Abstract
In vitro modeling of brain tissue is a promising but not yet resolved problem in modern neurobiology and neuropharmacology. Complexity of the brain structure and diversity of cell-to-cell communication in (patho)physiological conditions make this task almost unachievable. However, establishment of novel in vitro brain models would ultimately lead to better understanding of development-associated or experience-driven brain plasticity, designing efficient approaches to restore aberrant brain functioning. The main goal of this review is to summarize the available data on methodological approaches that are currently in use, and to identify the most prospective trends in development of neurovascular unit, blood-brain barrier, blood-cerebrospinal fluid barrier, and neurogenic niche in vitro models. The manuscript focuses on the regulation of adult neurogenesis, cerebral microcirculation and fluids dynamics that should be reproduced in the in vitro 4D models to mimic brain development and its alterations in brain pathology. We discuss approaches that are critical for studying brain plasticity, deciphering the individual person-specific trajectory of brain development and aging, and testing new drug candidates in the in vitro models.
Keywords
Introduction
Modeling the brain tissue in vitro is a challenging problem in modern neurobiology and neuropharmacology. Complexity of the brain structure and diversity of cell-to-cell communication in various conditions make this task almost unachievable. However, development of adequate in vitro brain models would ultimately lead to better understanding of brain plasticity, efficient selection of new drug candidates, and further progress in neurobioengineering.
Phenomenon of brain plasticity is based on an ability of brain to modify its structure or function in a response to the action of various exogenous or endogenous stimuli. 1 This is a fundamental property of the brain that is based on synaptic transmission, synapse establishment and elimination, growth and retraction of cellular processes, neurogenesis, gliogenesis and cell death, angiogenesis and recession of microvessels, modulation of intracellular pathways and intercellular communication, adaptation of metabolism to the current needs of the particular brain region or neuronal circuit.2,3 Therefore, reconstruction of brain tissue in vitro is facing to the problems related to the complexity in reproducing the numerous mechanisms leading to significant changes in the brain tissue architecture and functional activity upon action of numerous external stimuli. Even there is a recent technological leap from the simple planar (2D) in vitro models to more complicated 3D ones,4,5 it seems to be insufficient to achieve the main goal of the in vitro modeling—to recreate the brain activity as an integral result of information perception, processing and responding. This is particularly important in neuropharmacology where correct assessment of a drug pharmacodynamics and pharmacokinetics at the preclinical stage is crucial for choosing the appropriate strategy and further improvements.
Various approaches have been already established to model the brain tissue in the in vitro conditions either in the transwell format, on microfluidic chips, in 3D hydrogels, or as cerebral organoids derived from the human induced pluripotent stem cells. Thus, we are not going to analyze their advantages and limitations here just addressing the readers to several up-to-date reviews.6–16 Due to apparent difficulties in the reconstruction of the whole brain tissue in an in vitro system, significant attempts have been paid to establish the in vitro models of some brain compartments like neurovascular unit (NVU), blood-brain barrier (BBB), blood-cerebrospinal fluid barrier (BCSFB), neurogenic niches (NNs).17 –19 In this review we focus on mechanisms of brain plasticity that are controlled by the structural and functional integrity of tissue barriers, and how this phenomenon could be properly reproduced in the in vitro brain models.
Complexity of the neurovascular unit and key brain tissue barriers
NVU consists of brain microvessel endothelial cells (BMECs), pericytes, perivascular astrocytes, other glial cells (oligodendrocytes and microglia), neurons, and extracellular matrix. It is a platform for diverse intercellular communications in the brain. As an example, activated neurons release glutamate and potassium that are taken up by astrocytes, they produce lactate which is used by neuronal cells to support energy production, or by endothelial cells to control the blood flow and BBB integrity.20–22 Thus, NVU could be considered as a self-regulatory compartment within the brain tissue where almost all the basic neurobiological and plastic phenomena are realized: neuronal excitability, neuron-astrocyte metabolic coupling, gliovascular control, immune recognition, neurogenesis/gliogenesis. Therefore, the general goal to reproduce the changeable brain which is sensitive to the action of external stimuli in the in vitro conditions may be reduced to the task on modeling the NVU.
Structural and functional integrity of the blood-brain barrier
BBB is recognized as a part of the NVU with the main focus on functional and structural integration of BMECs and contacting cells (pericytes and astrocytes). BMECs are coupled via tight, gap and adherens junction proteins that control paracellular permeability. 23 In addition, BMECs express numerous transporters serving for membrane-assisted transcellular permeability. BMECs possess rather high density of mitochondria, low fenestration rate, transferrin and insulin receptors, and highly controlled permeability. 24 BMECs metabolism and functional activity affect the integrity of the BBB and control blood supply in active brain regions.25,26 Pericytes and perivascular astrocytes cover the layer of BMECs and provide tight regulation of their integrity, permeability and metabolic plasticity. Their contribution to the barrier functionality has been described in details elsewhere.21,22,27,28
The commonly accepted view on the BBB as a mechanical barrier in the brain is gradually replaced with the understanding that BBB and NVU, in general, are the regulated and interactive entity of the brain. 29 Such shift in a paradigm is partially related to the accumulating data on heterogeneity of NVU in various brain compartments. Particularly, gray matter is characterized by greater glucose consumption, higher vascular density, reduced expression of expression of junctional proteins occludin, claudin-5 and α-catenin in BMECs, lower structural integrity of endothelial layer, but higher resistance to the agents inducing BBB breakdown, protoplasmic astrocytes with numerous fine processes. 30 Within the neurogenic niches of the adult brain, astroglial coverage of BMECs is intrinsically defective in the subventricular zone (SVZ), but is rather tight in the hippocampal subgranular zone (SGZ). 31 Also, direct contacts of astroglial end-feet and BMECs that affect the efficiency of neurovascular coupling differ in various cortical layers. 32 The electrical resistance of BMECs is 1500–8000 Ω∙cm2 in vivo (Table 1), thereby suggesting high level of the BBB structural integrity.33,34
Key differences in the structure and function of the BBB and the BCSFB.

TEER: transendo(epi)thelial electrical resistance; BM: basal membrane.
Reported in vivo TEER values reflect data for amphibians (frog species) and mammals (rat species) as for BBB and only for amphibians (bullfrog species) as for BCSFB.
Reported in vitro TEER values reflect data for mammals: for human and rat species as for BBB and only for rat species as for BCSFB.
Structural and functional integrity of the BBB is under the control of NVU components, but, vice versa, BBB permeability affects the activation of neuronal and glial cells due to transport of cytokines, growth factors, metabolites and transmitters to the loci of stem/progenitor cells maintenance and recruitment.9,35 Almost all the central nervous system disorders (neurodevelopmental, neurodegenerative, inflammatory) are characterized by elevated BBB permeability which is recognized as a sign of severe brain tissue alterations.
Structural and functional integrity of the blood-cerebrospinal fluid barrier
Another barrier in the brain—blood-cerebrospinal fluid barrier (BCSFB)—consists of the barrier cells of the arachnoid membrane and choroid plexus epithelial cells that are linked with tight junctions. In contrast, choroid plexus capillary endothelial cells are not tightly-coupled, therefore, they can’t provide efficient barrier function. The choroid plexus (CP) locates in each of the two lateral ventricles of the brain, in the third and fourth ventricles. Ependymocytes, along with microvascular cells, form the choroid plexus, whereas the ependymal membrane (ependyma) separates the cerebrospinal fluid and the CNS parenchyma. Despite a similar embryological origin, ependymal cells and epithelial cells of the choroid plexus have certain differences. First, the choroid plexus epithelial cells are tightly-coupled near their apical surface. 36 Second, the differentiation of choroid plexus epithelial cells from their neuroepithelial progenitors during embryogenesis is accompanied by the expression of several types of enzymes and secretory proteins, the most important of which is transthyretin, a protein that provides thyroxine transport. 37 Third, the choroid plexus epithelium expresses specific receptors that act as keys for the transduction of humoral signals between the blood and the CNS, including growth hormones, insulin, insulin-like growth factor, prolactin, vasopressin, and leptin. 38 Forth, these cells express a huge number of specific transporters and ion channels on their apical surface. This polarized distribution of transport systems helps to move complexes of bioactive molecules across the BCSFB and wash out toxins and other potentially harmful substances from the CNS. 39
Choroid plexus provides synthesis and secretion of cerebrospinal fluid (CSF) (approx. 0.4 mL/min per gram of tissue, or up to 500 mL in adult humans per day), 40 and exchange of molecules between the blood and the CSF. BCSFB controls ultrafiltration of blood plasma components from capillaries into the lumen of brain ventricles and production of CSF, secretion of some peptides and proteins into CSF as well as absorption of various substances from CSF. 41 For instance, up to 20% of CSF in the human brain originates from the brain interstitial fluid (ISF), and there is an important mechanism to remove metabolites and potentially toxic substances from the brain parenchyma: CSF goes into the brain tissue alongside the perivascular spaces up to microvessels, then due to activity of AQP4 in perivascular astrocytes CSF is transferred into the interstitial space to refill ISF. 42 Finally, the fluid leaves the brain tissue at the level of veins: through ependymal cells into ventricles, via pia-glial membranes into the surface of the brain and spinal cord, and by means of glymphatic system into extracranial lymph nodes. 42
Choroid plexus epithelial cells (CPECs) are originated from the ventricular ependymal cells and are considered as modified ependymal cells. 43 Ependymal cells include multiciliated cells of the cubic epithelium lining the cavities of the ventricles of the brain and the central canal of the spinal cord of vertebrates. More than half-century ago, it was shown that due to the movement of cilia, ependymocytes create the flow of cerebrospinal fluid inside the lateral ventricles of the brain, 44 then, the protective function of the ependyma was clearly demonstrated as an ability of these cells to regulate the transport of molecules between the cerebrospinal fluid and choroidal capillary. 45 This ability is provided by the presence of different types of intercellular contacts and anchor proteins: (i) in the apical part, the cells are interconnected by means of adherens junctions; (ii) in the middle part, there are gap junctions formed by connexons; (iii) within the BCSFB, ependymocytes are connected with tight junctions.46,47 Electrical resistance of CPECs is just 150 Ω∙cm2 in vivo (Table 1). 34
CPECs cells express various types of ion channels and transporters for the transfer of glucose, fructose, lactate, urate, several ions, and for the tightly-controlled secretion of CSF. These cells are well-equipped with enzymes—cytochromes P450—needed for the metabolism of endogenous hydrophobic molecules and xenobiotics.34,48,49 Presumably, they should express wide spectrum of receptors sensitive to metabolites produced locally or transferred from the peripheral blood. For instance, recent data suggest that CPECs as well as ependymal cells express lactate GPR81 receptors, 50 thereby they are able to sense lactate which is considered as a key molecule in intercellular metabolic coupling and brain plasticity. 21
Ependymal cells (EpCs) locate at the boarder of the cerebral ventricles and the brain parenchyma (Figure 1). In the adult brain, ependymocytes express stem cell markers such as integrin-beta-1.51–53 Being the actual barrier between the CSF and brain cells, EpCs differ in the frequency of movement of their cilia, which is characteristic of a particular type of ependymocytes and remains constant throughout life. A decrease in cilia movement (e.g. when cells are exposed to toxic agents like alcohol) leads to the accumulation of CSF and an increase in the volume of the brain ventricles. 54 Disfunction of the ciliary apparatus leads to dramatic consequences: altered production and movement of cerebrospinal fluid, as well as impaired neurogenesis caused by deregulated production and secretion of regulatory molecules needed for the establishment of pro-neurogenic microenvironment in the nearest neurogenic niche—subventricular zone.55 –59

Principles of structural and functional organization of BBB and BCSFB. Blood-CSF barrier, consisting of choroid plexus (CP) endothelial cells (ECs) and choroid plexus epithelial cells (CPECs), ensures its barrier function by tightly coupled CPECs, whereas ECs are highly fenestrated. In contrast, integrity of the blood-brain barrier is determined by endothelium where brain microvessel endothelial cells (BMECs) are contacted via tight junctions. The ventricular barrier (VB), located right between BBB and BCSFB, has low integrity since ependymal cells (EpCs) are mainly coupled via gap junctions and have high paracellular permeability.
