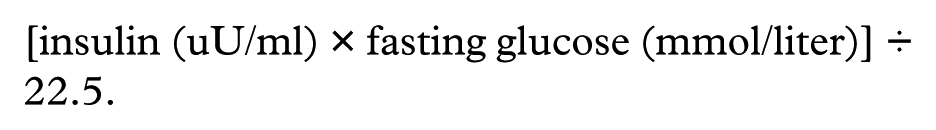Abstract
Background:
We investigate the effects of omega-3 plus vitamin E and vitamin C plus zinc supplementations on cardiovascular risk markers in postmenopausal women with type 2 diabetes.
Methods:
In this double-blind placebo-controlled clinical trial, 75 postmenopausal women with type 2 diabetes were randomly assigned to one of three groups to take, daily, for 12 weeks: (1) 1.8 g omega-3 fatty acids plus 400 mg vitamin E; (2) 5 mg zinc plus 300 mg vitamin C; or (3) a placebo.
Results:
Although the cardiovascular risk markers variations decreased at the end of intervention, we did not find any significant differences in cardiovascular risk markers between therapeutic groups and the control group. Results of repeated measures analysis of variance (ANOVA) of markers showed that total cholesterol, low-density lipoprotein (LDL) cholesterol, Apo A1 and Apo B100 in two time periods were significant (p = 0.001). The level of total and LDL cholesterol decreased significantly (p = 0.05) in patients diagnosed with diabetes equal or less than 7 years in the group receiving omega-3 plus vitamin E. However, decreased LDL cholesterol (p = 0.003) and increased high-density lipoprotein (HDL) cholesterol (p = 0.03) were predominant in patients who had been diagnosed with diabetes equal or less than 7 years in the group receiving zinc plus vitamin C.
Conclusion:
The effectiveness of nutraceutical supplementation was varied on biochemical biomarkers based on the kind of supplement or supplement pharmacogenomics, duration of diabetes affected and other pathophysiologic status in studied groups.
Introduction
Type 2 diabetes mellitus (T2DM) is associated with a high rate of complications related to cardiovascular disease and diabetic nephropathy, retinopathy, and neuropathy [Gu et al. 1996]. Antioxidant therapy might be of great benefit in these patients, and it has recently been suggested that diabetic subjects with complications might have defective cellular antioxidant response against the oxidative stress generated by hyperglycemia [Ceriello, 2006]. Several studies have shown that persons with T2DM have lower vitamin E and C levels. Lower tocopherol levels might increase the risk of oxidative stress (a cause) or levels of vitamin E might be lower because of greater oxidative stress [Penckofer et al. 2002].
Diets high in saturated fat are related to reduced insulin sensitivity. Substitution of n-3 fatty acids from fish oil [largely the long-chain highly unsaturated eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) fatty acids] with the safflower oil diet (rich in n-6 fatty acids) prevents the development of in vivo insulin resistance and improves insulin action in the liver and skeletal muscle. There is consistent evidence that n-3 unsaturated fatty acids improve insulin action and increase insulin sensitivity in rodents [McAuley and Mann, 2006]. Regular consumption and dietary supplementation with n-3 polyunsaturated fatty acid (PUFA) supplements, such as fish oil capsules, have health benefits in disease prevention, health promotion and the prevention and treatment of inflammatory diseases. They could play a key role in the prevention and management of several diseases such as coronary heart disease, dyslipidemia, T2DM, insulin resistance, and hypertension. However, some effects of n-3 PUFAs on physiological processes in humans and experimental animals still remain unclear and need further research [Garg et al. 2006; Lombardo and Chicco, 2006]. Furthermore, clinical trials have demonstrated that administration of vitamin E and other antioxidants and omega-3 PUFAs improve insulin sensitivity in patients with insulin resistance or diabetes. These supplements have a favorable effect on atherogenesis and the secondary complications of diabetes [McCarty, 1998; Schroder, 2007]. On the other hand, zinc supplementation in T2DM patients improves lipid and lipoprotein profiles [Partida-Hernandez et al. 2006], elevates serum zinc levels, and improves glycemic control [Al-Maroof and Al-Sharbatti, 2006].
