Abstract
Chronic wounds have become major clinical problems due to dysregulated inflammation, impaired angiogenesis, and abnormal extracellular matrix (ECM) remodeling. Noncoding RNA (ncRNA), including microRNA (miRNA), long noncoding RNA (lncRNA), and circular RNA (circRNA), serve as key molecules to regulate these pathological processes. These ncRNA also constructs a competitive endogenous RNA (ceRNA) network to precisely adjust miRNA activity and target gene expression. Through gene editing or chemical modification, exosomes achieve ncRNA delivery with high efficiency and targeting and show exciting promise in preclinical models. Combining with biomaterials such as hydrogels can prolong the exosome half-life and achieve the continuous release of ncRNA. In addition, researchers explore how machine learning (ML) and artificial intelligence (AI) could advance future therapeutic applications. Drug delivery system can be improved both by predicting personalized ceRNA networks for individual patients, and designing smart wound dressings that combine exosomes with hydrogel materials tailored to specific wound types. By integrating these recent advances, this review helps bridge basic research and the future creation of targeted chronic wound treatments.
Keywords
Introduction
The skin is the largest organ system in the human body and could effectively protect against mechanical damage, infection, and fluid and heat imbalances. After skin damage, wounds will form and start to heal. Wound healing is a complex process that requires a variety of cells to work together, with various cytokines and biomolecules involved. In general, this process can be divided into three main stages: inflammation, proliferation and remodeling. 1 The inflammatory phase is the beginning of wound healing, when the skin is injured, inflammatory cells are recruited to the site of wounded area. The proliferative phase is characterized by the accumulation of new cells and connective tissue. Keratinocytes (KCs) and endothelial cells become key players in this stage. The remodeling phase is the final step and requires a precise balance between apoptosis of existing cells and the generation of new cells. Alterations at any stage can affect normal wound healing under the influence of various internal and external factors. Chronic diseases such as diabetes reduce the rate of wound healing and create chronic wounds such as diabetic foot ulcers (DFUs), which cause microbial infections and often lead to amputations. 2 Excessive wound healing, including hypertrophic scars and keloids, often leads to itching and pain, which reduces quality of life. Traditional therapies often fail to cope with this complexity. This highlights the urgent need for new molecular understanding and targeted interventions.
With the rapid development of technologies such as qPCR, microarrays and next-generation sequencing, as well as the cross-application of molecular biology and big data science, endogenous noncoding RNAs (ncRNAs) are key contributors to the evolution and development of biological complexity by acting as regulators of gene expression at multiple epigenetic, transcriptional, and post-transcriptional levels.3,4 NcRNAs can generally be functionally divided into two categories, housekeeping ncRNAs and regulatory ncRNAs. regulatory ncRNAs, that is, long noncoding RNAs (lncRNAs), microRNAs (miRNAs), and circular RNAs (circRNAs) participate in wound-related processes by regulating different signaling pathways, thereby controlling the growth, differentiation, and apoptosis of various cells and tissues.5,6 For decades, ncRNAs have been shown to benefit wound healing by accelerating wound closure, modulating inflammation, enhancing epithelial reformation, increasing angiogenesis, and regulating extracellular matrix (ECM) remodeling.7–9 More and more literature shows that ncRNA, as a high-frequency keyword, is becoming a popular therapeutic candidate in the field of chronic wound healing.10,11 Notably, due to the inherent biological complexity of the ncRNA family, individual ncRNAs have only isolated effects on disease development, and it is necessary to decipher the coordinated interactions of multiple ncRNAs in the ncRNA network and to establish a fine network of competing endogenous RNAs (ceRNAs) mediated by ncRNAs in order to accurately target disease-related genes. 12 With the continued validation of the ceRNA network and the discovery of therapeutic targets and biomarkers for wound healing over the past decade, ncRNA-based therapeutic tools, and in particular miRNA-based products, have made great strides and are considered to be a promising component of the therapeutic marketplace. 13 These therapies are highly specific, precisely targeted and cost-effective, and hold great promise in the field of personalized medicine.
Despite significant advancements, the clinical translation of RNA therapeutics still depends on further interdisciplinary studies on delivery, stability, immune system activation and potential off-target effects. Exosomes have emerged in recent years as non-cellular therapeutic candidates to overcome these barriers due to their unique biological properties (low immunogenicity, low cytotoxicity, high stability, ability to cross the blood-brain barrier). 14 New treatment approaches like engineered exosomes, biomimetic vesicles, and exosome-biomaterial combinations aim to meet more personalized and customized clinical needs and improve exosome efficacy. To further support this narrative review, a comprehensive literature search was conducted in PubMed, Scopus, and Web of Science. The emphasis was on articles from the past 5 years, which highlight cutting-edge research. To improve retrieval sensitivity, the search strategy incorporated a combination of MeSH terms and free terms, including “exosomes,” “ncRNA,” “wound healing,” and other related keywords such as “DFU” and “skin regeneration.” Non-English articles and studies containing only sequencing data without functional validation were excluded. This review clarifies how wounds heal at the molecular level. It also helps identify biomarkers for predicting outcomes, find new treatment targets for chronic wounds and scarless healing, and improve exosomal ncRNA applications in tissue repair. This review integrates diverse ncRNA regulatory roles with advanced delivery systems and emerging technologies. It aims to establish a comprehensive framework that clarifies molecular mechanisms and connects basic science to translational applications in chronic wound therapy.
The roles of miRNA in the wound healing cascade
The 1993 identification of lin-4 in

Regulatory role of miRNAs in various stages of wound healing: (a) inflammation, (b) angiogenesis, (c) proliferation, and (d) remodeling. Major pathways, such as NF-κB, PI3K/Akt, MAPK/ERK, Wnt/β-catenin, TGF-β/SMAD can exert complex regulatory roles through miRNAs at different phases.
Inflammation
During the inflammatory phase of wound healing, a moderate inflammatory response is necessary to initiate tissue repair. However, excessive inflammation in the wound microenvironment greatly hinders the progress of healing. 26 NF-κB, a key transcriptional driver of tumor necrosis factor alpha (TNF-α) in macrophages, is regulated by PTEN—a known negative target of miR-21.27,28 PTEN enhances TNF-α expression through NF-κB activation, while miR-21 disrupts this pro-inflammatory positive feedback loop via PTEN-dependent silencing. 29 Programed cell death 4 (PDCD4), a pro-inflammatory tumorsuppressor, is another negative regulatory target of miR-21. 30 It boosts the pro-inflammatory pathway of NF-κB/TNF-α and modulates the c-Jun-AP1 pathway. These changes furthersuppressing the anti-inflammatory cytokine interleukin-10 (IL-10).29,31 This dual inhibitory mechanism establishes miR-21 as a central regulator of inflammatory homeostasis. It also predicts this molecule has broader functions in repair stages we’ll discuss later. In addition, multiple miRNAs (miR-19a/b, miR-20a, miR-223, miR-146a, and miR-132) create a synergistic anti-inflammatory effect by targeting different nodes of NF-κB signaling. miR-19a/b and miR-20a suppress Toll-like receptor 3 (TLR3)-mediated NF-κB activation through SHCSH2 domain-binding protein 1 (SHCBP1) and Semaphorin7A (SEMA7A) targeting, respectively. Together, they reduce the production of inflammatory chemokines and cytokines by KCs, including CXC chemokine ligand 8 (CXCL8), IL-8, and TNF-α. 32 miR-146a inhibits TLR2-induced inflammation by silencing critical NF-κB components such as IL-1 receptor-associated kinase 1 (IRAK1) and TNF receptor-associated factor 6 (TRAF6). 33 Overexpression of MiR-146a exerts anti-inflammatory and antioxidant effects by inhibiting reactive oxygen species (ROS) production in aged dermal fibroblasts (Fb). It alleviates age-related pathological burdens and fosters a pro-regenerative microenvironment. 34 Notably, cerium oxide-modified miR-146a enhances wound healing in diabetic Yorkshire pigs. 35 miR-132 mediates the downregulation of chemokines such as CXCL1, CXCL5, and IL-8 in KCs. It helps avoid persistent inflammation, which prevents wound healing from entering the proliferative phase. 36 Such timing-specific miRNA activity is key during the inflammation-to-proliferation transition. miR-223 could also suppress the classical NF-κB pathway via targeting TRAF6, TGF-β, and TAK1-binding proteins 1 (TAB1). 37 Collectively, these miRNAs exert anti-inflammatory effects. They modify upstream signals to inhibit NF-κB, thereby reducing the excessive production of pro-inflammatory cytokines and chemokines (e.g. IL-1β, IL-6, and TNF-α) by NF-κB. The mutual regulation of miRNA and NF-κB signaling pathway is essential to maintain the inflammatory balance during wound healing. Their dysregulation strongly contributes to chronic wound pathogenesis. Macrophages are another central inflammatory effector. After an injury, circulating monocytes are recruited to the wound site and differentiate into macrophages. Under the influence of pro-inflammatory and anti-inflammatory cytokines, mature macrophages are polarized and divided into pro-inflammatory M1 subtype or anti-inflammatory M2 subtype, which is called macrophage polarization. The M1 phenotype dominates the early inflammatory phase. It clears pathogens and debris by producing pro-inflammatory cytokines (e.g. TNF-α, IL-1β, IL-6) and ROS. M2 macrophages secrete anti-inflammatory cytokines (such as IL-10, TGF-β) and growth factors to support Fb proliferation, angiogenesis, and tissue remodeling in the later stages of the wound. 38 Therefore, promoting the transition of M1 macrophages to M2 macrophages is a crucial breakthrough in the treatment of chronic wound healing. miRNAs play a significant regulatory role in this process. Studies by Li et al. 39 demonstrate that miR-21 promotes Kruppel-like factor 6 (KLF6)-dependent M2 polarization, a process synergistically enhanced by apigenin. 40 More specifically, miR-9, miR-127, miR-155, and miR-125b have been shown to promote M1 polarization, whereas miR-124, miR-223, miR-34a, let-7c, miR-132, miR-146a, and miR-125a-5p induce macrophage M2 polarization through transcriptional and adaptor protein targeting. 41 Therefore, in-depth study of macrophage polarization and miRNA regulation helps reveal the molecular basis of wound healing. It also identifies therapeutic targets focus on macrophages. Through multilayered control such as gene silencing, pathway interactions, and cellular reprograming, miRNAs balance inflammation in tissue repair.
