Abstract
Bioscaffolds with characteristics of the tumor microenvironment is an important substrate to support the formation of multicellular tumor spheroids (MCTS). Conventional biomaterials promote MCTS formation using tumor extracellular matrix (ECM) components and exogenous growth factors, but lack individualized tumor cell niche construction which may restricts cell biology and drug discovery research. Herein, an ECM mimicked fiber composite dual-network hydrogel (FCDNG) was constructed. The 3D nanofibrous framework was introduced into the hydrogel by a “dispersion-swelling” method and an alginate/platelet-rich plasma (PRP) dual-network hydrogel was constructed by a simple “one-step” activation process. The scaffold can well control the physicochemical conditions that mimic the ECM structure and biomimetic mechanical properties. The fibrin formed by PRP can promote tumor cell proliferation, angiogenesis and cancer stem cell enrichment. Meanwhile, FCDNG has suitable degradation properties to meet the matrix remodeling requirements during the formation of MCTS. Tumor cells cultured in FCDNG had enhanced tumor stemness, proliferation, and resistance to chemotherapeutic agents, and thus providing an effective strategy for clinical drug evaluation.
Introduction
Solid tumors such as breast, prostate, and pancreatic cancer are 3D structures composed of extracellular matrix (ECM) and heterogeneous cells. 1 Tumor cells are in contact in all directions with the ECM, which is naturally composed of a continuously integrated fibrous network and a negatively charged proteoglycan matrix. 2 As one of the important components of the tumor microenvironment, ECM plays a key role in tumorigenesis and development. In view of its advantages in the development of anti-tumor drugs and personalized tumor therapy, in vitro construction of artificial ECM has become a research hotspot. 3 Overall, cancer cells grown in 3D artificial ECM showed different phenotypic and genotypic levels, and response to anticancer drugs compared to those in conventional three-dimensional (2D) cell culture. 4 However, they are not considered ideal models for cancer research because of their inconsistent formation, the challenges of handling them, the lack of tissue ECM components, and their controversial biological relevance.5,6
Scaffold-based 3D culture of multicellular tumor spheroids (MCTS), which are tumor avatars for cancer patients, has emerged as a potential preclinical model.1,7,8 The ideal MCTS should be established in an artificial ECM similar to that of the original tumor to achieve a high correlation of drug response. 9 It is an ecological niche of a special environment where tumor cells, including cancer stem cells (CSC), reside and receive signals that determine their fate. These signals take the form of intercellular contacts, cell-matrix interactions, and secreted factors.10,11 CSC are a subpopulation of tumor cells that are highly self-renewing, chemo- and radiotherapy-resistant, and key to tumor recurrence, making them attractive targets for the development of anti-cancer therapies. 12 Tissue engineering techniques offer a very promising approach for the 3D culture of tumor cells. Several studies have shown that scaffolds that mimic the properties of tumor stroma in vitro lead to CSC enrichment.13–15
Hydrogels are commonly used as tumor cell culture candidates due to their natural similarity to the ECM. A prime example is sodium alginate, a tunable and versatile natural hydrogel that has been investigated in numerous studies as a culture matrix for use in various tumor models.16,17 Unfortunately, these conventional nanoporous crosslinked polymer networks typically exhibit high water content and do not feature cellular-scale fibrous topography, have poor mechanical strength, and are unable to maintain a stable 3D structure during tumor cell culture. 18 Natural ECM has inherent fiber/gel morphology and nonlinear mechanics. The structure/composition defects of hydrogel lead to its inability to promote cell-cell and cell-substrate interaction.
The design of novel synthetic matrix scaffolds must address the relevant properties of the ECM in vivo. Collagen fibers in the ECM have a diameter of 10–300 nm and provide tumor tissue resistance to tension and compression. A gel-like substance, mainly composed of glycosaminoglycans, surrounds the collagen fibers, providing resistance to compression, storage of water, and growth factors (GFs). 19 Structurally, scaffolds should provide mechanical stability with elasticity close to the desired tissue, allowing cells to adapt through small interactions. In addition, the scaffolds must have an open porosity that allows cells to migrate into the material, facilitating the formation of 3D tissue structures. To mimic the fibrous structure of the matrix ECM, the scaffold must be composed of fibers with physiologically adjustable diameters in the submicron or nanometer range. Several research groups have shown that the fibrous structure facilitates the migration or invasion of cancer cells.20–22 On the other hand, the ECM components have a significant effect on cell behavior. MCTS cultured on scaffolds containing ECM components including matrigel, hyaluronan, collagan, and peptide-proteinshow a strong upregulation of EMT-associated transcription factors and CSC enrichment.23,24 However, it lacks control in mimicking the ECM because of its high batch variability, undefined composition, and murine origin. These features are important limitations to effectively screen and develop new treatments for cancer. 25 Platelet-rich plasma (PRP), a blood-derived product, is a concentrated platelet derived from whole blood that is activated by calcium/thrombin. 26 The activated platelets immediately degranulate and secrete a variety of pro-survival, pro-angiogenesis, and immunoregulatory factors in a non-contact manner to construct and maintain primary and metastatic tumor microenvironments.27,28 At the same time, plasma rich in fibrinogen is converted into a network of densely stacked polymerized fibrin that forms a gel capable of entangling factors. Fibrin and fibrinogen and their degradation products are actively involved in the pathophysiology of cancers, including blood vessel formation and cancer progression. 29 Moreover, it can be obtained from patient’s own blood to be used as an autologous scaffold, to reduce the potential risk of immunological reaction or infections. 30 In addition, the advantages of personal PRP made it suitable for artificial ECM, which can meet the requirement of personalized drug screening for patients with advanced cancers using exhaustion of conventional therapies. 31
In this study, we prepared composite scaffolds for tumor cell culture using highly porous electrostatically spun fiber mesh and PRP-based hydrogels. The mechanical properties of the fiber composite gel scaffolds were closer to those of natural tumor tissues, with a significant increase in the porosity. In addition, the scaffolds could continuously release growth factors during cell culture to promote cell proliferation. To facilitate the production of this in vitro biomimetic tumor scaffold standardized, repeatable, and easy to operate, we chose PLGA as the fibrous material, and sodium alginate and PRP were selected to form a dual network hydrogel through a one-step activation process, and they acted as sufficient substrates for 3D culture scaffolding capabilities, as shown in Scheme 1. In a proof-of-concept study, we determined tumor cell responses to different chemotherapeutics. We further probed the possible sources of drug resistance.
