Abstract
Extracellular vesicles (EVs) have emerged as a promising strategy for treating a wide spectrum of pathologies, as they can deliver their cargo to recipient cells and regulate the signaling pathway of these cells to modulate their fate. Despite the great potential of EVs in clinical applications, their low yield and the challenges of cargo loading remain significant obstacles, hindering their transition from experimental research to clinical practice. Therefore, promoting EV release and enhancing EV cargo-loading are promising fields with substantial research potential and broad application prospects. In this review, we summarize the clinical applications of EVs, the methods and technologies for their large-scale production, engineering, and modification, as well as the challenges that must be addressed during their development. We also discuss the future perspectives of this exciting field of research to facilitate its transformation from bench to clinical reality.
Introduction
Advances in nanotechnology have significantly impacted the biomedical field. 1 Over the past four decades, various nanomaterial-based drug delivery systems have been developed. 2 Traditional nanoparticles, such as carbon nanotubes, graphene oxide (GO), noble metal nanoparticles (NMNPs), and mesoporous silicon nanoparticles (MSNs), are characterized by controllable particle size, diverse morphologies, and ease of large-scale production. However, these traditional nanoparticles suffer from poor biocompatibility and high immunogenicity. 1 Extracellular vesicles (EVs) have emerged as a promising focus in nanotherapy, potentially addressing the limitations of traditional nanotechnology. EVs are nanoscale lipid bilayer membrane capsules secreted by cells, which can be taken up by recipient cells. 3 They play critical roles in cell communication, including signaling and the delivery of biological cargos. 4 EVs also exhibit immune tolerance, circulatory system stability, and the ability to traverse biological barriers, such as the blood-brain barrier, blood-CSF barrier, and blood-tumor barriers.5 –10 Furthermore, EVs derived from different donor cells inherit unique characteristics and functions from their source cells, making them ideal vectors for diagnosing and treating various diseases.11 –13 Consequently, interest in EVs has grown rapidly, as evidenced by the exponential increase in PubMed publications on EVs every year (Figure 1).
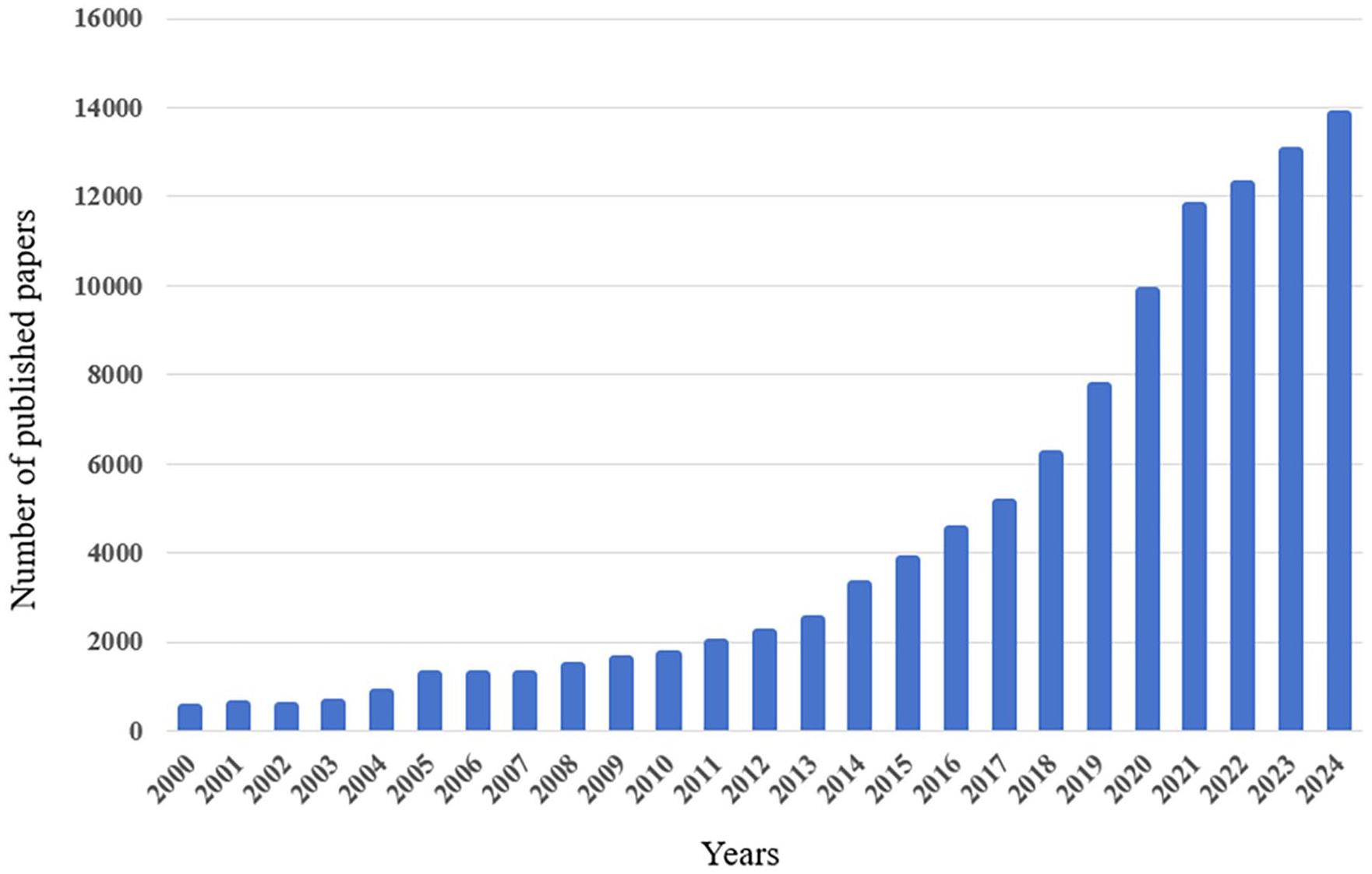
Number of PubMed entries using “EV” OR “exosome” OR “extracellular vesicles” as search terms. Search performed December 31, 2024.
EVs contain a variety of bioactive components, including proteins, 14 lipids, 15 and nucleic acids, 16 which exert biological functions by delivering specific biomolecules. 17 To date, EV-based therapies and vaccines have emerged.18,19 For instance, in July 2023, the first human clinical trial of platelet EVs for wound healing treatment was conducted. 20 EVs were decorated with a recombinant SARS-CoV-2 receptor-binding domain as an inhalable COVID-19 vaccine. Plasma EVs expressing ACE2 increased in patients with COVID-19, and EVs carrying ACE2 on their surfaces can inhibit SARS-CoV-2 infection. 21 The design of artificially engineered small EVs displaying ACE2 variants on their surfaces can protect against SARS-CoV-2 infection. 22 Additionally, EVs can encapsulate nucleic acids, proteins, lipids, and chemical drugs, showing positive effects in various aspects of cancer and metabolic diseases.23 –27 This indicates that the active molecules loaded into EVs play a crucial role in their clinical application, and loading specific cargo into EVs or onto their surfaces is key to achieving their therapeutic effect. It has been reported that this process of loading specific cargo into EVs or modifying their surfaces is also called EV modification or engineering, enabling the delivery of specific molecules to recipient cells. 28 The methods of producing engineered EVs mainly include cell modification to produce specially modified EVs and specific modification after EVs isolation. Although EVs have diverse biological functions, they still face challenges, such as low production, which is one of the key issues limiting their clinical application and a topic we will discuss in this review.