Proliferation
The proliferative phase of wound healing focuses on two goals: re-epithelization and vascular reconstruction. miR-21 relieves its inhibition of the PI3K/Akt pathway by targeting PTEN. 42 This activates VEGF expression to promote angiogenesis and drive cell proliferation, migration and collagen synthesis. 43 The mechanism has been validated in corneal epithelial repair via miR-21-enriched small extracellular vesicles (sEV) derived from umbilical cord mesenchymal stem cells (UCMSCs). 44 In addition, miR-21 participates in dendritic cell (DC) differentiation through the PTEN/PI3K/Akt axis. 45 DCs can trigger cell proliferation and accelerate wound healing by secreting proliferative factors. 46 This PTEN/PI3K/Akt/DC signaling establishes an immune-repair collaborative network. Thus, having helped to resolve the initial inflammatory response, miR-21 now directly promotes proliferation process. Another key miRNA, miR-17-5p, displays a broader effect. It protects endothelial cells from high glucose (HG)-induced damage and also exerts Fb protection by inhibiting neutrophil extracellular traps (NETs).47,48 When delivered through engineered exosomes combined with gelatin methacryloyl (GelMA) hydrogel, miR-17-5p silences p21 to delay cell aging via PTEN/PI3K/Akt pathway. Furthermore, it synergistically enhances angiogenesis and collagen deposition activation. 49 These roles highlight its clinical value as a multi-effect regenerative biomolecule. Innovative delivery systems have improved therapeutic outcomes significantly. Human adipose mesenchymal stem cell-derived exosomes (hADSC-Exos) carry miR-148a-3p, combined with PF-127 hydrogel, form a spatiotemporally controlled release system. This system optimizes the wound microenvironment and promotes neovascularization through PTEN/PI3K/Akt pathway modulation. 50 In contrast, miR-99 family members (miR-99a, miR-99b, miR-100) exert opposing regulatory effects on similar signaling pathways. Yi et al. 51 demonstrated that injury-induced downregulation of miR-99 family members activates mammalian target of rapamycin (mTOR), a key master regulator of cell growth and metabolism, as well as the upstream PI3K/Akt pathways. The activation of the PI3K/Akt/mTOR pathways stimulates the proliferation and migration of KC and accelerating wound closure.
During angiogenesis, engineered exosomes have unique delivery advantages. A study found that exosomes extracted from urinary source stem cells with hypoxia pretreated can carry miR-486-5p, which can be used to help angiogenesis by acting on serpin family E member 1 gene (SERPINE1). 52 There is also a synovial MSC-derived exosome (MSC-Exo) equipped with miR-126-3p, activating both MAPK/ERK and PI3K/Akt pathways. 53 This activation improved the growth and movement of skin blood vessel cells. However, interactions between signaling pathways make therapy more complex. Although miR-146a shows anti-inflammatory abilities as previously discussed, its overexpression unexpectedly hinders angiogenesis by inhibiting MAPK/ERK and PI3K/Akt pathways through ADP targeting. 54 This suggests the need for balancing modulation of miR-146a’s anti-inflammatory and pro-angiogenic effects in wound healing. To achieve this balance, targeted delivery of miR-146a inhibitors to endothelial cells via nanocarriers or engineered exosomes might be a solution.54,55 Consequently, the MAPK/ERK pathway is essential for fundamental cellular processes including proliferation, differentiation, and migration, making it a promising therapeutic strategy. 56 There are also studies that have found that if SRY-box transcription factor 2 (SOX2), a core transcription factor essential for maintaining cellular stemness in epithelial tissues, is overexpressed, it can promote the proliferation and migration of KCs through the EGFR/MEK/ERK pathway. 57 And if the m6A modification of miR-155 is blocked, it can also increase the level of SOX2. 58 Based on these findings, Gondaliya et al. 59 tried to load miR-155 inhibitors into MSC-Exo and found that the nanocarriers could promote angiogenesis and re-epithelialization. miR-21 also has multiple roles in this signaling pathway. Expression of miR-21 mimics via exosomal delivery can target both PTEN and Reversion inducing cysteine rich protein with kazal motifs (RECK), a negative regulator of matrix metalloproteinases (MMPs), in Fb. This targeting activates the MAPK/ERK pathway, which in turn promotes Fb function and angiogenesis.24,60 This approach significantly improves the quality of foot ulcer healing in a diabetic rat model. 61 Remarkably, engineered exosomes delivering miR-21-5p target the Wnt/β-catenin pathway to promote angiogenesis and collagen remodeling. 62 At the same time, exosomal miR-21-5p suppresses signal transducer and activator of transcription (STAT3), a transcription factor central to inflammation and cell survival, thus reducing MMP1 production. 63 The axis curbs excessive cell migration and minimizes scar formation, which lays the molecular foundation for scarless healing. Collectively, miR-21 coordinates wound healing processes across inflammatory and proliferative phases via multiple pathways including PTEN/NF-κB/TNF-α, PTEN/PI3K/Akt, and PTEN/MAPK/ERK.
Some miRNAs can have a negative effect on the proliferation. While Wnt/β-catenin signaling promotes cell proliferation and matrix remodeling, 64 inhibition of its key components by miR-124 slows the healing process. 65 In a HG environment, miR-29c-3p inhibits the activation of the Wnt signaling pathway by downregulating platelet-derived growth factor type BB (PDGF-BB), leading to impaired function of endothelial progenitor cells (EPCs). 66 Similarly, circulating exosomal miR-20b-5p inhibit angiogenesis in human umbilical vein endothelial cells (HUVECs) by disrupting normal Wnt9b/β-catenin signaling. 67 miRNA-24-3p, also from circulating exosomes, prevents HUVEC migration and tube formation by targeting phosphatidylinositol-3-kinase regulatory subunit 3 (PI3KR3). 68 It delays wound closure in full-thickness skin graft models and highlights the importance of microenvironmental regulation. miR-195-5p and miR-205-5p in wound fluid from ulcerative lesions of DFU patients negatively regulate angiogenesis in DFU patients by directly targeting VEGFA. 69 In addition, miR-210 hinders ischemic wound healing by silencing E2F3, a transcription factor essential for KC proliferation. 70 These findings suggest that it may be a good idea to develop treatment based on different miRNA expressions in each patient. The regulatory roles of miRNAs during wound proliferation phase offer great therapeutic potential.
Remodeling
Tissue remodeling in the final stages of wound healing faces key challenges in repairing damaged tissue while preventing abnormal scarring. During this phase, the ECM produced by Fb provides the cells with a suitable microenvironment, where type III collagen gradually transitions to type I collagen. 71 Driven by the TGF-β/Smad signaling pathway, Fb differentiates toward myofibroblasts, which further promotes ECM secretion. Hypertrophic scar (HS) is easily caused when ECM is over deposited. 72 Microarray and RT-PCR analyses reveal dysregulated miRNA profiles in HS tissues, with 92 miRNAs (e.g. hsa-miR-564, hsa-miR-936) upregulated and 13 miRNAs (e.g. hsa-miR-451, hsa-miR-223) downregulated. 73 It implicates that miRNA is closely related to the occurrence and evolution of HS. Specifically, miRNAs such as miR-128-1-5p, miR-138-5p, miR-146a-5p, miR-141-3p, and miR-29 all reduce pathologic scar formation in the skin, whereas miR-21 and miR-26b-5p promote fibrosis in the skin. Many signaling pathways mediate HS formation and promotion by inducing cell proliferation and inhibiting apoptosis. The TGF-β/Smad pathway, a central regulator of collagen synthesis in Fb and myofibroblasts, becomes dysregulated during fibrotic progression. 72 miR-128-1-5p can reduce skin scar formation in diabetic rats by inhibiting TGF-β1 and further decreasing the phosphorylation of Smad2/3. 74 Meanwhile, miR-138-5p and miR-146a-5p exert their effects by targeting sirtuins 1 (SIRT1) and C-X-C chemokine receptor type 4 (CXCR4), respectively. SIRT1 is a key deacetylase regulating cellular stress, while CXCR4 acts as a critical receptor for stem cell homing and leukocyte recruitment. These mechanisms work together to inhibit the abnormal proliferation of Fb and pathological remodeling of ECM.75,76 Therefore, miR-146a-5p exhibits a multiple functional balance of anti-inflammatory, delayed angiogenesis and anti-scarring requirements during wound healing. It needs precise regulation of expression level according to the type of wounds (e.g. burns, surgical incisions, diabetic ulcers) and the individual patient’s differences to design a personalized miR-146a-5p therapeutic regimen. For instance, treating diabetic ulcers focuses on controlling inflammation and boosting blood vessel growth, while burn care requires stronger efforts to prevent excessive scarring. miR-663a and miR-141-3p emerge as potential HS therapeutics by targeting TGF-β/Smad signaling to regulate Fb distribution, collagen alignment, and ECM deposition.77,78 miR-141-3p, delivered via dissolvable microneedle arrays, achieves sustained release in HS tissues. 78 Conversely, miR-21 presents a more complex function. While it is known for effectively resolving inflammation and promoting proliferation, this same molecule can drive pathological fibrosis by amplifying TGF-β1 signaling through Smad7 suppression. 25 miR-21 inhibitors or targeting the TGF-β1/miR-21/Smad7 axis could be promising strategies for HS and fibrotic diseases. The potential of miR-21 as a therapeutic target is being explored in several research areas, especially in cancer, fibrotic diseases, and wound healing. Current researches are exploring miR-21-based gene editing, nanocarriers, and cell therapies for chronic wound management.79,80 Within the fibrosis regulatory network, the miR-29 family (miR-29a, miR-29b, miR-29c) antagonizes miR-21’s fibrotic effects by targeting at least 16 genes associated with the ECM, acting as suppressors of scar formation. 81 Levels of miR-29, especially miR-29a are significantly reduced in keloid compared to healthy Fb. Knockdown of miR-29a results in increased expression of type I and type III collagen mRNAs and proteins in Fb. 82 More specifically, miR-29a derived from hADSC-Exo suppresses HS by inhibiting TGF-β2/Smad3 activation and downregulating α-smooth muscle actin (α-SMA), collagen I, and collagen III. 83 A Phase 1 clinical trial tested remlarsen, a synthetic miR-29b mimic. This double-blind, placebo-controlled study found remlarsen reduces collagen production and scar tissue growth in skin wounds, thereby preventing fibrosis in skin wounds. 84 In addition, activation of TGF-β downregulates miR-29, upregulating ECM proteins that drive fibrosis. 85 The role of the PI3K/Akt pathway in the fibrotic process should not be overlooked. Its activation promotes the synthesis of the ECM and cell proliferation and survival, which in turn drives the fibrotic process.86,87 miR-26b-5p enriched in hypoxic macrophage-derived exosomes, could exacerbate keloid progression by regulating the proliferation, migration, and invasive ability of keloid fibroblasts (KF) through PTEN/PI3K/Akt pathway. 88 Collectively, miRNAs critically regulate collagen dynamics, ECM remodeling, and Fb differentiation during tissue remodeling. Future miRNA-based therapies are expected to achieve scarless healing and functional tissue restoration through expression modulation. To provide a clearer overview of the complex role of miRNAs in chronic wound, the key molecules discussed in this section are summarized in Table 1. This table highlights their downstream targets and primary functions at different stages of healing. The table shows that miRNAs function as a coordinated network rather than in isolation. miRNAs such as miR-21 and miR-146a exhibit different effects. Their roles are highly dependent on the specific state and time phase of the wound healing process, emphasizing the importance of precise modulation.
