Abstract
Objective:
Bisphosphonates are the most effective therapeutic agents in patients with Paget’s disease of bone. As a result of their inhibition of osteoclastic activity, hypocalcemia of variable frequency and severity following intravenous bisphosphonate therapy has been reported. The present study assessed the effect of physician and patient education on adequate supplementation of calcium and vitamin D to reduce the potential risk of developing hypocalcemia following infusion of 5 mg zoledronic acid.
Methods:
This was an open-label, multicenter, controlled registry trial in which patients with Paget’s disease were treated with a single intravenous infusion of zoledronic acid. Physicians were provided with educational materials focusing on optimization of calcium and vitamin D supplementation following zoledronic infusion that they used to educate their patients. The primary safety variable was the percentage of patients with serum calcium level <2.07mmol/l 9–11 days after zoledronic acid infusion.
Results:
A total of 75 patients were evaluable in the post dose hypocalcemia safety analysis. Of these, only 1 patient had treatment-emergent hypocalcemia, with a serum calcium level of 1.92 mmol/l 4 days following therapy. Hypocalcemia-related symptoms were not reported in this patient and the serum calcium returned to normal range at 2.17 mmol/l within 1 week on oral calcium supplementation.
Conclusions:
These results suggest that, with optimization of calcium and vitamin D supplementation by physician and patient education, hypocalcemia is an infrequent occurrence following zoledronic acid infusion.
Introduction
Oral and parenteral aminobisphosphonates are the most effective therapeutic agents for Paget’s disease of bone [Polyzos et al. 2011; Ralston, 2013]. As a result of their potent osteoclast inhibition, instances of asymptomatic or symptomatic hypocalcemia as a potential side effect have been reported. Mild to moderate transient asymptomatic hypocalcemia has been reported in patients with Paget’s disease of bone when treated with bisphosphonate drugs. In some cases, there have been reports of symptomatic, severe and persistent hypocalcemia, especially in patients treated with intravenous (IV) aminobisphosphonates such as pamidronate and zoledronic acid [Fenton et al. 1991; Ferraz-de-Souza et al. 2013; Gutteridge et al. 1996; Papapetrou, 2009; Polyzos et al. 2011; Ralston, 2013]. A review of the available data also suggests that subnormal serum calcium levels were observed following treatment with bisphosphonates in patients with metastatic bone disease and multiple myeloma [Breen and Shane, 2004; Mehrotra, 2009; Segal et al. 2012], osteoporosis [Black et al. 2007; Lyles et al. 2007; Reid et al. 2002; Tsourdi et al. 2011], osteogenesis imperfecta [Vuorimies et al. 2011], renal insufficiency [Polyzos et al. 2011], malignancies [Chennuru et al. 2008; Major and Coleman, 2001; Sims et al. 1998], underlying vitamin D deficiency or insufficiency [Breen and Shane, 2004; Ralston, 2013; Rosen and Brown, 2003; Segal et al. 2012] and poor calcium intake and/or calcium malabsorption [Breen and Shane, 2004].
Supplementation with calcium and vitamin D has been recommended to prevent post therapy hypocalcemia [Polyzos et al. 2011]. The present study was conducted to assess the effect of physician and patient education on adequate supplementation of calcium and vitamin D, when necessary, following infusion of 5 mg zoledronic acid to reduce the potential risk of developing hypocalcemia. The study determined the frequency of hypocalcemia following zoledronic acid therapy in patients with Paget’s disease of bone. The symptoms associated with hypocalcemia in patients were also assessed using a questionnaire.
Subjects and methods
Study design and treatment
This was an open-label, multicenter, controlled registry trial conducted in 9 centers in the USA in which patients with Paget’s disease of bone were treated with a single IV infusion of zoledronic acid 5 mg. Evaluable patients were defined as those who had baseline serum calcium level ⩾2.07 mmol/l (normal range: 2.07–2.64 mmol/l) and at least one measurement for serum calcium 9-11 days post zoledronic acid infusion. The original plan was to enroll 300 patients. However, due to slow enrollment and lower than expected incidence of hypocalcemia, the protocol was amended to enroll a maximum of 100 evaluable patients and to implement a formal stopping rule in an interim analysis at about 75 evaluable patients. This report includes the results from 75 evaluable patients for the final analysis. The physician/investigators were educated about prescribing zoledronic acid in patients with Paget’s disease of bone by providing appropriate education material from the sponsor along with the US Prescribing Information issued in 2011 for zoledronic acid (Reclast®). All patients received written and verbal education from their physicians regarding the importance of calcium and vitamin D supplementation in maintaining serum calcium levels and regarding symptoms suggestive of hypocalcemia. To reduce the risk of hypocalcemia, all patients were instructed regarding a total intake of 1500 mg of elemental calcium daily and a minimum of 800 IU vitamin D daily, particularly in the immediate 2 weeks following zoledronic acid administration. The daily calcium intake (drug and food supplement) was recorded by each patient in a diary.
