Abstract
The selective permeability of the blood-brain barrier (BBB) enables the necessary exchange of substances between the brain parenchyma and circulating blood and is important for the normal functioning of the central nervous system. Ischemic stroke inflicts damage upon the BBB, triggering adverse stroke outcomes such as cerebral edema, hemorrhagic transformation, and aggravated neuroinflammation. Therefore, effective repair of the damaged BBB after stroke and neovascularization that allows for the unique selective transfer of substances from the BBB after stroke is necessary and important for the recovery of brain function. This review focuses on four important therapies that have effects of BBB tissue repair after stroke in the last seven years. Most of these new therapies show increased expression of BBB tight-junction proteins, and some show beneficial results in terms of enhanced pericyte coverage at the injured vessels. This review also briefly outlines three effective classes of approaches and their mechanisms for promoting neoangiogenesis following a stroke.
Introduction
The blood-brain barrier (BBB) separates circulating blood from the brain parenchyma, protecting the brain from toxic substances in the blood, and its selective permeability ensures that the brain receives needed nutrients and other functional molecules from the blood, as well as allows the transport of metabolic wastes from the brain’s interstitial fluid into the blood, to maintain a homeostatic environment in the central nervous system (CNS) for normal neuronal function.1,2 BBB consists of endothelial cells (ECs), basement membrane (BM), pericytes, and astroglial foot processes 3 (Figure 1b). Among these, the ECs within the BBB possess a unique structure distinct from the peripheral ECs, playing an irreplaceable role in maintaining BBB function. Firstly, tight junction proteins (TJPs) within EC, composed of transmembrane proteins such as claudin and occludin, ZO proteins, and cytoskeletal proteins, selectively mediate the diffusion of small ions and water molecules. This selective diffusion is a major contributor to maintaining the low paracellular permeability of BBB. Moreover, the interaction of TJPs between adjacent ECs also provides certain physical support.4,5 Secondly, transport proteins on EC regulate the dynamic influx and efflux of specific substrates, providing nutrients to the brain parenchyma, maintaining ion balance, and eliminating metabolic waste from the brain tissues (Figure 1c). Thirdly, the low-level expression of leukocyte adhesion molecule on EC restricts the infiltration of immune cells into the CNS. 6 The BM, an extracellular matrix (ECM), acts as a hub for intercellular signaling among BBB cells, regulating the homeostatic environment within the brain. 6 Pericytes, distributed along the capillaries and enveloping the capillary walls, embedded within the BM, participate in crucial physiological processes such as regulating cerebral blood flow, repairing BBB, neurogenesis, and angiogenesis. 7 Astrocytes surrounding the capillaries form the closest layer of the neurovascular unit (NVU), 6 providing energy to neurons and modulating their excitability, maintaining synaptic growth and plasticity, 8 and aiding in interstitial fluid clearance. 9 Under normal physiological conditions, BBB tissues interact with each other to maintain an effective functional barrier role.

(a) Schematic diagram of the cerebrovascular microenvironment, (b) schematic diagram of the structure of BBB, (c) schematic diagram of the transporter function of EC, and (d) the course of BBB injury after stroke.
At present, a large number of studies have shown that the occurrence, development, and prognosis of various CNS diseases, such as acute cerebrovascular diseases (mainly stroke), Alzheimer’s disease (AD), and epilepsy are closely related to the degree of damage to the structure and function of BBB. 10 Intravascular aggregation of the causative proteins Aβ and tau in AD causes leakage of BBB, which in turn causes the flow of toxic substances from the blood into the brain parenchyma, exacerbating neurological damage and promoting the development of AD. 11 It has been proposed that repairing BBB by restoring the normal morphology of EC, increasing the number of pericytes, decreasing the number of astrocytes activated, or restoring their normal morphology may be more beneficial for the treatment of AD patients. 12 Epilepsy is a neurological disorder triggered by a homeostatic imbalance in the CNS. 13 After a seizure, the structure and function of BBB is disrupted, which proximally results in seizure recurrence.14,15 And, it has been demonstrated that the degree of BBB damage is associated with the intensity of epilepsy. 16 Recent studies have found that a specific antagonist of sphingosine 1-phosphate receptor 1 increases TJPs in injured brain sites, attenuates BBB damage, and reduces seizure severity. 17 Furthermore, RepSox, as a regulator of claudin-5 protein expression, stabilizes BBB and prevents disease seizures in experimental epilepsy. 15 It can be found that the intact structure of BBB enables BBB to better perform the role of material transfer as well as energy transport to maintain normal vital signs. The BBB is closely associated with a wide range of CNS disorders and is involved in the whole process of the diseases. Therefore, stabilizing and repairing BBB to exert selective permeability of BBB is extremely important.
Stroke is one of the most common neurological diseases and a major cause of disability and death. 18 The prevalence of stroke is expected to increase as the human population ages, putting the health of humans at greater risk. Stroke is divided into ischemic stroke and hemorrhagic stroke, of which ischemic stroke patients account for 85%. 19 Ischemic stroke is a localized ischemic and hypoxic environment in the brain caused by blocked blood circulation caused by arterial occlusion caused by an embolus or embolism. 20 Hemorrhagic stroke presents parenchymal hemorrhage, which is usually caused by chronic hypertension and rupture of small penetrating vessels in the cerebral arteries supplying the deep brain structures, and intracerebral hemorrhage (ICH) also occurs in the elderly due to cerebral amyloid angiopathy to a large extent.21 –23
Stroke disrupts the integrity of BBB and causes BBB dysfunction, which is manifested by increased BBB permeability associated with alteration in the tight junction (TJ) structure.24,25 Within minutes of the onset of cerebral ischemia, Cl−, Na+, and water enter the intracellular compartment, inhibiting cellular volume regulation and leading to cell swelling. 26 Consequently, the functionality of ion transporters within BBB is restricted, resulting in an imbalance of ions in the brain, and triggering ionic edema. 27 As the BBB is further compromised, water and certain plasma proteins extravasate into the brain parenchyma through increased permeability pores, causing vasogenic edema. 26 Cerebral edema elevates the expression of vascular endothelial growth factor (VEGF), angiopoietin-2 (Ang-2), matrix metalloproteinases (MMPs) within the BBB, disrupts the integrity of TJ, and promotes vascular edema.26,28,29 When the BBB is significantly damaged, its high permeability allows red blood cells and other cells from the bloodstream to enter the brain tissue, leading to hemorrhagic transformation.26,30 Additionally, the damage to BBB is closely associated with adverse neurological outcomes. Following a stroke, the selective permeability of BBB is compromised, resulting in oxidative stress and excitotoxicity, activating microglia and astrocytes, leading to increased production of cytokines, chemokines, MMPs, reactive oxygen species, and VEGF in the ischemic area.31 –33 These inflammatory mediators and proteases can disrupt BBB and upregulate adhesion molecules in EC, causing blood-derived inflammatory cells such as neutrophils, macrophages, and lymphocytes to migrate across transendothelial transfer to the brain parenchyma, leading to subsequent neuronal cell death. 34 In addition, damaged neurons release damage-associated molecular patterns that lead to intensified neuroinflammation by further activation of microglia, astrocytes, and leukocytes, resulting in a cascade intensification in the cell death. 31 In summary, the loss of structural integrity in the BBB leads to ion imbalances, disruption of signaling pathways, and the attack of immune cells and inflammatory mediators on CNS, causing dysfunction and even substantial neuronal death 35 (Figure 1d).
Stroke can damage the function and structure of BBB, exacerbating the disease and worsening cognitive impairment. The extent of BBB damage triggered by stroke is associated with multiple factors. Firstly, timely treatment (within a 4.5-h window for intravenous thrombolysis and a 6-h window for thrombectomy) is crucial for preserving and restoring BBB integrity post-stroke.36,37 Thrombolytic therapy using tissue plasminogen activator (tPA) during the thrombolysis period can effectively improve ischemic stroke but may exacerbate BBB disruption, intensifying cerebral edema and posing a risk of hemorrhagic transformation. 38 Missing the thrombolysis and thrombectomy windows results in severe BBB damage, necessitating comprehensive consideration in treatment, including careful selection of drug dosage, administration method, and timing. Treatment becomes increasingly challenging. Secondly, age is a critical factor affecting the severity of BBB damage post-stroke. Advancing age typically induces more severe post-stroke BBB disruption, making BBB recovery more difficult. Researches by Bacigaluppi et al. 39 indicate that aged stroke mice exhibit weaker abilities to repair BBB damage, characterized by a decrease in upregulation of genes related to ECs, pericytes, and astrocytes. Furthermore, an individual’s health condition influences the extent of BBB damage post-stroke. Hypertension damages intracranial vessels, promoting arteriosclerosis, thereby increasing stroke risk. Hypertension also compromises BBB integrity, triggering neuroinflammation. 40 Consequently, after a stroke occurs, it’s imperative to tailor rational treatment plans to patients based on disease type, severity, and stage.
BBB plays an important role in maintaining normal CNS function, therefore, it is necessary to pay attention to the repair and protection of damaged BBB after stroke, as well as the important role of BBB in neural function recovery. The following will focus on the new therapies with injured BBB repair effects after stroke in recent seven years, including cell-mediated BBB repair therapies, molecular therapies, and Wnt/β-catenin signaling pathway and VEGF signaling pathway mediated BBB repair therapies, and briefly summarize effective methods for promoting neoangiogenesis after stroke (Figure 2).
