Abstract
With advances in surgical and medical treatment and the availability of assisted reproductive techniques, pregnancy in women with acromegaly is more frequently encountered. Diagnosis of acromegaly during pregnancy is difficult because of changes in growth hormone and insulin like growth factor-1 (IGF-1) axis secondary to placental production of growth hormone. The difficulty is compounded by the inability of routine hormone assays to detect placental growth hormone. In the majority of patients with acromegaly, pregnancy does not have an adverse effect on mother or fetus and pituitary mass does not increase in size. The level of IGF-1 usually remains stable because of the effect of estrogen causing a growth hormone resistant state. In patients with pituitary macroadenoma, the possibility of an increase in size of the pituitary mass needs to be kept in mind and more frequent monitoring is required. In case of tumor enlargement, pituitary surgery can be considered in the mid trimester. Experience with the use of medical treatment for acromegaly during pregnancy is increasing. Dopamine agonists, somatostatin analogs or growth hormone receptor antagonists have been used without any adverse consequences on mother or fetus. At present, it is advisable to stop any medical treatment after confirmation of pregnancy till more data are available on the safety of these drugs.
Keywords
Pituitary gland morphology during normal pregnancy
The shape and size of the pituitary gland varies during life. It is slightly increased around puberty and in reproductive age, when the height of pituitary reaches around 10 mm [Amar and Weiss, 2003]. During pregnancy, the size of the pituitary gland increases due to an increase in the size and number of lactotrophs secondary to the effect of estrogen [Gonzalez et al. 1988; Feldt-Rasmussen and Mathiesen, 2011]. Pregnancy has a differential effect on various anterior pituitary cells. There is a decline in gonadotrophs, whereas the number of thyrotrophs and corticotrophs does not change [Glinoer et al. 1990; Scheithauer et al. 1990; Karaca et al. 2010; Feldt-Rasmussen and Mathiesen, 2011]. The somatotrophs are suppressed because of high insulin like growth factor-1 (IGF-1) secondary to increased production of growth hormone (GH) from placenta and IGF-1 from maternal side [Eriksson et al. 1989].
Growth hormone, IGF axis during normal pregnancy
During early pregnancy, pituitary GH (also known as GH1) increases because of the stimulatory effect of estrogen with a significant contribution from relaxin (secreted by the corpus luteum of pregnancy) [Emmi et al.1991]. However, after the 25th week of pregnancy, GH1 decreases. Placental GH (also known as GH2 or GHV) secreted from the syncytiotrophoblast starts appearing in maternal circulation at 10 weeks and peaks at 25–27 weeks to a level of 10-20 mg/l [Frankenne et al.1988; Karaca et al. 2010]. Unlike GH1, it is produced in a nonpulsatile fashion. It is not released in response to growth hormone releasing hormone (GHRH) and is not detected by routine radioimmunological assay (RIA) or immunoradiometric assay (IRMA) assay. However, it is inhibited by glucose [Braunstein, 2011]. In addition, the response of GH1 and GH2 to insulin induced hypoglycemia and arginine infusion differ. There is enhanced GH response in first half of pregnancy (because of GH1) and decreased response in second half (because of GH2) [Foyouzi et al. 2004]. Table 1 summarizes the salient features of GH from pituitary and placental origin.
Summary of characteristics of pituitary and placental growth hormone.

GH, growth hormone; GHRH, growth hormone releasing hormone; IGF-1, insulin like growth factor-1.
During early pregnancy, production of IGF-1 initially decreases because of an intact feedback system. The IGF-1 level is maintained in the normal range or is increased [Schmid et al. 2001; Dias et al. 2014]. The maternal IGF-1 level is increased during the second half of pregnancy due to effects of GH2 and human placental lactogen (HPL); a high IGF-1 concentration results in suppression of maternal GH secretion [Handwerger and Brar, 1992; Fuglsang and Ovesen, 2006].
Pregnancy and acromegaly
Growth hormone producing tumors are commonly associated with infertility because of effect of tumor mass on gonadotropins and possible associated hyperprolactinemia causing anovulation [Kaltsas et al. 2007]. As acromegaly is being diagnosed in young women, the desire for pregnancy after surgical or medical treatment is increasing, more so because of improvement in in vitro fertilization techniques. Hence an updated understanding of the effects of pregnancy on maternal physiology and tumor size, and vice versa is important.