In contrast to BMECs and CPECs, the ependymal cells that line the ventricular wall to separate the CSF from the brain ISF are interconnected with gap junctions, making them permeable to most substances, including macromolecules. 34 EpCs express functional purinergic receptors (P2X7) that are sensitive to extracellular ATP, therefore, they could be activated by massive ATP release from damaged cells in neuroinflammation or brain lesions. 60 ATP itself or adenosine, which is a metabolite of ATP acting at A2B receptors in EpCs, stimulate ciliary beat frequency, thereby affecting CSF dynamics. 61 Stabilizing role of ependymal cells on the integrity of the ventricular wall has been shown: disruption of the ependyma leads to obliteration of the cerebral aqueduct and hydrocephalus. 62 Some specialized ependymal cells, such as tanycytes and ependymocytes of the CP, provides neuroendocrine and sensory function. 63 In the cooperation with CPECs, ependymal cells protect the brain fluid environment from toxic compounds which is particularly important in the developing brain. 64
Thus, the complexity of the structure of key barriers in the brain tissue is associated not only with the presence of cells that are diverse in morphology, physiology and expression profile, but also with the permanent changes in their functional activity that are determined by the brain plasticity. In other words, the reproduction of an adequate model of such complex systems is impossible without taking into account the action of factors that initiate short-term and long-term changes within the NVU: (i) excitation of neurons accompanied by glial activation; (ii) release of neuro- and gliotransmitters, metabolites, cytokines, growth factors, exosomes into the extracellular space; (iii) dynamic changes in the composition of extracellular matrix; (iv) modulation of neurogenesis and cerebral angiogenesis; (v) activity-driven changes in local microcirculation, production and flow of CSF and ISF.
Dynamic changes in the brain compartments affected by BMECs, CPECs, and EpCs
Establishment of microfluidic in vitro systems allows reproducing some characteristics of fluids dynamics within the tissue. Blood flow is an important regulator of BMECs viability and functional competence, whereas CSF/ISF production and flow are needed for the adequate functioning of brain parenchyma. Attempts to reproduce the latter phenomenon, as a rule, face serious gaps in understanding the general mechanisms of CSF/ISF dynamics.
Fluid dynamics in the brain, compartmentalization of water in the brain, and CSF flow show circadian/diurnal characteristics: water diffusibility is higher at night and associates with sleep midpoint (the circadian marker). 65 CSF flux is higher in the resting period compared to the active period. 66 Generation of circadian rhythms is provided by neurons in the suprachiasmatic nucleus (SCN). 67 Moreover, CP is now considered as a part of the circadian clock in the brain. The machinery of circadian rhythmicity is based on the transcriptional activator CLOCK-BMAL1 (Circadian Locomotor Output Cycles Kaput; Brain and Muscle ARNT-Like 1) that are needed for the transcription of clock proteins PER1 and 2 (Period 1 and 2) bound to CRY1 and 2 (Cryptochromes 1 and 2). 68 CP cells express all the components of the circadian rhythm machinery in a gender-dependent manner. 69 Deregulated expression of BMAL1 and PER2 has been detected in experimental Alzheimer’s disease. 70 In the in vitro co-cultures of CP and suprachiasmatic nucleus (SCN) cells, the long period of rhythms in the SCN is restored to the level of the behavioral circadian period in the presence of CP cells that are equipped with the connexins 43 (Cx43) and, probably, other connexins which support synchronization, therefore, SCN circadian clock is controlled by some signaling molecules released by CP cells into the CSF. 71 Circadian rhythms controlled by SCN and other brain structures (retina, CP, cortex, hippocampus, nucleus accumbens) could regulate brain fluids dynamics, CSF clearance, activity of membrane transporters, dopaminergic signaling and reward system feeding behaviors, drug metabolism.68,69,72 CP is an additional source of melatonin production (even with non-circadian pattern) in the brain. 73 There is a hypothesis that melatonin released from CP in nigdht could affect water permeability of the BCSFB. 69 Actually, BBB also demonstrates dependence on the circadian rhythmicity: transporters activity and BBB permeability undergo daily oscillations.74 –76 Expression of xenobiotic-metabolizing cytochromes P450 (Cyp4x1 and Cyp2c11) in BMECs shows circadian/diurnal dynamics, 77 so, the efficiency of drugs seems to be different at night or day time as it was demonstrated for anti-epileptic medicines. 76 BMECs express the genes encoding for the proteins regulating circadian rhythm that affect BBB permeability and xenobiotics metabolism.77,78
However, some other studies suggest that expression of junction proteins and transporters might have no dependence on circadian rhythms, but expression pattern of proteins involved in translation, angiogenesis and energy production demonstrate diurnal variations. 79 As it was shown on the in vivo chemogenetically activated rodent neurons, BMECs change their expression pattern after stimulation of NVU neurons: the expression of adherens junctions and focal adhesion proteins, regulators of cytoskeletal dynamics is upregulated, whereas neuronal activity inversely correlates with the expression of ABC efflux transporters (incl. Pgp), LRP1, and PAR bZip circadian-clock-regulated transcription factors. Thus, the net efflux transport through BMECs decreased with the activity in organisms demonstrating diurnal oscillations (e.g. in flies, mammals). 80 But in a contrast to endothelial microvascular cells located in peripheral organs, BMECs have much lower amplitude of clock gene expression, 79 therefore, in general, circadian rhythm-related event could be more expressive in the BCSFB, but not in the BBB.
It should be noted that for the appropriate functioning, BMECs, CPECs, and EpCs require high energy resources, for instance, CP receives its blood supply up to 4 mL/min per gram which is 5–10 times higher than the blood supply rate in the brain parenchyma 34 ; the density of mitochondria in BMECs and CPECs is up to 12%–15% of the total cell volume which is much higher than in peripheral endothelial or epithelial cells.58,81 Since energy production is coupled to dynamic changes in NAD+/NADH ration in cells, the barrier cells should depend on the availability of NAD+. Indeed, it was demonstrated in endothelial cells needed in NAD+ for angiogenesis and sprouting. 82 Availability of NAD+ in cells depend on NAD+ producing enzymes (e.g. NAMPT—nicotinamide phosphoribosyl transferase which provides a rate-limiting step in the NAD+ salvage pathway) and NAD+-consuming enzymes (e.g. CD38, CD157, PARP (poly(ADP)-ribose polymerase), SIRT (sirtuin/histone deacetylase), and is directly linked to the activity of glycolysis and oxidative phosphorylation (OXPHOS) providing intracellular NAD+ regeneration.7,24 NAD+ concentrations in the CSF are thought to reflect pathological changes in the brain tissue, but the data are rather controversial since they have been reported to be lowered in a case of aging, neurodegeneration, oxidative stress and inflammation or increased in some cases of neurodegenerative pathology like Huntington’s disease.83,84
NAD+ intracellular concentrations have been shown to regulate circadian rhythm by affecting the activity of SIRT1 and histone acetylation. It is connected to the competition of two enzymes—SIRT1 and PARP1—for the same substrate—NAD+. Thereby, excessive activation of PARP1 (e.g. caused by DNA damage) results in circadian phase advancements (stimulating effect on the circadian rhythm), and NAD+ levels and acetylated histones oscillate in antiphase. Moreover, it was found that NAD+ levels oscillate by approximately 40% during each circadian cycle in response to oscillations in NAMPT protein levels. 85 Energy metabolism also shows clear dependence on the circadian rhythm: glycolytic activity and lactate production in cells are increased, but OXPHOS is suppressed in the absence of BMAL1, but decreases when CRY1 and CRY2 are inactivated. All these effects are linked to the circadian regulation of NAD+-dependent SIRT3 activity and mitochondrial proteins acetylation. 86 NAD+ and NAMPT levels oscillate during the daily 24-h cycle. 87 It is interesting that in plant cells, another metabolite of NAD+ - cyclic ADP-ribose which is produced by CD38/NAD+-glycohydrolase—takes part in the circadian rhythm control (Ca2+-mediated feedback loop). 88 Thus, intracellular NAD+ pool is regulated by circadian clock and oscillates with 24-h rhythmicity due to rhythmic changes in the expression of sirtuins, CD38, and PARP1. 89 Transmission of circadian signals within the NVU requires cyclically expressed gap junctions, 76 thereby suggesting that synchronization of cells activity is a basis for circadian rhythmicity. Connexins supporting synchronization of CPECs (as described above) might operate through the NAD+ availability since Cx43 is functionally coupled to some NAD+-consuming enzymes (CD38), or mediate NAD+ transport to neighboring cells and its efflux to the extracellular space. 90
In sum, BMECs and CPECs are not only the part of two barrier systems in the brain, but serve as a key regulators of cyclic/rhythmic changes in brain fluids, metabolism and plasticity. Their functional activity affected with genetic and non-genetic approaches is in the focus of numerous studies aimed to develop new treatment solutions, 91 but still there is a shortage in available technological approaches that could allow precise reconstructing their rhythm-controlling action in the brain in vitro models.