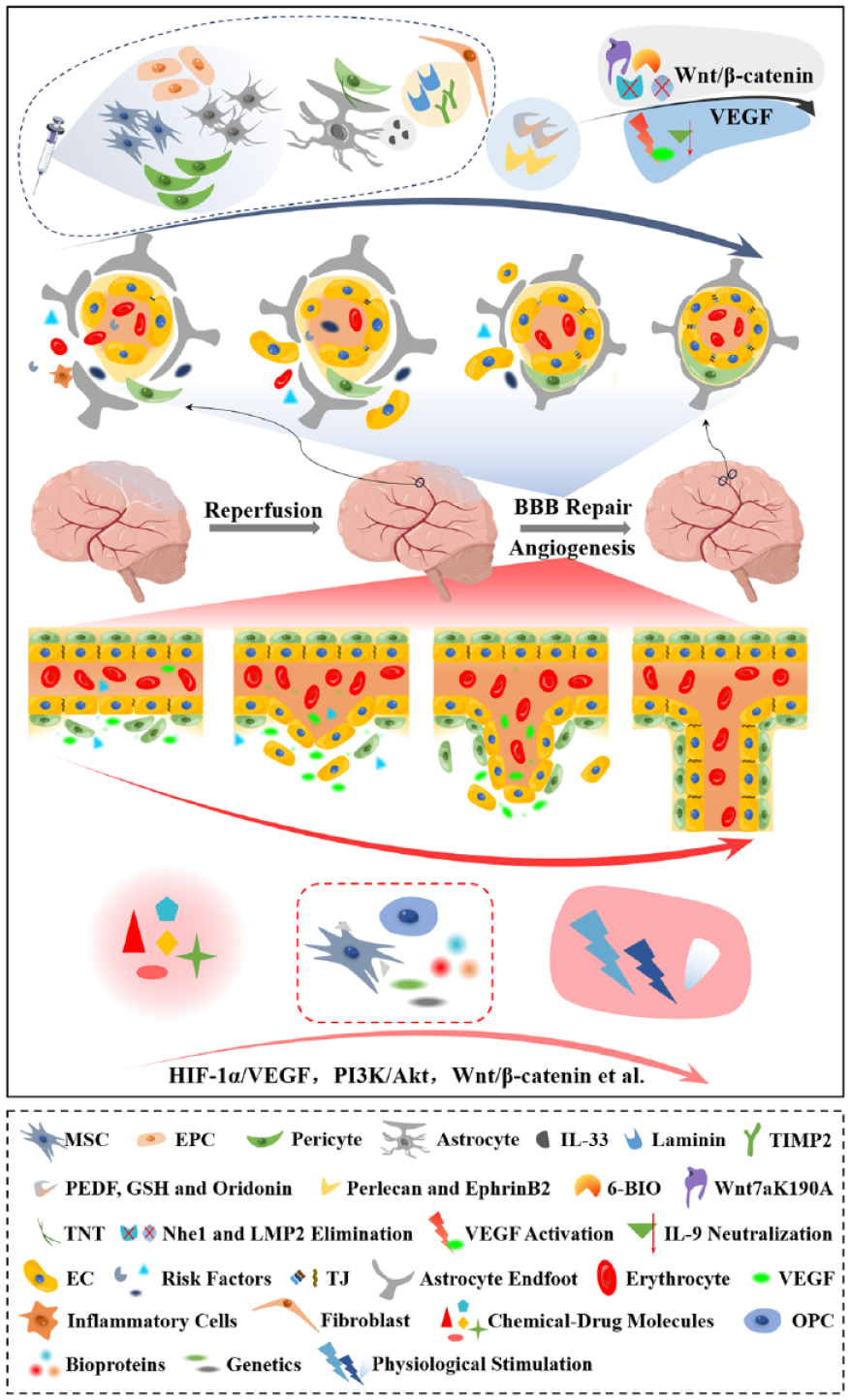
BBB repair and pro-neoangiogenic therapies after stroke.
BBB repair therapies
Cell-mediated BBB repair therapies
Thrombolytic therapy and thrombectomy can effectively alleviate blood circulation obstruction caused by thrombosis, but due to narrow time windows and serious complications, the number of patients receiving treatment is limited. Therefore, using drugs for neuroprotection, BBB repair, nerve regeneration, and neovascularization of the injured brain to prolong the treatment window or assist in thrombus clearance therapies is crucial for the recovery of brain function. Among them, cell-mediated BBB repair therapies have shown better results in repairing the damaged BBB after stroke through exogenous cell replacement and cell-cell interactions.
Cell transplantation therapies
Cell transplantation therapies use transplanted cells that differentiate into cells needed at the site of brain damage to replace missing or damaged cells or the secretome of transplanted cells to repair the damaged brain. In the study of cerebral ischemia models, mesenchymal stem cells (MSCs), endothelial progenitor cells (EPCs), neural stem cells (NSCs), and pericytes play a positive role in BBB repair through different modes of action in cell transplantation therapies.
MSC transplantation
MSC has significant advantages for the treatment of ischemic stroke due to their high availability, efficient isolation and culture, high immune tolerance, and few treatment complications. 41 Studies have shown that MSC can exert brain tissue protection and repair in the acute, subacute, and chronic phases of stroke, including alleviating inflammatory reactions, modulating immune function, inhibiting cell apoptosis, and promoting neurogenesis and angiogenesis.42 –44 The use of MSC in the treatment of stroke has been proven safe and effective, not only in animal models but also in clinical trial researches.45 –48
Namioka et al. 46 infused MSCs from the bone marrow of rat models of permanent middle cerebral artery occlusion (MCAO) after 8 weeks to investigate whether MSC infusion can improve functional outcomes in chronic ischemic stroke rats. It was found that only weak Evans Blue leakage was observed and pericyte coverage was higher in MSC-treated rats compared to vehicle-treated rats. In addition, vessel length measurements of RECA-1 ECs and PDGFRb+ pericytes within the lesion area around the ischemic core in the MSC-infused groups showed longer vessel lengths. Experimental phenomena indicate that MSC systemic infusion can promote BBB recovery in the chronic stroke phase and then improve neurological function. Studies suggest that MSC infusion can achieve effective treatment not only for acute cerebral ischemia, but also for chronic cerebral ischemia. Moreover, chronic cerebral ischemia has lower permeability of BBB compared to acute cerebral ischemia, so stereotactic drug delivery may have more significant therapeutic effects. 49 However, experiments of systemic administration for chronic cerebral ischemia still have excellent therapeutic efficacy, demonstrating the powerful therapeutic potential of MSC.
MicroRNA-126a-3p (miR-126) is an EC-specific microRNA (miRNA/miR) with important roles in promoting neoangiogenesis and maintaining vascular integrity.50,51 Wang et al. 52 injected miR-126-transfected MSCs into type VII collagenase-induced ICH rats and found that miR-126 accelerated the differentiation of MSC to vascular EC. And compared to the control group injected with untransfected MSCs, brain tissue injected with miR-126-modified MSCs showed lower expression of protease activated receptor-1 and matrix metalloproteinase-9 (MMP-9), and higher expression of ZO-1 and claudin-5, exhibiting a more complete BBB structure. Thus, miR-126-modified MSCs have unique advantages in repairing damaged BBB after ICH (Figure 3). However, the potential mechanisms underlying the BBB repair and neurological recovery effects of miR-126-modified MSCs in ICH still need to be further explored. The excessive differentiation of MSCs into vascular ECs observed in the experiments is an extremely important reason why transplanted MSCs can play a role in BBB repair. Inspired by these studies, the use of a pericyte-specific miRNA to modify MSCs may also be able to play a role in BBB repair, but pericyte-specific miRNA is yet to be explored.
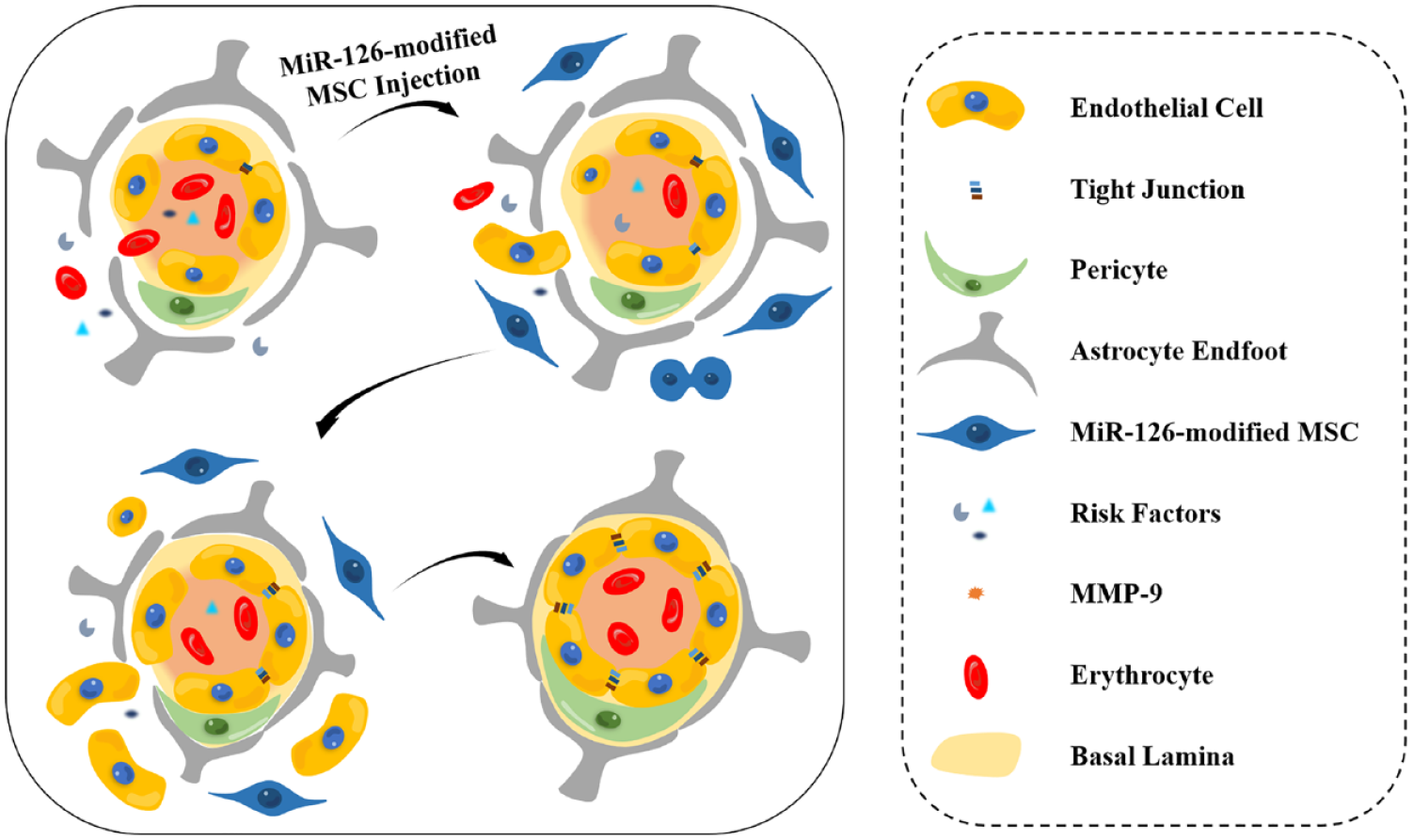
The drug treatment of MSC modified by miR-126 on ICH models can effectively repair the BBB damage caused by ischemia and hypoxia. MiR-126-modified MSCs replace apoptotic ECs by differentiating into healthy ECs, and attenuate the adverse effects of MMP-9 on BBB, in addition, MSCs may be able to repair damaged ECs by paracrine. MiR-126-modified MSCs can increase the expression of TJP in ECs and make the structure of BBB more complete.