The recommended dose of 5 mg/100 ml zoledronic acid was administered as an aqueous IV infusion over at least 15 minutes. The patients had no restrictions as to concomitant therapy and these were documented during the study.
Inclusion and exclusion criteria
Patients with Paget’s disease of bone were selected based on the approved indication (Reclast® US Prescribing Information). Patients with hypersensitivity to the bisphosphonate class of drugs, creatinine clearance <35 ml/min, hypocalcemia, and pregnant or lactating women were excluded from the study (Reclast® US Prescribing Information). Patient selection was not based on serum 25-hydroxy vitamin D (25-OHD) levels or vitamin D intake prior to baseline; however, the serum 25-OHD level was checked at baseline as a good clinical practice and, if necessary, patients were treated with appropriate vitamin D supplements.
Written informed consent was obtained from each patient before enrollment in the study. The study protocol was reviewed and approved by the Independent Ethics Committee or Institutional Review Board for each center. The study was conducted according to the ethical principles of the Declaration of Helsinki [ClinicalTrials.gov identifier: NCT00668200].
Safety assessment
The incidence of hypocalcemia was evaluated 9–11 days after zoledronic acid infusion. The study objective was to evaluate the effect of investigator and patient education on calcium and vitamin D supplementation in mitigating the potential for hypocalcemia following administration of zoledronic acid in patients with Paget’s disease of bone. This was assessed by comparing the incidence of hypocalcemia in this study with the lower limit of the 95% confidence interval (CI) (14.4%) for the incidence of hypocalcemia (20.4%) observed in the pivotal studies with zoledronic acid in Paget’s disease of bone [Reid et al. 2005].
The primary safety endpoint was the percentage of patients with hypocalcemia (serum calcium level <2.07 mmol/l) 9–11 days post zoledronic acid infusion. Serum calcium levels were determined at baseline and 9–11 days after drug infusion (i.e. Study Days 10–12). Any patient with an abnormal serum calcium level (<2.07 mmol/l) on Days 10–12 was required to have another serum calcium determination and to complete the hypocalcemia questionnaire on Day 30. The end of the study was defined as the day on which the last post baseline serum calcium level was measured (Days 10–12 and/or Day 30). In addition, the patients were asked to complete a standardized questionnaire on potential hypocalcemia-related symptoms prior to the infusion (Day 1) and on Days 10–12 (secondary endpoint). The questionnaire included the following symptoms (yes or no): tingling around mouth or fingers; cramps in hands, leg or feet; involuntary movements of hands, feet or facial muscles; and generalized (epileptic) seizures (Appendix Table 1). The patients were also monitored for all nonserious and serious adverse events (SAEs).
Statistical analysis
The intent-to-treat (ITT) and safety analysis populations included all subjects who received drug infusion. The number and percentage of patients with hypocalcemia 9–11 days post zoledronic acid infusion are presented using the safety population. The denominator for the percentage was the number of evaluable patients (patients whose response to a treatment can be measured) who had a baseline serum calcium level ⩾2.07 mmol/l and at least one measurement of serum calcium post zoledronic acid infusion. The 2-sided p-value for testing the null hypothesis that the event rate is 0.144 or higher was calculated based on the exact method. The O’Brien–Fleming spending function was used across the interim and final analyses to give an overall level of 0.05. The 95% CI for the rate was calculated based on the exact method (Clopper–Pearson) for descriptive purposes.
Results
There were 80 patients enrolled in the study. In addition, one patient inadvertently signed the consent form from another Paget’s disease study; this patient was excluded from the ITT population but included in the safety population. Thus, the ITT population included 80 enrolled patients; however, the safety population included 81 patients. All 81 patients received the complete 100 ml zoledronic acid 5 mg infusion and the average duration of infusion was approximately 27.8 minutes. Of those enrolled, 77 completed the study. Three patients discontinued their participation in the study; two patients withdrew their consent and one patient was lost to follow up. Among the enrolled patients, 75 patients met the inclusion criteria for the analysis of incidence of hypocalcemia. Six patients were excluded from the analysis, as one patient had a low (exclusionary) serum calcium level drawn at baseline and the other five had missing serum calcium data either at baseline or post baseline. At baseline, the mean ± standard deviation (SD) serum calcium level was 2.40 ± 0.15 mmol/l. Other demographic and baseline characteristics of the enrolled patients are shown in Table 1.