Integrity of brain barriers and control of adult neurogenesis
Maturation and acquisition of functional competence of brain cells greatly depend on efficient intercellular communication and availability of soluble regulatory molecules (transmitters, cytokines, metabolites) produced either locally or distantly. Neurogenesis within brain neurogenic niches (NNs) is a clear example of how the cellular and humoral microenvironment determines the fate of cells at different stages of their development. Recent data suggest that not only local blood supply, but also CSF movement from lateral and third ventricles to neurogenic niches might contribute to adjusting the rate of neurogenesis to actual demand of developing and active brain. 102 Since embryonic and adult neurogenesis has been extensively described in details elsewhere,103 –106 we are going to focus here on a role of BBB and BCSFB in the regulation of adult neurogenesis stimulated in experience-driven brain plasticity and repair of lesioned brain tissue.
Neural stem cells (NSCs) are present in the nervous system in the embryonic and the postnatal period. They are the main source of new neurons that could be further integrated into pre-existing or newly-established neural circuits. 107 In most mammals, within 1–2 weeks after birth, radial glia cells produce adult NSCs in the subventricular zone (SVZ), and ependymal cells. 108 Later throughout the life, two major neurogenic niches—SVZ and hippocampal subgranular zone (SGZ)—serve as sites of experience- or damage-driven neurogenesis. In addition, some non-conventional neurogenic niches in hypothalamus, cerebellum, or non-recently generated immature cortical neurons contribute to plasticity-associated changes in neurogenesis. Thus, brain plasticity critically depends on the ability of NSCs to keep their own pool, or to undergo effective recruitment upon action of external stimuli (e.g. in learning, recognition, memory consolidation, or in brain damage).
Microvascular support of neurogenesis in conventional neurogenic niches in the adult brain
It is clear now that neurogenesis in SVZ and SGZ depends on the functional activity of NVU as well as on the integrity of the BBB and the BCSFB. Vascular scaffold plays an important role in the maintenance of SGZ and SVZ homeostasis by controlling the access of the niche environment to various soluble regulatory molecules, nutrients, oxygen, and cells. 35 In early development, SVZ is formed due to terminal differentiation of telencephalic radial glial cells into either adult NSCs or EpCs. 109 B-cells of astroglial lineage locate in the SVZ and serve as NSCs and neuronal progenitor cells (NPCs) that are able to differentiate up to different types of neurons, and oligodendrocytes. 110 NSCs/NPCs in the SVZ produce stable contacts with local microvessels as well as receive the regulatory stimuli from the CSF compartment. 111 Newly-established neuroblasts further move to various brain regions using microvessels as a vascular “road” for proper migration, such as to olfactory bulbs, cortex, striatum, and this mechanism is accelerated or suppressed in pathological conditions. 112
In the hippocampal SGZ, neural stem cells may undergo cell division, thereby some daughter cells (DCX+PSA-NCAM+ intermediate progenitors) differentiate into migrating neuroblasts and further to mature granule neurons capable to integration into pre-existing neural networks within the hippocampal granule cell layer. 113 In the SGZ NSCs have prominent radial processes serving for either establishment of direct contacts with local microvessels and for sending multiple branches into the inner molecular layer. 114 Thus, SGZ NSCs may detect local changes in the microcirculation as well as actual neuronal activity within the granule cell layer (where neurons are tightly packed together and send their unmyelinated axons known as mossy fibers to the CA3 region), and inner molecular layer of the hippocampus dentate gyrus (which is enriched with numerous dendrites of granule cells, pyramidal basket cells and polymorphic layer cells, as well as axons coming from the entorhinal cortex). 115
Fate of stem and progenitor cells within the niches depends on the availability of numerous regulatory molecules coming from the blood. Well-established astroglial coverage of hippocampal microvessels predicts lower availability of blood-derived regulatory molecules within the SGZ, whereas loss of tight astroglial contacts in SVZ BMECs results in higher dependence of this neurogenic niche on cytokines, metabolites and growth factors coming from the peripheral blood. In addition, cells fate within the SVZ niche is affected by soluble factors of CSF origin. 31
In a neurogenic niche, enhanced permeability of the BBB might be rather beneficial for the delivery of pro-neurogenic factors. Moreover, even outside the niches, it might be recognized as a compensatory mechanism aimed to support the establishment of novel pro-neurogenic loci and restoration of the brain tissue. That is why it is not surprising that establishment of new neurogenic zones in the injured brain is seen in a close vicinity to periventricular area and microvessels with higher permeability than it is kept in the intact brain parenchyma. 35 Particularly, this phenomenon has been described in rats subjected to the ischemic brain injury: novel neurogenic niches enriched with highly proliferating cells and microvascular scaffold with increased BBB permeability have appeared on day 4–14 after artery occlusion along the walls of third and fourth ventricles. It is important to note that ischemia-enhanced cell proliferation and neurogenesis parallel BBB leakage. 116
Promotion of neurogenesis by the enhanced permeability of the BBB is mainly attributed to the SVZ, but not to the SGZ neurogenic niche. 116 Within the SVZ, pre-existing higher permeability of the BBB is further elevated in the conditions of ischemia, thereby providing the access for the systemic VEGF. The latter triggers DLL4-Notch-dependent interactions of BMECs and NSCs and increases SVZ neurogenesis for better tissue recovery. 117 Direct contacts of NSCs with endothelial cells, ependymal cells, and neurons are established due to different types of astroglial processes. The changes in the morphology of astrocytic processes affect the integrity of barriers, neurotransmitter clearance, K+ dynamics, and the supply of energy substrates within the NVU. 118 Presence of two types of astroglial processes, mitochondria-enriched branches and mitochondria-deprived leaflets, suggests that dynamic changes in astroglial metabolism (oxidative phosphorylation vs glycolysis) might correspond to the extension and retraction of processes and efficacy of astroglial coupling with other cells. 118 Particularly in neurons, sprouting dendritic filopodia have a short lifespan, but when they are in a contact with astroglial processes, they have got an increased stability and ability to develop. 119
In the lateral ventricles, CPECs produce cytokines and growth factors to support the pool of primitive neuroepithelial precursors (NSCs that are the radial glia-derived cells) within the SVZ. 120 Due to continuous proliferation, up to 80% of NSCs differentiate to neuroblasts, whereas 20% provide the maintenance of the local stem cells pool. 121 Being recruited, SVZ NSCs produce migrating neuroblasts that go along the rostral migratory stream (in rodents) up to olfactory bulbs to integrate into the pre-existing local neuronal circuits. 122 Thus, ependymocytes are the important part of the SVZ neurogenic niche. 123 Ependymal cells (type E) separating the subventricular zone from the lateral ventricle form rosettes, inside which cilia of neuronal stem cells of type B1 pass. The opposite process of type B1 cells is in contact with the vessel. When these cells divide, transitory amplifying cells (type C) appear to give rise to neuroblasts (type A). 124
CPECs secrete numerous molecules that act at proliferating and migrating cells, for instance, insulin-like growth factors, bone morphogenetic proteins, Notch ligands. OTX2, which is secreted transcription factor, is produced and released by adult CPECs, and regulates migration of neuroblasts being absorbed by guiding astrocytes. 125 Recent data suggest that amyloid precursor protein (APP) is produced by CPECs in a form of sAPPα and positively affects proliferation of NSCs in SVZ and SGZ. 126 Thus, secretory activity of CPECs results in the appearance of numerous regulatory molecules in the CSF that are sensed by receptors expressed in SVZ cells. NSCs located here are in tight interactions with ventricular ependymal cells and local blood microvessels to provide the microenvironment permissive for the NSCs/NPCs maintenance and recruitment.127,128 NSCs send a short apical process through the ependymal cell layer to directly access the CSF as a source of regulatory molecules. 129 At the same time, NSCs contacting with local vascular network remain their high proliferative activity up to late stages of ontogenesis (aging). 130 Radial glia in the SVZ senses local microcirculatory changes and gradients of various homing factors (e.g. growth factors and cytokines attracting newly-formed neuroblasts to the loci of brain tissue lesions).131,132 In addition, NSCs are sensitive to the functional activity of neurons residing within the SVZ. It was shown that optogenetic activation of choline acetyltransferase-positive neurons in rodent SVZ results in enhanced proliferation of NSCs and neuroblasts production. 133 The same could be achieved with the activation of 5HT serotonin receptors expressing in SVZ NSCs. 134 Moreover, SVZ is directly innervated from dopaminergic projections from the substantia nigra and the ventral tegmental area, even the data on the local dopamine action are still controversial. 135 Thus, NSCs within the neurogenic niche serve as a “hub” to coordinate the activity of mature neurons, local microcirculation and permeability of barriers to provide the pro-neurogenic microenvironment.
