin have been reported in the literature. This is possibly due to its relatively short existence on the market.
Of the 17 cases, six reports attributed the initial reaction to lispro, five to detemir, five to aspart, and one to glulisine. The majority of the cases (88.2%) discussed within the literature occurred in patients with type 2 diabetes, with only two of the cases occurring in patients with type 1 diabetes (11.8%). Reactions occurred after a few days in one case, but were delayed for years in others. Confirmatory testing was done in 14 of the 17 cases (82%), including SPT (29%), IDT (53%), antibody testing (65%), skin biopsy (12%) and patch testing (12%). It is important to note that some cases used multiple testing methods. Management of the HIA hypersensitivity reactions most commonly included use of a desensitization strategy (35%), followed by conversion to other non-insulin therapeutic options (23.5%), conversion to an alternative insulin (12%) and then use of immunotherapy (6%). Several cases did not discuss details regarding management of the reaction (23.5%; Table 3).
Summary of literature.

Ab, antibody; ACE, angiotensin-converting enzyme; CSII, continuous subcutaneous insulin infusion; D/C, discontinue; GLP-1, glucagon-like peptide 1; HMG-CoA, 3-hydroxyl-3-methylglutaryl coenzyme A; IDT, intradermal testing; NPH, neutral protamine Hagedorn; SPT, skin prick testing.
Data synthesis
This systematic review sought to provide a synopsis of documented hypersensitivity reactions to HIAs in insulin-naïve patients in the literature to encourage reporting of these events and provide an overview of key information that should be collected when documenting a potential or confirmed HIA hypersensitivity reaction. Evidence supporting HIA hypersensitivity reactions to insulin analogs was variable, with some analogs having multiple case reports noted within the literature and others being fairly sparse or even absent. This may be attributed to a variety of causes, including some HIAs being commercially available for a longer period of time compared with others, the overlap of the continued use of ‘older’ insulin preparations (e.g. NPH, animal insulin) with the ‘newer’ HIAs (e.g. lispro, aspart), the difficulty in confirming a true case of hypersensitivity reaction to HIAs, underreporting of identified cases in the published literature, and potential publication bias.
As noted in the cases described within this review, identification and management approaches were inconsistent. This was not unexpected as certain approaches may have been warranted based on the patient’s presentation (e.g. anaphylaxis requiring immediate intervention, minor urticarial allowing more time for investigation). However, even if time was available to do extensive investigation, the lack of a unified approach for accurately identifying true insulin hypersensitivity reactions likely contributed to the inconsistencies in evaluating and reporting cases. Aspects of patient insulin administration, including technique, were specifically discussed in two case reports,37,45 but not addressed in the majority of cases. In addition, there was variable use of different testing modalities for identifying true insulin hypersensitivity reactions (e.g. SPT, IDT, antibody evaluation), which appeared to be selected based on clinician preference and experience rather than a set standard. As noted earlier, IDT has been proposed as a more accurate assessment of insulin hypersensitivity reactions, though no consensus has been met regarding the preferred method. 32 When performing confirmatory testing, reactions to common HIA excipients (Table 1) should also be assessed in order to rule out hypersensitivity to individual excipients. If a positive reaction to an excipient does occur, using an HIA that does not contain that substance, or if unavoidable, one that has a lower concentration of the excipient, should be tried as was done by Kim and Baraniuk. 24 Some authors did not perform additional tests to rule out alternative causes for the reaction, instead relying solely on the temporal relationship between the insulin analog and the reaction. Regardless, use of a validated scale would assist in identifying the probability of the reaction being a result of the HIA (e.g. Naranjo scale 36 ), similar to what was done by Wang and colleagues. 6
Numerous strategies for managing HIA hypersensitivity reactions have been utilized in the literature. One of the most common strategies is the use of insulin desensitization protocols, including regular,28,46 glargine,47–50 aspart25,50 and lispro
51
Conclusion
Based on the available literature of hypersensitivity reactions to HIAs in insulin-naïve patients, it is clear that hypersensitivity reactions occur, but it is difficult to determine the ideal treatment approach. Obtaining a detailed history regarding the reaction, ensuring appropriate administration technique, ruling out other potential causes, considering a rechallenge if appropriate, performing confirmatory testing, and using a validated tool to evaluate probability, seem both practical and ideal. Future case reports on HIA hypersensitivity reactions should explicitly state the patient’s history of any prior insulin exposure; the specific insulin associated with the reaction and its duration of use prior to the reaction; the method of insulin administration (i.e. pen, syringe); and a clear timeline of the reaction with discussion of any precipitating events or confounding factors.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.