EPC transplantation
EPC, a precursor of vascular EC, can be obtained from circulating monocyte populations, umbilical cord blood, and BM.53,54 It was found that when vessels are exposed to ischemia and hypoxia, perfusion of BM-derived EPC enables them to migrate to the ischemic region and differentiate into mature vascular EC to repair damaged vessels and promote new vessel formation, or secrete cytokines, including VEGF and basic fibroblast growth factor, to indirectly participate in vascular remodeling.55,56
Garbuzova-Davis et al. 57 studied BBB recovery in rats that underwent intravenous transplantation of β-galactosidase prelabeled human bone marrow endothelial progenitor cells (HBMEPCs) 48 h after transient middle cerebral artery occlusion (tMCAO). Ultrastructural analysis of microvessels in the bilateral striatum and motor cortex of brain injury rats 5 days after cell therapy showed widespread vascular repair, which was mainly due to the extensive intravascular implantation of HBMEPCs. Moreover, the number of normal form mitochondria in ECs and perivascular astrocytes was significantly increased. In addition, many vesicles appear in the cells after HBMEPC implantation, and cells have transcytosis that allows rapid drainage of fluid from the damaged brain, which is consistent with the results of perivascular edema that was not detected in the experiments. The experimental results show that HBMEPC transplantation has a strong cellular therapeutic function, and preservation of mitochondria and increased transcytosis represent a new neural repair mechanism for BBB repair in the subacute phase after stroke.
EPC transplantation is an effective stroke therapeutic, but given the lack of adequate safety of cellular therapies in clinical translation with potential side effects, research into new approaches to advance the clinical treatment of stroke is necessary. 58 Loiola et al. 59 mass-produced EPC-secretome from stroke patient (scCM) to investigate their effects on post-stroke vascular barrier integrity. In vitro experiments revealed that scCM reduced the permeability of the EC monolayer models and upregulated the expression of the junctional proteins Occludin, VE-cadherin, and ZO-1. Furthermore, scCM avoided leakage in the BBB models caused by oxygen-glucose deprivation (OGD), which could be attributed to the upregulation of junctional proteins and the modulation of MAPK/VEGFR2 signaling activity. The experimental results show that scCM facilitates vascular maturation and functional recovery of the BBB.
EPCs positively affect vascular repair after stroke due to their ability to differentiate into few cell lines including ECs, but the scarcity of EPCs in both bone marrow and peripheral blood limits their therapeutic potential in ischemic stroke. 60 Abdulkadir et al. 61 performed in vitro cultivation of outgrowth endothelial cells (OECs) and investigated their ability to recover BBB after brain injury. Wound scratch assays showed that OECs have a greater proliferative and directional migration capacity compared to human bone marrow endothelial cells (HBMECs). In in vitro models of BBB established with astrocytes, pericytes, and HBMECs, exogenous OECs were effective in repairing damaged ECs caused by serum-free conditions. In addition, wound scratch experiments with a monolayer of HBMECs further confirmed that OECs have the endothelial repair effects. In vitro cellular assays show that OECs can effectively repair damaged BBB, thus suggesting that repairing ischemic stroke-induced BBB damage with these low-energy cells may be an effective therapeutic strategy.
NSC transplantation
NSC is a pluripotent cell with the ability to differentiate into a wide spectrum of cell lineages, including neurons and astrocytes. 62 The transplantation of NSCs stands out as a promising therapeutic strategy for the treatment of ischemic stroke. However, only a small fraction of transplanted NSCs manages to survive in the long run, with the majority of these surviving cells differentiating into astrocytes around the infarct core. This phenomenon restricts the therapeutic efficacy of the transplanted cells.63,64 To explore a solution, Ge et al. 65 loaded VEGF into gelatin and modified NSC by layer-by-layer assembly of the loaded gelatin and hyaluronic acid (LbL (VEGF)-NSC). Cell therapy following tail vein infusion of LbL (VEGF)-NSC in mice with distal middle cerebral artery occlusion was found to promote the survival of implanted NSCs and the ability of NSCs to differentiate into neurons, and transplanted LbL (VEGF)-NSC promoted angiogenesis and BBB repair in the penumbra of the stroke mice. In the experiment, the method of vein transplantation was used to transplant modified stem cells, which had the lowest invasiveness but resulted in fewer stem cells acting on the lesion area. 66 Local intracerebral injection of drugs can increase brain damage, although they can bring more stem cells to the site of the lesion. Therefore, optimizing the implantation method becomes crucial, striking a balance between the efficacy of damage repair and minimizing additional injury, a pivotal consideration in cell therapy approaches.
Pericyte transplantation
Pericyte replacement therapy holds promise for restoring the structure of the BBB, but the paucity of material sources greatly limits the use of primary pericytes in cell transplantation studies as well as in clinical therapy. Sun et al. 67 obtained pericyte-like cells with a cranial neural crest origin in human pluripotent stem cells (hPSC-CNC PCs) and transplanted hPSC-CNC PCs into mouse models of tMCAO. It was found that hPSC-CNC PC treatment significantly increased PC coverage and improved TJP expression compared to control groups. In addition, the transplanted hPSC-CNC PCs can migrate to the sites of BBB damage for more effective BBB reconstruction. These experiments suggest that hPSC-CNC PC has the potential to repair the damaged BBB following ischemic stroke.
Cell transplantation therapies have the potential for BBB repair after stroke, but suboptimal clinical outcomes and unclear molecular mechanisms demonstrated by certain cell transplantation therapies have limited their clinical translation. Among them, unsatisfactory clinical outcomes are directly related to the potential side effects of cell transplantation. The type, route, and timing of cell delivery and the regulation difficulty of cell dose are also strongly associated with poor clinical outcomes after stroke. 68 Therefore, developing the optimal cell therapy plan for stroke patients with different degrees of illness still requires considerable effort, and optimizing diagnosis and treatment plans may receive the help of artificial intelligence in the future. In addition, delving deeper into the intrinsic molecular mechanisms of cell transplantation therapies is crucial to advance safe and effective clinical applications.
Pericyte therapies
As an important component of the BBB, the pericyte plays active roles in maintaining the normal structure and function of the BBB. This is demonstrated not only by the barrier role of the pericyte in selective substance exchange in the BBB, but also by the ability of the pericyte to positively regulate the function of other BBB tissues. 69 It was found that pericyte-secreted angiopoietin-1 (Ang-1) mediates TJ formation by activating its receptor Tie-2 on the EC, 70 and that pericyte-derived neurotrophic factors can upregulate claudin-5 expression and increase BBB integrity. 71 Pericyte is also able to promote basal lamina formation by secreting extracellular matrix proteins. 69 After a stroke episode, pericytes at the sites of brain injury are capable of mediating BBB repair through the above pathways, in addition to the repair of adjacent injured cells through functional mitochondrial transfer.
Tunneling nanotubes (TNT) are elongated protrusions that enable long-distance connections between the cytoskeleton and plasma membrane of different cells and can transfer and communicate material between cells, which is essential for maintaining a healthy intercellular state and repairing adjacent damaged cells.72 –74 Previous studies have shown that in in vitro ischemia-reperfusion (I/R) models, mitochondria in MSC are able to transfer into damaged EC via TNT to rescue EC from apoptosis. 74 In contrast, Pisani et al. 75 showcased in vitro that BBB constituent cells can also achieve material transfer through TNT, as demonstrated by co-culture models of pericytes, ECs, and astrocytes and by intercellular transfer of mitochondria in 3D multicellular assemblies of human BBB. Under the condition of oxygen-glucose deprivation/reoxygenation (OGD/R), functional mitochondria in pericytes are transferred to injured astrocytes via TNT to rescue apoptosis. The ability of pericytes to rescue damaged astrocytes using TNT provides a closer elucidation of the mechanism of organismal repair of the BBB after stroke and provides theoretical support for the optimization of post-stroke BBB repair measures.
Pericyte interact synergistically with other components of the BBB, contributing to its repair after stroke. The absence of pericyte can lead to abnormal TJ structure, increased BBB permeability, and BBB integrity disruption. 76 Thus, pericyte plays an irreplaceable role in maintaining normal BBB function. However, the pathways and molecular mechanisms by which pericyte communicates signals with various components of BBB to maintain and repair BBB under physiological and pathological conditions in the brain have not been adequately clarified, and research on the pericyte-mediated repair of BBB damage has been unfavorable to the extent that there is insufficient theoretical support for clinical translation, posing challenges for clinical applications. Furthermore, during the acute phase of ischemic stroke, pericyte contributes to vascular constriction, leading to a no-reflow phenomenon post-reperfusion, causing further BBB damage. 77 Following vasoconstriction, pericyte death occurs, reducing pericyte coverage and exacerbating BBB injury. 78 Hence, in the acute phase of cerebral ischemia, appropriately managing pericyte vasoconstriction and fostering pericyte recruitment to enhance cellular coverage may serve as an effective research strategy.
Astrocyte therapies
Astrocyte is an important component of the BBB, stretching to envelop more than 90% of capillary ECs and pericytes, capable of expressing endothelial transport molecules such as glucose transporter protein-1, and their termini can release signaling molecules to support the formation and maintenance of TJ structure, playing an important role in the structure and function of the BBB.79,80 After a stroke, astrocyte is activated and transition from a resting state to a reactive state, exhibiting two distinct types of reactive astrocytes known as A1 pro-inflammatory and A2 anti-inflammatory types. 81 A1 astrocytes upregulate neurotoxins and pro-inflammatory factors such as TNF-α, IL-6, and IL-1β, negatively impacting the structural integrity of the BBB. Conversely, A2 astrocytes secrete IGF-1, IL-2, IL-10, and TGF-β, accelerating the anti-inflammatory process. 82 Research indicates that post-stroke A2 astrocytes, expressing increased levels of TGF-β and VEGF, are associated with beneficial outcomes, including reduced BBB permeability and vascular damage improvement. 83 The increased neurotrophic factors and cytokines from A2 astrocytes support neuronal survival and repair. 84 Studies have shown that metabolic shifts in reactive astrocytes can be a key target for recovery of brain function in the subacute phase after stroke. 85 More recently, studies have recently found that lipid metabolism in reactive astrocytes has a positive impact on the recovery of BBB after stroke.
IL-33 is an astrocyte-derived factor with neuroprotective effects after stroke, but its role in the BBB after cerebral ischemia is unclear. 86 Wei et al. 87 studied the tMCAO mouse models and found that astrocyte-specific fatty acid synthase (FASN)-dependent adipogenic responses were activated during the subacute phase of cerebral ischemia and that the level of IL-33 in the infarct area was increased. In contrast, inhibition of astrocyte lipogenesis reduced levels of ZO-1, Occludin, and Claudin-5, increased BBB permeability, and inhibited IL-33 production. Supplementation of IL-33 to mice with disrupted lipogenesis elevated TJP levels in the infarct region and rescued BBB destruction due to failed lipogenesis. Studies have shown that lipogenesis in astrocytes promotes the production of the downstream molecule IL-33 to repair BBB. It is theoretically observed that IL-33 may be an effective target for injured BBB repair after ischemic stroke. However, lacking agonists and activators of FASN, astrocyte-specific FASN-dependent adipogenesis failed to be addressed. It may be possible to use IL-33 as a therapeutic target for BBB repair.