As per previous research, achieving biological outcomes typically requires a dose of 106–1011 EVs per treatment in mice, which may necessitate liters of conditioned media and several months of collection.5,29 –31 In clinical experiments, the inhaled dose of EVs for mice is 105 particles/g, and for a mouse weighing 20–40 g, it requires approximately 2–4 × 106 particles per mouse. 31 For the regulatory effect of injecting EVs through the tail vein to treat liver disease, the injection amount was 109 particles per mouse. 32 Furthermore, mice were treated by intraperitoneal injection of EVs loaded with siRNA at a dose of 109 particles per time, three times a week. 33 Studies have shown that the optimal therapeutic dose of EV aerosol inhalation is 10⁵ particles/g in a mouse lung injury model. Further clinical trials have been conducted in healthy human volunteers using doses ranging from 2 to 16 × 10⁸. 31 It is evident that different clinical treatments require a large amount of EVs, and low yield is a major drawback of EV production at present. Therefore, increasing EV yield is essential. Recently, many methods for promoting EVs secretion and loading foreign cargo into EVs have been reported, such as the development of large-scale cell culture platforms, 34 cell stimulation to increase EV yield per cell, 35 and the production of EV-like nanovesicles. 36 These large-scale production methods bring hope for the clinical application of EVs.
In this review, we summarize the clinical application prospects of EVs, methods to enhance EV production, and the advancements in engineering EVs to create functional EVs. Additionally, we discuss the challenges of transitioning EVs from the laboratory to clinical application. This review offers new perspectives and insights into EVs-based nanotherapy and diagnosis.
Clinical applications of EVs
The understanding of the role of EVs in disease biology is still evolving, with a substantial increase in studies exploring their potential utility in diagnosing and treating various pathologies. 37 EVs carry distinct cargoes under different pathological conditions, enabling disease detection and monitoring through changes in specific cargoes. The unique properties of EVs in delivering functional cargo to cells also make them promising therapeutic vehicles for disease treatment and prevention. EVs are transitioning from academic research to biotechnology and advancing toward clinical applications. In this section, we will discuss the versatility of EVs in translational medicine, including their applications in disease treatment, prevention, and diagnosis (Figure 2).
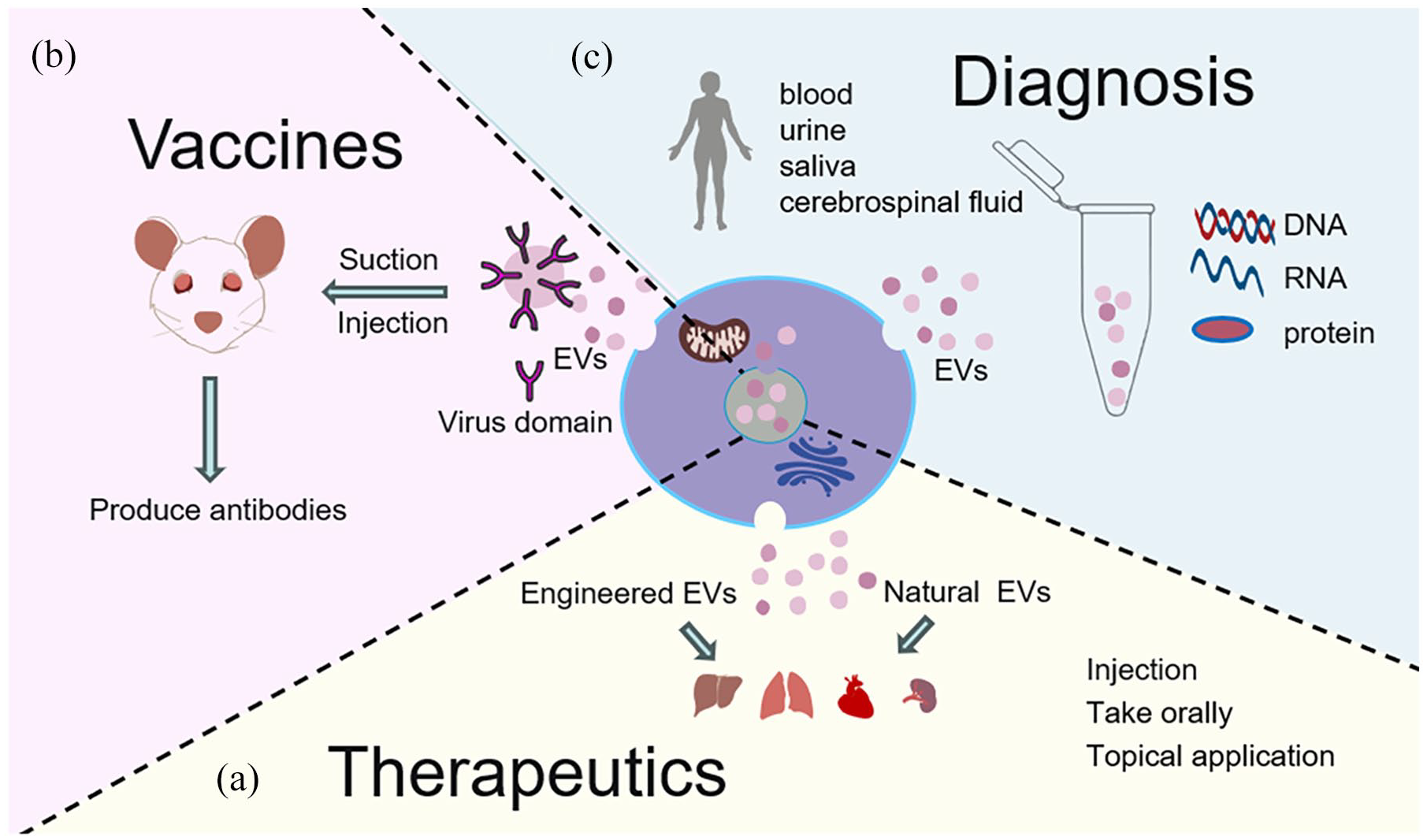
Application of EVs in disease diagnosis, prevention, and treatment: (a) natural or engineered EVs, administered orally, via injection, inhalation, or topical application, can impact multiple organs, including the liver, intestines, skin, etc., (b) injection or inhalation of EVs carrying viral antigens can induce an immune response in animals, and (c) EVs isolated from body fluids can detect changes in nucleic acid, proteins, and other components and can be utilized for liquid biopsy in diseases.
Therapeutic potential of EVs
EVs exhibit significant potential as drug delivery vehicles due to several key features that enhance their effectiveness. Firstly, their excellent biocompatibility, combined with their inherent ability to transport materials, ensures they can effectively carry therapeutic agents. Additionally, EVs possess low immunogenicity, minimizing adverse immune responses and making them safer for clinical applications. Their intrinsic long-term circulatory capability allows for sustained drug release, ensuring therapeutic effects are maintained over time. Moreover, the good biomembrane penetration capacity of EVs facilitates the delivery of drugs directly to target cells, further enhancing therapeutic efficacy. Collectively, these attributes position EVs as promising candidates in the field of drug delivery systems.38 –40 They are rich in bioactive substances and have demonstrated therapeutic potential in various applications. 41 Numerous studies have highlighted the significant roles of EVs in treating various diseases, including cancer, 42 nerve diseases, 43 tissue injuries, 44 and pathogenic infections. 45 Several EV studies have entered clinical trials, such as EVs in fibrin gel for cartilage repair (NCT06713902), nasal drop therapy with EVs for amyotrophic lateral sclerosis (NCT06598202), and the safety and efficacy of stem cell EVs in patients with retinitis pigmentosa (NCT06242379). Specifically, a large body of research has shown that cell-derived EVs,46 –48 EVs present in body fluids,49,50 bacterial outer membrane vesicles (OMVs),51 –53 and plant-derived EV-like nanoparticles (PELNs)54 –56 all hold significant potential in disease treatment. Through these advancements, EVs are emerging as a revolutionary approach to drug delivery, warranting further exploration and development in therapeutic applications.