In addition, antioxidants with different chemical properties may recharge each other in an antioxidant network. Antioxidants and other plant compounds might also improve the endogenous antioxidant defense through induction of antioxidant and phase 2 enzymes [Blomhoff, 2005]. Medical nutrition therapy is the first approach in the management of diabetes, and dietary manipulations that modify low-density lipoprotein (LDL) oxidative resistance could have an influence on the development of vascular disease [Rodriguez-Villar et al. 2004]. It has been postulated that a vitamin cocktail would provide stronger protection than each vitamin acting alone against diabetes [Hardy et al. 2003]. The results of an 11-year study confirmed that relative risks of conventional cardiovascular risk factors for the occurrence of myocardial infarction in postmenopausal women were higher than in men in all age groups [Mahmoodi et al. 2007]. Therefore, in this research, we assessed the effects of omega-3 plus vitamin E and vitamin C plus zinc supplementation on cardiovascular risk markers in postmenopausal women with T2DM and compared them with a control group.
Subjects and methods
Study subjects
A total of 75 postmenopausal women aged 50–65 years with type 2 diabetes participated from the Iranian Diabetes Association. Participants were randomly assigned to 3 groups, each containing 25 postmenopausal women taking (for 12 weeks): (1) 1.8 g/day of omega-3 (2 capsules each containing 0.9 mg) plus 400 mg/day vitamin E (one capsule per day); (2) 5 mg/day zinc plus 300 mg/day vitamin C (one capsule per day); (3) a 500 mg canola oil (one capsule per day) placebo control. During the intervention period, individuals consumed their normal diet.
Eligibility and study design
We conducted a 12-week randomized, double-blind, placebo-controlled clinical trial in Tehran, Iran, using the Iranian Diabetes Association as an educational, consulting and medical clinic. We assessed the effects of omega-3 plus vitamin E and vitamin C plus zinc supplementation on cardiovascular risk markers such as serum total cholesterol, LDL cholesterol, apolipoprotein (Apo) A1, Apo B100, fasting insulin, glycated hemoglobin levels, homeostatic model assessment insulin resistance (HOMA-IR), weight, and body mass index in postmenopausal women with type 2 diabetes compared with the control group. Randomization was performed by a statistics counselor with the use of Excel software. Sampling was performed as sequential at the first visit. The masked administration of either therapies or placebo began at the first visit and at 4-week intervals. In order to maintain equal intensity of treatment, all three supplements were placed in similar white plastic containers among the three studied groups, except for instructions and materials specific to each treatment approach. Six times during the 12-week intervention, 10–15 minute motivational telephone interviews were conducted with participant patients who were having difficulty adhering to the supplements. Compliance with supplements was assessed by unused pill counts.
The criteria for eligibility were: (1) willingness to participate in the study and sign the informed consent; (2) postmenopausal women aged 50–65 years; (3) the presence of type 2 diabetes at least for 1 year (according to the American Diabetes Association criteria); (4) noninsulin therapy; (5) patients receiving either diet therapy or diet therapy with a combination of oral antidiabetic medications; (6) nonsmokers; (7) no history of myocardial infarction, stroke, cardiovascular disease, active cancer, liver, kidney or thyroid dysfunction, or infectious diseases; (8) nonconsumption of antioxidant vitamin supplements and fish oil capsules, nonsteroidal anti-inflammatory drugs, corticosteroids, antiplatelets and anticoagulation drugs for the least 3 months before they were recruited; (9) no use of hormone replacement therapy. Exclusion criteria were: (1) change in dose of medications such as oral antidiabetic, antilipidemic, or antihypertensive drugs; (2) occurrence of myocardial infarction, stroke, active cancer, liver, kidney or thyroid dysfunction, or infectious diseases; (3) change in drug regimen or insulin therapy.
Each participant provided written informed consent before enrollment. The numbers of patients who underwent randomization, allocation, and completed the study are shown in Figure 1. After obtaining the informed consent, patients were assessed by dietary intake, medical history, anthropometric, and biochemical measurements.

Flow chart of enrollment of the participants for the study.
This trial was registered with the Iranian registry of clinical trials for IRCT000000002214N1. The trial was sponsored by National Nutrition and Food Technology Research Institute (NNFTRI), Vice chancellor for Research of Kerman University of Medical Sciences, and Belgian Minami Nutrition Company, and the protocol was approved by review panels and ethics committees (Ref. Num. 5830/47/25) of each research center. The funding organizations had no role in the design of the study; analysis, or interpretation of the data; or the writing of the manuscript.