Astrocytes, as cells essential for the maintenance of selective substance exchange in the BBB, can normalize the function of the BBB by interacting with other surrounding cells. The positive roles of astrocytes in the study of BBB repair after stroke are gradually being discovered, making some molecular signals therapeutic targets for BBB repair. In addition, astrocyte-mediated inflammatory responses after stroke are detrimental to BBB recovery, and their double-edged effects need to be looked at in research. There are relatively few studies on astrocyte-mediated BBB repair, which may be because the inflammatory response induced by astrocytes after stroke affects the BBB repair function of astrocytes, which increases the difficulty of experimentation.
Fibroblast therapies
Fibroblasts are a heterogeneous cell population. In the CNS, fibroblasts are mainly located in the meninges and perivascular spaces. 88 There is evidence that fibroblast can protect BBB integrity and reduce stroke-induced hemorrhagic transformation in ischemic stroke. 89 Recent discoveries have indicated that fibroblasts, along with their secreted proteins, also contribute to the repair of a damaged BBB following ICH.
Xu et al. 90 generated mouse mutants with ablated Col1α1+ fibroblasts by targeting the fibroblast-specific marker Col1α1 to explore the roles of fibroblasts in ICH, which also largely avoided misguided findings from non-fibroblast-specific markers. Mutant mice were found to have increased BBB injury caused by ICH and reduced pericyte coverage at the sites of brain injury. Correspondingly, in in vitro ICH models, fibroblasts promoted the integrity of BBB. The positive effects of Col1α1+ fibroblasts were found to be at least partially attributable to the upregulation of TJPs by fibroblast-secreted tissue inhibitor of metalloproteinases 2 (TIMP2), which is consistent with the administration of TIMP2 to the ICH models attenuating BBB damage and increasing the expression of ZO-1 and occluding. In these studies, the repair effects of Col1α1+ fibroblasts on the damaged BBB after ICH suggest that Col1α1+ fibroblasts and TIMP2 may be targets for ICH therapy. However, the injured BBB repair roles of perivascular fibroblasts, but not meningeal fibroblasts after ICH remain to be tested in the future using tools that specifically label perivascular fibroblasts.
Laminin is an extracellular matrix protein that plays an important role in the formation and maintenance of BBB integrity. 91 However, the function of laminins secreted by fibroblasts in maintaining the BBB remains unknown. Xu et al. 92 used the Col1α1-Cre line to generate mutant mice with lamininγ1 deficiency in fibroblast-like cells. ICH induction in mice revealed mutants showing more severe BBB injury and brain damage than control mice, and results such as enhanced BBB permeability, reduced TJ structural integrity and PDGFRβ coverage, and increased hematoma volume predict that fibroblast-derived laminins may play a role in BBB repair in ICH. In addition, fibroblast-derived laminins were found to significantly affect the migration of fibroblasts. Studies suggest that in the pathogenesis of stroke, fibroblast-derived laminins can repair the BBB more effectively by promoting fibroblast migration to the hematoma area. Laminin may be a valuable target in the treatment of ICH. Experiments have also confirmed that integrin β1 is not a receptor that regulates laminin-mediated fibroblast function.
Xu et al.90,92 conducted a series of studies on the role of fibroblasts in the treatment of ICH, showing positive roles for fibroblasts in the treatment of BBB after ICH. Laminins secreted by fibroblasts have a role in repairing BBB and brain injury after ICH and also promote fibroblast migration to areas of vascular hemorrhagic injury. Fibroblasts surrounding the hematoma are in turn able to repair the damaged BBB through other beneficial pathways, including molecular pathways mediated by the fibroblast secretion TIMP2 (Figure 4). Furthermore, in studies of BBB repair after stroke, attention needs to be paid to the negative effects that fibroblasts have, for example, the inhibition of neuronal regeneration after CNS injury by fibroblast-derived ECM proteins in scar tissue.
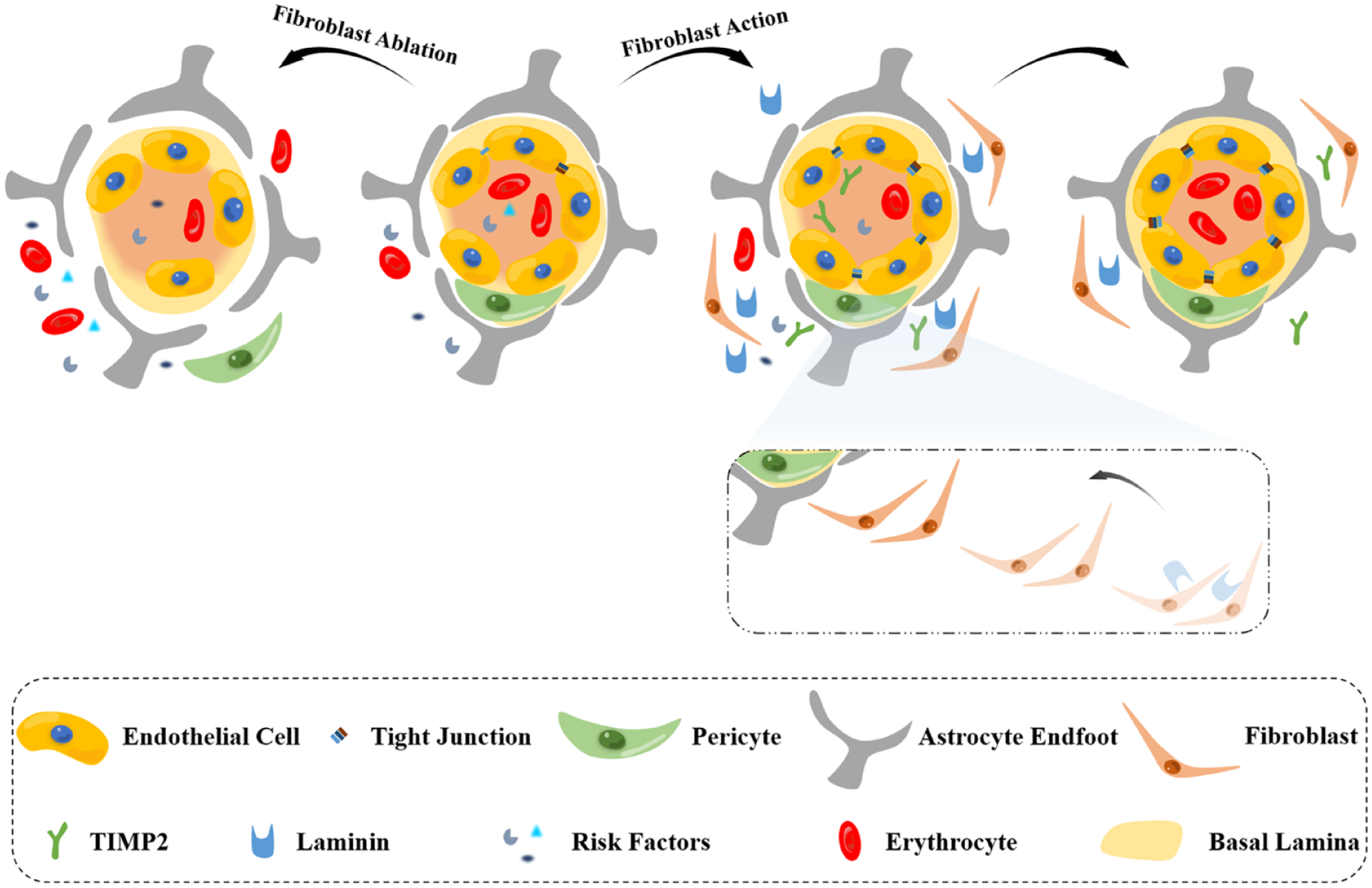
After ICH, fibroblasts at the brain injury site can secrete TIMP2 and laminin, protecting part of TJPs from the damage of inflammatory factors, and upregulating the expression of TJPs. Moreover, laminin secreted by fibroblasts can enable fibroblasts to migrate to the brain injury area, which further promotes BBB recovery. The use of BBB repair drugs for adjuvant treatment can more quickly restore the complete structure of the BBB. However, fibroblast ablation will make the BBB of the ICH brain face collapse.
Cell-mediated therapeutic approaches for post-stroke BBB repair can increase the number of beneficial cells in the damaged area, upregulate TJP expression, increase pericyte coverage, reduce BBB permeability, and improve brain function impairment. Different therapeutic methods repair BBB through diverse pathways. Among these, cell transplantation therapies utilize transplanted cells to differentiate into cells needed at the site of brain injury, replacing deficient cells or secreting nutrients beneficial for BBB repair, thus enhancing BBB integrity and ameliorating BBB dysfunction. Cell transplantation therapies appear relatively safe and effective, particularly the MSC transplantation methods, showing advantages in clinical trials. However, cell transplantation therapies still present some side effects, and the underlying mechanisms behind their BBB repair roles remain unclear. Additionally, the difficulty in regulating the type, route, timing of cell delivery, and cell dosage pose obstacles to the clinical application of cell transplantation therapies. Pericyte therapies demonstrate the beneficial effects of enhanced pericyte coverage at the ischemic site on BBB and pericyte’s reparative effects on other tissues within the ischemic and hypoxic microenvironment. Yet, the lack of molecular mechanisms that pericyte-mediated repair related to injured BBB after stroke and limited preclinical studies makes safe and effective clinical application more distant. Astrocyte and fibroblast therapies indicate that astrocyte and fibroblast secretions enhance the structural integrity of TJ and favor the functional recovery of BBB.
Furthermore, peripheral immune cells have a double-edged role in the pathological process of stroke, in which cellular phenotypes with anti-inflammatory properties favor the restoration of BBB integrity after stroke. 93 Research has found that polymorphonuclear neutrophils (PMNs) release anti-inflammatory molecules, such as annexin-1, lipoxin A4, and resolutions, to alleviate the inflammatory response after stroke, and PMNs can clear apoptotic cell debris.94,95 M2 microglia release anti-inflammatory factors such as IL-4, IL-10, and TGF-β to maintain the integrity of the BBB.93,96 Th2 cell subtypes release anti-inflammatory factors such as IL-4, IL-5, IL-10, and IL-13 and promote M2 polarization to maintain BBB integrity.93,97,98 The anti-inflammatory properties of peripheral immune cells in the disease progression of stroke favor the reduction of BBB damage by inflammatory mediators and deserve to be investigated in the repair of BBB after stroke.