For instance, EVs derived from stem cells can promote cell regeneration or repair57,58 and serve as substitutes for stem cells to avoid immune rejection and carcinogenic risks associated with stem cell therapy, offering significant application value in regenerative medicine. Mesenchymal stem cells (MSCs) are known for their excellent properties in cell therapy. However, the risk of immune rejection linked to cell transplant therapy limits their use. 59 MSC derived EVs function as effective therapeutic vehicles and appear to replicate the broad therapeutic effects observed in MSCs themselves,60,61 giving rise to the novel concept of “cell-free” stem cell therapy. 62 Additionally, MSC-derived EVs have shown potential therapeutic application in SARS-CoV-2 pneumonia,63,64 while M2 macrophage-derived EVs have been found to promote diabetic fracture healing by acting as immunomodulators. 65 In a study on osteoarthritis, EVs from human bone marrow MSCs (hBM-MSCs) were shown to promote extracorporeal cartilage regeneration by stimulating chondrocytes to produce proteoglycans and type II collagen. 66 Similarly, EVs from human umbilical cord MSC (huc-MSCs) ameliorate bone loss in senile osteoporotic mice. 67 Furthermore, adipose-derived mesenchymal stem cells (ADSCs)-derived EVs have demonstrated the potential to alleviate neuronal damage, promote neurogenesis, and rescue memory loss in mice with Alzheimer’s disease. 68 In addition to stem cells, various immune cells-derived EVs have been studied and characterized for their anti-tumor or tumor-promoting functions. 69 Studies have shown that immune cell-derived EVs possess immune activity. 70 EVs derived from CD4+T cells increase the antitumor response of CD8+T cells by enhancing their proliferation and activity without affecting regulatory T cells (Tregs). Moreover, EVs derived from interleukin-2 (IL2)-stimulated CD4+T cells induce a stronger antitumor response of CD8+T cells compared with that of IL2-unstimulated CD4+T cell-derived EVs. 71 Natural killer (NK) cell-derived EVs exert cytotoxic effects on melanoma cells and can inhibit tumor cells by delivering tumor suppressor microRNA (miR-186).72,73 Furthermore, NK cell-derived EVs carrying miR-1249-3p reduce insulin resistance and inflammation in mouse models of type 2 diabetes. 74
Vesicles produced by gram-negative bacteria are referred to as OMVs.75 –78 OMVs, with a structure similar to cell-derived EVs, are natural functional nanomaterials. 51 Additionally, various bacterial-derived OMVs have been reported to exhibit anti-cancer and inflammatory regulatory effects.79 –82 Research has demonstrated that OMVs possess a significant ability to effectively induce antitumor immune responses and completely eradicate established tumors without notable adverse effects. This antitumor response of OMVs is durable, with secondary and tertiary tumor re-challenges being fully rejected by mice cured from the primary challenge. Furthermore, systematically administered OMVs target and accumulate in tumor tissue, subsequently induce the production of antitumor cytokines CXCL10 and interferon (IFN)-γ. 83 Similar studies have demonstrated that E. coli OMVs lead to complete remission in two mouse syngeneic tumor models. Mechanistic investigations revealed that E. coli OMV treatment increases the infiltration and activation of CD8+T cells, particularly cancer antigen-specific CD8+T cells with high expression of TCF-1 and PD-1. 84 Additionally, Lactobacillus amylovorus derived OMVs significantly alleviate aflatoxin B1-induced intestinal injury by inhibiting the production of inflammatory factors, reducing intestinal permeability, and increasing the expression of tight junction proteins. 85 Therefore, OMVs hold great potential for application prospects in disease treatment, particularly in the field of tumor therapy.
Studies on vesicles from natural sources, including edible plants, are gaining momentum due to their biological implications. 86 Plant-derived vesicles, referred to as EV-like nanoparticles (ELNs), 87 have emerged as a novel and promising area of research in nanomedicine. These vesicles are structurally and functionally similar to exosomes found in mammalian systems but offer unique advantages, such as easy availability and high yield due to their plant origins.88,89 In recent years, EVs have been isolated from various plant species and their different parts (e.g. fruits, roots, seeds, leaves) using standardized isolation and purification methods, such as ultracentrifugation.56,90 –92 ELNs can be engineered to encapsulate various therapeutic agents, including anti-cancer drugs, antibiotics, and RNA molecules. Their inherent stability and biocompatibility make them suitable carriers for targeted drug delivery, potentially reducing side effects and enhancing therapeutic efficacy.93,94 For instance, ELNs derived from mulberry bark could prevent DSS-induced colitis through the AhR/COPS8 pathway. 95 Intravenous injection of Momordica charantia L.-derived ELNs has been shown to mitigate doxorubicin cardiotoxicity, improving cardiac function and myocardial structure. 56 Additionally, modifying cRGD-targeted doxorubicin (DOX) nanoparticles (DN) onto the surface of orange-derived ELNs significantly enhances tumor accumulation and penetration, efficiently inhibiting ovarian cancer growth. 96
EVs are present in almost all body fluids, including blood, milk, urine, and others.97 –101 Among these, milk EVs, in addition to the advantages of traditional EVs such as low immunogenicity and high biocompatibility, also exhibit high yield and partial resistance to digestion.102 –105 Based on these characteristics, we believe milk EVs are highly suitable for disease treatment. Studies have shown that the abundant proteins and miRNAs in milk EVs play a role in regulating immune and inflammatory pathways. Oral administration of milk EVs has been shown to prevent colon shortening, reduce intestinal epithelium disruption, and inhibit inflammatory cell infiltration and tissue fibrosis in a mouse model of ulcerative colitis. Mechanistically, milk EVs attenuate inflammatory responses by inhibiting the TLR4-NF-κB signaling pathway and NLRP3 inflammasome activation.106 –108 Additionally, milk EVs miR-148a protects against necrotizing enterocolitis by regulating p53 and sirtuin 1 .109,110 Beyond natural milk EVs, engineered milk EVs have also demonstrated significant therapeutic potential. For instance, miR-31-5p mimics were successfully encapsulated into milk EVs via electroporation, enhancing cellular uptake of miR-31-5p and providing resistance to degradation. These engineered EVs carrying miR-31-5p promoted angiogenesis and accelerated diabetic wound healing. 111 Similarly, studies have shown that encapsulating TNF-α siRNA into milk EVs via electroporation and orally administering these engineered EVs reduced colonic TNF-α expression and intestinal inflammation in mice. 112 Additionally, previous studies from our research group have demonstrated the potential therapeutic effects of oral milk EVs, including altering the miRNA expression profile in the serum of piglets, 113 promoting the level of intestinal secreted immunoglobulin A (sIgA) in mice, increasing the expression of intestinal polymeric immunoglobulin receptor (pIgR) in IPEC-J2 cells to regulate intestinal immune function, 114 and alleviating DON- and LPS-induced intestinal injury in piglets through oral administration of milk EVs in vivo.115,116 Furthermore, miRNA-loaded milk EVs have shown antiviral effects in vivo and in intestinal organoids, 45 while pituitary EVs have demonstrated potential in alleviating liver damage caused by carbon tetrachloride through a novel endocrine regulatory system.117,118 These studies highlight the versatility of EVs, with EVs from various sources exhibiting therapeutic effects on different diseases, underscoring their significant potential for clinical application.
In addition to natural EVs, specifically modified EVs, such as those with protein modifications, nucleic acid alterations,111,119 and chemical drug loading, 120 have also demonstrated therapeutic potential. For example, inhalable cell-derived EVs used as paclitaxel carriers for lung cancer treatment have shown enhanced targeted antitumor effects while reducing adverse effects on the liver, kidney, and intestine. 121 Furthermore, injecting EVs loaded with siRNA has demonstrated the ability to cross the blood-brain barrier and knock down specific mRNA in the brains of mice, with increased knockdown efficiency.122,123 Topical application of EVs containing sodium alginate precursors or miR-21 has been found to accelerate skin wound healing.124,125 Additionally, the immune response and targeting can be modulated by designing genetically engineered EVs with surface-displayed antibody targeting groups and immunomodulatory proteins.126,127
EVs are actively being explored as therapeutic agents, with both natural and engineered EVs demonstrating therapeutic potential across various diseases. Furthermore, due to their low immunogenicity and ability to cross barriers,8,128 EVs hold significant promise for development and application in the field of disease therapy. However, the large-scale production of EVs remains a critical challenge for their clinical use, which will be discussed later.
