Abstract
Diabetic retinopathy is a common microvascular complication of prolonged diabetes. It is one of the leading causes of vision loss in working age adults and a significant source of morbidity. To reduce the incidence and severity of diabetic retinopathy, it is important to identify patients at risk and promptly implement intensive glycemic and hypertensive control. To date, there are many interventions that can limit moderate and severe vision loss in people with diabetes, including laser photocoagulation, vitrectomy surgery, and intravitreal pharmacological injections. This review aims to define the different stages of diabetic retinopathy and the important therapeutic advances developed to address the major causes of vision loss in each stage.
Keywords
Introduction
Diabetes affects 200 million people worldwide, including 23.6 million people in the USA alone (7.8% of the population) [Center for Disease Control and Prevention, 2007]. Diabetic retinopathy is one of the leading causes of preventable blindness among working-aged Americans [Klein et al. 1984a, 1984b]. According to the Center for Disease Control, diabetic retinopathy causes between 12,000 and 24,000 new cases of blindness each year. Diabetic retinopathy is the primary cause of blindness in patients who develop diabetes before the age of 30, and the primary cause in one-third of cases of blindness among those developing diabetes after the age of 30. The prevalence of diabetic retinopathy increases with the duration of diabetes. The Wisconsin Epidemiologic Study of Diabetic Retinopathy Study found the rate of retinopathy to be 98% in patients with type 1 diabetes and 78% in patients with type 2 diabetes with disease duration greater than 15 years [Klein et al. 1984a, 1984b]. In the USA, the reported cost of diabetic-related blindness in adults (aged 40 and over) approaches $500 million annually [Rein et al. 2006].
In recent years, advances in surgical techniques and pharmacotherapy have shown significant promise in the treatment of diabetic retinopathy. This article reviews the major clinical trials and therapeutic advances in diabetic retinopathy.
Diabetic retinopathy
The pathogenesis of diabetic retinopathy is not completely understood. Hyperglycemia is the primary insult, resulting in damage to the microvasculature, and subsequently to retinal tissue. Diabetic retinopathy is separated into two stages: nonproliferative and proliferative diabetic retinopathy.
Nonproliferative diabetic retinopathy is the earlier form of retinopathy with microvascular damage characterized by microaneurysms, retinal hemorrhages, and capillary closure. Progressive damage is marked by nerve fiber layer infarcts (i.e. cotton-wool spots), venous beading, and intraretinal microvascular abnormalities (Figure 1).
Nonproliferative diabetic retinopathy: microaneurysms, retinal hemorrhages, and cotton wool spots (color fundus photograph).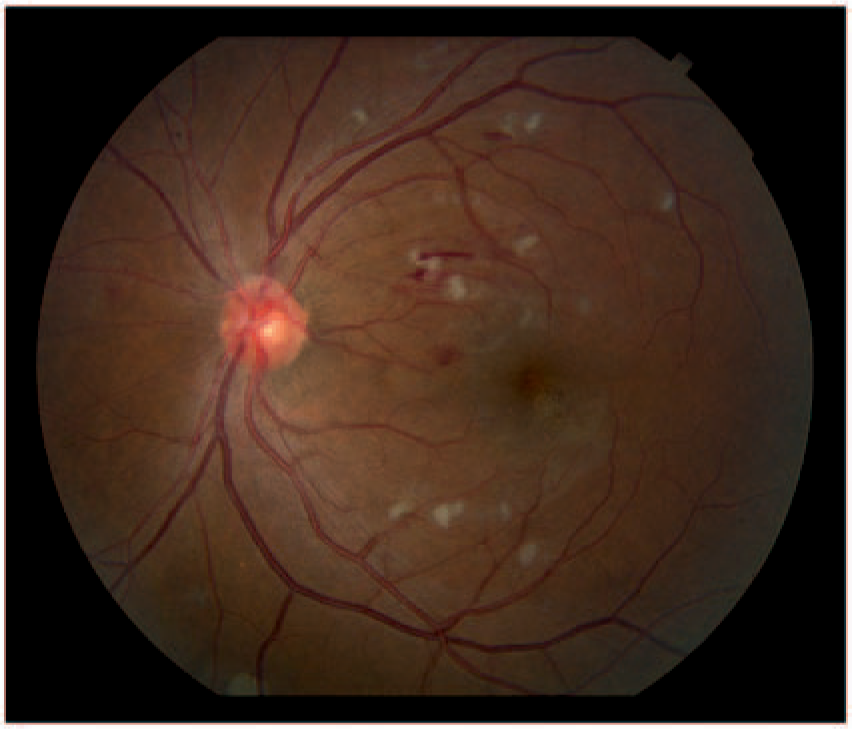
Capillary closure results in retinal hypoxia, leading to increased production of vascular endothelial growth factor (VEGF) and other angiogenic factors. Proliferative diabetic retinopathy is characterized by proliferation of fine new abnormal blood vessels on the retina or optic nerve, stimulated by angiogenic factors from ischemic retinal tissue (Figure 2(a)). This neovascularization can lead to retinal or vitreous hemorrhage from rupture of abnormal vessels (Figure 2(b)), or to fibrosis resulting in tractional retinal detachment (Figure 2(c)). Continued ischemia can cause abnormal blood vessel growth over the iris and trabecular meshwork, the drainage system of the eye, resulting in elevated intraocular pressure in neovascular glaucoma (Figure 2(d)).