Effect of pregnancy on maternal hormonal levels in acromegaly
During early pregnancy, there is hepatic resistance to action of GH, because of high concentration of estrogen [Valassi, 2013]; subsequently, production of IGF-1 decreases and there is some improvement in the clinical features of acromegaly during first trimester [Leung et al. 2004]. During the second trimester, placental GH production results in an increase in IGF-1 generation. The pituitary gland in pregnant women seems to be responding to negative feedback because of increased IGF-1 production, secondary to increased GH production from the placenta [Caron et al. 2010; Caron, 2011; Wiesli et al. 2006].
In a retrospective analysis of data of 13 new cases of acromegaly, IGF-1 levels were reduced, stable or mildly increased during pregnancy [S Cheng et al. 2012]. In another multicenter study of 59 pregnancies, IGF-1 levels were significantly less than pre conception levels. Levels of GH remained stable [Caron et al. 1995]. In a prospective study of acromegaly and pregnancy involving eight cases, where GH was measured by interference-free immunofluorometric assay and IGF-1 by IMMULITE 2000 assay, mean IGF-1 levels before and after pregnancy were similar. The paradox of stable IGF-1 in late pregnancy despite high placental GH is explained by a process of blunted maximum response of hepatic IGF-1 production. In the same study, pituitary GH levels as measured by immunofluorometric assay did not change [Dias et al. 2014]. It is believed that pregnancy does not stimulate the secretion of GH from GH secreting adenoma.
Effect of pregnancy on growth hormone producing pituitary adenoma
Normally, the pituitary gland enlarges up to 2–3 fold during pregnancy. The increase in gland size is mainly because of hypertrophy and hyperplasia of lactotrophs secondary to high maternal estrogen level [Gonzalez et al. 1988; Valassi, 2013]. Growth hormone producing pituitary adenoma may theoretically increase in size because of the effect of stimulating hormones, hemorrhage, infarction or discontinuation of previous medical treatment [Caron, 2011]. In one large study, magnetic resonance imaging (MRI) was performed in 27 patients with acromegaly within 6 months of delivery. Tumor size increased only in three patients, who needed either bromocriptine or surgery. In 22 cases, the tumor was stable and in 2 patients its size decreased [Caron et al. 2010]. In another series of 13 patients, none was detected to have tumor growth, though one patient required medical treatment during pregnancy [S Cheng et al. 2012]. In a recent prospective study of eight patients, none developed pituitary enlargement or required treatment during pregnancy [Dias et al. 2014]. Table 2 summarizes the case series of patients of acromegaly and pregnancy published in past 15 years.