Vaccines
Nanoparticle vaccine formulations have emerged as an innovative antigen delivery system capable of activating both local and systemic cell-mediated and antibody responses, while ensuring the gradual release of immunogens. 129 EVs represent a new and essential source of immune response antigens and molecules that can be utilized for animal vaccination.57,130,131 EVs can be engineered to display viral antigens, inducing strong and specific CD8(+) T cell and B cell reactions. 132 For instance, EVs were decorated with a recombinant SARS-CoV-2 receptor-binding domain as an inhalable COVID-19 vaccine, which, after lyophilization, can be stably stored at room temperature for over 3 months. 18 EVs loaded with mRNAs encoding immunogenic forms of the SARS-CoV-2 Spike and Nucleocapsid proteins elicited long-lasting cellular and humoral responses to both. 133 Furthermore, EVs were genetically engineered to express either the SARS-CoV-2 delta spike or the more conserved nucleocapsid protein on their surface. In two different animal models, including rabbits and mice, these engineered EVs were injected to produce potent antibodies and a robust T-cell response with nanogram quantities of proteins without any adjuvant. 134 These studies indicate that EVs modified through the engineering of viral domains can effectively generate immune responses, offering a promising new direction for the development of novel genetically engineered vaccines.
EVs derived from cancer cells are particularly appealing for cancer immunotherapy as they can present tumor-specific antigens to the immune system, potentially triggering robust anti-tumor immune responses.135 –137 They hold promise as a cancer vaccine. Several studies have emphasized the potential of EVs as cancer vaccines in preclinical models and early-phase clinical trials. EVs loaded with tumor antigens have been demonstrated to elicit strong anti-tumor immunity in mouse models. For instance, EVs derived from melanoma cells carrying antigens successfully activated CTLs and inhibited tumor growth. 138 A phase I clinical trial using dendritic cell-derived EVs loaded with tumor antigens in patients with advanced melanoma demonstrated safety and feasibility, with some patients showing immune responses. 139 These findings highlight the significant potential of EVs for development and application, whether as a viral vaccine or a cancer vaccine.
Diagnostic potential of EVs
Currently, circulating tumor DNA (ctDNA), circulating tumor cells (CTCs), and EVs represent the three main branches of liquid biopsy. CtDNA and CTCs have been approved for clinical use by the U.S. Food and Drug Administration (FDA). 140 Compared to ctDNA and CTCs, EVs exhibit greater advantages and unique characteristics in liquid biopsy. First, the abundance of EVs in biofluids makes them relatively easy to obtain. Second, EVs are secreted by living cells and carry a variety of biological information from their parental cells. Third, EVs are inherently stable due to their lipid bilayer structure, allowing them to circulate under physiological conditions, even within the harsh tumor microenvironment. This high biological stability also enables the long-term storage of specimens for EV isolation and detection. 141 We conducted a PubMed search using the keywords “Extracellular Vesicle” OR “exosome” and “Plasma” OR “Serum” OR “Urinary” OR “Milk” OR “Sputum” OR “Semen.” Several representative studies on the role of EVs in disease diagnosis were identified and are listed (Table 1).
Research on EVs in disease diagnosis.
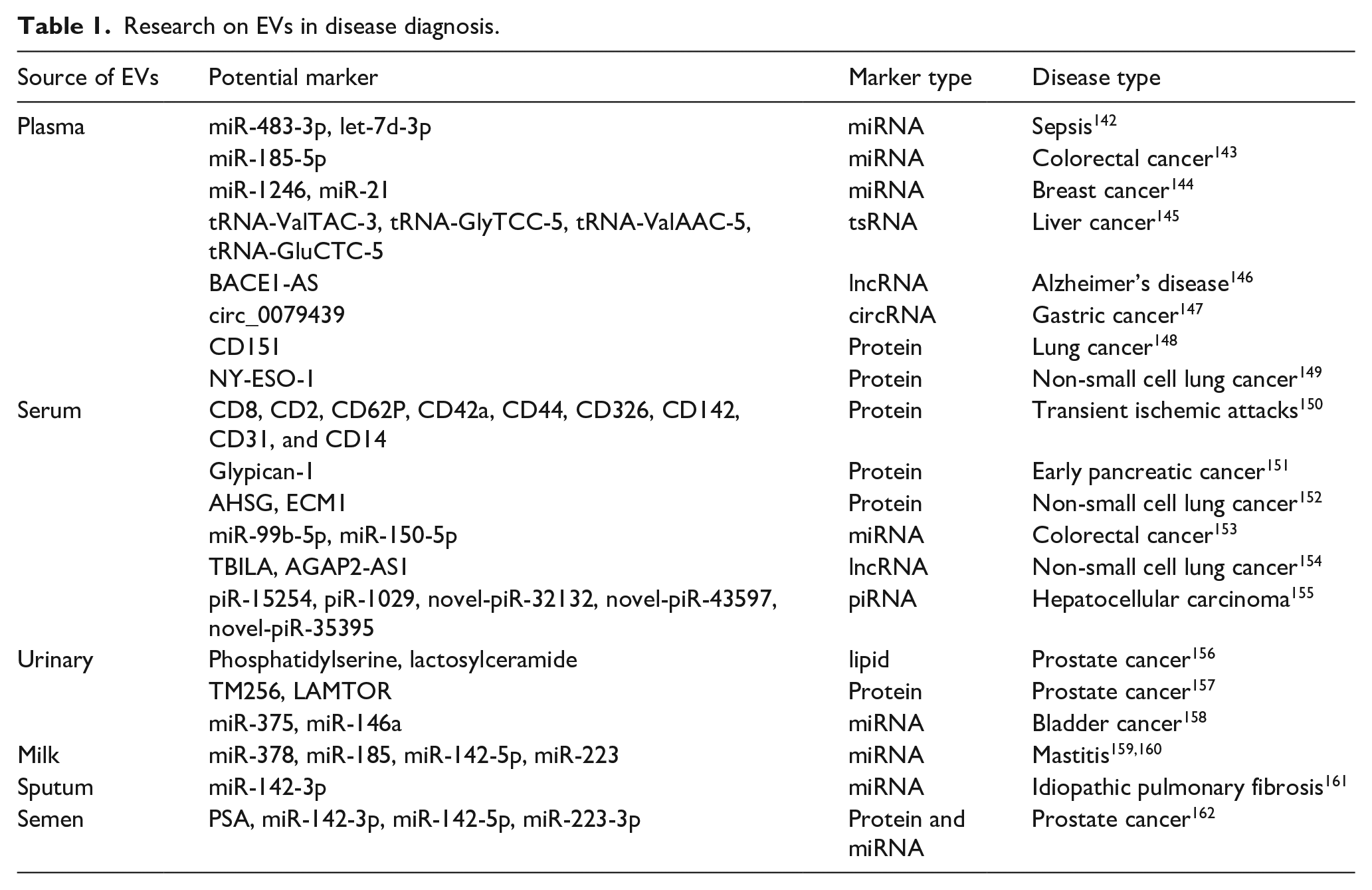
One potential clinical application of EVs involves their use as diagnostic and prognostic biomarkers.163,164 EVs can carry biomolecules from their original cells, which may contain indicators of pathophysiological conditions, and can be detected in various body fluids such as urine, 165 blood, 166 saliva, 167 and cerebrospinal fluid, 168 serving as a liquid biopsy. 169 They have demonstrated effectiveness in diagnosing and treating cancer,170,171 cardiovascular disease, 172 tuberculosis, 173 and central nervous system diseases. 174 EVs are structurally stable and rich in content, containing numerous specific cytokines, functional mRNA, growth factors, lipids, and other substances involved in intercellular material transport and information exchange. They are closely associated with the onset and progression of various diseases. Changes in EV composition, including specific proteins, nucleic acids, signaling molecules, and lipids, may reflect the state of parental cells.175 –177 Markers detected using EVs mainly include changes in their protein and nucleic acid molecules. Research indicates that circulating EV mRNA signatures can assist in the early diagnosis of clear cell renal cell carcinoma and the detection of prostate cancer.178,179 EV-encapsulated miRNA can serve as a biomarker for breast cancer diagnosis.144,180 The serum GPC1 content of EVs in early pancreatic cancer patients was significantly higher than in healthy individuals. GPC1(+) circulating EVs were identified in the serum of pancreatic cancer patients with absolute specificity and sensitivity, distinguishing healthy individuals and patients with benign pancreatic disease from those with early- and late-stage pancreatic cancer. 151
In recent years, numerous companies have invested in research related to EV diagnosis, including EV Diagnostics, EV Plus, and Exosomics. In May 2023, EV Diagnostics, a molecular diagnostics company, announced interim results from a prospective randomized study involving over 1000 patients to evaluate the clinical utility of its ExoDx™ prostate test over a 5-year period. 181 The emergence of innovative technologies is advancing the application of EVs in clinical diagnosis, such as flow cytometry, confocal and non-confocal (fluorescence) microscopy, scanning electron microscopy (SEM), atomic force microscopy (AFM), Raman spectroscopy, surface plasmon resonance (SPR), mass spectrometry (MS), and micro nuclear magnetic resonance (μNMR). 182 As a novel in vitro diagnostic technology, EVs hold significant potential and value for research and development in the screening and diagnosis of tumors and other chronic diseases.
