Abstract
Hydrogels, composed of three-dimensional polymer networks, are excellent delivery carriers and have been extensively employed in the biomedical field. Inflammation acts as a protective mechanism to prevent harmful substances from entering living organisms, but chronic, long-lasting inflammation can cause oxidative stress, which damages tissue and organs and adversely affects patients’ quality of life. The aberrant expression of microRNAs (miRNAs) has been found to play a significant part in the etiology and progression of inflammatory diseases, as suggested by growing evidence. Numerous hydrogels that can act as gene carriers for the intracellular delivery of miRNA have been described during ongoing research into innovative hydrogel materials. MiRNA hydrogel delivery systems, which are loaded with exogenous miRNA inhibitors or mimics, enable targeted miRNA intervention in inflammatory diseases and effectively prevent environmental stressors from degrading or inactivating miRNA. In this review, we summarize the classification of miRNA hydrogel delivery systems, the basic strategies and mechanisms for loading miRNAs into hydrogels, highlight the biomedical applications of miRNA hydrogel delivery systems in inflammatory diseases, and share our viewpoints on potential opportunities and challenges in the promising region of miRNA delivery systems. These findings may provide a new theoretical basis for the prevention and treatment of inflammation-related diseases and lay the foundation for clinical translation.

Introduction
Hydrogels are usually materials with a three-dimensional (3D) polymer network structure composed of a significant amount of water without dissolving, typically formed from natural or synthetic hydrophilic materials through physical or chemical crosslinking, and that can maintain their shape and volume under specific conditions while facilitating the diffusion and penetration of water molecules within their structure.1,2 Hydrogels are very promising biomedical engineering materials with advantageous physical and chemical properties, such as biocompatibility, biodegradability, low toxicity, and enhanced retention effect.3–6 Generally, hydrogels change the physicochemical properties by changing the material composition, concentration, functional group modification, crosslinking mechanism, density, and preparation methods, or by integrating with other nanocarriers to synergistically control spatial and temporal drug release for specific requirements in various applications.7–9 Furthermore, modulating the degree of crosslinking and the polymer’s interaction with water in the matrix can modify the hydrogel’s porosity. Its structure and shape can be adjusted in response to environmental stimuli such as pH, temperature, and light.10–12 Hydrogels can exist in the form of a macroscopic network or be confined to smaller sizes, such as microgels or nanogels, and can be administered in a variety of modes, including oral, regional injection, and transdermal, chosen primarily to maximize overall efficacy and patient compliance.13,14
Inflammation serves as a defense mechanism of an organism against stimulation, involving vascular changes, the recruitment of immune cells and the release of a variety of molecular mediators, and its main pathological changes are degeneration, exudation, and proliferation of tissues.15–17 Fighting infections or hazardous stimuli, healing damaged tissues, and re-establishing homeostasis are the main objectives of inflammation, but persistent chronic inflammation can accelerate the progression of many diseases by damaging tissues and organs through oxidative stress.18–22 Currently, the most commonly used anti-inflammatory drugs in clinical practice are nonsteroidal anti-inflammatory drugs and glucocorticoids.23,24 However, the abovementioned drugs have a variety of limitations, so the development of new anti-inflammatory drugs is urgent.
MicroRNAs (miRNAs) are significant adjustors of gene expression in many eukaryotes.25–28 Many reported studies have explored the important roles of the differential expression of miRNAs in regulating the pathogenesis of inflammatory diseases.29,30 Recent studies have found that miRNAs are used as nucleic acid drugs for the treatment of inflammatory diseases, but the rapid degradation and inactivation of miRNAs in the adverse disease microenvironment remains an unavoidable obstacle in many inhibitory or alternative therapies using targeted miRNAs, thus limiting the application of miRNAs.31,32 Therefore, the efficient delivery of miRNAs to target genes or cells remains a persistent challenge in developing efficient and safe delivery systems and applying them to the treatment of inflammatory diseases. Schematic representation of miRNA biogenesis and the regulatory role of miRNAs in inflammation was shown in Figure 1.
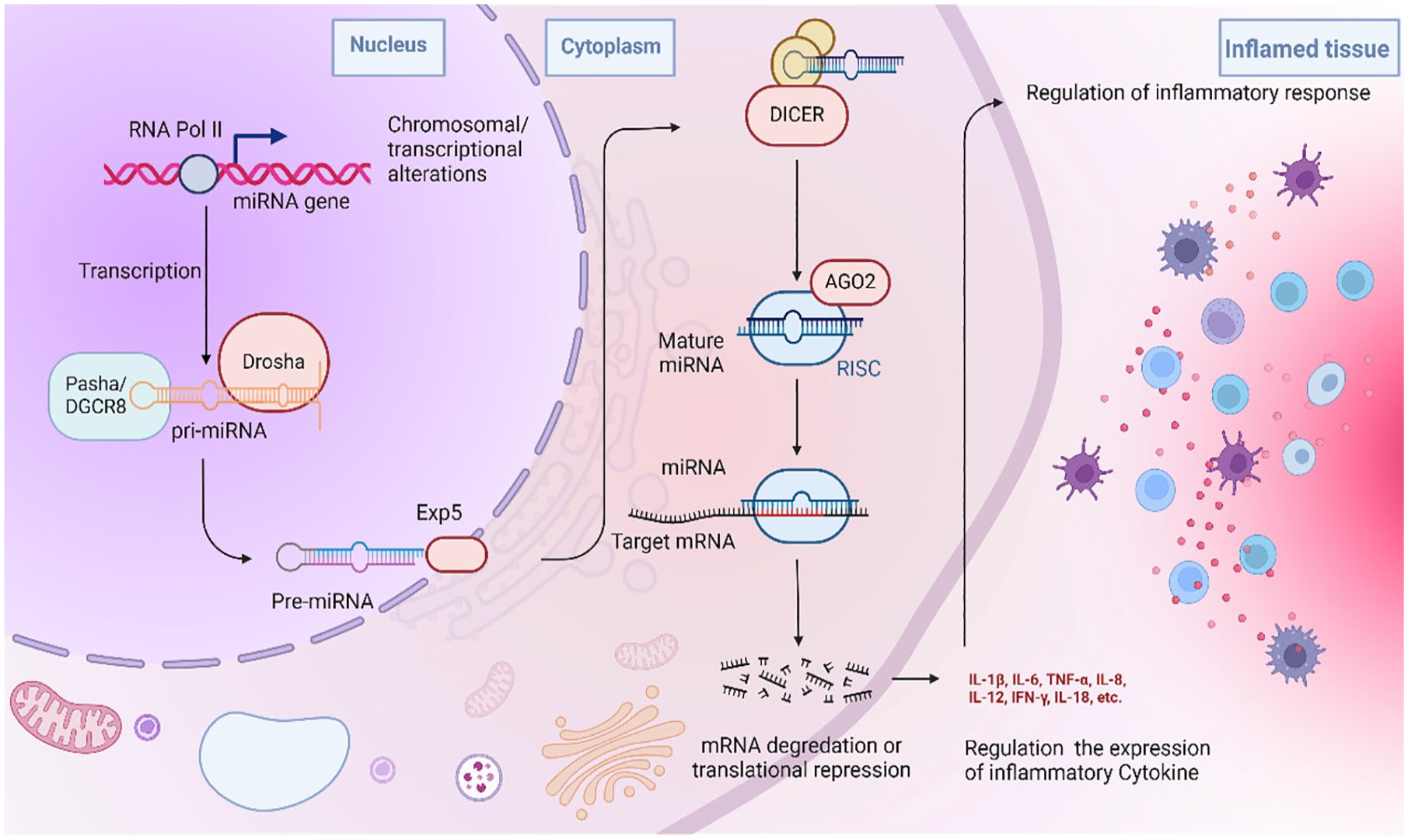
Schematic representation of miRNA biogenesis and the regulatory role of miRNAs in inflammation. Most miRNA biogenesis starts with the transcription of primary miRNA (pri-miRNA) by RNA polymerase II. Drosha cleaves pri-miRNA in the nucleus to produce precursor pre-miRNA of approximately 70–110 nt, while Dicer processes the precursor pre-miRNA into a mature miRNA of approximately 22 nucleotides in the cytoplasm. The expression levels of mRNAs and proteins can be regulated by the synthesis and silencing of miRNAs, accordingly, adjusting the physiological function of cells and inflammatory cytokines and influencing the progression of inflammation-related diseases.
