Abstract
Sellar and parasellar masses are a common finding, and most of them are treated surgically via transsphenoidal approach. This type of surgery has revolutionized the approach to several hypothalamic-pituitary diseases and is usually effective, and well-tolerated by the patient. However, given the complex anatomy and high density of glandular, neurological and vascular structures in a confined space, transsphenoidal surgery harbors a substantial risk of complications. Hypopituitarism is one of the most frequent sequelae, with central adrenal insufficiency being the deficit that requires a timely diagnosis and treatment. The perioperative management of AI is influenced by the preoperative status of the hypothalamic–pituitary–adrenal axis. Disorders of water metabolism are another common complication, and they can span from diabetes insipidus, to the syndrome of inappropriate antidiuretic hormone secretion, up to the rare cerebral salt-wasting syndrome. These abnormalities are often transient, but require careful monitoring and management in order to avoid abrupt variations of blood sodium levels. Cerebrospinal fluid leaks, damage to neurological structures such as the optic chiasm, and vascular complications can worsen the postoperative course after transsphenoidal surgery as well.
Finally, long-term follow up after surgery varies depending on the underlying pathology, and is most challenging in patients with acromegaly and Cushing disease, in whom failure of primary pituitary surgery is a major concern. When these pituitary functioning adenomas persist or relapse after neurosurgery other treatment options are considered, including repeated surgery, radiotherapy, and medical therapy.
Keywords
Introduction
Several masses can involve the hypothalamic–pituitary region, including pituitary adenomas (accounting for the majority of cases), craniopharyngiomas, Rathke’s cleft cysts, meningiomas, other rare brain tumors, or infiltrative, infectious, or vascular disease. Pituitary and craniopharyngeal duct tumors are the second most common group of brain neoplasms in the USA (pituitary adenomas most common in adults, and craniopharyngiomas in children and adolescents), accounting for approximately 16% of all primary brain tumors, and being the most common affected sites in adolescents and young adults (about 32%). 1
While the majority of pituitary adenomas are nonsecreting, the most frequent hormone-secreting tumors are prolactinomas. While prolactinomas are usually treated medically, other secretory pituitary adenomas and nonfunctioning sellar and parasellar masses that cause mass effect symptoms are treated surgically, with a majority of cases performed via the transsphenoidal approach. Postoperative care by a multidisciplinary team including neurosurgeons, endocrinologists, and intensive care teams is a crucial component of the management. 2 In this review we will analyze the possible complications of pituitary surgery and their management, focusing on the most frequent: hypopituitarism and disorders of water metabolism. Other aspects of long-term surveillance have to be tailored according to the cause that led to pituitary surgery, and this goes beyond the aim of this review. We will focus only on the postsurgical management of acromegaly and Cushing disease (CD), as failure of primary surgery is a major clinical problem and the decision on the best approach for persistent and recurrent disease requires careful evaluation.
Hypopituitarism
One of the most common complications after pituitary surgery is hypopituitarism. 3 This can be partial or total (panhypopituitarism, where all hormonal axes are involved), affecting the anterior pituitary, the posterior pituitary, or both. Moreover, hypopituitarism can be transient or permanent. In this section anterior pituitary hormone deficiency is analyzed, whereas disorders of posterior pituitary are reported later in the manuscript (Table 1).
Management of anterior hypopituitarism after pituitary surgery.

ACTH, adrenocorticotropic hormone; AI, adrenal insufficiency; AM, ante meridiem; CD, Cushing disease; DI, diabetes insipidus; FSH, follicle-stimulating hormone; GC, glucocorticoids; GH, growth hormone; GHRH, growth-hormone-releasing hormone; IGF-1, insulin-like growth factor type 1; L-DOPA, levodopa; LH, luteinizing hormone; LT4, levothyroxine; rhGH, recombinant human growth hormone; T4, thyroxine; SHBG, sex hormone-binding globulin; TRH, thyrotropin releasing hormone; TSH, thyroid stimulating hormone (thyrotropin).
The risk of postoperative hypopituitarism varies according to case series and the etiology, ranging from 5–25% for pituitary adenomas, and its occurrence varies depending on the operating neurosurgeon’s experience.4,5 It peaks to approximately 75% for craniopharyngiomas. 6 For pituitary adenomas, in addition to experience, the size and consistency of the tumor, the extension of surgical manipulation, and surgery for recurrent disease play a role in the occurrence of hypopituitarism. 4 The rate of pituitary insufficiency is higher for patients operated for CD, and several factors can account for this observation: the long-term need for glucocorticoids (GCs) postoperatively for the onset of adrenal insufficiency (AI), a larger pituitary manipulation during surgery, and the frequent median localization of adrenocorticotropic hormone (ACTH)-secreting pituitary adenomas. Finally, endogenous hypercortisolism can cause growth hormone (GH) deficiency and central hypogonadism per se, and these deficits can last for months/years after successful treatment of CD (Table1).
