Abstract
Hydrogels, hydrophilic and biocompatible polymeric networks, have been used for numerous biomedical applications because they have exhibited abilities to mimic features of extracellular matrix (ECM). In particular, the hydrogels engineered with electrospinning techniques have shown great performances in biomedical applications. Electrospinning techniques are to generate polymeric micro/nanofibers that can mimic geometries of natural ECM by drawing micro/nanofibers from polymer precursors with electrical forces, followed by structural stabilization of them. By exploiting the electrospinning techniques, the fibrous hydrogels have been fabricated and utilized as 2D/3D cell culture platforms, implantable scaffolds, and wound dressings. In addition, some hydrogels that respond to external stimuli have been used to develop biosensors. For comprehensive understanding, this review covers electrospinning processes, hydrogel precursors used for electrospinning, characteristics of fibrous hydrogels and specific biomedical applications of electrospun fibrous hydrogels and highlight their potential to promote use in biomedical applications.
Biomedical applications of fibrous hydrogels fabricated via electrospinning techniques
There have been efforts to mimic characteristics of extracellular matrix (ECM), three-dimensional (3D) fibrous networks that involve diverse biomolecules and play important roles in various cell behaviors and responses,1–3 with biomaterials for biomedical applications, and one way to mimic ECM is using hydrogels that are hydrophilic polymer networks and amendable through chemical modifications.3,4 For instance, appropriate mechanical properties (stiffness and elasticity) of hydrogels, which might be similar to target tissue, could be determined by variation of hydrogel composition and density.5–7 Additionally, hydrogels could be degraded by cells and adhesive to cells via introducing degradable crosslinkers and adhesive ligands, respectively, to hydrogels.5,8–10 Geometry of ECM, nanofiber complex, has been also regarded as an important feature influencing directional cell migration, 11 cell spreading, 12 and proliferation. 13 Diverse biofabrication techniques such as electrospinning,14,15 solution blowing,16,17 self-assembly,18,19 and drawing20,21 have been developed to fabricate various polymers including hydrogels in fibrous form to mimic ECM structures. Advantages and disadvantages of these techniques used for fabrication of fibrous hydrogels are shown in Table 1. In particular, electrospinning techniques and its variants (co-axial electrospinning, 22 wet electrospinning, 23 and needleless electrospinning 24 ) were utilized to generate diverse types of fibrous hydrogels.
Advantages and disadvantages of techniques for fabrication of fibrous hydrogels.

Electrospinning is a technique to produce polymeric micro/nanofibers and have been used to generate fibers with various polymers for diverse applications. Typically, diameters of electrospun fibers, controlled by electrospinning setup (voltage applied to a needle, flow rate of precursors, and distance between a needle and a collector)32–35 and viscosity of precursors, 36 vary from some 10 nm to 10 µm, 37 and various approaches (rotating collector, gap electrospinning, and magnetic field-assisted electrospinning) were developed to manipulate alignment of fibers.38–41 These micro/nanofibers were electrospun with various materials such as poly (ε-caprolactone) (PCL),42–44 polylactic acid (PLA),45,46 poly (D, L-lactic-co-glycolic) acid (PLGA),47–50 polyurethane (PU),51–53 and polyacrylonitrile (PAN)54,55 and used for drug delivery, wound dressings, and vascular graft applications.
In particular, electrospun micro/nanofibers fabricated with hydrogels (methacrylated dextran (DexMA), gelatin methacryloyl (GelMA), and poly (vinyl-alcohol) (PVA)) have been used for various biomedical applications because they had potential to mimic ECM and their characteristics could be expanded by variation of hydrogel types. For example, electrospun fibrous hydrogels soft enough for cells to deform were used to study cell behaviors and responses.56–60 Fibrous hydrogels cells could adhere were developed as scaffolds and used to regenerate tissues.61–67 Degradable fibrous hydrogels involved drugs and biomolecules and delivered them through degradation of micro/nanofibers after implantation.61,64,65 In addition, electrospun fibrous hydrogels that could exhibit pH-sensitive volume expansion 68 or glucose-sensitive enzymatic activities 69 were used to develop biosensors.
To promote use of electrospun fibrous hydrogels for biomedical applications, this review focuses on fabrication of electrospun fibrous hydrogels and their biomedical applications. Specifically, general electrospinning setup, hydrogel precursor types, and hydrogel crosslinking methods were included to provide information on fabrication methods and types of fibrous hydrogels already used for biomedical applications. Additionally, their biomedical applications such as cell culture platform used for cell-related studies, implantable scaffolds to regenerate tissue after implantation, wound dressings to recover wounded skins, and biosensors to sense pH or glucose levels (Figure 1) were described for further applications to biomedical fields.

Crosslinking methods for fibrous structure stabilization and biomedical applications (cell culture platforms, implantable scaffolds, wound dressings, and biosensors) of electrospun fibrous hydrogels.
Fabrication of electrospun fibrous hydrogels
Electrospinning techniques have been used to generate micro/nanofibers that were made of diverse polymers including hydrogels and exploited for numerous biomedical applications. In general, electrospinning setup is composed of a power supply to apply voltage to a metal needle, a collector grounded, and a syringe pump to constantly supply polymer precursors through the metal needle in a controlled flow rate (Figure 2(a)). At a constant pumping of polymer precursors with application of voltage to a needle, a droplet of polymer precursors, formed at a needle tip, is stretched out by electrostatic repulsion applied to the droplet and ejected as micro/nanofibers toward the grounded collector when the electrostatic repulsion overcomes surface tension of the droplet (Figure 2(b)).70,71 Solvents of precursors evaporate during flight of the liquid jet, and solid fibers are finally collected on the collector. Additionally, there are modified electrospinning techniques to fabricate various fibrous hydrogels with hydrogel precursors (Figure 3). For instance, co-axial electrospinning technique was used to generate fibers with two different materials separated at the core and the sheath of the fibers. In wet electrospinning, grounded collector was immersed in a coagulation bath to fabricate fibrous hydrogel mats with relatively low fiber packing density. Needless electrospinning setup could generate multiple jets to increase productivity of hydrogel fibers. Advantages and disadvantages of these electrospinning techniques are described in Table 2. Typically, fibers in diameters of some hundred nanometers were obtained, and the diameter can be controlled by manipulating voltage, distance from a needle to a collector, and flow rate and viscosity of polymer precursors.36,72,73 Additionally, thickness74,75 and porosity76,77 of fibrous mat and alignment 78 of fibers can be controlled by various approaches.