Multiple strategies have been devised for transporting miRNA, encompassing viral vectors, liposomal formulations, cationic polymers, and modified exosomes to enhance therapeutic efficacy. Nonetheless, these carriers still encounter challenges, such as poor biocompatibility, potential biosafety concerns, and insufficient stability, 33 it is imperative to explore more effective delivery methods. Hydrogel delivery systems with porous structures mimic the native extracellular matrix (ECM) and allow for the incorporation of various miRNAs, commonly utilized for successful miRNA delivery in drug therapy.34–40 This delivery method offers distinct advantages such as improving miRNA stability, enabling local and sustained release, ensuring biocompatibility and degradability, precise release control, versatility, and wide applicability. However, the basic strategy of miRNA hydrogel delivery system preparation and its application in inflammatory diseases needs to be further elucidated. This review is intended to summarize the classification of miRNA hydrogel delivery systems, strategies for incorporating miRNA into hydrogels, and the mechanisms for controlling miRNA release, highlighting recent progress in miRNA hydrogel delivery systems in the therapy of various inflammatory diseases, such as osteoarthritis, diabetic foot ulcer, myocardial infarction, and spinal cord injury. Here, advanced application of miRNA hydrogel delivery systems in various inflammatory diseases was shown in Figure 2. We also offer our perspectives on the future promise and potential limitations facing problems and potential in this blooming scientific field.
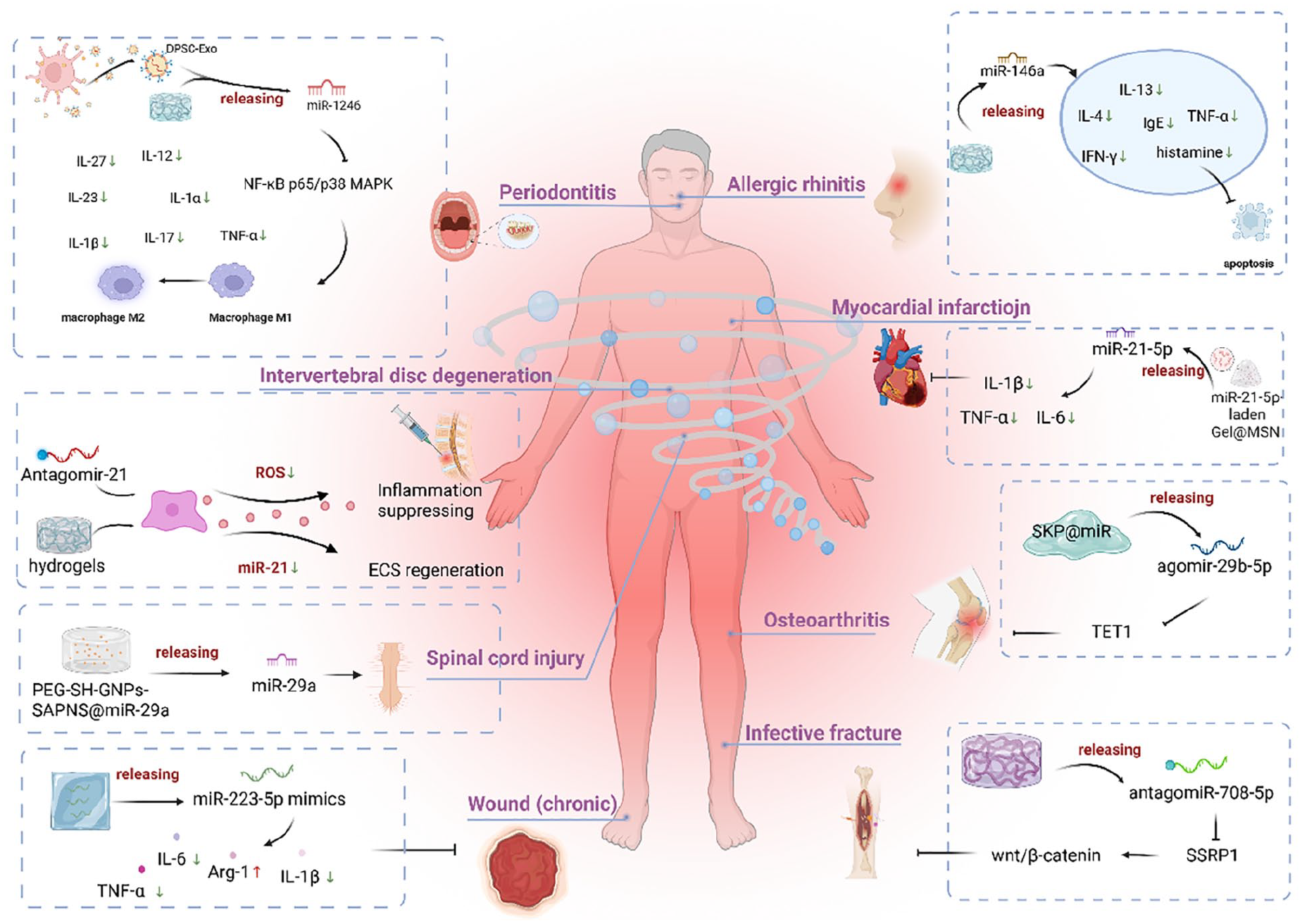
Advanced application of miRNA hydrogel delivery systems in various inflammatory diseases. MiRNA hydrogel delivery systems are a promising platform for delivering miRNA. They have the potential to be developed into targeted gene delivery systems and therapeutic drugs for treating inflammatory diseases. These systems can effectively restrict multiple inflammatory factors, including IL-1β and TNF-α, making them valuable for treating conditions such as osteoarthritis, diabetic foot ulcers, and other inflammatory diseases.
Classification of miRNA hydrogel delivery systems
Appropriate hydrogel delivery systems could protect miRNA from premature nuclease degradation and promote the cellular uptake of miRNA at target locations.41,42 Generally, miRNA-loaded hydrogel materials are classified into natural hydrogels, synthetic hydrogels, complex hydrogels and supramolecular hydrogels (Figure 3).1,43 Chitosan (CS), hyaluronic acid (HA), alginate, gelatin, and silk fibroin are examples of natural hydrogels that demonstrate remarkable biocompatibility and biodegradability; however, their poor mechanical properties restrict the range of applications. 44 Synthetic polymers (such as PEG and acrylamide) have more desirable mechanical properties.45,46 Composite hydrogels are composites of different types of polymers or hydrogels with added functional nanomaterials, such as gold nanoparticles and graphene. 47 Supramolecular hydrogels have been proposed as smart materials due to their excellent dynamic responsiveness in response to various external or internal stimuli.48,49 Overall, a summary of typical miRNA-loaded hydrogel materials and their physicochemical properties is provided (Table 1) to offer a systematic impression of miRNA-loaded hydrogels.
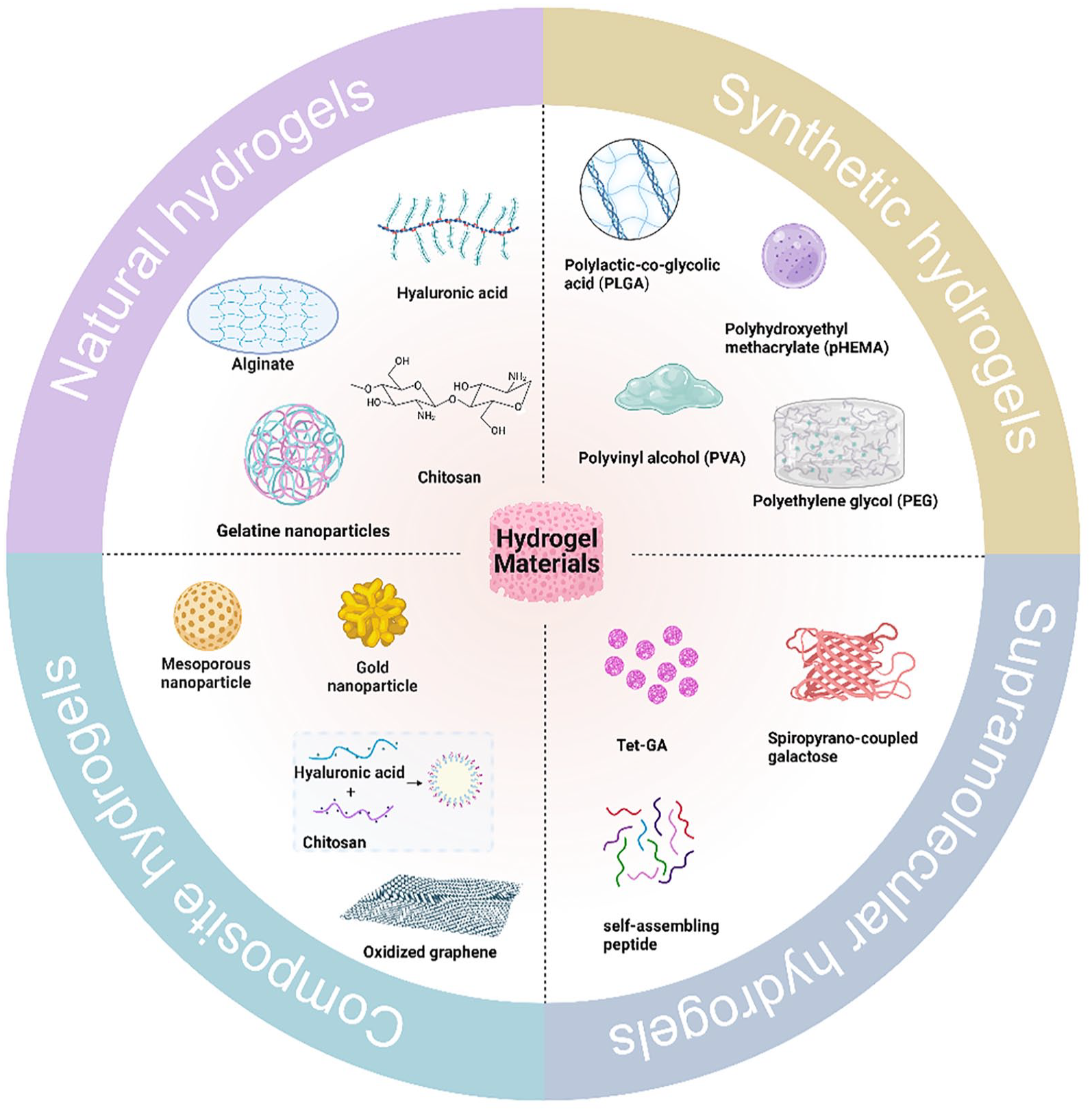
Materials commonly used in the synthesis of miRNA hydrogel delivery systems. According to the type of materials and their structural properties, hydrogels can be categorized in a variety of ways, and miRNA hydrogel delivery systems can be divided into natural hydrogels, synthetic hydrogels, complex hydrogels, and supramolecular hydrogels.