Provided that the postoperative period has been uneventful and no acute complications have occurred (e.g. central AI; see below), the first evaluation for the anterior pituitary function should be performed approximately 4–6 weeks after pituitary surgery (Table 1). Thyroid axis evaluation requires measurement of free thyroxine (T4). Morning serum testosterone is measured in males, while menstrual history (and possibly estradiol measurement) is used in premenopausal women. A low or normal follicle-stimulating hormone (FSH) in a postmenopausal woman is a strong indicator of gonadotrophic dysfunction suggesting hypopituitarism. GH axis can be initially assessed by serum insulin-like growth factor (IGF)-1 level. However, unless IGF-1 is low in the setting of panhypopituitarism, the diagnosis of GH deficiency usually requires failure of at least one GH stimulation test. 3 Patients with a preoperative diagnosis of pituitary insufficiency should be reassessed to confirm the need of hormonal replacement therapy, as this may improve after adenomectomy in a significant percent of patients. Jahangiri and colleagues reported normalization of thyroid, male gonadal, female gonadal, cortisol and GH axis in 36%, 18%, 41%, 29%, and 22% of cases respectively. 7 Such recovery may take time, and it has been demonstrated to be higher at 1 year compared with 3 months after surgery. Interestingly, recovery of adrenal function seems more likely in acromegaly patients compared with nonfunctioning pituitary adenomas.8,9 The timing for further evaluations should be carried out upon clinical judgment.
Management of AI
Central AI is the most important anterior pituitary deficit that can follow pituitary surgery. It should be promptly recognized, as untreated AI can be a life-threatening condition. The status of the hypothalamic–pituitary–adrenal axis (HPAA) before pituitary surgery accounts for differences in the management of central AI perioperatively and in the long term (Table 2).10–17
Management of central AI perioperatively and after pituitary surgery.
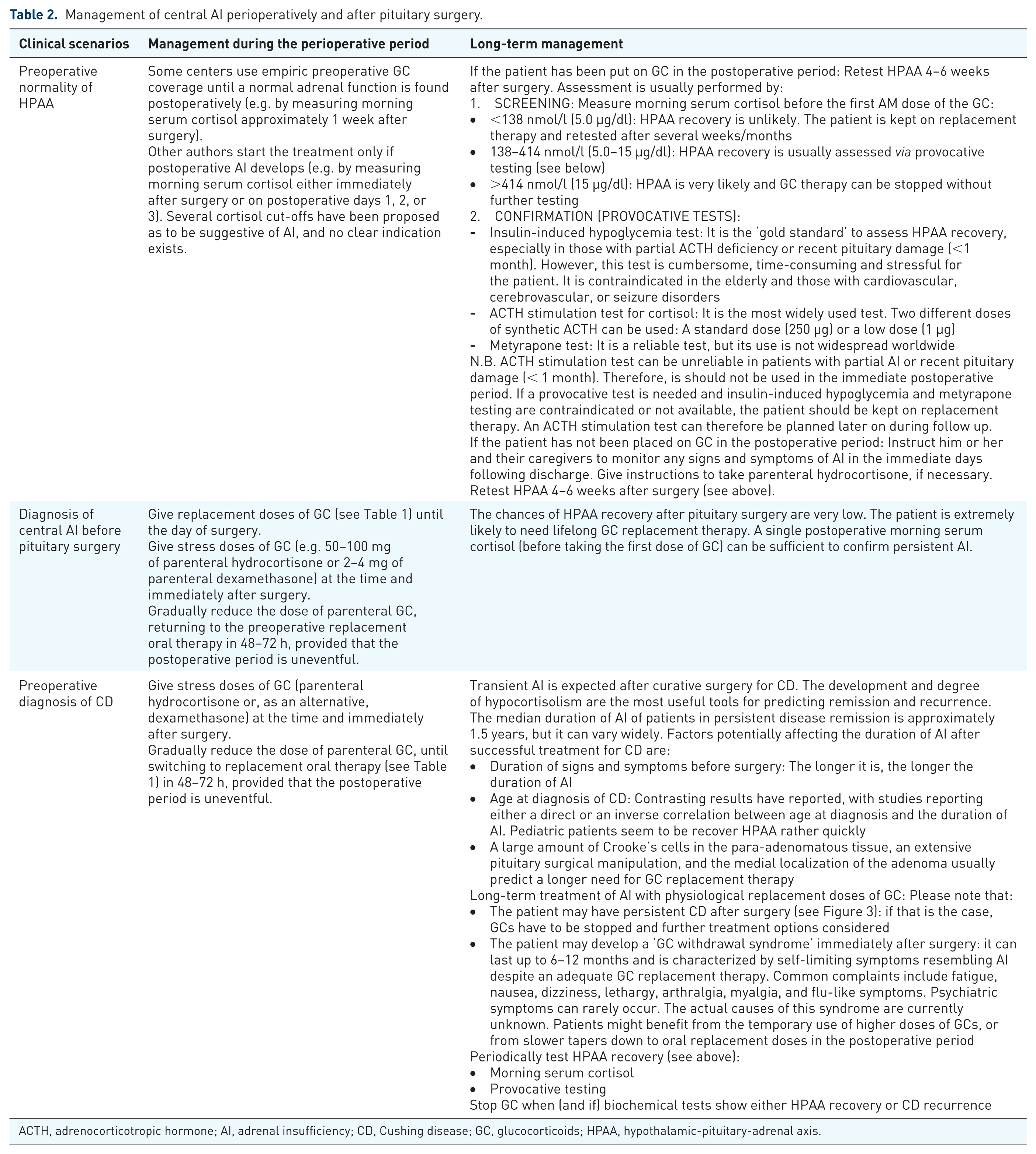
ACTH, adrenocorticotropic hormone; AI, adrenal insufficiency; CD, Cushing disease; GC, glucocorticoids; HPAA, hypothalamic-pituitary-adrenal axis.