Anthropometry assessment
The participants were weighed without shoes and minimal clothing to the nearest 0.1 kg at baseline and at the end of the study with the use of the Seca scale. With the use of a wall-mounted Seca stadiometer, height was measured to the nearest 0.5 cm at baseline and the end of the study to calculate the body mass index (the weight in kilograms divided by the square of the height in meters).
Biochemical measurement
Blood samples were drawn into ethylenediaminetetraacetic acid (EDTA) tubes after a 12-hour fast at baseline and the end of the study and immediately stored on ice until centrifugation at 3000 rpm for 10 minutes. Plasma samples were stored at −80°C until an assay for glycemic and lipoprotein biomarkers could be performed. Levels of glycated hemoglobin were measured in fresh samples. The levels of glycated hemoglobin were determined by ion exchange method (Glycohemoglobin kit, Inter Medical Co., Italy). Plasma insulin levels were measured by an enzyme-linked immunosorbent assay (ELISA; Mercodia, Uppsala, Sweden) with the use of an automated analyzer (Sunrise, Tecan, Austria), with a sensitivity of mU/lit. The HOMA-IR was calculated according to the following equation:
Plasma levels of total cholesterol were determined enzymatically (Pars Azmoon kit) with the use of a Selectra 2 autoanalyzer, with a sensitivity of 3 mg/dl. Plasma levels of LDL cholesterol were calculated using the Friedwald equation. Plasma levels of apolipoprotein A1 and B100 were measured by ELISA (Total Human Apo B & Apo A, Alerchek, ink, USA). Laboratory personnel were unaware of the treatment assignments.
Statistical analysis
In order to calculate the sample size (type I error = 0.05, type II error = 0.1), data from published tissue plasminogen activators indicated that a minimum of 18 participants per group would be needed to detect significant differences between groups.
Statistical analysis was performed using SPSS software, version 16.0 (SPSS Inc.). Normal distribution of biomarkers was investigated by Kolmogorov–Smirnov test. Significance was assumed at p < 0.05. We compared the dietary intake values between three groups with the use of an analysis of variance (ANOVA) in which all pairwise comparisons among the three groups were performed with the use of Tukey’s honestly significant difference (HSD) procedure. ANOVA followed by Tukey’s HSD for multiple comparisons was used to determine significant mean differences between the start and the end of the study. Comparisons of mean differences for each biomarker between the start and the end of study in each group were analyzed by paired t-test. In order to clarify individual differences and diminishing intragroup variances and increasing power analysis, mean differences were compared by one-way repeated measures ANOVA. As the mean and median of the independent variable duration of diabetes in affected participants (7 years) closely corresponded, this variable was dichotomized. Then, general linear models (univariate ANOVA) were used to examine associations between selected dependent variables and the duration of diabetes and the start and the end of the study.
Supplement characteristics
EPA and DHA capsules (PluShinzO-3 Cardio Omega-3) and matching placebos were donated by Belgian Minami Nutrition Company; vitamin E capsules (Nature Made) were donated by Iranian Poura Teb Medical and Pharmaceutical Company; and vitamin C and zinc capsules (EuRho OTC Pharma GmbH) were donated by Hakiman Teb Pharmaceutical Company.