Regulatory role of key miRNAs in chronic wound healing.

Emerging frontiers in miRNA therapeutics
In recent years, studies combining molecular biology, genetics, pathology, oncology, cardiovascular science, and neuroscience have pushed miRNA research to the forefront. It has been evidenced by exponential growth in publications and increasing patent filings and funding allocations over the past two decades.
89
The unique gene-regulatory capacity and extracellular delivery potential of miRNAs make them powerful tools for disease biomarker and targeted therapies. Some call them the “crown jewel” of molecular medicine. Technological advancements further expand miRNA applications in translational medicine. Emerging sequencing platforms, genome-editing tools, and artificial intelligence (AI) -driven big data analytics have broadened research horizons of miRNA.
90
For example, clustered regularly interspaced short palindromic repeats (CRISPR)/Cas9 systems enable precise miRNA editing in donor cells to modulate miRNA profiles.
91
With the help of machine learning (ML) algorithms and deep learning models, researchers can more accurately predict the miRNA-target interactions and achieve personalized treatment through miRNA expression profiling.
92
This journey highlights miRNA’s scientific importance and medical value, transitioning from bench to bedside within two decades. Current miRNA therapies mainly use chemically modified antisense inhibitors (anti-miRs) or synthetic mimics (overexpression of miRNAs). These treatments are delivered through localized injections or skin patches to target wounds directly.
93
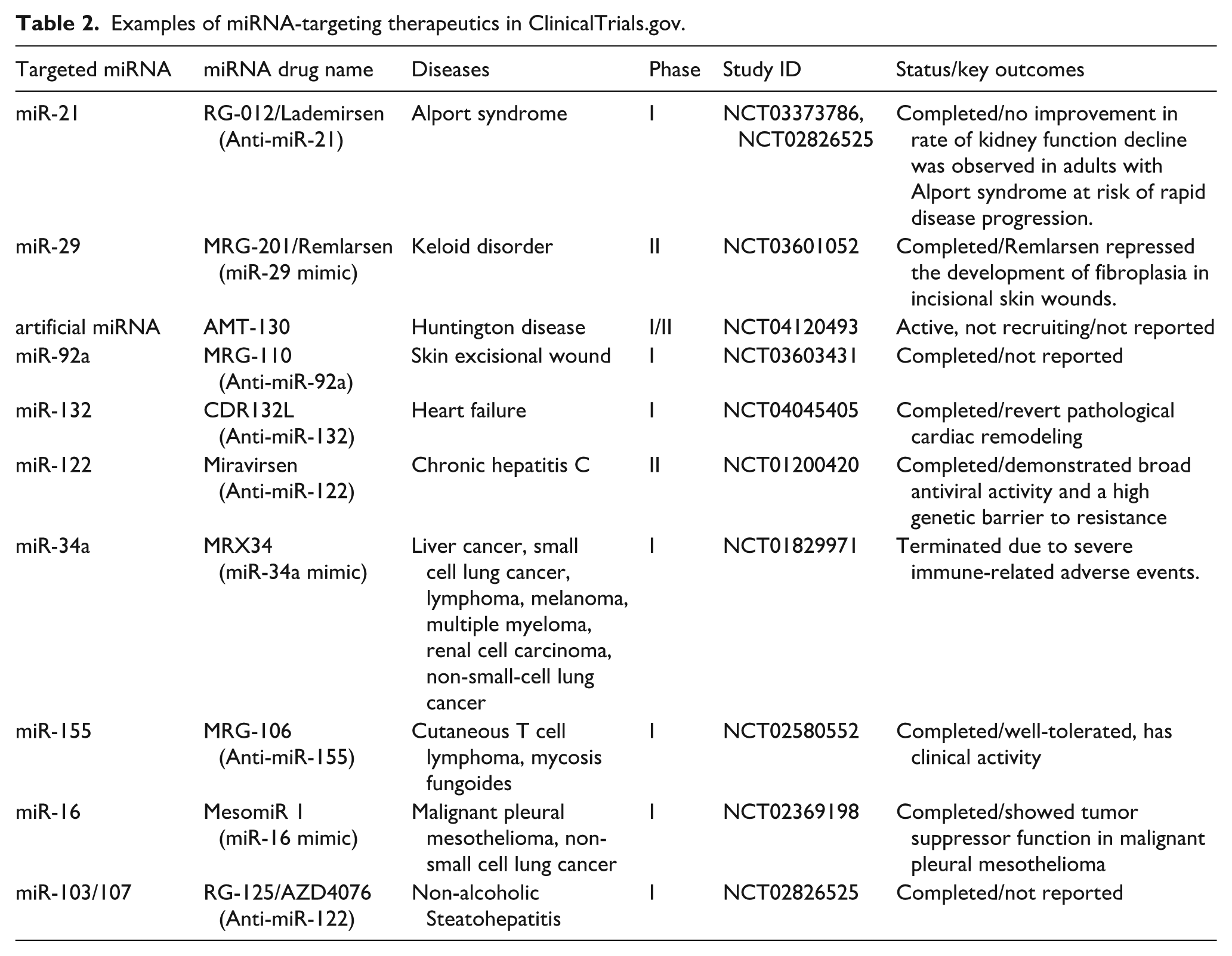
To clarify the prospects of miRNA-targeted therapy, we screened representative clinical trials from
Examples of miRNA-targeting therapeutics in ClinicalTrials.gov.
The roles of lncRNA in the wound healing cascade
The discovery of miRNAs in the 1990s set the stage for the subsequent rapid identification of lncRNAs over the following 25 years. The development of lncRNAs research demonstrates how technological advancements drive scientific understanding. Initially regarded as “transcriptional noise,” lncRNAs are now recognized as multifunctional regulators of gene expression, a realization driven by advances in sequencing technologies. 99 Defined as transcripts exceeding 200 nucleotides, lncRNAs participate not only in cell proliferation and differentiation, but also manage complex gene networks through epigenetic modifications, chromatin remodeling, and post-transcriptional mechanisms.100,101 Notably, lncRNAs can act as oncogenes or tumor suppressors, exerting precise regulatory roles in cancer initiation and progression. 102 Distinct from the highly conserved targeting mechanisms of miRNAs, lncRNAs show unique molecular interaction characteristics. They regulate gene expression at epigenetic, transcriptional, and post-transcriptional levels by interacting with DNA, chromatin-modifying complexes, transcriptional regulators, or regulating the stability of mRNAs and the efficiency of translation.103,104 More specifically, lncRNAs act as ceRNAs by acting as miRNA “sponges” and reducing miRNA targeting to mRNAs. 105 This theory has also been established as a novel post-transcriptional regulatory paradigm with a fine-tuning effect in wound healing. Emerging evidence highlights the critical regulatory functions of lncRNAs in chronic wounds. Key molecules being investigated include H19, MALAT1, GAS5, XIST, and WAKMAR. Comparative analyses by Yu et al. 106 revealed 58 upregulated and 42 downregulated lncRNAs in DFU skin samples compared to healthy controls. Similarly, Tu et al. 107 identified 7 upregulated and 7 downregulated angiogenesis-related lncRNAs in HG-cultured human dermal fibroblasts (HDF). These findings emphasize the prognostic potential of lncRNAs in diabetic wound pathophysiology, particularly DFU. In the future, with the deep integration of multi-omics analyses and synthetic biology technologies, lncRNAs are expected to become revolutionary therapeutic targets in wound repair after miRNAs.
Inflammation
While miRNAs exhibit well-defined roles in inflammatory regulation, how lncRNAs regulate macrophage polarization remains an evolving field. H19, the first imprinted lncRNA discovered in 1990,
108
has attracted sustained attention for its roles as a tumor suppressor, oncogene, and regulator of mammalian embryonic development.
109
Recent studies highlight its involvement in chronic wound healing. Peroxisome proliferator-activated receptor gamma (PPARγ) activation is known to drive monocyte differentiation into M2 macrophages.
110
ADSC-derived exosomal H19 enhances M2 polarization by downregulating miR-130b-3p, thereby upregulating PPARγ and STAT3 to accelerate cutaneous repair.
111
Concurrently, H19 strengthens macrophage infiltration in the early stage of wound healing by suppression of Fb-derived growth and differentiation factor 15 (GDF15), accelerating tissue regeneration.
112
The NOD-like receptor family pyrin domain containing (NLRP3) inflammasome is often overactivated in diabetic wounds and HG-induced macrophages, exacerbating inflammatory pathology.
113
H19-enriched exosomes from hair follicle mesenchymal stem cells (HF-MSCs-Exo) reduce diabetic skin inflammation by inhibiting NLRP3 inflammasome activation.
114
However, the effectiveness of this healing promotion depends on the circumstances. Cheng et al.
115
demonstrated that H19 inhibition alleviates HG-induced endothelial inflammation and oxidative stress via the miR-29b/VEGFA/Akt/eNOS axis, underscoring the need to further investigate its cascading regulatory networks.
116
This bidirectional functionality suggests that H19 may be a dynamic modulator of inflammatory homeostasis. This regulation is not unique, as other classical lncRNAs are equally multifunctional. XIST, initially linked to X-chromosome inactivation (XCI) in 1991,
117
promotes M2 polarization via miR-19b suppression and subsequent IL-33 upregulation. This axis also enhances human skin Fb proliferation, migration, and ECM synthesis.
118
Thus, the XIST/miR-19b/IL-33 axis plays a key role in all three phases of inflammation, proliferation and remodeling in the repair of degenerated dermis after thermal injury. Following XIST, technological advances revealed additional fundamental lncRNAs. This period saw the discovery of lncRNA NEAT1 and NEAT2 (also known as MALAT1). They are named for their high abundance in the nucleus and regulate nuclear architecture and transcriptional programs.
119
As a popular and highly conserved lncRNA, MALAT1 is noted as one of the most abundant RNAs in the nucleus. Its discovery emerged from cancer research, where it was found to play a regulatory role in cancer metastasis, cell migration and cell cycle.