Proliferative diabetic retinopathy: (a) neovascularization elsewhere, cotton wool spots (color fundus photograph); (b) preretinal vitreous hemorrhage, neovascularization of the optic disc (color fundus photograph); (c) tractional retinal detachment (color fundus photograph); (d) neovascular glaucoma, anterior segment photo showing leakage of fluorescein dye from peripupillary neovascular vessels.
Diabetic macular edema can occur at any stage of diabetic retinopathy. Diabetic macular edema occurs as a result of increased vascular permeability due to a breakdown of the blood–retinal barrier, often with deposition of hard exudates in the macula (Figure 3(a)). Fluorescein angiography of diabetic macular edema shows leakage of the blood vessels (Figure 3(b) and (c)) resulting in retinal edema, which can be seen in cross section with optical coherence tomography (Figure 3(d)). The Early Treatment Diabetic Retinopathy Study defined clinically significant macular edema (CSME), which requires treatment, as retinal thickening that involves or threatens the center of the macula. This is characterized as retinal thickening within 500 µm of the fovea, hard exudates within 500 µm of the fovea if associated with adjacent thickening, or a zone of retinal thickening at least 1500 µm in size within a 1500 µm perimeter of the fovea [Early Treatment Diabetic Retinopathy Study Research Group, 1985].
(a) Clinically significant macular edema (color fundus photograph); (b) clinically significant macular edema in same eye (fluorescein angiography, early phase, leakage of dye); (c) clinically significant macular edema in same eye (fluorescein angiography, late phase, leakage and staining of dye); (d) clinically significant macular edema (optical coherence tomography).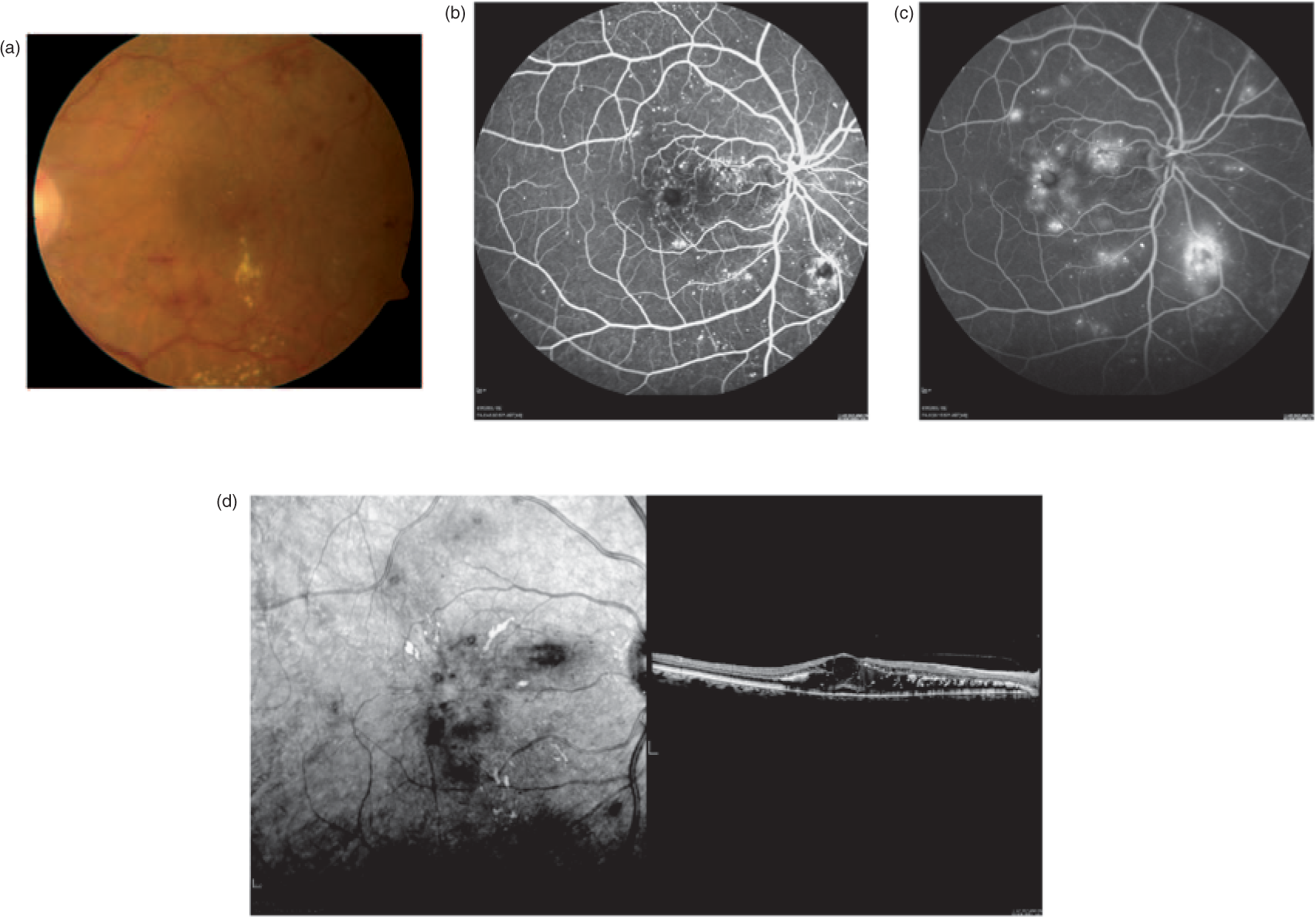
The most common causes of moderate and severe vision loss from diabetic retinopathy result from diabetic macular edema and proliferative diabetic retinopathy, respectively.