Large-scale production of EVs
While EVs have shown therapeutic potential in various diseases,183 –185 their natural production is limited, and generating a substantial quantity of EVs within a short timeframe remains a challenge. 186 This limitation results in high production costs, which currently impede the widespread production and application of EVs. Consequently, it is crucial to explore new methods to enhance EV production and reduce associated costs. Recent studies have proposed various strategies to improve EV production, including the development of large-scale cell culture platforms, 34 increasing the quantity of EVs secreted by individual cells through conditional stimulation or genetic modification,35,187 and producing EVs with nanovesicle-like structures by promoting cell disruption.188,189 We have summarized these methods for the large-scale production of EVs and EV mimetics (Figure 3).
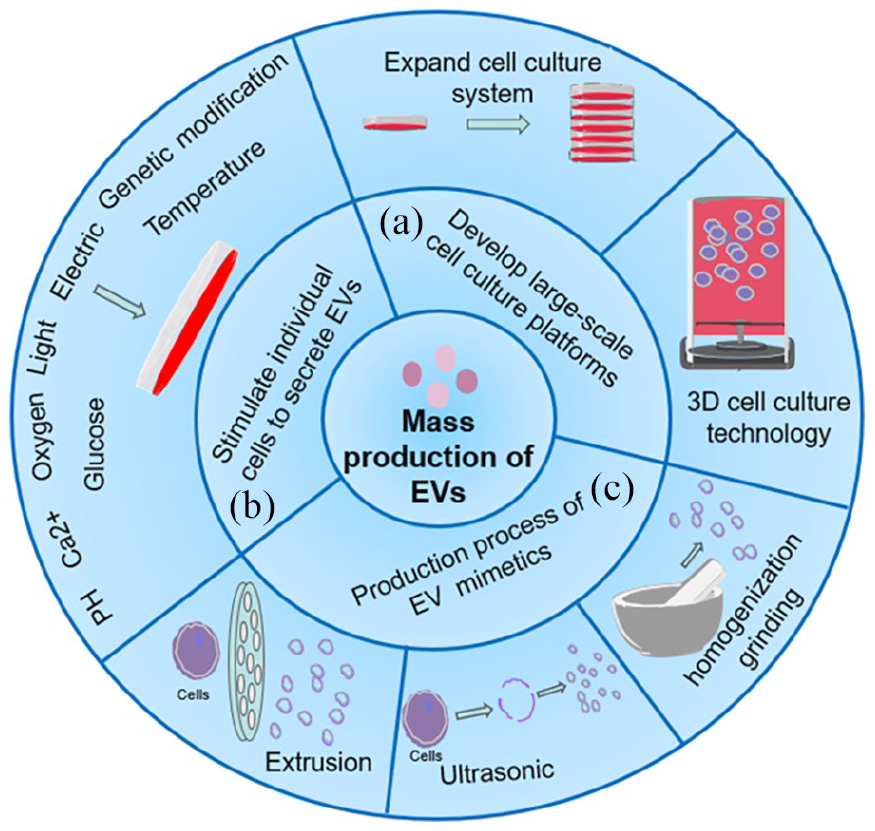
Mass production of EVs and EV mimetics: (a) expanding cell culture to collect more EVs, (b) adjusting pH, calcium ion concentration, glucose content, oxygen levels, and other culture conditions, as well as applying physical stimuli such as increased temperature, light exposure, or electric shock, can effectively enhance EV secretion, and (c) using physical forces (such as ultrasound, grinding, or extrusion) to disrupt the cell membrane, followed by the reformation of a large number of vesicles with structures similar to EVs.
Large-scale cell culture platforms
The traditional EV production protocols rely on two-dimensional (2D) flask cultivation, which requires substantial amounts of cell culture plastics, media, and space. In contrast, three-dimensional (3D) cell culture employs gels, microcarriers, hollow fiber bioreactors, or synthetic scaffolds to enable cell growth from a 2D plane to a 3D state.190 –193 Research has demonstrated that the yield of MSCs EVs from 3D cultures is over 20 times higher than that from 2D cultures. 194 Moreover, applying flow stimulation to dental pulp or adipose-derived stem cells on 3D constructs, or subjecting them to mechanical stretching within 3D-engineered skeletal muscle tissues, has been shown to significantly enhance EV production compared to static conditions. 195 Additionally, 3D bioreactors offer an alternative approach with the benefit of increased scalability, with systems currently ranging from less than 1 L to over 80 L in capacity. 196 A microcarrier-based bioreactor culture system, specifically a scalable S/XF microcarrier bioreactor culture system, has been developed for the efficient production of MSC-EVs using MSCs from three different human tissue sources. This system achieves higher concentrations and productivity of MSC-EVs compared to conventional static culture systems. 197 Furthermore, the use of the CELLine cell culture system increases EV production by tumor cells. 198 It is important to note that this method primarily focuses on increasing EV yield by expanding the cell culture volume, and does not appear to affect the amount of EVs secreted by individual cell. In addition to the availability of larger quantities of EVs, EV function also changes under varying culture conditions. The study demonstrated that EVs derived from 3D cultures of umbilical cord mesenchymal stem cells in a hollow-fiber bioreactor exhibit enhanced osteochondral regeneration activity. 199 Similarly, 3D culture of MSCs generates exosomes with higher yields and improved therapeutic efficacy for cisplatin-induced acute kidney injury. 200 Thus, we hypothesize that different culture conditions influence not only EV production but also EV function, potentially by altering EV components.