120
This foundational role in basic cell processes makes MALAT1 a pivotal regulator in multiple pathologies, including wound healing, as discussed in later sections. Numerous studies have shown that MALAT1 is a pro-inflammatory RNA.121,122 However, a new study reported its opposite role by polarizing macrophages. Kuang et al.
123
reported that MALAT1 downregulates miR-1914-3p to elevate Milk fat globule-epidermal growth factor 8 (MFG-E8) expression, a critical inhibitor of TGFβ1/SMAD3 signaling. In addition to alleviating inflammation by inducing M2 macrophage polarization and inhibiting NLRP3 inflammasomes,124,125 MFG-E8 could also promote angiogenesis and improves Fb migration in wound healing.126,127 As mentioned previously, the TGFβ1/SMAD3 pathway can exacerbate wound fibrosis, so it is reasonable to hypothesize that the MALAT1/miR-1914-3p/MFGE8 pathway can also reduce scar formation during wound healing by downregulating TGFβ1/SMAD3. Indeed, MALAT1 functions beyond inflammation by also promoting proliferation and reducing fibrosis. These roles will be detailed later in Sections
Proliferation
During the proliferative phase of wound healing, lncRNAs including H19, MALAT1, WAKMAR2, XIST, GAS5, and HOTAIR collectively regulate critical processes such as Fb and KC proliferation, angiogenesis, and re-epithelialization. Among them, the regulation of PI3K/Akt signaling pathway was particularly prominent. This pathway regulates angiogenesis, and its dysfunction directly contributes to delayed healing in diabetic wounds. HG levels impairs the insulin-PI3K-Akt cascades, severely weakening the body’s ability to regenerate blood vessels. 137 IGF1R, a key initiator of this pathway, activates PI3K/Akt signaling through tyrosine kinase activity to drive cellular proliferation, differentiation, and migration. 138 Hypoxic microenvironment studies reveal that hypoxia-inducible factor-1α (HIF-1α) activates PI3K/Akt signaling by suppressing LINC02913 transcription, thereby alleviating IGF1R downregulation and enhancing angiogenic functions. 139 This HIF-1α/LINC02913/IGF1R axis is similar to the regulatory mechanism mediated by H19. H19 directly activates PI3K/Akt to promote endothelial function, 140 while enhancing the methylation modification of HIF-1α histone by recruiting enhancer of zeste homolog 2 (EZH2). 141 As a master regulator of hypoxia adaptation, HIF-1α activates downstream pro-angiogenic gene expression to accelerate cutaneous wound repair. 142 Notably, in mouse pulmonary artery endothelial cells, H19 can also relieve its inhibitory effect on HIF-1α by acting as a sponge for miR-20a-5p. 143 It creates a synergy between epigenetic regulation and miRNA sponging. As a result, H19 integrates multiple signaling pathways to support angiogenesis, cell proliferation, and migration, offering comprehensive therapeutic support. It has been found that the use of extracellular vesicle-mimetic nanovesicles (EMNVs) delivering H19 exhibits scalable production advantages. 140 It is expected to be an effective nanodrug delivery system for lncRNAs. In particular, the lncRNA/HIF-1α regulatory axis provides critical insights into wound healing mechanisms. 144 For instance, MALAT1 from hADSCs stimulates Fb proliferation and migration through synergistic activation of HIF-1α/VEGF signaling and a MALAT1/nuclear factor-erythroid 2-related factor 2 (Nrf2)/HIF-1α positive feedback loop.145,146 Nrf2, an antioxidant transcription factor, may reinforce this loop by reducing ROS-induced inflammation via the Nrf2/VEGF axis. 147 In addition, KLF2 has been shown to promote the proliferation and migration of traumatic vascular endothelial cells. 148 MALAT1 could relieve the inhibitory effect of miR-92a on KLF2, thus upregulating KLF2 expression and enhancing neovascularization. 149 This multi-target regulation pattern was more prominent under oxidative stress conditions. The exosomal NEAT, derived from oxidative stress-induced endothelial cells-derived exosomes, activates Wnt/β-catenin signaling to boost EPCs angiogenesis and random skin flap survival. 150 Corresponding to is the regulation of re-epithelialization. WAKMAR2 serves as a key promoter of KC migration, with its knockdown resulting in the blockage of re-epithelialization in isolated cutaneous wounds. 136 Li et al. 151 further demonstrated that WAKMAR1 suppresses methylation of the E2F Transcription Factor 1 (E2F1) promoter by isolating DNA methyltransferases (DNMTs), accelerating KC migration in human ex vivo wound models. XIST, beyond its role in miR-19b/IL-33-mediated HSF regulation, enhances HG-induced human immortalized keratinocytes (HaCats) proliferation and migration via the miR-126-3p/EGFR axis. 152 This aligns with the classical mechanism of EGFR signaling, which coordinates multicellular functions through the PI3K/Akt pathway. 153 GAS5 modulates both proliferation and survival. 154 In chronic wounds, activation of the transcription factor cellular-myelocytomatosis viral oncogene (c-Myc) leads to depletion of epidermal stem cells. As a proto-oncogene driving cell cycle progression and proliferation, the activity of c-Myc in this context consequently establishes itself as a biomarker of chronic wounds. 155 GAS5 binds to c-Myc mRNA to block its translation and effectively reverses the depletion of epidermal stem cells in chronic wounds. 156 GAS5 could also promote HG-induced endothelial cell proliferation through activation of the HIF-1α/VEGF pathway mediated by the TATA cassette-associated factor 15 (TAF15). 157 GAS5 operates in both inflammatory and proliferative phases. Its ability to regulate multiple signaling pathways across cell types and environments highlights its multifaceted role, offering new therapeutic targets for diabetic wounds. In addition to being activated by GAS5, the HIF-1α/VEGF pathway is also positively regulated by lncRNA ANRIL. ANRIL positively regulates the stability of HIF-1α mRNA, which in turn regulates VEGFA to promote angiogenesis in vivo. 158 The study also found that HIF-1α transcriptionally controls ANRIL and VEGFA in EPCs. This interaction forms a feedback loop, enabling EPCs to adapt to hypoxic environment. Also of interest is the bidirectional regulatory properties of HOTAIR in wound repair. HOTAIR is a well-characterized lncRNA involved in cell proliferation, apoptosis, migration, and cancer cell invasion, 159 and has been shown to play an important role in the maintenance of cellular stemness. 160 HOTAIR shows cell-type-specific duality in wound healing. In epidermal stem cells, overexpressing HOTAIR enhances stemness and re-epithelialization. 161 in blood vessel cells, it exerts anti-angiogenic effects via sponging miR-126. 162 This precise control across time and space highlights lncRNA networks as complex molecular hubs. By coordinating miRNAs, gene-modifying proteins, and signaling pathways, lncRNAs dynamically balance proliferative phase progression, offering new breakthroughs for the development of therapeutic strategies.
Remodeling
Collagen, the principal component of ECM, serves as both a structural scaffold for cellular proliferation and migration and a key material basis of skin functional integrity. 163 A novel lncRNA was found to be highly expressed in diabetic skin, named lnc-URIDS. lnc-URIDs indirectly inhibit the synthesis of collagen, especially collagen I, by binding to procollagen-lysine 1,2-oxoglutarate 5-dioxygenase 1 (Plod1) to reduce its stability. This process exacerbates abnormal collagen metabolism in diabetic skin, leading to delayed wound healing. 164 Conversely, lncRNA-ASLNCS5088, enriched in M2 macrophage-derived exosomes, acts as an endogenous sponge for miR-200c-3p in Fb to upregulate α-SMA and collagen expression. 165 The process was reversed with the exosome inhibitor GW4869. lncRNAs also play a multiple regulatory role in pathologic scar formation. From the regulation of Fb proliferation, abnormal deposition of ECM to related gene expression networks, there are molecular imprints of lncRNA activity. As a representative example, the lncRNA H19 has been demonstrated to participate in fibrotic diseases such as pulmonary and hepatic fibrosis. 166 It suggests the potential pro-fibrotic role of H19 in pathological scar formation. This hypothesis is validated in keloid studies. The mTOR signaling pathway contributes to keloid development. 167 H19, which is overexpressed in keloid tissues and Fb, drives pathologic keloid progression through activation of the mTOR signaling pathway and up-regulation of VEGF expression. 168 Knockdown of H19 significantly inhibits Fb proliferation and excessive ECM deposition. As a central hub of fibrosis regulation, TGF-β signaling pathway-associated lncRNAs are particularly prominent. Functioning as a TGF-β-activated effector, lncRNA-ATB promotes TGF-β2 self-secretion by sponging miR-200c. 169 This creates a loop that continuously stimulates scar tissue growth. Similarly, LINC01116 reinforces TGF-β1/Smad3 signaling via miR-3141 inhibition, positively facilitating KF migration and subcutaneous scar growth in vivo. 170 In contrast to the mechanisms described above, the discovery of maternally expressed gene 3 (MEG3) provides a new direction for therapeutic strategies. MEG3, downregulated in keloids, exerts anti-fibrotic effects by activating the TP53 pathway to suppress KF activity and collagen deposition when delivered via BMMSC-Exo. 171 In the remodeling phase, lncRNAs ensure balanced synthesis and breakdown of ECM by regulating key molecules and pathways. This improves tissue structure and functional recovery while preventing pathological scarring. The multiple regulatory functions of hot lncRNAs in wound healing are summarized in Table 3. These lncRNAs often act as central hubs that integrate signals from multiple pathways. Unlike miRNAs, lncRNAs act more like versatile platforms. They can interact with chromatin modifiers, transcription factors, and miRNAs at the same time. This structural feature allows a single lncRNA to coordinate multiple processes from gene expression to cell state. This context-dependent characteristic is both an attractive feature and a major challenge for lncRNA therapy.
Regulatory role of key lncRNAs in chronic wound healing.