A schematic illustration of general electrospinning setup and processes. (a) General electrospinning setup. It is mainly composed of a syringe pump ejecting hydrogel precursors in a controlled flow rate, a power supply to apply voltage to a metal needle and a collector to collect polymeric fibers. (b) Overall processes to generate electrospun fibers. A droplet of polymer precursor at a needle tip is ejected toward a grounded collector when electrostatic repulsion exceeds surface tension of the droplet. Reproduced from Han et al., 79 Chen et al., 65 and Marginean et al. 80
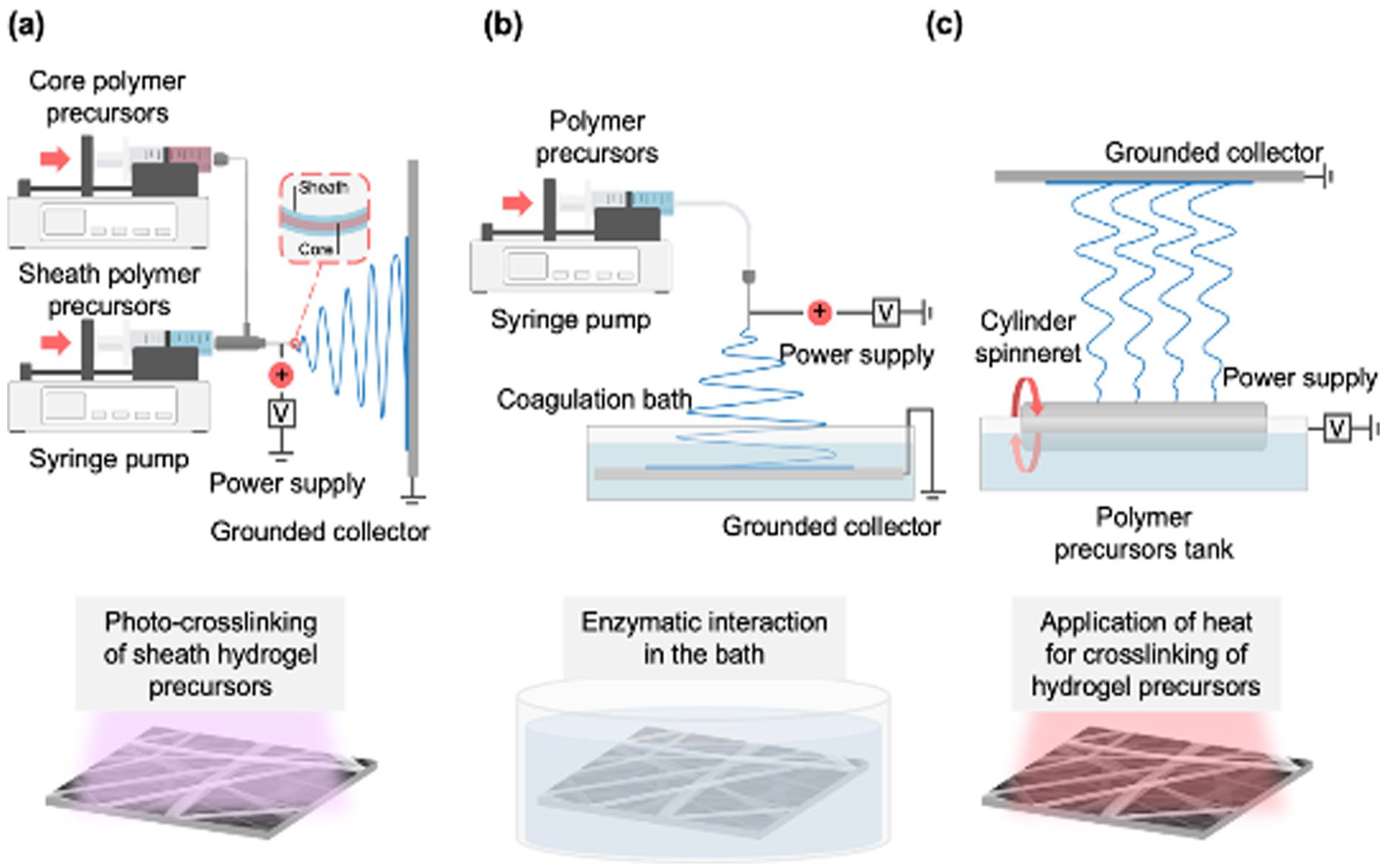
Various types of electrospinning techniques with crosslinking steps used for fabrication of electrospun hydrogel fibers. A power supply and a grounded collector are commonly required for electrospinning techniques: (a) co-axial electrospinning technique uses a co-axial needle to obtain fibers composed of one material at the core and another one at the sheath, (b) wet electrospinning technique uses a coagulation bath containing a grounded collector to fabricate fibrous hydrogel mats with relatively low fiber packing density, and (c) needleless electrospinning technique generates multiple jets to increase productivity of fibrous hydrogels. Reproduced from Chen et al. 65
Advantages and disadvantages of the electrospinning techniques used for fabrication of fibrous hydrogels.

In general, hydrogel precursors loaded into syringes are electrospun to collect dried micro/nanofibers, and crosslinking steps are followed for stabilization of fibrous structures. For biomedical applications, diverse types of hydrogel precursors have been used to generate fibrous hydrogels with various characteristics (Table 3). Specifically, photo-crosslinkable hydrogel precursors such as precursors containing MeHA, 58 NorHA, 60 HEMA-HA, 61 GelMA,63,64 or γ-PGA-Nor/γ-PGA-SH 66 with photoinitiators and with/without crosslinkers have been exploited for fabrication of fibrous hydrogels. As photoinitiator, Irgacure 2959 has been widely used. 84 Other than that, POH/POA precursors and PVA/CS precursors were crosslinked through in situ crosslinking.59,65 Gel-HPA 57 and fibrinogen 62 precursors were crosslinked through enzymatic reaction by HRP with H2O2 and thrombin, respectively. Gelatin/PLGA precursor was crosslinked through coupling reaction of EDC/NHS. 67 Lastly, PVA/PAA and PVA/β-CD precursors were crosslinked through heat treatment.68,69 Additionally, viscoelasticity is an important property of the precursors to generate electrospun micro/nanofibers, and PEO, water-soluble and biocompatible polymer, are often added to the precursors to meet viscoelasticity proper to implement electrospinning.
Composition of hydrogel precursors used for electrospinning, crosslinking methods for the electrospun micro/nanofibers, diameter of fibers, and their biomedical application.