Cell stimulation or genetic modification to increase EV yield per cell
Several factors can promote EV release, including adjustments to the culture environment, physical stimulation, and molecular interference. 201 Studies have demonstrated that hydrogen ion concentration202,203, calcium ion concentration, 204 glucose concentration, 205 and oxygen content206,207 can influence EV secretion. Notably, an acidic pH (pH = 4) has been reported to enhance the stability of EVs in vitro, thereby improving their separation and collection rates. In an acidic environment, the concentration of EV proteins and RNA collected has been shown to increase fivefold. 208 The transport and fusion of multivesicular bodies (MVBs) with the plasma membrane are regulated by calcium ions. 209 Increased intracellular Ca2+ levels promote EV secretion 204 and affect the expression of proteins associated with EV release, such as members of the Rab protein family. 210 Additionally, glucose concentration can influence EV secretion, as both insufficient and excessive glucose levels can promote EV release. For instance, under low-glucose conditions, EV secretion from H9C2 cardiomyocytes was increased. 205 Similarly, studies have shown that the amount of EVs secreted by trophoblast cells increases in high-glucose environments. 211 Oxygen content is another factor affecting EV secretion. Under hypoxic conditions, the expression level of Rab27, a key regulatory factor involved in EV secretion, increases, leading to enhanced EV release. 212
Furthermore, physical stimuli such as temperature, light, and electric shock can also enhance the production of EVs. Temperature, a critical factor in maintaining the normal life activities of all organisms, also regulates cell EV secretion. 213 Changes in temperature have been shown to affect the release of EVs by breast cancer cells, with twice as many secreted EVs at 39°C compared to 35°C. 213 Light has also been found to impact EV secretion, with studies demonstrating that immune function EV secretion can be increased by over 13 times when dendritic cells are stimulated with light at 365 nm, while maintaining overall quality, biocompatibility, cell internalization ability, and immunogenicity. This method has shown similar effects on other cell types. 35 Additionally, low-energy electric fields have been observed to activate intracellular signals, including Rho-GTPase and endocytosis, both involved in EV formation mechanisms. 214 Studies indicate that low-level electricity can enhance EV secretion by activating Rho-GTPase, without altering EV morphology or surface-labeled proteins, and without inducing cytotoxicity. 215 In addition to physical stimulation, regulating the expression of proteins related to EV genesis can also affect EV secretion. For instance, regulating the expression of key EV genesis proteins such as Rab (Rab27, Rab37, Rab39) and Phospholipase D (PLD) can influence EV secretion.187,216 –218 Activation of H1R1 in HeLa cells increases Ser110 phosphorylation of SNAP23, promoting MVB-plasma membrane fusion and the release of CD63-enriched EVs. 219 KIBRA promotes EV secretion by inhibiting the proteasomal degradation of Rab27a. 220 Additionally, inhibition of Sirtuin 1 expression has been shown to increase the number of exosomes secreted by cells. 221 PLD has been shown to participate in the biogenesis of EVs, with EV biogenesis and budding into MVBs controlled by ARF6 and PLD2. 222 Our study also demonstrated that inhibiting Rab27 protein expression significantly reduces EV secretion. 223
Above research shows that moderate pH reduction, adjustment of calcium ion concentration, glucose content, oxygen content, and other culture conditions, along with the exogenous application of physical stimuli such as temperature, light, and electric shock, can effectively enhance the formation and secretion of EVs, serving as a viable approach to increase EV production. Our previous study demonstrated that GHRH stimulation promotes the secretion of pituitary extracellular vesicles and influences the content of its cargo miR-375-3p, thereby regulating the proliferation and IGF-1 expression of hepatocytes. 223 However, whether these aforementioned stimuli affect the cargo, function, and stability of EV remains to be further studied.
Broken cell membrane assembles into vesicle particles
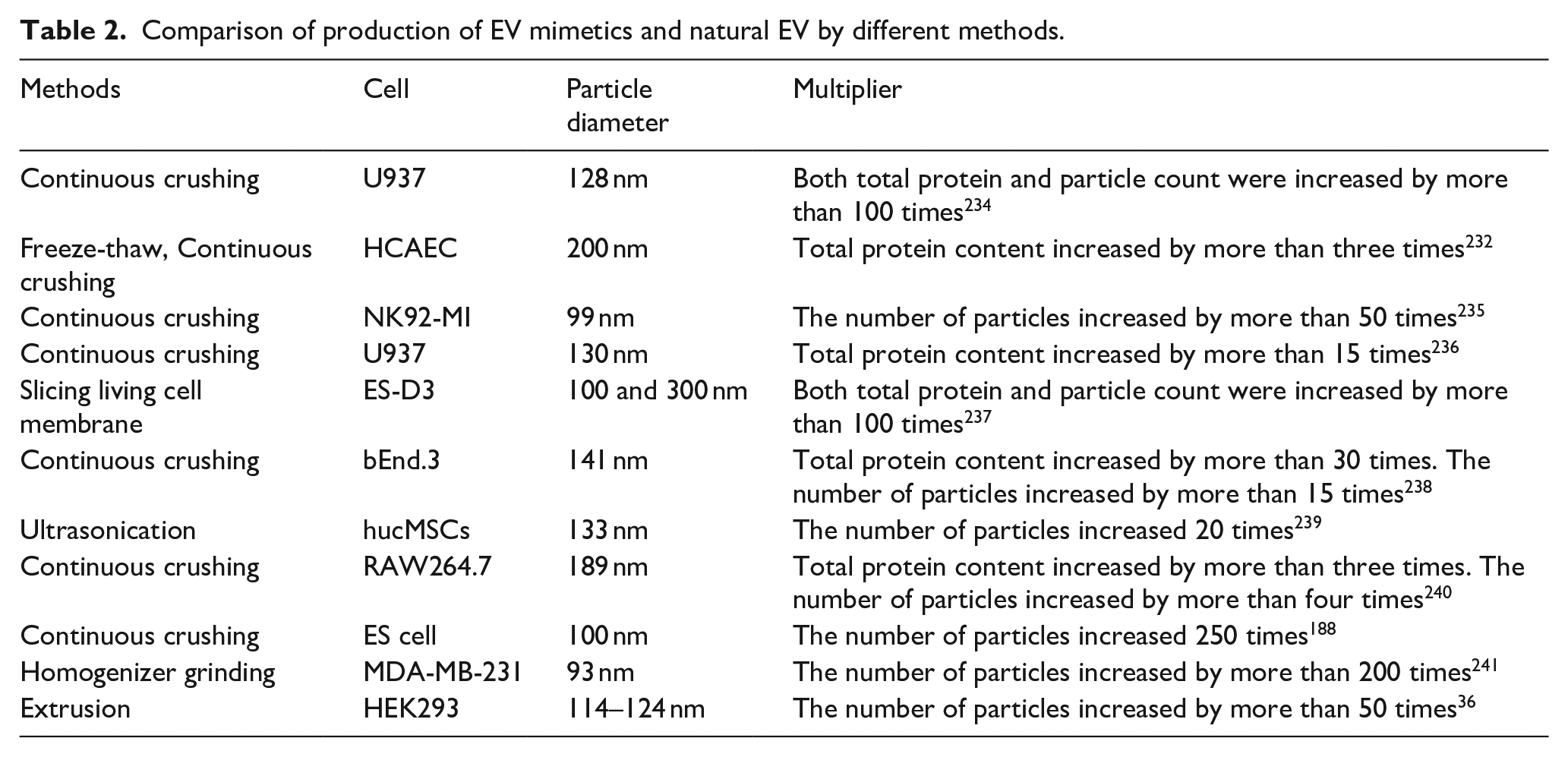
While populations of EVs can be isolated from EV-secreting cell lines,1,224 the low yield of EVs and the complexity of their purification have driven an increase in studies using EV mimetics. Recently, artificial EVs have been developed to address the limitations of natural EVs, emerging as new theragnostic biomaterials with potential clinical applications.225,226 Various methods have been identified to disrupt cell membranes and reassemble them into vesicle particles, including extrusion through micro-sized pore filters,36,188,227 ultrasonic techniques, 228 nitrogen cavitation, 229 and homogenization grinding. 189 According to the “2023 MISEV guidelines,” EV-like particles produced through direct artificial manipulation are termed EV mimetics. 230 Among these, cell fragmentation by shear force is the most widely used method for producing EV mimetics. Extruded cells can yield EVs that closely resemble naturally secreted EVs in shape and content, with a productivity rate 1000 times higher than that of natural cells. This is achieved by sequentially passing cells through nanofilters with progressively smaller pore sizes, such as 10–5–0.22 μm filters successively. 231 Additionally, it has been demonstrated that the amount of EVs produced using 10–1–0.2 μm filtration membranes is three times greater than that of naturally secreted EVs. 232 Vesicles can also be produced by mechanically grinding cells, for example, using homogenizers and filters of 0.8 and 0.22 μm, which produce vesicles similar in morphology and structure to EVs. 189 Similarly, vesicular structures can be formed through membrane fusion induced by ultrasound following hypotonic and mechanical cell disruption. 228 EV mimetics exhibit a vesicular structure comparable to EVs and contain CD63 and TSG101 proteins. 233 Currently, numerous methods for preparing EV mimetics have been published, all achieving higher production rates compared to natural EVs. Using the keywords “Exosome mimetic,” “Engineered Extracellular Vesicles,” “Cell-derived nanovesicles,” “exosome-like vesicles,” or “Engineering exosomes,” we conducted a PubMed search and screened several representative studies on EV mimetic preparation (Table 2).