Since CP possesses a role of circadian rhythm regulator, it is not surprising that development of NSCs/NPCs in both neurogenic niches depends on circadian oscillations: circadian clock genes are not required for neurosphere formation in vitro, but their absence suppresses neurosphere growth, suppresses differentiation of cells along the neuronal lineage, and promotes apoptosis. 136 Whether or not it might be attributed to the regulatory role of CPECs in the in vivo conditions, remains to be assessed. Taking into consideration diurnal variations in the actual concentrations of neurotransmitters (noradrenaline, histamine, orexin, glutamate, gamma-aminobutyric acid (GABA), serotonin, acetylcholine, and dopamine), 137 one may suggest that NSCs/NPCs might be sensitive to the local concentrations of neurotransmitters produced by neuronal activity governed by CPECs and SCN.
In the SGZ, recruitment and proliferation of NSCs are mainly under the control of local neurotransmitters (like glutamate and GABA). 138 For instance, NSCs reside closely to GAD67+ terminals of parvalbumin-expressing (PV+) interneurons and respond tonically to GABA released from these neurons: GABA signaling reduces their proliferation and induces quiescence. 139 Excitation of proliferating NPCs is produced by the activation of NMDA receptors and Cav1.2/1.3 channels which leads to prominent changes in gene expression and cell differentiation, 140 whereas loss of Kv1.1 channel activity causes proliferation of neural progenitor cells in the SGZ. 141
In recent decades, the role of EpCs as NSCs has been widely discussed. The expression profile of EpCs resembles those of NSCs (Sox2, Nestin, CD133), but neurogenic capacity of ependymal cells might be suppressed at the transcriptional or post-transcriptional levels. 142 Thus, several studies have shown that this cell type does not proliferate under normal conditions, and thus does not fully meet the characteristics of stem cells. 143 However, ependymal stem cells may control various aspects of adult neurogenesis and even are able to generate neuronal and astroglial cells being activated in the ischemic brain. 143 Under certain conditions, EpCs can behave like NSCs: in nerve tissue injury, in stroke, in carcinogenic transformation, in blockade of Notch1 signaling or being exposed to various growth factors. 144 Besides, ependymal cells express Noggin which positively controls neurogenic differentiation and suppresses gliogenesis. 145 Rostral migration of neuroblasts depends in part on the Slit2 gradient which is maintained by ciliary beating of ependymal cells. 146 It has also been shown that ependymal cells express the ankyrin protein, which is necessary for maintaining the SVZ structure and producing new neurons. 147 However, partial destruction of the ependyma by injection of neuraminidase into the subventricular zone leads to an increase in the production of neuroblasts, which proves the role of ependymocytes as negative regulators of NSC proliferation. 148
Arachnoid barrier cells express various efflux transporters (like Pgp), thereby contributing to the regulation of concentration of their ligands in the brain parenchyma and CSF. 149 Moreover, the complex of choroid plexus, CSF, ependyma, and brain parenchyma controls distribution of neurotransmitters, peptides, hormones, and xenobiotics to different brain regions. 150 As an example, SVZ enriched with neural stem cells (NSCs) and neuronal progenitor cells (NPCs) is fenced off the CSF via the layer of ependymal cells (Figure 1), which are “sister” cells to the NSCs and are able to provide trophic and metabolic support, to participate in the synthesis and secretion of CSF, to mediate steroidogenesis, and to regulate the selective passage of water. 41 Specialized EpCs known as tanycytes locate in the third and fourth ventricles and communicate with the hypothalamic median eminence serving as a diet-regulated neurogenic niche important for the feeding behavior and metabolic control. 151 EpCs coordinate CFS dynamics in (patho)physiological conditions by generating a directional CSF flow controlled by ciliary beating.109,152 –154 Neuroblast migration parallels CSF flow, 146 thereby migration of SVZ-born neuroblasts depends on the fluid dynamics, EpCs and CPECs physiology and metabolism.
Another intriguing mechanism of microvascular control within the SVZ relates to the existence of fractones that are the extracellular matrix-originated structures providing compartmentalization of the niche being, probably, the anchoring points for NSCs and regulatory molecules (growth factors, cytokines) as well as a platform for juxtacrine signaling. Ependymal cells and GFAP-expressing cells are responsible for their establishment. 155 These structures are tightly coupled with local capillaries,156,157 even the protein composition of vascular basement membrane and fractones is different. 158
In sum, there is a coordinated activity of BMECs, EpCs, and CPECs which is required for the local blood supply in neurogenic niches, or CSF production and movement toward sites of extensive cells proliferation, differentiation and maturation in the developing and adult brain. It is reasonable that changes in the permeability of BBB and BCSFB would significantly affect the fate of cells at different stages of neurogenesis.
Barrier permeability and neurogenesis in non-conventional neurogenic niches in the adult brain
In the brain parenchyma, BBB breakdown caused by brain tissue alterations promotes development of neuroinflammation, brain edema, and secondary tissue injury.159 –161 This mechanism might be supportive for neurogenesis in non-conventional neurogenic niches (e.g. in the amygdala, hypothalamus, or cerebellum) 162 or even in the brain cortex. Indeed, another mechanism of plastic changes is based on the direct conversion of reactive astrocytes into mature neurons, or on the differentiation of so-called non-recently generated (dormant) premature DCX+PSA-NCAM+ neurons into mature ones in the brain cortex.2,163 –167 Even the data on such mechanisms are rather controversial, several attempts have been paid to reprogram local astrocytes in the brain tissue to treat neurodegeneration, for instance, based on NeuroD1 overexpression in reactive astrocytes resulting in increased neuronal density and regeneration within the lesioned loci in the ischemic monkey brain. 168 It was demonstrated that resting astrocytes can’t be efficiently transformed into neurons, whereas reactive astrocytes that are activated due to neuroinflammation are the “ideal” target for the desired transformation. 169 This finding corresponds to previous data on regenerative potential of activated astrocytes. 170 Thus, it is tempting to speculate that transitory BBB breakdown causing activation of astrocytes within the NVU would be beneficial in the generation of astroglial population sensitive to triggers of neuronal transformation. 171 One may assume that optogenetic or chemogenetic activation of astrocytes either in vitro or in vivo might be useful not only for the controlled release of gliotransmitters and promotion of neuron-astroglial interactions,172,173 but also for the production of reactive glial cells susceptible for further conversion into mature neurons.
Maturation of DCX+PSA-NCAM+ dormant neurons (non-recently generated immature neurons that can stay in the undifferentiated status for decades in the primate brain) has been detected in the pyriform cortex.174,175 Presumably, their differentiation up to the mature neurons might be helpful for higher order processing of sensory inputs in the brain. 176 Precise mechanisms of induction of these events are not clear yet, however, it was found that enzymatic depletion of PSA promotes the final stages of development of immature neuronal population, 175 the number of cells expressing PSA-NCAM induced by a dopamine receptor agonist decreases due to the differentiation of these cells associated with the loss of PSA-NCAM expression. 177 Then, newly-formed neurons become to be functionally integrated into the pre-existing neuronal ensembles in the cortex. 174 Almost similar mechanism seems to exist in the amygdala where dormant immature DCX+PSA-NCAM+ neurons persist for several years, but then may develop into mature neurons during adolescence. 178
On the other hand, recent data suggest that some mature SGZ neurons undergo so-called dematuration in the conditions of hyperexcitation or neuroinflammation, and start to express the markers of immature neurons or NPCs coupled to the cell cycle re-entry. 179 Since mature neurons are post-mitotic cells, they can’t complete the cell cycle, but may respond to any cell stress via cell cycle re-entry mechanism as a way to support apoptosis or mitotic catastrophe, DNA repair, and synaptic plasticity.180,181 It was proposed, that cell cycle-related molecular mechanisms serve as regulators of synaptic activity in neurons whose inability to proliferate is required for making the synaptic connections. 180 In some cases, increased dematuration of neurons and enhanced cortical neurogenesis are the associated processes in the primate brain, 182 thereby suggesting involvement of the pool of DCX+PSA-NCAM+Ki67 as dormant immature neurons in these events. 183
Thus, changes in BBB permeability may affect neurogenesis in various brain regions that serve as a platform for the development of new neuronal and glial cells (Figure 2). Immature neurons may migrate from meninges consisting of external dura mater, arachnoid and internal pia mater to the brain parenchyma and further differentiate up to mature neurons or oligodendrocytes. 184 Meninges project to the hippocampus and choroid plexus (pia matter wraps the CP), and such projections might be important for functional coupling of conventional and non-conventional neurogenic niches in the brain, presumably, due to activity of the blood-meningeal barrier (BMB). Whitin the BMB, the perivascular space is separated from the brain parenchyma by the pia mater basal membrane and astrocytes end-feet forming glia limitans. In physiological conditions, arachnoid and pia contain neural precursors, stromal cells (pericytes, telocytes, smooth muscle cells, fibroblasts, and pial cells), immune cells (macrophages, dendritic cells, and mastocytes). 184 Pial cells do not express tight junctions but are joined by desmosomes and gap junctions, however, activated astrocytes may express tight junction machinery. 185 NSCs have been identified in the perivascular area endowed with a thin sheath of meningeal cells surrounding cerebral arterioles. 184 In a contrast to pyramidal neurons, granule cells in the dentate gyrus of hippocampus are generated mainly postnatally. 186 Immature cells have been shown to migrate in a CXCR4-dependent manner from the newly-discovered neurogenic niche (subhippocampal zone, SHZ) existing in the medial walls of the lateral ventricles between the dorsal surface of the hippocampus and the CP through the fimbria-dentate junction along the meninges in the direction of the hippocampal dentate gyrus. 187 In the adult rodent brain, proliferation of progenitor cells in the dentate gyrus has been shown to be suppressed after fimbria–fornix lesions but not after entorhinal deafferentation (in physiological conditions, major and minor projections carrying sensory information come from the entorhinal cortex to the hippocampus), therefore, proliferation and/or differentiation in the SGZ seem to be controlled by factors reaching the hippocampus via fimbria-fornix afferents. 186 Thus, CP is not only the part of the ventricular-SVZ neurogenic niche, it also affects the development of new cells in the adult hippocampal SGZ either via secretion of numerous regulatory molecules (growth factors, cytokines and metabolites), or being involved into the regulation of immature cells migration from the SHZ along the meninges to the dentate gyrus.