Baseline demographics (ITT population).

Vitamin D deficiency was determined based on the active medical history of patients at enrolment.
ITT, intent-to-treat; N, number of subjects in the analysis population; n, number of subjects meeting the criterion (for categorical variables) or the number of subjects with a measurement (for continuous variables); SD, standard deviation.
Concomitant medications
Prior to start of the study, 82.5% (n = 66) of the patients took various forms of calcium. Most common was calcium with vitamin D (31.3%). The other concomitant medications were ergocalciferol (51.3%), multivitamins (33.0%), acetylsalicylic acid (30.0%), simvastatin (26.3%) and omeprazole (25.0%). Prior use of bisphosphonates included zoledronic acid (11.3%), pamidronate (5.0%), alendronate (3.8%) and etidronate (1.3%). After enrollment into the study, 95% (n = 76) of the patients took various forms of calcium; commonest were calcium citrate (33.8%) and calcium with vitamin D (33.8%). The other concomitant medications included ergocalciferol (47.5%), paracetamol (38.8%), multivitamins (33.8%), acetylsalicylic acid (31.3%), simvastatin (26.3%) and omeprazole (25.0%).
Calcium and vitamin D intake
The average calcium intake in patients between baseline and Days 10–12 was 1706.7 mg/day. Average calcium intake was >1500 mg/day in 52 (64.2%) patients, 1000–1500 mg/day in 21 (25.9%) patients and <1000 mg/day in 4 (4.9%) patients during the study. Exposure to vitamin D was not quantified due to the possibility of the patients receiving monthly injections of vitamin D prior to, or in addition to, or in place of oral vitamin D supplementation. The combined calcium intake of the patients from drug and food supplement is shown in Table 2.
Daily calcium (drug and food) intake in the study (safety population).

One patient has taken excess amount of calcium (5600 mg/day). The serum calcium level (2.35 mmol/l) was within the normal range on Days 10–12 and no adverse event was reported.
Data represent mean ± standard deviation (median);
N, number of subjects in the analysis population; n, number of subjects with a measurement.
Safety analysis
Patients who had baseline serum calcium of at least 2.07 mmol/l and at least one serum calcium measurement 9–11 days following zoledronic acid infusion were included in the analysis of incidence of hypocalcemia. Of the safety analysis population of 81 patients, 75 met the inclusion criteria for evaluation for hypocalcemia. Among these patients, treatment-emergent hypocalcemia (serum calcium <2.07 mmol/l, 1.3%, 95% CI: 0–7.2%) was observed in only 1 patient. Based on the physician’s judgment, serum calcium was analyzed in this patient on Day 4. The patient had low calcium levels (1.92 mmol/l) on Day 4. Although this patient was taking the recommended amount of calcium (1500 mg, daily), the physician added calcium citrate 500 mg + vitamin D 400 IU combination tablets thrice daily to the regimen. Subsequently, the patient’s calcium levels were within the normal range at 2.17 mmol/l on Day 11 (normal range: 2.07–2.64 mmol/l). No hypocalcemia-related symptoms were reported in this patient. The event was reported as an adverse event (AE) of hypocalcemia with moderate severity and suspected to be related to the study drug. No other instances of hypocalcemia were reported. The statistical hypothesis that the incidence rate of hypocalcemia was 14.4% or higher was rejected (2-sided p = 0.0002). Zoledronic acid had minimal effects on the serum calcium levels at post baseline measurements including Days 10–12, Day 30 and end of study (Table 3).
Change from baseline in serum calcium (mmol/l) (safety population).

Data represent mean ± standard deviation (median).
End of study: last post baseline serum calcium value (Days 10–12 and/or Day 30);
N, number of subjects in the analysis population; n, number of subjects with measurements within the visit window.
Adverse events
Approximately 50% of the patient population reported AEs during the study. Most of the common AEs reported after zoledronic acid infusion were associated with well-described post dose symptoms which included chills (n = 10, 12.3%), pain (n = 9, 11.1%), acute phase reaction (n = 6, 7.4%), malaise (n = 6, 7.4%) and pyrexia (n = 5, 6.2%). No patients died or discontinued participation due to AEs during the study. One patient reported an SAE of metastatic neoplasm which was not suspected to be related to study medication. A summary of AEs is presented in Table 4.