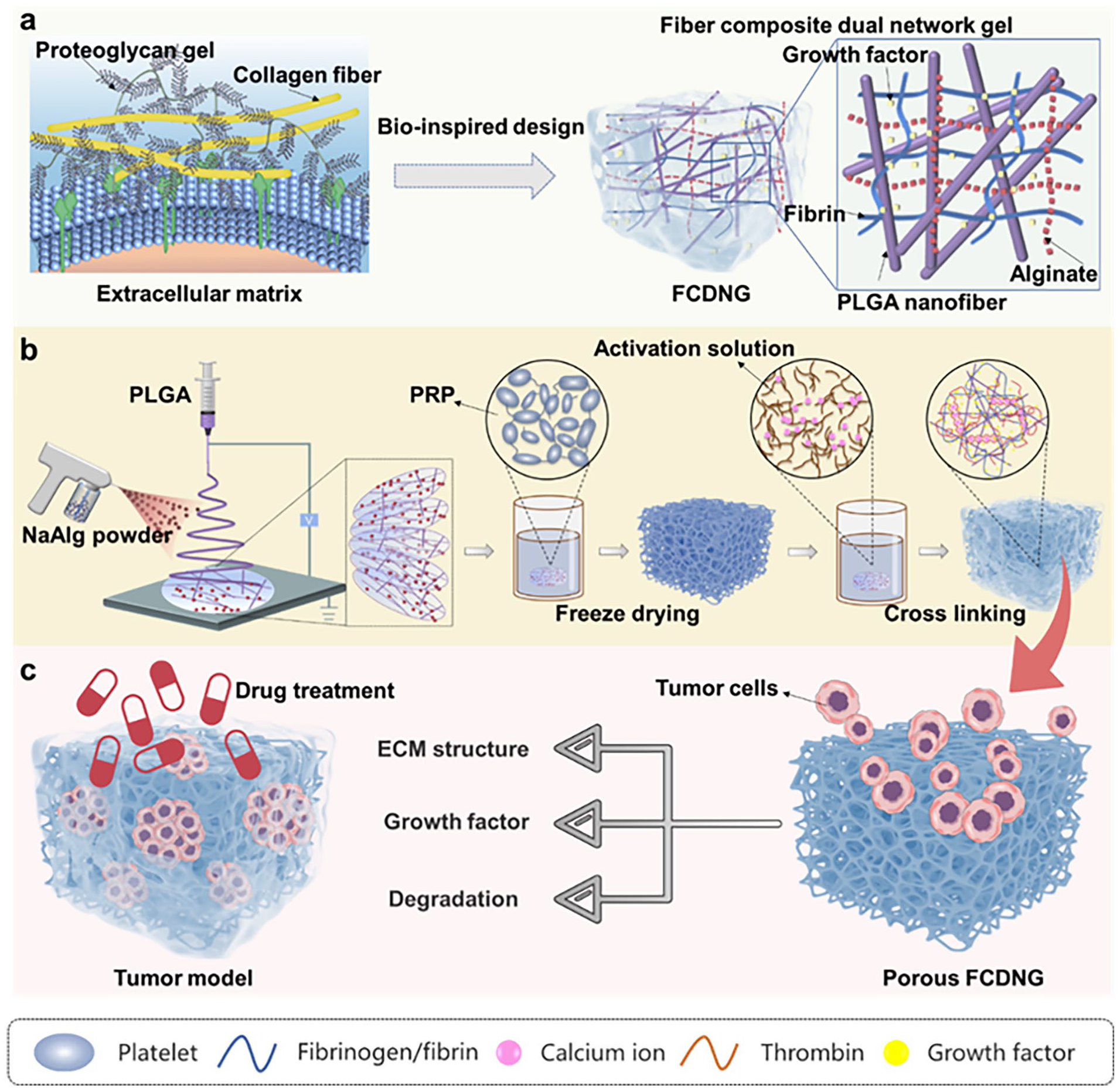
Schematic illustration of the fiber composite hydrogel and its preparation procedure. (a) Inspired by tumor ECM, a fiber composite dual network gel (FCDNG) that comprises a continuous electrospun fibers and a NaAlg/fibrin dual network gel is engineered for tumor cell culture. (b) The fabrication of FCDNG. (c) The resultant FCDNG displays multifunction including bionic structure, favorable mechanical performance, prolonged release of growth factors, and the appropriate degradation rate, therefore offering an advantageous microenvironment for formation of cell spheroid.
Material and methods
Materials and cells
Sodium alginate (SA, viscosity of 10 g/L dissolved in water at 20°C ⩾ 0.02 Pa s, Mw = 400 kDa), calcium chloride (CaCl2), N,N-dimethylformamide (DMF), and chloroform (CF) were purchased from Sinopharm Chemical Reagent Co, Ltd (Shanghai, China). Platelet-rich plasma was purchased from Guangzhou Hongquan Bio-technology Co, Ltd (Guangdong, China), thrombin was purchased from Rhön Reagent, PLGA (lactide/glycolide, 50/50; Mn = 100,000) was purchased from Jinan DaiGang Bio-Engineering Co, Ltd (Shandong, China). DU-145 cells, MCF-7 cells, Dulbecco’s modified Eagle’s medium (DMEM), and Minimum Essential Medium (MEM) were provided by the Stem Cells Bank, Chinese Academy of Sciences (Shanghai, China).
Preparation of samples
The clinically used SA and PRP were chosen as the skeleton from the point of view of safety and easy reproducibility.
Preparation of single network gels: Using deionized water as solvent, 10 mg/mL concentration of SA solution was prepared, 1 mL/well of SA solution was added to the 12-well plate, after freeze-drying, 0.8 mL of 20 mM CaCl2 solution was added to each well to crosslink for 30 min, and then freeze-drying was performed to obtain the single-network gel (SNG).
Preparation of dual network gels: PRP was diluted with deionized water (the volume ratio of PRP solution to water is 100% v/v, 50% v/v, 35% v/v, and 20% v/v, respectively). Adding 1 mL/well of above solution into the 12-well plate after primary freeze-drying of SA. Then, 0.8 mL of cross-linking solution containing 1 U mL thrombin and 20 mM CaCl2 was added to each well, and cross-linking was performed for 1 h at 37°C in a constant temperature shaker, and then the dual network gel (DNG) samples (i.e. DNG-100, DNG-50, DNG-35, and DNG-20) were obtained after secondary freeze-drying.
Preparation of PLGA fiber mesh: PLGA was dissolved in a mixture of chloroform (CF) and DMF (VCF: VDMF = 4:1) with mass ratio of 8%, 10%, and 12% (w/v). The obtained solution was subjected to electrospinning (TL-Pro, Shenzhen Tongli Micro-Nano Technology Co., Ltd., China) under the following process parameters: flow rate of 1, 1.5, and 2 mL/h, voltages of 18, 20, and 22 kV, respectively, and a collection distance of 20 cm.
Preparation of fiber composite single network hydrogel (FCSNG): The FCSNG were prepared by adding SA powder on-line during the electrostatic spinning process, wetted with deionized water, freeze-dried once, cut into 24-well plates, crosslinked with 0.8 mL of 20 mM CaCl2 for 30 min and freeze-dried. The FCSNG composite scaffolds were prepared with fiber contents of 5%, 10%, and 15% (w/w) were prepared by adjusting the spinning time.
Preparation of fiber composite dual—network hydrogel (FCDNG): The FCDNG were prepared on the basis of FCSNG scaffolds by immersing each FCSNG into PRP solution for complete absorption, then crosslinking the scaffolds with 800 μL crosslinking agent containing 1 U/mL thrombin and 20 mM CaCl2 for 1 h at 37°C in a shaking bed, and then freeze-drying as shown in Scheme 1.
Physico-chemical characterizations
The microstructure of the lyophilized hydrogels was examined using field emission scanning electron microscopy (FESEM, SU8010, Japan), while energy dispersive spectroscopy (EDS) component mapping (Escalab 250Xi, Thermo Fisher Scientific, USA) was used to measure the cross-sectional C, O, N, and Ca elements. A stress-controlled rheometer (MARS 60, Thermo Fisher Scientific, USA) with a 20 mm plate was utilized to measure the rheological properties of the hydrogels. The energy storage modulus (G′) and loss modulus (G″) were measured at 37°C and at a constant frequency of 1 Hz (in the linear viscoelastic region) in the 0.01%–10% strain during the amplitude sweep, while the oscillation frequency experiments were carried out at a 1% fixed strain along 0.1–100 rad/s. The pore size of the scaffolds was established using a pore size analyzer for porous materials (CFP-1100AI, Porous Materials, USA). The compression properties of the scaffolds were evaluated using the compression elasticity tester (LLY-06D, Laizhou Electronic Instrument Co., Ltd., China). A cylindrical indenter with a diameter of 20 mm was used to compress the samples at a rate of 5 mm/min until a strain of 50% was reached. At least three specimens of each stent type were tested. Swelling rate was tested after impregnation in phosphate buffer solution (PBS). The contact angle tester (OCA15EC, Dataphysics, Germany) was used to measure the static contact angle of the scaffolds.