Classification and properties of hydrogel carriers for miRNA delivery.

: In this table, miR-223* indicates the abbreviation for miR-223-5p mimic.
Natural hydrogels
HA is an anionic polymer present in the natural ECM. It is a linear polysaccharide consisting of alternating units of D-glucuronic acid and N-acetylglucosamine, possessing active sites that can interact with functional groups to meet specific requirements for miRNA delivery.69,70 Hyaluronic acid hydrogel has excellent biocompatibility and biodegradability, but the single component HA has low mechanical strength and uncontrollable degradation. Moreover, HA cannot be independently cross-linked, therefore, different chemical modifications of HA polymer chains are recommended before designing suitable hydrogels for different specific applications.71–73 Chemical modification of HA has been extensively studied, mainly targeting glucuronic carboxylic acid, primary and secondary hydroxyl groups, and N-acetyl groups (after deamidation). Sun et al. 50 adopted a multi-delivery strategy to construct a HA hydrogel modified by methacrylic anhydride (HAMA) combined with di(ethylene glycol) monomethyl ether methacrylate (DEGMA), and introduced exosomes enriched in miRNA 24-3p for cell-free therapy of corneal epithelial regeneration. The HA hydrogel addressed several important factors related to exosome stability, retention time on the ocular surface and the efficacy of miRNA 24-3p, which was particularly beneficial because it allowed for the sustained release of exosomal miRNA 24-3p, resulting in the promotion of corneal epithelial wound repair in vivo.
Cationic CS is able to form a miRNA delivery system through electrostatic interaction. 74 CS, a natural linear polysaccharide, has many highly desirable properties, such as antibacterial effects, biodegradability, biocompatibility, non-cytotoxicity and protein affinity, and holds immense promise for significant applications in gene drug delivery systems.75,76 However, CS has weak mechanical strength, difficulty in adjusting its pore size, poor long-term stability, and a short life span ranging from a few days to a month in physiological media. The above problems are usually solved by combining with other materials or optimizing cross-linking agents, preparation methods, and domain modifications to improve and adjust the chemical properties and structure of CS hydrogels. For example, CS has many active amino groups and hydroxyl groups that can be used for chemical modification to change its solubility, quaternization to enhance its antibacterial activity, and carboxymethylation to enhance the membrane permeability of biomacromolecule drugs.77–79 In general, CS has been frequently used as a carrier for loading exosomes because it can extend the duration of exosome presence at the site of injury, thus safeguarding them from immune system clearance.54,80 A low-temperature polymerized CS hydrogel wound dressing was customized by Tao et al. 55 and loaded with miR-126-3p-overexpressing synovium mesenchymal stem cell (SMSC) exosomes to promote angiogenesis. The results showed that wounds treated with CS hydrogel loaded with SMSC-126 exosomes (CS-SMSC-126-Exos) had better wound closure on day 14 than those in the untreated or CS-treated groups. Apart from the abovementioned common hydrogel materials for miRNA delivery, there are many other natural materials, such as gelatin, alginate, and silk fibroin, which also show high potential for miRNA delivery (Table 1).57–59
Synthetic hydrogels
Synthetic polymers are particularly promising for miRNA delivery, and well-known examples include polylactic-co-glycolic acid (PLGA), polyhydroxyethyl methacrylate (pHEMA), polyvinyl alcohol (PVA), and PEG.81,82 PEG is the most extensively used highly biocompatible synthetic polymer. 83 Different chemical modifications of PEG are necessary for different specific applications; for example, it is common to attach functional groups such as p-toluenesulfonate, carboxyl, and aldehyde groups onto the PEG polymer backbone. 84 Additionally, the ether bond (–O–) on the PEG chain can form strong hydrogen bonds with water, whereas the –CH2–CH2– on the PEG chain makes it hydrophobic; thus, PEG can be dissolved in water and some organic solvents, offering the possibility of PEG synthesis in polar and nonpolar solvents.85,86 The conjugated or noncovalent attachment of PEG molecules to other molecules or nanoparticles is known as PEGylation. 87 PEGylation may enhance biocompatibility and optimize the pharmacokinetic properties of nucleotide drugs.88–90 Nguyen et al. 60 pointed out that the local and continuous delivery of siRNA against noggin (si-Noggin) and miRNA-20a by the in situ formation of PEG hydrogels effectively promoted the osteogenic differentiation of wrapped human bone marrow-derived mesenchymal stem cells. Therefore, PEG has become one of the preferred materials for the design and preparation of continuous controlled drug delivery systems since it has desirable characteristics. 91
Composite hydrogels
A single hydrogel material might be unable to provide good biological properties, and therefore, a given hydrogel material is commonly integrated with other polymers or inorganic materials, giving it unique physical and chemical properties to enable new functions and new applications. Hence, composite hydrogels with efficient drug delivery capacity and bioactive performance might be excellent candidates for advanced miRNA delivery applications. Wu et al. 62 constructed a nano/hydrogel composite system prepared by combining CS/β-GP hydrogels loaded with stromal cell derived factor-1α (SDF-1α) and CS/tripolyphosphate/HA/antimiRNA-138 nanoparticles (CTH/antimiR-138 NPs) to deliver antimiRNA-138 for the regeneration of critical size cranial bone in rats.
Supramolecular hydrogels
Some small-molecule organic compounds can form nanofibers by gelling with water or organic solvents in a low-concentration environment, which then function as a network of supramolecular hydrogels.92–94 Small organic compounds, known as low-molecular-weight gel factors (LMWGs), form solid-like supramolecular hydrogels. 95 Xiao et al. 66 prepared a supramolecular hydrogel from spiropyrano-coupled galactose for the photo-controlled release of miR-122 and delivery to HepG2 cells cultivated on the hydrogel through the ligand-receptor interaction-mediated release of miRNA inhibiting the expression of target genes. Furthermore, Li et al. 67 fabricated a stable hydrogel utilizing the small-molecule hydrogelator Tet-GA formed by the dipeptide Gly-Ala linked with biphenyl-substituted tetrazole at neutral pH as a 3D matrix for live cells and hybrid materials while loaded with miRNAs. More than 95% of the Tet-GA hydrogel-coated cells remained viable during 3D culture for 24 h. This high viability rate provided researchers with the opportunity to investigate the delivery of miRNA to live cells within the hydrogel matrix.