One of the gray areas in the management of non-CD patients with normal preoperative HPAA is whether to give empiric GC coverage after surgery or to apply a steroid-sparing method, giving GC only if AI develops. To date, existing evidence has not demonstrated the superiority of one of the two strategies, and they are both used successfully in clinical practice. 2 Whatever approach is chosen, we recommend to take several factors into account: the percentage of postoperative AI at each center or each surgeon, the expertise of the team taking care of the patient after surgery, the extent of surgery, the postoperative course, and the turnaround time of serum cortisol measurements at the institution. Because postoperative AI, although relatively rare in those with an intact HPAA before surgery who are referred to experienced neurosurgeons, remains a life-threatening condition if not promptly recognized and treated, we give parenteral hydrocortisone to all patients in the first 24 h after pituitary surgery (e.g. 50 mg every 6 h), rapidly tapering to the standard oral replacement dose (15–20 mg/daily) during the following 48 h if surgery is uncomplicated. This dose is similar to what was recommended in a study looking at urinary free cortisol secretion in patients with intact HPAA (150 mg/day). 18 We then check an AM prehydrocortisone dose serum morning cortisol in an outpatient setting about 1 week after surgery. A level <138 nmol/l (5.0 µg/dl) highly likely predicts AI, whereas values >414 nmol/l (15.0 µg/dl) virtually rule out this diagnosis in all patients.2,14 However, different diagnostic cut-off points for morning serum cortisol have been proposed by various investigators, with the optimal diagnostic cortisol cut-off point remaining an issue of debate.
For low (<138 nmol/l, 5.0 µg/dl) or intermediate (138–414 nmol/l, 5.0–15.0 µg/dl) values we continue GC and retest the patients some weeks later. It is important to remember that if ACTH stimulation test is chosen for further assessment, at least 6 weeks from surgery should have passed to allow for adrenal atrophy to develop if ACTH secretion is abnormal. The standard (250 µg) and low (1 µg) dose ACTH stimulation tests have similar accuracy in the diagnosis of secondary AI, 19 with the latter not providing evident benefits 4–6 weeks after pituitary surgery. 20 Management of CD will be discussed below.
Disorders of water metabolism
Disruptions in water regulation can be linked to anatomic injury to the hypothalamus, pituitary stalk, or posterior pituitary gland during surgery. This damage alters the physiology of water metabolism controlled by the antidiuretic hormone (ADH) (see Figure 1).21–24 These disorders of water metabolism can occur due to a decrease in ADH release, leading to central diabetes insipidus (DI), or excess ADH release leading to water retention and to the syndrome of inappropriate ADH secretion (SIADH). Very rarely, a cerebral salt-wasting syndrome (CSWS), an ADH-independent condition, can occur after pituitary surgery.