Primary interventions
Glycemic control
Poor glycemic control is directly related to diabetic retinopathy. Two large randomized controlled trials provided evidence of the efficacy of tight glycemic control in reducing the incidence and progression of diabetic retinopathy.
The Diabetes Control and Complications Trial study group conducted a trial from 1983 to 1993 in 1441 patients with type 1 diabetes and nonproliferative diabetic retinopathy randomized to receive intensive glycemic control [median glycosylated hemoglobin A1c (HbA1c) 7.2%] or moderate control (median HbA1c 9.1%). The intensive treatment group had a reduction of diabetic retinopathy incidence of 76% and progression of 54% compared with the moderate treatment group [Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications Research Group, 2008; Diabetes Control and Complications Trial Research Group, 1993]. The study found two significant clinical drawbacks to tight glycemic control in patients with type I diabetes: an increased number of hypoglycemic episodes and early worsening of retinopathy. The effect on retinopathy was reversed by 18 months and did not result in serious visual loss. A careful ophthalmoscopic exam should be performed prior to institution of intensive glycemic control and at regular intervals of 3–6 months during this time [Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications Research Group, 2008]. Egger and colleagues showed that intensive therapy is associated with a threefold increase in hypoglycemic episodes and 70% higher risk of ketoacidosis compared with conventional treatment. Thus, tight glycemic control should be instituted under the close supervision of a primary care physician [Egger et al. 1997].
The UK Prospective Diabetes Study randomized 3867 patients with newly diagnosed type 2 diabetes to intensive or conventional therapy. They found that intensive glycemic control reduced the progression of diabetic retinopathy by 21% and reduced the need for laser photocoagulation of the retina for diabetic retinopathy (discussed later in this review) by 29% compared with conventional therapy after 12 years of follow up [UK Prospective Diabetes Study Group, 1998a]. These effects persisted 10 years after study completion [White et al. 2008].
Many of the landmark trials establishing the foundations for management and treatment of diabetic retinopathy were performed two to three decades ago. More recently, the Action to Control Cardiovascular Risk in Diabetes Eye Study randomized over 10,000 patients with type 2 diabetes to receive intensive glycemic control (median HbA1c 6.4%) or standard therapy (median HbA1c 7.5%); 2856 participants with nonproliferative diabetic retinopathy were followed for 4 years. Progression of diabetic retinopathy was significantly lower in patients in the intensive therapy group (7.3%) compared with the standard therapy group (10.4%). The rates of moderate vision loss among the groups were similar at 16.3% and 16.7% in the intensive and standard groups, respectively [Chew et al. 2010].
These trials provided compelling evidence that strict glycemic control is needed to prevent vision loss associated with diabetes. Innovations in monitoring diabetes such as home monitoring of glucose levels have contributed greatly towards more effective control of diabetes. Further, significant advances in treatment options, such as an insulin pump and a variety of oral antiglycemic agents, continue to improve the management of diabetes.
Hypertensive control
Tight control of blood pressure has been shown to reduce the progression of diabetic retinopathy in patients with type 2 diabetes [Schrier et al. 2002]. The UK Prospective Diabetes Study performed a subgroup analysis of 1148 patients with hypertension and type 2 diabetes; patients were randomized to less tight (<180/105 mmHg) and tight (<150/85 mmHg) blood pressure control with a mean follow-up period of 8.4 years. The tightly controlled patients had reduced progression of retinopathy in 34% and reduced deterioration in visual acuity by three lines in 47% compared with the conventional control group. The reduction in vision loss was primarily due to a 42% reduction in the incidence of macular edema. These effects were independent of glycemic control [UK Prospective Diabetes Study Group, 1998b].
Several antihypertensive agents have been studied for their effect on diabetic retinopathy. Angiotensin-converting enzyme inhibitors and angiotensin receptor blockers have been shown in separate trials to reduce diabetic retinopathy progression in patients with normotensive type 1 diabetes and mild diabetic retinopathy [Mauer et al. 2009; Chaturvedi et al. 2008, 1998].
In the Action to Control Cardiovascular Risk in Diabetes study, 1263 patients with type 2 diabetes and nonproliferative diabetic retinopathy were assigned to either intensive blood pressure control (systolic blood pressure <120 mmHg, median 117 mmHg) or standard therapy (systolic blood pressure <140 mmHg, median 133 mmHg). The rates of diabetic retinopathy progression in this study were similar between the groups at 10.4% and 8.8% in the intensive and standard groups, respectively. The rates of moderate vision loss were also similar at 19.4% and 15.8%, respectively [Chew et al. 2010]. The definition of tight and standard blood pressure control were much more stringent in this study than previous ones, which may account for the lack of significance in the role hypertension played on the progression of diabetic retinopathy.
Secondary interventions
Proliferative diabetic retinopathy: laser and surgical interventions
Proliferative diabetic retinopathy is one of the major causes of severe vision loss. The Wisconsin Epidemiologic Study of Diabetic Retinopathy showed a rate of progression to proliferative diabetic retinopathy of 42% in patients with type 1 diabetes over 25 years [Klein et al. 2008]. A similar rate of 48% was found among patients with type 1 diabetes in a separate study in Pittsburgh [Conway et al. 2009]. A recent meta-analysis pooled data from 28 studies and reported a trend of lower rates of proliferative diabetic retinopathy (cumulative: 11%; 1975–1985: 19.5%; 1986–2008: 2.6%) and severe vision loss (cumulative: 7.2%; 1975–1985: 9.7%; 1986–2008: 3.2%); this is likely related to improved diabetic monitoring and available therapy [Wong et al. 2009].
