Abstract
Diabetic wound healing presents a significant clinical challenge due to the interplay of systemic metabolic disturbances and local inflammation, which hinder the healing process. Macrophages undergo a phenotypic shift from M1 to M2 during wound healing, a transition pivotal for effective tissue repair. However, in diabetic wounds, the microenvironment disrupts this phenotypic polarization, perpetuating inflammation, and impeding healing. Reprograming macrophages to restore their M2 phenotype offers a potential avenue for modulating the wound immune microenvironment and promoting healing. This review elucidates the mechanisms underlying impaired macrophage polarization toward the M2 phenotype in diabetic wounds and discusses novel strategies, including epigenetic and metabolic interventions, to promote macrophage conversion to M2. Hydrogels, with their hydrated 3D cross-linked structure, closely resemble the physiological extracellular matrix and offer advantageous properties such as biocompatibility, tunability, and versatility. These characteristics make hydrogels promising candidates for developing immunomodulatory materials aimed at addressing diabetic wounds. Understanding the role of hydrogels in immunotherapy, particularly in the context of macrophage reprograming, is essential for the development of advanced wound care solutions. This review also highlights recent advancements in immunotherapeutic hydrogels as a step toward precise and effective treatments for diabetic wounds.
Introduction
Managing diabetic wounds presents an ongoing challenge in clinical practice. Various factors contribute to the complexity of diabetic wound healing, including peripheral neuropathy, insufficient local wound care, hyperglycemia, and peripheral vascular disease. These factors disrupt a range of biological responses, including restriction of cell migration and proliferation, production of healing related factors, persistent production of pro-inflammatory cytokines, and dysangiogenesis, all of which affect immune homeostasis in diabetic wounds, thereby delaying the wound healing process.1,2 Consequently, there is an urgent need to implement an immunomodulatory strategy to mitigate the multifaceted complications associated with diabetic wounds.
Macrophages, as a crucial component of the body’s innate immune system, are involved in a wide range of functions, including tissue repair and regulation of immune homeostasis. Macrophages exhibit a binary M1/M2 polarization pattern that governs diverse immune functions through intrinsic signaling cascades. During the early stages of healing, they incite inflammation with a pro-inflammatory M1 phenotype, followed by a transition to an anti-inflammatory M2 phenotype, which facilitates tissue repair.3–5 The phenotypic transition of macrophages is evidently crucial for the healing of chronic wounds. 6 In diabetic wounds, the local microenvironment hampers the conversion of macrophages to M2. This leads to sustained chronic inflammation, inadequate angiogenesis, diminished collagen deposition, and compromised wound healing.4,7 The hindered transition from M1 to M2 in diabetic wounds is influenced by several factors, including hyperglycemia, oxidative stress, and bacterial infection. These factors collectively contribute to heightened inflammation. Hence, stimulating or reprograming macrophages in diabetic wounds emerges as a promising therapeutic strategy to modulate the inflammatory response and improve clinical outcomes. In addition, although macrophages can be precisely regulated in wound repair, the immunomodulatory pathways involved remain poorly understood.
Macrophage reprograming is a precisely orchestrated process encompassing multiple pivotal signaling pathways, pre-transcriptional epigenetic modifications, and post-transcriptional metabolic regulatory networks.8,9 Epigenetic remodeling denotes a reversible mechanism that controls gene expression without modifying the DNA sequence.10,11 Epigenetic remodeling has been acknowledged for its role in modulating tumor progression by orchestrating the transcriptional regulation of genes related to immune activation/suppression and establishing cellular memory of cell fate.12–16 Metabolic reprograming is a swift adaptive response of cells to alterations in the surrounding microenvironment, aimed at fulfilling their energy requirements.17–20 The intimate interconnection between immune cell function and metabolism has given rise to a burgeoning field termed immunometabolism. In this field, metabolites and intermediates assume pivotal roles in governing the proliferation, differentiation, activation, and function of immune cells.21,22 The immunoregulatory strategy of reprograming macrophages to reinstate their metabolic pathways and pro-regenerative phenotypes has surfaced as a promising approach for augmenting wound healing at a profound level. This aspect will be meticulously scrutinized in the present comprehensive review.
At present, the management and treatment of diabetic wounds predominantly hinge on localized dressing application, systemic glycemic control, and antimicrobial interventions. Hydrogel-based wound dressings have become a highly regarded and promising material in wound care, and have been extensively explored in the field of wound management for a long time. 23 The distinctive advantages of hydrogels over conventional excipients include: (a) maintaining the wound moist while absorbing exudate, (b) covering sensitive underlying tissues without adhesion, (c) enabling the exchange of water and gas as well as the nutrient transport, (d) alleviating pain through cooling properties, and (e) exhibiting excellent biocompatibility that enables mimicry of the natural extracellular matrix.24–26 Rational design of hydrogel materials to regulate the immune microenvironment has become a key approach to accelerate tissue repair and regeneration. Hydrogel dressing with immunomodulatory ability has great application prospect in the clinical practice of diabetic wound. In recent years, extensive research on the pathogenesis of diabetes wounds risen various functional hydrogel dressings that have demonstrated promising effects, including the capability of inducing phenotype transformation in macrophages. On one hand, the three-dimensional network structure and intrinsic biological activity create an optimal immune microenvironment. On the other hand, hydrogels synergistically enhance the immune activation when accompanying with drug or cellular active ingredient delivery, thereby accelerating the healing process of chronic wounds. This review comprehensively summarizes potential immunotherapy modalities for hydrogels and explores emerging trends focused on enhancing efficacy and safety through multiple therapeutic avenues. Specifically, the paper delves into novel strategies aimed at promoting macrophage reprograming toward M2, while elucidating the correlation between the physical and chemical properties of hydrogels and their influence on macrophage reprograming function. These insights offer promise for advancing the design of advanced hydrogels to enhance diabetic wound repair, with potential translation into clinical applications in the near future.
Macrophages in different wound microenvironments
Normal phenotypic transition during regular healing
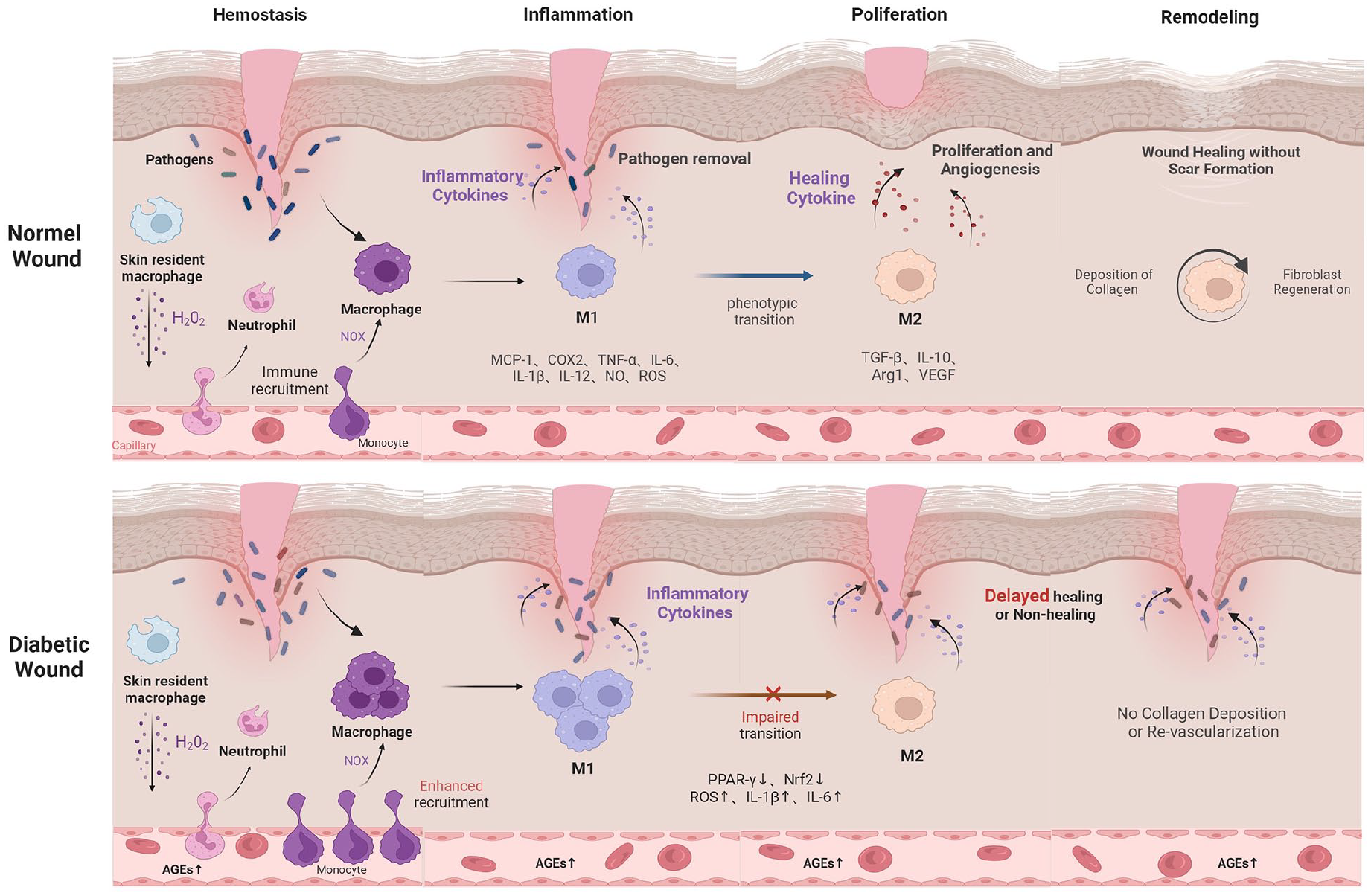
The crux of normal wound healing lies in the transition from the inflammatory to the proliferative phase. In this process, the phenotypic and functional transformation of macrophages is vital for the initiation and resolution of the inflammatory phase. 27 Tissue-resident macrophages initiate an early inflammatory response following skin injury by releasing hydrogen peroxide, which sequentially recruit blood neutrophils and monocytes, the latter further differentiate into macrophages under the influence of NADPH oxidases (NOX).6,28,29 Afterwards, the activation of M1 macrophages is triggered by pathogen-associated molecular patterns (PAMPs) or damage-associated molecular patterns (DAMPs), such as lipopolysaccharide (LPS), Th1 cytokines like Interferon-γ (IFN-γ), and tumor necrosis factor-α (TNF-α). Moreover, under the influence of NF-κB, NLRP3, TLR4, STAT1, and other signaling molecules, M1 macrophages demonstrate enhanced production and secretion of pro-inflammatory cytokines including TNF-α, IL-6, IL-1β, monocyte chemotactic protein-1 (MCP-1), and cyclooxygenase-2 (COX-2).30–32 Functionally, M1 macrophages upregulate oxygen and nitrogen reactive substances by increasing NOX-2, inducible nitric oxide synthase (iNOS), synaptotagmin-binding cytoplasmic RNA interacting protein (SYNCRIP), and TNF receptor associated factor 6 (TRAF-6), thereby thrust the inflammatory response and promoting phagocytosis and clearance of pathogenic microorganisms. 33 As healing progresses moving forward, the expression of lysine methyltransferase Setdb2 increases in macrophages, which acts as a “brake” of inflammation by enhancing histone H3K9 trimethylation at the NF-κB binding site on inflammatory cytokine gene promoters, leading to the resolution of inflammation and transition to a reparative phase. 34 Th2 cytokines IL-4 and IL-13 activate the IL-4Rα/JAK/STAT6 pathway to induce polarization toward M2 macrophages characterized by high levels of mannose receptor CD206, IL-10, arginase 1 (Arg1), TGF-β expression while down-regulating NOX-2, iNOS, SYNCRIP to counteract oxidative stress, thus playing a crucial role in suppressing inflammation as well as promoting tissue repair and regeneration.33,35,36
Impaired polarity conversion in diabetic wounds
Hyperactivation of inflammatory cells and persistent chronic inflammation are the primary pathogenic characteristics of impaired wound healing in diabetes.37–39 This is closely associated with the accumulation of pro-inflammatory mediators and deficiency of anti-inflammatory mediators due to disruption in macrophage phenotypic shift within the peri-wound area (Figure 1). 4 Under hyperglycemic conditions, elevated advanced glycation end products (AGEs) in the circulation induce high levels of reactive oxygen species (ROS) and reactive nitrogen species (RNS), rendering peri-wound monocytes more susceptible to transformation into M1 macrophages. 40 ROS triggers the activation of NF-κB, leading to elevated production of matrix metalase-9 (MMP-9), which can degrade extracellular matrix (ECM) components involved in tissue remodeling and is considered contributing to impaired diabetic wound healing.41,42 Previous studies have demonstrated that peroxisome proliferator-activated receptor-gamma (PPAR-γ) negatively regulates various inflammatory pathways mediated by M1 through interference with AP-1, NF-κB, or STAT-3 transcriptional activity. 43 Conversely, a high-glucose environment induces sustained production of IL-1β in the peri-wound region, leading to downregulation of PPAR-γ expression levels and impairing the switch in macrophage to pro-healing phenotype. 44 Moreover, impaired nuclear factor erythroid-2-related factor 2 (Nrf2) activity in hyperglycemic conditions leads to elevated levels of ROS and reduced expression of quinone oxidoreductase 1 (NQO1) and heme oxygenase (HO1), accompanied by enhanced secretion of pro-inflammatory cytokines such as IL-1β, IL-6, and MCP-1, thereby hindering macrophage phenotypic transformation. 45 As previously described, Setdb2 is a key epigenetic enzyme in the macrophage-to-repair mode transition. However, in diabetic wound tissues, the IFNβ-Setdb2 axis is impaired in macrophages by hyperglycemia and oxidative stress, resulting in a significant decrease in Setdb2 expression and failure of macrophage conversion.34,46 There’s also research that confirms excessive ferrous ions generated by diabetic wounds elevate intracellular iron levels in macrophages leading to an inflammatory response and impaired phenotypic transformation. 47 With the recent advancements in epigenetic and metabolic research, an increasing number of intricate mechanisms have been identified as being implicated in the alteration of macrophage polarization. For example, in diabetic macrophages, TGF-β enhances COX-2/PGE2 production by suppressing DNMT3b-mediated hypermethylation of the COX-2 promoter. Simultaneously, the histone methyltransferase MLL1 synergistically promotes the up-regulation of COX-2/PGE2 through increased expression of cPLA2 gene, resulting in a sustained pro-inflammatory phenotype of macrophages.48,49
Dysregulation of macrophage polarization from M1 to M2 in diabetic wounds. Figure created with Biorender.com.