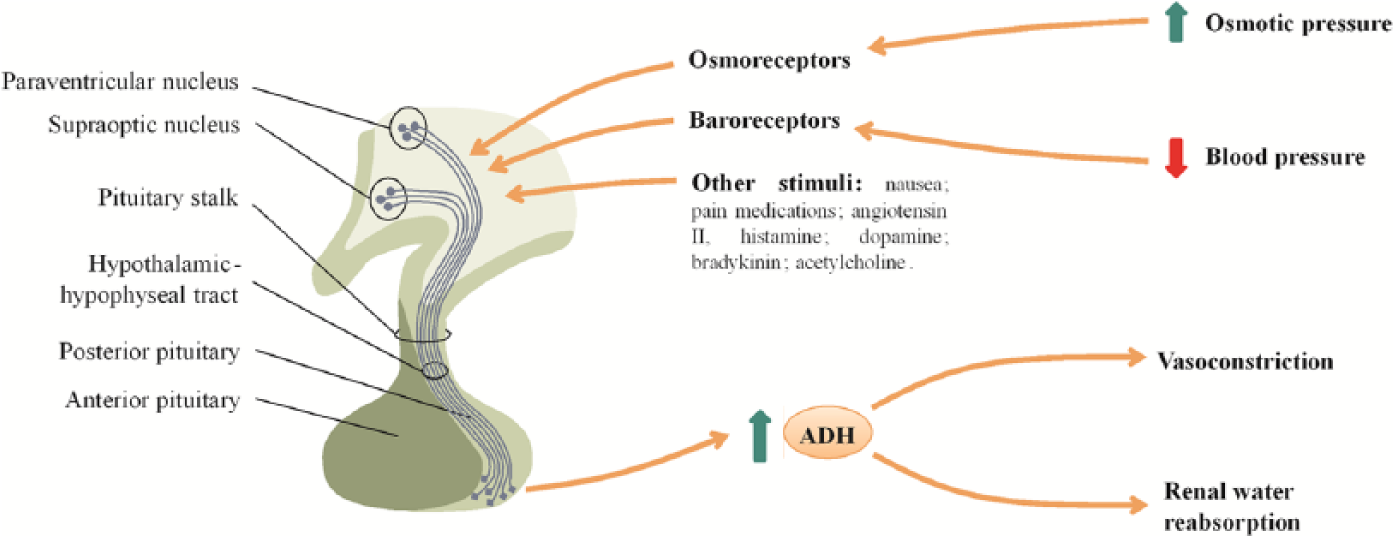
Physiology of ADH release and action. ADH is synthetized in the magnocellular neurons of the paraventricular and supraoptic nuclei, transported along the axons of hypothalamic neurons, and stored in the posterior pituitary. Its release has two major stimuli, a raise in plasma osmolality and a decrease in blood pressure and circulating blood volume. After ADH is secreted in the circulation, it acts both on kidneys (by promoting water reabsorption at the renal collecting duct) and on blood vessels (by stimulating vasoconstriction).
Central DI and SIADH
DI is defined as the concomitant presence of inappropriate hypotonic polyuria (urine output > 3 l/24 h and urine osmolality < 300 mOsm/kg) in the presence of high or normal serum sodium. 25 Other causes of postoperative polyuria should be considered, including intraoperative administration of large amounts of fluids, hyperglycemia caused or worsened by GC therapy, and, in acromegaly patients, a rapid drop in GH levels. The presence of both serum hyperosmolality and hypernatremia is highly suggestive for DI, but these laboratory alterations can be absent if the patient is conscious and has free access to water. 26 Central DI can be transient or permanent, and partial or complete, depending on the kind and extent of the damage to hypothalamic magnocellular neurons. According to most case series, DI is the most common complication after pituitary surgery. 27 It occurs in approximately 10–30% of patients undergoing pituitary surgery, but it persists long term only in 2–7%,4,28,29 with approximately 50% of patients remitting in 1 week and about 80% in 3 months. 30 The risk of permanent DI is higher in young patients, males, those with large intrasellar masses and postoperative CSF leak,26,31 in those with a preoperative diagnosis of DI,32,33 following surgery for craniopharyngioma or Rathke’s cleft cysts, 34 and after repeated pituitary surgery (e.g. for CD). 29
The onset of polyuria is usually abrupt, occurring within the first 12–24 h after surgery. Acute disorders of water metabolism can manifest in a triphasic pattern (in approximately 3% of patients): an initial polyuric phase, a subsequent antidiuretic phase (the patients can temporarily concentrate urine and SIADH and hyponatremia develops), and a final polyuric phase that is usually chronic. 35 Persistence of DI implies that at least 85–90% of hypothalamic magnocellular neurons have been damaged by surgery. Finally, the antidiuretic phase described above is sometimes isolated and represents SIADH. This temporary, late-onset (peaking 5–8 days after surgery) hyponatremia may be heightened by concomitant hypocortisolism as patients with unreplaced AI may also present with hyponatremia. 36 Figure 2 reports the pathophysiology management of the postoperative disorders of water metabolism.