Results
Patient characteristics
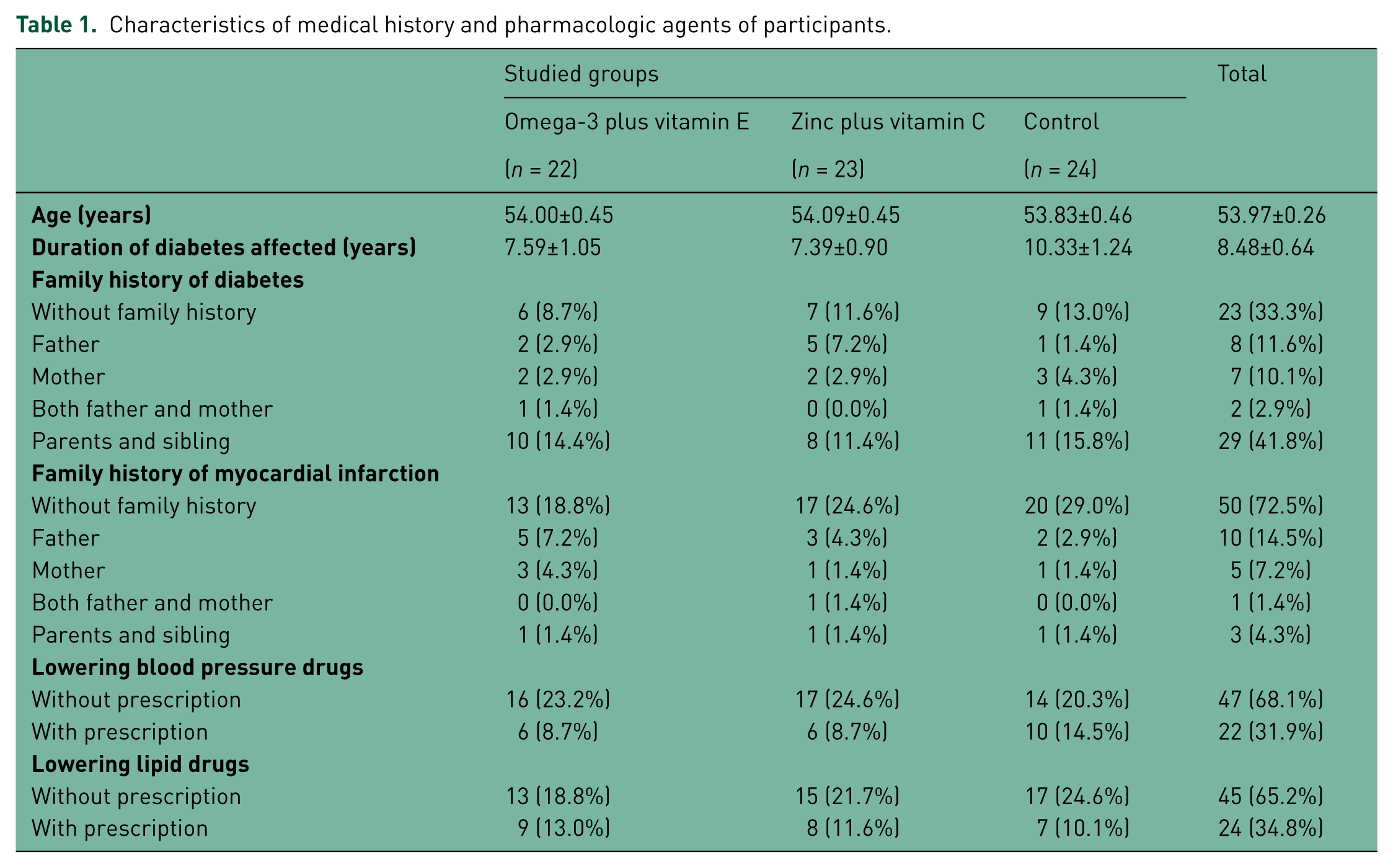
The rate of adherence to the study was 92% (69 subjects) at 12-week intervention. Six subjects were excluded due to noncompliance with intervention (Figure 1). The mean (±SD) age of participants was 53.97 ± 2.14 years. The patients had a mean and a median duration of disease of 7 years. Baseline characteristics of the medical history of diabetic patients are shown in Table 1. There were no significant differences among the groups due to the random allocation of patients. Table 1 shows that one third of participants did not have any family history of diabetes and more than of two thirds of participants had no family history of myocardial infarction.
Characteristics of medical history and pharmacologic agents of participants.