In addition, the self-assembling peptide (SAP) composed of amino acids, as a type of supramolecular hydrogels with building units including natural amino acids and/or hydrophobic fatty acid chains, can self-organize into ordered nanofibers and further form ordered scaffolds in water, which are also extremely attractive nanomaterials to be used as miRNA carriers. 96 The self-assembly of polypeptide molecules is driven mainly by internal driving forces, which are mainly non-covalent forces, such as electrostatic interactions, hydrogen bonds, л-л stacking, and hydrophilic/hydrophobic interactions. SAP hydrogels have the ability to decompose into pure biological components, exhibiting lower toxicity compared to synthetic nanoparticles and polymer nano-systems. Peptides and proteins can self-assemble into nanostructures on various compound surfaces. Essential factors like pH, temperature, ionic strength, and the physicochemical characteristics of the substrate surface play crucial roles in shaping nanostructures during the self-assembly process. Consequently, these parameters can be skillfully modified to govern the characteristics of the nano-materials. By simple structural modifications of synthetic peptides, supramolecular hydrogels were fabricated, making them responsive to ligand-receptor interactions. With the help of supramolecular hydrogels, miRNAs are delivered into cells to perform biological functions and suppress the expression of their target genes concurrently. In physiological environments, SAPs have the ability to self-assemble and gelate into hydrogels. These hydrogels typically exhibit excellent biocompatibility and bioactivity, making them suitable for treating local inflammation. Furthermore, they can prolong drug retention time and reduce side effects without compromising drug efficacy. As a typical ionic complementary SAP, RADA16-I, Ac-(RADA)4-NH2, has been extensively studied across different disciplines. The SAP is capable of creating a nanoscale environment in water similar to the natural ECM. It exhibits self-assembly and adaptive properties, facilitating easy binding to host tissues. Moreover, its versatility in functionalization with growth factors and other biological motifs has made it a promising candidate for various biomedical applications.97–100
It has been previously reported that modified RADA16-I self-assembly hydrogel can load miRNA and play a role in sustained release precision therapy. Zhu et al. 68 designed an injectable, stem cell-homing hydrogel to deliver miRNA (SKP@miR), which was formed by integrating agomir-29b-5p with functionalized SAPs containing the stem cell-homing sequence SKPPGTSS. The introduction of cationic functional peptides (SKP peptides) into the hydrogels formed by SAP enabled endogenous stem cell recruitment. Agomir-29b-5p binded to the SKP peptide primarily through hydrogen bonding and electrostatic interactions. In the presence of the SKP peptide, agomirs exhibited a preference for binding to the functional motif SKPPGTSS over RADA sequences, due to the positively charged lysine. Thus, high affinity binding between agomir-29b-5p and SKP results in sustained release. Furthermore, the inclusion of agomirs does not impact the β-sheet structure or nanofiber formation of SAPs. The injectable SKP@miR hydrogel created a suitable in situ microenvironment to allow controlled and minimally invasive gene vector delivery with spatiotemporal precision and reduce intraarticular vector diffusion and therapeutic gene agent loss. Research on supramolecular hydrogel is still in its infancy at present stage, mainly focuses on the biomedical field. As it responds to specific environmental stimuli, further exploration of miRNA functions using this supramolecular hydrogel represents a new direction for the development of anti-inflammatory hydrogels.
Methods for the preparation of the miRNA hydrogel delivery system
The miRNA encapsulation capacity of hydrogel is contingent upon the chemical composition and characteristics of their constituent structural elements. Hydrogels can encapsulate miRNAs in two ways: either by directly incorporating the miRNA without any protective covering or by employing miRNA nanocarriers that bind to the hydrogel (Figure 4).

Schematic representation of methods for the preparation of the miRNA hydrogel delivery system.
Hydrogels directly encapsulate miRNA
MiRNAs are capable of binding to the structural components of hydrogels through physical adsorption or chemical bonding. Physical adsorption involves the non-covalent binding of miRNA to the hydrogel, facilitated by interactions such as electrostatic forces and hydrogen bonds. On the other hand, chemical bonding occurs when specific functional groups of miRNA chemically react with corresponding groups on the hydrogel, forming stable chemical bonds. Gan et al. 101 reported that an injectable poly(ethylene glycol) (PEG) hydrogel was prepared and conjugated with a cholesterol-modified noncoding miRNA (Chol-miR-26a) through an ultraviolet (UV)-cleavable ester bond for bone regeneration. Specifically, injectable PEG hydrogels are formed by a copper-free “click” reaction between the terminal azide groups of 8-arm PEG and the dibenzocyclooctyne-functionalized PEG. Simultaneously, the UV-light cleavable cholesteryl-miR-26a is encapsulated into the hydrogel through a Michael addition reaction.
miRNA pre-complexation hybrid systems in hydrogels
The most commonly reported continuous miRNA delivery systems are hydrogels in combination or separately with liposomes, polymers, nanoparticles, and exosomes mixtures. These hybrid systems can be tailored based on factors such as particle size, surface charge, crosslinking methods, incorporation techniques, stability, and biocompatibility to optimize their miRNA delivery capabilities, ultimately influencing their behavior and therapeutic effects in vivo.
Liposomes, consisting of a double lipid layer surrounding an internal aqueous core, are ideally sized between 80 and 150 nm for cellular uptake. 102 The lipid bilayer of these liposomes permits the incorporation of lipophilic substances, enhancing their solubility and enabling their inclusion within hydrophilic hydrogels. 103 The integration of liposomes into hydrogels confers sufficient viscosity to the local drug delivery dispersion. This hydrogel matrix serves to hinder rapid clearance of the liposomes from the skin site and prevents rapid degradation by maintaining their membrane integrity. The combination of these two delivery systems represents a promising approach for achieving controlled drug delivery and effective localized therapy. Rajagopal and colleagues have devised a 3D transfection protocol, where miR-140 is encapsulated within cationic liposomes (AmCTOC) to produce functional miRNA liposomes. 104 These are then loaded onto hydrogels containing human mesenchymal stem cells (MSCs). Notably, miR-140 significantly suppressed the expression of the hypertrophy marker COL10A1 on both the 14th and 21st days, while concurrently inducing a stable chondrogenic phenotype characterized by high expressions of COL2A1, SOX9, and ACAN.
Cationic polymers are used for the delivery of negatively charged nucleic acids. 105 Polyarginine has been shown to have cell-permeable properties and can be used for miRNA delivery. Gribova et al. 106 developed a delivery system for miRNA transport to macrophages via the in vivo coassembly of polyarginine with a polymerization degree of 30 (PAR30) with a HA hydrogel based on electrostatic interactions. Positively charged PAR30, as a nucleic acid vector, was assembled with negatively charged miRNA into a complex, which was then successfully loaded into polyanionic HA hydrogels and transported into the cell. Subsequently, the electrostatic interaction between miRNA, PAR30, and hyaluronic acid is disrupted, resulting in the release of miRNA into the cell.
Nanoparticle-hydrogel composites are frequently employed for miRNA delivery. NPs have the potential to improve the stability and solubility of the encapsulated cargo, facilitating transmembrane transport, lengthening cycle times, and improving availability, which may have benefits such as low cytotoxicity, high blood stability, low immune reactivity, biodegradability, and controlled release.107–110 Inorganic materials, such as graphene oxide, gold NPs and mesoporous silicon NPs, have been extensively studied for the codelivery of miRNA with hydrogel assembly.111,112 A PEG- and polyethylenimine (PEI)-functionalized graphene oxide (GO)-based CS hydrogel (GO-PEG-PEI@CS) was prepared by Qin et al. 63 to deliver miR-29b. The results indicated that miR-29b was effectively delivered using the GO composite hydrogel, which significantly aided in the differentiation of bone marrow mesenchymal stem cells in osteogenic tissue and bone regeneration without causing an inflammatory response. Van et al. 64 engineered self-assembled polymer nanoparticle (PNP) hydrogels by encapsulating functionalized AuNPs with PEG, has-miR-214 and HA peptide to deliver has-miR-214. AuNP-miR-214 retained functionality in a 3D in vitro model of human calcific aortic valve disease and was successfully cleared 11 days after subcutaneous injection of the PNP-AuNP-miRNA hydrogel delivery system in mice. A mesoporous silica nanoparticle (MSN)-embedded core-shell structured poly (ethylene glycol)-b-poly (lactic-co-glycolic acid)-b-poly (N-isopropylacrylamide; PEG-PLGA-PNIPAM) hydrogel was reported by Lei et al. 65 for the local and lasting codelivery of microRNA-222 and aspirin (ASP; miR222/MSN/ASP hydrogel), and this hydrogel successfully promoted bone regeneration. The main disadvantage of nanoparticles for miRNA delivery is their inability to degrade in organisms, which might bring a variety of negative health consequences. Several studies have emphasized this issue, and more research is still needed.
Exosomes, ranging in size from 30 to 150 nm, represent a subset of the smallest extracellular vesicles discharged by cells, and they demonstrate facile and efficient cellular uptake. These vesicles offer numerous advantages, including the potential for targeted engineering, high biocompatibility, relatively low cytotoxicity and immunogenicity, and the ability to transport various nucleic acids, including miRNAs.113,114 Exosomes are secreted into the microenvironment to mediate intercellular communication by effectively transporting miRNAs to their target cells.33,114,116 In the realm of exploring miRNA delivery technologies, exosome-based delivery has demonstrated its distinct advantages. However, exosomes face challenges during the delivery process, such as high clearance rates and short half-lives, which to some extent limit their therapeutic efficacy.117,118 To overcome these limitations, researchers have proposed an innovative strategy—using hydrogel to deliver exosome-encapsulated miRNA, aiming to enhance delivery efficiency and stability, thus paving the way for new applications of exosomes in the field of drug delivery. The preparation strategy for stem cell exosomes-loaded hydrogel to deliver miRNA effectively combines the dual advantages of exosomes and hydrogels. By encapsulating the miRNA payload within the exosomes and then enveloping these exosomes in the hydrogel material, it not only effectively circumvents the rapid clearance of exosomes in vivo but also harnesses the therapeutic potential of exosomes and hydrogels simultaneously. This approach offers an optimal platform for targeted delivery and sustained release of exosomes, ensuring that they can fully exert their therapeutic effects in vivo. 119 For example, Hu et al. 120 fabricated a UV-shielded injectable gel-nanohydrogel with good biomechanical properties using gelatin methacrylate (GelMA) as a carrier loaded with therapeutic human umbilical cord mesenchymal stem cells-derived small extracellular vesicles (hUC-MSCs-sEVs) that transferred miR-23a-3p to promote cartilage regeneration. The nanoclay was mixed with the GelMA solution and incubated after SEVs with high-level miR-23a-3p were added, and the composite hydrogels were obtained after chemical cross-linking by ultraviolet radiation.