The scaffold degradation was performed at 37°C in PBS with continuous shaking at 60 rpm. During degradation, the solution was replaced every 2 days. Samples were collected at predetermined intervals, washed thoroughly with deionized water, lyophilized, and weighed. The degradation rate was calculated by assessing the mass variation using the previous method. 32
Release Profiles of Hydrogels: To study the related GFs release behavior of hydrogels, DNG (containing 800 μL of PRP) were immersed in PBS buffer (1 mL) at 37℃. About 250 μL of PBS was collected at predetermined time intervals, while 250 μL of new PBS was replenished. The content of bFGF and VEGF was measured using the ELISA kit.
Cell culture
DU-145 cells were cultured in DMEM medium with 10% FBS and 1% P/S. MCF-7 cells were cultured in MEM medium with 10% FBS, 1% penicillin-streptomycin solution (P/S), 1.12% non-essential amino acids (NEAA), and 0.01 mg/ml of human recombinant insulin. All cells were incubated at 37°C in a 5% CO2 environment.
Cell proliferation assay
To determine the biocompatibility of the scaffolds, the cell viability of the scaffolds using MCF-7 cells was first tested before cell proliferation assay. The cell viability and proliferation stimulated by hydrogels was evaluated using Alamar Blue agent according to the reported procedure. 33
Cell viability assay. MCF-7 cells were inoculated into 24-well plates at a density of 2 × 104 cells/mL. After cell attachment to the wall, the sterilized samples of the 24-well tissue culture plate (TCP) sizes were placed in the culture medium, with an additional group serving as a blank control without samples. After 24 h, 1 mL of Alamar Blue (Yeasen Biotechnology Co., China) working solution (VDMEM: VAlamar Blue = 9:1) was added to the well plates and incubated for 10 h. The optical density (OD) value of 570/600 nm was determined using a microplate reader (Sunrise Tecan, Austria), and the cell viability of each group was calculated following the manufacturer’s instructions.
Cell proliferation assay. The steps for seeding cells into the scaffolds were as follows: 5 × 104 DU-145 or MCF-7 cells were first dispersed in 200 μL medium to create a cell concentrate. The cell concentrate was slowly added drop by drop to the surface of the scaffold using a 10-μL pipette. After cell addition, these scaffolds were gently transferred to the cell incubator. After 4 h of incubation, 800 μL of cell culture medium was slowly and uniformly added to each sample. It was then gently transferred to the cell culture incubator. At pre-determined intervals (days 4, 7, and 10), the reduction values for each group were calculated using the Alamar Blue kit according to the experimental method described above. In addition, cell viability (%) was calculated for each group as a description of cell proliferation performance, using the reduction values obtained on day 4 of the 2D culture as a reference.
Besides, to observe the spread morphology of cells on the sample surface, the cells were visualized by FESEM after culture, according to the previous experimental procedure. 32 After 7 days, cells were fixed by adding 2.5% glutaraldehyde at 4°C overnight and protected from light for FESEM test. The cells were dehydrated with a gradient of ethanol (30%, 50%, 75%, 90%, 95%, and 100% v/v). After dehydration, the cells were dried using mixtures of ethanol and hexamethyldisilazane with different v/v ratios (ethanol:HMDS = 2:1, 1:1, 1:2, and 100% HMDS). Finally, cell morphology was observed by FESEM.
Immunofluorescence staining assay
Live-dead cell staining. The cultured cells were washed with PBS three times, followed by Calcein AM/PI Double Staining Kit staining (Shanghai Keygen Biological Company, China) according to the provided instructions. Afterwards, apoptosis was observed using an inverted fluorescence microscope (Leica DMIL LED, Germany).
Cell morphology staining. After culture, the samples washed with PBS were fixed with 4% paraformaldehyde for 1 h, then permeabilized with 0.1% Triton X-100 solution for 5 min. After blocked with 5% bovine serum albumin (BSA) for 30 min, the actin and nucleus were marked with rhodamine-labeled phalloidin (Invitrogen, China) and 4,6-diamidino-2-phenylindole (DAPI, Beyotime, China), and visualized under a confocal laser scanning microscope (CLSM).
Tumor markers staining. After 7 days of culture under 2D and 3D conditions, the cells were fixed and permeabilized at room temperature, and then 1 mL 1% BSA serum was added to block the cells for 1 h. Diluted primary antibody solution N-Cadherin (1: 300, Abcam, UK), MMP2 (1: 300, Abcam, UK), SOX2 (1: 300, Abcam, UK), Integrinβ1 (1: 100, Bioss, China), and YAP (1: 500, Invitrogen, USA) was added and incubated at 4°C overnight. On the second day, the first antibody was sucked out and the samples were washed with PBS. The secondary antibody was diluted in 1 % BSA at a ratio of 1: 200, and 800 μL of the solution was added to each sample. Finally, the nucleus was marked with DAPI and captured by CLSM.
RNA isolation and quantitative real time-polymerase chain reaction (qPCR) analysis
The cells after 7 days of culture were collected and mechanically disrupted and the total RNA was isolated using TRIzol reagent (Thermo Fisher Scientific, USA). RNA quantity and purity were determined using a NanoDrop 2000 UV-Vis spectrophotometer (Thermo Scientific, USA). The primer sequences of specific genes are shown in Supplemental Table S1. Calculation of relative gene expression levels was conducted using the 2−ΔΔCT method, and β-actin was used as the reference; results were presented as relative RNA levels, compared with 2D culture (n = 3 for each sample).
Chemosensitivity assay
To assess chemosensitivity, the 3D and 2D samples were treated with various concentrations of different drugs. As shown in Supplemental Table S2, after 4 days of proliferation, the samples were incubated in a culture medium supplemented with various concentrations of drugs for another 3 days. The group with a drug concentration of 0 as the control group. Relative inhibition rates of the cells were calculated using the equation: (1 − ODsample/ODcontral) × 100%.
Western blot analysis
The total protein was extracted and quantitated using the BCA method. Tumor cells were homogenized in RIPA solution (Beyotime, Shanghai, China) containing protease inhibitor cocktail. Next, protein concentrations were determined using a BCA Protein Assay kit (Beyotime, Shanghai, China). The equal amounts of protein extracts were resolved on SDS-PAGE kit and then transferred onto nitrocellulose membranes (NC membranes). The membranes were then blocked using Western blocking solution (Beyotime, P0023B, Shanghai, China) and incubated with the desired primary antibodies including Anti-pAKT antibody (Bioss, bs-2720R, 1:1000, China), Anti-AKT antibody (Proteintech, 60203-2-Ig, 1:5000, USA), Anti-pERK antibody (Affinity, AF1015, 1:1000, USA), Anti-ERK antibody (Abcam, ab32537, 1:1000, UK) at 4°C overnight. The membrane was then washed and incubated with appropriate secondary antibodies at room temperature for 1 h. Immunoreactivities were visualized using the ChemiDoc Imaging System (Bio-Rad, CA, USA).
Statistical analysis
The data were presented as the mean ± standard deviation (SD). All experimental groups were analyzed in technical triplicates unless otherwise noted. The data were analyzed by t-tests or one-way ANOVA followed by Tukey’s test. A p-value of less than 0.05 was considered statistically significant (*p < 0.05, **p < 0.01, and ***p < 0.001).