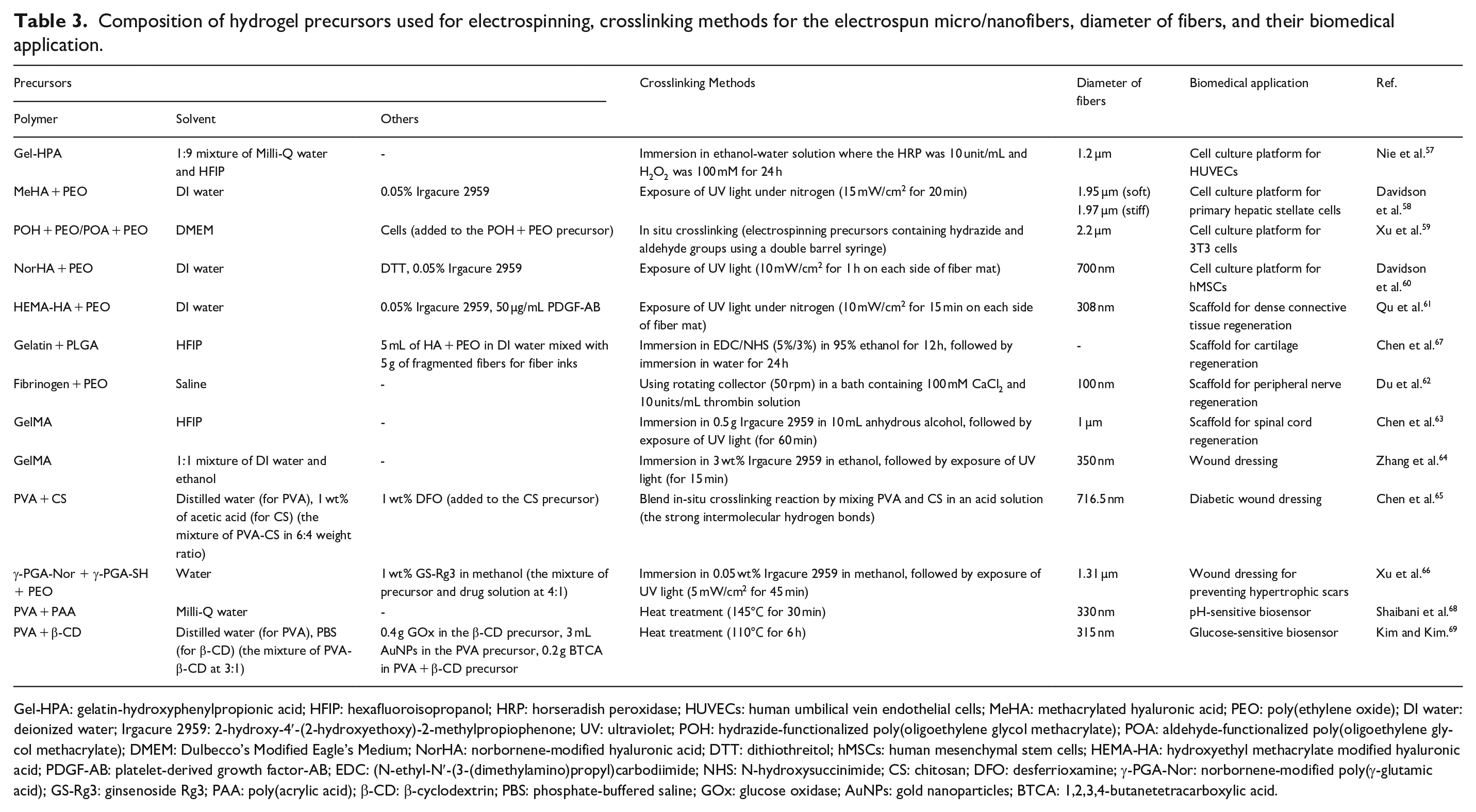
Gel-HPA: gelatin-hydroxyphenylpropionic acid; HFIP: hexafluoroisopropanol; HRP: horseradish peroxidase; HUVECs: human umbilical vein endothelial cells; MeHA: methacrylated hyaluronic acid; PEO: poly(ethylene oxide); DI water: deionized water; Irgacure 2959: 2-hydroxy-4′-(2-hydroxyethoxy)-2-methylpropiophenone; UV: ultraviolet; POH: hydrazide-functionalized poly(oligoethylene glycol methacrylate); POA: aldehyde-functionalized poly(oligoethylene glycol methacrylate); DMEM: Dulbecco’s Modified Eagle’s Medium; NorHA: norbornene-modified hyaluronic acid; DTT: dithiothreitol; hMSCs: human mesenchymal stem cells; HEMA-HA: hydroxyethyl methacrylate modified hyaluronic acid; PDGF-AB: platelet-derived growth factor-AB; EDC: (N-ethyl-N′-(3-(dimethylamino)propyl)carbodiimide; NHS: N-hydroxysuccinimide; CS: chitosan; DFO: desferrioxamine; γ-PGA-Nor: norbornene-modified poly(γ-glutamic acid); GS-Rg3: ginsenoside Rg3; PAA: poly(acrylic acid); β-CD: β-cyclodextrin; PBS: phosphate-buffered saline; GOx: glucose oxidase; AuNPs: gold nanoparticles; BTCA: 1,2,3,4-butanetetracarboxylic acid.
Characteristics of electrospun fibrous hydrogels
Fibrous hydrogels fabricated with various electrospining techniques exhibit distinct characteristics compared to those of bulk hydrogels, due to the difference in scale and structure of those hydrogels (Figure 4). In general, diameters of fibrous hydrogels obtained through electrospinning techniques were in micro/nanoscales while hydrogels in millimeter to centimeter scales were defined as bulk hydrogels. 85 Overall structures of bulk and fibrous hydrogels can be determined by various fabrication techniques. 3D printing,86,87 lithography, 88 and molding89,90 techniques allowed fabrication of bulk hydrogels in user-defined and complex structures. Likewise, double layers of fibrous hydrogels were obtained by electrospinning two different materials successively,59,91 and bundles62,63 and a hollow conduit 92 were prepared by rolling electrospun fibrous hydrogel mats. Additionally, fibrous hydrogels in user-defined 3D structures were formed by 3D printing of jammed fragmented fibrous hydrogels. 60
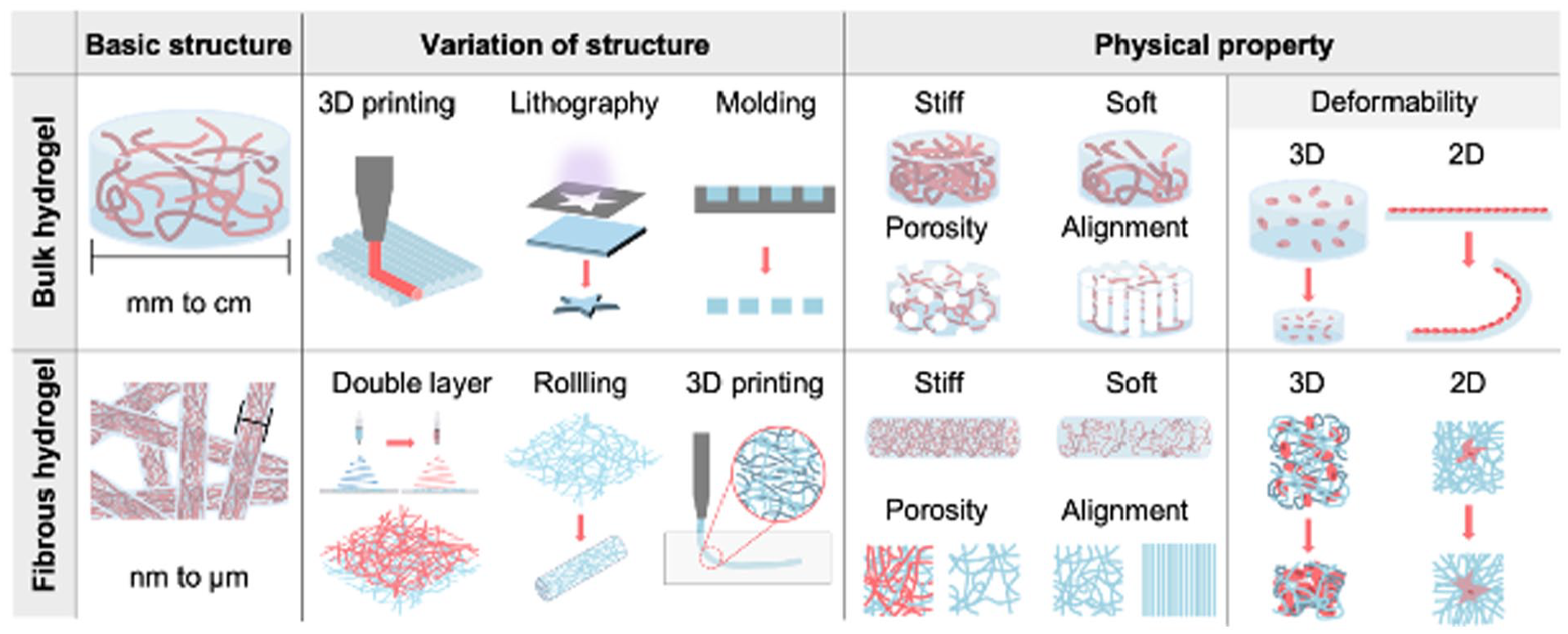
Comparison between characteristic of bulk hydrogel and fibrous hydrogel. Bulk hydrogels were referred to hydrogels in millimeter to centimeter scales, and diameters of electrospun fibrous hydrogels were generally in nano/micrometer scales. Fabrication methods could determine overall shapes of bulk and fibrous hydrogels. Physical properties (Stiffness, porosity, alignment and deformability) of fibrous hydrogels might induce cellular behaviors different from cellular behaviors influenced by those properties of bulk hydrogels.