Release mechanism of miRNA from hydrogel matrices
The mechanism of hydrogels applied in miRNA delivery systems release miRNAs is primarily contingent upon the properties and design of the hydrogel material. The transport of miRNA within hydrogels can be categorized based on its rate-limiting steps, which include diffusion, swelling, and controlled release, serving to regulate the release process, as shown in Figure 5. 14

Schematic representation of release mechanism of miRNA from hydrogel matrices.
Diffusion release stands as the predominant mechanism widely employed in miRNA hydrogel delivery systems. This mechanism refers to miRNA entering the region of action by means of simple diffusion. 121 Van et al. 64 fabricated an injectable shear-thinned self-assembled PNP hydrogel delivery platform loaded with AuNP-miRNAs that conjugated miRNA mimics and PEG peptide gold nanoparticles (AuNP) and exerted its effect by diffusion release.
Swelling release will occur when the rate of drug diffusion is higher than the swelling rate of the hydrogel. 122 Swelling release mechanism denotes the process wherein the hydrogel undergoes shape modification upon encountering a conducive environment. 14 The shape transition is contingent upon the quantity of polymer (gel), crosslinker, and porosity. 123 Penetration of water into the hydrogel interior through its pores induces swelling of the dosage form and subsequent release of the miRNA. Radmanesh et al. 124 developed PEI-DA polyplexes as a promising non-viral polymeric platform, loaded into PEG hydrogels. The delivery system causes network swelling through degradation, facilitating the release of PEI-DA/LNA-92a, and subsequently releasing the hsa-miR-92a-3p inhibitor.
Controlled release may be defined as a method that allows controlling the rate and timing of miRNA release through internal automatic regulation or external stimuli (such as temperature, pH, enzymatic presence, magnetic field, light, and other stimuli). The hydrogel material was chemically modified and special functional groups were introduced to achieve the purpose of changing the release mechanism of the hydrogel. Thermosensitive hydrogels commonly contain hydrophobic groups. 125 pH-responsive hydrogels comprise polymers containing acidic, alkaline, and amphoteric groups that react to alterations in the pH of the external environment.126,127 The hydrogel possesses glucose responsiveness through conjugation to an active substance bound to glucose (such as phenylboronic acids).128,129,130 Enzyme-sensitive crosslinkers or hydrogel materials with enzyme-sensitive degradation sites are appropriate for achieving enzyme responsiveness of hydrogels. 130 The introduction of borate bonds into reactive oxygen species (ROS)-responsive hydrogels exhibits ROS sensitivity, and polymers with ROS-responsive bonds can consume excess ROS and relieve local oxidative stress in addition to being able to release contents via ROS triggering. 131 Chromophores are introduced in hydrogel structures to confer photosensitivity. 132 Using a light-controlled release mechanism, Gan et al. 101 engineered an injectable UV-controllable released hydrogel based on a photosensitive group containing o-nitrophenyl-modified miRNA through a light-controlled release mechanism. The release of miRNA from the UV-light cleavable hydrogel was subsequently detected after ultraviolet irradiation. In addition, Ji et al. 133 developed a hydrogel triggered by NIRII light delivering miR-196c-3p mimics to deep tissue. This biocompatible hydrogel could be used for therapy in situ after ischemia/reperfusion (I/R). Huynh et al. 134 developed a photodegradation PEG-based hydrogel system based on Michael addition catalyst, which provided a means of controlling the activity of the miRNA loaded through the release of the RNA/PEI complex by applying external ultraviolet light. The bioactivity of the released cargo was measured, and the results showed that there was no difference in the degree of expression of the target between the “UV free” and “UV” hydrogels, indicating that ultraviolet light is benign in terms of the bioactivity of the cargo. Hence, it is fundamental to verify that the release mechanism of hydrogel system does not interfere with this inherent bioactivity of the cargo.
In general, when determining the appropriate mechanism for releasing miRNA cargo, it is necessary to carefully assess the entire pathological environment and the subsequent changes in the biological activity of the miRNA drug. It is worth noting that these mechanisms are not isolated, but can be used in combination to achieve more precise, effective, and controllable miRNA release.
Role and mechanism of miRNA hydrogel delivery system in inflammatory diseases
In gene therapy, the symptoms of inflammatory diseases can be relieved by regulating the expression of inflammation-related miRNAs, which avoids the limitations of current treatments. In particular, miRNA-loaded hydrogels with unique physicochemical properties could be used to construct targeted systems for the delivery of therapeutic genes and drugs. Here, we review the mechanism of the anti-inflammatory components and the recent advances in miRNA hydrogel delivery systems for the therapeutic application of inflammation-associated diseases, such as osteoarthritis, diabetic foot ulcers, and other inflammatory diseases.
MiRNA hydrogel delivery system for inflammatory disease of bones and joints
The inflammatory response in skeletal muscles and joints may lead to related diseases or prevent bone and tissue from repairing themselves. Inflammatory diseases of the bones and joints encompass infections as well as immune-mediated local and systemic diseases. 135
Osteoarthritis (OA) is a chronic degenerative disease accompanied by inflammation and pain, ultimately forming a vicious cycle if no medical intervention occurs.136,137 Previous studies have revealed that miRNA is one of the most effective targeted therapeutics for OA. 138 Zhu et al. 68 proposed a miRNA delivery strategy based on stem cell-homing hydrogels and further prepared an injectable hydrogel (SKP@miR) for the delivery of miR-29b-5p, which was demonstrated to alleviate cartilage degeneration and repair cartilage damage in OA (Figure 6(a)). Notably, knee joints with OA induced by anterior cruciate ligament transection surgery (ACLT) exhibited a reduction in inflammation comparable to that of healthy joints after treatment with SKP@miR. Furthermore, the treatment led to decreased defects and synovitis in the knee joints while maintaining cartilage integrity and reducing cartilage degeneration. These findings highlight the effectiveness of using hydrogels to deliver miRNA as an intervention for OA.

MiRNA hydrogel delivery system for inflammation occurred in the skeletal muscles and joints. (a) SKP peptide hydrogel loaded with agomir-29b-5p (SKP@miR) for osteoarthritis. Adapted with permission from Acar et al. 100 Copyright 2022 American Association for the Advancement of Science. (b) GNP-incorporated exosome (GNP-Exo) hydrogel to deliver miR-451a to inhibit inflammation around the bone defect. Reprinted with permission from Tao et al. 55 Copyright 2022 BMC. (c) Schematic representation of the bacterial-sensitive antagomiR-708-5p-loaded HA hydrogel that promotes the healing of infected fractures. Adapted under the terms of American Association for AMER CHEMICAL SOC license from Zhou et al. 52 Copyright 2022, the authors. (d) HA hydrogel-wrapped miRNA plasmid/NPs attenuated the occurrence of tendon adhesions. Reprinted with permission from Saleh et al. 51 Copyright 2018 Elsevier.
Fracture repair depends on controlled inflammatory responses followed by the recruitment of bone stem/progenitor cells, deposition of cartilage and bone matrix and remodeling of bone tissue.139,140 Unresolved inflammation is a major cause of protracted fracture nonhealing, and miRNAs have a considerable impact on the modulation of the inflammatory response at fracture defects. 53 Li et al. 58 demonstrated the treatment of inflammation in fracture defects with gelatin nanoparticle (GNP) hydrogels loaded with exosomes from adipose-derived stem cells (ADSC-Exos) to deliver miR-451a, which accurately reached its target location to perform its functions (Figure 6(b)). Hydrogels can also be loaded with miRNA antagomir, that is, the reverse complementary sequence of a mature miRNA chain. A bacterial-sensitive HA hydrogel was designed by Yu et al. 53 to deliver antagomiR-708-5p to infected fractures. Mechanistically, the hydrogel loaded with antagomiR-708-5p greatly accelerated fracture healing by targeting structure-specific recognition protein 1 (SSRP1) and activating the Wnt/β-catenin signaling pathway to promote osteoblast differentiation and inhibit inflammation (Figure 6(c)).
Hydrogels can also be designed to improve the inflammatory microenvironment in the wounded tendon. Zhou et al. 52 engineered a novel HA hydrogel incorporating PLGA nanoparticles loaded with cyclooxygenase (COX-1 and COX-2)-engineered miRNA plasmids to treat flexor tendon adhesion. This targeted method shows promise in downregulating the expression of COX-1 and COX-2 in the injured tendon microenvironment and has proven to be an effective therapeutic strategy for reducing tendon adhesion (Figure 6(d)).