Pan-retinal photocoagulation
Pan-retinal photocoagulation (PRP) was one of the first treatment modalities shown to be effective in preventing visual loss from proliferative diabetic retinopathy. PRP involves placement of laser burns throughout the retina, preserving the optic nerve and macula. The procedure causes regression of new blood vessels by reducing the ischemic load on compromised retinal tissue through destruction of the peripheral retina, limiting the production of VEGF [Shinoda et al. 2000].
The strongest evidence for the efficacy of PRP is from two randomized controlled trials performed in the 1970s and 1980s. The Diabetic Retinopathy Study randomized 1758 patients with proliferative diabetic retinopathy to receive PRP or no treatment. This trial showed that PRP decreases the rate of severe vision loss (visual acuity <5/200) in high-risk proliferative diabetic retinopathy patients by 50% (6.4% of treated eyes versus 16.4% of untreated eyes) [Diabetic Retinopathy Study Research Group, 1976]. The greatest benefit occurred in eyes with high-risk characteristics: neovascularization at the optic disc, vitreous hemorrhage, or 1500 µm of neovascularization elsewhere in the retina [Diabetic Retinopathy Study Research Group, 1987]. Similarly, the Early Treatment Diabetic Retinopathy Study Research Group randomized 3711 patients with less severe diabetic retinopathy to early PRP or observation, and showed that PRP reduces risk of high-risk proliferative diabetic retinopathy by 50%.
The adverse effects of PRP include loss of peripheral vision, night blindness, color vision changes, exacerbation of macular edema, uveal effusion, and tractional retinal detachment. The application of PRP can be limited by pain, requiring several sessions for completion. Newer lasers, namely the Pascal (OptiMedica, Santa Clara, California), allow more efficient and less painful laser photocoagulation. Infrequently, an inadvertent laser burn in the macula can result in central visual loss [Fong et al. 2007]. Currently, PRP remains the gold standard of treatment for proliferative diabetic retinopathy.
Pars plana vitrectomy
Pars plana vitrectomy, innovated in the 1970s, is a surgical procedure that allows removal of the vitreous and manipulation of the retina. The procedure involves creating three small ports at the pars plana, an avascular zone of nonretinal tissue in the anterior globe. Conventionally, one port is for an infusion line to maintain the intraocular pressure. A light pipe typically occupies one of the remaining ports, allowing endoillumination. The final port allows access for a vitrector or other intraocular instruments. PRP can be performed intraoperatively with an endolaser.
The indications for vitrectomy in proliferative diabetic retinopathy include removal of media opacities (such as nonclearing vitreous hemorrhage or preretinal hemorrhage) and repair of tractional retinal detachments. Studies on diabetic eyes before and after vitrectomy show that vitreal oxygen tension increases significantly following removal of the vitreous gel [Holekamp et al. 2005]. The Diabetic Retinopathy Vitrectomy Study randomized 616 eyes of patients with type 1 diabetes and new vitreous hemorrhage accompanied by severe visual loss (<5/200) of 1 month’s duration to either early vitrectomy (within 6 months) or observation. After 2 years, 25% of the early vitrectomy group were found to have significant visual improvement (20/40 or better) compared with 15% of the observation group, with benefits maintained at 4 years [Diabetic Retinopathy Vitrectomy Study Research Group, 1990, 1988a, 1988b, 1985].
Fibrovascular proliferation on the retinal surface can result in retinal traction and subsequent retinal detachment. Such traction can predispose to retinal tears, owing to combined tractional and rhegmatogenous retinal detachments. Vitrectomy with dissection of preretinal membranes can relieve undo traction and allow the retina to reattach. Endolaser is usually applied to seal breaks and to complete PRP. The vitreous cavity is typically left filled with a tamponade, such as gas or silicone oil.
Vitrectomy has advanced significantly since the Diabetic Retinopathy Vitrectomy Study with smaller-gauge, potentially sutureless transconjunctival surgery. Small-gauge vitrectomy (23 or 25 gauge) is capable of addressing most, if not all, of the indications for traditional 20-gauge vitrectomy in proliferative diabetic retinopathy, with the benefit of improved postoperative patient comfort [Smiddy et al. 1999]. Complications of pars plana vitrectomy include vitreous hemorrhage, endophthamitis, retinal tears and detachment, cataract progression, and glaucoma.
More recently, intravitreal bevacizumab, an anti-VEGF agent, has been used as a preoperative- or perioperative adjunct to vitrectomy. Anti-VEGF agents injected preoperatively can reduce neovascularization and fibrovascular adherence to the retina, which can reduce intraoperative bleeding and facilitate dissection of membranes; however, it has been associated with increased risk of traction and retinal tears [Figueroa et al. 2009; Oshima et al. 2009]. See the section below for more detailed information on anti-VEGF agents.
Diabetic macular edema: laser, surgical and medical interventions
Diabetic macular edema is the principal cause of moderate vision loss (doubling of visual angle, i.e. 20/20 to 20/40) among patients with diabetes and nonproliferative diabetic retinopathy [Klein, 1992]. The pathophysiology of diabetic macular edema is multifactorial and includes increased levels of VEGF, loss of endothelial tight junctions, and an increase in inflammatory mediators, which all result in increased breakdown of the blood–retinal barrier and accumulation of extravascular fluid in the macula [Funatsu et al. 2002; Pelzek and Lim, 2002; Ozaki et al. 1997; Aiello et al. 1994].