Results and discussion
Preparation and characterization of hydrogels
The SA single network gels (SNG) and SA-PRP dual network gels (DNG) was successfully synthesized using a simple “one-step” activation process are shown in Scheme 1 and Figure 1(a). SA aerogel was first formed, followed by impregnation of SA aerogel in PRP solution. A mixture of thrombin and CaCl2 was then added to the solution as one gel medium for both networks. The addition of thrombin and CaCl2 to the PRP solution causes plasma to coagulate and fibrinogen to polymerize into a fibrin network, resulting in the formation of a PRP gel. On the other hand, Ca2+ ions can interact with the carboxyl groups present in the alginate backbone to form an “egg carton” ionic cross-linked network. Within the DN gel, which is widely used as a surgical aid, the PRP gel was prepared according to the classical activation procedure.34,35 As can be seen by visual observation and SEM (Figure 1(b) and Supplemental Figure S1a–c), the porosity of the four DNG scaffolds was over 80%. By gradually decreasing the PRP content, the DNG transforms from a dense structure to a typical 3D structure with interconnected pores. The scaffolds with lower PRP content (DNG-35 and DNG-20) had a little higher porosity. The average pore size of DNG scaffolds gradually increased with decreasing PRP content, which were 13.22 ± 0.95, 22.35 ± 1.09, 41.63 ± 1.150, and 58.50 ± 1.57 μm, respectively. Higer porosity and lager pore size facilitates cellular uptake of nutrients and transport of metabolic waste. In addition, a fibrous network structure was found in the pore junctions and walls, suggesting that the fibrin network was derived from activated PRP. The presence of a nanoscale fibrous network structure in the internal pore of gel controls intercellular interactions and improves cell-matrix interactions.36,37

Preparation and characterization of gels. (a) Schematic showing the fabrication of SNG and DNG based on a simple “one-step” activation method. SEM images of (b) DNG and SNG. (c) FTIR spectrum. (d) XPS spectrum. (e) Cell viability after incubation with DNG. (f) Rheological analysis.
The formation of the DNG was further investigated analytically by FTIR (Figure 1(c) and Supplemental Figure S2a). The peaks in SNG located at 1597 cm−1 (carbonyl group) and 1417 cm−1 (carboxyl group) were attributed to antisymmetric and symmetric stretching of carboxylates, respectively. 38 PRP gels showed characteristic peaks of amide groups, including amide II (1548 cm−1, C=O stretching vibration) and amide I (1644 cm−1, C–N stretching vibration and N–H bending vibration). 39 The characteristic peaks of both NaAlg and fibronectin were observed in DNG. FTIR spectra showed the presence of both sodium alginate and fibrin fractions in DNG scaffolds, confirming the successful preparation of composite DNG. Specific N-elements derived from activated PRP were identified in the DNG using XPS (Figure 1(d) and Supplemental Figure S2b) and energy dispersive spectroscopy (EDS) elements, also suggesting that the fibrin network was uniformly dispersed in DNG (Figure Supplemental Figure S2c).
PRP-derived scaffolds are well suited for 3D cell culture because of the large number of immunomodulatory proteins, and platelet-activated endogenous growth factors (GFs) that stimulate cellular activity. 40 However, previous studies have shown that the dose-response curve for PRP is not linear and that high levels of PRP may even have an inhibitory effect on cell growth. 41 To understand the effect of PRP content on cell proliferation, cell viability of MCF-7 cells on DNG scaffolds with different PRP content was evaluated (Figure 1(e)). MCF-7 cells were cultured on 2D and DNG hydrogels for 7 days, and the cell viability of each group was calculated. The cell viability on the fourth day of 2D culture was used as a reference. At the detection time point, the cell viability in DNG with low PRP content (DNG-35 and DNG-20) was always higher than that in DNG hydrogels with high PRP content (DNG100 and DNG-50). On the seventh day, DNG-35 hydrogels showed the highest level of cell proliferation. This is partly due to the small pore size and low porosity of DNG hydrogels at high PRP content. Increasing the fibrin content would significantly decrease the pore size and fluid permeability of the fibrin gels, potentially impacting cell proliferation by hindering nutrient uptake and waste transport. 42 Conversely, when the fibrin gel content was too high, it resulted in a denser fibrin gel, which also prevented cell migration and proliferation. 30 Due to the low content of fibrin and growth factors in DNG-20 hydrogel, its effect on cell proliferation is not as good as that of DNG-35. Rheological characterization also showed that DNG-35 formed a more stable gel than SNG (Figure 1(f)). Therefore, the follow-up studies used DNG-35 to prepare fiber-reinforced hydrogel scaffolds.
Preparation and characterization of fiber composite hydrogels
The ECM surrounding cells serves a complex nanoscale structural protein architecture, including collagen, laminin, and fibronectin, to form the preexisting mechanical properties within the cell microenvironment. In this study, PLGA electrospinning continuous filaments (length: diameter > 10,000:1) were used to construct fiber composite hydrogels to achieve effective stress transfer. The structure of the scaffolds was observed via SEM, and the resulting images are presented in Figure 2(a) and (b). To provide a scaffold related to the structure of solid tumors, in this study, PLGA fibers with a diameter of 246.22 ± 58.33 nm were optimized by adjusting the voltage and the flow rate (Figure 2(b) and Supplemental Figure S3–S5). This is comparable to the diameter of collagen fibers in tumor tissue (10–300 nm). Prefibrillar networks (marked by yellow arrows) were found in the pore junctions and walls (Figure 2(b) and Supplemental Figure S6), indicating fibrin networks from activated PRP. 33

Preparation and characterization of fiber composite gels. (a) Photos and (b) SEM images of PLGA, SNG, DNG, FCSNG, and FCDNG. (c) Porosity and (d) pore size of PLGA, SNG, DNG, FCSNG, and FCDNG (The blue rectangle in the figure is the size range of MCF-7 and DU-145 tumor cells: 16–25 μm). (e) FTIR spectrum and (f) XPS spectrum of PLGA, SNG, DNG, and FCDNG. Rheological analysis of (g) SNG, FCSNG and (h) DNG, FCDNG. The (i) compression stress-strain curve and (j) compression modulus of samples.
The composite scaffolds have interconnected pores and the porosity exceeded 80% (Figure 2(c)), that provided ample space for nutrient exchange, waste transfer, and the release of growth factors from PRP. Thus the composite scaffold can promote tumor cell proliferation migration, and proliferation. 43 The average size of DU-145 was about 20 μm and the size of MCF-7 cells ranged from 16 to 25 μm.44,45 Therefore, the FCSNG and FCDNG composite scaffolds with suitable pore size allow cell infiltration without affecting cell adhesion and compromising the mechanical properties of the scaffolds (Figure 2(d)).
FTIR (Figure 2(e)) and EDS results confirmed the composition of the scaffold (Figure 2(f) and Supplemental Figure S7), validate the successful preparation of fiber-composite single-network gel (FCSNG) and fiber-composite dual-network gel (FCDNG) composite scaffolds using a “one-step” activation method.
Rheological analysis was performed to characterize the viscoelasticity of the hydrogels. The energy storage modulus (G′) values of all hydrogels significantly exceeded their loss modulus (G″) values as frequency gradually increased, indicating their elastically dominant properties (Figure 2(g) and (h)). The incorporation of fibers increased the G′ of the SNG and DNG. This suggests that fibers play a role in the elasticity of the composite scaffolds. This benefits from the mechanically self-locking structure formed by the fibers in the hydrogel, which can dissipate energy during deformation. 46 The three network components are interspersed and uniformly distributed at the microscopic scale, which contributes to the overall macroscopic mechanical properties. All 3D scaffolds demonstrate a typical “J-curve” loading profile (Figure 2(i)), which is consistent with the most biological tissues. 47 Once the cells are seeded, the scaffold should maintain its integrity and strength until the loaded cells adapt to the environment and secrete sufficient ECM. 48 The advantage of PLGA fiber reinforced hydrogel is the mechanical properties can be effectively regulated by adjusting the PLGA fiber content without changing its porosity (Figure 2(j) and Supplemental Figure S8). The adjustable stiffness can meets prostate tumors modulus ranging from 24.1 to 88 kPa. 45 Previous studies revealed that hydrogels with a modulus of 5 kPa optimally support the growth and expression of tumor stemness in MCF-7 cells. 49 Therefore, fiber contents 10% and 15% of FCDNG were selected as culture models for breast and prostate tumor cells, respectively.