Review of maternal and fetal outcome in reported patients with acromegaly and pregnancy.
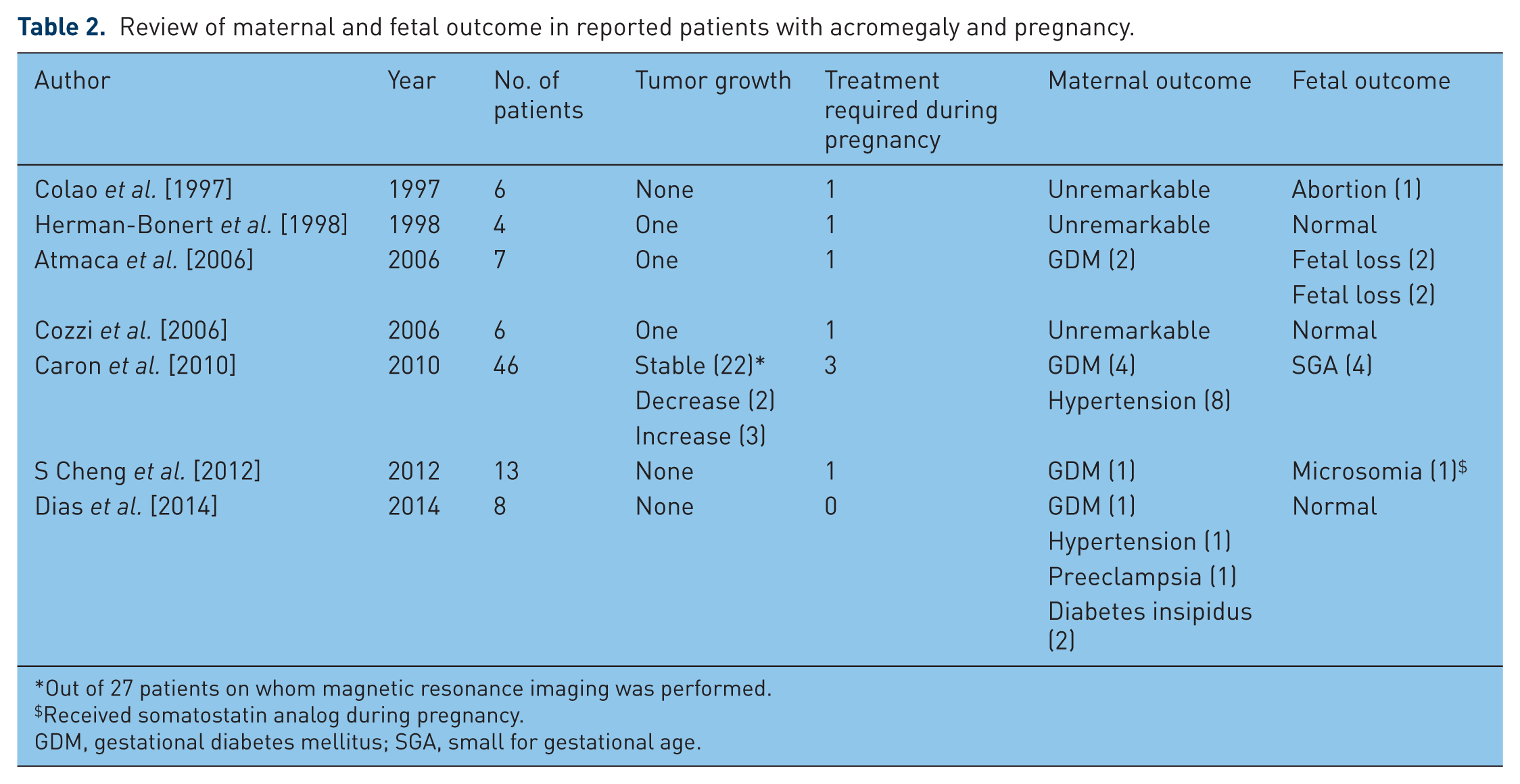
Out of 27 patients on whom magnetic resonance imaging was performed.
Received somatostatin analog during pregnancy.
GDM, gestational diabetes mellitus; SGA, small for gestational age.
From the results of published studies it can be concluded that patients with microadenoma (tumor size <10 mm) can be followed by trimester intervals for any headache and visual symptoms because the tumor usually does not increase in size, where as those with tumor size >10mm and medical treatment for <1 year before conception need to be followed monthly for clinical signs of tumor syndrome such as headache and visual defects. In case of tumor enlargement, MRI without contrast can be performed and medical treatment in form of dopamine agonists (bromocriptine or cabergoline) instituted. In case of medical treatment failure, surgery can be performed after the first trimester. These women should be advised against breast feeding [Caron, 2011; Laway and Mir 2013].
Maternal and fetal outcome
GH being an anti-insulin hormone, possible adverse effects on blood pressure, glucose tolerance and atherosclerosis could be anticipated. However; the effects are not as adverse as expected. Only a few cases of mild gestational diabetes have been reported [Okada et al.1997; Atmaca et al. 2006; Verhaeghe, 2008]. The status of GH/IGF-1 secretion may be an important determinant of occurrence of these complications during pregnancy.
Caron and colleagues found that a slightly higher percentage of women with acromegaly had gestational diabetes and hypertension compared with age-related general pregnant women without acromegaly. Overall, 13.6% women developed hypertension as against 5–15% in controls and 6.8% developed diabetes as against <6% in controls. The occurrence of both hypertension and diabetes was related to control of GH/IGF-1 secretion before pregnancy [Caron et al. 2010; Caron, 2011]. In another recent study, only 1 out of 13 new cases developed gestational diabetes and none developed hypertension or cardiovascular diseases [S Cheng et al. 2012].
The results of prospective, multicenter study were published recently. Out of 10 pregnancies in 8 women, only 1 patient developed gestational diabetes at 26 weeks which was detected by oral glucose tolerance test, 2 patients developed diabetes insipidus which recovered after delivery and 1 developed preeclampsia [Caron et al. 2010].