In the Wisconsin Epidemiologic Study of Diabetic Retinopathy Study, the prevalence of diabetic macular edema was 20.1% in patients with type 1 diabetes, and 25.4% and 13.9% in patients with type 2 diabetes on insulin and not on insulin, respectively. Over a 10-year period, 14% of patients with noninsulin-dependent diabetes will develop diabetic macular edema [Klein et al. 1995], and of these, 50% will lose two lines of visual acuity within 2 years [Ferris and Patz, 1984].
Macular laser treatment
Macular laser treatment was the first treatment shown to be of benefit for diabetic macular edema. It involves application of focal laser photocoagulation to localized leaking microaneurysms or in a grid pattern within a region of macular edema. The mechanism by which macular laser therapy improves diabetic macular edema is unknown, however it is hypothesized that it reduces retinal hypoxia, thus reducing the VEGF load and subsequent vascular permeability in the eye [Nguyen et al. 2006].
In the 1980s the Diabetic Retinopathy Study Research Group conducted a large randomized controlled trial comparing focal laser photocoagulation (754 eyes) with observation (1490 eyes). Patients undergoing focal laser therapy had a 50% reduced risk of moderate vision loss compared with no treatment and an improvement of vision in some patients [Diabetic Retinopathy Clinical Research Network, 2008; Early Treatment Diabetic Retinopathy Study Research Group, 1985]. The Early Treatment Diabetic Retinopathy Study showed that laser photocoagulation reduced the risk of moderate visual loss in eyes with clinically significant macular edema by 50% over 3 years. Figure 4 shows a fluorescein angiogram of an eye with clinically significant macular edema after grid laser treatment.
Clinically significant macular edema after grid laser treatment (fluorescein angiography, no leakage of dye).
Adverse effects include minor visual field losses, inadvertent laser burn to the fovea, and infrequently, expansion of laser scar over time into the fovea with loss of central vision.
Pars plana vitrectomy
More recently, vitrectomy has been shown to have a role in the treatment of a specific subtype of diffuse diabetic macular edema resulting from vitreous traction on the central retina. Contraction of the posterior face of the vitreous, the hyaloid, over the macula can result in edema, which does not respond to focal laser photocoagulation. In some cases, an epiretinal membrane also contributes to traction and macular edema. Vitrectomy allows peeling of epiretinal membranes, often with peeling of the underlying internal limiting membrane, the innermost layer of the retina.
Diabetic macular edema can be exacerbated by traction from the vitreal–retinal interface, the posterior hyaloid. Surgical release of the traction can often improve diabetic macular edema anatomically and clinically improve visual acuity [Harbour et al. 1996].
Most of the randomized controlled trials for vitrectomy in diabetic macular edema have had small sample sizes and short follow up; results have been inconclusive. A randomized trial of 24 patients with diffuse diabetic macular edema reported improved visual acuity and decreased central retinal thickness after vitrectomy with internal limiting membrane peel at 6 months compared with grid photocoagulation [Yanyali et al. 2005]. A similar randomized trial with 24 eyes showed improvement of central retinal thickness but no significant difference in visual acuity between the vitrectomy and grid laser groups at 6-month follow up [Kumar et al. 2007]. A randomized trial of 56 eyes did show a significant improvement in visual acuity and central retinal thickness in eyes with diffuse diabetic macular edema treated with vitrectomy with membrane peel compared with observation [Stolba et al. 2005].
Corticosteroids
Corticosteroids have potent anti-inflammatory and antiangiogenic effects [Nauck et al. 1998]. The accumulation of fluid in the retina in diabetic macular edema is a result of increased vascular permeability factors, including VEGF [Funatsu et al. 2002], loss of endothelial tight junctions [Antonetti et al. 1998], and production of inflammatory mediators such as prostaglandins. Corticosteroids can inhibit these processes without the destruction of retina, an unavoidable consequence of laser photocoagulation.
Injection of steroids into the vitreous cavity is effective in treating diabetic macular edema, however steroids can have significant side effects, including cataract progression and steroid-induced secondary glaucoma.
Triamcinolone
Intravitreal triamcinolone (Kenalog 40, Bristol-Myers Squibb, Princeton, New Jersey) has been used more frequently since 2002 for the treatment of diabetic macular edema. Gillies and colleagues [Gillies et al. 2006] conducted a randomized controlled trial in 69 eyes with persistent diabetic macular edema to receive either 4 mg intravitreal triamcinolone or sham injection; after 2-year follow up, 56% of triamcinolone-treated eyes had an improvement in visual acuity compared with 26% in the control group. However, many eyes required reinjection (mean 2.2). The incidence of steroid-induced glaucoma was significant in the treatment group, with an intraocular pressure rise of 5 mmHg occurring in 68% of eyes receiving intravitreal triamcinolone compared with 10% in the sham group; 44% of these eyes required glaucoma medications. In addition, cataract progression requiring cataract surgery occurred in 55% of triamcinolone-treated eyes over the course of 2 years compared with none in the control group.