Biochemical measurement
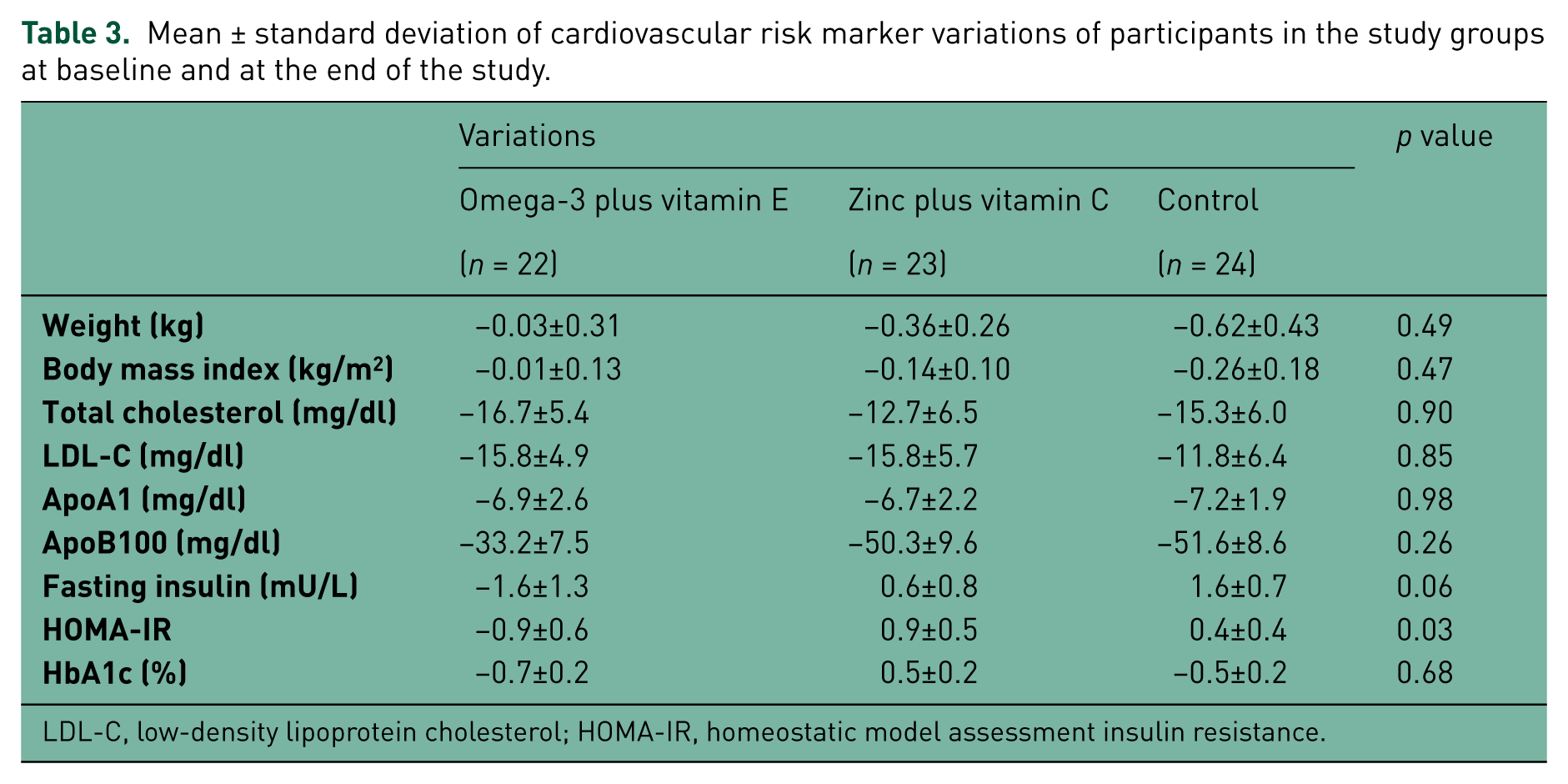
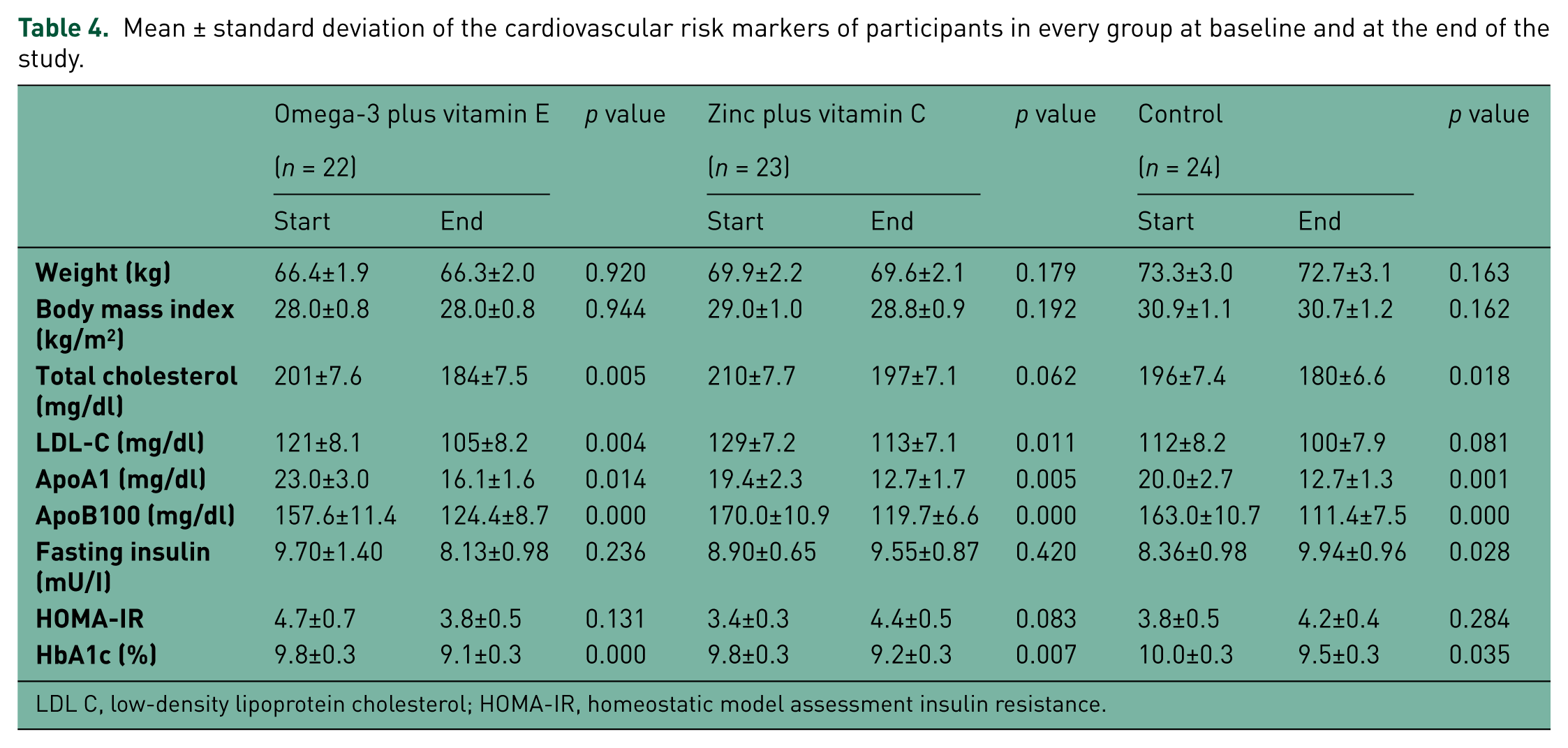
There were no significant differences in baseline cardiovascular risk markers among the groups. There were no significant differences in cardiovascular risk markers among the groups after the 12-week intervention (Table 2). Table 3 indicates the mean (±SD) differences of cardiovascular risk marker variations of the participants in the studied groups during the intervention. Although the cardiovascular risk markers variations decreased at the end of intervention, we found a significant difference in HOMA-IR and a difference close to significant in plasma insulin as cardiovascular risk markers between therapeutic groups and the control group. Table 4 indicates the mean (±SD) of cardiovascular risk markers of diabetic patients in every group at baseline and at the end of intervention.
Mean ± standard deviation of cardiovascular risk markers of participants in the study groups at baseline and at the end of the study.

LDL-C, low-density lipoprotein cholesterol; HOMA-IR, homeostatic model assessment insulin resistance.
Mean ± standard deviation of cardiovascular risk marker variations of participants in the study groups at baseline and at the end of the study.
LDL-C, low-density lipoprotein cholesterol; HOMA-IR, homeostatic model assessment insulin resistance.
Mean ± standard deviation of the cardiovascular risk markers of participants in every group at baseline and at the end of the study.
LDL C, low-density lipoprotein cholesterol; HOMA-IR, homeostatic model assessment insulin resistance.
Results of one-way repeated measures ANOVA of markers (Mauchly’s test had not been significant and the assumption of compound symmetry had been met in order to clarifying individual differences) showed that total cholesterol, LDL cholesterol, Apo A1 and Apo B100 in the two time periods were significant (p = 0.001). However, interaction between these markers and therapeutic groups was not significant.