Anti-inflammatory miRNA hydrogel delivery system in wound healing application
The wound healing process consists mainly of hemostasis, inflammation, proliferation, regeneration and remodeling, but sustained inflammation can hinder tissue healing.141–143 The persistent state of proinflammatory macrophages (M1) in chronic diabetic wounds, without transitioning to an anti-inflammatory phenotype (M2), is one of the significant factors in ongoing inflammation and hindered tissue repair. 144 Studies have shown that miRNAs are differentially expressed in diabetic wound healing, which has been confirmed to exert an important regulatory role. 145 Hydrogels have good hydrophilicity, biocompatibility, and three-dimensional porous structures, which can provide an environment conducive to cell attachment and proliferation. 146 The introduction of various anti-inflammatory ingredients, including miRNAs, into hydrogels can effectively promote wound healing. 147 Some miRNA hydrogel delivery systems have been reported to participate in macrophage phenotypic polarization, mediating inflammatory responses during wound healing. Saleh et al. 51 developed a GelMA-based adhesive hydrogel encapsulated in HA-based nanoparticles for local delivery of miR-223-5p mimics (miR-223*), thereby driving the polarization of macrophages toward anti-inflammation and shortening the wound healing process. The results showed that the adhesive hydrogel can adhere and cover the wound, and local delivery of miR-223* can effectively promote the development of vascularized skin in a model of acute wound resection, which indicated that the hydrogel miRNA delivery system could effectively control the polarization of tissue macrophages during the healing process (Figure 7A). In another study, a triple active PAR30/HA hydrogel was designed by Gribova et al., 106 which also had a positive effect on wound healing. PAR30 itself demonstrates anti-inflammatory functions and can also serve as a nucleic acid carrier to effectively load into negatively charged HA hydrogels. The PAR30/miRNA-loaded hydrogel in the wells also showed no Staphylococcus aureus growth even after 24 h, indicating that the antibacterial property of PAR30 was maintained in the presence of miRNA (Figure 7B). Overall, the miRNA HA hydrogel delivery system has anti-inflammatory, antibacterial, and miRNA carrier functions and is versatile enough to be used in diabetic ulcers.

miRNA hydrogel delivery system for wound healing. (A) MiR-223-5p mimic (miR-223*)-loaded NPs in an adhesive hydrogel are used to regulate the polarization of macrophages for wound healing. (a) Diagram showing the synthesis of NPs/miR-223*-loaded GelMA hydrogels and their application in wound healing. (b) Typical images of wounds treated with different groups after 0 and 12 days. Reproduced with permission from Zhu et al. 68 Copyright 2019 Wiley. (B) PAR30/miRNA-loaded HA hydrogels offered antibacterial, immunomodulatory, and miRNA delivery functions. Reproduced with permission from Wang et al. 105 Copyright 2022 Wiley.
MiRNA hydrogel delivery system in rhinitis treatment
Allergic rhinitis (AR) is a high-incidence respiratory inflammatory disease. 148 Recently, miRNA expression has been studied in AR due to the important regulatory role of miRNAs in immune system function. 149 To overcome the issues of naked miRNA, such as poor transmission ability and faster removal speed through nasal cilia, Su et al. 56 prepared Gel/NPs/miR-146a, a thermosensitive CS hydrogel loaded with miR-146a, which can rapidly form a binary hydrogel with good mucosal adhesion in the nasal cavity at 34°C, stably and continuously delivering miR-146a into the nasal mucosa after nasal administration. In terms of treating rat rhinitis induced by ovalbumin, the delivery ability of nucleic acids and the pharmacodynamic results were significantly enhanced in the Gel/NPs/miR-146a binary preparation compared to the other preparations (Figure 8).

A thermosensitive CS hydrogel loaded with Gel/NPs/miR-146a was prepared to treat allergic rhinitis(a) Mechanisms of miRNA-146 hydrogel delivery system in nasal mucosa. (b) Schematic representation of the temperature-sensitive miRNA-NPs/CS hydrogel for targeted delivery of miRNA-146 to the nasal mucosa. (c) The therapeutic effect was particularly visible in the AR + gel-NPs/miR-146a group. Reprinted with permission from Yu et al. 53 Copyright 2020 MDPI.
MiRNA hydrogel delivery system in periodontitis treatment
Periodontitis is a chronic inflammatory disease arising from the interaction between microorganisms and the host immune system characterized by gum inflammation and alveolar bone loss.150,151 MiRNAs are important regulators of inflammation and are involved in the pathogenesis of periodontitis. 152 Shen et al. 54 prepared an anti-inflammatory CS hydrogel loaded with dental pulp stem cell-derived exosomes (DPSC-Exos) for periodontal inflammation. DPSC-Exos/CS mixtures were translucent liquid at 4°C and transformed into hydrogels at 37°C. This special hydrogel could deliver DPSC-Exos, resulting in the conversion of macrophages from a proinflammatory to an anti-inflammatory phenotype in periodontal tissues and the repair of alveolar bone and periodontal epithelium in periodontitis mice. Further studies indicated that miR-1246 was enriched in DPSC-Exos and mediated the effect of DPSC-Exos (Figure 9).

MiRNA hydrogel delivery system in periodontitis. (a) A DPSC-Exo/CS hydrogel was synthesized to treat periodontitis. (b) Synesis of CS hydrogel. (c) RT‒qPCR showed that miR-1246 levels were higher than those of other miRNAs in DPSC-Exos. (d–e) Levels of the anti-inflammatory marker CD206 and the proinflammatory marker CD86 in BMDMs treated with DPSC-Exos + miR-1246 inhibitor. (f–g) Expression levels of CD206 and CD86 in BMDMs treated with the miR-1246 mimic. Reprinted with permission from Guo et al. 80 Copyright 2020 Elsevier.
MiRNA hydrogel delivery system in myocardial infarction treatment
Myocardial infarction (MI) is a common cardiovascular disease arising from myocardial ischemia.153,154 The heart reacts with an intense inflammatory response to remove the necrotic tissue caused by ischemic injury in MI, while inflammatory dysregulation or prolonged inflammation may lead to adverse cardiac remodeling and major adverse clinical events (MACEs).155- 157 MiRNAs have emerged as inflammatory regulators exhibiting great potential in MI. 158 An injectable polyethylene glycol hydrogel (Gel@MSN/miR-21-5p) containing the MSN/miR-21-5p complex was prepared by Li et al. 61 with a focus on treating myocardial infarction by exploiting the synergistic effects of combined inflammation inhibition and angiogenesis augmentation. This system provided the delivery and pH-triggered release of MSN/miR-21-5p complexes on demand from the hydrogel matrix in acidic infarct regions and then intracellularly delivered miR-21-5p from the complexes. The results demonstrated that Gel@MSN/miR-21-5p actively protected cardiomyocytes and reduced the production of several important inflammatory cytokines (TNF-α, IL-1β, and IL-6; Figure 10).

Gel@MSN/miR-21-5p significantly reduced the expression of inflammatory cytokines, such as (a) TNF-α, (b) IL-1β, and (c) IL-6, in the infarct area 1 day after MI. (d) Quantification was performed on the percentafes of cells that wre double positive for F4/80 and TNF-α, IL-1β, or IL-6. Adapted with permission from Su et al. 61 Copyright 2021 American Association for the Advancement of Science.