Macrophage interactions in wound healing
During the proliferative phase of wound repair, macrophages play a crucial role in coordinating the proliferation, differentiation, and migration of various cell types, including keratinocytes, fibroblasts, and endothelial cells. They achieve this through the secretion of cytokines and growth factors. 50 This process facilitates re-epithelialization of the wound and promotes angiogenesis, leading to the eventual formation of new tissue. 50 Macrophages and fibroblasts engage in a reciprocal exchange of growth factors to support their respective survival. Macrophages supply fibroblasts with platelet-derived growth factor (PDGF) ligands essential for fibroblast viability, while fibroblasts produce colony-stimulating factor 1 (CSF1) to promote macrophage survival, proliferation, and differentiation. 51 M2 macrophages play a pivotal role in promoting fibroblast proliferation and migration, as well as facilitating extracellular matrix synthesis. They achieve this by releasing growth factors such as vascular endothelial growth factor (VEGF) and TGF-β1. Conversely, in diabetic wounds, M1 macrophages significantly reduce the expression levels of crucial healing factors for fibroblasts, including matrix metalloproteinases (MMPs), Chitinase-3-like protein 1 (CHI3L1), and VEGF-A. This downregulation impairs fibroblast activation and hampers wound healing. 52
The formation of a vascular network is crucial for delivering essential nutrients and oxygen to support neighboring cell proliferation during tissue repair. Macrophages play a significant role in this process by secreting increased levels of TGF-β and VEGF. This promotes the survival of vascular endothelial cells (ECs) and stimulates myofibroblast differentiation, thus facilitating wound healing. 53 The activation of ECs has been shown to coordinate the transport of lactic acid to macrophages via vascular endocrine mechanisms. This process polarizes macrophages toward the M2 and induces significant production of VEGF, thereby further enhancing angiogenesis. 54 The positive feedback loop between ECs and macrophages plays a crucial role in enhancing the ischemia-reperfusion response and promoting regeneration in damaged muscle tissue. 54 M2 macrophages are widely recognized for their pivotal role in tissue angiogenesis. It’s noteworthy that during the initial stages of wound healing, M1 macrophages act as a significant source of VEGF. However, the prolonged retention of M1 macrophages leads to a heightened inflammatory response, which subsequently hampers endothelial cell function in the later stages of wound healing. This impairment affects vascular regeneration and overall wound healing process.55,56
Reprograming macrophages to promote diabetic wound healing
Epigenetic remodeling
Epigenetic mechanisms regulating gene expression encompass DNA methylation, post-translational histone modifications (PTHMs), and the non-coding RNAs (ncRNAs). 57 In diabetes, high glucose levels, oxidative stress, and inflammatory mediators active the aberrant epigenetic expression that induce the upregulation of pro-inflammatory genes, and these underlying mechanisms are strongly associated with macrophages subpopulation along with impaired wound healing in diabetic patients.58,59
DNA methylation
DNA methylation involves the addition of methyl groups to cytosine residues in cytosine-phosphate-guanine dinucleotides (CpG sites), a process commonly associated with gene silencing. 60 DNA methylation is primarily regulated by three members of the DNA methyltransferase (DNMTs) family, namely DNMT1, DNMT3a, and DNMT3b. The pivotal role played by the DNMT family in promoting macrophage polarization through DNA methylation has been demonstrated to enhance diabetic wound healing. Yang et al. 61 found DNMT3b facilitated M1 macrophage polarization and adipose tissue inflammation by increasing DNA methylation at the PPARγ1 promoter, resulting in downregulation of PPARγ1. Subsequent investigations successfully switched macrophage polarization toward a selectively M2 phenotype through complete knockdown of DNMT3b, leading to reduced expression of inflammatory factors and restoration of insulin sensitivity in adipocytes. 61 In a study utilizing genetic (db/db) and diet-induced obesity (DIO) mouse models of type 2 diabetes (T2D), an elevation in DNMT1 expression was observed in bone marrow-derived hematopoietic stem cells (HSCs), which facilitated the development of a pro-inflammatory macrophage phenotype. 62 The authors reported that NOX-2-induced oxidative stress upregulated DNMT1 by inhibiting microRNA let-7d-3p in diabetic HSCs, resulting in DNA hypermethylation of Notch1, PU.1, and Klf4 genes responsible for HSC differentiation toward monocytes/macrophages. Consequently, this downregulation impaired their expression levels within HSCs and ultimately promoted M1-type macrophage polarization, thereby hindering the wound healing process. Another investigation revealed that pharmacological administration of 5-aza-2′-deoxycytidine or genetic inhibition of DNA methylation through DNMT1 deletion facilitated the alternative activation of macrophages while inhibiting inflammation. Interestingly, exhibited protection against obesity-induced inflammation and insulin resistance were found in these mice. 63 Furthermore, DNMT1 plays a crucial role in wound tissue angiogenesis. Increased DNMT1 expression in endothelial cells and blocked transcription of vascular homeostasis genes (Ang-1 and Ang-2) during hyperglycemia result in decreased regeneration of blood vessels in wounds and impaired wound healing.64,65 Conversely, inhibiting DNMT1 modulates the Ang-1/NF-κB signaling pathway to promote angiogenesis and accelerate diabetic wound healing. 66
Additionally, DNA demethylation also occurs in diabetic wounds. TET2 is a well-known DNA demethylation protein that induces the demethylation of the MMP-9 promoter in diabetic skin tissue. lncRNA TETILA, as a molecular scaffold, increases the stability of TET2 protein through the ubiquitin-proteasome pathway and indirectly activates the demethylation of MMP-9 promoter. 67 Inhibiting TETILA and subsequently down-regulating the expression of MMP-9 can improve the ability of wound healing in vitro.67,68 Collectively, targeting the machinery responsible for DNA methylation emerges as a promising for addressing non-healing diabetic wounds. However, compared to DNA methylation, much less is known regarding the role of DNA demethylation in diabetic wound healing and further study is warranted.
Histone modifications
Histone modifications occur at specific amino acid residues of core histones, including acetylation, methylation, phosphorylation, and ubiquitination. The activity of histones can be determined by specific modification states, leading to either the activation or silencing of the transcription for corresponding genes, in contrast to DNA methylation, which consistently associates with transcriptional suppression. 69
Methylation is the most extensively investigated histone modification. Aberrant histone methylation under diabetic conditions leads to a prolonged inflammatory phenotype in macrophages during wound healing. During the early stages of wound healing, the Histone H3K27 demethylase Jumonji domain containing-3 (JMJD3) downregulates trimethylation of histone H3 at lysine 27 (H3K27me3) via the JAK1,3/STAT3 signaling pathway, which in turn enhances the expression of NF-κB inflammatory factor genes and induces pro-inflammatory effects in macrophages (Figure 2). Afterwards, this effect is attenuated in the later stages of wound healing, allowing the transition of the wound from an inflammatory to a reparative state. However, in diabetic wounds, there is a sustained JMJD3 expression that impedes macrophage transition toward a regenerative phenotype, thereby adversely affecting wound healing (Figure 2). 70 Importantly, Audu et al. 70 have also demonstrated that STING regulated by JMJD3 through the IRF3/IFN-I pathway exerts anti-inflammatory and facilitates tissue repair; however, in diabetic wounds, its role is altered to elevate NF-κB cytokine pathway that results in sustained inflammation (Figure 2). Additionally, SET7/9 acts as an important coactivator of NF-κB and increases methylation of H3K4 on the NF-κB p65 promoter in peripheral blood PBMCs from T2 diabetic patients, leading to higher expression of inflammatory factors such as MCP-1, iNOS, ICAM-1, and COX-2, which contribute to M1-type macrophage production in peripheral tissues and negatively affect wound repair. 71 Another member of the histone methyltransferase family, SET and MYND domain-containing protein (SMYD), plays a regulatory role in various downstream target genes and immune-related signaling pathways through histone lysine methylation. SMYD2 acts as a negative regulator of macrophage M1 polarization by upregulating TNF and IL-6 promoter H3K36me2, inhibiting NF-κB and extracellular regulated protein kinases (ERKs) signaling, and reducing pro-inflammatory cytokine production. 72 Stender et al. 73 demonstrated that SMYD5, associated with the nuclear receptor co-repressor (NCoR) complex, attenuates macrophage-mediated inflammatory responses by catalyzing inhibition of Toll-like receptor4 (TLR4) gene expression via H4K20me3. In contrast to the first two, SMYD3 has been observed to exhibit down-regulation under hyperglycemic conditions and possesses the ability to induce inflammation. 74

Schematic of the mechanism of JMJD3 activity in wound macrophages. (Left) In non-diabetic wounds, macrophages activated by IFN-β enhance the transcription of JMJD3 via the JAK/STAT3 pathway, contributing to early inflammation. Concurrently, STING is regulated by JMJD3 through the IRF3/IFN-I pathway, which exerts anti-inflammatory and facilitates tissue repair. (Right) In diabetic wounds, IL-6 promotes persistent expression of JMJD3 in macrophages, impeding the transition to a regenerative phenotype and ultimately impairs the wound repair. This sequence primes STING to elevate NF-κB cytokine pathway that results in sustained inflammation and hinders wound healing. Reproduced under terms of the CC-BY license. 70 Copyright 2022, Audu et al, published by Springer Nature.
Moreover, histone acetylation plays a crucial role in the epigenetic regulation of macrophages. Histone deacetylase (HDACs) and histone acetyltransferase (HATs) jointly regulate the acetylation state of histones, controlling the “on” and “off” of gene transcription, respectively. 75 Lysine acetyltransferase 8 (KAT8), a member of the MYST family, primarily targets H4K16ac acetylation to facilitate gene transcription. 76 In diabetic skin wounds, TNF-α stimulates the excessive expression of KAT8 in macrophages, which promotes the expression of inflammatory factors, such as IL-1β, TNF, and NOX-2, leading to aberrant macrophage phenotypic transformation and persistent chronic inflammation. 77 Additionally, KAT8 also selectively inhibits IFN-I production by acetylating Interferon regulatory factor 3 (IRF3) at site K359 while influencing macrophage functions. 78 Zhang et al. 79 demonstrated that the recruitment of HDAC2 selectively inhibits IL-6 transcription, thereby promoting inflammation resolution, while HDAC2 loss induces up-regulation of various inflammatory mediators during late lipopolysaccharide reaction. Conversely, deletion of HDAC3 increased expression of cathepsin B in, resulting in significant degradation of serine-threonine kinase 1 (RIP1), which interferes with TNF-α-mediated NF-κB activation and inflammatory response. 80 Therefore, the pivotal role played by histone-modifying enzymes (e.g. HDACs) and their inhibitors (e.g. HDACi) in modulating the progression of inflammatory processes may render them promising targets for therapeutic intervention in diabetic trauma.
MicroRNAs
MicroRNAs (miRNAs) are small, endogenous, non-coding RNAs of ∼22 to 26 nucleotides in length that function primarily as post-transcriptional regulators.81,82 MiR-9, miR-127, miR-155, and miR-125b promote M1 polarization of macrophages, while miR-124, miR-223, miR-34a, miR-132, miR-146a, and miR-125a-5p induce M2 polarization by targeting various transcription factors and adaptor proteins. 82 However, contrasting views have been held regarding the role of miR-21 in mediating macrophage polarization. In a study conducted by Caescu et al., 83 knockdown of miR-21 resulted in a reduction of M2 phenotypic genes, including arginase 1, mannose receptor 1, IL-4Ra, and FIZZ in macrophages. Furthermore, these authors found that miR-21 inhibited the expression of SIRPb1, an activator of the MEK/ERK1/2 pathway, thereby further promoting M2 polarization. Nevertheless, Wang et al. 84 reported that miR-21 promotes M1 polarization and inhibits M2 polarization in peritoneal macrophages. Mechanistically, miR-21 acts as an endogenous brake to block PGE2-mediated M2 macrophage generation by targeting STAT3. However, Sheedy et al.’s 85 findings reconcile these conflicting views by proposing that pri-miR-21 primarily drives macrophage polarization toward the M1 phenotype during the early phase of inflammation, while mature-miR-21 exhibits anti-inflammatory properties and promotes macrophage polarization toward the M2 phenotype during the regressive phase. Indeed, further research is warranted to validate this conclusion.