Summary of common adverse events (⩾5%) (safety population).
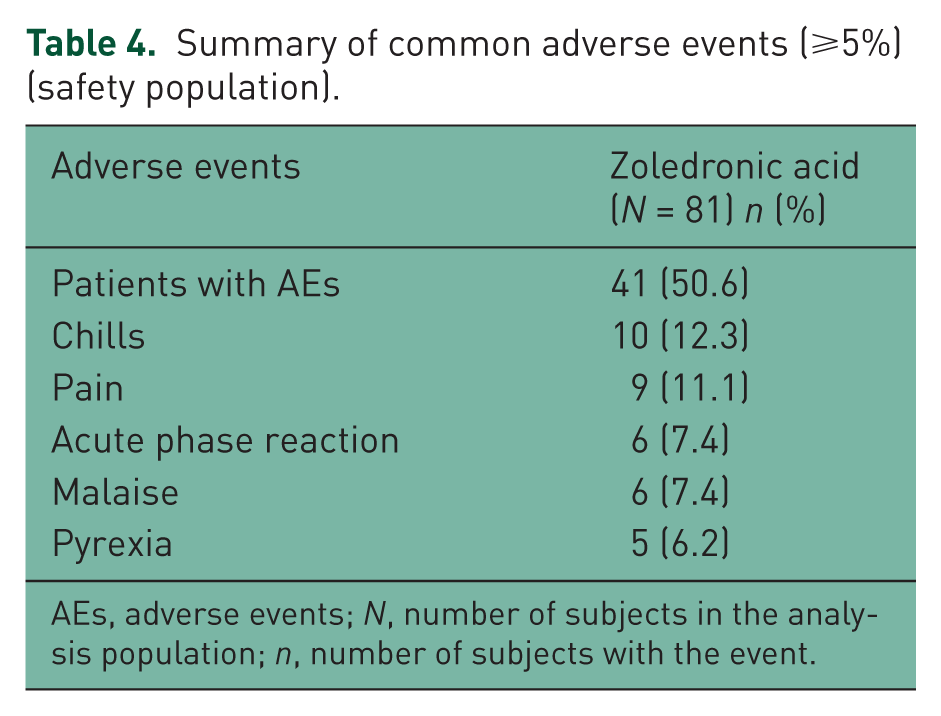
AEs, adverse events; N, number of subjects in the analysis population; n, number of subjects with the event.
Based on the hypocalcemia questionnaire, a total of eight patients reported newly occurring post baseline symptoms suggestive of hypocalcemia. In three of these patients, AEs were reported in association with the symptoms (e.g. tingling around mouth, involuntary movements of hands, tingling in fingers). In all three cases, the AEs were mild and not suspected by the investigator to be related to the study medication or serum calcium levels. For the remaining five patients, the physician did not report AEs in association with hypocalcemia symptoms. None of the 8 patients had serum calcium levels below the reference range (2.07–2.64 mmol/l) (Appendix Table 2).
Discussion
Clinically significant hypocalcemia following bisphosphonate therapy has been considered by some authors to be a rarity or an uncommon occurrence, and by others a frequent or expected finding [Fenton et al. 1991; Ferraz-de-Souza et al. 2013; Gutteridge et al. 1996; Papapetrou, 2009; Polyzos et al. 2011]. Hypocalcemia has been reported more frequently following IV rather than oral bisphosphonate therapy. Hypocalcemia following bisphosphonate therapy has been reported in patients with Paget’s disease of bone [Papapetrou, 2009; Polyzos et al. 2011; Reid et al. 2005], juvenile Paget’s disease of bone [Polyzos et al. 2010], osteoporosis [Black et al. 2007; Lyles et al. 2007; Reid et al. 2002; Tsourdi et al. 2011], malignancies with metastatic bone disease and multiple myeloma [Mehrotra, 2009; Segal et al. 2012], osteogenesis imperfecta [Vuorimies et al. 2011], and in patients with bone loss following liver transplantation [Crawford et al. 2006]. In many instances, patients have had comorbid conditions which can increase the risk of hypocalcemia including pre-existing hypoparathyroidism, hypomagnesaemia, renal insufficiency, vitamin D deficiency or insufficiency, gastrointestinal malabsorption, and repeated bisphosphonate infusions in patients with malignancy [Breen and Shane, 2004; Chennuru et al. 2008; Peter et al. 2004; Polyzos et al. 2011; Rosen and Brown, 2003]. In a number of reports, suboptimal calcium intake and/or vitamin D supplementation appear to have been important underlying risk factors [Breen and Shane, 2004; Polyzos et al. 2011; Segal et al. 2012; Whitson et al. 2006].