Stability characterization of fiber composite hydrogels
When utilizing hydrogels for tumor model, insufficient swelling can restrict the capability to exchange substances, while excessive swelling can lead to hydrogel breakdown and disintegration, rendering it impractical for further use. The swelling rate and the appearance of the scaffolds was measured after hydration in PBS for 24 h (Figure 3(a) and (b) and Supplemental Figure S9). The swelling rate of DNG (2733%) was lower than that of SNG (3468%) owing to the increase in crosslinked network. SNG showed an unstable structure morphological structure of during the swelling process, adding PLGA fiber can improve its stability. This is because evenly dispersed PLGA fibers restricted excessive swelling of the hydrogel (Supplemental Figure S10). Fiber component assisting the scaffolds in maintaining long-term structural stability for cell culture. 50
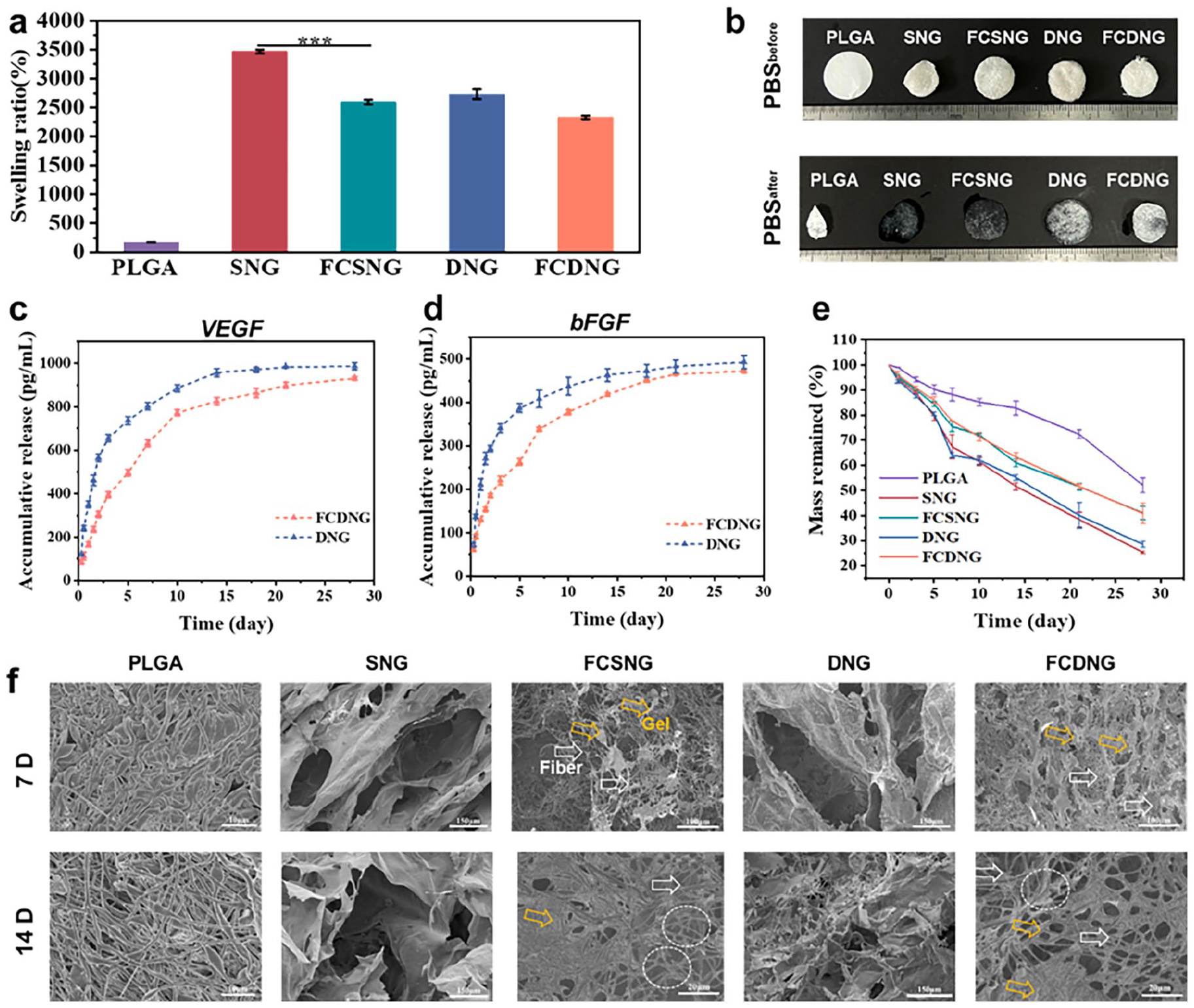
The wettability and stability characterization of fiber composite hydrogels. (a) Swelling behavior. (b) The stability of the scaffold after immersion in PBS for 24 h. (c) bFGF and (d) VEGF release profiles of DNG and FCDNG. (e) Mass change and (f) morphology of PLGA, SNG, DNG, FCSNG, and FCDNG during the degradation process.
Calcium-activated PRP provides many GFs and bioactive substances (e.g. VEGF and bFGF), which can regulate the cell growth process. VEGF plays an important role in tumor angiogenesis, and bFGF stimulates cell proliferation and migration. GFs release from DNG and FCDNG scaffolds was measured and the results are shown in Figure 3(c) and (d). Approximately 57 % of the GFs was released from the DNG in the first 2 days, followed by a sustained release of 14 days. Compared to single-network hydrogels in the reference (the release rate reached 75% within 10 h), DNG exhibit slow-release behavior with more durable supply of growth factors. 33 The release of growth factors in FCDNG scaffolds was slower in the early stage than DNG, and about 32 % of GFs were released in the first 2 days, and a sustained release for up to 21 days. This could be attributed to the fact that PLGA fibers elongate and bend the path for drug diffusion, and also provide an additional diffusion barrier due to their hydrophobic nature, which limits hydrogel degradation. 51 For these reasons, FCDNG composite scaffolds exhibit more stable and prolonged release profiles.
Typically, scaffolds that maintain structural integrity are essential for tumor cell growth, while appropriate degradability allows for cellular migration, proliferation, matrix remodeling, and MCTS formation. The degradation rates of PLGA membranes, hydrogels, and fiber composite hydrogel scaffolds over 28 days are presented in Figure 3(e). The mass loss of SNG and DNG were faster than that of FCSNG and FCDNG. The incorporation of PLGA nanofibers promotes the retention of the network structure within the hydrogel (marked with arrow). This led to improved integrity and stability, resulting in a reliable support for long-term (more than 7 days) cell cultivation. SEM images (Figure 3(f) and Supplemental Figure S11) revealed that around 50% of the electrospun scaffolds remain after 21 days. Thus, during cell culture, FCSNG and FCDNG can still maintain ECM-like structures. 52
Cell cultures within gel and fiber composite gel
The cells morphology within gels and fiber composite gel was studied and the cells grown within PLGA and 2D tissue culture plate (TCP) as a control group. The results showed that the scaffolds had good cell compatibility, and the cell viability of all scaffolds was more than 80 % compared with TCP (Supplemental Figure S12). Figure 4(a) and (b) presents the cell viability of human prostate tumor cells DU145 and human breast tumor cells MCF-7 at the time points tested. The cell viability of cells in each group consistently increased over 10 days, suggesting that the cells were capable of proliferation in both 2D and 3D cultures. The number of cells under 3D culture consistently remained smaller than that of 2D culture throughout the experiment, which is closer to the slower growth curve of tumor natural growth. This observation is consistent with previous studies.53–55 By comparing cell viability between the scaffold with and without PRP, it can be found the presence of PRP led to a significant increase in cell proliferation (Supplemental Figure S13), in line with previous studies. 56 This was attributed to fibronectin can provide an adhesive surface and a more stable environment for cell growth.56,57 The comparison of cell viability between FCSNG and SNG, FCDNG and DNG showed that the addition of fiber components could significantly improve cell proliferation. This can be attributed to two reasons. One is that nanofibers formed by electrospinning are able to highly mimic the fiber networks of natural ECM, which have been evidenced to provide morphological cues that improve cell adhesion and proliferation. 58 Another is that the elastic modulus of the fiber composite gel scaffold is closer to the tumor tissue. Increased matrix stiffness has been reported to enhance tumor cell-ECM interactions. 59 The results above suggest that the FCDNG composite scaffold can mimic the tumor extracellular matrix both chemically and physically, which serves as a more appropriate scaffold than hydrogel for in vitro tumor cell culture.