In the aberrant microenvironment of diabetic wounds, there is a significant downregulation in the expression levels of miRNA-497 and miR-185-5p. Restoration of these specific miRNAs has demonstrated effective mitigation of wound inflammation, acceleration of wound healing, and promotion of epithelialization.86,87 Furthermore, myofibroblasts secrete exosomes enriched with miR-224 and transfer them to macrophages within an inflammatory milieu, thereby inhibiting M2 polarization, while the M1 secretome further stimulates myofibroblast proliferation and induces an exacerbated inflammatory response. 88 Alternatively, stem cells involved in wound repair also modulate macrophage polarization by releasing exosomes enriched with specific miRNAs. For example, exosomes derived from epidermal stem cells (EpiSC-EXOs) overexpressing miR-203a-3p activate the SOCS3/JAK2/STAT3 signaling pathway and induce M2 macrophage polarization, 89 and mesenchymal stem cell-derived exosomes (MSC-EXOs) enhance M2 macrophage polarization and promote vascular regeneration through upregulation of miR-146a levels. 90 In conclusion, miRNA-based cellular reprograming technology holds promise as a potential therapeutic strategy for modulating the diabetic wound healing response.
Metabolic reprograming
Glucose metabolism
Glycolysis serves as the primary cellular energy supply mechanism. M1 and M2 utilize distinct glucose metabolism pathways for energy acquisition: the former rapidly relies on glycolysis, whereas the latter predominantly utilizes oxidative phosphorylation (OXPHOS) and fatty acid oxidation (FAO).91–93 In diabetic wound tissue, the glycolytic activity in M1 macrophages exhibits similarities to the Warburg effect observed in tumor cells, characterized by increased glucose consumption and lactic acid release, as well as decreased oxygen consumption. 94 Pyruvate kinase M2 (PKM2) is a critical determinant of the activation of LPS-induced inflammatory response in M1 macrophage. 95 Lactate inhibits the Warburg effect by activating PKM2, thus promoting the transition of pro-inflammatory macrophages to a reparative phenotype and facilitating wound healing in mice, and further exploration revealed that the promoting effect of lactate occurs during acetylation of the K62 site of PKM2, which prevents its tetrameric to dimeric transition, enhances PKM2 activity, and reduces nuclear distribution. 96 Moreover, upregulation of the glucose transporter GLUT1 induces enhanced glucose uptake for metabolic reprograming, whereby IL-10 exerts its by modulating GLUT1 translocation and regulating the expression of glycolytic enzyme genes (Hk1, Hk3, Pfkp, and Eno2) to suppress glycolytic flux while promoting OXPHOS. 97
Once produced in glycolysis, pyruvate is decarboxylated to acetyl-CoA, which is oxidized through a series of reactions known as the Kreis cycle or Tricarboxylic Acid (TCA) cycle to generate hydrogen, which reduces NAD+ and FAD to NADH and FADH2. These hydrogen delivering bodies produce large amount of ATP by oxidative phosphorylation in the inner mitochondrial membrane. It is well known that M2-like macrophages display a functional and intact TCA cycle, a process that produces ATP to satisfy the high glycosylation levels of lectin and mannose receptors, which are essential for M2 macrophage function. 98 In contrast to M2 metabolism, proinflammatory macrophages exhibit an augmented glycolytic flux and a concurrent disruption of the TCA cycle at two crucial junctures, facilitating citrate and succinate, which are also pivotal for sustaining the inflammatory response. Excessive succinate induces macrophage transition to the M1 phenotype by stabilizing hypoxia inducible factor-1α (HIF-1α), a process attributed to succinate’s inhibition of prolyl hydroxylase (PHD) activity and promotion of HIF-1α stability (Figure 3).91,99 Another study also showed that increased glycolytic flux in LPS-stimulated M2 macrophages resulted in a high intracellular accumulation of succinate, which in turn augmented HIF-1α-dependent IL-1β expression and ultimately exhibited significant pro-inflammatory effects. 100 Notably, HIF-1α also upregulates the activity of GLUT1 and PKM2, indicating its important role in the metabolic reprograming of M1 (Figure 3). 101 Citrate further generates itaconate, which conversely inhibit the Krebs cycle enzyme succinate dehydrogenase (SDH), thereby blocking ROS production in complex I and inhibiting HIF-1α activity.102,103 Itaconate has also been reported to activate NRF2 signaling and reduce the p38 signaling pathway to hinder inflammation and cause the abrogation of the pro-inflammatory phenotype in macrophages.104,105 However, contrary to previous findings, Runtsch et al. 106 indicated that itaconate blocks alternative activation of macrophages by inhibiting JAK1/STAT6 phosphorylation and downstream signaling.
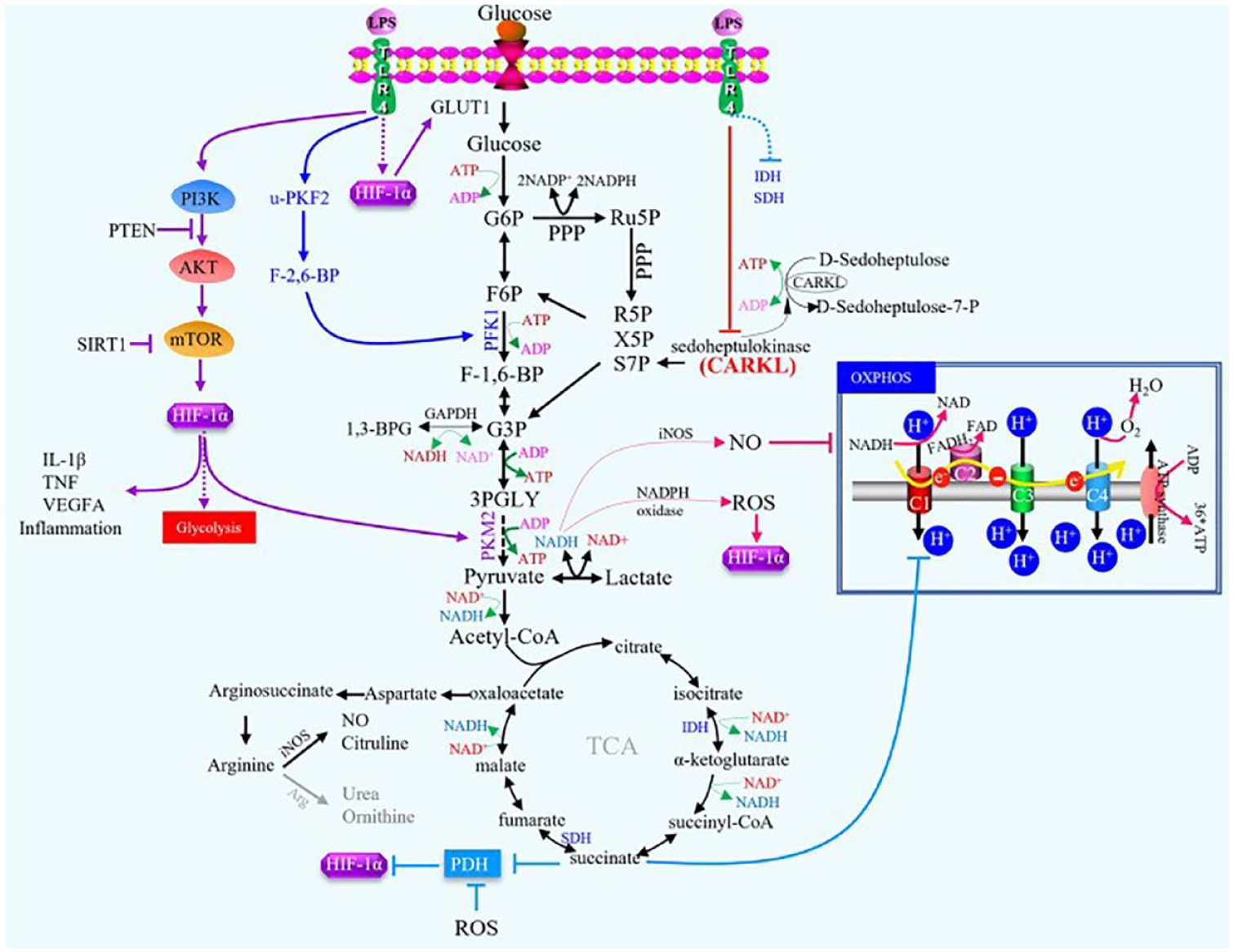
Schematic diagram of glucose metabolism in inflammatory macrophages. LPS-induced M1 macrophages increase HIF-1α via the P13K/AKT/mTOR pathway and promote glycolysis. Reduced oxidative phosphorylation (PPP) that allows the intermediate metabolite succinic acid to accumulate in the cell, which exerts pro-inflammatory effects by inhibiting the activity of prolyl hydroxylase (PHD) and HIF-1a stability. In addition, HIF-1α also acts directly on the glucose transporter GLUT1, causing cells to take up more glucose for glycolysis and PPP. These results illustrate the key role of HIF-1a in the metabolic reprograming of M1 macrophages. Reproduced under terms of the CC-BY license. 101 Copyright 2023, Li et al., published by Springer Nature.
In addition to glycolysis, upregulation of the pentose phosphate pathway (PPP) is another glucose catabolism pattern that triggers inflammation in classically activated macrophages. Glucose-6-phosphate, an intermediate metabolite of glycolysis, flows into the pentose phosphate pathway and promotes the synthesis of reduced nicotinamide adenine dinucleotide phosphate (NADPH), which transfers electrons to oxygen molecules under NOX-2, upregulates ROS levels in macrophages and modestly stimulates the expression of pro-inflammatory genes. 107 Itaconic acid is produced by activating the enzyme encoding immune response gene 1 (IRG1) in macrophages. Another mechanism by which itaconic acid acts as an anti-inflammatory metabolite is to significantly promote the pentose phosphate pathway, thereby up-regulating the production of NADPH oxidase and ROS, which increase the expression of anti-inflammatory gene A20 and subsequently reduced the production of inflammatory cytokines IL-6, IL-1β, and TNF-α. 108 Glucose-6-phosphate dehydrogenase (G6PD) is a key rate-limiting enzyme in the pentose phosphate pathway, G6PD is involved in pro-inflammatory responses accompanied by oxidative stress in macrophages. Among various environmental factors, FFAs and LPS promote high expression of G6PD in macrophages, which in turn activates downstream MAPKs and NF-κB signaling pathways, causing intracellular oxidative stress and expression of pro-inflammatory cytokines, which switch toward the M1 polarization. 109 Notably, Haschemi et al. 110 employed a kinase screening strategy to discover that sedoheptulose kinase CARKL of the pentose phosphate pathway coordinates the metabolic state of macrophages. Metabolomics analysis revealed that M2-like metabolic reprograming required the up-regulation of CARKL, while M1-like metabolic reprograming did the opposite, refocusing cell metabolism to a high-redox state (Figure 3). Intriguingly, CARKL functioned as a negative regulator of LPS-induced SOCS3 expression but resulted in enhanced STAT3 phosphorylation, which possibly exact molecular mechanism by which CARKL regulates M1/M2 activation.
Lipid metabolism
Lipids and their metabolites are increasingly recognized as a profound effect in regulating macrophages, including modulating their response to inflammation. While M1 engage in lipid accumulation and phagocytosis primarily through lipogenesis, M2 possibly rely on fatty acid β-oxidation as their dominant energy source. 111 Sterol regulatory element-binding protein-1 (SREBP-1) is one of the momentous nuclear transcription factors for lipid metabolism and mainly modulates the biosynthesis of fatty acids, triglycerides, and cholesterol. LPS-induced M1 macrophage responses to SREBP-1a-depe lipogenesis, inflammatory cytokine production and phagocytosis directly via TLR4/mTORC1 signaling pathway (Figure 4).112,113 Moreover, the uptake and utilization of glucose in macrophages also supports lipid synthesis, for example, citrate produced by the broken TCA cycle is transferred to acetyl-CoA pools in the cytoplasm needed for FA synthesis. The pentose phosphate pathway is also upregulated by LPS and reduces power in the form of NADPH required for FA synthesis (Figure 4). 113 Caspase-11, a protease with increased expression upon stimulation by TLR4 agonists such as LPS or IFN-γ, is part of the atypical activation of inflammatory vesicles and pyroptosis, which is positively correlated with SREBP activation. Recent studies have shown that SREBP-1a activation is dependent on caspase-11 interacting with SREBP processing via site 1 protease (S1P). 114 Early experiments demonstrated that oxidized low-density lipoprotein (ox-LDL) increased ROS production, NLRP3 inflammasome activation and release of active IL-1β, followed by overexpression of sterol regulatory element binding protein 1 (SREBP-1) and its downstream protein fatty acid synthase, contributing to lipid overaccumulation and foam cell formation in macrophages. Among them, the NLRP3 inflammatory vesicle is likely to be responsible for the upregulation of SREBP-1, as siRNA silencing of NLRP3 decreases the level of SREBP-1. 115 In contrast, HDL is a repressor of macrophage induction toward the M1 phenotype by the specific mechanism that HDL suppresses ERK1/2 and STAT3 phosphorylation by declining caveolin-1 in M1 macrophages, and caveolin-1 is necessary for HDL to inhibit M1 induction. 116 Taken together, the SREBP-1 signaling pathway-mediated lipogenesis contributes to the pro-inflammatory response and phagocytic activity of M1.