Physical properties of bulk and fibrous hydrogels, such as stiffness, porosity, alignment, and deformability, can possibly affect various cellular behaviors in different manners. For instance, elevation of fibrous hydrogel stiffness decreased cellular proliferation and spreading while elevation of bulk hydrogel stiffness increased the proliferation and spreading.56,58 Similarly, porosity and alignment of fibrous hydrogels, different from those of bulk hydrogels in scale and structure, might induce cellular behaviors that are different from cellular behaviors influenced by pores and aligned structures of bulk hydrogels. Due to the different scale and structure of bulk and fibrous hydrogels, deformability of those hydrogels was also different to each other. Through the cellular contraction, a bulk hydrogel in a cylindrical shape shrank and a hydrogel membrane was rolled.93–95 On the other hand, cells on and within fibrous hydrogels significantly deformed adjacent fibrous hydrogels irregularly56,60 although overall structures of fibrous hydrogels might be also deformed like bulk hydrogels (It was shown that cells within fibrous hydrogels changed overall fibrous hydrogel shapes via contraction 60 ). These all imply that cellular behaviors on and within fibrous hydrogels can be different from cellular behaviors on and within bulk hydrogels. Specifically, 3T3 fibroblast cells cultured within fibrous POA + POH hydrogels showed higher viability than cells cultured in flat bulk hydrogels (Figure 5(a)). 59 Differentiation of myofibroblasts in bulk dextran vinyl sulfone (DexVS) hydrogels was decreased with elevation of hydrogel stiffness while fibroblasts in the hydrogels containing fibrous hydrogels were primed for activation into myofibroblasts via interaction with fibrous hydrogels (Figure 5(b)). 15 Additionally, human pulmonary artery smooth muscle cells (hPASMCs) on fibrous hydrogels infiltrated deeper than cells on bulk hydrogel platforms (Figure 5(c)). 96 Possibly, fibrous hydrogels can be useful in biomedical applications because relatively high cell viability and infiltration are expected from fibrous hydrogels. The fibrous hydrogels exhibiting distinct features were used to study various cellular behaviors although a few systemic comparisons between cell behaviors with bulk and fibrous hydrogels were implemented. Specifically, spreading (Figure 6(a)),56,58 infiltration (Figure 6(b))46,97 cell viability (Figure 6(c))57,59,98 and alignment (Figure 6(d))62,63 were studied with fibrous hydrogel environments.

Comparisons of cellular behaviors on/within bulk and fibrous hydrogels. (a) Live/dead staining after 7 days of 3T3 cell culture in bulk hydrogels (top) and within fibrous hydrogels (bottom) (Scale bar: 200 µm). Reproduced from Xu et al. 59 Myofibroblast differentication study in soft/stiff bulk hydrogels (top, scale bar:200 µm) and in hydrogels with 0.0% and 5.0% fiber density (bottom, FD: fiber density, scale bar: 100 µm). Stiffness of hydrogels was controlled by controlling concentration of crosslinkers (GCVPMS↓MRGGCG, VPMS). Reproduced from Matera et al. 15 Distributed under the Creative Commons Attribution NonCommercial License 4.0 (CC BY-NC, https://creativecommons.org/licenses/by-nc/4.0/). (c) Infiltration thickness of hPASMCs on bulk (left) and fibrous (right) hydrogels after 1 day of seeding. Reproduced from Ding et al. 96

Cells on and within electrospun fibrous hydrogels were observed to investigate various cellular behaviors and responses. (a) Cell spreading on soft (top) and stiff (bottom) fibrous hydrogels after 7 days of cell culture. Reproduced from Davidson et al. 58 (b) Cell infiltration through fibrous hydrogels after 3 days of cell culture. Reproduced form Wang et al. 97 (c) Viability of cells on fibrous hydrogel membranes. Reproduced from Sun et al. 98 (d) Cells on aligned fibrous hydrogels (top) and randomly oriented ones (bottom) after 1 day of cell culture. Reproduced from Du et al. 62
Taken together, electrospun fibrous hydrogels exhibit distinct characteristics compared to those of bulk hydrogels and were used for investigation of cellular behaviors.
Biomedical applications
In vitro cell culture platforms mimicking in vivo fibrous environments
2D platforms to study cellular behaviors
Diverse cell culture platforms have been developed and utilized for various applications. For example, traditional platform such as cell culture dishes providing wide and flat reservoirs to contain cells and cell culture media with relatively easy handling have been widely used although their mechanical properties and geometry that can possibly influence cell behaviors and fate are different from in vivo microenvironments. To meet needs of improvement in cell culture platforms, hydrogels to mimic ECM and microfabrication techniques to create in vivo-like microenvironments were applied to develop the platforms. Cells were grown on various hydrogels,99–105 and cell differentiation and migration were studied by analyzing cells on hydrogels with stiffness alteration.106,107 Microfluidic chips providing in vivo-like cell culture platforms were also used to study various cellular mechanisms108–111 and even developed as organ-on-a-chip by mimicking features of specific organs.112,113
Alternatively, electrospun fibrous hydrogels sheets that have ECM-like topography and potential to mimic various ECM features have been used for 2D cell culture and cell behavior studies.114,115 For instance, fibrous gelatin (relatively stiff) and Gel-HPA (relatively soft) hydrogel microfibers were fabricated by electrospinning and human umbilical vein endothelial cells (HUVECs) were cultured on each hydrogel microfiber scaffold for 3 days (Figure 7(a)). 57 Higher cell viability and more cell spreading were observed on Gel-HPA fibers in comparison to the gelatin fibrous hydrogels, indicating that soft fibrous environments can enhance the cell adhesion and cell spreading.