Cell cultures within gel and fiber composite gel. In vitro cell proliferation assays of (a) DU-145 and (b) MCF-7. *p < 0.05, **p < 0.01, ***p < 0.001. FITC and DAPI staining of (c) DU-145 and (d) MCF-7 to demonstrate cell morphology after 10 days of culture. Scale bar: 50 μm.
Cell nuclei labeled by DAPI and actin labeled by FITC were observed through CLSM. In 2D culture and on PLGA membranes, DU-145 cells took on a shuttle-shaped, polygonal form, and were seen beginning to stack and grow apically by day 7 (Figure 4(c), Supplemental Figures S14a and S15). MCF-7 cells attached to 2D and PLGA electrostatically spun scaffolds displayed a triangular or polygonal shape and fused into flattened sheets. As the incubation time increased, the cells gradually formed a monolayer (Figure 4(d), Supplemental Figure S14b). In 3D scaffolds, MCF-7 and DU-145 cells formed spheres observed through with z-stack imaging over time similar to those found in vivo (Figure 5(a)). Each sphere consisted of many cells, indicating that single cells can form multicellular tumor spheres in scaffolds after multiple cell divisions. Sphere formation is typically associated with poor prognosis and enhanced metastatic potential. 60 At day 4, part of the cells in the SNG scaffold aggregated but did not form multicellular tumor spheres due to the oversized pores of the SNG scaffolds, which limit cell proliferation. 61 Notably, multicellular tumor spheres are mostly single independent individuals and there are more individually distributed cells in gel scaffold, with spheres farther apart from each other. In contrast, the fiber-containing FCSNG and FCDNG scaffolds showed several adjacent multicellular tumor spheres, indicating that cells tended to interact with each other. This could be attributed to the varying depths at which the fibers are dispersed in the hydrogel, restricting cell settling and a more compact cell distribution. 62 Additionally, the fibers permit cellular communication through biochemical signals, as demonstrated by the distribution of actin between neighboring spheres. 63 As a consequence, these tumor spheres progressively grow in size over time, and neighboring spheres can combine to create bigger clusters of tumor cells (Figure 5(b) and (c)).

Characterization of cell spheroid within gel and fiber composite gel. (a) Diagram and (b and c) depth of Z-stack. **p < 0.01. (d) SEM images of cell spheroid.
SEM photographs of tumor cells demonstrated in Figure 5(d). Cells on PLGA membrane were spread out on a plane without forming spheres. A number of multicellular tumor spheroids were noted on 3D scaffolds, and DU-145 cells resembled those in solid tumors exhibiting the typical cobblestone morphology of epithelial cancer cells. The observation of pseudopods on the cell surface connected to PLGA fibers (marked with blue rectangles) within the FCDNG scaffolds suggests cell anchoring to the fiber network, promoting cell adhesion, proliferation, and signaling. 50 The cell surface on composite scaffolds of FCSNG and FCDNG exhibited microvilli structure (marked with blue arrows). Microvilli are typical of epithelial cancer cells and impact sensitivity to cytotoxic compounds, such as chemotherapeutic agents, while the pore-like features may indicate the formation of duct-like structures within the spheres.64,65 In contrast, MCTS formed on scaffold without fiber presented a smooth surface. MCF-7 cells cultured on various scaffolds presented similar results (microvilli structure marked with yellow arrows, tubular structure marked with yellow rectangles). The results above demonstrate that fiber-hydrogel composite scaffolds can enhance the interaction between tumor cells and matrix, and their microenvironment bestows growth benefits for tumor cells.
Changes in the expression of tumor markers
Metastasis of breast and prostate tumors has a significant impact on patient survival rates. The process of tumor metastasis involves the epithelial-mesenchymal transition (EMT), during which epithelial cells show characteristics of tumor cells, including enhanced stemness, motility, metastasis, and drug resistance. 66 The process of EMT, which promotes cell motility and tumor metastasis, is characterized by the down-regulation of E-cadherin and up-regulation of N-cadherin. As shown in Figure 6(a) and (b), FCDNG scaffolds significantly increased N-cadherin expression in DU-145 cells by 40-fold, 15.66-fold, and 1.76-fold compared to 2D culture, PLGA membrane, and DNG. Furthermore, the 3D scaffold reduced E-cadherin expression. Figure 6(c) and Supplemental Figure S16a showed that the significant upregulation of N-cadherin expression in MCF-7 cells using FCDNG scaffolds as well. Other study have reported a similar trend for tumor cells cultured in alginate composite gel systems. 67 This implies that the fiber and hydrogel components have potent synergistic effects on epithelial cell transformation into mesenchymal stromal cells, and that both are essential for this process.

Changes in the N-cadherin, E-cadherin, SOX2, and OCT4 expression of cells. (a) N-cadherin staining of DU-145 cells. N-cadherin and E-cadherin expression of (b) DU-145 cells and (c) MCF-7 cells. (d) SOX2 staining of DU-145 cells. SOX2 and OCT4 expression of (e) DU-145 cells and (f) MCF-7 cells.
Tumors comprise a diverse collection of cells, among which a small proportion of CSC, or cancer-initiating cells, possess stem cell-like properties and drive tumorigenesis, advancement, and metastasis. Research on CSCs has elucidated the mechanisms underlying drug resistance and tumor recurrence. Tumor cells that undergo EMT display stem cell-like characteristics and express biomarkers SOX2 and OCT4. These biomarkers’ expression levels can be utilized to evaluate tumorigenesis. 68 The expression of SOX2 and OCT4 significantly increased in the FCDNG composite scaffolds compared to 2D culture and DNG. A 1.91-fold increase in SOX2 and a 2.22-fold increase in OCT4 expression were observed in DU-145 cells (Figure 6(d) and (e)), while a 2.45-fold increase in SOX2 and a 3.04-fold increase in OCT4 expression were observed in MCF-7 cells (Figure 6(f) and Supplemental Figure S16b). Similar results were observed in terms of the expression of CSC markers, which contributing to the self-renewal and pluripotent differentiation of tumor cells.67,69 The composite scaffold showed a more pronounced effect on the expression of OCT4, indicating the significant role of the microenvironment in the expression of distinct stem cell markers.
MMP-2 and MMP-9 are essential proteins for degrading ECM during the invasion and metastasis of numerous tumor cells. These proteins are significantly expressed in aggressive prostate and breast tumors, causing the migration of tumor cells into nearby tissues. 70 Compared to 2D culture, DU-145 cells in FCDNG scaffolds exhibited significant increases of 10.69-fold and 10.86-fold in MMP-2 and MMP-9 transcripts, respectively (Figure 7(a) and (b)). In addition, compared with DNG, FCDNG also significantly up-regulated the expression levels of MMP-2 and MMP-9. Similarly, MCF-7 cells in FCDNG scaffolds showed significant increases of MMP-2 and MMP-9, respectively (Figure 7(c) and Supplemental Figure S17a). These results suggest that FCDNG scaffolds further promote tumor cell invasion. The expression of MMPs was linked with matrix stiffness, resulting in higher MMP expression in FCSNG and FCDNG scaffolds compared to SNG and DNG.

Changes in the MMP2, MMP9, VEGF, and bFGF expression of cells. (a) MMP2 staining of DU-145 cells. MMP2 and MMP9 expression of (b) DU-145 cells and (c) MCF-7 cells. (d) VEGF staining of DU-145 cells. VEGF and bFGF expression of (e) DU-145 cells and (f) MCF-7 cells.