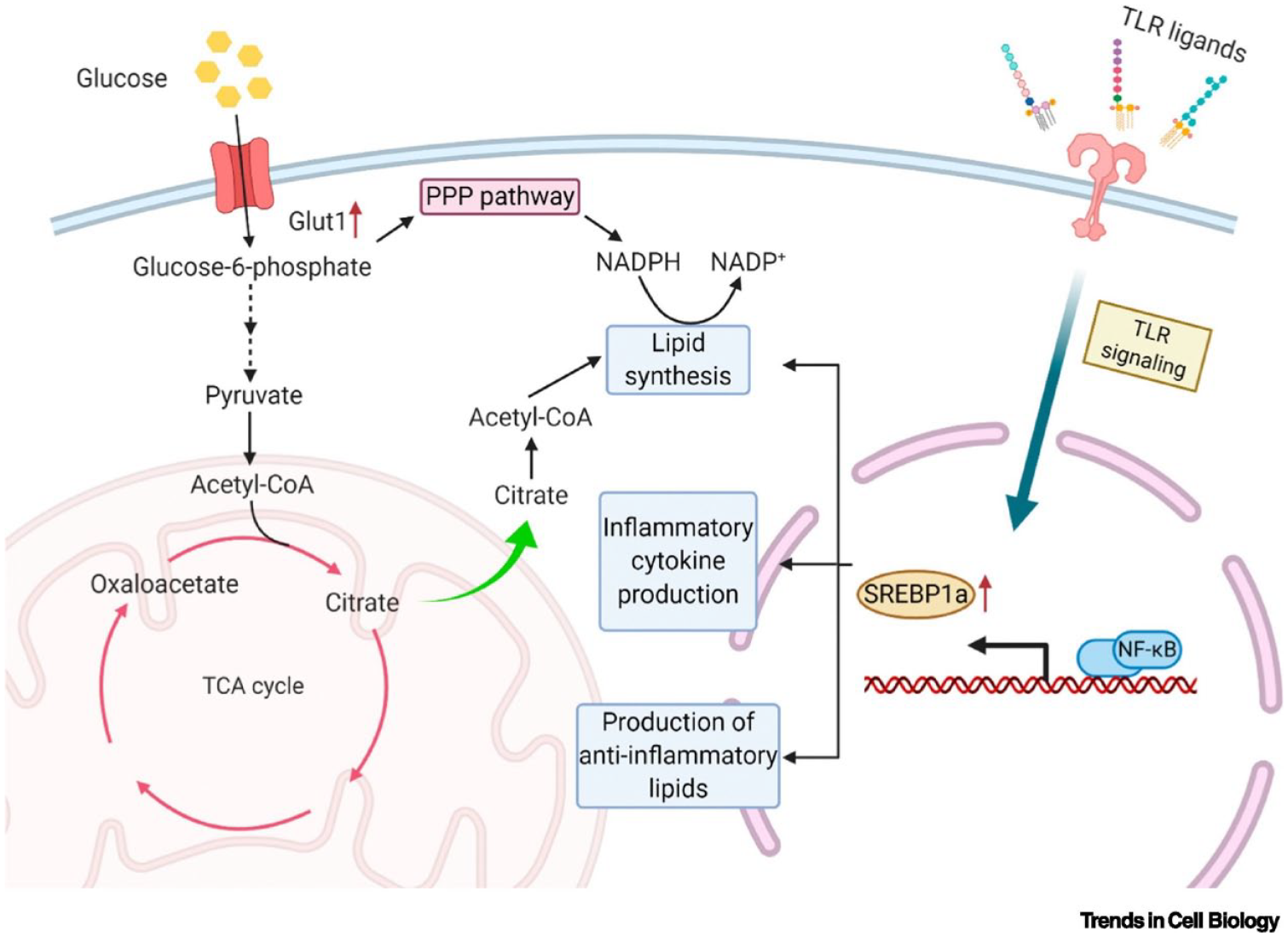
Lipid synthesis in M1 macrophages. M1 macrophage induced by Toll-like receptors (TLR) signaling promote fatty acid (FA) synthesis and inflammatory cytokine production by activating SREBP1α. At the same time, the broken TCA cycle in the mitochondria of M1 macrophages leads to the accumulation of citrate, which is transferred to the cytoplasm to produce acetyl-CoA needed for FA synthesis. The pentose phosphate pathway (PPP) is also upregulated to support the NADPH required for FA synthesis. Reproduced with permission. 113 Copyright 2020, Elsevier.
IL-4 signaling activates transcriptional activator 6 (STAT6) to support fatty acid transport and oxidation in alternately activated macrophages (Figure 5). This process is also regulated by other signaling pathways; for example, nuclear receptor peroxisome proliferator-activated receptor γ (PPAR-γ) upregulates fatty acid transporter protein CD36 to increase fatty acid influx, followed by binding of medium/long-chain fatty acids to carnitine and their transport to mitochondria for fatty acid oxidation (FAO) by carnitine palmitoyl transferase 1A (CPT1A) and eventually promotes the reprograming of macrophages from pro-inflammatory to pro-absorptive phenotype (Figure 5). 117 FATP1, an integral membrane protein with long-chain and ultra-long-chain fatty acid acyl-CoA synthetase activity, exerts a facilitatory role in FFA influx. In oleic acid-treated macrophages, activation of PPAR-α and PPAR-γ attenuated FFA influx via repressing Fatp1 expression, and subsequently reducing total FFA and TG accumulation. 118 Taken together, therapeutic strategies to reduce total FFA and TG accumulation through activation of PPAR signaling, repressing FFA input and TG synthesis, and facilitating FFA oxidation are effective approaches to alleviate wound inflammation, and notably, the negative effects of PPAR should not be overlooked, such as the upregulation of CD36 that promotes FFA uptake. 118
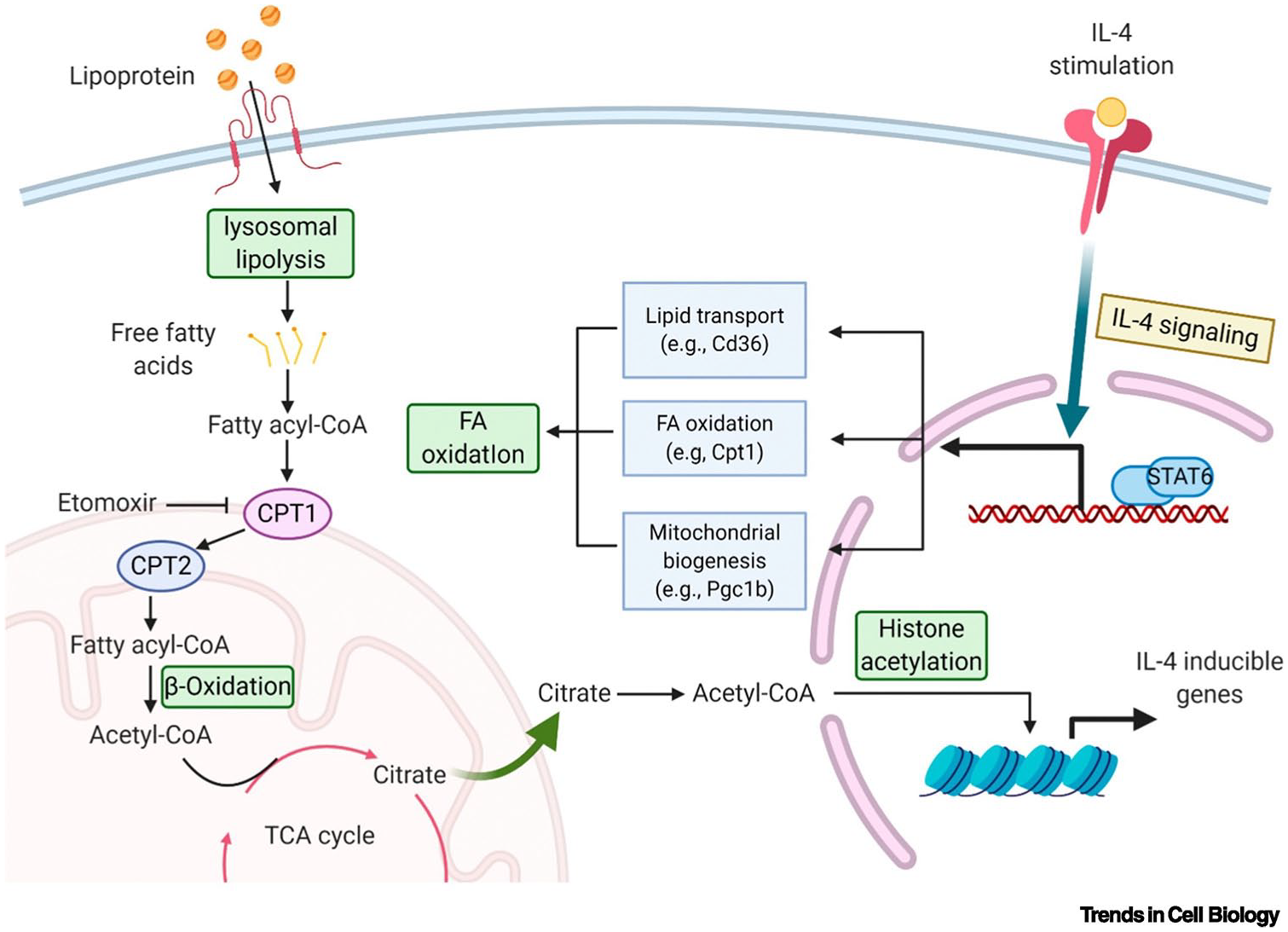
Lipid Metabolism in M2 macrophages. IL-4 signaling promotes lipid transport and fatty acid oxidation (FAO) by activating transcriptional activator 6 (STAT6). Carnitine palmitoyl transferase 1/2 (CPT1/2) transports free fatty acids to mitochondria for FAO, which is regulated by peroxisome proliferative activated receptor gamma (PPAR-γ). Some studies have shown that FAO partially drives histone acetylation and IL-4 induced gene expression through the production of acetyl-CoA pool, thereby activating M2 macrophages. Reproduced with permission. 113 Copyright 2020, Elsevier.
Furthermore, cholesterol levels in macrophages are also influenced by various factors such as SREBPs, PPARs, and LXRs, which play a role in regulating inflammatory responses. Cholesterol crystals act as potent mediators of inflammasome activation and DAMP expression in primary human macrophages, thereby driving M1 polarization. 119 Liver X receptor (LXR), a crucial transcription factor for cholesterol efflux and inflammatory gene responses in macrophages, has been found to promote the polarization of macrophages toward the M2 through enhancing ATP-binding cassette transporter protein A1 (ABCA1) and ABCG1-dependent cholesterol efflux. 120 Recently, a study showed that the LXR-dependent transcriptional pathway controls macrophage cholesterol efflux and curtails the aggravated inflammatory response by modulating Cav-1 expression and intramembrane compartmentalization. 121 In addition, PPAR-γ also promotes cholesterol removal by activating the nuclear receptor LXRα to induce ABCA1 expression in macrophages. 122 SREBP-1a upregulates the expression of cholesterol 25-hydroxylase transcripts, leading to enhanced production of 25-hydroxycholesterol (25-HC), an endogenous agonist of hepatic X-receptor alpha (LXRα) that increases cholesterol efflux to limit cholesterol accumulation and M1 polarization, 123 meanwhile, the production of 25HC by cholesterol-25-hydroxylase (CH25H) negatively influences further cholesterol biosynthesis. CH25H gene deletion causes cholesterol accumulation in macrophage, leading to reduced cholesterol-dependent mitochondrial respiration and release of mitochondrial DNA into the cytoplasm, which activates inflammasome of absent in melanoma 2 (AIM2) to trigger IL-1β production and promote pro-inflammatory macrophages. 124 Lipid accumulation in macrophages is known to be an important pathological event in the development of atherosclerosis, which may occur equally in diabetic trauma. Therefore, modulating macrophage function by reprograming its lipid metabolism may become a promising therapeutic strategy.
Amino acid metabolism
Increasing evidence supports the critical role of cellular amino acid metabolism in regulating inflammatory function and highlights the central involvement of the arginine metabolic pathway. Arginine catabolism enters two distinct metabolic pathways via two encoded enzyme genes, iNOS and ARG1. M1 activate nitric oxide synthase through the high expression of iNOS, which converts arginine into NO and citrulline. Then, NO induces the transformation of macrophage metabolism to glycolysis through oppressing mitochondrial respiration, thus enhancing their pathogen-killing ability and defense against infection. Additionally, citrulline can be efficiently recycled for NO synthesis via the citrulline-NO cycle. In contrast, in M2, arginine is broken down into ornithine and urea by the arginase ARG1, afterwards, the metabolites of ornithine, polyamines, and proline, foster cell proliferation and collagen synthesis, which play a pivotal role in wound healing and tissue repair. 93
Glutamine metabolism potently interacts with macrophage metabolic and epigenetic reprograming, thereby orchestrating immune response. Glutamine is converted to glutamate by glutamine synthetase (GS1), which can further be transformed into α-KG, a substance that is essential for OXPHOS and FAO. Liu et al. 125 demonstrated that α-ketoglutarate enhances activation of M2 macrophages through JMJD3-dependent demethylation of H3K27 on M2-specific mark genes promoter (Figure 6). This pro-M2 activation mechanism is regulated by the ratio of α-ketoglutarate to succinate, with a high ratio enhancing M2 macrophage activation and a low ratio promoting M1 macrophage polarization. Furthermore, it is further shown that α-ketoglutarate may exert a regulatory role in restraining M1 activation by interfering with the NF-κB pathway through the activation of PHD and inhibition of IKK activation (Figure 6). Glutathione (GSH), a crucial antioxidant safeguarding cells against ROS, has shown potential in scavenging ROS by inducing Nrf2-mediated heme oxygenase-1 (HO-1) signaling pathway and is able to modulate M1 macrophage polarization to enhance immune activity. 126 Serine glycine, which is involved in glutathione metabolism, also plays a prominent role in the process of macrophage polarization. Inhibition of phosphoglycerol dehydrogenase (PHGDH), a key enzyme in the serine synthesis pathway, as well as restriction of exogenous serine or glycine, significantly enhance M1 macrophage polarization. This is attributed to the defect of serine metabolism, which diminishes the histone H3K27me3 modification in the IGF1 promoter region by down-regulating the level of S-Adenosyl-Lmethionine (SAM) in macrophages, thereby augmenting IGF1 expression. Subsequent activation of IGF1 relies on the p38-JAK-STAT1 axis to facilitate M1 polarization. 127 Methionine, an indispensable amino acid involved in crucial metabolic processes, plays an important role in maintaining redox homeostasis and regulating macrophage metabolism and activation. In the context of pro-inflammatory macrophage activation, methionine sulfoxide reductase B1 (MsrB1) effectively reduces oxidized methionine and protects GAPDH from ROS, consequently sustaining GAPDH activity for glycolysis. Notably, knockdown of MsrB1 in LPS/IFN-γ-induced macrophages leads to the loss of enzymatic activity of GAPDH, resulting in impaired glycolysis and hindered transition toward a pro-inflammatory phenotype. 128 Therefore, while LPS initially establishes the metabolic profile of macrophages and determines their transition to a pro-inflammatory phenotype, the modified redox status can feed back into the metabolic pathways of macrophages and reprogram them.