The Diabetic Retinopathy Clinical Research Network compared eyes with diabetic macular edema randomized to macular laser treatment, 1 mg intravitreal triamcinolone, or 4 mg intravitreal triamcinolone. The study found that although mean visual acuity was improved in the short term (4-month follow up) with the higher dose of intravitreal triamcinolone, at 1 year there was no difference between the groups; at 2 years, visual acuity was better in the laser group. In addition, the intravitreal triamcinolone groups had an increased risk of an intraocular pressure rise (4%, 16%, and 33%), and a high rate of cataract surgery (13%, 23%, and 51%) in the laser group, the 1 mg intravitreal triamcinolone group, and the 4 mg intravitreal triamcinolone group, respectively [Diabetic Retinopathy Clinical Research Network, 2008].
Thus, although intravitreal triamcinolone is useful in short-term anatomic and visual acuity improvement in diabetic macular edema, it requires repeat injections that subject the eye to significant ocular morbidity and the potential for endophthalmitis. The effect of intravitreal triamcinolone is short lived, unlike macular laser treatment. It is possible that combination treatment with intravitreal triamcinolone followed by macular laser treatment may be more effective than either treatment alone.
Fluocinolone acetonide implant
Fluocinolone acetonide (Retisert, Bausch & Lomb, Rochester, New York) is a surgically placed implant in the eye, designed to release 0.59 µg of drug per day for extended steroid delivery. The US Food and Drug Administration (FDA) approved it for use in chronic, noninfectious posterior uveitis (inflammation of the posterior chamber of the globe). Pearson and colleagues randomized 97 patients with diabetic macular edema to receive the implant or standard care (macular laser treatment or observation). They found that at 3 years, diabetic macular edema resolved in 58% of eyes with implants compared with 30% of control eyes [Pearson et al. 2006]. However, cataract progression and glaucoma continued to be a problem in steroid-treated eyes, with 5% of study patients requiring implant removal for adequate glaucoma control.
A second fluocinolone acetonide implant, Iluvien (Alimera Sciences, Alpharetta, Georgia) is a sustained drug delivery system similar to Retisert. It is a small, nonbiodegradable, cylindrical tube that can be delivered into the vitreous cavity through a 25-gauge needle. It was designed to release fluocinolone acetonide at a rate of 0.5 or 0.2 µg/day initially, with a subsequent slow release of the drug for up to 2–3 years. Campochiaro and colleagues conducted a randomized controlled trial in 37 eyes with refractory diabetic macular edema (previously treated with macular laser treatment) to receive one of two dosages of the implant (0.2 or 0.5 µg/day). They observed improvement of visual acuity and reduction of central retinal thickness with the higher dose implant, although there was a mild increase in intraocular pressure [Campochiaro et al. 2010]. This implant is still under study for use in diabetic macular edema.
Extended-release dexamethasone implant
The FDA recently approved the extended-release dexamethasone biodegradable drug delivery system (Posurdex, Allergan, Irvine, California) for treatment of macular edema following retinal vein occlusion. The implant affords a rapid increase in vitreous concentration of dexamethasone, sustained for up to 6 months; of note, intraocular pressure in eyes receiving the implant were found to be higher, but the results were not of statistical significance [Williams et al. 2009; Kuppermann et al. 2008, 2007].
Haller and colleagues reported a subanalysis on 171 eyes with persistent macular edema secondary to diabetic retinopathy that were treated with observation or a 350 µg or 700 µg dexamethasone implant. The 700 µg implant group was found to have improvement in visual acuity and decreased central retinal thickness compared with observation at 3 months. The most common adverse event was anterior chamber inflammation and vitreous hemorrhage. The group found that the rate of cataract progression was not increased by the implant during the study duration (6 months). However, this follow-up period may not be sufficiently long to assess the rate of cataract progression. In addition, the number of eyes that experienced an increase in intraocular pressure greater than 25 mmHg (average intraocular eye pressure is 16 ± 5 mmHg) was lower with the implant (8%) compared with the reported rates for intravitreal triamcinolone [Haller et al. 2010].
Antivascular endothelial growth factor
VEGF inhibitors have revolutionized the treatment of neovascular age-related macular degeneration. Studies have shown that VEGF levels are elevated in eyes with proliferative diabetic retinopathy, and these levels decline following PRP [Aiello et al. 1997]. Blocking VEGF has been shown to prevent development of neovascular disease in mouse models of ischemic retinopathy [Robinson et al. 1996].
Of note, the majority of the safety data for anti-VEGF medication is derived from studies involving neovascular age-related macular degeneration patients. Patients with diabetic macular edema tend to be younger with more cardiac and renal disease. In addition, diabetic eyes may have retinal neovascularization and fibrovascular proliferation that can contract following anti-VEGF injection, leading to retinal traction and tears. Further long-term randomized controlled trials are required to establish the safety of anti-VEGF agents for diabetic retinopathy [Nicholson and Schachat, 2009].
Adverse events, including infectious endophthalmitis, noninfectious inflammation, retinal detachment, vitreous hemorrhage, and lens injury, can occur following any intravitreal injection. In one of the pioneering studies for pegaptanib (see below), the complication rates for intravitreal injection in 1186 eyes were: endophthalmitis (0.06–0.16% per injection), retinal detachment (0.03–0.17% per injection), and lens injury (0–0.7% per injection) [Gragoudas et al. 2004].