Results of univariate ANOVA of cardiovascular risk markers based on the duration of diabetes affected in studied groups at baseline and at the end of study are shown in Table 5. The level of total cholesterol and HbA1c decreased significantly (p = 0.05 and p = 0.001, respectively) in patients who were diagnosed with diabetes equal or less than 7 years in the group receiving omega-3 plus vitamin E. The level of LDL cholesterol also decreased significantly (p = 0.04) in the same group. Moreover, a decrease in LDL cholesterol (p = 0.003) was predominant in patients diagnosed with diabetes equal or less than 7 years in the group receiving zinc plus vitamin C and a decrease in HbA1c (p = 0.04) was found in patients who were diagnosed with diabetes more than 7 years in the group receiving zinc plus vitamin C.
Univariate analysis of variances of cardiovascular risk markers of the participants in the study groups at baseline and at the end of the study.

LDL C, low-density lipoprotein cholesterol; HOMA-IR, homeostatic model assessment insulin resistance.
Discussion
The results of our study indicate that supplementation with omega-3 plus vitamin E has the greatest effect on reducing HOMA-IR and plasma insulin concentration variations. These findings show a reduction in HOMA-IR and plasma insulin concentration in the group receiving omega-3 plus vitamin E supplementation in comparison with an increase in these cardiovascular markers in the group receiving zinc plus vitamin C supplementation. Moreover, the other cardiovascular risk markers variations decreased at the end of intervention, but we found no significant differences in cardiovascular risk markers between the therapeutic and control groups. On the other hand, when we dichotomized samples based on the duration of diabetes affected, the levels of total cholesterol, LDL cholesterol and HbA1c were decreased significantly in patients diagnosed with diabetes equal or less than 7 years in the group receiving omega-3 plus vitamin E, and in the group receiving zinc plus vitamin C the level of LDL cholesterol and HbA1c was decreased in patients diagnosed with diabetes equal or less and more than 7 years, respectively.
In this study, there was no significant variation in HbA1c. Ebbesson and colleagues [Ebbesson et al. 2005] demonstrated that a high consumption of omega-3 was correlated inversely with plasma levels of insulin and HOMA-IR, and positively affected insulin sensitivity. In contrast with our results, other researchers did not find any significant differences in insulin concentration and insulin sensitivity [Shidfar et al. 2008; Woodman et al. 2002; Puhakainen et al. 1995; Kesavulu et al. 2002; Brady et al. 2004]. However, the effect of omega-3 supplementation on HbA1c was not significant in some studies [Shidfar et al. 2008; Woodman et al. 2002; Puhakainen et al. 1995; Petersen et al. 2002]. In Ble-Castillo’s study [Ble-Castillo, 2005], alpha-tocopherol administration did not affect glycated hemoglobin. In our study, the effect of supplementation on the markers studied was the result of simultaneous administration of omega-3 and vitamin E.
The results of previous studies revealed that supplementation with omega-3 had disparate and inconsistent influences on cardiovascular markers that might be due to the diversity, mixture, composition, and purity of omega-3, as well as the duration of supplementation. Pathophysiology status of samples in these studies might have particular importance. Age, gender, obesity, and predisposing disorders, such as hyperlipidemia, can affect the results of studies.
On the other hand, one of limitations of our study was that we could not determine haptoglobin (Hb) genotypes in our patients and we could not assess the effect of supplementation on cardiovascular biomarkers based on Hb genotypes in these patients. Therefore, both Hb genotypes of patients and type of supplements will be effective in the response of biomarkers. Blum and colleagues assessed the relationship between Hb type, vitamin E supplementation, and cardiovascular end points in diabetic participants [Blum et al. 2010]. The results of their meta-analysis indicated that vitamin E supplementation could reduce the number of cardiovascular events in diabetic patients with Hb 2-2 genotypes [Blum et al. 2010].
In our study, zinc plus vitamin C supplementation resulted in increased plasma insulin, HOMA-IR, and glycated hemoglobin. Chen and colleagues [Chen et al. 2006] investigated the effects of high-dose oral vitamin C in altering insulin resistance in 32 diabetic subjects in a randomized, double-blind, placebo-controlled study of vitamin C (800 mg/day for 4 weeks). No significant changes in plasma insulin and insulin resistance were observed after vitamin C treatment in their study. The second part of the Al-Maroof and Al-Sharbatti’s study [Al-Maroof and Al-Sharbatti, 2006] showed that HbA1c% concentration decreased significantly after 3 mg per day zinc supplementation at the end of the 3 months of follow up.