Emerging frontiers in lncRNA therapeutics
Novel RNA-targeting technologies such as CRISPR/Cas13 are accelerating the resolution of lncRNA function. Unlike protein regulators, they exert translation-independent effects. The feature enables them to respond rapidly to changes in the microenvironment and precisely coordinate the spatial and temporal dynamics of gene expression programs. 172 Recent advancements in RNA-targeting technologies, such as CRISPR/Cas13, have accelerated the process of lncRNA function resolution. Through large-scale screening of more than 6200 lncRNAs, researchers have identified 778 lncRNAs with cell type-specific or broad-spectrum functions. 173 lncRNAs regulate genetic information to dominate the entire healing process from inflammation to remodeling, including epidermal regeneration, angiogenesis, and ECM remodeling. It reveals intricate balances in cellular state dynamics. Clinically, lncRNAs’ bidirectional regulation offers new opportunities for targeted therapies. Most current drugs may already have lncRNA binding sites, which lays the foundation for the development of targeted drugs. For example, melatonin inhibits MALAT1-mediated NLRP3 inflammasome activation and TGF-β1/Smad signaling to reduce cardiac fibrosis in diabetic cardiomyopathy. 174 Topical mevastatin stimulates KCs to secrete GAS5-enriched exosomes that suppress c-Myc expression, accelerating DFU healing. 175 In particular, the tissue-specific expression patterns and miRNA/mRNA network-regulating capacity of lncRNAs could reduce off-target effects compared to traditional therapies. 176 However, clinical translation faces challenges including low sequence conservation (except rare cases like MALAT1 and H19), inefficient in vivo delivery systems, and complex mechanisms due to intricate regulatory networks. 177 These challenges may explain the absence of lncRNA-based clinical trials for chronic wounds. In the face of these issues, the integration of cutting-edge technologies is generating solutions. Progress in nanocarrier-based delivery systems is particularly notable. Smart nanoparticle-based delivery systems enable localized, targeted lncRNA delivery without viral toxicity. 178 This “carrier-ncRNA” strategy overcomes conservation limitations and allows dynamic modulation of the wound microenvironment. With the deep application of single-cell sequencing and spatial transcriptome technology, it is expected to construct a specific regulatory map of lncRNAs in trauma repair in the future, laying the foundation for precision therapy.
The roles of circRNA in the wound healing cascade
The research history of circRNAs can be traced back to 1976, when they were first discovered and identified in plant-like viruses. 179 By the 1990s, circRNAs were identified and characterized as molecules produced from endogenous RNAs, 180 gradually emerging as critical subjects in ncRNA research. However, due to the predominance of linear RNAs in cells and technological limitations, circRNAs were initially considered as by-products of RNA splicing. Since then, with the advent of next-generation sequencing technology and related bioinformatics tools, circRNA research has gained substantial attention since 2010. The biological properties of circRNAs have been gradually revealed. circRNAs are closed covalent circular RNAs formed by reverse splicing of pre-mRNAs. 181 This circular molecule’s unique structure has challenged the traditional belief of the linear RNA world. The advantage of circRNAs is that they are not affected by RNA exonucleases. They also have tissue/cell-specific expression, structural stability, and sequence conservation. 182 Being widely expressed in mammalian cells, circRNAs can also be detected in extracellular body fluids and in exosomes. 182 This suggests that they are expected to serve as biomarkers for noninvasive diagnosis. Functionally, circRNAs regulate gene expression by modulating transcriptional processes in the nucleus and interacting with protein factors.183,184 Like lncRNAs, circRNAs can also act as miRNA sponges to build ceRNA networks and inhibit miRNA-mediated gene silencing. A seminal example is circCDR1as (also known as ciRS-7), which contains over 60 complementary binding sites for miR-7 and can tightly regulate the availability of miR-7. 185 This discovery established the classic paradigm of the “miRNA sponge” and led to an explosion of related research. In the past few years, the number of circRNAs reported in academic journals has increased almost exponentially. Star molecules including circCDR1as, circHIPK3, and circMbl have been identified, most of which act as miRNA sponges to function. 186 New research reveals circRNAs’ promising medical applications in vaccines, 187 cancer treatment, 188 protein therapy, 189 and wound healing. 190
Inflammation
Endothelial cells form the innermost vascular layer, play critical roles in regulating blood-tissue exchange, vasodilatory tension, and blood flow. Chronic HG conditions exacerbate oxidative stress, apoptosis, and inflammatory factor expression, leading to endothelial dysfunction. 191 Studies demonstrate that inhibition of NF-κB and NLRP3 inflammasome pathways effectively reduces HG-induced oxidative stress and inflammation. 192 TLR4 drives diabetes development by activating these pathways. 193 Cheng et al. 194 revealed that hsa_circ_0068087 acts as a key regulator in TLR4/NF-κB/NLRP3 signaling. Silencing this molecule reverses HG-induced HUVEC dysfunction and inflammation by suppressing TLR4 overexpression. Adipocyte-derived exosomal circ_0075932 interacts with the RNA-binding protein Pumilio2 to activate the Aurora A/NF-κB pathway, promoting KC inflammation and apoptosis. 195 These findings reveal a new “RNA-protein-kinase” system for NF-κB signaling modulation. The above studies demonstrated the regulatory role of circRNAs in cellular inflammation. Diabetic wound models further reveal circRNA’s medical value. In vivo delivery of circ-Snhg11 enhances wound healing in diabetic mice by suppressing HG-induced secretion of IL-6, IL-1β, and TNF-α. Circ-Snhg11 also promotes STAT3 expression and acts on major effector cells in inflammation by regulating the miR-144–3p/STAT3 axis, thereby driving M2-like macrophage polarization. 196 Similarly, circRps5 overexpression accelerates murine wound healing by sponging miR-124-3p to induce M2 polarization, transitioning wounds from inflammatory to proliferative and remodeling phases. 197 Both studies applied ADSC-Exo for circRNA delivery. It suggests that exosomes, as biocompatible carriers, can significantly affect the intensity and duration of inflammatory responses by delivering circRNAs. These findings provide a technical paradigm for overcoming delivery barriers in wound management, emphasizing the therapeutic promise of exosomal circRNA systems.
Proliferation
Fbs in the skin primarily cover injuries with granulation tissue during the proliferative phase, a process modulated by circRNAs. In a murine excisional wound model, Yang et al. 198 demonstrated that circ-Amotl1 overexpression promotes Fb proliferation and migration by sponging miR-17-5p to elevate STAT3 protein levels. Mice injected with circ-Amotl1 exhibited increased cellular density and accelerated wound healing. Mediating epidermal stereotyping, development and differentiation is a key role of the transcription factor p63, which has been widely demonstrated by mouse models. 199 Su et al. 199 found that circAMD1 modulates functional and phenotypic alterations in p63-mutant HDFs by suppressing miR-27a-3p. Overexpression of circAMD1 enhanced proliferation, collagen synthesis, and myofibroblast differentiation in p63-mutant HDFs. This study suggests that circRNAs may be involved in the individualized repair mechanism under the genetic background differences. In UVB stress-induced premature senescence (UVB-SIPS) models, circRNA_100797 was found to accelerate Fb proliferation and reduce cell cycle arrest, revealing its role in light-damaged skin repair. 200
Proliferation and migration of epidermal KC are essential for wound re-epithelialization. The impaired migratory capacity of KC and abnormal overgrowth at the wound margin lead to the progression of DFU. 201 Dysregulation of circRNAs contributes to this pathological process. Upregulated circRNA-0080968 in DFU degrades miR-326 and miR-766-3p, suppressing KC migration while promoting its proliferation. 202 This contradictory effect indicates circRNA’s complex control over repair processes. hsa_circ_0084443 acts as a negative regulator of KC migration. 203 He and Xu 204 further demonstrated that overexpression of hsa_circ_0084443 inhibits TGF-β1-induced KC migration and proliferation via the miR-17-3p/ Forkhead box O 4 (FOXO4) axis. Knockdown of circ_PRKDC, the parental gene of hsa_circ_0084443, promotes KC migration through the miR-31/fibrillin 1 (FBN1) axis. 205 circ_PRKDC also directly targets miR-20a-3p to regulate RAS p21 protein activator 1 (RASA1) expression, inhibiting HaCaT KC migration. 206 This bidirectional regulation implies circRNAs act as molecular switches to precisely regulate cellular behavior at different stages of epidermal repair. Huang et al. 207 identified circCDK13 (also named hsa_circ_0079929) directly interacts with insulin-like growth factor 2 mRNA-binding protein 3 (IGF2BP3) to form a circRNA-protein-mRNA ternary complex. This circRNA promotes the proliferation and migration of HDF and KC by upregulating c-Myc. This study also constructed engineered small extracellular vesicles overexpressing circCDK13 and demonstrated that circCDK13 could promote wound healing in diabetic mice and diabetic rats.
Active angiogenesis is also positive for wound repair. circRNAs regulates endothelial cell function through multiple dimensions. Differentially expressed circRNAs are known to be closely associated with HG-induced endothelial cell damage. circ-IGF1R promotes angiogenesis in ulcerated tissues by down-regulating miR-503-5p to activate the HIF-1α/VEGFA axis. 208 At The hexokinase activity regulated by circ-IGF1R may improve impaired glucose metabolism in diabetic wounds. In the previously mentioned Nrf2/VEGF axis, circHIPK3 enhances angiogenesis under HG condition by sponging miR-20b-5p to relieve Nrf2 suppression. 209 EPCs are precursor cells of vascular endothelial cells that can be directed to the site of ischemic injury. There they form new blood vessels through proliferation and differentiation. 210 circ-Erbb2ip inhibits EPC damage and reduces inflammation in diabetic mice by targeting the miR-670-5p/Nrf1 pathway. 211 These findings suggest circRNAs integrate metabolic and epigenetic regulation to maintain vascular regenerative homeostasis. Vascular smooth muscle cells (VSMCs) are a type of smooth muscle that controls the diameter of medium and large blood vessels. From a regenerative medicine perspective, VSMCs enhance angiogenesis. 212 In an exploration of the role of circRNA on VSMC, knockdown of circSFMBT2 inhibites VSMC proliferation and migration. Histone deacetylase 5 (HDAC5), a downstream target of miR-331-3p in VSMC, reduces the transcription rate of angiogenic factor with G-patch and FHA domains 1 (Aggf1), which plays an important role in regulating the transition of VSMC from synthetic to contractile. 213 The circSFMBT2/miR-331-3p/HDAC5/Aggf1 axis thus offers novel insights into circRNA-driven vascular remodeling. Collectively, these studies show how circRNAs actively control Fb behavior, KC activity, and vascular regeneration, making them promising targets for personalized wound treatments.