The most feared complication of intravitreal injections is endophthalmitis, and stringent protocols for prevention are of utmost importance as these injections become more common in the treatment of many eye conditions. McCannel presented a meta-analysis of over 100,000 intravitreal injections; he found an endophthalmitis rate of 0.049%, of which 58.1% were culture positive [McCannel, 2010]. Shah and colleagues presented a prospective study of nearly 28,000 injections, finding that neither lid speculum use, hemisphere of injection, nor displacement of conjunctiva affected risk of endophthalmitis. These results reinforce the importance of betadine and sterile technique [Shah, 2010].
Pegaptanib
Pegaptanib is a pegylated RNA aptamer directed against VEGF-A 165 isoform (Macugen, OSI/Eyetceh, Melville, New York). Pegaptanib is FDA approved for the treatment of neovascular age-related macular degeneration. Its safety and efficacy have been established in the VEGF Inhibition Study in Ocular Neovascularization (VISION) trial, which found no systemic side effects associated with its use over a 3-year follow-up period [Gragoudas et al. 2004]. In a phase II trial, 172 eyes with diabetic macular edema were randomized to three different dosages of pegaptanib or sham injection; over the course of 9 months, pegaptanib eyes had improved visual outcomes, decreased central retinal thickness, and were less likely to require MLT [Cunningham et al. 2005]. Newer, more potent anti-VEGF agents that target all isoforms of VEGF, as discussed below, have largely replaced pegaptanib.
Ranibizumab
Ranibizumab is a recombinant humanized antibody fragment active against all VEGF isoforms, FDA approved for the treatment of neovascular age-related macular degeneration. The Ranibizumab for Edema of the Macula in Diabetes (READ-2) Study examined a subgroup of 126 patients with diabetic macular edema randomized into groups receiving the ranibizumab alone (monthly for 6 months), the drug in combination with focal/grid laser photocoagulation, or laser alone. Results show that at 6 months, the intravitreal ranibizumab group improved the most in visual acuity compared with laser or laser–drug combination [Nguyen et al. 2009]. This cohort of patients was treated with ranibizumab as needed after 6 months; improved visual acuity and decreased central retinal thickness were maintained up to 2 years [Nguyen et al. 2010]. The study showed that benefit from laser treatment occurred slowly; ranibizumab injections can provide greater visual improvement and reduced central retinal thickness more rapidly than laser alone.
The safety profile for ranibizumab was established in the three large randomized controlled clinical trials for neovascular age-related macular degeneration with no significant systemic effects at 2 years for patients treated with ranibizumab compared with controls [Abraham et al. 2010; Brown et al. 2009; Rosenfeld et al. 2006].
Bevacizumab
Bevacizumab is a full-length recombinant humanized antibody specific for all VEGF-A isoforms. Initially FDA approved for colon cancer treatment, bevacizumab is readily available and cost effective; it has been widely used for various ophthalmic conditions, including neovascular age-related macular degeneration, retinal vein occlusion, and diabetic retinopathy treatment.
The Diabetic Retinopathy Clinical Research Network studied diabetic macular edema eyes treated with intravitreal bevacizumab compared with focal/grid laser alone or a laser–bevacizumab combination. The results show improvement in central retinal thickness and visual acuity in eyes treated with bevacizumab compared with laser only, with a decrease in drug effectiveness between the 3- to 6-week postinjection period. Other trials have shown similar results [Goyal et al. 2010; Diabetic Retinopathy Clinical Research Network, 2007]. Faghihi and colleagues compared 130 treatment-naïve diabetic macular edema eyes randomized to intravitreal bevacizumab, bevacizumab/intravitreal triamcinolone, or focal/grid laser photocoagulation with similar results [Faghihi et al. 2008].
Recent data from the BOLT study, which randomized 80 eyes with nonischemic refractory diabetic macular edema with prior laser treatment to either intravitreal bevacizumab injections or macular laser treatment, demonstrated the efficacy of bevacizumab in the treatment of diabetic macular edema. Eyes receiving bevacizumab had improved visual acuity and reduced central retinal thickness over 1 year. Eyes required a median of nine bevacizumab injections compared with three macular laser treatments [Michaelides et al. 2010].
Studies in the systemic use of bevacizumab in combination with chemotherapy have shown an increased risk of thromboembolic events [US Food and Drug Administration (FDA) MedWatch, 2005]. However, the administered dose of bevacizumab in an intravitreal injection is 1.25 mg, 400-fold less than the systemic dose. A recent retrospective review of safety data in bevacizumab and ranibizumab use for age-related macular degeneration in Medicare beneficiaries showed no increase in mortality from myocardial infarction, bleeding, or cerebral vascular accident in comparison to prior FDA-approved therapies: photodynamic therapy (laser-activated intravenous verteporfin, a non-anti-VEGF treatment modality) and pegaptanib [Curtis et al. 2010].
VEGF Trap
VEGF Trap is a 115 kDa recombinant fusion protein with a combination of human IgG1 Fc domain fused to the binding domains for VEGF 1 and 2 receptors. In animal studies, VEGF Trap has been demonstrated to have a longer half life in the eye, and stronger binding affinity for VEGF. It has the potential to limit the frequency of intravitreal injections required to inhibit VEGF and the resulting neovascularization and breakdown of the blood–retinal barrier [Nguyen et al. 2006; Saishin et al. 2003].
Do and colleagues recently presented the results of phase II testing on the safety and efficacy of the VEGF-Trap intravitreal injection in eyes with diabetic macular edema; VEGF-Trap 0.5 mg or 2 mg injections were given monthly for three doses, followed by injections as needed or laser treatment. At 24 weeks, the injected eyes showed significantly improved visual acuity and reduced central retinal thickness compared with laser-treated eyes [Do, 2010]. Thus, VEGF-Trap may offer a longer-term alternative to monthly anti-VEGF injections.