Effect of drug exposure during pregnancy
Three classes of drugs (somatostatin analogs, dopamine agonists and growth hormone receptor ligands) have been used in acromegaly. The effects of early fetal exposure because of use of long-acting somatostatin (SST) analogs, dopamine agonists (bromocriptine and cabergoline) and GH receptor antagonists have been studied. Although dopamine agonists, SST analogs and GH receptor ligands cross the placenta, no major adverse effects have been reported in mother and fetus [Caron et al. 2010; Valassi, 2013]. Use of dopamine agonists during pregnancy in some cases has been associated with fetal macrosomia [Yap et al.1990; S Cheng et al. 2012]; otherwise use of these drugs throughout pregnancy has not been associated with any fetal malformation or any adverse postnatal development [Lebbe et al. 2010; Molitch, 2010]. SST analogs have been found to cross the placenta and are detected in fetal circulation and fetal biological fluids [Caron et al. 1995; Maffei et al. 2010]. Octreotide has sometimes been used during pregnancy to control headache associated with GH producing adenoma [Colao et al. 1997; Fassnacht et al. 2001; Mikhail, 2002]. Children born to mothers who were exposed to SST analogs during pregnancy have been shown to have a shorter length, though in the normal range [Colao et al. 1997]. A high concentration of octreotide is maintained in maternal serum during drug treatment and transplacental passage occurs by diffusion [Caron et al. 1995]. All five types of SST receptor subtypes bind octreotide, with high affinity to SST2 and 5, and low with SST4 receptor. A high capacity and low affinity to placental cells has been described [Tsalikian et al. 1984; Caron et al. 1997]. Maffei and colleagues demonstrated a functional placental barrier to octreotide because of its low level of binding to the placenta and umbilical cord [Maffei et al. 2010]. The low affinity of SST analogs to SST receptor in placenta might explain the absence of its effect on GH production [Caron et al. 2010]. In the same study, however, a transient decrease in uterine artery blood flow and its systolic velocity was observed after injection of short-acting octreotide. This may partly explain the microsomia associated with the use of SST analogs during pregnancy [Brian et al. 2007; van der Klaauw et al. 2012]. Otherwise exposure to SST analogs during pregnancy has not been shown to be associated with malformation or adverse postnatal growth and development. Pituitary function has been documented to be normal up to 6 years of age in some studies [Mikhail et al. 2002; Boulanger et al. 2004].
Pegvisomant, a GH receptor antagonist, has not been found to be teratogenic in animal studies [Caron et al. 2010]. Before 2014, only two cases of use of pegvisomant during pregnancy had been reported [Qureshi et al. 2006; Brian et al. 2007]. Van der Lely and colleagues reported the largest number of patients who were on pegvisomant before pregnancy [van der Lely et al. 2014]. The majority of patients discontinued pegvisomant upon confirmation of pregnancy apart from three patients who continued it throughout pregnancy. A minimal transplacental passage of the drug has been documented and drug has not been detected in breast milk [Brian et al. 2007; Fleseriu, 2015]. Babies born were reported to be normal and continued to be for up to 6 months of follow up [V Cheng et al. 2012].
Conclusion and recommendations
The normal pituitary gland increases in size during pregnancy; GH and IGF-1 levels in maternal circulation are also increased secondary to increased production of placental GH. In the majority of women with pituitary microadenoma, tumor growth does not occur; also high levels of GH and IGF-1 do not have a marked adverse effect on blood pressure and glucose status in the mother. However, patients with acromegaly need to be carefully followed during pregnancy. Women taking long-acting SST analogues when not pregnant need to use a short-acting analogue before planning a pregnancy and all the drugs need to be stopped upon confirmation of pregnancy. In women with macroadenoma, visual field testing is required every month. In case of symptoms and visual field abnormalities, MRI without contrast can be performed after the first trimester. If the tumor has increased in size, dopamine agonists such as bromocriptine or cabergoline can be instituted. Use of drugs during pregnancy is only limited to controlling an expanding mass and headache [Katznelson et al. 2014]. If there is no relief, trans-sephenoidal surgery can be performed during the second trimester. In case of microadenoma, evaluation is required every 3 months instead of monthly. Women with tumor expansion during pregnancy should be advised against breast feeding.
Footnotes
Acknowledgements
The author is grateful to Mr Altaf Hussain Shah for his secretarial assistance.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