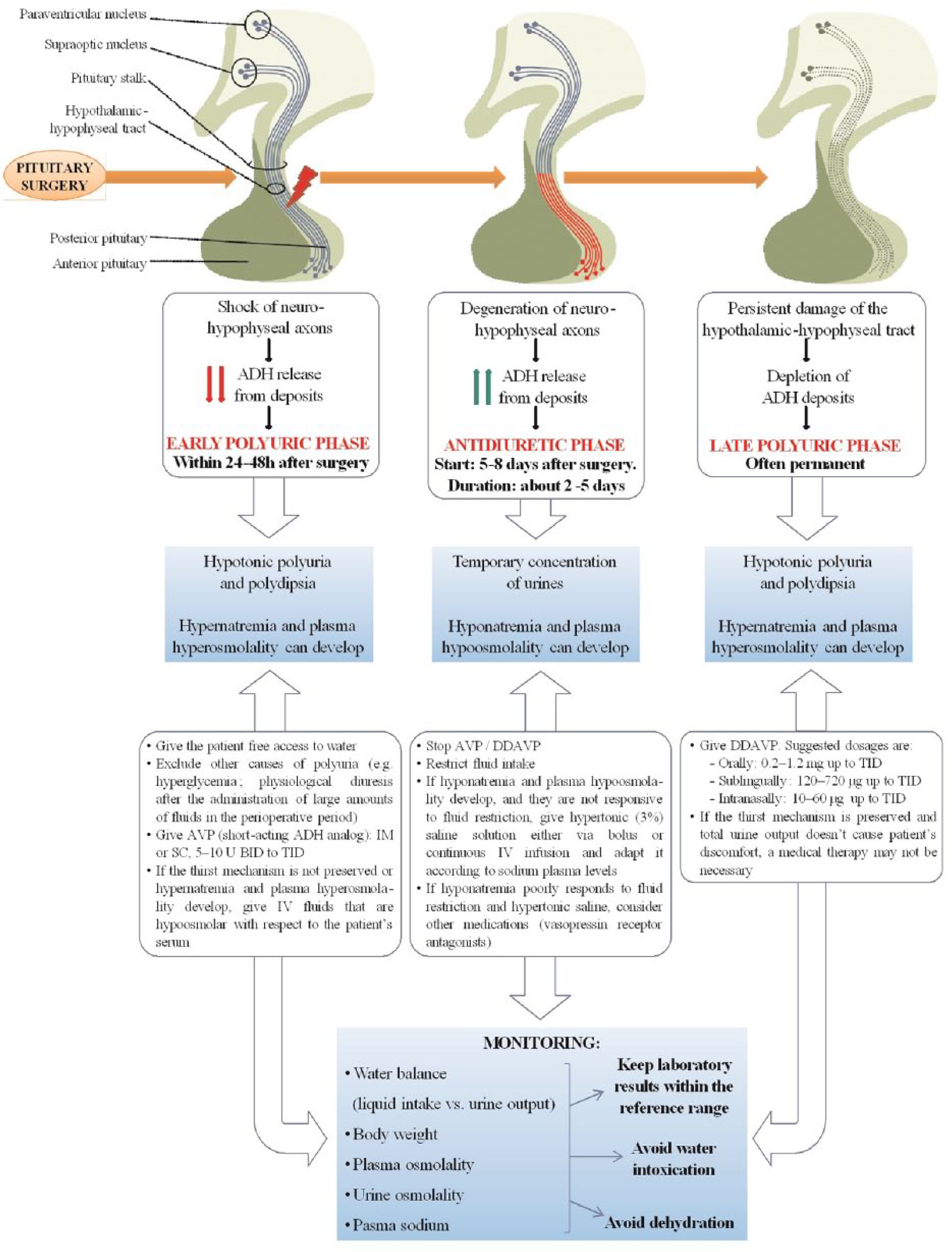
Pathophysiology and management of water metabolism imbalance after pituitary surgery.