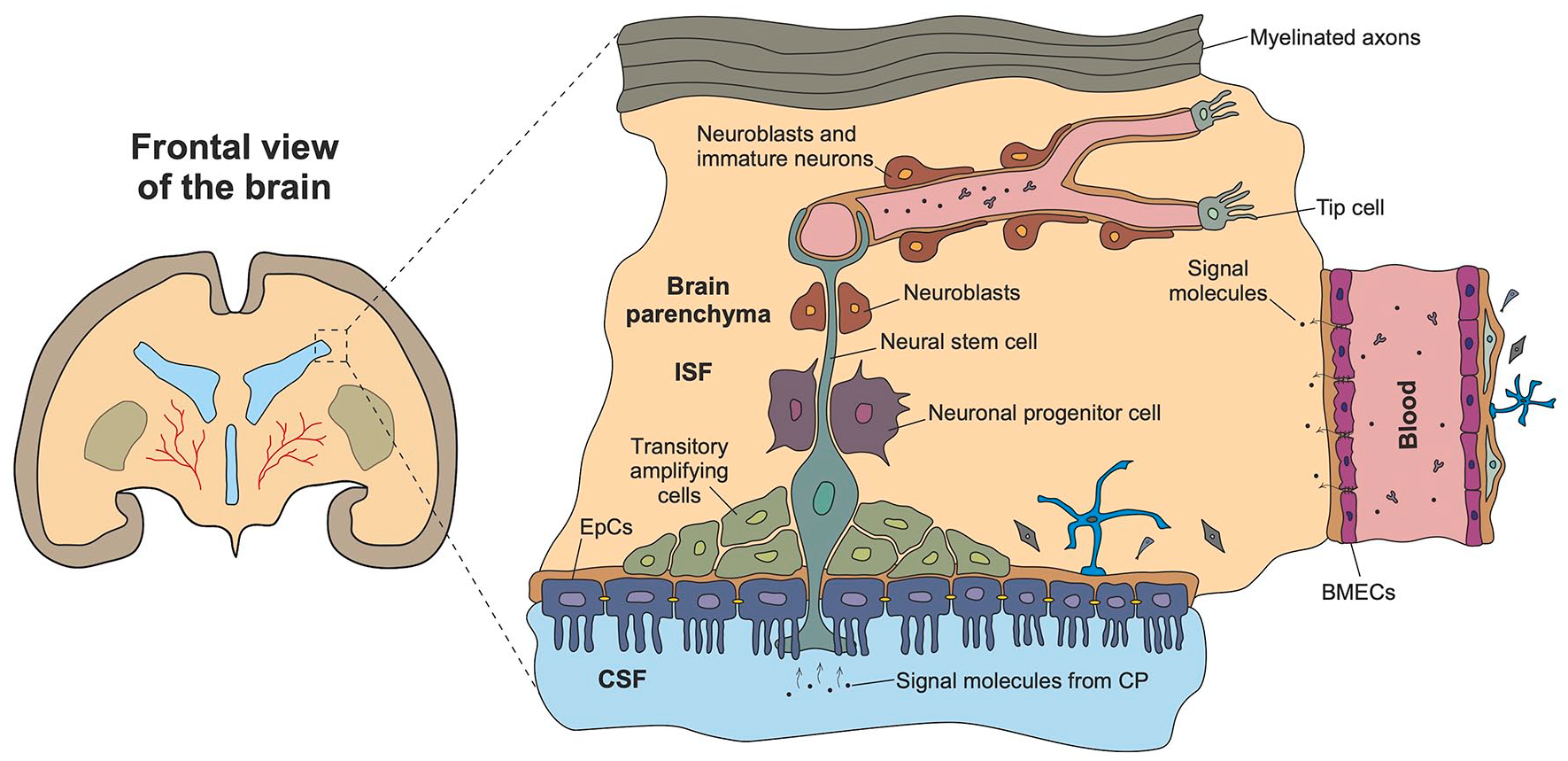
Brain plasticity in the SVZ. Plasticity of neurogenic niche consisting of ventricular barrier ependymal cells (EpCs), neural stem cells (NCSs), transitory amplifying cells, neuronal progenitor cells (NPCs), neuroblasts, immature neurons, astrocytes, extracellular matrix proteins and other components, is under the control of blood-brain barrier (BBB) and blood-CSF barrier (BCSFB) integrity. Appropriate stimuli coming either from the blood in a case of BBB disruption or from the CSF due to choroid plexus (CP) activity induce two key long-term mechanisms of brain plasticity: angiogenesis and neurogenesis.
Development of a changeable brain: 4D neurogenic niche in vitro models
Evolution of the brain barriers in vitro models: Searching for the optimal microarchitecture
Development of the brain in vitro models started from the in vitro reconstruction of BBB using various sources of cells and different conditions for their co-culture. Particularly, the simplest model of the BBB in vitro is Transwell, which is a static two-dimensional culture system with a microporous membrane. Transwell models very conventionally translate key barrier parameters in vivo, such as TEER and barrier permeability, whose values appear respectively below and above the generally accepted ranges of the norm. To solve this problem, numerous comparative studies of dynamic and static systems have been conducted, showing the primary role of flow-induced shear stress in increasing TEER and decreasing permeability—new dynamic models catched on, allowing to obtain more close to physiological values of key BBB in vitro parameters.188 –191 These systems used the technology of the so-called «hollow fibers», inside which a flow was created, determining the formation of tight contacts between endothelial cells (EC), as well as the inhibition of their cell cycle, resulting in a characteristic for brain capillaries monolayer of cells without fenestrations between them.188,190
The main disadvantages of the hollow fiber model are the considerable wall thickness (an order of magnitude greater than the membrane thickness in Transwell systems), which limits intercellular interactions, as well as the long establishment of stable TEER values, which increases the experiment time. 192
Further attempts to improve in vitro BBB models have been focused on the use of well-established microfluidic technologies. In 2012 Ross Booth and Hanseup Kim representatively pointed out the advantages of microfluidic BBB models over standard ones and proposed an original PDMS chip design with two perpendicular channels, a polycarbonate membrane between them and two glass substrates with embedded electrodes for TEER registration. 192 There were no obvious disadvantages of hollow fibers, therefore, it was the first step in extensive research and dissemination of physiologically more relevant microfluidic models of the BBB.
In the same year, a simplified chip model using immortalized hCMEC/D3 EC line was presented. 193 Cells were cultured there for 7 days and expressed tight junction proteins (ZO-1) on day 4. Barrier function was studied by mechanical (shear stress) and biochemical (tumor necrosis factor—TNF) stimuli. Mechanical stimulation resulted in a threefold increase in TEER, and addition of TNF resulted in a 10-fold decrease.
All known types of co-cultures involved in BBB were used in the work. 194 Neurons differentiated from iPSC in multi-well plates, as well as isolated brain endothelial cells, pericytes and astrocytes were co-cultured in a microfluidic PDMS bioreactor based on two chambers separated by a porous membrane. Perfusion of both compartments was ensured. Pericyte-conditioned medium increased EC polarization (alignment of actin filaments in the flow direction), which is one of the main markers of barrier integrity (along with the expression of tight junction proteins). Fluorescent dextrans with different molecular weight were used to test the barrier permeability, in particular, the diffusion of FITC-dextran was studied. It was experimentally shown that one of the key mechanisms of transfer of compounds across the barrier besides diffusion is active transport.
Around the same time, a different approach was developing, involving a more accurate reproduction of the 3D geometry of the capillary network in vitro based on hydrogel 3D printing. 195 The system, based on hollow fiber technology, is a collagen matrix with four channels obtained by slow removal of micro-needles from the hydrogel and subsequent seeding bEnd.3 (completely covered the surface in 3–4 days). Thus, each channel is a lining of endothelial cells on some analog of the basal membrane (fibronectin coating) in the collagen hydrogel. The main goal of the model is to reproduce a more accurate capillary architecture without the use of PDMS and a synthetic porous membrane. Non-physiological mechanical properties of PDMS (high modulus of elasticity), as well as its adsorption of hydrophobic compounds strongly limit the relevance of organ-on-chip platforms and microfluidic bioreactors of BBB based on it. An alternative material is collagen 1, which has a close to extracellular matrix (ECM) in vivo elastic modulus (about 100 Pa). A system similar to the one described above, however, using a PDMS channel covered with an adhesive layer of poly-l-lysine and a layer of collagen, is presented in the work. 196 The main drawback of these models is the incomplete reconstitution of the neurovascular unit as a multicellular ensemble.
Sufficient material for analysis has already been accumulated, about 10 different microfluidic models of the BBB have been developed, a comprehensive comparative analysis of which is presented in an excellent review. 98 Among other things, this review justifiably emphasizes the importance of validating in vitro BBB models by key parameters (TEER, permeability coefficient, shear stress, protein expression, barrier co-culture composition) and provides quantitative estimates to standardize measurements.
A year later, 197 a new membrane-free microfluidic bioreactor design for the BBB with 4 horizontally divided channels—for nutrient medium, for the gel with neurons, for the gel with astrocytes, and for endothelial cells—was demonstrated. Cell-to-cell interactions within the NVU have been monitored using calcium intracellular imaging. Direct contacts between astrocytes and brain microvessels without the use of membranes were also reproduced in the study. 198
However, membrane chips were not completely replaced by membrane-free ones, and new vertical PDMS chip with a polycarbonate membrane between the upper and lower layers was developed an year later. 199 A feature of the design here was the use of parallelism for potential high-throughput screening—four channels were made in each PDMS layer, forming a total of 16 individual BBB cells on the chip.