Molecular therapies
Several molecules such as amino acids, proteins, herbal extracts, and sugars show potential for BBB repair after stroke.
Glutathione (GSH) is an intracellular thiol tripeptide present in all mammalian tissues. Chen et al. 99 performed gastric perfusion of GSH in tMCAO rats. The assays found that GSH administration reversed the protein expression downregulation of ZO-1 and claudin-5 in the brain induced by I/R surgery, reduced toxin levels in the brain and endomycin levels in the plasma of rats with cerebral ischemia, and repaired hypoxia-induced cellular damage. The results of the study show that oral exogenous GSH is effective in repairing ischemic stroke-induced BBB damage.
Pigment epithelium-derived factor (PEDF) is a neurotrophic factor with anti-angiogenic and anti-permeability effects. 100 Riabinska et al. 101 performed intraventricular PEDF infusion in tMCAO mouse models and found that brain edema and brain cell death were significantly reduced in the onset mice after administration of the drug. These results may be due to an increase in the Claudin-5, which improves the paracellular pathways and thus gradually restores the function of the BBB. Furthermore, the ability of PEDF administered after cerebral ischemia to counteract the VEGF-induced increase in BBB permeability suggests that the timing of PEDF administration is important for stroke treatment. In these regards, moderate administration of PEDF in the acute phase of ischemic stroke plays a beneficial role at least in maintaining BBB integrity. In the vascular remodeling stage after stroke, PEDF attenuates VEGF-mediated neoangiogenesis, which is detrimental to the recovery of neurological function. However, in the late stage of BBB recovery, PEDF administration may be able to avoid the negative effects of excessive VEGF on neointimal maturation and repair of damaged vessels.
Oridonin, a natural diterpenoid compound derived from a Chinese herb, has demonstrated anti-oxidative stress effects across various disease models. 102 Li et al. 103 administered oridonin to tMCAO and OGD/R models and detected increased expression of occludin, claudin-5, and ZO-1 in TJP. Oridonin has been shown to promote BBB repair and has the potential as a post-stroke therapeutic agent.
Perlecan is a large multidomain heparan sulfate proteoglycan present in the BBBECM. 104 Nakamura et al. 105 studied tMCAO models and found that conditioned perlecan-deficient mice exhibited more significant BBB leakage as well as pericyte deficiency compared to controls. In contrast, the increased number of pericytes at the site of cerebral ischemia in control mice is dependent on perlecan C-terminal domain V enhancing phosphorylation of Src homology 2 domain-containing phosphatase 2 and FAK via PDGFRβ in concert with pericyte integrin α5β1, promoting pericytes migration and contributing to BBB maturation. Researches suggest that perlecan plays an important role in the BBB recovery phase of ischemic stroke by promoting the recruitment of pericyte in the infarcted area to the microvasculature to enhance the integrity of the BBB.
The transmembrane glycoprotein ephrinB2 and its receptor EphB4 are more active at developmental stages and are involved in vascular remodeling. 106 Ghori et al. 107 further explored its potential role in tMCAO cerebral ischemia models. It was found that activation of ephrinB2 resulted in increased pericyte recruitment at the site of injured vessels and increased EC-pericyte interactions, allowing effective repair of ischemia-induced neurovascular injury. In contrast, inhibition of ephrinB2/Ephb4 signaling resulted in increased brain swelling. Studies suggest that drugs with a role in promoting the endogenous expression of ephrinB2 have greater research significance in the repair treatment of damaged BBB after ischemic stroke.
Administration of PEDF, GSH, and oridonin to ischemic stroke models increased the expression of TJPs at the site of brain injury, which in turn enhanced the structural integrity of the BBB. The beneficial effects of perlecan and ephrinB2 on BBB repair were to promote pericyte recruitment at the site of injury. In BBB repair therapy, it is necessary to pay attention to the appropriate selection of drug delivery methods to minimize the ratio of accessible brain injury to BBB repair effects.
Wnt/β-catenin signaling pathway-mediated BBB repair therapies
Wnt/β-catenin signaling pathway has the function of promoting BBB formation and maturation and stabilizing BBB in the physiological structure regulation of BBB, which is of great significance for BBB recovery after CNS diseases.108,109 Wnt ligands are mainly secreted by astrocytes and neurons, and Wnt ligands stabilize β-catenin in the cytoplasm by activating the endothelial receptor complex formed by Frizzled and LRP5/6. 110 Signaling by β-catenin can induce TJP expression and contribute to the repair of damaged BBB. 108 Given this, therapies that activate the Wnt/β-catenin signaling pathway to achieve repair of the damaged BBB have an important role in the stabilization and repair of the BBB after stroke.
Wnt7a/b ligands are produced by neuroepithelial cells in the CNS and can regulate BBB maturation.108,111,112 Therefore, targeting Wnt7a/b pathway to regulate BBB integrity is a reasonable strategy. However, drug delivery of Wnt7a/b is difficult due to the poor results caused by the mixed signaling pattern of Wnt7a/b and Frizzled receptors.113,114 In view of this, the Free University of Brussels transformed the Wnt7a ligand into a Gpr124/Reck-specific agonist (Wnt7aK190A), making it unable to activate Frizzled receptors without Gpr124/Reck. The agonist specifically targets the Gpr124/Reck co-receptor complex enriched on EC in BBB and enables the activation of downstream molecular signals such as Frizzled. The important significance of this method is to reduce the toxic effects caused by the signal communication between Wnt7a ligand and Frizzled at other sites. Mice after agonist administration were subjected to tMCAO surgery with control groups. It was found that mice treated with Wnt7aK190A showed smaller infarct volume, and the vascular leakage caused by reperfusion was also limited, and the BBB function was improved 115 (Figure 5a). These beneficial results can’t explain the direct therapeutic effects of Wnt7aK190A on BBB, which need further research to clarify. In addition, the lipophilicity of Gpr124/Reck agonists complicates large-scale recombinant production. Overall, large-scale induced Wnt7aK190A increases Wnt7a signaling in brain damaged sites, which can control Wnt/β-catenin dependent BBB maturation.108,111,112

(a) Molecular mechanisms of drug delivery and gene silencing-mediated Wnt/β-catenin pathway and (b) Wnt7a is expressed in ovarian cancer cells and promotes ovarian tumor growth.
6-Bromoindirubin-3′-oxime (6-BIO) is a cell-permeable, non-toxic molecule that specifically activates the classical Wnt pathway in stem cells. LeBlanc et al. 109 found that 6-BIO promoted the expression of claudin-3/5 and inhibited the expression of the plasmalemma vesicle associated proteins to reduce BBB breakdown after activating Wnt pathway. Studies have shown that 6-BIO can effectively promote the Wnt pathway and repair the damaged BBB after stroke. In addition, delaying recombinant tissue plasminogen activator (rtPA) administration to mice administered 6-BIO 6 h after stroke reduced the incidence of hemorrhagic transformation and side effects of the drug. The effects of 6-BIO administration on post-stroke rtPA treatment suggest that activation of the Wnt pathway after ischemic stroke can repair part of the damaged BBB and contribute to the maintenance of BBB integrity allowing an extended time window for rtPA treatment. 6-BIO may be able to be used as an ancillary drug to rtPA treatment.
Nhe1 is an ion exchange protein that is widely distributed in astrocytes. The post-stroke ischemic environment leads to sustained activation of Nhe1, which results in Na+ overload in astrocytes and triggers astrocyte swelling, 116 and the function and structure of astrocytes are thus destroyed. Begum et al. 117 performed selective deletion of the Nhe1 gene in astrocytes in mice subsequently subjected to tMCAO stroke induction. It was found that Nhe1 elimination attenuated astrocyte hypertrophy, reduced BBB damage and possessed a more intact TJ structure. Recently, the team further explored and found that expression of Wnt7a/b in the astrocyte and Wnt7a/b and β-catenin in the EC of the ischemic region was significantly increased in the brains of Nhe1 gene deletion, with a substantial reduction in paracellular permeability. Administration of XAV-939 (a β-catenin inhibitor) revealed that the protective effects of the gene deletion on BBB were eliminated. 118 Experimental phenomena suggest that the astrocyte maintains BBB integrity after ischemic stroke by stimulating Wnt/β-catenin signaling. Studies suggest that targeting the Nhe1 proteins is a novel approach to functionally convert abnormal astrocytes into endogenous drugs with BBB repair after ischemic stroke. While studies have shown that Nhe1 gene deletion prior to stroke allows astrocytes to retain most of their function and structure after stroke, it may also be possible to normalize astrocyte function to some extent by inhibiting Nhe1 activity during the acute phase of ischemic stroke, allowing them to activate the Wnt/β-catenin signaling pathway to repair BBB damage.
Proteasome inhibitors help to maintain the integrity of the BBB after stroke, but their indiscriminate inhibition of protein degradation can cause damage to cells.119,120 Chen et al. 121 found that inhibition of low molecular mass peptide 2 (LMP2), the major catalytic subunit of the immunoproteasome, promoted angiogenesis in tMCAO rats. The team further studied the connection between LMP2 and the integrity of BBB and related molecular mechanisms. Experiments showed that LMP2 inhibition in immunoproteasome of rats significantly increased Occludin, Claudin-1, and ZO-1 levels in EC, upregulated Wnt/β-catenin signaling and improved BBB integrity. In addition, silencing LMP2 gene expression also promoted proliferation and migration of rat brain microvascular ECs after OGD/R, which is important for vascular repair and BBB maturation. The disruption of BBB protection by LMP2 gene silencing caused by combined silencing of the β-catenin gene demonstrates that the enhancement of BBB integrity caused by inhibition of LMP2 after stroke is associated with activation of the Wnt/β-catenin pathway. 122 Both in vitro and in vivo experiments were conducted to inhibit the expression of LMP2 gene before the establishment of stroke models, which is different from the exploration of the repair mechanisms of BBB after stroke, but still can provide effective theoretical support for the study of tissue repair of BBB after stroke.
The effects of Wnt/β-catenin signaling pathway to promote the enhanced expression of TJPs can effectively repair the damaged BBB after stroke. In recent years, many studies have identified new ways to achieve BBB repair by mediating Wnt/β-catenin signaling pathway and these methods are valuable for the recovery of injured vessels and the maturation of new vessels after stroke. The endogenous activation of Wnt/β-catenin signaling has an overall high safety profile in stroke therapy; in contrast, exogenous Wnt ligand administration needs to avoid adverse outcomes such as organ damage and tumor development caused by high levels of Wnt protein. Therefore, exploring new pathways to promote Wnt/β-catenin signaling is of great significance for BBB repair after stroke. In addition, advancing studies on exogenous therapy using Gpr124/Reck agonists and developing new methods for delivering Wnt proteins into the brain are crucial steps forward.