2D cell cultures on electrospun fibrous hydrogels. (a) Live/dead staining (left, middle) and F-actin staining (right) of human umbilical vein endothelial cells (HUVECs) cultured on fibrous gelatin and Gel-HPA hydrogels on Day 1 and 3. Fibrous gelatin hydrogel was stiffer than fibrous Gel-HPA hydrogels. Reproduced from Nie et al. 57 Distributed under the Creative Commons Attribution 4.0 International License (CCBY 4.0, http://creativecommons.org/licenses/by/4.0/). (b) Activated stellate cells on soft/stiff MeHA hydrogel nanofibers. Representative masks of cell outlines on soft/stiff nanofibers (left, scale bar: 50 µm) and fluorescent images of soft/stiff nanofibers under the cells (right, scale bar: 50 µm). Reproduced from Davidson et al. 58
Activated stellate cells were also cultured on soft and stiff MeHA hydrogel nanofibers functionalized with adhesive ligands, and their spreading area and phenotype were observed (Figure 7(b)). 58 The cells on soft nanofibers displayed larger spreading than cells on stiff nanofibers, and phenotype of the cells became quiescent on stiff nanofibers. In addition, soft and stiff DexMA hydrogel microfibers coupled with adhesive ligands were prepared via electrospinning of precursors and variation of crosslinking degree and used to study how geometry and stiffness of microfibers affect adhesion area and proliferation of human mesenchymal stem cells (hMSCs). 56 For comparison, spreading area and proliferation of those cells on type Ⅰ collagen matrices with low and high concentration were also analyzed. On bulk hydrogels, spreading area and proliferation of hMSCs increased with an elevation of hydrogel stiffness. On the contrary, cell on fiber networks exhibited decreased spreading area and proliferation with a stiffness increase of fiber networks, similar to a trend observed from cell on collagen matrices.
These all imply that topography and stiffness of fibrous hydrogel sheets can greatly influence cells and need to be considered for cell cultures.
3D platforms to encapsulate cells within fibrous environments
Electrospun fibrous hydrogels in sheet forms have been used as 2D cell culture platforms in various studies because they were relatively easy to fabricate and handle. Those fibrous sheets were generally obtained by tightly stacking numerous electrospun fibers on collectors. The stacked micro/nanofibers often interfered cell migration through the fibrous sheets, 116 which could a huddle for them to be used as a 3D cell culture platform. In order to create fiber-based 3D microenvironments that can encapsulate cells for cell culture, strategies electrospinning precursors containing cells 59 and fabricating structures with fragmented nanofiber suspensions involving cells 60 were suggested.
To encapsulate 3T3 mouse fibroblast cells within electrospun hydrogel nanofibers, POH and PEO precursors including cells and POA and PEO precursors were electrospun together through a double barrel syringe with a relatively low voltage, 10 kV (Figure 8(a)). 59 Subsequently, electrospun nanofibers were stabilized via aldehyde and hydrazide interaction, and immediately transferred into cell culture media for cell culture. Viabilities of cells encapsulated in bulk hydrogel and fibrous hydrogel were also analyzed on day 3 and 7, and cells within fibrous environment exhibited higher viabilities than cells in bulk hydrogels (Figure 7(b)).

3D cell culture within fibrous hydrogels. (a) A schematic illustration of processes to encapsulate 3T3 mouse fibroblast cells within fibrous hydrogels. (b) 3D confocal images of the cells encapsulated in bulk hydrogel (left) and electrospun hydrogel nanofibers (right) after 3 and 7 days of incubation (scale bar: 200 µm). Reproduced from Xu et al. 59 (c) Representative fluorescent images of NorHA nanofibers with varied nanofiber densities (75%, 50%, and 20%) (scale bar: 10 µm). (d) Representative inverted phase-contrast images of mesenchymal stem cells (MSCs) encapsulated within nanofibers on day 1 and 3 with varied nanofiber densities (75%, 50%, and 20%) (scale bar: 2 mm). (e) A schematic illustration (top) and representative images (bottom) of 3D-printed cell-laden nanofibers with 75% (gray) and 20% (pink) nanofiber densities (scale bar: 1 mm). Reproduced from Davidson et al. 60 Distributed under the Creative Commons Attribution NonCommercial License 4.0 (CCBY-NC, https://creativecommons.org/licenses/by-nc/4.0/).
Alternatively, nanofiber suspensions were prepared by fragmenting electrospun NorHA nanofibers and suspending them with varied fiber density (Figure 8(c)), 60 and cells were encapsulated and cultured within nanofiber-based structures by forming and crosslinking nanofiber suspensions containing cells (Figure 8(d)). Interestingly, the overall structures containing MSCs with 20%, 50%, and 70% nanofiber densities contracted overtime through interaction between crosslinked nanofibers and the cells, and degree of contraction increased with a decrease of nanofiber density. Exploiting the differences in contraction degree, curved membranes were also fabricated (Figure 8(e)). Specifically, the suspensions containing cells with 20% and 70% nanofiber densities were extruded within a support bath to form relatively flat and layered membranes, and the membranes were curved through the differences in contraction degree on day 1. This research demonstrated that 3D fibrous cell culture platforms could be fabricated with nanofiber suspensions and the suspensions had a potential to generate complex structures similar to tissues and organs.
Taken together, cells were cultured well within electrospun fibrous hydrogel-based 3D platforms, and further studies regarding cell behaviors within these platforms and development of platform in tissue-like structures are expected.
Scaffolds for tissue engineering
Implantable scaffolds for tissue regeneration
Electrospun hydrogel micro/nanofibers have been also applied to develop scaffolds that provide tissue-like structures to support tissue regenerations, exploiting their ECM-like geometries and abilities to mimic mechanical and chemical characteristics of local tissues.61–63,117
For example, HEMA-HA and PEO nanofibers together with PCL nanofibers were used to present strategies to regenerate knee meniscus (Figure 9(a)). 61 Specifically, the strategies included controlled release of biomolecules. PEO nanofibers containing collagenase dissolved rapidly within tissue to improve cell migration through scaffolds by enlarging pores at scaffolds and implement burst delivery of collagenase, followed by a sustained delivery of PDGF-AB involved in HEMA-HA nanofibers. Rapid release of collagenase was to improve local cell migration via degrading tissue around implantation site, and relatively slow release of PDGF-AB was to recruit cells around the scaffolds. As a result, the scaffolds releasing collagenase and PDGF-AB improved cell migration through scaffolds, expecting rapid regeneration of meniscus tissues (Figure 9(b)).

Implantable scaffolds developed with fibrous hydrogels for dense connective tissue regeneration. (a) A scheme of knee meniscus regeneration strategy using PEO and HA nanofibers releasing collagenase and PDGF-AB, respectively. (b) H&E staining and collagen type Ⅰ and Ⅱ immunostaining of the wounded meniscus tissues without scaffolds and with scaffolds containing PDGF-AB only and PDGF-AB with collagenase after 4 weeks of subcutaneous implantation (scale bar: 100 µm). Reproduced from Qu et al. 61 Distributed under the Creative Commons Attribution 4.0 International License (CCBY, http://creativecommons.org/licenses/by/4.0). (c) Electrospun gelatin/PLGA fibers and (d) the fragmented fibers. (e) Scaffolds 3D printed with gelatin/PLGA hydrogel fibers. Analysis of (f) GAG content and (g) total collagen content for evaluation of cartilage regeneration after 8 weeks of implantation. Reproduced from Chen et al. 67
Similarly, gelatin/PLGA fibers obtained by electrospinning (Figure 9(c)) 67 were used to regenerate cartilages. Specifically, the fibers were fragmented by centrifuging fibers in tert-butanol (Figure 9(d)) and kneaded in HA and PEO solution to form 3D printable inks. Then, scaffolds were fabricated by molding with the inks and freeze-drying (3DF) and the other scaffolds with relatively large pores and high porosity were fabricated by 3D printing with the inks and freeze-drying (3DP). The scaffolds were implanted in cartilages of mice and evaluation of cartilage regeneration was implemented after 8 weeks. As a result, glycosaminoglycan (GAG) and total collagen content in the 3DP group were significantly higher than those in the 3DF group (Figure 9(f) and (g)). This result implied that cartilages in the 3DP group were regenerated more than those in 3DF group because of relatively high porosity and large pores in 3DP group.