Recently, one of the most successful commercially oriented in vitro BBB model projects was considered to be OrganoPlate by MIMETAS. The original study, invested by Biogen pharmaceutical company, is a platform of 40 or 96 compactly arranged microarrays for antibody transcytosis testing. 200 A special feature of the platform is that bi-directional perfusion is performed by rocking it without the use of microfluidic pumps, which makes an extensive implementation of the technology more attractive. The obvious advantage of the system is the absence of membranes and PDMS, as well as the flexibility and high degree of parallelism. At the same time, the shear stresses generated are rather low, which is a disadvantage.
Active development of the iPSC technology allowed the differentiation of brain endothelial cells according to standard protocols, the necessary components were selected and validation experiments were performed.201 –203 Progress in this field led to the introduction of iPSC-derived endothelial cells into microfluidic devices. One of such devices was the development of D. Ingber and colleagues, structurally identical to the lung-on-chip presented by them earlier. 204 The stated idea was to make a simple and most functional platform for drug delivery and screening systems. Among the achievements of the model are the long-term maintenance of the barrier function (for 2 weeks) and its continuous measurement using the built-in electrodes for TEER.
In one of subsequent reports, a membrane construction for more detailed study of nanoparticle transport was presented. 205 The 3D astroglial network implemented there reproduces the physiological expression of aquaporin-4, thus it is a model for studying water transport (and ion homeostasis) within the BBB. Scientists were able to simulate eHNP-A1 transcytosis through the barrier, which defines eHNP-A1 as a potential drug delivery system for the CNS.
Receptor-mediated transcytosis as a way to deliver the drugs to the target regions of the brain parenchyma is currently a key tool in nanomedicine for BBB -associated diseases and has significant potential for implementation in the clinic.206–209 Analysis of available literature data shows that there is a problem in increasing the targeting concentration of a drug,210,211 thus, in vitro modeling of drug transport through the BBB and verification of such experiments is of particular relevance. However, so far only two 3D in vitro models of the BBB have been used to find correlation with in vivo data,205,212 which indicates that the field of nanomedical applications in the development of drug delivery systems for CNS is at the stage of databases formation and first experiments on microfluidic and spheroidal models. The key reference point here is the attempt to use receptor-mediated transcellular drug transport using nanocarriers, 213 while paracellular pathways can only be used effectively if there is a prior exposure to the BBB that increases its permeability, that is, fenestrations naturally absent between endotheliocytes of brain microvessels.
One of the latest achievement in the field is the development of pump-free, open-microfluidic BBB in vitro model with integrated fully transparent ITO electrodes presented by Andreas Hierlemann group. 214 Chip design and electrodes configuration enable simultaneous high-resolution imaging and TEER measurements. Scientists were aimed to study how BBB reacts to external stimuli and established ischemic conditions with oxygen-glucose deprivation, leading to morphological changes in endothelial cells associated with BBB disruption. In contrast to the membraneless OrganoPlate, where cell perfusion is also performed by bidirectional flow by rocking the platform, this chip represents barrier model with vertical cell-to-cell interactions via a membrane between the lower vascular compartment and the upper parenchyma compartment.
The large number of devices under development has contributed to the systematization of advances in the field of bioreactors for BBB-on-chip MPS, the search for new approaches, and the development of new challenges. Given the accumulated experience, in recent years scientists have sought not only to reproduce 3D barrier architecture and create physiological conditions for BBB component cells, but also to integrate various biosensors into such platforms to automate quantitative experiments to improve the clinical applicability of in vitro models and to apply these platforms to model CNS diseases and consider the role of the BBB in their pathogenesis.214 –218
In contrast to numerous BBB-on-chip models, rather few BCSFB models have been created. In BCSFB there are two main types of barrier cells, namely tightly coupled CPECs, determining barrier properties, and fenestrated ECs. Modeling the BCSFB in vitro is more complex than the BBB because it is quite difficult to obtain primary epithelial cells due to a lack of suitable cell lines and problems in establishing appropriate conditions for their functioning in vitro (incl. filtration of blood plasma and production of CSF). However, over the last decade, models of the BCSFB have evolved, similar to those of the BBB, from static cell epithelium monolayers and cell culture filter inserts (Transwell-like barriers) to dynamic ones on microfluidic platforms.219 –224
One representative example of Transwell BCSFB model is illustrated in the work. 222 An inverted model of BCSFB designed to study the cellular and molecular mechanisms mediating the migration of immune cells through BCSFB. Primary CPECs growing on inverted Transwell filters were defined as a reliable in vitro model for experiments mimicking the T cell migration from the blood vessel side to the apical side facing the CSF. In addition, TEER and barrier permeability were evaluated for different membranes: TEER decreases by 50% when membrane pores are increased by a factor of 10, while barrier permeability changes only slightly. Given the large variability of materials and pore sizes of membranes used in brain-on-chip models, it is worth considering this parameter separately when analyzing the obtained TEER values.
The microfluidic approach in the establishment of BCSFB in vitro models was introduced in 2023, when a previously developed standard chip geometry was used to create the dynamic BCSFB in vitro model. 225 The main advantages of the model are the co-culture of human CPECs and BMECs and the application of shear stress. Barrier properties, including modeling of neuroinflammation (TNF-treated cells), were assessed by immunofluorescence imaging, permeability measurements and transcriptional profiling of epithelial cells. Thus, it was confirmed that TNF perfusion impairs the barrier function of BCSFB. Development of a more complex co-culture system incorporating immune cells or brain parenchyma cells, as well as measurement of TEER, might improve the presented model for further investigations.
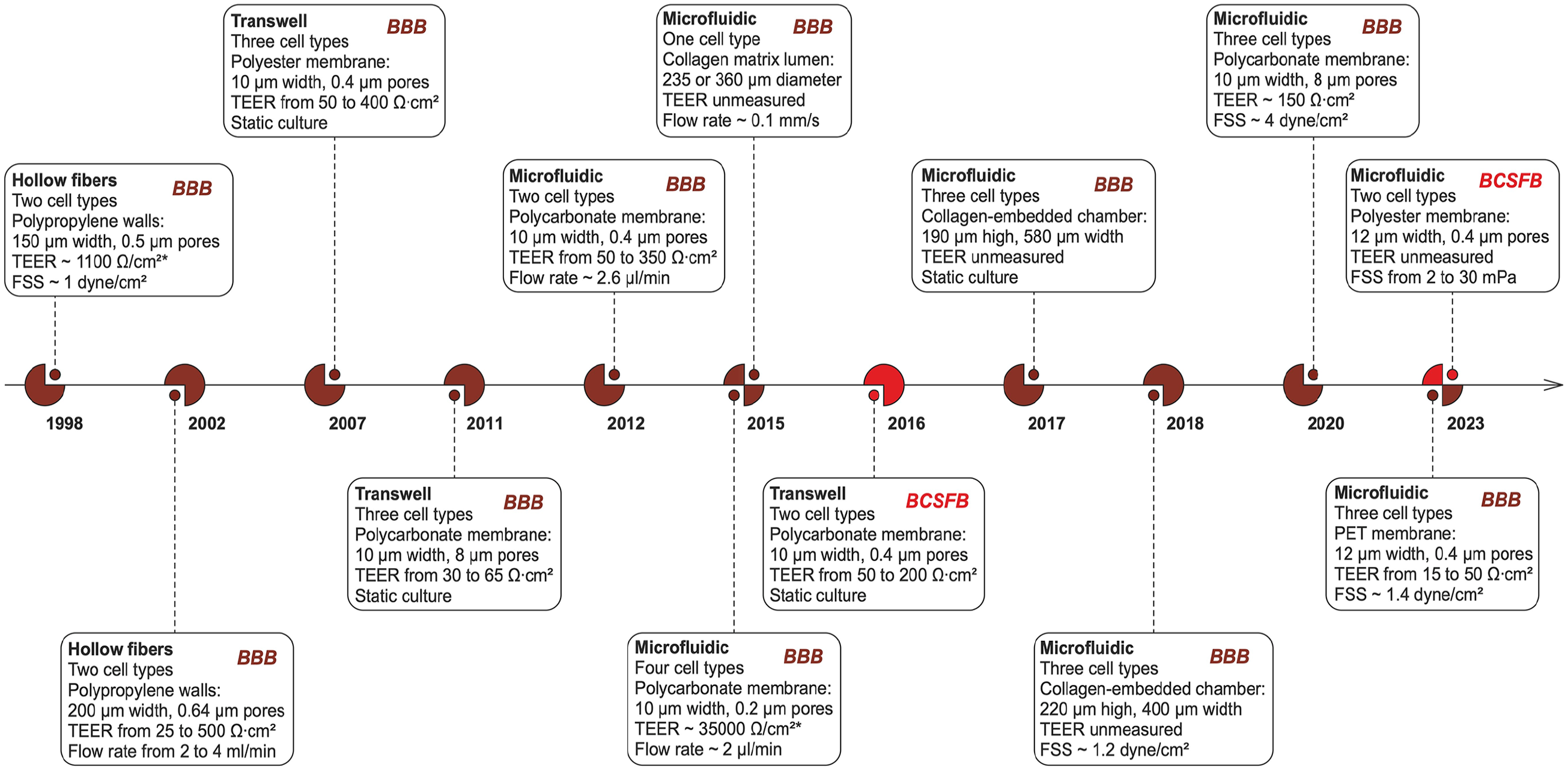
Thereby, characteristic stages of evolution in the field of in vitro modeling of the BBB and BCSFB have been presented, with an emphasis on the biological and technical features of the systems being developed in this field. The reader is referred to the reviews for additional information.17,31,33,213,226 –231 The most representative works were analyzed and systematically reproduced in Tables 2 and 3. Timeline of technology evolution with key excerpts is visualized in Figure 3.
In vitro models of BBB representing major milestones in the technology evolution.

FSS: flow shear stress; DIV-BBB: dynamic in vitro BBB; NDIV-BBB: new dynamic in vitro BBB; NVU: NeuroVasular Unit; NVC: Neurovascular chip; MW: molecular weight; TEER: transendothelial electrical resistance.