MiRNA delivery to cancer foci using hydrogel
Gel/hydrogel-based in situ biomaterial platforms show promise for postoperative cancer treatment and recovery, reducing the risk of adverse outcomes and poor prognosis. These techniques are well reviewed in the article by Feng et al. 159 If hydrogels can serve as delivery platforms for effective transport of cellular components such as DNA, RNA, and proteins, they have the potential to be used as cancer therapeutic agents. In this case, miRNA undoubtedly also holds significant promise as a cancer therapeutic agent. By virtue of the precise targeting and sustained release characteristics of the hydrogel, miRNA can be directly released in the postoperative tumor area, effectively inhibiting the growth and metastasis of tumor cells, while reducing systemic side effects. Combining this technique, the delivery of miRNA via hydrogels for cancer treatment demonstrates unique therapeutic advantages. Early studies associated with cancer and inflammation examined the procancer effects of inflammation. 160 Chronic inflammation caused by persistent infection and metabolic disorder-induced angiogenesis and genomic instability are known to lead to increased cancer risk and accelerated cancer progression. 161 MiRNAs may not only participate in signaling pathways affecting a range of cellular phenotypes by acting as inflammatory factors but also serve as effective regulators of immune cell function. 162 The secretion/uptake of particular miRNAs by immune cells may decrease the immune surveillance of cancer cells, leading to a loss of control over cell proliferation and differentiation and further promoting tumor formation and metastasis.163,164 Tumor-associated macrophages, an important component of the tumor matrix, are a key mediator of the connection between inflammation and cancer.165,166 Direct evidence has demonstrated that miRNA, in addition to the important regulatory role in TAMs, may also link inflammation and tumorigenesis. In addition, the CD44 receptor, an integral membrane glycoprotein on macrophages, can specifically bind to HA, which has a wide variety of applications in tumor treatment. 167 HA with different molecular weights has attracted tremendous attention in delivering various anticancer drugs, siRNAs and miRNA targeting to cancer because of its properties, such as pH sensitivity, enzymatic degradation, and especially tumor targeting ability. 168 Therefore, utilizing HA-delivered miRNAs has reversed macrophage polarization to obtain favorable phenotypes as an effective strategy for treating certain cancers. In non-small cell lung cancer, M2-type macrophages are dominant. Parayath et al. 169 prepared HA-PEI NPs to deliver miR-125b for treating a non-small cell lung cancer model. After intraperitoneal injection, HA-PEI nanoparticles containing miR-125b target CD44 receptors and are thus phagocytosed by abdominal macrophages and carried to the lung, where they can repolarize lung tumor-associated macrophages from M2 to M1 and contribute to the antitumor efficacy of therapeutic agents for non-small cell lung cancer. In addition, the use of hydrogels to deliver miRNAs for cancer therapy can avoid causing inflammation at the surgical site. For example, Ding et al. respectively customized three hydrogels for in vivo delivery of a miR mimic (tumor suppressor miRNA)/an antagomiR (oncomiR inhibitor), tumor suppressor miRNA-205/miRNA-221, and miR-96/miR-182 for treating breast cancer.170–172 The absence of inflammation at the surgical site before and after hydrogel implantation was noted, indicating good in vivo biocompatibility of these materials without eliciting severe immune or toxic reactions. At present, while previous studies have revealed part of the mechanism of miRNA hydrogel delivery systems for cancer treatment, the answer to this question remains incomplete. The specific treatment of cancer and the rational prevention of inflammation through miRNA hydrogel delivery systems is indeed an important issue that urgently requires further exploration.
Applications in other inflammatory diseases
A persistent local inflammatory response appears early during intervertebral disc degeneration (IDD). 173 Wang et al. 174 synthesized an injectable gelatin-based hydrogel to deliver curcumin, a naturally occurring anti-inflammatory, and antagomir-21, a cholesterol-modified miRNA-21 inhibitor, for locally and continuously treating IDD. This delivery system was successfully prepared by mixing mPEG-TK-PLGA (ROS-responsive amphiphilic polymers) and curcumin in dimethyl sulfoxide (DMSO) to prepare micelles@curcumin (MIC@Cur), followed by wrapping MIC@Cur and antagomir-21 in hydrogel. Interestingly, the sustained release of the coated antagomir-21 could modulate MAPK/ERK signaling by controlling the transcription of PTEN, thereby inhibiting ECM degradation and reestablishing ECM metabolic balance in nucleus pulposus cells (Figure 11). In a subsequent report, Wang et al. 175 further developed an in situ crosslinked tannic acid nanoparticle (TA NP) hydrogel delivery system for sustainably releasing TA NPs@antagomir-21 on demand, which significantly alleviated local inflammation and relieved symptoms of IDD (Figure 12).

An injectable inflammatory reactive gelatin-based hydrogel was used to deliver naturally derived anti-inflammatory curcumin and antagomir-21 for IDD. (a) Schematic representation of inflammation-responsive hydrogels loaded with curcumin and miRNA. (b) The expression of M2 marker genes (CD206 and CD163) and M1 marker genes (TNF-α and IL-1β) was treated with curcumin-loaded hydrogels. (c) Image showing that the curcumin-loaded hydrogel upregulated the expression of CD206. Reproduced with permission from Wang et al. 174 Copyright 2022 Elsevier.

An injectable hydrogel containing TA NPs was developed for on-demand delivery of antagomir-21 for IDD. (a) Schematic diagram of the hydrogel design. (b) qRT‒PCR, (c) western blotting, (d) heatmap, and (e) immunofluorescence staining showed that the TNF-α expression level was downregulated after treatment with TA NPs/hydrogel, indicating their anti-inflammatory potential. Adapted with permission from Wang et al. 175 Copyright 2023 Elsevier.
Spinal cord injury (SCI) can be caused by inflammation, which hinders its functional recovery. Wan et al. 176 designed the gold nanoparticles and self-assembling peptide hydrogel composite scaffold (PEG-SH-GNPs-SAPNS@miR-29a delivery system) to repair damaged spinal cord by constructing a regenerative microenvironment for recruiting endogenous neural stem cells (Figure 13). Sulfhydryl polyethylene glycol (PEG-SH) is utilized to enhance the biocompatibility and stability of gold nanoparticles (GNPs), enabling them to more effectively penetrate and function within damaged spinal cord tissue. Gold nanoparticles (GNPs) are effective nano-carriers capable of carrying miR-29a to the target site and protecting it from enzymatic degradation at the site of spinal cord injury due to their high drug loading capacity and targeting abilities. The self-assembling peptide nanofiber scaffold (SAPNS), a composite scaffold consisting of self-assembling peptide hydrogel, creates a protective microenvironment that enhances the stability and bioactivity of miR-29a. The PEG-SH-GNPs-SAPNS delivery system enables the efficient and precise delivery of miR-29a to the site of spinal cord injury. As a result, miR-29a induces a variety of biological effects, such as promoting nerve regeneration and inhibiting inflammatory responses. In conclusion, the PEG-SH-GNPs-SAPNS@miR-29a delivery system presents a novel approach and method for treating spinal cord injury, demonstrating potential clinical application value and prospects.

PEG-SH-GNPs-SAPNS@miR-29a delivery system for spinal cord injury. (a–c) The image of the five groups in the length, density, and the number of neurites in vivo. (d) The recovery of hind limb motor function in each group. (e–f) The SEP latency and MEP latency in each group were detected before and 56d after the operation. Adapted with permission from Wan et al. 176 Copyright 2023 Elsevier.
As mentioned above, relevant studies have shown that miRNAs exert effects on the regulation of inflammatory diseases through anti-miRNA inhibitory therapies inhibiting specific endogenous miRNAs or miRNA replacement therapies using miRNA mimics to provide an exogenous source of miRNAs. The hydrogels can achieve a better therapeutic effect on miRNA encapsulation. Hence, for the miRNA hydrogel delivery system to treat inflammatory diseases, clever coordination among the three elements of miRNA, cytokines, and hydrogel materials is needed to determine a strategy suitable for the repair of inflammatory tissue, thereby benefiting human beings through hydrogel engineering.
Progress and challenges in clinical transformation of miRNA hydrogel delivery system
Clinical translation remains a major goal in the development of drugs and their delivery systems. Hydrogel delivery systems have a wide range of potential applications in drug delivery, with several systems having already been clinically translated, including oncology therapy, wound dressing, tissue engineering, and drug-eluting scaffolds.14,177 In recent years, hydrogels have matured considerably in the field of biomedicine and are widely utilized as carriers for nucleic acid drugs. Hydrogel delivery systems show promise as a platform for miRNA delivery, yet there have been no direct instances of clinical translation. The hydrogel delivery system is a promising miRNA delivery platform, although there are currently no direct clinical translation cases. However, with its excellent stability, controlled release, targeted distribution, and biocompatibility, the hydrogel method for miRNA delivery has great potential for clinical translation. For example, Fan et al. 178 developed a dextran-based hydrogel to encapsulate hADSCs transfected the reverse sequence of miR-29A (miR-29A inhibitor), with the purpose of promoting chondrogenesis of hADSCs and new cartilage matrix production in vitro and in vivo. The encapsulated hADSCs showed good viability, bioactivity, and differentiation properties in glucan-AN hydrogel. The hydrogel promoted chondrogenesis within 8 weeks. 178 In a separate study, Xiao et al. 179 investigated an injectable triblock polymer (hydrogel) that is composed of polyglycolic acid copolymer and polyethylene glycol loaded miR-138 modified stem cell exosomes (Exos-138). This system could reduce the level of inflammation in BV-2 cells through the NLRP3-caspase1 signaling pathway, and reduce neuronal apoptosis by downregulating intracellular reactive oxygen species levels through the Nrf2-keap1 signaling cascade. The results showed P-Exos-138 hydrogel promoted nerve recovery in SCI rats.
The research on miRNA hydrogel delivery systems has shown promise, but the clinical translation of miRNA hydrogel drug delivery system therapy still faces significant barriers. In the clinical translation of miRNA hydrogel delivery systems, although hydrogels provide various advantages as delivery vehicles, including favorable biocompatibility, controllable degradation rates, and ease of local drug administration, the inherent characteristics of miRNAs significantly influence the overall system’s success.