Treatments of stroke mice with Wnt7aK190A, 6-BIO drug delivery and gene expression inhibition increase the integrity of BBB by increasing the amount of Wnt proteins in sites of injury as well as stabilizing β-catenin. The molecular mechanism is that binding of Wnt protein to the Frizzled and LRP5/6 receptor complexes phosphorylates the cytoplasmic region of LRP5/6 by GSK3β, resulting in the recruitment of Dvl and Axin proteins to the cytoplasmic regions of Frizzled and LRP5/6 receptors, respectively. Furthermore, disruption of the disruptive complexes by Dvl, as well as inhibition of the phosphorylation of β-catenin, which in turn inhibiting its degradation. β-catenin accumulates in the cytoplasm and translocates to the nucleus and binds to TCF/LEF transcription factors to regulate gene transcription. 123
The advantage of Wnt7aK190A drug therapy is that only when the Gpr124/ Reck endothelial receptor complex is present, Wnt7aK190A is able to communicate with the Fz receptor, which in turn carries out the signaling of Wnt/β-catenin pathway to avoid the harmful effects of Wnt overexpression on other organs and tissues. King et al. 124 found that Wnt7a in mouse ovarian cancer cells activates the Wnt/β-catenin signaling pathway and promotes tumor growth in the ovary.
VEGF signaling pathway mediated BBB repair therapies
VEGF is angioneurins expressed by damaged neurons, astrocytes, macrophages, and vascular ECs after ischemic stroke.125,126 Cerebral ischemia triggers increased expression of VEGF in the brain parenchyma, which in turn may cause increased activity of MMP-9 to damage the integrity of BBB. 127 In the acute phase of ischemia, the upregulation of endogenous VEGF level increases the permeability of the BBB, which is prone to exacerbate neuroinflammation, brain edema, and hemorrhagic transformation. 128 However, in the recovery period of ischemic stroke, VEGF participates in angiogenesis and plays a beneficial role in the functional recovery of BBB. 126
Ghori et al. 129 operated immediately activating VEGF signal in tMCAO mice. Their findings revealed that early activation of VEGF signaling, compared to control groups, led to decreased vascular permeability and increased coverage of Claudin-5 and pericytes in the affected mice. Thus, early overexpression of VEGF accelerates early vascular repair and enhances BBB integrity. It is suggested that the time window of VEGF activation has a greater impact on BBB recovery after stroke.
IL-9 is a pleiotropic cytokine. Tan et al. 130 applied an anti-IL-9-neutralizing antibody to tMCAO models in the acute phase, which significantly reduced the cerebral infarct volume and BBB destruction in rats. Compared with the control groups, part of TJP was repaired in rats treated with an anti-IL-9-neutralizing antibody, and the expression of astrocyte-derived VEGF-A was downregulated in the ischemic area. Studies have shown that the BBB repair effects of anti-IL-9-neutralizing antibodies through downregulation of astrocyte-derived VEGF-A may be due to the synergistic effects of alleviated VEGF-A damage and the body’s own repair.
Studies on VEGF pathway-mediated BBB repair typically focus on the acute phase of cerebral ischemia due to the different roles of VEGF at different times in stroke. In the acute stage of stroke with extremely damaged BBB, upregulation of endogenous expression of VEGF has negative impacts on stroke therapy, therefore, VEGF signaling should be appropriately inhibited at this stage. In contrast, activation of VEGF immediately after cerebral ischemia demonstrated the role of BBB repair, again demonstrating that VEGF-mediated BBB repair therapies need to take into account the pathological stage of stroke.
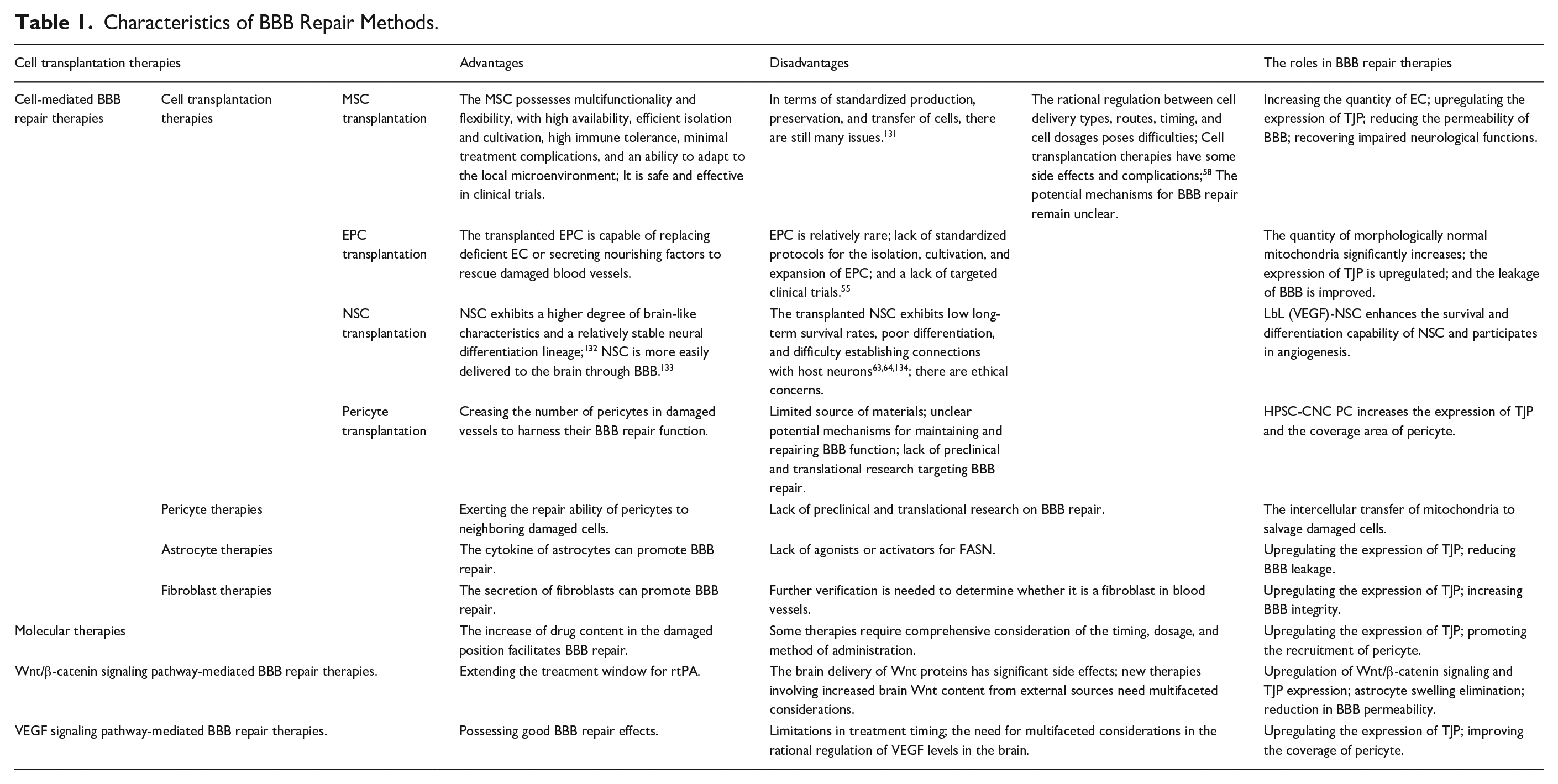
BBB is a bridge for the exchange of substances between brain parenchyma and circulating blood, and its selective permeability can maintain a normal homeostatic environment within the brain, which plays an important role in the normal operation of the CNS. Stroke exposes the BBB to a hypoxic-ischemic microenvironment, which disrupts the structural integrity of the BBB so that normal physiological functions of the BBB are lost. Severe BBB damage can put patients at risk of brain hemorrhage. Therefore, treatment of the integrity of the BBB after stroke is essential. In recent years, studies of BBB repair after stroke have identified multiple pathways that are effective in repairing BBB. Cell-mediated therapies for BBB repair demonstrate various cells rescuing brain damage post-stroke through different pathways, but unclear potential mechanisms and limitations of some treatments have hindered progress in their application. Molecular therapies aim to enhance BBB repair molecules at the site of brain injury, improving BBB dysfunction. However, treatment approaches require comprehensive consideration regarding administration timing, methods, and drug dosages based on the extent of stroke damage. Wnt/β-catenin signaling-mediated BBB repair therapies involve increasing brain Wnt protein levels and stabilizing β-catenin to regulate gene transcription, promoting TJP expression and facilitating BBB structural restoration. Exogenous addition of Wnt proteins shows significant therapeutic effects but demands multifaceted considerations to ensure the safety of Wnt administration. VEGF signaling-mediated BBB repair therapies involve regulating VEGF levels at different stages to enhance brain self-repair capabilities, promoting BBB integrity restoration. However, the timing of treatment administration poses limitations. The advantages and limitations of the BBB repair methods are shown in Table 1.
Characteristics of BBB Repair Methods.
Additionally, certain physical techniques are beneficial for post-stroke BBB integrity and brain function recovery, such as optogenetics. Optogenetics is a technology used to manipulate cell activity with high specificity and temporal precision, playing a breakthrough role in neuroscience research. 135 Previous studies have shown that optogenetic stimulation can increase levels of brain-derived neurotrophic factors, nerve growth factors, and neurotrophin-3 in stroke models, promoting brain function recovery. 136 Recent research indicates that optogenetically activated astrocyte-derived IL-10 can reduce matrix metalloproteinase-2 activity and facilitate TJ formation, favoring BBB integrity. 137 Targeting appropriate methods to repair BBB damage at different stages post-stroke is crucial for disease management.
Angiogenesis
The harsh microenvironment after stroke causes the loss of normal structure and function of the BBB. Thereafter, exogenous harmful substances as well as toxic factors secreted by cells and tissues in the brain harm neural cells and contribute to their apoptosis. Excessive nerve cell death tends to trigger poorer neurological outcomes, producing irreversible damage to the CNS. The neurovascular unit is composed of ECs, pericytes, BM, astrocytes, microglia, and neurons. 138 Among them, the close connection between vascular tissues and neurons is essential for the maintenance of neuronal function in health and the protection and regeneration of neurons after stroke.139,140 Therefore, in the pathological state after stroke, vascular repair, and angiogenesis in the infarcted area of the brain play important roles in the recovery of neurological function and the repair of damaged tissues.