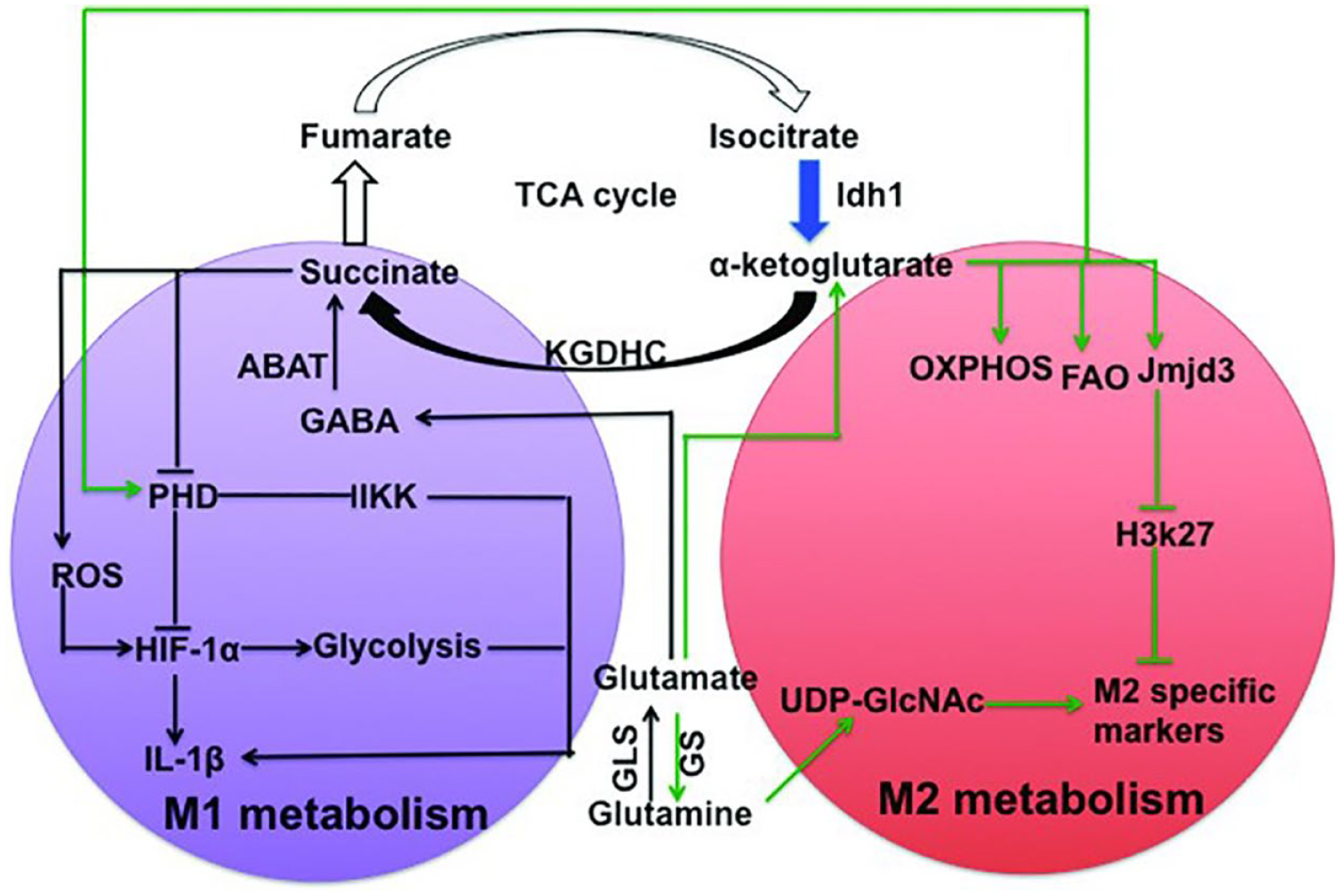
Glutamine metabolism involved in macrophage polarization. Succinate stabilizes HIF-1α by inhibiting PHD or promoting ROS production, thereby regulating the expression of IL-1β and other HIF-1α-dependent genes, including enzymes required for glycolysis, ultimately initiating M1 macrophages. Alpha-ketoglutarate from glutamine hydrolysis is essential for OXPHOS and FAO and activates M2 macrophages through demethylation of H3K27 on the Jmjd3-dependent M2-specific marker gene promoter and inhibition of IKK activation via PHD. The black pathway enhanced the activation of M1 macrophages, the green pathway promoted the differentiation of M2 macrophages, and the blue pathway are impaired. HIF-1α, hypoxia inducible factor 1α; ROS, reactive oxygen species; OXPHOS, oxidative phosphorylation; FAO, fatty acid oxidation; Jmjd3, Jumonji domain-containing 3; IKK, inhibitor of NF-κB kinase; PHD, prolyl hydroxylase domain. Reproduced under terms of the CC-BY license. 129 Copyright 2019, Ren et al., published by American Society for Nutrition.
Reprograming of macrophages by immunomodulatory hydrogels facilitates tissue regeneration
Hydrogels, constructed from the crosslinking of hydrophilic polymers, are three-dimensional mesh-like materials. They are regarded as highly promising for mimicking the natural extracellular matrix due to their porous structure, significant water content, excellent biocompatibility, and affinity for cells. Leveraging these inherent properties, hydrogels promote wound healing in a moist environment and can function as both permanent and temporary dressings for various types of wounds, aiding in the regeneration of both epidermal and dermal layers.130–132 Diabetic wound healing is hampered by a number of factors, including bacterial infection, excessive inflammatory cytokines, high levels of active oxides, and persistent hypoxia, all of which lead to impairment of wound immune homeostasis.133,134 Immunomodulatory behavior is focused on regulating the immune system’s activity in order to maintain a well-balanced response to external environmental stimuli. Achieving an appropriate balance between M1 and M2 macrophages to regulate the immune microenvironment has become a key pathway to accelerate tissue repair and regeneration. 135 Consequently, there is growing interest in developing hydrogels with precise immunomodulatory properties that can effectively regulate macrophage phenotypes, holding great promise for enhancing the therapeutic effectiveness of chronic wound treatments. In recent years, several immunomodulatory hydrogels based on macrophage reprograming have been developed for regenerative medicine, such as immunomodulatory hydrogels dependent on the intrinsic properties, hydrogel reservoirs for delivering immunomodulators, and nanohydrogel scaffolds for in situ regulation of immune cells. These versatile hydrogels accelerate wound healing in a variety of ways, including reducing inflammation, promoting angiogenesis, antibacterial, and antioxidant stress, and have confirmed that macrophages play an important role in the final outcome. Figure 7 and Table 1 summarize recent advances of these smart hydrogels in reshaping the immune microenvironment of diabetic wounds by reprograming macrophages to promote healing.
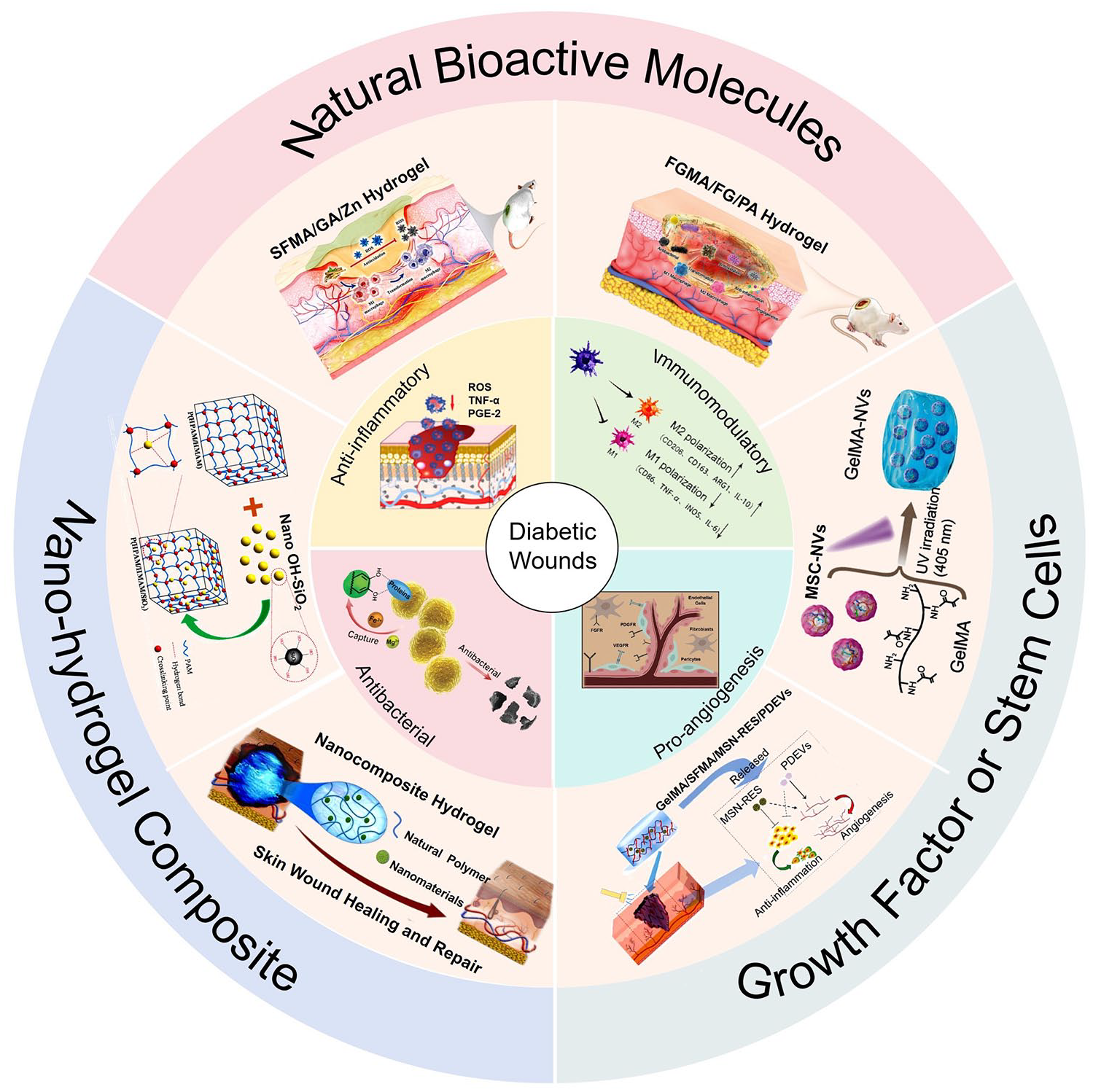
The schematics of contemporary applications of hydrogels that accelerate diabetic wound healing by reprograming macrophages, encompass hydrogels containing natural bioactive molecules, hydrogels supplemented with stem cells or growth factors, and nanohydrogel composites, which possess anti-inflammatory, immunomodulatory, pro-angiogenesis, antibacterial properties. Reproduced with permission. 136 Copyright 2023, Wiley-VCH GmbH. Reproduced with permission. 137 Copyright 2022, Wiley-VCH GmbH. Reproduced with permission. 138 Copyright 2023, Wiley-VCH GmbH. Reproduced with permission. 139 Copyright 2022, Elsevier. Reproduced with permission. 140 Copyright 2023, American Chemical Society. Reproduced with permission. 141 Copyright 2022, Royal Society of Chemistry. Reproduced with permission. 142 Copyright 2022, Elsevier. Reproduced with permission. 143 Copyright 2019, Elsevier.
Different kinds of hydrogels for immunomodulation.

HA: hyaluronic acids; TA/Fe3+: tannic acid/ferric ion; BSP: bletilla striata polysaccharide; FG: fish gelatin; MA: methacrylate; PA: protocatechuic aldehyde; SF: photo-crosslinked methylacrylated silk fibroin; GA: glycyrrhizic acid; MSC-NVs: MSC-derived nanovesicles; DPL: dendritic polylysine; hADSCs-Exo: human adipose-derived stem cell exosomes; ADAS: adipose-derived adult stem cells; NIR: near-infrared irradiation; MSN-RES: resveratrol nanoparticles; BP: black phosphorus; bFGF: basic fibroblast growth factor; CS: chitosan methacryloyl; HA-MA: hyaluronic acid methacryloyl; PDEVs: platelet-derived growth factor; SFMA: silk fibroin glycyl methacrylate; lipoSDF-1α: liposomal stromal cell-derived factor-1α; MSN: mesoporous silica nanoparticles; AE: artemisia absinthium extract.