Remodeling
In skin trauma repair models, Fb achieves dynamic functional regulation through phenotypic plasticity: covering the injury with granulation tissue during the proliferation phase and differentiating into a myofibroblast phenotype during the remodeling phase. 214 Recent studies reveal that circRNAs regulate the repair-fibrosis balance by modulating Fb phenotypic transitions. Hyperproliferation of KF is one of the major causes of keloid formation. 215 It has been previously shown that circRNAs are aberrantly expressed in KF compared to healthy controls. 216 Complex circRNA/miRNA networks are involved in the process of keloid scarring. 217 For instance, circ-PDE7B promotes KF proliferation, invasion, migration, and ECM accumulation via the miR-331-3p/cyclin-dependent kinases 6 (CDK6) axis, identifying it as a potential therapeutic target. 215 circ_101238 accelerates KF proliferation through miR-138-5p/CDK6 signaling. 218 The above two studies exemplify the synergistic nature of the CDK6 regulatory pathway. Similarly, dual regulatory nodes are also reflected in the ceRNA network. circ_0057452 sponges miR-7-5p to upregulate Grb2-associated binding protein 1 (GAB1), driving KF proliferation, apoptosis resistance, collagen synthesis, and cell cycle progression. 219 circCOL5A1 also sponges miR-7-5p, activating the PI3K/Akt pathway to exacerbate keloid progression. 220 It reflects the hierarchical and synergistic nature of ncRNA regulatory networks. Through in-depth exploration of Gene Expression Omnibus (GEO) data, Yang et al. 221 demonstrated that circ_064002 upregulated fibronectin 1 (FN1) expression and enhanced KF invasiveness by competing for miR-30a/b-5p. As the parent gene of circ_064002, FN1, which is a key ECM component, mediates epithelial-mesenchymal transition (EMT) and cellular functions such as migration and differentiation.222,223 The EMT process by which epithelial cells acquire mesenchymal Fb-like features is an indispensable event for wound healing and scar formation. Therefore, the use of anti-EMT strategies during specific repair phases may achieve better scar treatment results. Aligning with this, Chen et al. 224 developed an anti-scarring strategy by silencing circ_0008450 to upregulate runt-related transcription factor 3 (Runx3, a keloid suppressor gene), thus inhibiting KC proliferation and migration. This scheme also inhibits the TGF-β1-induced EMT process and TGF-β1/Smad signaling pathway to achieve reversible EMT modulation. Table 4 summarizes the emerging roles of representative circRNAs in coordinating the wound healing process. These molecules increase regulatory complexity mainly by acting as ceRNAs. Their stable structure and presence in exosomes make them promising biomarkers and therapeutics. However, circRNAs’ exact functions in human chronic wounds are still not well understood.
Regulatory role of key circRNAs in chronic wound healing.

Emerging frontiers in circRNA therapeutics
These findings confirm circRNAs actively control cellular processes, not just being splicing byproducts. But most circRNA functions remain in early research phases. Their newly discovered activities keep expanding scientific understanding. A recent study revealed a novel role for circRNAs as miRNA protectors. Unlike the traditional miRNA sponge model, circASH1L(4,5) protects miR-129-5p from Nuclear Receptor subfamily 6 group A member 1 (Nr6a1) mRNA-triggered degradation, enhancing KC migration and re-epithelialization. 225 This finding provides a novel perspective on circRNA regulation of miRNA, suggesting the role of circRNAs as ceRNAs in miRNA needs to be further explored. In therapeutic innovation, the circARHGAP12-engineered MSCs developed by Meng et al. 226 team demonstrated significant therapeutic advantages. circARHGAP12-expressing MSCs exhibit enhanced survival in wound beds compared to standard MSCs, though this effect is reversible via miR-301b-3p. This reveals the mutual repair role between circRNA and MSC. In addition, emerging protein replacement therapies featuring mRNAs have previously been available, for example, by delivering VEGF-A mRNA into the body to express the desired protein. 227 However, the application of linear mRNAs has been limited due to their easy degradation, poor stability, and short expression time. The highly stable covalent closed-loop structure of circRNAs, along with their low immunogenicity which helps to avoid the activation of innate immune mechanisms, make circRNAs a valuable alternative to conventional protein therapies. Based on this, Liu et al. 189 developed an engineered circRNA lipid nanoparticle (LNP) system encapsulating encoded VEGF-A for sustained production and release of VEGF-A. This resulted in sustained protein expression (>7 days) and enhanced angiogenesis in diabetic wounds. Despite these advances, clinical translation faces challenges including delivery systems optimization and production standardization. To overcome these challenges, researchers are developing more efficient circRNA platforms to reduce the introduction of exogenous sequences and decrease the risk of immunogenicity. 228 Improved delivery methods could make circRNA a safe, versatile and effective therapeutic approach. In circRNA delivery, although the use of virus-based delivery (like lentivirus/adenovirus) has achieved good therapeutic efficacy in preclinical studies, safety concerns still persist in clinical applications. 198 Huang et al. 207 reported that sEVs overexpressing circCDK13 accelerate wound healing. But how these vesicles pack circRNAs remains unclear. Therefore, it is particularly important to explore a safe and effective circRNA delivery strategy. Unlike other methods, exosomes emerge as ideal candidates due to high biocompatibility and enriched circRNA content (sixfold higher than parent cells). 229 It suggests that exosomes are capable of efficiently gathering and transporting circRNAs. Exosomal circRNAs play critical roles in intercellular communication and gene regulation. Therefore, the establishment of standardized methods for exosome purification and circRNA delivery may promote a cell-free treatment modality for chronic wounds.
The CeRNA network in wound healing
The ceRNA hypothesis has emerged as a frontier in ncRNA regulatory mechanisms. This framework mainly refers to the fact that diverse ncRNAs, such as lncRNAs and circRNAs, act as miRNA “sponges.” They competitively bind to miRNAs by sharing common miRNA response elements (MREs). This downregulates miRNA molecules and reduces inhibitory activity of miRNAs against downstream target genes. 230 This regulatory network, which binds mRNAs to ncRNAs through the bridging role of miRNAs, is known as the ceRNA regulatory network (ceRNET). The network is highly dynamic and tissue-specific This is demonstrated by the fact that one ceRNA can bind to multiple miRNAs and, conversely, one miRNA can repress the expression of multiple transcripts. 231 For example, both lncRNA PVT1 and lncRNA H19 can competitively bind to miR-29a, increasing high mobility group box 1 (HMGB1) and FBN1 expression, respectively.232,233 This creates a one-to-many or many-to-one regulatory relationship between miRNAs and target genes, forming a fine and complex ceRNET centered on miRNAs. In the field of wound healing, current research focuses on analyzing the dynamic regulation of the ceRNA network at different stages of wound repair, particularly its coordination of cell proliferation, angiogenesis, and ECM remodeling through miRNA-mediated crosstalk (Figure 2).

lncRNAs and circRNAs regulate key signaling molecules to affect the overall activity of the pathway related to wound healing. This model illustrates how lncRNAs and circRNAs operate within ceRNA networks to coordinately regulate major wound healing processes. The diagram is structured around core biological themes: Inflammation regulation (left, e.g. JAK/STAT, NF-κB/NLRP3, macrophage polarization), Proliferation and Angiogenesis (center, e.g. HIF-1α/VEGF, Keap-Nrf2, PI3K/Akt, Wnt/β-catenin), and Tissue Remodeling (right, e.g. TGF-β/Smad, ECM deposition). Key ceRNA pairs (e.g. MALAT1/miR-142, H19/miR-19b) are highlighted to demonstrate how sponging specific miRNAs can fine-tune these pathways.
In studies targeting the proliferative phase of wound healing, ADSC-derived exosomal H19 accelerates re-epithelialization and tissue repair by sponging miR-19b to relieve its inhibition of SOX9, thereby activating the Wnt/β-catenin pathway. 234 Similarly, ADSC-derived exosomal MALAT1 promotes proliferation and migration of HaCaT and HDF cells in H2O2-damaged models via miR-124 inhibition and Wnt/β-catenin activation. 235 MALAT1 from ADSC-Exos also regulates wound healing through the miR-378a/FGF2 axis, where its silencing suppresses FGF2 expression and impairs murine Fb migration and proliferation. 236 Platelet-rich plasma-derived exosomal MALAT1 could also enhance DFU healing by targeting miR-374a-5p to inhibit Fb apoptosis. 237 Liang et al. 238 further demonstrated that MALAT1 upregulates zinc finger protein 217 (ZNF217) by sponging miR-141-3p, activating the TGF-β/SMAD pathway to drive Fb proliferation and migration. The ZNF217/TGF-β/SMAD pathway also enhances the expression of matrix proteins and promotes the reconstruction of the ECM. Notably, for RNAs with long nucleotides, the regulation of their expression levels is the basis for various experiments. Therefore, the use of CRISPR/Cas9 technology to precisely intervene lncRNAs could lead to a better understanding of the mechanism in wound healing. Shi et al. 239 innovatively engineered MALAT1-overexpressing stem cells, revealing its role as a miR-142 sponge to upregulate Nrf2 and promote angiogenesis in diabetic ulcers. BMMSC-derived exosomal lncRNA KLF3-AS1 induces angiogenesis in diabetic mice by sponging miR-383 to upregulate VEGFA. 240 In serum from sepsis patients, NEAT1 activates TGF-β/SMAD signaling via miR-495-3p inhibition, enhancing HUVECs proliferation and suggesting its therapeutic potential for burn injuries. 241 In addition to angiogenesis, slow lymphangiogenesis has been identified as one of the major factors in chronic wound healing. Lymphangiogenesis is also regulated by ceRNETs. Prospero homeobox 1 (Prox1) is a major transcription factor that regulates the development and differentiation of lymphatic endothelial cells. 242 GAS5 enhances the expression of Prox1 by downregulating miR-217, which promotes lymphatic vessel formation. 243 Similarly, lncRNA ANRIL also binds miR-181a and promotes lymphangiogenesis by deregulating the inhibitory effect of miR-181a on Prox1, thus improving diabetic wound outcomes. 244 Meanwhile, lncRNAs acting as ceRNAs also show excellent potential in the remodeling phase of wound healing. By integrating the secondary mining and bioinformatics analysis of the GEO database, two significantly up-regulated lncRNAs (DLEU2, AP000317.2) and four significantly down-regulated lncRNAs (ADIRF-AS1, AC006333.2, AL137127.1, LINC01725) were found in keloid-susceptible individuals. 245 These varying lncRNAs indirectly regulate Fb proliferation, differentiation, apoptosis, and collagen deposition through competing for binding miRNAs to form a regulatory network. MALAT1, a key fibrosis-associated lncRNA, involved in the progression of fibrosis by regulating signaling pathways and influencing the synthesis and deposition of ECM mainly through the ceRNA mechanism. Although the specific mechanism of MALAT1 in fibrosis in different organs varies, its value as a potential therapeutic target has been gradually recognized.246–248 It has been demonstrated that MALAT1 is upregulated in HS and promotes HS progression by acting as a miR-29a-3p sponge to promote Smad ubiquitin regulatory factor 2 (SMURF2) expression, driving TGF-β signaling and ECM overaccumulation. 249 GNAS-AS1, upregulated in keloids via STAT3 induction, promotes KF proliferation and migration by targeting miR-196-5p to derepress CXC chemokine ligand 12 (CXCL12) inhibition. 250 The upregulation of CXCL12 could further activate STAT3 to form a positive feedback loop, which allows the continued amplification of key signaling pathways during scar formation and accelerates keloid formation and development. 250 The high expression of the popular molecule H19 is closely associated with scar formation. Researchers found that H19 inhibits Fb apoptosis in HS by acting as a ceRNA targeting the miR-3187-3p/GAB1 axis. 251 It is expected that ideal therapeutic strategies will be developed by inhibiting H19 expression or directly regulating its downstream molecules. Collectively, these findings underscore lncRNAs as pivotal ceRNA regulators in wound healing.