To screen for potential development of postoperative DI and SIADH, we recommend measurement of urine output and fluid intake, urine specific gravity daily, and serum sodium every 6–12 h until discharge. For treatment of DI during the immediate postoperative period, we recommend as-needed use of short acting subcutaneous vasopressin (rather than desmopressin, DDAVP), with frequent reassessment of response and need to avoid administering an ADH when a SIADH phase is occurring. We favor the use of vasopressin at this stage because of its shorter duration of action, in case DI is transient and it reverts to SIADH. Hyponatremia usually develops between postoperative days 5–8, 2 and therefore these patients should routinely have a serum sodium level check approximately on postoperative day 6 or 7. Postoperative SIADH seems more frequent in patients with cardiac, renal or thyroid disease, older age and low body mass index, and in patients who receive a postoperative lumbar drain. 37
Given its often transient course, it is important not to over-treat early postoperative DI, to reduce the risk of precipitating hyponatremia if the second SIADH phase were to occur. Finally, patients should be instructed to only drink to thirst, and periodically (every couple of weeks) stop their DI treatment during the 6 months after surgery to verify whether they still need it.
Mild (134–125 mmol/l) hyponatremia can be treated in outpatient setting with fluid restriction and frequent sodium checks, while more severe hyponatremia (<125 mmol/l) requires hospitalization with possible short-time use of hypertonic saline or ADH receptor antagonist drugs, being careful to avoid over-correction. 2
CSWS
Very rarely, hyponatremia after pituitary surgery may be caused by CSWS, characterized by excessive natriuresis and extracellular volume depletion. While SIADH is characterized by euvolemic hyponatremia (and thus treated optimally by fluid restriction), CSWS is characterized by brain natriuretic peptide-mediated hypovolemic hyponatremia (and thus managed with hypertonic fluid administration). The differential diagnosis between SIADH and the rarer CSWS is sometimes difficult. Weight (increased/unchanged in SIADH and decreased in CSWS), serum osmolality (decreased in SIADH and increased/normal in CSWS), serum protein levels (normal/high in CSWS), and hematocrit (low/normal in SIADH and increased in CSWS) may help the differential diagnosis. 38
Other possible complications
Apart from hypopituitarism and disorders of water metabolism, damage to parasellar structures can lead to other complications after pituitary surgery, including CSF leak, epistaxis, damage to the parasellar visual system, and damage to internal carotid arteries. Mortality rate for pituitary surgery in the USA is reported between 0.2–1.2%, with lower rates in neurosurgical units with more experience.4,39
A CSF leak usually manifests as rhino-liquorrhea and headache that is typically worse when sitting up and improves lining down. It may be associated with light sensitivity, nausea, and neck stiffness. CSF leak usually occurs in 0.5–4% of patients undergoing pituitary surgery, 4 although rates up to 40% have been reported. 40 A non-adenomatous disease, the need for surgical revision, tumor margins, size and increased consistency have been associated to the risk of intra and postoperative leaks.41,42 CSF leaks have to be promptly recognized and treated, as they expose the patient to the risk of ascending infectious meningitis (Table 3).43,44
Management of postoperative CSF leaks.