Additionally, aligned fibrin hydrogel nanofibers (AFG) were developed as scaffolds to regenerate peripheral nerves (Figure 10(a)). 62 Larger gap at peripheral nerve often requires autograft to relay signals to avoid disorders in motor function and sensory perception. Instead of autograft, strategies using aligned nanofibers to guide axonal regeneration eventually for bridging the large gap were introduced in this research. AFG and randomly oriented fibrin hydrogel nanofibers (RFG) were implanted into 10 mm-nerve gaps at left femur of rats, and the AFG and autograft groups showed few hyperplastic collagen fibers in comparison to the RFG groups after 12 weeks of implantation (Figure 10(a)). Likewise, density, mean diameter of myelinated nerve fibers and thickness of myelin sheath were similar in both groups, implying that AFG could replace autograft in regeneration of peripheral nerves (Figure 10(b)).
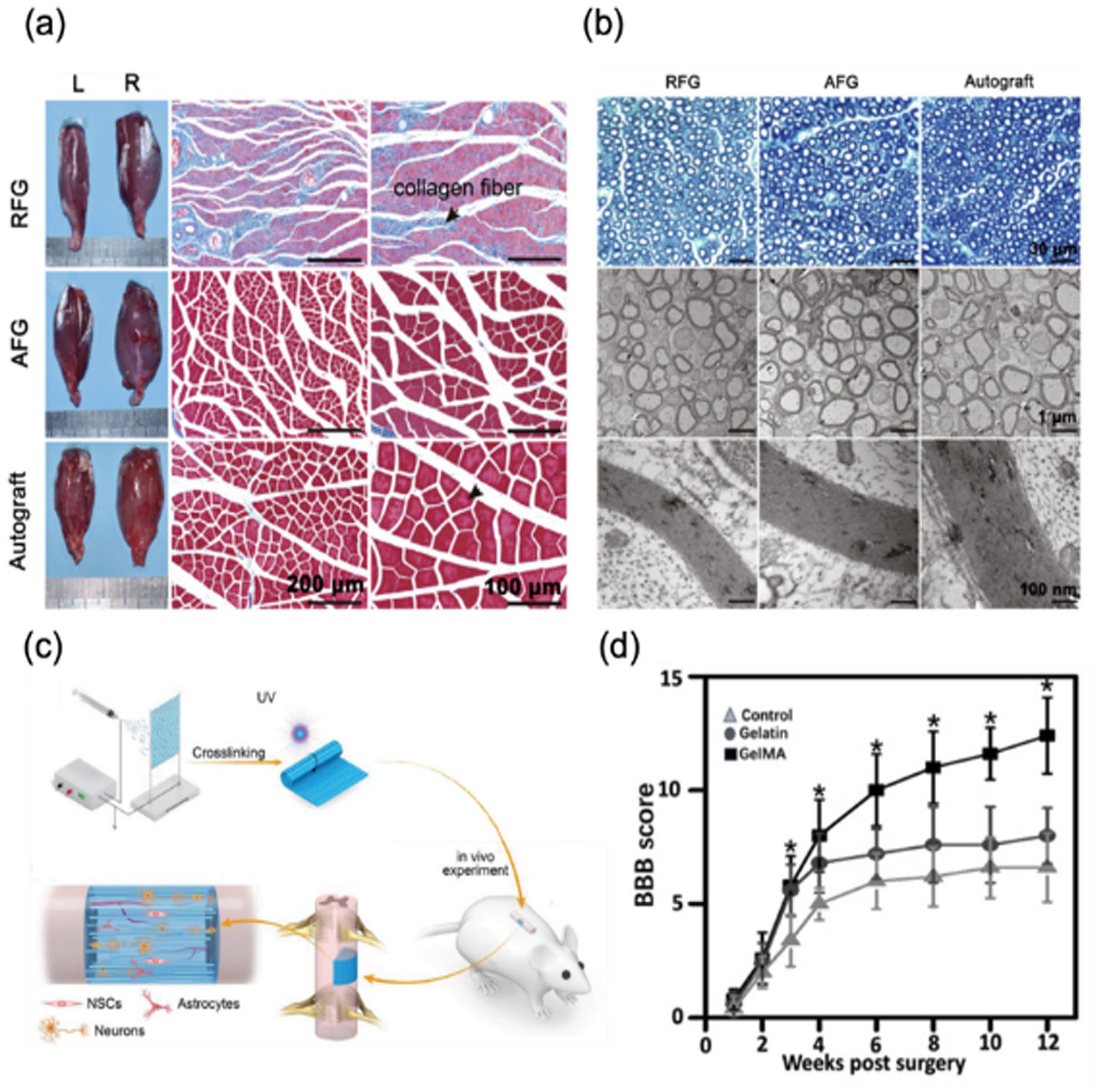
Implantable scaffolds developed with fibrous hydrogels to guide axonal regeneration. (a) Reinnervation of gastrocnemius at 12 weeks after implantation (left limb). Pictures of the isolated muscle and images of Masson’s trichrome staining of the sectioned muscles from the injured limbs. RFG: randomly oriented fibrin hydrogel nanofibers, AFG: aligned fibrin hydrogel nanofibers. (b) Images of regenerated nerve fibers isolated from middle of the implanted site at 12 weeks after surgery. Toluidine blue-stained transverse sections (top) and transmission electron microscopy (TEM) images (middle and bottom) of the regenerated sciatic nerve. Reproduced from Du et al. 62 (c) Processes of spinal cord injury repair study using fibrous GelMA scaffold. (d) Evaluation of rat limb motor function with Basso, Beattie, and Bresnahan (BBB) score after implantation of the GelMA scaffold. Reproduced from Chen et al. 63
Similarly, aligned GelMA microfibers obtained by electrospinning with customized parallel electrodes were developed as scaffolds to repair spinal cord (Figure 10(c)). 63 Promoted cell adhesion and guided directional growth were expected from microstructures of the scaffolds, and the scaffolds showed high cell viabilities. After 12 weeks of scaffold implantation to right hemisected spinal cord defect of rats, motor function recovery of hind limb was evaluated with the Basso, Beattie, and Bresnahan (BBB) locomotor rating scale (Figure 10(d)). Compared to a control groups, the GelMA groups exhibited better recovery of motor functions.
Electrospun hydrogel micro/nanofibers were developed as implantable scaffolds, and tissues were regenerated by implanting those scaffolds. Considering characteristics and potential, these scaffolds can be further applied to regenerate other various tissues that have fibrous structures or require fibrous structures for efficient tissue regeneration.