In an early study of Paget’s disease of bone, asymptomatic low calcium levels occurred in all 26 patients who were treated with IV pamidronate [Fenton et al. 1991]. Similar observations were subsequently reported by Gutteridge and colleagues [Gutteridge et al. 1996], in which mean serum ionized calcium levels fell with a nadir between 4 and 10 days after IV pamidronate administration with mean levels returning to pre therapy values within 6–12 weeks in 71 patients with Paget’s disease of bone. More striking changes in the serum calcium level occurred in patients with higher levels of disease activity. In a report based on 2 pivotal trials comparing a single infusion of zoledronic acid with 60 days of oral risedronate therapy in Paget’s disease of bone [Reid et al. 2005], hypocalcemia was reported in 8 (out of 177) patients in the zoledronic acid group during study days 3 through 13. Two of these patients who were mildly symptomatic had not taken their calcium and vitamin D supplements. In addition, one patient in the risedronate group had severe symptomatic hypocalcemia requiring IV calcium. Avramidis and colleagues reported a significant but transient decrease in mean serum calcium of 10% in 9 patients with Paget’s disease of bone all of whom were asymptomatic following zoledronic acid therapy [Avramidis et al. 2008].
In the present study of 75 patients with Paget’s disease of bone who were treated with zoledronic acid, 1 patient had asymptomatic hypocalcemia with a subnormal serum calcium level of 1.92 mmol/l on the fourth day postinfusion. Calcium and vitamin D supplement (combination tablet of calcium citrate + vitamin D 400 IU) thrice daily was added to the patient’s regimen and, a week later, serum calcium level was normal (2.17 mmol/l). Based on the hypocalcemia questionnaire results, newly occurring post baseline symptoms suggestive of hypocalcemia such as tingling about the mouth or fingers and involuntary hand movements were reported in eight patients; however, serum calcium levels in all these eight cases were normal.
Other AEs following zoledronic acid treatment included the well-described acute phase reaction with symptoms such as malaise, pyrexia, chills and pain.
There are two main limitations of this study. Being an open-label single arm study, the effect of investigator and patient education on calcium and vitamin D supplementation in mitigating the potential for hypocalcemia following administration of zoledronic acid in patients with Paget’s disease of bone was evaluated using historical controls. This study was originally designed to enroll 300 patients. However, due to slow enrollment and lower than expected incidence of hypocalcemia, target enrollment was reduced to 100 patients and the study was stopped early when 75 patients met the inclusion criteria. Despite these limitations, the present study results provide support for the validity of the recommendations that were evaluated.
Conclusion
The present study shows that, with physician and patient education on adequate calcium and vitamin D intake, the incidence of hypocalcemia following zoledronic acid infusion in this trial was lower than that reported in other clinical trials of bisphosphonate therapy in Paget’s disease of bone. The importance of adequate clinical and laboratory assessment of patients prior to zoledronic acid therapy cannot be overstated nor can the necessity to optimize total daily elemental calcium intake (approximately 1500 mg) and appropriate vitamin D supplement to ensure a serum 25OHD level of at least 30 ng/ml.
Footnotes
Appendix
Change in serum calcium level from baseline in eight patients who reported newly occurring post baseline hypocalcemia symptoms by questionnaire.