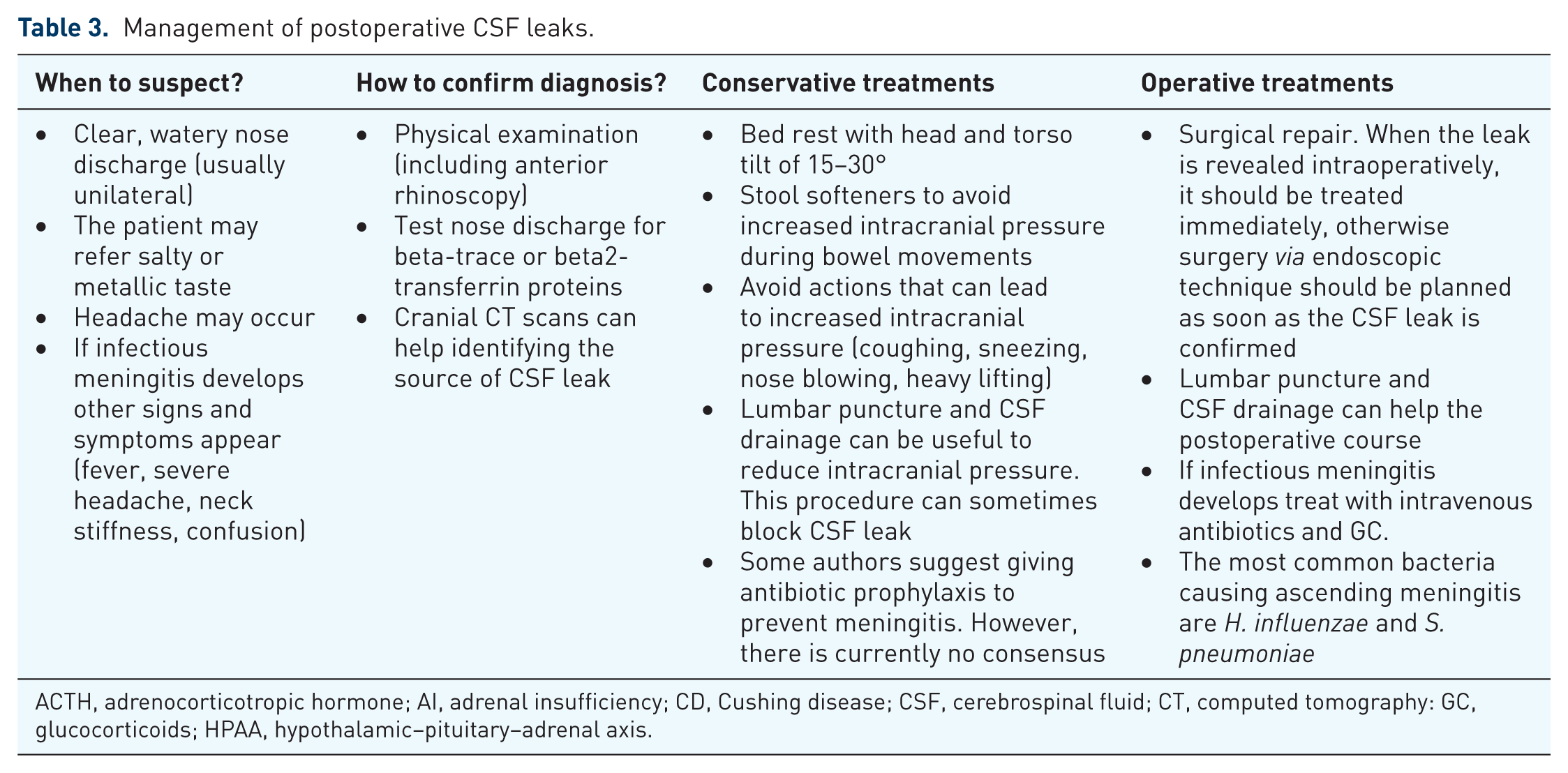
ACTH, adrenocorticotropic hormone; AI, adrenal insufficiency; CD, Cushing disease; CSF, cerebrospinal fluid; CT, computed tomography: GC, glucocorticoids; HPAA, hypothalamic–pituitary–adrenal axis.
Loss of vision or ophthalmoparesis occur in approximately 0.5–2.5% of patients undergoing pituitary surgery in the USA. 4 They can be either caused by a direct surgical damage or by postoperative compression of optic structures by hemorrhage in the surgical bed. Delayed vision loss has been described as well, and it is generally attributed to herniation of the optic chiasm within the sella (secondary empty sella). 45
Internal carotid artery injury is uncommon (0.5–1.5% of surgeries), but can lead to massive life-threatening bleeding. 4 Finally, postoperative venous thromboembolism can occur, especially in patients with CD, who harbor a higher thrombotic risk that persists for at least 3 months after surgery. 46 Therefore, postoperative thromboprophylaxis is advised by some authors in patients with CD although the optimal drug choice, dosing and durations are unknown. 47
Special situations: CD management after primary surgery
After primary surgery for CD, the initial remission rate is between 25–100%. 29 Higher remission rates are observed for magnetic resonance imaging (MRI)-visible microadenomas without cavernous sinus invasion, and in centers where 36–40 patients were operated per year. 48 Nonetheless recurrent and persistent disease is a major issue, and second-line (and sometimes third-line) treatment options have to be considered, including repeated neurosurgery, radiotherapy, medical therapy, bilateral adrenalectomy, or a combination of these (Figures 3 and 4). The recurrence rate after neurosurgery is highly variable, ranging between 0–51.2% according to case series.27,49,50 A recent meta-analysis places the risk of recurrence at 10%. 51

Cured, persistent and recurrent CD.