Wound dressings to repair skin
Skins play important roles in sensory perception, homeostasis and protecting internal organs and tissues from infection and physical impact, 118 which can be interfered by severe skin wounds that might require large amount of time to recover. Thus, wound dressings with various recovery strategies including prevention of moisture loss and infection at wound sites have been developed to aid skin repair,119–122 and electrospun hydrogel micro/nanofibers were also applied to fabricate wound dressings. Compared to conventional bulk hydrogel wound dressings, electrospun fibrous hydrogel wound dressing might provide larger surface area for cell adhesion and allow more cell infiltration through the dressing.
Electrospun GelMA nanofibers grafted with particles releasing drugs in response to increase of temperature were developed as wound dressings, and wound healing with these dressings was evaluated with full-thickness mouse skin defect models (Figure 11(a)). 64 On day 12 of wound healing assay with the skin defect models, wounds covered by GelMA nanofibers releasing drugs at body temperature (GelMA-polydopamine (PDA)-aspirin (ASP) group) showed more wound closure than wounds just recovered with cotton gauzes (Control group). Additionally, H&E staining on the day 12 allowed to verify that the skin repaired with the dressings showed high reepithelization and structure similar to normal skin (Figure 11(b)).
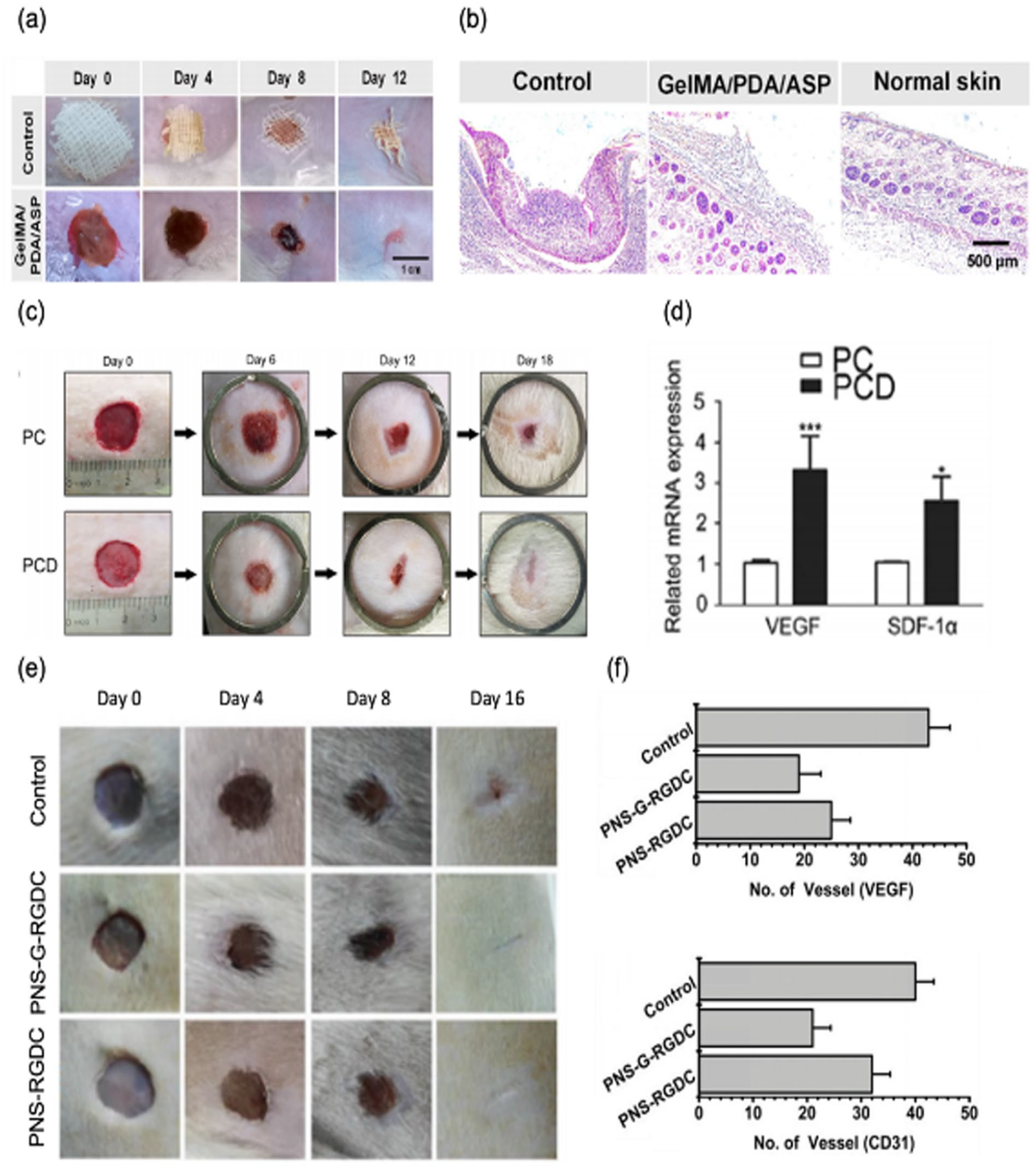
Wound dressings developed with fibrous hydrogels. (a) Pictures of wounds covered with gauze (Control) and GelMA-PDA-ASP nanofibers on day 0, 4, 8, and 12. (b) H&E images of sectioned wound site after 12 days of treatment. Reproduced from Zhang et al. 64 (c) Pictures of wounds treated with PVA/CS (PC) nanofibers or PC nanofibers loaded with DFO (PCD) on diabetic rats on day 0, 6, 12, and 18. (d) mRNA expression of VEGF and SDF-1α after 6 days of PC and PCD treatment. Reproduced from Chen et al. 65 (e) Pictures of the wounds from control, PNS microfibers with GS-Rg3 and cell adhesion peptide (PNS-G-RGDC) and PNS microfibers with cell adhesion peptide (PNS-RGDC) groups on day 0, 4, 8, and 16. (f) Immunohistochemical staining analysis of VEGF (top) and CD31 (bottom) after 16 days of treatment. Reproduced from Xu et al. 66
In addition, PVA/CS (PC) nanofibers loaded with PFO (PCD) expected to accelerate formation of blood vessels were used to treat full thickness of wounds on dorsum of diabetic rats (Figure 11(c)). 65 Promoted angiogenesis can be helpful for recovery of diabetic wound because formation of blood vessels delivering oxygen and nutrients required for wound healing can be inhibited by high level of glucose. As a result, wound closure was enhanced by using PC nanofibers releasing DFO. Increased expression of vascular endothelial growth factor (VEGF) and stromal cell-derived factor 1alpha (SDF-1α) was also verified when DFO was delivered to wound sites (Figure 11(d)).
γ-PGA-Nor/ γ-PGA-SH (PNS) microfibers involving GS-Rg3 and added with cell adhesion peptide (arginine-glycine-asparate-cysteine (RGDC)) (PNS-G-RGDC) were also used to treat wounds at back skin of rats (Figure 11(e)). 66 Gs-Rg3 that could suppress proinflammatory cytokines and downregulate VEGF expression was expected to inhibit hypertrophic scar formation. As a result, the dressings promoted wound healing and relatively inhibited scar formation. Additionally, angiogenesis regarded as an indicator of scar formation123,124 was reduced when the dressings were used to treat wounds (Figure 11(f)).
Wounds at skins were effectively treated with electrospun fibrous hydrogel-based wound dressings developed with healing strategies, showing potential to treat various types of wounds at skins. Development of diverse target-specific healing strategies as well as relatively easy fabrication methods might be useful for wide application of these electrospun fibrous hydrogel-based dressings.