TEER values are not normalized properly in the original model description.
Short summary of available in vitro BCSFB models.

CPECs: choroid plexus epithelial cells; BMECs: brain microvascular endothelial cells; ECM: extracellular matrix; TEER: transepithelial electrical resistance; FSS: flow shear stress.
Development of 4D neurogenic niche in vitro models
As we have mentioned above, neurogenesis in the embryonic and adult brain represents the good example of the brain plasticity phenomenon. During embryogenesis, massive development and migration of cells result in the establishment of very heterogenous brain tissue. Currently, iPSCs-derived cerebral organoids grown in the in vitro conditions are considered as best models for studying the early steps in brain developmental plasticity. Even they have numerous limitations (e.g. loss of vascularization and absence of microglia), but they are able to recapitulate some crucial mechanisms of fetal brain development. 234 As to the adult neurogenesis, establishment of conventional neurogenic niches (NNs) is based on tight structural and functional coupling of neural, glial and endothelial cells with different stages of maturity. All key events within the niche—maintaining the pool of NSCs, their recruitment and proliferation, maturation of NPCs, migration and differentiation of neurobalsts—are driven by the changes in the tissue stiffness and local microenvironment which is provided by the secretory activity of resident cells or by the permeability of the BBB (either in SGZ or SVZ), BCSFC and VB (in SVZ).28,35,235,236
The NNs in vitro model should include the following compartments: (i) a chamber for NSCs/NPCs, neuroblasts and resident mature cells with regulatory activity, e.g. astrocytes; (ii) a channel covered with BMECs for reproducing the bulk blood flow and BBB activity in niche microvessels; (iii) a channel covered with CPECs for reproducing the filtration of blood plasma and production of CSF for its further diffusion through the BCSFB and VB into the compartment with NSCs/NPCs and (im)mature neuronal cells; (iv) a chamber for mature neuronal cells with the well-established synaptic connections and glial cells as an analog of brain parenchyma. The geometry of the model should provide the possibility for the short-distance migration of newly-born cells along the chains of endothelial or astroglial cells, and their controlled maturation and interaction with mature neurons. In vivo, stimulus-driven proliferation of NSCs and their conversion into NPCs may take up to 3 days, followed by the differentiation within 1 week and further development of mature neurons within 4–6 weeks. 237 There is a difference in the time-course of adult neurogenesis in two niches: development of newborn neurons in the SGZ takes more than1.5 month before their complete maturation and functional integration into the pre-existing neuronal circuits, whereas development of newborn neurons in the SVZ occurs within 1 month. 238
Therefore, the following key aspects of NNs in vitro modeling should be taken into the consideration: (i) selection of appropriate extracellular matrix-mimicking 3D scaffolds (e.g. made of degradable biopolymers) with the appropriate stiffness, porousity, and biocompatibility, including those used for the reproction of BBB, BCSFB, and VB within the models; (ii) tightly-controlled addition or removal of extracellular molecules (e.g. neurotransmitters, growth factors, metabolites) needed for the regulation of cells fate at different stages of their development; (iii) establishment of concentration gradients of molecules in transwell systems or within the microfluidic platforms for the controlled proliferation, differentiation and migration of newly-born cells; (iv) maintaining the long-lived multicellular ensembles recapitulating the whole cycle of neurogenesis in vitro with the permanent monitoring of cells expression pattern, their proliferation and differentiation capacity.239–243
Thus, modeling the adult NN in vitro is a non-trivial task, and only few attempts have come close to creating a physiologically relevant NNs in 3D or 4D conditions. They include neurospheres cultured in the conditions preventing cells attachment to the surface (e.g. droplet-based microfluidics), monolayers or hydrogel-embedded cultures of NSCs/NPCs grown with or without other cell types, for example, astroglial cells, microglial cells, and BMECs.244–249
Figure 4 illustrates the technological leaps in BBB and BCSFB modeling. Graphical examples and comparative analysis of existing in vitro brain-on-a-chip models are provided.

Technological leaps in BBB, BCSFB and NN modeling. Transwell inserts, hollow fiber bioreactors and microfluidic chips as available tools to recreate brain tissue in vitro.
Establishment of an “ideal” brain in vitro model: Shifting from 2D/3D to 4D conditions
Since developmental and adaptive plasticity is the most critical property of the brain, establishment of an “ideal” brain tissue in vitro models requires precise reproduction of events that reflect dynamic changes in the brain structure and function. Particularly, modulation of excitation of neuronal cells and activity of glial cells, neurogenesis, cell migration and physiological cell death, vasculogenesis/angiogenesis and recession of microvessels, synaptogenesis and competitive elimination of synapses, barriergenesis and dynamic changes in the permeability of barriers, dynamics of brain fluids (blood plasma, interstitial fluid, and CSF) between brain compartments. It is obvious that such changes are implemented with different time-courses (ms – h/days),250–253 therefore, physiologically relevant brain in vitro models should provide the possibility to monitor either fast or long-lasting events with good time resolution and accuracy.
A retrospective look at the development of the technology for the brain in vitro modeling highlights the following starting points for future research:
– As 3D architecture has been reproduced in various formats (spheroids, organoids, hydrogel cultures, microfluidic chips), attempts to realize the dynamics of brain tissue histogenesis by relying on simulation of key brain barriers may lead to a new class of models, 4D models in vitro, capable of reproducing neuro- and angiogenesis processes on chip. Thus, it might be possible to establish changing brain in vitro which could reproduce the most important mechanisms of brain development, maturation, repair, and aging. Particularly, special attention should be paid to the development of multi-sensor platforms for permanent registration of events induced by the action of external stimuli with the software for real-time analysis of huge dataset. As an example, one could propose the establishment of long-lived complex systems with the components of brain-on-chip barriers with the brain region-specific parenchyma sensitive to the action of various neurotransmitters, gliotransmitters, neuropeptides, and cytokines, and demonstrating detectable changes in the synaptic transmission, neurogenesis, and neoangiogenesis. Future research in this area will enable a qualitative leap in disease modeling, taking into account brain plasticity as its fundamental in vivo phenomenon, and consequently improve the validity of in vitro models for drug screening targeting neurodegenerative diseases in particular, and BBB/BCSFB-associated CNS diseases in general;
– The inclusion of molecular profiling methods (omics technologies) will allow, on the one hand, better identification of molecule targets at the preclinical trials stage by identifying differences between genomic, transcriptomic, proteomic and metabolomic profiles for samples of sick and healthy subjects and, on the other hand, evaluation of therapeutic effects of candidate molecules on omics information obtained from the target area of target cells in the body. Thus, if omics technologies are integrated into the cycle of high-throughput drug screening systems, the predictive value of in vitro models can be improved to accelerate clinical translation of candidate drugs;
– Full reconstruction of (patho)physiological brain fluids dynamics (either bulk flow or diffusion) is still unresolved problem in available brain in vitro models. Even there are some recent attempts to the CSF flow measurements in vitro 254 or to the creation of in vitro platforms that provide the microenvironment needed for the control of the brain hydrodynamics, 255 they mainly deal with the establishment and monitoring of one separate type of the flow (e.g. CSF or blood);
– The trade-off between technological complexity (physiological relevance) and high performance of microfluidic systems for drug screening is one of the stumbling blocks between the transition of laboratory research (academia) to commercial technology implementation (industry). However, instead of searching for a compromise, it is advisable to focus on the target function of the project and match all parameters of the developed system to its final application. In particular, for modeling neurodegenerative diseases in vitro and studying their pathogenesis, the structure and function of the BBB and BCSFB, and the fundamental properties of the brain, systems with increased complexity and low reproducibility will certainly be a priority. On the other hand, simpler systems with high repeatability and reproducibility of research results are more suitable for simultaneous screening of drugs and performing a large array of quantitative in vitro assays.
In December 2022, the FDA de facto equated preclinical testing of drugs in vivo (using animals) and in vitro testing (using standard Transwell systems, microfluidic organ-on-a-chip or other models). This means that the preclinical trials phase could be significantly shortened in both time and cost in the coming years by more detailed in vitro screening and the complete abandonment of animal testing before the human clinical trial phase. We can state that the primary goal of developing organ-on-a-chip technology and, in particular, developing microfluidic BBB/BCSFB systems on a chip to meet one of today’s major healthcare needs (management of CNS diseases) has fully justified itself and, moreover, receives a new impetus for development and wide implementation into standard practice by pharmaceutical companies.
Obviously, there are some preferences in using the cheaper in vitro models instead of animals. Nevertheless, a complete rejection of animal drug testing is unlikely in the coming years, and there are a number of good reasons for this. The main stopping factor here is the paucity of work (only two on BBB modeling) with validation of in vitro models by comparative in vivo experiments. The second reason relates to the current unavailability of real 4D (where fourth dimension is time) brain-on-chip models fully representing the changing/developing brain in vitro. The relevance of the problem of creating a relevant in vitro model of the NN to become a reliable platform for obtaining repeatable and reproducible results in the study of barrier function, disease modeling, drug screening is thus confirmed not only by the accumulated experience in the field, but also by the trend in recent years to reduce the use of animals in the preclinical trials and the appearance of the first in vivo-in vitro correlated data.
In sum, it is quite obvious that reproducing brain plasticity in the in vitro models is a non-trivial task both in terms of the variability of cells whose interactions must be taken into account, and composition/dynamics of tissue barriers and extracellular fluids that regulate brain’s activity. Numerous attempts have been made to achieve the reconstruction of the brain tissue in vitro which could properly reflect the real network of brain cells and their ability to undergo plastic changes upon receiving the external stimuli like it happens in learning, memory encoding and consolidation.218,256–259 However, we are still far from the complete solving the problem of constructing the 4D models where the tissue undergoes dynamic changes similar to developing or experience-activated brain. Let’s summarize which tools are available now, and what characteristics should have an “ideal” brain in vitro model:
1. There is a diversity of cells used for brain tissue modeling in vitro. Particularly, primary cell cultures, cell lines, stem cells-derived terminally differentiated cells, cerebral organoids spontaneously organized in vitro are currently in use.259 –262 Still there are some technical difficulties in the establishment of tissue models made of human cells, in getting the correct phenotype of iPSC-derived cells in vitro, in the co-culture of different cells (e.g. neurons, astrocytes, BMECs, or CPECs) in rather simplified and unified conditions, as well as in monitoring of cell-specific functional activity within the multicellular ensembles.