Hydrogels with intrinsic immunomodulatory properties
Natural hydrogel dressings hold significant potential in tissue repair applications due to their exceptional cytocompatibility, safety, and biodegradability. 136 Some natural polymers, such as hyaluronic acid, alginate, chitosan, etc., possess immunomodulatory properties that effectively regulate macrophage phenotype and behavior. Consequently, hydrogels formulated with these substances have gained considerable popularity within the field of remediation.
Hyaluronic acid (HA), a naturally occurring polysaccharide, possesses diverse biological activities, and its therapeutic role in managing various inflammatory diseases is well-established. Its anti-inflammatory and pro-inflammatory properties are pivotal in this regard.144,157 Studies have shown that various molecular weights of HA can modulate macrophage phenotype. Specifically, high molecular weight HA (3000 kDa) promotes M2 polarization, whereas low molecular weight HA (5 kDa) enhances M1 polarization. 158 Hence, selectively increasing the molecular weight of HA to construct scaffolds may present a promising strategy for enhancing immunosuppressive efficacy.
Chitosan (Ch), a natural polysaccharide derived from the deacetylation of chitin, is characterized by a high content of modified amino groups and inherent antibacterial activity. 159 In addition, Ch can influence the immune response around implanted biomaterials. It has been reported that macrophages have significantly different responses to Ch 3D porous scaffolds with different degrees of acetylation. Ch scaffolds with lower DA (5%) can induce repair response of M2 macrophages, while Ch scaffolds with higher DA (15%) can induce severe inflammatory response of M1 macrophages. Furthermore, lower levels of acetylation in Ch scaffold are more likely to alter regenerative medicine outcomes. 160
Indeed, due to the biodegradability of natural polymers, proteolysis triggered by chronic wound inflammation is considered a significant hurdle in utilizing biomaterials for tissue engineering or medical implants. Additionally, the mechanical stability of a singular hydrogel matrix as a biomaterial is often suboptimal along with limited immunomodulatory capacity. Strategically, the incorporation of synthetic polymer hydrogels and the integration of other bioactive agents can augment their biological activity while bolstering their resilience to facilitate immune modulation and tissue regeneration. Natural bioactive ingredients have attracted more and more interest in the application of wound healing due to their multi-functional intrinsic bioactivity, excellent biocompatibility, rich sources and easy processing.
Protocatechuic aldehyde (PA), a natural compound extracted from salviorrhiza, has demonstrated potent anti-inflammatory effects and is utilized as an active ingredient in the treatment of coronary heart disease. 161 Fu et al. 162 synthesized a biocompatible hydrogel comprising a physically entangled network formed by FG/PA and a chemically cross-linked network formed by photo-crosslinked methacrylate gelatin (FGMA). The physical cross-linking allows the hydrogel to adapt to deformations during wound motion, while the optical cross-linking enhances its structural stability. Additionally, the complex microenvironment of diabetic wounds is prone to bacterial invasion, and the catechol groups present in PA effectively interact with bacterial surface membrane proteins, thereby impeding bacterial activity and proliferation. After treatment with the hydrogel, LPS-stimulated M1 macrophages reprogram to M2, which effectively shortens the inflammatory period while stimulating angiogenesis and epithelialization (Figure 8(A)). Additionally, wounds treated with the hydrogel showed a well-defined epidermal layer with a higher proportion of dermal appendages and fresh collagen volume. 136
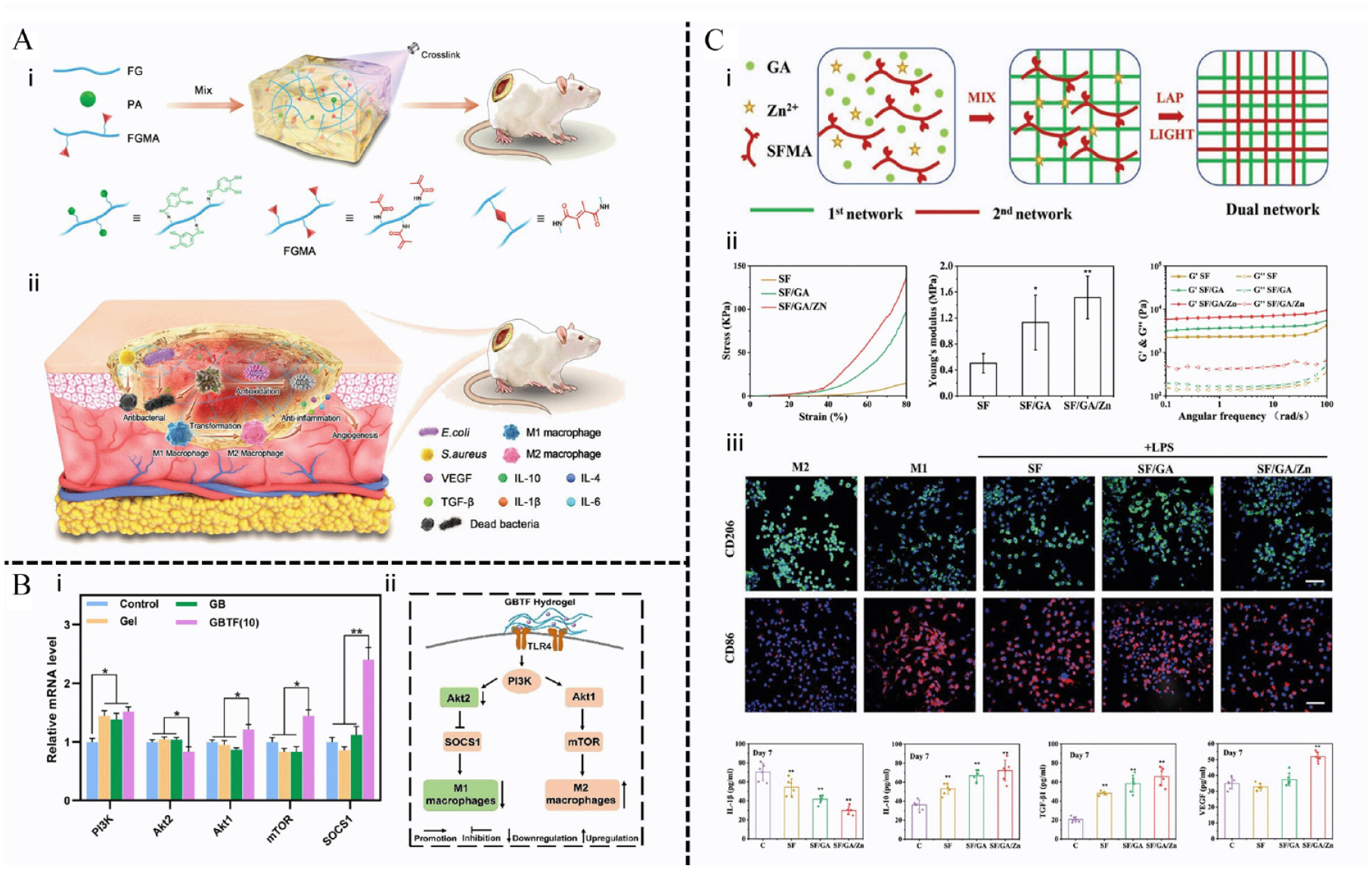
Hydrogels with natural bioactive molecules for diabetic wound healing. (A) All-natural hydrogel (FGMA/FG/PA) for treating diabetic wounds: (i) scheme illustration of the preparation of the all-natural hydrogel; (ii) illustration of the mechanism for promoting wound healing, including antibacterial effect, anti-inflammation, angiogenesis. Reproduced with permission. 136 Copyright 2023, Wiley-VCH GmbH. (B) GBTF hydrogel regulate macrophage polarization: (i) the relative mRNA of Akt1, Akt2, PI3K, mTOR, and SOCS1 in macrophages co-cultured with control, Gel, GB, GBTF hydrogel; (ii) the proposed mechanism of M2 macrophage polarization by the interaction with GBTF hydrogel. Reproduced with permission. 146 Copyright 2023,Elsevier. (C) Effects of multifunctional Hybrid Hydrogel for wound healing: (i) the schematic of the two-step fabrication process for obtaining the SF/GA/Zn hybrid IPNs hydrogel; (ii) the mechanical properties of SF/GA/Zn hydrogel were quantified, showing better elastic, mechanical properties, and stability; (iii) SF/GA/Zn hydrogel promote M2 macrophage polarization and reduce the production of inflammatory factor. Reproduced with permission. 137 Copyright 2022, Wiley-VCH GmbH.
Bletilla striata polysaccharide (BSP) is a naturally active polysaccharide extracted from the traditional Chinese herb Bletilla striata. Historically, it has been utilized for its hemostatic, wound healing promotion, and anti-inflammatory properties. 163 D-mannose residues on the BSP backbone play a crucial role in orchestrating the reprograming of macrophages during both inflammatory and proliferative phases. They achieve this by selectively engaging mannose receptors, which in turn promote collagen deposition and facilitate wound healing. 164 An increasing number of studies have demonstrated the utilization of BSP in the preparation of hydrogel scaffolds, showcasing its high potential for development in wound healing applications.165,166Indeed, various natural ingredients or metal ions can be incorporated to modify the BSP substrate material. For instance, Xu et al. 146 synthesized the GBTF hydrogel using gelatin methacrylate (Gel-MA), BSP, and tannic acid/iron ion (TA/Fe3+) through photopolymerization. The photothermal conversion of TA/Fe3+ endows the biomaterial with excellent antimicrobial properties and compressive strength. In addition, GBTF hydrogels reprogram macrophages to M2 via activating the PI3K/Akt/mTOR signaling pathway to accelerate angiogenesis and promote diabetic wound regeneration (Figure 8(B)). Mechanistically, mTORC1 and Akt are involved in the regulation of macrophage glycolysis. As previously mentioned in Figure 3, the Akt-mTORC1 axis in M1 macrophages is enhanced by HIF-1α-mediated glycolysis flux in response to LPS signaling. It is worth reiterating that among Akt members, Akt2 participates in M1 and Akt1 supports M2. 167 Research has shown that Akt1-mTORC1 may increase the expression of n-glycosylated M2 markers on cell surface by promoting the hexosamine pathway and glutamine metabolism. 168
Glycyrrhizic acid (GA), derived from the licorice root, is a naturally occurring compound known for its inherent anti-inflammatory activity. It holds significant promise as an ideal component of advanced hydrogel dressings with intrinsic immunomodulatory properties. 147 For example, Qian et al. 137 formulated a mixed hydrogel based on GA for enhancing diabetic wound healing (SFMA/GA/Zn). The low concentration self-assembly of GA induced by Zn2+ effectively mitigated the concentration-dependent cytotoxicity associated with GA (⩾1%) and synergistically demonstrated anti-inflammatory properties. Moreover, the incorporation of photocrosslinked SFMA significantly enhanced the mechanical integrity of the hydrogel. This immunomodulatory hydrogel expedites the three stages of diabetic wound repair (inflammation, proliferation, and remodeling) by effectively promoting the phenotypic transformation of macrophages toward M2 (Figure 8(C)). This results in an increased production of VEGF to recruit endothelial cells and promote angiogenesis, as well as elevated levels of TGF-β1 to further recruit fibroblasts to the wound site and stimulate ECM synthesis. An innovative GA-functionalized biomimetic (HA-GA) hydrogel was developed with a GA-mediated secondary cross-linking network and primary hydrazine network, resulting in superior mechanical properties and a lower enzyme degradation rate. HA-GA gels attract more myeloid cells (CD45+CD11+) to produce large amounts of IL-10, which polarizes macrophages into an immunosuppressive M2 phenotype through the IL-10/STAT3 pathway. 145 In our opinion, this pathway may be involved in glycolytic reprograming of macrophages, as IL-10 inhibits glycolytic flux by regulating GLUT1 translocation. In addition, activated STAT3 has been shown to control the expression of IL-10 promoters, resulting in a positive feedback mechanism. 169 Recruitment of immune cells also leads to complete degradation of HA-GA on the 10th day of implantation, thereby reducing fibrotic envelope deposition.
In summary, hydrogels with inherent immunomodulatory properties demonstrate considerable potential in regulating immune responses, offering the advantages of cost-effectiveness, environmental friendliness, and straightforward synthesis processes. However, it’s essential to note that proteins and polysaccharides derived from natural sources may raise concerns regarding cytotoxicity, thus necessitating thorough biosafety verification for hydrogel systems. 170 Moreover, it is critical to employ rational design strategies that integrate suitable initiators and crosslinkers to improve the physical and chemical properties of hydrogels while addressing challenges related to inadequate mechanical performance and instability.