Angiogenesis is necessary for tumors to provide nutrients for growth and metastasis. VEGF and bFGF are known to positively regulate angiogenesis. The results showed that cells cultured in 3D scaffold showed upregulated levels of VEGF and bFGF expression compared to 2D culture (Figure 7(d)–(f) and Supplemental Figure S17b). It was further observed that VEGF and bFGF expression was upregulated in DNG and FCDNG compared to SNG and FCSNG after the addition of PRP. This outcome can be attributed to the synergistic effect of the abundant growth factors in PRP and the 3D microenvironment built by fiber-hydrogel. VEGF can directly target tumor cells, especially cells CSCs, thereby promoting tumorigenesis, progression, and recurrence. Therefore, the scaffolds can imitate the in vivo tumor microenvironment and stimulate tumor angiogenesis.
MMPs aid in angiogenesis, infiltration of cancer cells, metastasis, along with EMT, and that MMPs promote tumor growth and stimulate neovascularization to help tumor cells spread. 71 Malignant tumors typically demonstrate elevated VEGF expression with a coinciding increase in VEGF secretion in vivo, which subsequently promotes breast cancer cell survival and invasiveness. Furthermore, bFGF secretion induces endothelial cell migration and stimulates the expression of ECM remodeling enzymes in vivo. These results demonstrate the positive impact of the FCDNG microenvironment on secretion of MMP, VEGF, and bFGF, which can be utilized to model tumorigenic phenotypes like tumor migration.
Responses to treatment in spheroid-containing FCDNG and potential mechanisms
To evaluated the drug resistance of the composite scaffold, different concentrations of doxorubicin (DOX, nucleic acid formation inhibitors), paclitaxel (a cell mitotic arrest inducer drug) were added to the culture medium, and drug resistance was assessed after 72 h of incubation. Compared to the 2D culture, the IC50 values for DOX in DU-145 and MCF-7 cells cultured in FCDNG was 10.22-fold and 16.22-fold higher, respectively (Figure 8(a) and Supplemental Table S3). Increased resistance was significant in MCF-7 cells on the FCSNG and FCDNG as well. The effect of the drug inhibition in the 3D cultures appeared to be less marked than that in the 2D cultures, which is consistent with previous reports. 72 In addition, the IC50 value of 3D MCF-7 spheroids cultured in fibrin was found to be 13 times higher than that of the 2D culture. 73 This is closer to our results. However, the diameter of the spheroids cultured in fibrin was 1.4 mm, which was much larger than the spheroids obtained in this study, and highlighting the positive impact of fibers in the drug resistance of spheroids. This is because the cell-cell tight interaction in MCTS constitutes an infiltration barrier, as evidenced by the live-dead staining results as well (Figure 8(b)). The situation similar to the real tumor environment. Whereas 2D cultured cells do not have such a microenvironment, the cells can immediately take up the same dose of drug. The similar results were found in the cellular resistance test to paclitaxel (Figure 8(c)). At the maximum paclitaxel concentration utilized in the experiment, DU-145 cell viability on FCSNG, DNG, and FCDNG scaffolds was 56.19%, 53.08%, and 56.39%. The corresponding MCF-7 cell viability on FCSNG, DNG, and FCDNG scaffolds was 60.59%, 62.89%, and 64.89%, respectively (Figure 8(a) and (c), Supplemental Table S4). The results suggests that the addition of fiber and PRP can further increase the resistance of MCTS. Previous studies have shown that drug uptake within gel is unrealistically high, resulting in uptake values similar to those in 2D cell cultures. 25 Thus, we conclude that cellular responses in FCSNG and FCDNG are more physiological than gel. Limited penetration of therapeutic agents in solid tumors is a potential cause of drug resistance. The amplified resistance in composite scaffolds may also be due to increased stiffness. Research has indicated that tumor cells grown on rigid substrates are more resistant to conventional chemotherapeutic drugs than cells cultured on soft 3D substrates. 74 In addition, the introduction of PRP increased the proportion of CSCs in MCTS, resulting in higher drug resistance of tumor cells in FCDNG and DNG compared to FCSNG and SNG. These results collectively indicate that FCDNG is a competent tissue-mimicry culture system suitable for anticancer drug screening.

Comparison of the sensitivities of the cancer cells grown on the different scaffolds to anticancer drugs. (a) Cell viability after drug treatment. Typical live-dead staining images of (b) DU-145 cells and (c) MCF-7 cells after DOX drug treatment.
Many studies have shown that drug resistance in MCTS in 3D culture systems involves multiple intracellular cascade responses that elicit the expression and activation of key proteins such as integrin β1, PI3K, AKT, ERK, and YAP. Integrin β1 is a major regulator affecting cell-ECM interactions and can serve as a mediator of tissue sclerosis-induced drug resistance in tumor cells by mediating cellular responses to ECM stiffness. 75 Integrin β1 can interact with growth factor receptors to activate the growth factor signaling pathway, promoting tumor cell proliferation in situ. To explore the differences in drug sensitivity observed between 3D scaffolds, the signaling within tumor cells on different scaffolds was evaluated. Immunofluorescence staining was used to assess the expression of Integrin β1 (Figure 9(a)). The results showed that the 3D scaffold exhibited higher fluorescence intensity than 2D. Comparison of DNG and FCDNG revealed that Integrin β1 intensity was higher which may be the difference in stiffness caused by nanofibers (Figure 9(b)). Comparison of FCSNG and FCDNG showed that Integrin β1 intensity was higher in FCDNG, which could be a combination of hardness and growth factors caused by presence of ECM-derived components.

Expressions of proteins in the heart sections in different groups through immunostaining and western-blot analysis. (a) Immunofluorescence staining and (b) relative fluorescence intensity of integrin β1. (c) Immunofluorescence staining, (d) the activated cells and (e) the relative gene expression of YAP. (f) Western-blot analysis of p-PI3K, PI3K, p-Akt, and Akt. (g) Quantitative analysis of p-PI3K, PI3K, p-Akt, and Akt assessed using Image J software to analyze the western blot results.
YAP acts as an effector molecule of the Hippo pathway, which is activated by translocation from the cytoplasm to the nucleus to regulate gene expression and promote tumorigenesis. YAP is often involved in the resistance patterns of multiple antitumor therapies, including the induction of resistance to paclitaxel and DOX in tumor cells. 76 To this end, this study first analyzed the expression of YAP in different culture systems by immunofluorescence (Figure 9c). The results showed that the percentage of YAP-activated cells in 3D was relatively low due to the formation of MCTS, where only the cells in the outer layer of the spheroid were in an active proliferative state (Figure 9(d)). However, the percentage of YAP-activated cells was also higher in FCSNG and FCDNG than in DNG due to the presence of more CSCs with self-renewal ability, and among them, FCDNG was even higher than 2D (Figure 9(e)). It further suggests that both the ECM structure and composition in FCDNG could play a positive role in the proliferation of tumor cells.
In addition, we used western blotting to analyze the signaling pathways associated with drug resistance in tumor cells. PI3K/AKT/mTOR signaling pathway is one of the major signaling pathways regulating cancer cell proliferation and apoptosis. 77 As shown in Figure 9(f) and (g), both AKT and p-AKT protein expression in 3D scaffolds were significantly higher than that in 2D. AKT and p-AKT protein expression was gradually increased in group of DNG, FCSNG, and FCDNG. These results suggest that the addition of nanofiber and ECM components can promote cell proliferation by increasing the activation of the PI3K/AKT/mTOR signaling pathway. ECM stiffness also induces epithelial tumor cell growth through the Rho/ERK signaling pathway. 78 In addition, the expression of p-ERK, ERK protein was relatively increased in DNG, FCSNG, and FCDNG. It suggests that increased scaffold stiffness can activate the Rho/ERK pathway to trigger the mechanical properties of cancer cell behavior, further up-regulate MMP expression and contribute to promote EMT.