Research on circRNAs as ceRNAs initially progressed slowly, but has accelerated significantly with advancements in next-generation sequencing and bioinformatics tools. Since 2013, circRNAs have been recognized as miRNA sponges. 185 Although their roles in ceRNETs across wound healing phases are well-documented, their full potential remains underexplored. Tian et al. 252 identified differentially expressed circRNAs (DEcircRNAs) between normal and DFU tissues, highlighting circRNA_072697 and circRNA_405463 as key modulators. In a rare example of traditional Chinese medicine integration, the Sheng-ji Hua-yu (SJHY) formula was shown to focus on circRNA-Krt13 and circRNA-Krt14 by analyzing gene expression differences between normal and diabetic wounds, providing mechanistic support for its clinical efficacy. 253 The abundance of bioinformatics data extends the scope of combinatorial use of ceRNET. Many databases such as starBase, SomamiR, LncACTdb, AFCMEasyModel, and HumanViCe now catalog circRNA-ceRNA interactions and underscore their vast regulatory potential. 254
In summary, ceRNAs have recently been increasingly highlighted as key coordinators of gene expression regulation. The complex sponge effect in the ceRNA network can be reflected by the effects summarized in Table 5. While both lncRNAs and circRNAs can act as molecular sponges for miRNAs, their specific roles, especially the sponge function of circRNA, have been detailed in previous sections. Table 5 focuses specifically on well-studied lncRNAs to deeply reveal the ceRNA-miRNA-mRNA axis in wound healing. However, the field still faces technical barriers such as insufficient resolution of network complexity and unclear tissue-specific regulatory mechanisms. Most of the existing studies are limited to animal models or in vitro experiments, lacking multi-omics validation of clinical samples. ceRNET construction needs to pay attention to the following factors: (1) The relative concentration of ceRNA/miRNA. Most miRNAs and ceRNAs can only adjust mRNAs and maintain RNA homeostasis in a physiological environment. Small changes in miRNA/ceRNA are unlikely to produce significant biological alterations in the levels of the target RNAs, and too much or too little miRNA/ceRNA reduces the competitive activity of ceRNAs. 255 (2) The subcellular localization of ceRNAs may also affect the availability of ceRNAs to shared miRNAs, thereby affecting ceRNA activity. 256 (3) RNA-binding proteins may affect miRNA binding to targets by occupying the MREs. 257 An alteration in any of the above factors may lead to an imbalance in ceRNET. Due to the intricate mechanisms and high experimental costs, the key to overcome the clinical translational barriers depends on combining advanced technologies with innovative precision control methods. ML and AI are emerging as pivotal tools to understand the complexity of the ceRNA network. For instance, deep learning models such as LncRNAnet could discover new lncRNAs by analyzing raw genetic sequence data. 258 The tool LncRNAnet combines both recurrent and convolutional neural networks. This model was trained on human and mouse transcriptome data and demonstrated superior performance over traditional ML methods. Thereby, the ML approach provides a high-throughput method to expand the catalog of potential ceRNAs. With the expansion of high-throughput omics and wound healing genomic databases, ncRNA/miRNA/mRNA ceRNETs are increasingly utilized to study regulatory mechanisms in chronic wounds. Targeting ncRNAs within ceRNETs thus offers novel methods for diagnostic biomarker discovery, drug development, and clinical interventions.
Representative examples of lncRNAs functioning as ceRNAs in wound healing.

Exosome
In the field of regenerative medicine, extracellular vesicles (EVs) are transforming treatment approaches as ideal biological carriers. These membranous structures, secreted by living cells, not only carry biological codes for intercellular communication but also serve as core vehicles for cell-free therapy due to their exceptional drug delivery capabilities. 259 EVs are classified into three subtypes based on biogenesis, size, cargo composition, and functionality, including exosomes (30–150 nm), microvesicles (100–1000 nm), and apoptotic bodies (50–5000 nm).260,261 In a survey of the number of publications on these three types of vesicles over the last decade, exosomes have been studied much more than the other two types of vesicles, suggesting that exosomes have become the absolute research hotspot in advancing regenerative therapies. 260
Exosomes act as ncRNA delivery carriers
Exosomes are a class of double-membrane extracellular vesicles generated through inward budding of the plasma membrane, serving as mediators of intercellular communication and signal transduction. 262 Their cargo includes bioactive molecules such as proteins, genetic materials (e.g. DNA, mRNA, and ncRNAs including miRNAs and lncRNAs), and lipids. 263 On one hand, exosomes serve as key mediators of the paracrine pathway of MSCs. Extensive studies have demonstrated that exosomes promote angiogenesis and accelerate wound healing by stimulating critical cells such as Fb and KC through interactions between their surface ligands and recipient cell receptors. 264 On the other hand, exosomes transfer internal cargo, including ncRNAs, to recipient cells for precise regulation of gene expression. 265 This integration of therapeutic RNAs with a natural delivery system offers innovative solutions to overcome limitations of conventional RNA therapies. However, as a fast-emerging field of personalized medicine, there are still several major obstacles for RNA therapies to achieve the desired results: high negative charge, large molecular size, susceptibility to endogenous nuclease degradation, and immunogenicity-driven immune recognition. 266 How to expand the potential of ncRNAs for clinical applications in wound healing is an upcoming focus. Exosomes are natural nanoparticles from a wide range of sources, exhibiting lower immunogenicity, higher cellular uptake efficiency, and more stable safety compared to synthetic carriers, lipid nanoparticles, and viral vectors, respectively. 267 They can protect ncRNAs from degradation and improve targeting specificity, making them superior RNA delivery vehicles. Notably, exosomes loaded with high concentrations of ncRNAs show better healing effects compared to wild exosomes. Wang et al. 268 reported that ADSC-Exos engineered with mmu_circ_0000101 (circ-Astn1) reduced HG-induced EPC apoptosis via the miR-138-5p/SIRT1/ FOXO1 signaling axis. These circ-Astn1-enriched exosomes significantly enhanced wound repair in diabetic mice compared to wild-type ADSC-Exos. In conclusion, exosomes have shown great promise as drug delivery vehicles. How to load therapeutic RNAs into exosomes so as to precisely target specific cells is one of the challenges for clinical translation of exosomes.
Engineered exosomes
Over the past decade, engineered exosomes have shown great promise for biomedical applications. These natural nanocarriers, which have been genetically or chemically “domesticated,” not only retain the inherent low immunogenicity and high biocompatibility of exosomes, but also upgrade their functions through targeted modification and therapeutic molecular loading. 269 Current engineering strategies are primarily divided into two categories: parent cell-based exosome engineering and direct exosome modification. The former regulates exosome biogenesis through genetic editing or environmental preconditioning of parent cells prior to isolation. The latter focuses on post-isolation physicochemical modifications to improve exosome performance. 270
Exosome engineering based on parental cells can be divided into genetic engineering of parental cells and pretreatment of parental cells. Genetic engineering has demonstrated strong programmability in parental cell engineering strategies and has been widely used for the creation of therapeutic nanoparticles. Specifically, by employing viral vectors, CRISPR/Cas9 systems, or plasmid transfection of specific gene fragments, parent cells are genetically modified to enhance or silence the expression of exsomal ncRNAs. 271 Ge et al. 272 constructed engineered exosomes from miR-132-overexpressing ADSCs via lentiviral transfection. The synergistic action of miR-132 and exosomes promoted M2 macrophage polarization, reducing inflammation in diabetic wounds. Despite the efficiency of viral vectors, complications such as their immunogenicity and toxicity limit their further application. 273 Based on this, nonviral plasmid transfection has emerged as another practical option. A recent study used this approach to promote overexpression of lncRNA HOTAIR in parental cell MSC to promote angiogenesis in diabetic mice. 274 Additionally, CRISPR/Cas9, a revolutionary genome editing technology, has been actively explored for wound healing. With superior targeting precision, reduced costs, and higher editing efficiency compared to conventional methods, CRISPR/Cas9 enables precise modulation of ncRNA expression in parent cells. 275 Since ordinary vectors cannot transport lncRNAs with excessive molecular weight, Shi et al. 239 used CRISPR/Cas9 technology to overexpress MALAT1 in parental cells. Overall, engineered exosomes can effectively break through the therapeutic limitations of natural exosomes.
Preconditioning involves altering the external physical environment or culture conditions of parental cells to promote the secretion of therapeutic exosomes and increase exosome production. Hypoxia, pharmacological agents, and cytokines have been shown to improve exosome efficacy. 276 For example, BMMSC-Exos exposed to Fe3O4 magnetic nanoparticles and static magnetic fields showed elevated miR-1260a abundance, promoting angiogenesis and bone regeneration. 277 The operation of pretreatment is simpler compared to genetic engineering. However, the drawbacks are lower loading efficiency and possible cytotoxicity of the drug. 278 It prompts researchers to turn their attention to direct exosome engineering.
Direct exosome engineering strategies are categorized into chemical and physical approaches. In general, this approach is more efficient than parental cell-based exosome engineering because the encapsulation efficiency (EE%) and loading capacity (LC%) of exosomes are manipulated more. 279
Chemical methods include co-incubation, saponin permeation, and click chemistry. 280 Co-incubation enables passive cargo loading via spontaneous molecular diffusion into exosomes. 281 Saponin uses surfactants to induce the formation of small pores in lipid membranes, thereby facilitating cargo permeation. 282 Click chemistry is a covalent binding modality that couples functional ligands to the surface of exosomes. 283 Although chemical modification of exosomes offers fewer options than genetic engineering, its rapid and high yielding advantages make it a popular tool for biomolecule modification. Physical methods encompass electroporation, sonication, and extrusion. 284 Electroporation employs pulsed electric fields to generate nanopores in exosomal membranes, enabling cargo diffusion. 285 Xiong et al. 286 loaded miR-542-3p into BMMSC-Exos via electroporation, enhancing neovascularization and wound remodeling. Similarly, sonication uses mechanical shear forces to disrupt membrane integrity for cargo entry. 287 The disadvantage is that high-power sonication is the most destructive technique for exosome membrane integrity. 288 Therefore a carefully designed sonication program is required. Extrusion is a method of extruding exosome-cargo mixtures through a membrane filter of a specific pore size to generate exosome-mimetic vesicles. 289 Different drug loading strategies exhibit different drug loading rates, but all of them show different degrees of improvement compared to natural exosomes. Electroporation achieves a 3.96-fold enhancement in loading efficiency compared to passive drug loading, while low-frequency sonication achieves a 19.6-fold increase. 290 To provide a clear comparison, the major characteristics of parent cell-based and direct exosome engineering strategies are summarized in Table 6. Small changes in isolation or pretreatment methods can greatly affect experimental outcomes. Researchers should compare drug-loading approaches and define key parameters like drug concentration before testing. These steps will enhance the reproducibility of engineered exosome studies.