Other medications
Anti-platelet agents
Studies have not shown a beneficial effect of aspirin (650 mg/day) on diabetic retinopathy progression [Chew et al. 1995; Early Treatment Diabetic Retinopathy Study Research Group 1991]. Patients receiving aspirin and dipyridamole in combination [Dipyridamole, Aspirin, Microangiopathy of Diabetes (DAMAD) Study Group, 1989] or ticlopidine [Ticlopidine Microangiopathy of Diabetes (TIMAD) Study Group, 1990] were shown to have a mild decrease in microaneurysms on fluorescein angiography compared with placebo; both may slow the progression of microaneurysm evolution in early diabetic retinopathy.
Lipid-lowering agents
Studies on fenofibrate monotherapy have demonstrated a potential effect of the drug in reducing diabetic retinopathy progression, and the need for laser treatment in CSME in patients with type 2 diabetes [Keech et al. 2007]. Other small, randomized controlled clinical trials have suggested a benefit of statins on reduction of diabetic retinopathy severity of diabetic retinopathy [Gupta et al. 2004; Sen et al. 2002], however the sample sizes in these studies were of insufficient power.
More recently, the Action to Control Cardiovascular Risk in Diabetes conducted a subanalysis of 1593 patients with type 2 diabetes with dyslipidemia and nonproliferative diabetic retinopathy to receive simvastatin (to reduce low-density lipoprotein cholesterol) in combination with either fenofibrate (reduction of triglycerides and increase in high-density lipoprotein cholesterol) or placebo. The rate of diabetic retinopathy progression at 4 years was significantly lower in the fenofibrate group (6.5%) in comparison to the placebo group (10.2%). The rates of moderate vision loss were similar between the groups: 16.0% in the fenofibrate group and 15.2% in the control group [Chew et al. 2010]. Thus lipid control with fenofibrate can slow the progression of diabetic retinopathy.
Protein kinase C inhibitors
Ruboxistaurin (Arxxant, Eli Lilly and Company, Indianapolis, Indiana) is a daily orally administered protein kinase C beta-isozyme-selective inhibitor. Microvascular alterations are thought to arise from hyperglycemia, which induces VEGF-mediated activation of protein kinase C. Animal models of diabetic retinopathy have shown that the protein kinase C beta-isoform activation results in neovascularization and retinal vascular permeability. This effect is reduced with oral ruboxistaurin [Aiello et al. 1997; Ishii et al. 1997; Xia et al. 1996]. Similarly, following laser coagulation treatment of the retina for neovascular disease, expression of protein kinase C was found to be markedly reduced in rabbit eyes [Ghosh and Gjörloff, 2005].
In a large randomized trial of 685 patients with diabetic macular edema, daily oral ruboxistaurin treatment (32 mg/day) was found to reduce vision loss, need for laser treatment, and diabetic macular edema progression, while increasing occurrence of visual improvement in patients with nonproliferative diabetic retinopathy over 3 years of follow up [Davis et al. 2009; PKC-DMES Study Group, 2007; PKC-DRS2 Group, 2006]. The trial is still ongoing to evaluate the effectiveness of this drug in diabetic macular edema.
Hyaluronidase
Intravitreal purified ovine hyaluronidase (Vitrase, ISTA Pharmaceuticals, Irvine, California) causes enzymatic vitreolysis. The dissolution of the vitreous is useful in the treatment of vitreous hemorrhage as a result of proliferative diabetic retinopathy, as the only two options currently available are observation or vitrectomy. Two large combined phase III randomized controlled clinical trials looked at 1125 eyes with vitreous hemorrhage of greater than 1 month duration and poor visual acuity (less than 20/200) treated with intravitreal ovine hyaluronidase compared with saline. The injected eyes had improved visual acuity and decreased vitreous hemorrhage density at 1 month which was sustained up to 3 months [Kuppermann et al. 2005a, 2005b]. Earlier clearing of the vitreous hemorrhage allows treatment (PRP) for the source of the bleeding. The most common adverse effect of this treatment was a self-limited iritis. This drug is still under evaluation for FDA approval.
Conclusion
In summary, there is strong evidence that tight glycemic and blood pressure control reduces the incidence and progression of diabetic retinopathy, reducing vision loss. In patients with proliferative diabetic retinopathy, PRP significantly reduces the risk of severe vision loss by 50%, especially in eyes with high-risk proliferative diabetic retinopathy. Early vitrectomy should be considered in patients with persistent vitreous hemorrhage from proliferative diabetic retinopathy. In patients with diabetic macular edema, there is compelling evidence that macular laser treatment reduces the risk of moderate vision loss by 50% and increases the chance of visual improvement. Intravitreal steroids have shown to be of benefit in refractory cases of diabetic macular edema. Ongoing studies have shown a promising role for intravitreal anti-VEGF agents in the treatment of diabetic macular edema, although current usage is off label.
Although diabetic retinopathy remains one of the leading causes of significant vision loss in the USA, primary and secondary interventions have been shown to be effective in reducing visual loss. Timely ophthalmoscopic screening and identification of high-risk patients remain the most effective tools to prevent vision loss in patients with diabetes.
Footnotes
Funding
This article received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors have no commercial affiliation or conflict of interest with the material presented.