Hydrogel delivery systems that encapsulate immunomodulatory molecules
Therapies based on stem cells and bioactive factors have gained considerable attention in the field of wound repair, but the low cell implantation rate and the instability of exogenous growth factors greatly limit their clinical applications. Subsequently, scientists have predominantly directed their endeavors toward creating effective cell delivery vehicles as a means to surmount the obstacles linked with the low survival and efficacy of directly injected stem cells. Hydrogel scaffolds designed for the delivery of immunomodulatory molecules, including stem cells, exosomes, growth factors, etc., represent a compelling category of biomaterials for localized and sustained delivery, aiming to enhance therapeutic efficacy while minimizing systemic side effects. 170 Moreover, hydrogel delivery systems can be engineered to finely tune the release kinetics of multiple bioactive agents and cells, allowing for effective synergistic immune regulation. 171
Mesenchymal stem cells (MSCs) are widely employed in regenerative medicine owing to their remarkable ability to differentiate into various functional cell types and exhibit potent immunomodulatory properties. Exosomes derived from MSCs are emerging as a promising cell-free therapeutic tool due to their diminished immunogenicity and enhanced tissue penetration capabilities.172,173 Taking advantage of this characteristic, Pang et al. 138 combined GelMA with MSC-derived nanovesicles (MSC-NVs) to produce GelMA-NVS for osteoarthritis under ultraviolet irradiation. The uniform and porous mesh structure of GelMA-NVS facilitates cell adhesion, diffusion, and growth, as well as the sustained release of MSC-NVs over an extended period, ensuring effective treatment. Moreover, the presence of miR-146 within MSC-NVs promotes epigenetic reprograming of macrophages to M2, exerting immunosuppressive effects and alleviating inflammation progression (Figure 9(A)). Zhu et al. 148 introduced an innovative injectable hydrogel carrier composed of dendritic lysine and HA crosslinked by a reversible Schiff base which endowed shear thinning and self-healing properties. ECM biomimetic components and highly symmetrical nanostructures derived from peptide dendrimers offer structural and mechanical support for cell growth and phenotypic regulation. Compared to naked ADSCs, ADSCs-laden hydrogels exhibit prolonged retention time in vivo, leading to enhanced reprograming of macrophage phenotype toward anti-inflammatory characteristics, as well as the inhibition of migration and invasion of rheumatoid arthritis (RA) fibroblast-like synovial cells (Figure 9(B)). Furthermore, temperature also influences the physical characteristics of biomaterial scaffolds. A low-temperature biodegradable scaffold composed of glycol chitosan and bifunctional polyurethane is employed to encapsulate adipose-derived adult stem cells (ADAS). 149 Frozen gels possess high porosity attributed to the formation of ice crystals, creating a rich internal space conducive to cell proliferation, thereby enhancing ADAS retention. ADAS-seeded cryogels contribute to ameliorating the inflammatory state and promoting tissue repair through various mechanisms, including the activation of C3a and C5a, upregulation of stromal cell-derived factor-1 (SDF-1) and TGF-β1 secretion, as well as the promotion of M2 macrophage transformation.
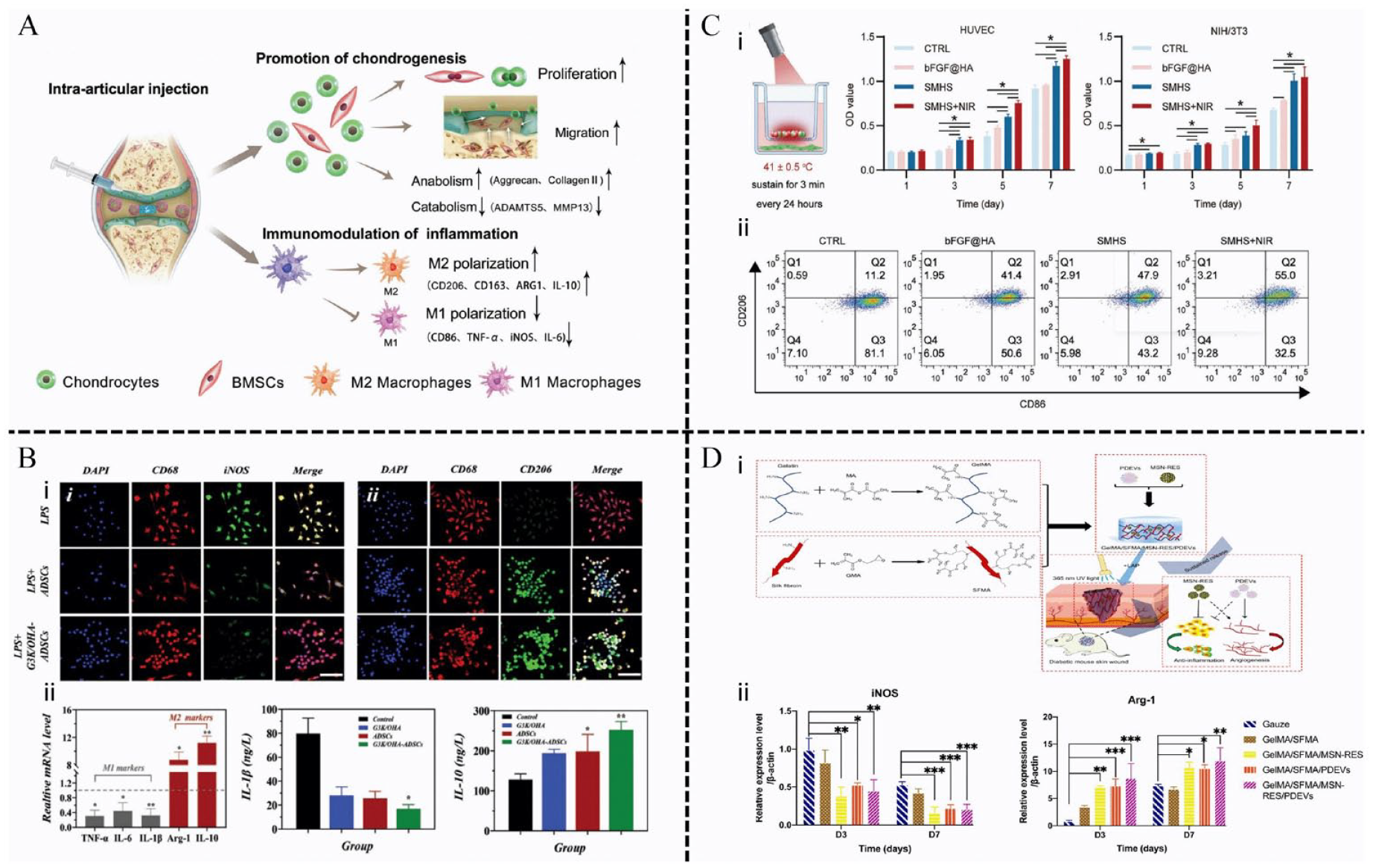
Hydrogels loaded with growth factors or stem cells promote M2 macrophage polarization. (A) MSC-NVs-encapsulated hydrogel was injected to treat osteoarthritis. Reproduced with permission. 138 Copyright 2023, Wiley-VCH GmbH. (B) G3K/OHA-ADSCs hydrogels regulate macrophage polarization: (i) immuno-fluorescence staining of CD68 (red), iNOS (green) and CD206 (green), and nuclei (blue) on macrophages without or with the treatment of G3K/OHA-ADSCs hydrogels; (ii) the mRNA levels of IL-1β and IL-10 in macrophages without and with the treatment of G3K/OHA-ADSCs hydrogels. Reproduced with permission. 148 Copyright 2023, Wiley-VCH GmbH. C)FGF loaded microsphere hydrogel improve wound healing under NIR irradiation: (i) The proliferation of HUVECs and NIH/3T3 cells was significantly accelerated after preparation of SMHS; (ii) the proportion of M2 macrophages (CD206) in the SMHS+NIR group was the highest among the four groups. Reproduced with permission. 150 Copyright 2023, Wiley-VCH GmbH. (D) A composite hydrogel loading MSN-RES and PDEVs for diabetic wounds: (i) schematic diagram of hydrogel preparation and application; (ii) effects of hydrogel on the secretion factors from macrophages. western blotting analysis of iNOS and Arg-1 levels in macrophages after 24 h of co-stimulation with treatments in a transwell system. Reproduced with permission. 139 Copyright 2022, Elsevier.
Growth factors, polypeptides, or proteins, regulate cell growth, migration, and differentiation, playing a pivotal role in tissue homeostasis and remodeling. Typically, under physiological conditions, growth factors have a short half-life and limited stability. Hydrogels effectively isolate the growth factor within the matrix, shielding it from rapid proteolysis or inactivation by enzymes. 174 For example, VEGF and PDGF-BB recombinant factors are covalently cross-linked with fibrin hydrogel to confer protection against enzymatic degradation, until the degradation of fibrin itself, thereby facilitating therapeutic arteriogenesis and expediting diabetic wound healing. 175 Certain hydrogels are engineered to respond to stimuli and environmental cues, which trigger the release of loaded growth factors. For example, Luo et al. 150 developed a hydrogel delivery platform (SMHS) that releases basic fibroblast growth factor (bFGF) in response to temperature changes. This system shows promise for the treatment of diabetic wounds. SMHS is self-assembled from positively charged black phosphorus nanosheets (BP) loaded chitosan microspheres and negatively charged bFGF loaded HA microspheres through electrostatic interaction. The concentration of BP influences the temperature change of biological materials upon near-infrared (NIR) irradiation, and SMHS degrades in response to this temperature change, releasing bFGF. In vitro experiments have shown that under the synergistic effect of photothermal therapy, SMHS promotes human umbilical vein endothelial cells (HUVECs) migration, neovascularization, and re-epithelialization by acting on the PI3K/AKT pathway. Additionally, it induces M2 macrophage polarization, thereby reducing inflammation (Figure 9(C)). A photo-responsive supramolecular hydrogel with adjustable crosslinking density has been created as a controllable delivery system for epidermal growth factor (EGF), aimed at promoting wound healing. The photo-switching property enhances EGF retention on the skin, resulting in a substantial promotion of granulation tissue formation and vascular regeneration. 176 Platelet-derived MVs reprogram macrophage gene expression and function by increasing Mir-126-3p levels in the macrophage genome. 177 Zhu et al. 139 developed a composite hydrogel by loading resveratrol nanoparticles (MSN-RES) and PDEVs with GelMA and silk fibroin glycyl methacrylate (SFMA) as raw materials. The complex hydrogel facilitates the in vivo transition of macrophages from M1 to M2 by upregulating CD73 and A2A-R expression (Figure 9(D)). CD73 converts immune-activating ATP into adenosine, which then interacts with the A2A receptor on immune cell surfaces. This interaction induces an immunosuppressive phenotype in macrophages by affecting Toll-like receptors, thereby shortening the inflammatory process and promoting wound healing.178,179 Suh et al. 180 proposed a new delivery model using a gelatin hydrogel loaded with the protein transduction domain (PTD) of recombinant human bone morphogenetic protein-2 (rhBMP2) formulated with lipid nanoparticles (LNP) that can prolong the half-life of BMP2 and demonstrate greater angiogenesis for effective treatment of diabetic wound healing.
The potential of hydrogels that encapsulate stem cells and growth factors to simultaneously achieve multifunctional wound treatment through a synergistic combination of physical (photothermal) and chemical (drug delivery) modalities holds great promise in the field. A key factor of the hydrogel as a delivery system: optimizing their release dose. Designing hydrogels with highly selective and specific binding sites to increase cell induction power is one strategy, and using affinity ligands in the vector can enhance the effect. In addition, the retention and release rates of growth factors are regulated by changing the parameters of the hydrogel network, such as crosslinking density, composition, or degree of functionalization. 174 Despite these technological advances, further research is needed on how to precisely control and optimize various growth factors in order to optimize therapeutic efficacy: (i) Exploring the use of other drugs in hydrogels in combination with growth factors requires further development to amplify the therapeutic effect and minimize the side effects of growth factors; (ii) Different tissues may respond differently to some growth factors; (iii) A deeper understanding of activation mechanisms at the molecular level and the environmental conditions that affect their function is needed. Anything else, the production process of this skin substitute is intricate and costly, posing challenges for clinical translation, while necessitating further verification of its long-term safety and efficacy. 174
Nanohydrogel composites with enhanced properties
Nanomaterials demonstrate exceptional mechanical properties in the wound environment, and their nanoscale properties are well suited to effective interaction with cells.181,182 Compared with single hydrogels, nanocomposite hydrogels have high encapsulation efficiency, that is, they can encapsulate and deliver biomolecules of various sizes, and the layer-by-layer surface functionalization promotes receptor-mediated targeted drug release, and realizes the adjustment of spatio-temporal and sequential control, which has a wider range of applications in wound management.183,184 At present, macrophage-based nano-composite hydrogels mainly exist in the form of fibers and micro-nanoparticles.
Nanofiber hydrogel composites 3D scaffolds have been commonly employed in tissue engineering owing to their resemblance to ECM in terms of morphology, as well as their adjustable chemical and physical characteristics that govern cellular behaviors and functions.185,186 ECM comprises immunomodulatory domains that selectively bind to immune cell receptors, facilitating adhesion and regulating their function. 187 Additionally, ECM acts as a structural scaffold promoting cellular migration, thereby creating an immune microenvironment conducive to tissue regeneration.170,187 Li et al. 154 reported a nanofiber-hydrogel composite material that replicates the microstructure and mechanical properties of adipose tissue ECM by binding the interfacial bond between the electrospun poly (ε-caprolactone) fiber and the hyaluronic acid hydrogel network. This interconnected network of micropores has been demonstrated to facilitate cell infiltration, revascularization, and integration of host tissue into the scaffold. After subcutaneous injection in a rat model, this complex allows host macrophages to infiltrate and enter the pro-regenerative phenotype by secreting MMPs (MMP2, MMP3, and MMP19), VEGF-D, and fibronectin 1 (Figure 10(A)). Consequently, these polarized macrophages gradually remodel the wound, allowing for its replacement with vascularized soft tissue. A photosensitive 3D HA-nanocomposite was developed to immobilize the Arg-Gly-Asp (RGD) adhesion peptide into an HA-based ECM and control its release under 302 nm UV irradiation. This coupling of ECM with the RGD peptide periodically activates the αvβ3 integrin of macrophages, crucial for maintaining the anti-inflammatory M2 phenotype and expediting endogenous tissue repair responses. 188 Feng et al. 151 designed a bioinspired nanofibrous glycopeptide hydrogel dressing possessing high water content, porosity, and self-repairing. This gel was demonstrated to upregulate the ERK / STAT6 signaling pathway via activation of the mannose receptor (MR), resulting in macrophage polarization to the M2, and full-layer skin defect models show accelerated wound healing rates and epidermal tissue regeneration (Figure 10(B)). IL-4 and MR co-activate the downstream signal STAT6 via JAK1 and ERK, respectively, to support fatty acid transport and oxidation, thereby promoting macrophage reprograming to M2. Henn et al. 152 developed an injectable nanofibrous hydrogel composite enriched with fractionated fat, which exhibits long-term structural integrity and minimal shrinkage upon in vivo implantation. Adipose-derived mesenchymal stem cells infiltrated into the composite along nanofibers enhanced the reprograming of M1-M2 macrophages in regenerated soft tissues and induced robust vascularization and tissue remodeling, effectively improving the limitations of fat transplantation for soft tissue reconstruction. Yu et al. 153 developed an innovative nanocomposite hydrogel system from which the pro-healing chemokine liposomal SDF-1α was released in a controlled manner to induce M2 macrophage recruitment and polarization to improve skin wound closure. Research confirms that the role of SDF-1α/CXCL12 extends beyond the recruitment and retention of blood monocytes. It also triggers a specific pathway for M2 macrophage differentiation by inducing the secretion of angiogenic factors VEGF and CCL1. 189 It thus appears that the structural and functional characteristics of ECM confer certain advantages to nanocomposite hydrogels, making them a promising approach for macrophage reprograming.