Angiogenesis refers to the growth of new vascular structures from existing vessels, a process that involves the proliferation and migration of ECs, vascular budding, lumen formation, and BBB maturation. 141 Angiogenesis increases the density of microvessels in the infarcted area of the brain and enhances blood microcirculation. Neovascularization uses the selective substance exchange of the BBB to deliver nutrients and energy to the damaged brain parenchyma and to impede neurotoxic substances to brain tissue, allowing for more efficient neurological recovery after stroke. Post-stroke angiogenesis can effectively reduce infarct size and promote neuronal survival 140 (Figure 6). Many approaches have been found to promote angiogenesis after stroke. In these regards, three types of methods with pro-angiogenic effects in the last seven years have been summarized to assist in subsequent studies related to pro-angiogenesis after stroke.

(a) BM degradation, the onset of angiogenesis, (b) EC proliferation, vascular budding, (c) EC proliferation, migration, and pericytes coverage, and (d) lumen formation, BBB maturation.
Reperfusion greatly reduces the obstruction of blood flow by the thrombus, but due to the severe damage to part of the vessels, it is more difficult for blood to enter the vessels, so this part of the vessels is in a prolonged state of ischemia and hypoxia, resulting in apoptosis of perivascular neuronal cells. And, some of the blood vessels are not able to deliver enough nutrients and energy to the damaged cells even when blood flow is restored. As a result, neoangiogenesis occurs in vessels without reflux as well as in some vessels with severe injury. New blood vessels provide additional nutrients and energy to the damaged cells and are essential for the recovery of brain function.
Chemical drug therapies
Studies on pro-angiogenic drugs after ischemic stroke have shown that some herbs and herbal extracts are effective in promoting angiogenesis in the area of cerebral ischemia and improving neurological function in the deficit. Research has shown that Buyang Huanwu decoction (BYHWD) increases microvascular density in ischemic brain areas, upregulates the expression levels of connexin 43 (Cx43), VEGF, and Ang-1, and has significant proangiogenic effects. The positive effects shown by BYHWD treatment are mediated by the upregulation of VEGF and Ang-1 expression by Cx43. 142 Naoluo Xintong decoction (NLXTD) treatment increases the number of EC and the expression of HIF-1α, VEGF, and VEGFR2 in the ischemic penumbra region, and its pro-angiogenic effects are closely related to the upregulation of the HIF-1α/VEGF signaling pathway. 143 Ginseng Yangrong decoction (GSYRD) was proved to promote the proliferation and migration of human umbilical vein EC in a dose-dependent manner. 144 New researches show that GSYRD can activate HIF/VEGF/Notch signaling pathway through miR-210 to achieve enhanced angiogenesis after cerebral ischemia. 145
Paeoniflorin, the active component of Paeoniae Radix, promotes the proliferation, and migration of bone marrow-derived EPC and tube formation in vitro and increases the secretion of pro-angiogenic factors and cerebral vascular density in vivo. 146 Ginsenoside Rg1, which is completely encapsulated by nanoparticles, and notoginsenoside R1, a unique component of Panax notoginseng, can promote the proliferation, migration of HBMEC and tube formation during the treatment of cerebral ischemia.147,148 Among them, notoginsenoside R1 shows pro-angiogenic effects partly dependent on the activation of the NAMPT-SIRT1 cascade, 148 while the therapeutic effects of ginsenoside Rg1 nanoparticles are achieved by inducing the demethylation of H3K27me3 within the promoter region of VEGF-A and Jagged1 genes. 147 In the regulation of angiogenesis in the treatment of cerebral ischemia, the mechanism of action of Astragaloside IV lies in the HIF/VEGF/Notch signaling pathway activated by miRNA-210. 149 The pro-angiogenic process of Shengui Sansheng San (SSS) extract is associated with Akt/mTOR and ERK1/2 signaling pathways. 150 The molecular mechanism of Skullcapflavone II, an extract of Scutellaria baicalensis, lies in the inhibition of NF-ĸB. 151 The positive effects of berberine are mediated by AMPK signaling that regulates microglia polarization to the M2 type. 152 In contrast, chlorogenic acid exerts its pro-angiogenic effects by regulating the PI3K/Akt signaling pathway. 153 The danshen extract danshensu has been shown to increase new arteries as well as restore cerebral blood flow in ischemic areas in the treatment of ischemic stroke. 154 In addition, 2,3,5,6-tetramethylpyrazine, the active ingredient of Ligusticum chuanxiong Hort, also has the effects of promoting angiogenesis after stroke. 155
Some plant extracts and clinical drugs can also act to promote angiogenesis at the site of brain injury during post-stroke treatment. The active compound DL-3-N-butylphthalide (NBP) in celery seeds has been shown to have pro-angiogenic capacity after stroke.156,157 The use of cerium oxide nanoparticles loaded with NBP also has pro-angiogenic effects after cerebral ischemia. 158 The flavonoid compound Morin isolated from mulberry plants promotes angiogenesis by enhancing the expression of angiogenic proteins such as Ang-1, Tie-2, Wnt3α, and β-catenin. 159 Nicotine promotes angiogenesis in the area of the hematoma without triggering a deterioration in vascular permeability caused by ICH. 160 In addition, zinc can promote angiogenesis during recovery from cerebral ischemia by facilitating the astrocyte-mediated HIF-1α/VEGF signaling pathway. 161 In contrast, Fraxetin may promote angiogenesis after ischemic stroke through activation of the PI3K/Akt pathway. 162 Xiaoshuan enteric-coated capsule (XSEC) is effective in the treatment of ischemic cerebrovascular diseases, and the combination therapy of the enriched environment (EE) with it significantly increases the expression levels of Netrin-1 and Robo-1, and proximally promotes angiogenesis. 163 Fingolimod intraperitoneal injection can increase the number of microvessels in the brain injury site of diabetic stroke mice. However, the aggravation of brain edema caused by fingolimod treatment and the low-level expression of ZO-1 and occludin proteins makes it have certain requirements in the treatment selection of brain injury severity in patients with acute ischemic stroke. 164 In addition, studies have proved that atorvastatin not only contributes to angiogenesis, but also matures the BBB in neovascularization by promoting the formation of TJ. These positive effects are associated with the PI3K/Akt pathway, the TGF-β/Alk1/5 pathway, and the pericyte-mediated endothelial TJ formation. 165
Biotherapies
Cell therapies, protein therapies, and gene therapies have all shown beneficial effects in promoting angiogenesis after stroke. Cell therapies increase the number of beneficial cells in the brain injury sites by transplanting beneficial cells to these sites, achieving proangiogenic effects after stroke. Studies found that human amniotic fluid stem cells play a therapeutic role in ischemic stroke by promoting angiogenesis in the ischemic region of the brain and reducing the inflammatory response in this region. 166 Human bone marrow-derived MSC (BMSC) can improve neurological behavior after stroke, enhance angiogenesis, and promote brain function recovery. 167 The combined treatment of tetramethylpyrazine and BMSC can improve the suboptimal homing of BMSC with monotherapy and promote angiogenesis in the area of cerebral ischemia to enhance tissue repair and function recovery after ischemic stroke. 168 Transplantation of the oligodendrocyte precursor cell (OPC) promotes angiogenesis in the ischemically injured brain via the Wnt/β-catenin signaling pathway. 169
Some proteins such as cytokines, bacterial toxins, and bioactive enzymes also exhibit promoting effects on neovascularization in the brain injury sites after stroke. Adiponectin and Ang-2 have been shown to promote angiogenesis after cerebral ischemia.170,171 Overexpression of α5 and Ang-1 synergistically promotes angiogenesis in the early stages of ischemic stroke and improves vascular structural integrity. 172 Mesencephalic astrocyte-derived neurotrophic factor (MANF) increases the total vascular surface area and the number of microvascular branch points, and these pro-angiogenic effects may be related to the activation of VEGF pathway. 173 In addition, the delivery of VEGF in the brain using gelatin methacryloyl (GelMA) microneedle (MN) can improve the needle injury and uneven VEGF distribution caused by conventional delivery methods, and show high VEGF expression and enhanced angiogenesis in the brain injury site. This method doesn’t cause obvious inflammatory reactions and has good biocompatibility. 174 Ras-associated C3 botulinum toxin substrate 1 (Rac1) in EC enhances angiogenesis in ischemic area through PAK1 signaling. 175 In addition, delayed treatment of pyruvate kinase M2 (PKM2) can restore cerebral blood flow in the infarcted area to a greater extent and promote angiogenesis in this area, which involves the activation of STAT3 and FAK signals. 176
Gene therapies show effective pro-angiogenic effects after ischemic stroke, in which miRNA have been widely studied. Different miRNAs can enhance angiogenesis after cerebral ischemia by regulating different signaling pathways. Studies have shown that overexpression of miR-126 in EPC and EPC transplantation can promote neoangiogenesis at the site of cerebral ischemia. 177 MiR-26a exerts pro-angiogenic effects through PI3K/Akt and MAPK/ERK signaling pathways, 178 while the pro-angiogenic effects of miR-181b are mediated by activation of the PTEN/Akt signaling pathway. 179 MiR-15a/16-1, miR-103, and miR-377 in EC inversely regulate angiogenesis at the site of brain injury after ischemic stroke.180 –182 ADAMTS9-AS2 is a long noncoding RNA whose overexpression enhances angiogenesis at the site of cerebral ischemia, and the effects may be associated with the regulation of the miR-185-5p/IGFBP-2 axis. 183 FAM3A in EC improves blood perfusion at the site of cerebral ischemic injury and increases capillary density by activating VEGF-A transcription, showing the ability to promote angiogenesis. 184
Physical therapies
Physical means have also shown the effects of promoting angiogenesis, reducing the infarcted area of the brain and restoring the defective neurological function in the treatment of ischemic stroke. Among them, electroacupuncture (EA) therapy acting on different parts of diseased mice can activate different pathways to promote angiogenesis. Research has shown that applying EA at GV26 and Yin meridian acupoints can effectively promote angiogenesis. The mechanism of the former is to activate the Src and VEGF signaling pathways mediated by EPO, 185 while the latter may be related to the increased expression of VEGF, P-Akt, and p-eNOS. 186 In addition, applying EA at GV20 can activate the expression of VEGF mediated by Wnt/β-catenin signaling pathway, thereby promoting angiogenesis and restoring blood perfusion in ischemic areas. 187 The miR-210 extracted from the serum of diseased mice treated with EA (EA-EXO) can increase the levels of HIF-1α, VEGF, and Notch1 proteins in rats with cerebral ischemia and promote angiogenesis. 188
Low-intensity pulsed ultrasound treatment significantly increases the number of CD31-positive blood vessels in the cerebral ischemic area. 189 Repetitive transcranial magnetic stimulation (RTMS) promotes angiogenesis after photothrombotic stroke and reduces apoptosis of EC in new and existing vessels, and these beneficial results are partly attributed to the enhancement of signaling of HIF-1α and the polarization shift of astrocyte associated with blood vessels. 83 Encephalomyosynangiosis has the beneficial effects of improving angiogenesis and vascular recovery after ischemic stroke. 190 Physiological ischemia training can promote angiogenesis in the rehabilitation process after ischemic stroke. 191 While rehabilitation exercise in the early stage of ischemic stroke can upregulate the expression of VEGF and laminin, thereby increasing brain microvessel density and positively regulating angiogenesis. 192 The enriched environment promotes angiogenesis after ischemic stroke by increasing the expression of IL-17A in astrocytes. 193
Multiple therapies have shown significant pro-angiogenic effects in the treatment of stroke, as evidenced by upregulation of pro-angiogenic factors, increase in vessel density, reduction in infarct size, and restoration of cerebral blood flow and neurological function. Different treatments usually mediate post-stroke angiogenesis through different pathways (Table 2), such as HIF-1α/VEGF, PI3K/Akt, MAPK, and ERK signaling pathways (Figure 7). VEGF plays roles in promoting EC proliferation, migration and survival in the process of neoangiogenesis, which involves several “destructive” behaviors such as degradation of the BM.194,195 Above a certain level, VEGF expression causes excessive degradation of the BBB in existing vessels and in neovascularization, exacerbating stroke-induced BBB damage and preventing neovascularization. Therefore, VEGF expression needs to be appropriately adjusted during post-stroke vascular remodeling to avoid VEGF overexpression being detrimental to stroke recovery. Recent studies have found that Sema3E-Plexin-D1 signaling may downregulate VEGF expression during the critical period of vascular remodeling to improve uneventful vascular repair as well as vascular maturation in the late stages of stroke. 196 Although VEGF can effectively promote angiogenesis in ischemic areas, overexpression of VEGF tends to increase the risk of bleeding in immature vessels. Therefore, it is meaningful to find an alternative strategy that can not only promote angiogenesis, but also discard the adverse results caused by VEGF. In these regards, studies have indicated that anti-Nogo-A antibodies have a strong pro-angiogenic capacity and do not increase vascular permeability due to cerebral ischemia. 197 The creation and maturation of new blood vessels facilitate blood microcirculation to the stroke brain, allowing blood to exchange material and energy with the brain parenchyma in the infarcted area via the BBB to rescue damaged nerve cells and normalize neurological function. During the treatment of ischemic stroke, newly generated vessels show excessive permeability due to the lack of TJP. Therefore, promoting the formation of TJ in new vessels and the maturation of new vessels for rapid angiogenesis is essential for the establishment of BBB integrity.