Comparison of production of EV mimetics and natural EV by different methods.
EVs by themselves or as vehicles for the delivery of drug payloads are being actively explored as therapeutic agents. Natural EVs from various sources perform diverse biological functions but exhibit batch effects and functional inconsistencies.242 –244 The core component of EVs is derived from the cell membrane. These methods utilize physical forces to disrupt the cell membrane, and the disrupted cell membrane then reforms into small vesicles containing proteins or drugs. Consequently, engineered EVs may offer improved consistency to ensure batch-to-batch reproducibility of their therapeutic efficacy, presenting greater potential for production and application. This physical method of producing EVs addresses two major limitations of natural EV production, including low yields and inconsistency in production. Loading specific proteins or nucleic acid drugs and while leveraging the rapid mass production of EV mimetics is another promising approach to complement natural EVs.
Production of engineered EVs
The composition of EVs, including proteins, lipids, non-coding RNA (ncRNA), and other components, can vary depending on the tissue source or physiological state. In recent years, there has been growing interest in engineering EVs or modifying them to specifically alter their cargo. When protein or nucleic acid drugs are introduced directly into the body, they are susceptible to elimination by the immune response. However, the vesicle structure of EVs can protect the drug molecules from the internal environment. By loading specific nucleic acids or proteins for different purposes, the components of EVs can be altered to perform specific biological functions.22,245 Additionally, the engineering modification of EVs can enhance targeting efficiency and specificity.246 –248 For example, EVs conjugated with anti-HER2 antibodies have demonstrated selective delivery to HER2-positive breast cancer cells. 249 Similarly, EVs derived from cells engineered to express Lamp2b fused with an RVG peptide (targeting the acetylcholine receptor) have been shown to deliver siRNA specifically to neurons. 122 EV engineering has been extensively researched in fields such as targeted tumor therapy,250,251 bone injury repair, 252 gene editing,28,253 and other areas. Its development will lead to the creation of new technologies and products for human and animal health. Methods for engineering EV modification can be categorized into two groups: endogenous modification and exogenous modification (Figure 4).
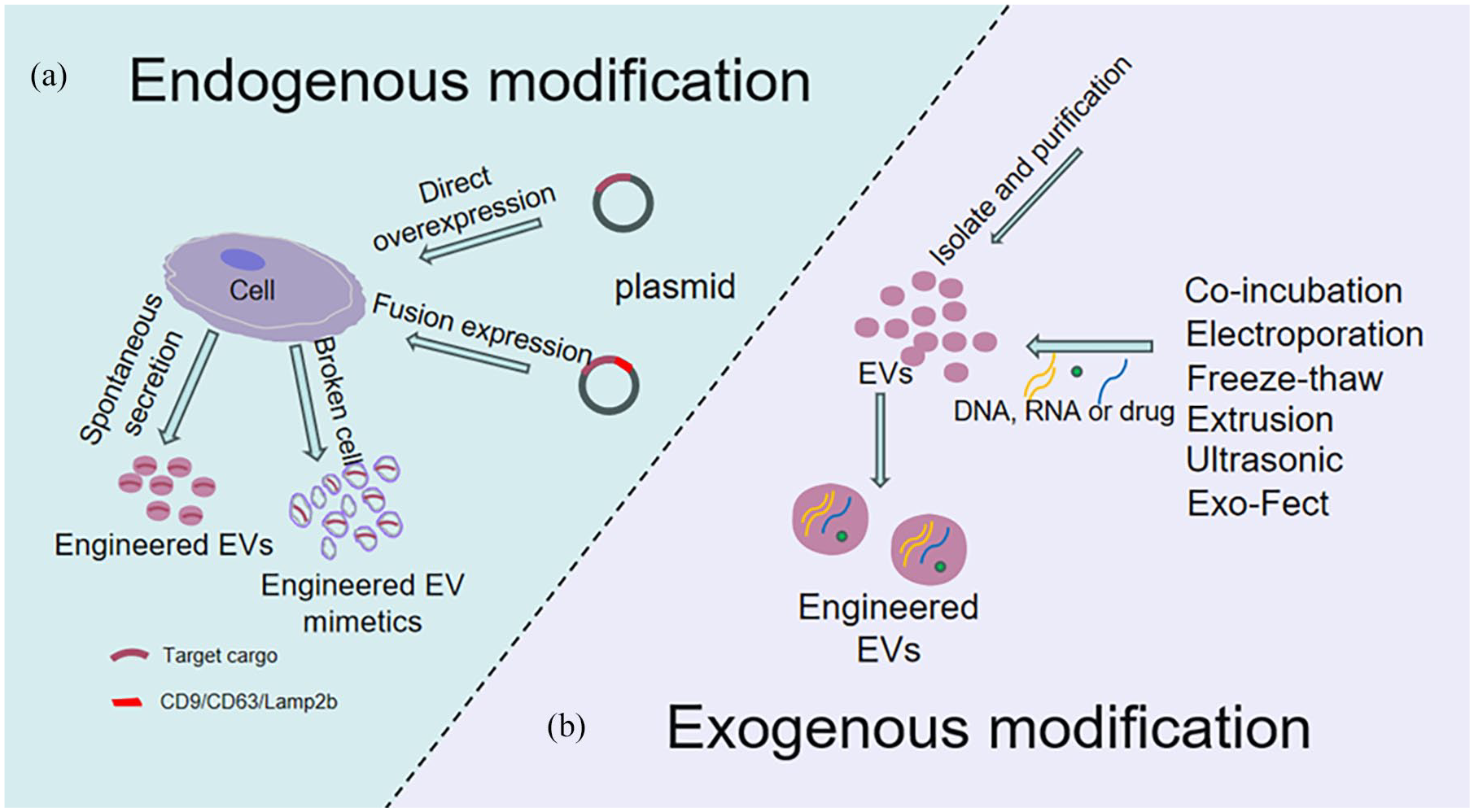
Two modification methods of engineered EVs: (a) genetically modifying the donor cells to achieve EV modification. This can be done by directly overexpressing or fusing the target protein with EV proteins (such as CD9, CD63, Lamp2b) to efficiently load the target protein into EVs. Another approach is to induce cell disruption after overexpression to generate EV mimetics that carry the target molecule and (b) after isolating EVs, exogenous cargo can be loaded into EVs using techniques such as electroporation, co-incubation, ultrasound, or transfection reagents.