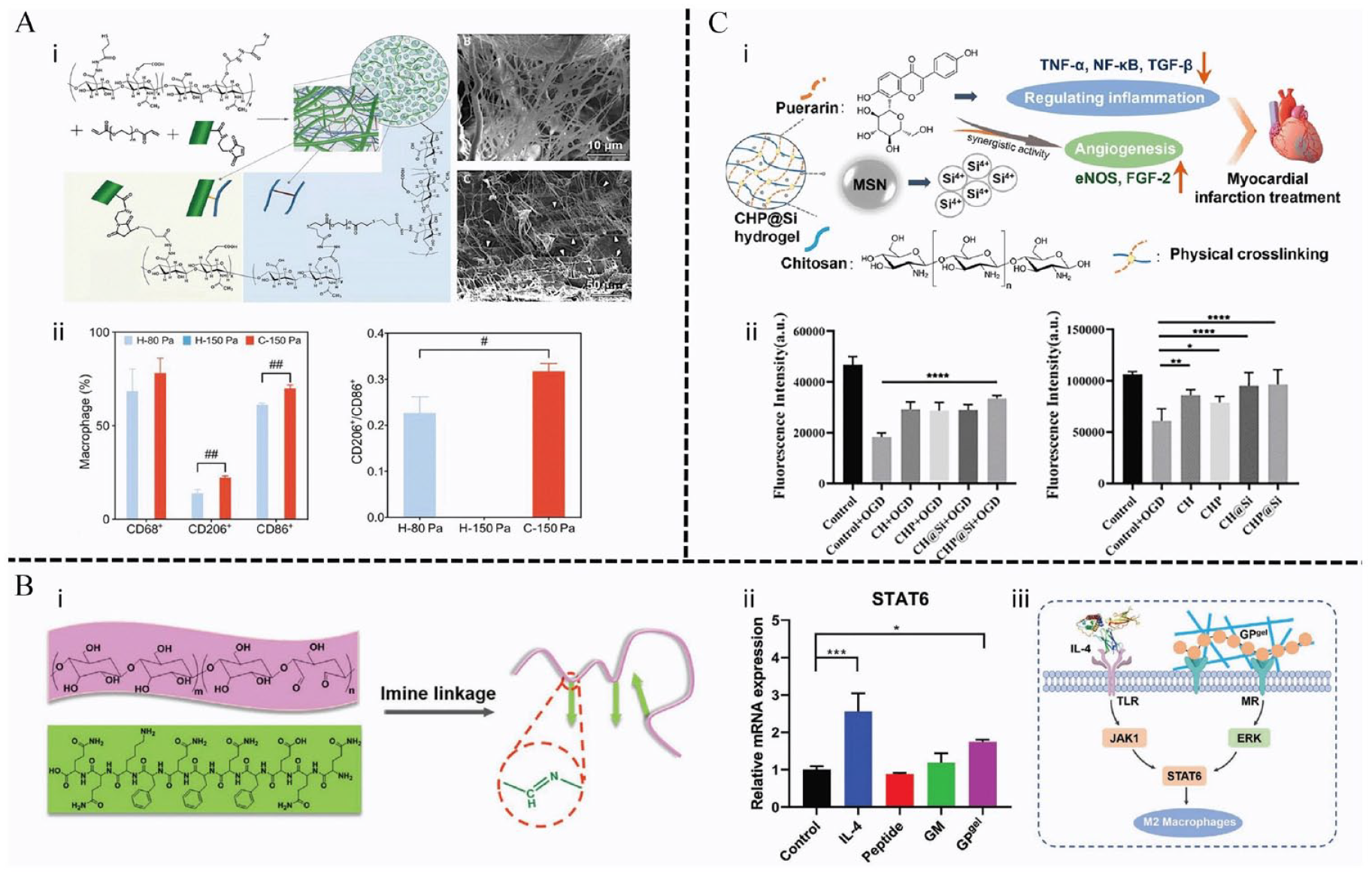
Nanohydrogel composites for wound healing. (A) Nanofiber-hydrogel composite simulated extracellular matrix: (i) the schematic diagram of the PCL nanofiber surface bonding with the HA hydrogel network. SEM image shows that the nanofiber is embedded in the HA hydrogel network (arrow); (ii) The hydrogel showed increased macrophage infiltration and M2-type polarization. Reproduced with permission. 154 Copyright 2019, The American Association for the Advancement of Science. B) Glycopeptide hydrogel regulated macrophage polarization: (i) scheme illustration of the fabrication process of glycopeptide hydrogel via dynamical imine bonds; (ii) relative mRNA expression of STAT6 in macrophages treated with IL-4, peptide, glucomannan, and glycopeptide hydrogel; (iii) the mechanism of M2 macrophage polarization by the interaction with glycopeptide hydrogel. Reproduced with permission. 151 Copyright 2020, Wiley-VCH GmbH. (C) Nano-silica hydrogel modulated macrophage polarization: (i) Silica ions and puerarin released from the hydrogel showed synergistic activity to improve angiogenesis and inhibit M1 macrophage polarization; (ii) the viability of HUVECs co-cultured with CH, CHP, CH@Si, and CHP@Si hydrogels under 4 h of OGD conditions and followed by 1 day of culture. Reproduced with permission. 156 Copyright 2023,Oxford University Press.
Nanoparticle hydrogel composites, as a novel class of nanomaterials, have exhibited remarkable advancements in the field of biomedicine. Nanoparticle hydrogel superstructures are highly tunable, demonstrating enhanced rates of cell-nanoparticle interaction and internalization. 190 As carriers for drug delivery systems, nanoparticle hydrogels can confine immunomodulators to specific sites and release them upon environmental stimuli, thereby enhancing the safety and therapeutic potential of immunotherapy. In addition, the nanoparticles can act as a binder between hydrogels and biological tissues, giving them superior mechanical properties. For instance, silica can serve as a binder to augment the affinity between water molecules and colloidal phases, thereby bolstering the strength and stability of the hydrogel. 142 It has been reported that the addition of silica improves the ductility and modulus of polyvinyl alcohol nanocomposite hydrogels, and inhibits the inhomogeneity of the composite gels. 191 Moreover, owing to its exceptional physical and chemical adsorption capacity, silica has remarkable potential to enhance drug stability and prolong drug administration duration, making it an optimal carrier for loading various drugs and proteins. 192 Artemisia absinthium extract (AE) has excellent antimicrobial and anti-inflammatory properties. Xue et al. 155 developed AE-loaded composite hydrogel scaffolds based on GelMA/HAMA and mesoporous silica nanoparticles (MSN) as slow-release drug carriers. The results showed this hydrogel effectively modulates M1-M2 macrophage polarization by up-regulating the expression of IL-4, TGF-β1, CD31, and α-SMA and down-regulating the expression of TNF-α and IFN-γ. Further analysis confirmed the integral role of MSNs in improving bioavailability, which may lead to a more favorable therapeutic effect in the treatment of chronic wounds associated with the regulation of macrophage polarization. Feng et al. 156 reported a novel injectable hydrogel (CHP@Si) containing puerarin and chitosan with simultaneous delivery of mesoporous silica nanoparticles. Prolonged puerarin modulates macrophage M2 polarization through inhibition of NF-κB and MAPK signaling cascade responses. Furthermore, the combined presence of silica ions and puerarin synergistically enhances HUVEC migration and promotes neovascularization in both conventional and hypoxic/glucose environments (Figure 10(C)).
Nanocomposites can achieve a variety of stimulus responses, sensing biomolecular or environmental changes, and regulating cell differentiation and proliferation. Meanwhile, they also have significant antibacterial effects, which can reduce the risk of infection and speeding up wound healing. However, high concentration nanoparticles have certain cytotoxicity, and how to strike a balance between anti-inflammatory properties and biosafety is an issue that needs to be considered. Furthermore, the heterogeneity exhibited by diverse wounds poses limitations on both the regenerative potential of nanomaterials and the optimization of manufacturing processes. 53
Conclusion and future perspectives
The article provides an overview of the fundamental mechanisms underlying the blockade of M1 macrophage to M2 macrophage polarization in diabetic wounds. It also explores various approaches to promote macrophage reprograming toward the M2 phenotype, highlighting the immunomodulatory effects and therapeutic efficacy of different hydrogel systems in tissue regeneration. Dysfunction in macrophages among diabetic patients not only reduces the polarization of reparative M2 macrophages but also impairs their ability to clear and engulf pathogens, thereby increasing the risk of wound infection and amputation. Efforts to restore this immune homeostasis have led to increased interest in methods to regulate macrophage reprograming, including interventions targeting metabolic pathways and epigenetic regulation. Understanding the relevant molecular mechanisms serves as a basis for developing macrophage-based diagnostic and therapeutic strategies. However, several challenges remain to be addressed. Macrophages exhibit biphasic functions during the healing process, and the M1 and M2 polarization states are not mutually exclusive. Determining the optimal timing to promote polarization switching for enhanced healing poses a challenge for future research. Moreover, consideration must be given to the long-term effects of reprograming. If M2 macrophages persist beyond the healing phase, it raises concerns about their impact on fibrosis formation in traumatic scars. Although various subtypes of macrophages have been identified, discussions often focus on the impact of reprogrammed macrophages on M1 and M2 polarization states for convenience. Future research should aim to further understand the potential impacts of other functional subsets on diabetic wounds. This knowledge will undoubtedly contribute to promoting more accurate and efficient wound healing in the future.
Hydrogels exhibit excellent biocompatibility, adjustable physical and chemical properties, and pleiotropic characteristics, making them promising for immune regulation in various applications. This paper emphasizes the immunomodulatory role and therapeutic efficacy of diverse hydrogel systems in tissue regeneration, particularly in macrophage reprograming. These include hydrogels with intrinsic immunomodulatory properties, hydrogel delivery systems loaded with immunomodulatory molecules, and nanohydrogel composites of biomimetic ECM. Achieving ideal immunomodulatory effects can be realized by regulating the physical and chemical properties of hydrogels, such as porosity, topological structure, electrostatic effect, hydrophilicity, drug composition, and environmental responsive degradation. This enables accurate spatio-temporal regulation of the immune system. Despite significant advancements in regenerative medicine facilitated by hydrogel-assisted immune regulation, it’s important to acknowledge their limitations and explore alternative approaches for future investigations. Firstly, chronic wounds are closely associated with biofilms, and hydrogels, as degradable biomaterials, have limited anti-infective properties due to susceptibility to degradation and compromised integrity. Incorporating metal ions such as Zn2+, Fe3+, and Ag+ can significantly enhance antibacterial efficacy. The charged matrix components can be bonded to the cell wall of bacteria through electrostatic adhesion to play a bactericidal role. The internal structure of hydrogels with high cross-linking density is more stable, which is conducive to isolating the invasion of external bacteria. Additionally, evaluating hydrogel immunogenicity is crucial, as foreign body reactions leading to the formation of fibrous capsule that impedes effective release and distribution of encapsulated payload, thus impacting therapeutic efficacy. Finally, biosafety, adequate experimental validation, and preclinical evaluation to minimize the undesired response induced by hydrogels are the most basic requirements for achieving clinical transformation.
Recent advances in cellular metabolism have highlighted the importance of understanding the regulation of immune cell metabolism, particularly regarding macrophages’ ability to adapt to changes in their microenvironment. This includes the polarization of M1 macrophages toward M2 macrophages, accompanied by metabolic reprograming from oxidative phosphorylation, and fatty acid oxidation to glycolysis and fatty acid synthesis. However, there is still a lack of research on the correlation between hydrogel biomaterials and immune cell metabolism. In the field of immunomodulatory hydrogels, several challenges remain to be addressed. These include elucidating the properties of hydrogels that govern immune regulation, defining and quantifying the physical and chemical signals perceived by cells, and understanding the regulatory mechanisms between hydrogels and immune cells. These challenges present new opportunities for the development of immunomodulatory materials, and a comprehensive understanding of these properties is expected to reveal crucial immune cell behaviors and enable the design of advanced guiding hydrogel materials for the treatment of diabetic wounds.
Footnotes
Author contributions
Writing—original draft preparation, D.S. (Dan Sun); writing—review and editing, Q.C. (Qiang Chang); Funding acquisition, F.L. (Feng Lu) and Q.C. (Qiang Chang).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the fund program of National Natural Science Foundation of China (Grant Nos. 81772101, 82072196, and 82002066) and the Science Fund for Distinguished Young Scholars of Southern Medical University (Grant No. 2020J009).