PH- and glucose-sensitive biosensors
Conventionally, hydrogel-based biosensors can detect biological events in two different ways. 125 The first one uses stimulus (e.g. pH, temperature, and electrical field)-sensitive changes in hydrogel swelling degrees for the sensing of biological events. In the other way, the biological events were detected via using bioreceptors that were added to hydrogels and categorized as enzyme, antibody, biomimetic, cell, and DNA. 125 Likewise, biosensors fabricated with fibrous hydrogels and used for biomedical applications also worked with these principles.
Specifically, working principles of fibrous hydrogel-based biosensors that were sensitive to pH and glucose are described in Figure 12. The pH-responsive biosensors were working with pH-sensitive swelling/deswelling of PVA/PAA nanofibers. In response to variation of media pH, the sensitive layer (PVA/PAA nanofibers) of the biosensor swelled/deswelled and capacitance of the layer varied. As a result, changes of media pH could vary photocurrents under light exposure. For the glucose responsive biosensors, the glucose oxidase enzyme was encapsulated in fibrous hydrogels. Thus, the currents could be increased via oxidation when there was glucose. In this case, the enzyme was used as a bioreceptor.

Illustration of working principles of pH-sensitive and glucose-sensitive biosensors fabricated with fibrous hydrogels.
As applications, PVA/PAA nanofibers sensitive to pH were integrated with a light addressable potentiometric sensor (LAPS) (NF-LAPS) to measure acidification of cancer cells and response of them to drugs (Figure 13(a)). 68 As a result, the NF-LAPS was able to sense variation of extracellular acidification when concentration of cancer cells in glucose-rich media was varied (Figure 13(b)). Additionally, effects of doxorubicin and enzyme inhibitors alone or together on cancer cells resistant to some drugs were verified by measuring variation of extracellular acidification.

Biosensors developed with electrospun fibrous hydrogels. (a) A structure of a light addressable potentiometric sensor integrated with PVA/PAA nanofibers (NF-LAPS). (b) Measurement of pH shift with varied concentration of cancer cells. Reproduced from Shaibani et al. 68 (c) A wearable biosensor fabricated with PVA/BTCA/β-CD/GOx/AuNPs hydrogel nanofibers. (d) Analysis of enzymatic activity of PVA/BTCA/GOx, PVA/BTCA/β-CD/GOx, and PVA/BTCA/β-CD/GOx/AuNPs hydrogel nanofibers. Reproduced from Kim and Kim. 69 Distributed under the Creative Commons Attribution License 4.0 (CCBY, http://creativecommons.org/licenses/by/4.0).
Additionally, patch sensor developed with PVA/BTCA/β-CD/GOx/AuNPs nanofibers were used to measure concentration of glucose (Figure 13(c)). 69 Enzymatic activity of those nanofibers was maintained relatively high via GOx/β-CD inclusion complex and AuNPs although their fabrication processes involved using high voltage and temperature (Figure 13(d)). It appears that structures and sizes of electrospun hydrogel nanofibers, compared to those of bulk hydrogels, helped enhancing performance of sensors. Similar to aforementioned hydrogel nanofiber-based applications, sensors with various practical functions for biomedical applications are expected if diverse characteristics of hydrogels in electrospun nanofiber forms are exploited for development of biosensors.
Future outlook
Various polymers, other than hydrogels, have been used for electrospinning to obtain polymeric micro/nanofibers mimicking geometries of ECM, and those fibers have been intensively used for various biomedical applications. In particular, PCL fibers, relatively easy to fabricate, biocompatible and degradable, have shown great outcomes in biomedical fields although their stiffness and adhesiveness to cells were quite different from those features of natural ECM. Recently, hydrogels that exhibited their ability to mimic various characteristics of ECM were used to fabricate micro/nanofibers, and studies used those fibers have shown distinct cellular behaviors and responses that were not known before. Especially, fabrication of fibers soft enough for cells to deform and remodel allowed to study how stiffness of fibers could affect cells, and cellular behaviors and responses were significantly influenced by fiber stiffness. These clearly implied that fibrous hydrogels have potential to draw great outcomes in biomedical fields by exploiting various characteristics and easy modification of hydrogels.
In 2D/3D cell culture systems, fibrous hydrogels might be able to provide ECM-like environment by modifying their stiffness, adhesiveness to cells, degradability and growth factor presentation in addition to geometry and topography. Strategies permitting cell migration through fibrous hydrogels via deformation and degradation of fibers can be useful for 3D cell culture. Tightly stacked fibers and fragmented fibers linked with non-degradable crosslinkers might not be ideal for culturing encapsulated cells. The strategies enhancing cell migration through fibrous hydrogels, which can vary through target tissues or type of diseases, can be also applied to implantable scaffolds and wound dressings for rapid tissue regeneration. Various hydrogels displaying stimuli-sensitive responses can be applied for development of sensors performing biomedical functions with precise measurement.
On the other hand, there exist some limitations on electrospun fibrous hydrogels developed for biomedical applications. For instance, productivities of electrospinning techniques used for fabrication of fibrous hydrogels were low because flow rates of hydrogel precursors were mostly low during the fabrication processes. Needless electrospinning technique, of course, enhanced productivity by generating multiple jets. 24 In addition, fibrous hydrogels in many researches were collected as densely packed mat forms that often limited cell infiltration through fibers and cell encapsulation within fiber networks. Likewise, some techniques to generate 3D environments with fibrous hydrogels (described in Table 4) were developed to improve this issue.22,23,59–63,67,126 Lastly, solvents toxic to cells were used for fabrication of some fibrous hydrogels. To ensure biocompatibility of fibrous hydrogels, removal of solvents were performed via washing with PBS 63 or drying steps.22,66,67
Methods for fabricating 3D environments with fibrous hydrogels, control of physical properties of the fibrous hydrogels and biomedical applications of the 3D environments.

Overall, fibrous hydrogels fabricated with electrospinning techniques in recent studies have shown great potential in biomedical applications. Exploiting diverse characteristics of hydrogels and developing efficient strategies would be useful to expand their biomedical applications and enhance their performances.
Conclusion
Polymeric micro/nanofibers mimicking geometries of ECM have been fabricated with electrospinning techniques and intensively used for various biomedical applications. In particular, various hydrogels that can be modified to mimic diverse ECM features were also used to generate electrospun hydrogel fibers, and the fibrous hydrogels were applied to biomedical fields. For instance, fibrous hydrogels fabricated with electrospinning techniques were used to develop 2D/3D cell culture platforms to study cellular behaviors and responses, implantable scaffolds to regenerate tissues and wound dressings to enhance recovery of wounded skin. In addition, fibrous hydrogels sensitive to stimuli were used to develop biosensors. Overall, fibrous hydrogels have exhibited great performances within those biomedical applications. In the future, applications of the fibrous hydrogels in biomedical fields can be expanded through exploiting various characteristics of hydrogels, modifying their compositions and developing effective strategies.
Footnotes
Author contributions
J.W.L. investigated review contents and wrote original draft. K.H.S. supervised writing this review and reviewed and edited the draft.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Incheon National University (International Cooperative) Research Grant (2020-0351) in 2020 and the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (NRF-2022R1A4A5034121).