Indirect engineering of EVs by modifying EV-producing cells
Endogenous modification involves altering parent cells before isolating EVs so that the secreted EVs carry specific molecules. Available data suggest that specifically modified EVs can be generated by modifying donor cells.254,255 A vector containing the target gene is transfected into donor cells using genetic engineering technology, and the proteins and nucleic acids are loaded into EVs. Proteins or ncRNAs encoded by the inserted genes are produced by donor cells and secreted into EVs through a natural packaging process. EVs enriched with target proteins or nucleic acids can then be obtained by isolating and purifying the cell culture supernatant.252,256 However, during EV biosynthesis, cells utilize a complex sorting system to determine which substances are packaged into EVs.257 –261 This system limits specificity when directly expressing target proteins or nucleic acids in donor cells, often resulting in random packaging into EVs and low loading efficiency. 38
Reports indicate that different nucleic acid sequences exhibit varying sorting efficiencies in EVs. 262 Sequences with high GC content are more likely to be sorted into EVs. For example, miRNAs containing GGNG sequences are reportedly more likely to be sorted into EVs. 263 Additionally, miRNAs with CNGGAG and CGGGNG sequences (both containing the mentioned GGNG) were sorted into EVs 24 and 80 times more than those in AML12 cells and SVEC cells, respectively. Furthermore, introducing the CGGGAG sequence at the end of miRNAs with low GC content can enhance the loading of target miRNAs into EVs by 10–20 times. 264 Studies on miRNA sequences have also shown that miRNAs containing GGCU and GGAG sequences are preferentially sorted into EVs. This is primarily due to the RNA-binding proteins SYNCRIP and hnRNPA2B1 in EVs, which bind to GGCU and GGAG sequences. 261 Similar studies have shown that the pre-miR-451 backbone can efficiently load siRNA into EVs. 265 Moreover, target RNA loading can be facilitated by introducing RNA-binding proteins. 266 For example, the RNA-binding protein HuR can bind to the specific AUAU sequence on miR-155, and when adsorbed by HuR, this interaction enables the successful generation of miR-155-rich EVs through plasmid transfection. 267 These findings on miRNA or siRNA loading highlight that efficient loading can be achieved by incorporating specific short cluster sequences or a simple loading skeleton, as well as enriching target RNA using RNA-binding proteins.
The protein sorting efficiency of EVs varies with different protein types, with membrane-localized proteins being more easily sorted into EVs. 245 For non-membrane-localized target proteins, fusing the target protein with the constitutive EV proteins (Lamp2b,267,268 CD63,38,269 CD9 28 ) can enhance the abundance of the protein of interest in EVs. This process is mediated by the expression of the constitutive protein, which can also improve the specificity of negative loading of the target protein into EVs. Another method was to use the late-domain (L-domain) pathway to load foreign proteins into EVs. Studies have shown that WW-Cre recombinant enzyme proteins can be recognized by Ndfip1 protein in the L-domain pathway, leading to ubiquitination and subsequent uptake into EVs. 270 Furthermore, while genetic modification of donor cells and direct overexpression of the target protein do not always achieve efficient loading, fusion with EV marker proteins can result in significant loading of specific functional proteins. However, it remains to be verified whether the fusion protein affects the function and structure of the target protein, as well as the morphological structure and function of EVs.
Direct engineering of EVs
Direct engineering of EVs, also known as exogenous modification, involves loading target molecules directly onto or into the surface or cavity of isolated and purified EVs through passive or active methods. Current techniques for delivering therapeutic agents into EVs primarily include co-incubation,120,271 electroporation, 272 extrusion, 273 ultrasonic treatment, 274 calcium chloride-mediated transfection, 275 saponin permeabilization, 276 freeze-thaw cycles, 277 and the use of the Exo-Fect commercial kit. 123 For instance, the isolated and purified curcumin can be successfully loaded into EVs via co-incubation. EVs isolated by ultracentrifugation from cell supernatant were incubated with curcumin for 5 min, resulting in the encapsulation of curcumin within the EVs. 120 In addition, studies have demonstrated the translocation of target substances into EVs using electroporation. Following EV isolation, Cas9-sgRNA complexes were loaded by electroporation and administered through the mouse tail vein system to treat liver disease by knocking out the target gene. 278 Furthermore, comparisons of Exo-Fect, electroporation, heat shock, and saponin methods for miRNA loading into EVs after EVs separation and purification have shown that Exo-Fect improves the efficiency of miRNA loading into EVs. 279 These methods generally involve separating and purifying EVs, isolating and purifying substrates, and loading the target substance into EVs, particularly for proteins and miRNAs. However, it is important to note that these physical or chemical methods may cause membrane damage or leave behind chemical residues.280,281 While these methods are highly feasible, more efficient technologies may need to be further developed for clinical applications.
Challenges in EV industry
EVs are an important class of vesicles in vivo and have garnered significant attention since the field of vesicle trafficking was awarded the Nobel Prize in Physiology or Medicine in 2013. Numerous studies have since highlighted the substantial clinical potential of EVs in diagnostics and therapeutics. Despite these advancements, several challenges remain to be addressed, particularly concerning their immunogenicity, safety for long-term use, mass manufacturing, and purification. While EVs are generally considered biocompatible and less immunogenic than synthetic nanoparticles, their immunogenicity can vary depending on the source, cargo, and modification methods. Engineered EVs, in particular, may pose additional immunogenic risks due to alterations in their surface molecules or cargo, which could trigger unwanted immune responses. 282 Likewise, artificial vesicles or synthetic analogs of EVs, while advantageous for scalable production and precise functionalization, may lack the sophisticated immunological compatibility that natural EVs possess, raising concerns regarding biocompatibility and immunotoxicity.283,284 Long-term safety studies are equally imperative.
As drug carriers and diagnostic tools, EVs face additional challenges related to efficient drug loading and stability. Current physical and chemical loading methods can damage EV membranes or leave chemical residues, while the co-incubation method, though preserving EV integrity, suffers from low efficiency. 273 Genetically engineering parent cells to produce EVs with specific target molecules offers a more robust and scalable alternative, but achieving high-purity EVs remains difficult due to their distribution in complex biological systems. Common isolation methods like ultracentrifugation are cost-effective but time-consuming and yield impure EVs, whereas techniques such as size-exclusion chromatography and immunoaffinity capture achieve higher purity but are costly and labor-intensive.285 –287 Additionally, high-quality reference specimens with consistent purity and well-characterized markers are essential for research and clinical applications, but batch variability and scalability issues remain unresolved. 288 Currently, some companies researching EVs have developed products for the standardized separation of EVs (Hansa BioMed Life Science, Vesiculab, etc.). EV diversity further complicates the identification of universal markers, as commonly used markers like CD9, CD81, and CD63 are not universally present.289,290 Specifying detection standards for EV from different sources may be the direction of future development. Current detection methods also lack the sensitivity and specificity needed for low-concentration EV markers or disease-specific EVs. Finally, the cost-effectiveness of EV diagnostics compared to traditional methods like ELISA is uncertain, emphasizing the need for innovative approaches to improve efficiency, scalability, and accuracy while reducing costs. Addressing these challenges, particularly those related to immunogenicity and long-term safety, will be critical for the successful clinical translation of EVs and engineered EVs.
Concluding remarks and future perspective
EVs have introduced a promising new approach for disease surveillance, drug delivery, and gene therapy across a wide range of diseases. Over three decades have passed since EVs were first described, and significant progress has been made in understanding their biology and functions. Naturally occurring EVs, engineered EVs, and EV mimetics all demonstrate biological therapeutic effects. From the perspective of production cost and maintaining batch efficiency consistency, endogenous modification and the production of EV mimetics are considered more likely to achieve clinical application. The loading of specific drugs into EVs may reflect their particular functions, with the loading of nucleic acid or protein drugs into EVs potentially being more cost-effective and efficient through endogenous modifications, while the loading of chemical drugs may require exogenous modifications. Despite the significant progress in this field, unanswered questions remain that could guide future research. For instance, current research on the production of EV mimetics lacks investigation into their stability, which is crucial in determining whether EV mimetics exhibit similar stability to EVs. Furthermore, there is a lack of technical standards for the production of EVs, engineered EVs, and EV mimetics, as well as detailed standardized processes for the collection, preservation, separation, and purification of EVs from different samples. Another major challenge is achieving precise and efficient targeting of EVs to specific tissues or cell types. While surface modification with ligands, peptides, or antibodies has shown promise, these approaches often face challenges such as off-target effects, limited binding affinity, and potential immunogenicity of the added targeting moieties. Researchers are encouraged to address these key issues to facilitate the transition of EVs from the laboratory to clinical use.
Footnotes
Data availability
No new data have been generated in this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National key research and development program (2022YFD1300904, 2022YFD1300401); National Natural Science Foundation of China (32372958, 32072812); and Natural Science Foundation of Guangdong Province (2024A1515010510, 2023A1515012127)