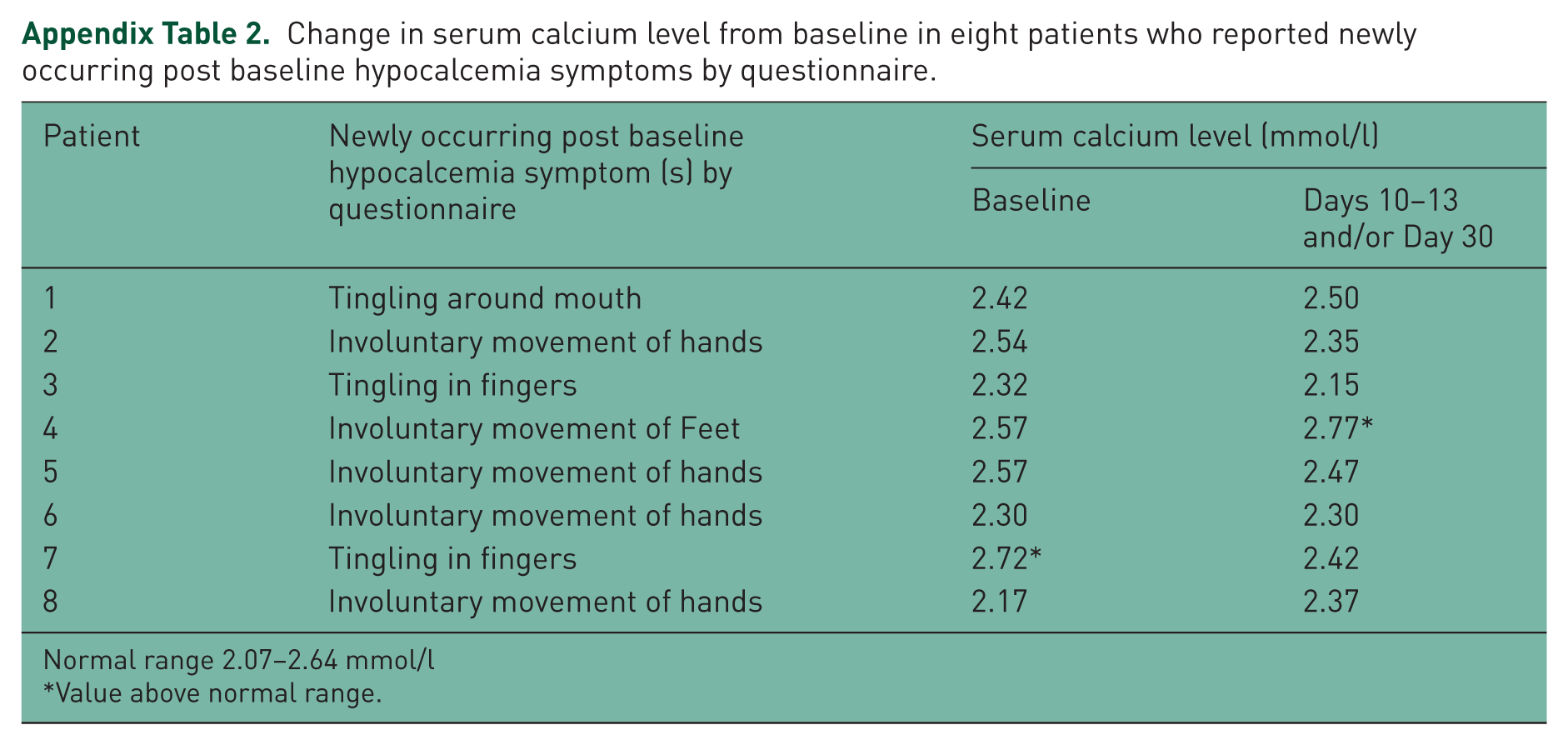
| Patient | Newly occurring post baseline hypocalcemia symptom (s) by questionnaire | Serum calcium level (mmol/l) |
|
|---|---|---|---|
| Baseline | Days 10–13 and/or Day 30 | ||
| 1 | Tingling around mouth | 2.42 | 2.50 |
| 2 | Involuntary movement of hands | 2.54 | 2.35 |
| 3 | Tingling in fingers | 2.32 | 2.15 |
| 4 | Involuntary movement of Feet | 2.57 | 2.77* |
| 5 | Involuntary movement of hands | 2.57 | 2.47 |
| 6 | Involuntary movement of hands | 2.30 | 2.30 |
| 7 | Tingling in fingers | 2.72* | 2.42 |
| 8 | Involuntary movement of hands | 2.17 | 2.37 |
Normal range 2.07–2.64 mmol/l
Value above normal range.
Declaration of Conflicting Interest
J.R.T has received research support from Novartis. H.G.B. has received research support, and consulting fees from Amgen, Novartis and Merck, and speaker fees from Amgen and Merck. G.S., M.T., Z.E.O. and R.P.A. are employed by Novartis Pharmaceuticals Corporation.
Funding
Financial support for this study was provided by Novartis Pharma AG. The authors acknowledge writing/editorial support from Ganesh Sangle (formerly of Novartis) and Deepika Kajarekar (Novartis) in the preparation of this manuscript.