Results of previous studies revealed that the role of water- or oil-soluble antioxidants in improving insulin resistance were different in type I and II diabetes.
Nevertheless, another study revealed that supplementation with vitamin E reduced high-density lipoprotein (HDL) oxidation and corrected HDL dysfunction in Hb 2-2 diabetic participants as compared with non-Hb 2-2 individuals [Asleh et al. 2008]. In contrast, vitamin C supplementation increased the oxidative activity of Hb 2-2-hemoglobin bound to HDL and not only increased HDL oxidation but also resulted in a proinflammatory and proatherogenic status [Asleh et al. 2008].
In our study, lipid and lipoprotein risk markers decreased in therapeutic groups; however, there were no significant differences in these risk markers between therapeutic groups and the control group after 12 weeks intervention. These effects were similar to other researchers [Shidfar et al. 2008; Woodman et al. 2002; Petersen et al. 2002; Finnegan et al. 2003; Mori et al. 2000]. Supplementation with omega-3 at different doses and various intervention periods did not produce significant differences in total cholesterol and LDL cholesterol. Axelrod and colleagues showed a low dose of omega-3 fatty acids (2.5 g/day) in 20 diabetic persons caused a significant but small increase in total cholesterol but had no effect on LDL cholesterol levels [Axelrod et al. 1994]. Hartweg and colleagues in a meta-analysis from 23 randomized clinical trials revealed that 3.5 g/day omega-3 with mean treatment duration of 8.9 weeks could raise LDL cholesterol [Hartweg et al. 2008]. Farvid and colleagues also showed that supplementation with vitamins C and E or minerals magnesium and zinc had no significant effect on total cholesterol and LDL cholesterol [Farvid et al. 2004].
In our study, Apo A1 and B100 decreased after intervention; however, there were no significant differences in these risk markers between therapeutic groups and the control group. Dietary supplementation with 2 g/day purified omega-3 fatty acids for 10 weeks on 50 type 2 diabetes patients revealed a significant decrease in ApoB-100, but no significant effect was found on Apo A1 [Shidfar et al. 2008]. A significant decrease in plasma ApoB-48 was observed following fish-oil supplementation [Lovegrove et al. 2004]. DHA supplementation in Theobald and colleagues’ study lead to increased Apo B100 [Theobald et al. 2004]. Vitamin and mineral supplementation in Farvid and colleagues’ study lead to increased plasma Apo A1 concentration [Farvid et al. 2004]. On the other hand, in our study, a decrease in Apo A1 and Apo B100 was not easily interpretable. However, supplementation in patients diagnosed with diabetes equal or less than 7 years had a more considerable influence.
The diversity in apolipoprotein and lipoprotein response after supplementation with omega-3 or fish oil alone and antioxidants was due to the variations in composition and purity. In our study, one of the most important factors was the simultaneous supplementation with omega-3 and vitamin E, due to the decrease in oxidative stress from omega-3 consumption as PUFAs. Therefore, imagine of decreasing or increasing a marker may relate to simultaneous supplementation with supplements. The other reason, supplementation effectiveness was based on duration of diabetes affected. Independent and effective variables such as body mass index and waist-to-hip ratio are valuable for the dichotomization of studied samples.
Conclusions
The effectiveness of nutraceutical supplementation on biochemical biomarkers was varied based on the type of supplement or supplement pharmacogenomics, duration of diabetes affected, and other pathophysiologic statuses in the studied groups.
Footnotes
Acknowledgements
The authors are grateful to the NNFTRI and Vice chancellor for Research of Kerman University of Medical Sciences for supporting this study. The authors are also grateful to Mr Jo Wyckmans, the founder of the Belgian Minami Nutrition Company, for sending EPA and DHA capsules and placebos, and the Iranian Poura Teb Medical and Pharmaceutical Company and Hakiman Teb Pharmaceutical Company for preparing vitamin E and vitamin C plus zinc supplements, respectively.
Conflict of interest statement
The authors have no potential conflicts of interest relevant to this article to report.
Funding
This conjoint research was supported by grants from NNFTRI and Vice chancellor for Research of Kerman University of Medical Sciences.