A comparative overview of the two primary exosome engineering strategies.

In addition to the drug-carrying efficiency of exosomes, how to realize large-scale production is also a challenge to be solved in the clinical translation of exosomes. Bionic exosome systems are emerging as innovative solutions to overcome this obstacle with faster manufacturing, lower costs, and personalized clinical benefits. 294 On the one hand, bionic exosomes can enhance the natural secretion of exosomes by optimizing the culture conditions of the cells. Dynamic 3D culture platforms, such as hollow-fiber bioreactors or microcarrier-based stirred-tank systems, generate large numbers of cells and exosomes in a short period of time by maximizing the culture surface area.295,296 Physical stimuli such as hypoxic conditions, pH and temperature changes can also increase exosome production, 297 which is a technique similar to the pretreatment of parental cells described above. On the other hand, bionic exosomes based on optimized reconfiguration of cell membranes can skip the natural secretion of exosomes and induce the production of synthetic exosome mimics. Extrusion and microfluidic devices are widely used to generate such exosome-mimicking nanovesicles, which have promising applications.298,299 Engineered exosomes are highly versatile, but using them in patients faces major challenges. We need to solve problems in large-scale production, batch-to-batch consistency, and reliable potency assays. Furthermore, their genetic or chemical engineering methods also bring regulatory questions.
Exosomes combined with hydrogels
Although engineered exosomes improve the some limitations of natural exosomes, unresolved technical problems still exist. As with synthetic nanocarriers, locally administered exosomes are rapidly cleared by the body. 300 Chronic wounds, especially DFUs, tend to take a long time to heal. The application of biomaterials can greatly stabilize exosomes and prolong their duration at wound sites. It has emerged as a critical strategy to increase the utilization of exosomes and accelerate their clinical translation. 290 Hydrogels are the most widely used biomaterial for exosome encapsulation. They have a high emergence intensity due to their favorable anti-inflammatory, antimicrobial, and antioxidant properties, and are still a high-frequency keyword in wound healing research. 301 Serving as exosome carriers, hydrogels avoid problems such as poor mechanical strength, low bioactivity, short duration of action, and uncontrolled exosome release. 302 Consequently, how to efficiently tailor hydrogels for desired exosome delivery and drug loading has become a hot topic in current research.
Hydrogels are broadly classified into natural and synthetic categories. Among them, natural hydrogels include polysaccharide-based hydrogels (e.g. alginate, hyaluronic acid, and chitosan) versus protein-based hydrogels (e.g. gelatin and silk protein). 303 These hydrogels exhibit excellent biocompatibility and low immunogenicity, and can mimic the original ECM very well. 304 Tao et al. 305 used lentiviral transfection to overexpress miR-126-3p from synovial MSC-Exos and combined the exosomes with chitosan to accelerate the re-epithelialization and angiogenesis of diabetic wounds in rats, which shows better therapeutic effects than chitosan hydrogel alone. However, the poor biomechanical strength, the difficulty in modifying material properties, and the high heterogeneity due to biological origin limit natural hydrogels’ further development. 290 Synthetic hydrogels mainly include polyethylene glycol (PEG) and polyvinyl alcohol (PVA). 303 The uniform preparation process is much cheaper and effectively overcomes the reproducibility of natural materials, but the biocompatibility is relatively poor. Therefore, chemical modification of natural polymers or 3D-printed functional hydrogels can enhance mechanical properties while retaining high biocompatibility, which has a wide therapeutic potential in the field of regenerative medicine. For instance, gelatin methacryloyl (GelMA) hydrogels, derived from methacrylated gelatin, have become popular materials for biomedical applications. GelMA hydrogel was incorporated into exosomes as wound dressings to achieve continuous exosome release. 306 At the same time, it can also accommodate irregular diabetic wounds for more efficient delivery of circ-Snhg11, which protects endothelial function in diabetic mice by sponging miR-144-3p. 302 In another study, bilayered thiolated alginate/PEG diacrylate (BSSPD) hydrogels were used as carriers for BMMSC-sEVs to sequentially release miR-29b-3p-enriched sEVs, accelerating scarless wound healing. 307 Compared to traditional hydrogel fabrication methods, 3D bioprinting allows for the rapid construction of adjustable three-dimensional scaffolds that fit the shape of the wound and resemble natural human tissues, enhancing the scaffold’s exosome-releasing ability. 308 In recent years, therapeutic applications of 3D bioprinting on exosome-containing hydrogels have been increasingly reported in the field of wound healing. Hu et al. 309 utilized 3D-printed bioactive glass scaffolds coated with GelMA/nanoclay hydrogels to deliver sEVs enriched with miR-23a-3p, inducing osteogenesis and angiogenesis. These innovations position 3D-bioprinted exosome-hydrogel systems as transformative platforms for regenerative medicine. In addition, AI and algorithmic predictive modeling are increasingly applied to improve hydrogel design, automate parameter adjustment, and discover novel materials. It guides the design and preparation of advanced hydrogels and drives breakthroughs in smart wound management. 310 These experiments show that the combination of exosomes and biomaterials can boost RNA therapies’ effectiveness and provide a new exosome-based wound treatment for clinical use. However, many aspects still need to be further explored, including hydrogel safety, fabrication complexity, and high costs. 311 It is suggested that more hydrogels composed of natural peptides should be developed in the future for diabetic wound applications.
Clinical translation of exosomal ncRNAs faces unresolved issues such as delivery efficiency, scalable production, purification standards, whether the yield of exosomes varies with the staging of chronic wounds, and a deeper understanding of molecular mechanisms. Despite these challenges, the intrinsic therapeutic properties of exosomes make them promising drugs. Recently, several clinical trials using exosomes as therapeutic agents for the treatment of wounds have been initiated. Key details of these trials are summarized in Table 7. Except one study (NCT05475418), which showed effectiveness in treating wounds, the results of other studies were not reported. 312 Researchers still need to conduct more randomized, double-blind trials to evaluate the safety and efficacy of exosomes, ultimately unlocking their full therapeutic potential in wound care.
Clinical trials investigating exosomes as therapeutics for wound management.

Conclusions
The long-term nature and the tendency to cause disability of chronic wounds have brought a huge burden to patients and their families. There is an urgent need to enhance the efficiency of chronic wounds to relieve patients’ suffering. This review traces the development from ncRNA discovery to ncRNA-based therapy for chronic wounds. It shows how miRNAs, lncRNAs and circRNAs help to identify potential treatment targets. It also discusses how advanced delivery systems like engineered exosomes and hydrogels are crucial to overcoming translational challenges. Important molecules such as miR-21, miR-29 family, H19, and MALAT1 play important roles across inflammatory, proliferative, and remodeling phases in wound healing. This review also focuses on ncRNAs’ modulation of key cellular players in wound healing like macrophages, KCs, Fbs, and vascular endothelial cells. They also affect critical signaling pathways like PI3K/Akt, MAPK/ERK, Wnt/β-catenin, NF-κB, and TGF-β/Smad. Certain ncRNAs can act as ceRNAs to construct an intricate molecular network that breaks the traditional gene regulation model and enriches our understanding of the potential role of ncRNAs at the transcriptome level. Preclinical studies of ncRNA therapies show promise, but their path to clinical use faces clear challenges. For example, the MRX34 trial was closed early due to unexpectedly severe immune-mediated toxicities, which resulted in four patient deaths in expansion cohorts. Such outcomes remind us of the challenges about off-target effects, immunogenicity, and delivery safety. Despite the difficulties, the development of miRNA drugs has never stopped. In therapeutic targeting, exosomes have emerged as revolutionary nanocarriers, enabling safe and efficient delivery of ncRNA mimics to target cells via genetic or chemical engineering. The combination of hydrogel with exosomes further optimizes wound repair, but the scalable applications remain constrained by inconsistent production quality and safety concerns. In the long run, exosome technology needs to find a balance between precision, safety and scale-up. Future trends in ncRNA clinical translation will focus on technological innovation. Synthetic biology may enable engineered exosomes or CRISPR dynamic systems for precise regulation of multiple genes, accelerating ncRNA therapy development. ML and AI can analyze single-cell transcriptomics and clinical data to build patient-specific ncRNA networks, predicting optimal therapeutic targets and dosages for precision medicine. 313 AI-driven smart hydrogels can achieve on-demand release of ncRNAs in drug delivery systems. 313 However, current research still faces serious challenges. In wound repair, different RNA molecules need to act precisely at specific times and locations, which puts higher demands on drug delivery systems. Genetic differences and metabolic characteristics of patients, such as those in diabetic wound microenvironments, require AI to analyze a large amount of medical data and develop a personalized and dynamic treatment. 314 In addition, two major challenges still need to be solved. One is to establish a long-term tracking mechanism to comprehensively assess the potential risks of RNA therapies. The other is to reduce the cost of high-end vector preparation through automated production and standardization to achieve large-scale application.315,316 Ultimately, the transition from bench to bedside for ncRNA-based therapies will depend on the in-depth integration of bioinformatics, engineered materials, and clinical medicine. Only through such collaboration can the complex RNA regulatory network be transformed into practical and usable trauma treatments.
Footnotes
Acknowledgements
No.
Author contributions
LY participated in the work, contributed to the conceptualization, wrote the original draft, and created visualizations. YL conceived and designed the study, acquired funding, supervised the work, and performed validation. All authors reviewed and approved the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the following grants: Shanghai Sailing Program (Grant No. 21YF1418800), Special Project for the Elderly of Shanghai Municipal Health Commission (Grant No. 2020YJZX0143), and Construction Project of High-level Local Universities, Shanghai University of Medicine & Health Sciences Commission (Grant No. E1260123201006).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All data or related information supporting the conclusions of the review is included in the article.