In summary, it can be assumed that the drug resistance mechanism of tumor cells in 3D scaffolds may be enhanced through the following aspects (Figure 10). Firstly, non-cell-autonomous mechanism. FCDNG scaffolds have ECM structure and components, which prompt tumor cells to form MCTS, and the MCTS formed a permeability barrier, which makes it difficult for chemotherapeutic drugs to enter the core of the tumor spheres. Besides, changes in cellular phenotypes. MCTS has a significantly enhanced invasive and CSC phenotype, with superb self-renewal ability and rapid replenishment of tumor cells after chemotherapeutic drug treatment. In addition, intracellular signaling. Through signaling pathways related to matrix stiffness and chemotaxis, the up-regulation of YAP nuclear translocation, SOX2, CDH1, and CDH2 expression, which ultimately leads to increased drug resistance.
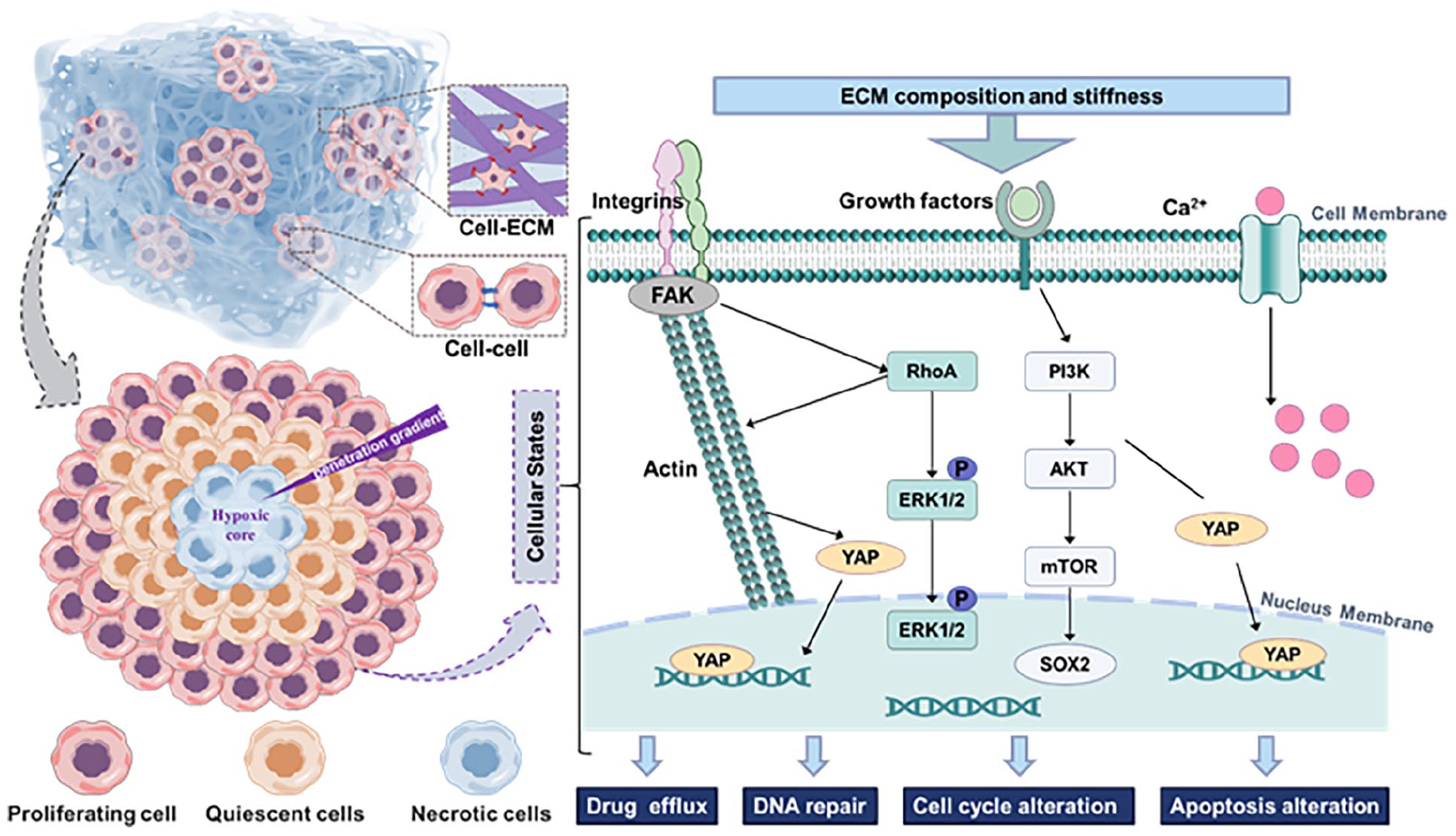
Schematic diagram of signaling responses and potential mechanisms of chemoresistance in tumor cells.
All the above results demonstrated superior tumor cell culture efficiency of FCDNG. The above results also suggest that FCDNG can be used for drug screening and personalized medicine by adjusting the scaffold properties to mimic individual tumor characteristics. However, the FCDNG may not be able to fully simulate the complexity of the in vivo tumor microenvironment. The structure and the bioactive components of the scaffold can be further optimized to Mimics the tumor microenvironment in vivo. Besides, patient-derived PRP could be applied in the scaffold in the future, which may promote the personalized drug screening for cancer patients.
Conclusion
Inspired by the natural tumor ECM, a fiber composite dual-network gel (FCDNG) was prepared by a “dispersion-swelling” method combined with calcium-ion-mediated “one-step” activation, and used for 3D culture of prostate tumor cells and breast tumor cells. Gel was filled in the interstitial space of the fibers to act as a functional mesenchyme and provide active factors. Three-dimensional nanofiber framework dispersed in gel endowed scaffold with fiber components, structural stability, and prolonged the release of active factors. Thus, FCDNG was able to mimic the function of natural ECM by recapitulating both the structural and signaling microenvironment of tumors. FCDNG artificial ECM could improve tumor stemness and form 3D tumor spheroids with excellent structure, cell viability and EMT behavior. Based on the ECM in this study, prostate and breast 3D tumor model exhibited improved drug resistance. These results suggest that the current FCDNG has great potential for application in clinical drug evaluation.
Supplemental Material
sj-docx-1-tej-10.1177_20417314241299941 – Supplemental material for Enhancement structural and signaling microenvironment of artificial tumor extracellular matrix for enhanced drug screening
Supplemental material, sj-docx-1-tej-10.1177_20417314241299941 for Enhancement structural and signaling microenvironment of artificial tumor extracellular matrix for enhanced drug screening by Xingxing Liu, Yueying Ren, Sijia Fu, Yongjie Jiao, Xinan Chen, Fujun Wang, Lu Wang, Mengbo Hu and Chaojing Li in Journal of Tissue Engineering
Footnotes
Acknowledgements
We gratefully acknowledge our colleagues, both locally, nationally, and internationally, who have contributed to numerous discussions, idea exchanges, and collaborations in the field of tumor cell culture over the years.
Author contributions
Xingxing Liu: Writing—original draft, Visualization, Investigation, Data curation. Yueying Ren: Investigation, Data curation. Sijia Fu: Visualization, Data curation. Yongjie Jiao: Data curation. Xinan Chen: Visualization, Data curation. Fujun Wang: Supervision, Resources. Lu Wang: Supervision, Resources, Project administration. Mengbo Hu: Writing—review & editing, Investigation, Data curation, Project administration. Chaojing Li: Writing—review & editing, Supervision, Visualization, Investigation, Data curation, Project administration.
Data availability statement
The data required to reproduce these findings can be made available upon reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work is supported by grants from the National Natural Science Foundation of China (NSFC; 32201080, 81802569), Shanghai Science and Technology Development Fund (21S31900700), China Postdoctoral Science Foundation (2023M732187), the Fundamental Research Funds for the Central Universities (2232023G-01), 111 Project 2.0 (Grant No. BP0719035), and Donghua University 2024 Cultivation Project of Discipline Innovation.
Ethics approval and consent to participate
Not applicable.
Supplemental material
Supplemental material for this article is available online.