2. Tissue architecture could be reproduced in static (transwell) or dynamic (microfluidic), 2D (planar) or 3D (spheroids, organoids, or cells embedded into the gels) formats. Even simple static models are still useful for studying intercellular communications in the brain tissue.17,28,219,263 –265 Application of microfluidic systems opens new opportunities for the establishment of changeable microenvironment, reconstruction of brain fluids flow and cells behaviors that are controlled by fluids movement and their chemical composition (e.g. fluidic shear stress enabling BMECs to develop and to function with higher efficacy), achievement of better barrier structural integrity as well as continuous monitoring of brain cells metabolism and performing the high throughput high content imaging and analysis.257,258,266,267 Implementation of 3D models allows recapitulating the brain complex microarchitecture, dimensionality, reconstructing different types of cell-to-cell and cell-to-extracellular matrix (ECM) communications, ECM- or vascular-derived tissue compartments, and assessing the self-organization of brain multicellular ensembles.8,268
3. Compartmentalization is achieved by reconstructing the brain tissue within the specially designed microchambers, microchannels usually separated with porous membranes or existing in a membraneless format. This approach allows establishing the integral brain tissue structure and intercellular interactions that depend on cell location, dynamic changes in the environment, for example, to provide optimal conditions for cell functional activity and metabolism, connectivity of various brain regions.10,256,258,269 Also, such compartmentalization might be useful for getting the multiscale architecture of the brain tissue existing as a “network of networks” in vivo.256,270
4. Introduction of various sensors to monitor cell activity within the model, for example, multielectrode assay for the assessment of neuronal excitability and synaptic transmission, optical, impedance and electrochemical sensors for monitoring cell migration, proliferation, metabolism, chemical composition of the extracellular fluids, and multiple intermolecular interactions, and BBB/BCSFB permeability.11,271,272 Other approaches include optical imaging of cells growing in vitro, analysis of subcellular events that are key for cell functionality (e.g. mitochondrial activity, calcium and cytoskeletal dynamics), molecular profiling of cells and extracellular fluids by means of transcriptomic, proteomic, metabolomic approaches in (patho)physiological conditions.11,218,273 –275
5. Application of novel materials and scaffolds mimicking ECM results in the improvement of cells viability, adhesion, interactions, functional activity, and communication. As an example, substrates with the desired stiffness, permeability, degradability, compatibility, and dimensionality improve neural tissue engineering by providing appropriate electrical stimulation of cells,276,277 promote recruitment, proliferation and differentiation of NSCs,278,279 support controlled release of proneurogenic or proangiogenic factors, 271 protect the cells exposed to unfavorable in vitro conditions. 280
6. Further progress in automatization and data analysis is extremely important, because complex reproduction of brain plasticity phenomenon in vitro will ultimately lead to the appearance of a problem with massive data analysis for the assessment of brain plasticity and establishment of appropriate feedback mechanisms in a model.
7. In addition to all above mentioned characteristics, the models aimed to reproduce key events in neurogenesis and neurogenesis-coupled brain plasticity should consider the following additional tasks: 7.1. establishment of multicellular ensembles made of NSCs/NPCs and their progeny as well as fully-differentiated cells (astrocytes, BMECs, CPECs, EpCs), tight integration of functionally and phenotypically distinct compartments (neurogenic niche, NVU/brain parenchyma, and associated brain tissue barriers) on a chip mimicking the regulation of neurogenesis in (patho)physiological conditions31,135,281
–283; 7.2. reproduction of ECM composition, oxygen and nutrients supply for better cell viability and functionality, reproduction of metabolic plasticity and local humoral microenvironment supporting cell-to-cell communications, establishment of conditions supportive for neurogenesis and angiogenesis204,284
–286; 7.3. reconstitution of the chip microarchitecture, fluids exchange and establishment of chemical gradients that are supportive for NSCs/NPCs maintenance due to dynamic changes in the concentrations of local and “systemic” regulatory molecules (neurotransmitters, gliotransmitters, growth factors, cytokines, alarmins, metabolites) produced by the cells themselves, infused into the microfluidic device artificially, or embedded into the ECM and scaffolds13,287; 7.4. achieving the controllable and reproducible recruitment, proliferation, differentiation, apoptosis of NSCs/NPCs, migration of neuroblasts, maturation of newly-formed neurons and their functional integration within the NVU/brain parenchyma compartment242,244,247,264,288,289; 7.5. establishment of the molecular machinery responsible for dynamic/phasic changes in the tissue (for instance, circadian/diurnal rhythms and/or mitochondrial dynamics) that are important for determining the stem cells fate or barrier functions136,290; 7.6. achievement of prolonged viability of the neurogenic niche in vitro model enabling long-lasting recording of “developmental” and plastic changes in the brain tissue.
The generalized characteristics of the brain tissue models are summarized in Figure 5.

Summary of brain tissue models. The following parameters should be taken in consideration for reproducing the changeable brain in vitro: tissue composition, ECM mimicking, compartmentalization, tissue architecture, key brain plasticity parameters and monitoring techniques.
In the context of multi-organ-on-chip methodology, continuous media circulation and inter-tissue interactions are required for the establishment of microphysiological systems suitable for studying molecular pathogenesis, drugs pharmacokinetics and pharmacodynamics, even in a patient-specific (personalized) mode. 291 Brain plasticity based on neurogenesis, maturation and functional integration of newly-born cells into pre-existing circuits might be considered as an analog of “multi-organ-on-chip” model. In this model, different brain compartments are interconnected in the appropriate manner to provide balanced development of new neurons further disseminating within the compartment to provide a substrate for brain plasticity seen in development, learning, cognition, or memory encoding and consolidation. Thus, development of the changeable brain in vitro model consisted of the neurogenic niche (as a compartment for stimuli-driven neurogenesis), the brain parenchyma (as a compartment for neurogenesis control and assimilation of new cells), and brain tissue barriers like BBB and BCSFB (as compartments providing the adjustment of local microenvironment to actual functional and metabolic needs) is one of the most ambitious tasks in neurobioengineering and, presumably, in regenerative neurology.
Conclusions
Current progress in brain tissue modeling has made it possible to mimic biological, biochemical, and even physical (mechanical) properties of the mammalian brain in the in vitro conditions. The main goal of such mimicking is to ensure proper physiological correspondence between in vivo and in vitro processes for receiving more reliable data in neurobiology, regenerative neurology and neuropharmacology. However, there are still some significant challenges whose solving would greatly increase the relevance of existing brain tissue in vitro models. Neuroscience development as well as technology evolution allows reconstructing the 2D and 3D microarchitecture with rather accurate cell composition, physiologically relevant mechanical stimulation, oxygen and nutrients supply. The question is whether we understand correctly the crucial mechanisms underlying brain plasticity. It is a multifaceted phenomenon whose reconstruction in vitro requires the development and long-term maintenance of 4D models whose components are sensitive to the action of stimuli initiating plastic changes in the brain tissue. Thus, instead of separately assembled NVU/BBB, BCSFB or NN, we should focus on a combination of all these elements in a microfluidic platform. It might shed a light on the entire mechanisms of brain plasticity: stimulus-driven neuronal activity, synaptic transmission, glial activation, induction of neurogenesis and angiogenesis, and associated changes in blood, CSF and ISF movement. Also, being created with the patient-derived cells, such 4D models would be helpful in testing the individual trajectory of brain development and aging, or in assessing the efficacy of drugs.
Footnotes
Acknowledgements
The authors thank all respondents who took the time to participate and further distribute the survey.
Abbreviations
APP: amyloid precursor protein; AQP4: aquaporin-4; ATP: adenosine triphosphate; A2B: adenosine receptor; BBB: blood-brain barrier; BCSFB: blood-cerebrospinal fluid barrier; BM: basal membrane; BMAL: brain and muscle arnt-like; BMB: blood-meningeal barrier; BMEC: brain microvessel endothelial cell; CD: cluster of differentiation; CLOCK: circadian locomotor output cycles kaput; CNS: central nervous system; CP: choroid plexus; CPEC: choroid plexus epithelial cell; CRY: cryptochromes; CSF: cerebrospinal fluid; Cx: connexin; DCX: doublecortin; EC: endothelial cell; ECM: extracellular matrix; EpC: ependymal cell; FSS: flow shear stress; GABA: gamma-aminobutyric acid; GPR: G-protein-coupled lactate receptor; ISF: interstitial fluid; JAM: junctional adhesion molecule; MW: molecular weight; NAD: nicotinamide adenine dinucleotide; NAMPT: nicotinamide phosphorybosyltransferase; NCAM: neuronal cell adhesion molecule; NeuroD: neurogenic differentiation; NN: neurogenic niche; NPC: neuronal progenitor cells; NSC: neural stem cell; NVU: neurovascular unit; OXPHOS: oxidative phosphorylation; P: day of postnatal development; PARP: poly(ADP)-ribose polymerase; PECAM: platelet endothelial cell adhesion molecule; PER: period; PSA: polysialylated; SCN: suprachiasmatic nucleus; SGZ: subgranular zone; SHZ: subhippocampal zone; SIRT: sirtuin/histone deacetylase; SVZ: subventricular zone; TEER: transendo(epi)thelial electrical resistance; TfR: transferrin receptor; ZO: zonula occludens.
Author Contributions
A.B.S., S.N.I., and S.O.Y. contributed to the study conception and design. M.R.S. prepared figures and tables. All authors contributed to literature search, writing of the first draft of the manuscript, and commenting on previous versions of the manuscript. All authors read and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