No miRNA drugs have yet successfully entered Phase III clinical trials in the research and development process of miRNA therapy. Therefore, the clinical translation of miRNA therapy still faces some challenges. 180 The successful delivery of miRNA molecules is one of the main challenges. Negatively charged miRNA molecules have difficulty crossing negatively charged cell membranes alone, leading to the blockage of miRNA uptake by cells. MiRNAs may exhibit multiple targets, a short half-life, and adverse immune responses under physiological conditions, making them prone to undesired targeting or off-target effects. 31 Among endogenously generated miRNAs, one miRNA may have several targets, resulting in poor specificity for intrinsic mechanisms. 181 Thus, humans cannot easily choose specific silencing tasks, which makes it difficult to achieve the precise targeting of miRNAs for gene silencing. And endogenous miRNAs cannot be accurately extracted and quantitatively determined. Therefore, accurate monitoring during miRNA delivery is crucial for identifying any potential off-target effects that may arise in the miRNA system delivery process. Another important point is that the physical embedding of miRNA easily causes problems such as miRNA difficulty or early release and the need for transfection agents, which may have potential cytotoxicity.
Additionally, for this hydrogel carrier itself, it remains a great challenge of further clinical translation, including hydrogel production and storage, material characterization, and biological effect. 182 It is worth mentioning that current chemically cross-linked hydrogel systems are not absolutely bioinert to the surrounding tissues due to adverse effects on the metabolic activity of cells or the bioactivity of growth factors during in situ hydrogel formation. Several approaches have been developed to minimize secondary harm from hydrogel formation and degradation. Good biocompatibility is a prerequisite to avoid immune rejection or the production of toxic substances. 183 Exosomes which contained miR-4645-5p being delivered through a hydrogel was toward the activity of sterol response element binding protein 2 (SREBP2) inhibition to regulate the polarization of macrophages to the M2 phenotype and further accelerate and promote diabetic wound healing by Shi et al. 184 The hydrogel was synthesized from highly biocompatible and naturally-derived biological compounds including gallic acid and CS. Biodegradable and controllable degradation rate are important for the hydrogels used as miRNA carriers, because the hydrogel carriers need to support soft tissue formation and minimize long-term foreign body reactions, and the release rate can be regulated by rate.179,185 Hydrogels made from synthetic polymers are excellent alternatives for natural materials because as they offer the key advantage of precisely controlled degradation times. 186 At last, implanted scaffolds that tend to be cytotoxic or to be harmful to the tissue are often created in chemical cross-linking, such as with glutaraldehyde. 187 Physical cross-linked methods (crosslinking using physical interactions, such as ionic interactions, hydrogen bonds, or hydrophobic interactions) for preparing hydrogels are alternative solution to the toxicity of crosslinkers. 188 Sener et al. 189 demonstrated a biomaterial system of amphiphilic ionic cryohydrogels (i.e. hydrogels polymerized at freezing temperature) enriched with cerium oxide nanoparticles (CNPs)-miR146a based on physical crosslinking by removing the crosslinker molecules from the reaction and polymerizing the hydrogels at freezing temperatures. The above hydrogels were locally applicable, injectable, self-healing, and provided a sustained release of miRNA-146a-loaded CNPs, which were proved to accelerate the healing time of diabetic wounds and significantly reduce inflammation occurs.
Moreover, in the context of miRNA hydrogel delivery systems as a whole, the ideal transition kinetics should be explored to prevent the effect of dilution of the solution by body fluids in vivo after in situ injection of the hydrogel in the solution state. The hydrogel biodegradation and miRNA loading capacity of the hydrogel delivery system are also worthy of attention. The selection of suitable hydrogel materials and the composite ratio of nucleic acids and polymers have become pivotal. In addition, due to the intricacy of the disease recovery process, multiple processes, and the factors involved also change constantly, making it challenging for a miRNA to simultaneously serve the requirements of the whole process. For instance, regulating the polarity of macrophages is an important tumor suppressor measure, but tumor-associated macrophage phenotypes change with tumor progression. Thus, miRNAs and other drugs loaded into hydrogels are commonly needed only at specific times, whereas at other times, they may even have opposite effects.
In conclusion, the specificity, stability, and immunogenicity of miRNA are critical factors influencing its therapeutic efficacy, while the biocompatibility, drug release kinetics, and the preparation process and cost of hydrogels affect their efficiency and feasibility as delivery systems. Moreover, attention should be paid to the potential challenges in clinical translation that may arise from the combination of miRNA and hydrogels. In general, the clinical translation challenges of miRNA hydrogel delivery systems exist in both miRNA itself and the hydrogel as a delivery system, as well as the complexity arising from their combined application. Hence, for the successful clinical translation of miRNA hydrogel delivery systems, a comprehensive consideration of these challenges is essential during their development and application. Overcoming these difficulties necessitates interdisciplinary collaboration, rigorous preclinical and clinical validation, adherence to regulatory requirements, optimization of economic and technical strategies, and enhancement of patient acceptance and market demand.
Conclusion
With the advancement of biomedical technologies, carriers such as Lipid nanoparticles have been successfully utilized for mRNA delivery, opening possibilities in the field of gene therapy, as described by Kubiatowicz et al. 110 In particular, hydrogel technology offers a novel approach for miRNA delivery, with its stable delivery and slow-release properties being particularly suitable for long-term and sustained gene expression regulation, indicating broad potential applications in the biomedical field. Using hydrogels as carriers for nucleic acid drugs in the in vivo delivery of miRNA can effectively reduce adverse factors, provide necessary protection for easily degradable miRNA, and further optimize the therapeutic effects of miRNA, offering strong support for the future of gene therapy. Hydrogels have emerged as a high-profile, highly versatile novel drug delivery system for miRNAs that can combine different miRNA physiological functions with the benefits of hydrogels to achieve the targeted delivery of miRNAs.
Hydrogel delivery systems offer advantages in that they allow the selection of different characteristic polymeric materials based on the needs of different inflammatory diseases, making them highly targeted and effective. With the deepening of research, there is a growing interest in studying the functions of various types of miRNA hydrogel delivery systems to achieve more effective interventions. In conclusion, optimizing hydrogel formulation and preparation parameters, using more mature disease models, and establishing more perfect in vitro and in vivo screening methods, resulting in local and sustained miRNA delivery for crucial gene silencing, will greatly increase progress in the treatment of inflammatory diseases in the future study of miRNA hydrogel delivery systems.
Footnotes
Abbreviations
3D, three-dimensional; miRNA, microRNA; ECM, extracellular matrix; CS, chitosan; HA, hyaluronic acid; HAMA, methacrylic anhydride; DEGMA, di(ethylene glycol) monomethyl ether methacrylate; SMSC, synovium mesenchymal stem cell; CS-SMSC-126-Exos, CS hydrogel loaded with SMSC-126-Exos; PLGA, polylactic-co-glycolic acid; pHEMA, polyhydroxyethyl methacrylate; PVA, polyvinyl alcohol; si-Noggin, siRNA against noggin; SDF-1α, stromal cell derived factor-1α; CTH/antimiR-138 NPs, CS/tripolyphosphate/HA/antimiRNA-138 nanoparticles; LMWGs, low relative molecular mass gel factors; SAP, self-assembling peptide; PEG, poly(ethylene glycol); Chol-miR-26a, cholesterol-modified noncoding miR-26a; UV, ultraviolet; MSCs, mesenchymal stem cells; PAR30, polymerization degree of 30; PEI, polyethylenimine; GO, graphene oxide; PNP, polymer nanoparticle; MSN, mesoporous silica nanoparticle; PEG-PLGA-PNIPAM, poly(ethylene glycol)-b-poly(lactic-co-glycolic acid)-b-poly(N-isopropylacrylamide); ASP, aspirin; GelMA, gelatin methacrylate; hUC-MSCs-sEVs, human umbilical cord mesenchymal stem cell-derived small extracellular vesicles; AuNPs, gold nanoparticles; ROS, reactive oxygen species; I/R, ischemia/reperfusion; OA, osteoarthritis; ACLT, anterior cruciate ligament transection; GNP, gelatin nanoparticle; ADSC-Exos, exosomes from adipose-derived stem cells; SSRP1, structure-specific recognition protein 1; COX, cyclooxygenase; M1, proinflammatory macrophages; M2, anti-inflammatory phenotype; miR-223*, miR-223-5p mimics; AR, allergic rhinitis; DPSC-Exos, dental pulp stem cell-derived exosomes; MI, myocardial infarction; MACE, major adverse clinical event; IDD, intervertebral disc degeneration; DMSO, dimethyl sulfoxide; MIC, micelle; Cur, curcumin; TA NP, tannic acid nanoparticle; SCI, spinal cord injury; PEG-SH, sulfhydryl polyethylene glycol; GNPs,gold nanoparticles; SAPNS, the self-assembling peptide nanofiber scaffold; SREBP2, response element binding protein 2; CNPs, cerium oxide nanoparticles.
Author contributions
SH and YL conceived the idea, analysis of literature, and writing of the manuscript; JC, XG, YZ, and LW collected and read the literature and revised the article. MZ and ML read through and corrected the manuscript. All authors contributed to the article and approved the submitted version.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant numbers 82370419, 81800434), Sichuan Science and Technology Program (2022YFS0627), and the Natural Science Foundation of Sichuan Province (2022NSFSC0364). Figures were created with ![]() .
.
Data availability statement
All data generated or analyzed during this study are included in this published article and its Additional Files.