Management of patients with CD.
How to establish CD remission after pituitary surgery?
If patients are not administered GCs, frequent (every 6 h) serum cortisol levels should be checked starting 12 h after completion of surgery. While typically serum cortisol nadirs 24–36 h after completion of surgery, cases of ‘late cure’ are reported. 52 We start GC therapy when serum cortisol drops below 138 nmol/l (5.0 µg/dl), or patients present symptoms of AI. The development of AI after pituitary surgery is the most immediate sign that neurosurgery has achieved a total or near-total resection of corticotroph tumor cells (see Table 2). Other biochemical tests can be used to confirm remission, and the most commonly used are report in Table 4.29,53
The most common ways to assess CD remission after neurosurgery.

ACTH, adrenocorticotropic hormone; AI, adrenal insufficiency; CD, Cushing disease; DDAVP, desmopressin; GC, glucocorticoids; IV, intravenous.
Morning serum cortisol is the most widely used tool to assess CD remission, although late-night salivary cortisol and DDAVP provocative testing have recently shown promising results.54,55 Whatever test is used, it is worth noting that they lack reliable accuracy in predicting long-term CD remission. For example, 10% of patients with delayed relapse have very low postsurgical morning serum cortisol, and conversely, transient normal-to-elevated cortisol values in the immediate postoperative period are sometimes found in patients who will later go into persistent remission. 56 Therefore patients successfully treated for CD after pituitary surgery require lifelong follow up and retesting, according to clinical judgment.
Management of recurrent and persistent CD
The decision regarding the best treatment for recurrent and persistent CD is challenging. Several options are available, and the decision has to be tailored to each patient, taking into account the severity of symptoms, comorbidities, neuroimaging, surgical risks, desire for pregnancy, response and tolerance to medications. 57
Surgical reintervention (when feasible) should be the first choice in cases of recurrent or persistent CD if postoperative MRI shows a clear, resectable tumor residue.29,53 However, repeated surgery is characterized by lower success rate and a higher risk of complications (chiefly hypopituitarism, DI and CSF leaks) in comparison with primary surgery. If reintervention is not a viable option or is expected to be unsuccessful (as judged by an experienced pituitary surgeon), second-line options (medical therapy, radiotherapy or bilateral adrenalectomy) have to be taken into account.
Special situations: acromegaly management after primary surgery
Similarly to CD, higher surgical remission rates in acromegaly are observed for microadenomas versus macroadenomas (particularly if extrasellar extension in present), and in more experienced surgical hands.58,59 Cure of acromegaly can cause significant diuresis, due to the drop of blood levels of GH and IGF-1 that cause fluid retention when inappropriately high. 60 This has to be taken into account in the differential diagnosis if postoperative polyuria develops.
Although early postoperative measurement of GH level can be a good predictor of cure, no obvious cut-off level has been identified.61,62 Conversely, IGF-1 may take several months to normalize even in patients who are completely cured. 63 Serum GH and IGF-1 should be measured 12 weeks after surgery or later. 64 A postoperative 75 g oral glucose load showing GH < 0.4 ng/ml within 2 h is currently considered the best test to assess remission in patients with abnormal IGF-1 or GH after pituitary surgery. 65
Up to approximately 50% of acromegaly patients show persistence of the disease after pituitary surgery. 66 These patients require a multimodal approach to control GH excess, and possible strategies are repeated neurosurgery, medical therapy (somatostatin analogs, dopamine-receptor agonists, and GH-receptor antagonists), and radiotherapy.
Conclusion
The postoperative management of patients undergoing transsphenoidal pituitary surgery requires a multidisciplinary approach involving neurosurgeons, endocrinologists, and often intensive care teams. A preoperative hormonal assessment will help guide management decisions in the perioperative period, and intra and postoperative complications have to be promptly diagnosed and treated as to improve patient’s outcomes and prognosis. Most complications resolve in the short term, but some persist and will require lifelong surveillance and treatment. In the light of such a multifaceted picture, we strongly suggest to refer patients requiring transsphenoidal surgery to tertiary centers where neurosurgeons, endocrinologists, and neurointensivists have the expertise to deal with these diseases.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