Molecular Mechanisms Associated With Pro-Angiogenic Approaches.


Part of the VEGF-mediated pro-angiogenic signaling pathways. ERK signaling pathway participates in cell proliferation, PI3K/Akt signaling pathway supports cell survival, and MAPK signaling pathway mediates cell migration.
In addition, some methods for promoting angiogenesis have significant effects, although not targeted at BBB, but still have certain reference significance. Gao et al. 198 investigated the ability of induced pluripotent stem cells (iPSCs) generated microvasculature (iMV) to promote vascular regeneration and tissue repair through in vivo transplantation. Research has found that vascular cells derived from iMV can directly promote vascular reconstruction and immune microenvironment regulation, thereby promoting tissue repair in kidney and skin injury models. Moreover, intact iMV can form a large number of perfusion vessels and promote early muscle regeneration. Therefore, using appropriate substances to induce iPSC, inducing them to differentiate into brain microvascular ECs and pericytes. In vivo transplantation of cerebrovascular constituent cells or generated cerebral microvessels may be beneficial in promoting post-stroke neoangiogenesis. The safety of intracerebral transplantation of iPSC-formed cerebrovascular tissue needs to be considered. Hong et al. 199 discovered that injection of the self-assembled peptide RADA16 was able to reduce inflammatory cells in the lesion lumen and increase the number of blood vessels around the damaged spinal cord, and that the effects were more pronounced in the case of RADA16 containing the neuropeptide substance P. Exploring the proangiogenic effects of neuropeptide substance P-modified RADA16 after stroke or other CNS diseases could be a promising avenue for study. Moreover, understanding the correlation between injury severity, drug dosage, and the method of drug delivery is crucial in this context.
Stroke induces BBB leakage, and reperfusion exacerbates BBB damage, leading to disease progression. Therefore, post-stroke management requires timely repair of BBB, employing appropriate treatment methods according to the extent of BBB injury. Post-reperfusion therapy necessitates BBB repair following damage. Patients who miss reperfusion therapy often exhibit severe BBB damage. Apart from anticoagulation, neuroprotection, and BBB protective treatments, adjusting the dosage of BBB repair drugs might be necessary. Among the treatments with BBB repair potential, cell transplantation (particularly MSC transplantation) and Wnt/β-catenin-mediated BBB repair therapy (specifically drug delivery of Wnt7aK190A) demonstrate relatively safe and effective therapeutic outcomes. MSC transplantation has undergone clinical trials, offering hope for extending the time window for thrombolytic therapy in clinical settings. However, currently, no drugs or treatments significantly restore BBB and substantially improve BBB dysfunction post-stroke. Moreover, there are currently no drugs in clinical practice that can significantly repair BBB. Research must intensify to explore safe and notably effective novel methods for BBB repair post-stroke. Furthermore, regulating neovascularization in the stroke-affected region based on the extent of BBB and brain functional damage in stroke patients is crucial. During stroke recovery, BBB repair treatment not only restores damaged BBB but also promotes BBB maturation in newly formed blood vessels. When repairing BBB tissue damaged after stroke, adjunctive BBB-protective therapies to reduce further damage to the BBB by inflammatory mediators are largely beneficial to BBB repair therapy.
The BBB repair and neovascularization therapies presented in the article commonly employ tMCAO rat models. Although the animal and human BBB structural components are similar, molecular differences persist. 200 Establishing human-like organ models of the BBB holds crucial significance in fully understanding stroke pathology and investigating new post-stroke treatment methods. Lancaster et al. 201 cultured three-dimensional brain organoids from human iPSC, while Pham et al. 202 further vascularized brain organoid tissues using patient-derived iPSC-derived ECs. Recently, a three-dimensional BBB sphere model composed of human brain ECs, pericytes, astrocytes, microglia, oligodendrocyte progenitor cells, and neurons has been reported. This model demonstrated expression of TJ, adhesion junctions, related proteins, and cell-specific markers, exhibiting functional responses to hypoxia and neurotoxicity. 203 Further research into human-like organ models of the BBB is imperative. The development of an in vitro model that closely resembles the structure and function of the human BBB not only contributes to understanding stroke pathology and exploring new treatments for post-stroke injuries but also propels pathological research and the management of other CNS diseases. This advancement holds immense potential for clinical translation in therapies targeting CNS diseases. Moreover, the article predominantly features BBB repair studies using in vivo tMCAO, permanent MCAO, ICH, and in vitro BBB models subjected to OGD/R. Most studies typically conduct both in vivo and in vitro tests. Different stroke models target distinct pathological processes; hence, treatment methods applicable to various stroke types may not be effective for all. However, they offer valuable insights for one another.
Conclusion and perspective
The selective permeability of the BBB allows for the exchange of substances and energy between the brain parenchyma and circulating blood to maintain normal brain function. Stroke disrupts the BBB’s structure, leading to a loss of its selective permeability. This breach allows harmful substances from the bloodstream to infiltrate the brain parenchyma, adversely affecting nerve cells. Effective repair of damaged BBB tissue after stroke is of great significance to the recovery of brain function. In recent years, studies have found many new methods to show the roles of BBB repair, mainly manifested in the enhanced expression of TJP and the decreased permeability of BBB in vivo, as well as the recovery of injured EC and the proliferation of EC in vitro. In the studies of BBB tissue repair, the increased expression of TJP in EC indicates that EC has been effectively repaired. However, there are relatively few studies on the effective repair of damaged pericytes, astrocytes, and BM after stroke treatment. Strengthening research on the repair of other BBB tissues, such as morphological recovery of astrocyte endfeet post-stroke, remains imperative.
Neoangiogenesis after stroke increases the blood microcirculation at the site of cerebral ischemic injury, reduces the infarct size, and rescues nerve cells facing apoptosis. The remarkable effects of the newly formed blood vessels in restoring brain function are due to the ability of BBB in the new vessels to transfer substances and energy and impede the attack of toxic substances. Neoangiogenesis transiently increases existing BBB permeability, emphasizing the need to appropriately bolster BBB repair during this process. In addition, accelerating neoangiogenesis has beneficial effects on the recovery of brain function after stroke.
Several therapies have shown promise in effectively repairing the damaged BBB after stroke and promoting angiogenesis at the site of brain injury. However, their clinical translation outcomes haven’t met ideal standards, possibly due to various factors. The limitations of preclinical stroke models are an important reason for the failure of clinical translation of treatment methods. Studies on BBB repair and neoangiogenesis after ischemic stroke mostly use MCAO animal models, but this does not cover all types of ischemic stroke in clinic. Moreover, the general lack of microglia and leukocytes in in vitro models will largely affect the results of drug treatment. In addition, some stroke patients also have diabetes and hypertension, and there are relatively few studies on the treatment of comorbidities after stroke. Future researches into effective and safe BBB repair therapies and pro-angiogenic therapies in multiple stroke models still need to be strengthened. In addition, when delivering drugs for stroke, it is necessary to choose the appropriate dosage, delivery method, and delivery timing based on the disease development period, degree of brain injury, and comorbidity type, in order to better treat the disease. Reasonable treatment plans for stroke still need to be further improved through continuous scientific research to obtain theoretical support.
Footnotes
Abbreviations
blood-brain barrier (BBB); central nervous system (CNS); endothelial cells (ECs); basement membrane (BM); tight junction proteins (TJPs); extracellular matrix (ECM); neurovascular unit (NVU); Alzheimer’s disease (AD); intracerebral hemorrhage (ICH); tight junction (TJ); vascular endothelial growth factor (VEGF); angiopoietin-2 (Ang-2); matrix metalloproteinases (MMPs); tissue plasminogen activator (tPA); mesenchymal stem cells (MSCs); endothelial progenitor cells (EPCs); neural stem cells (NSCs); middle cerebral artery occlusion (MCAO); microRNA-126a-3p (miR-126); microRNA (miRNA/miR); matrix metalloproteinase-9 (MMP-9); human bone marrow endothelial progenitor cells (HBMEPCs); transient middle cerebral artery occlusion (tMCAO); EPC-secretome from stroke patient (scCM); oxygen-glucose deprivation (OGD); outgrowth endothelial cells (OECs); human bone marrow endothelial cells (HBMECs); gelatin loaded with VEGF and hyaluronic acid is assembled layer by layer to modify NSC (LbL (VEGF)-NSC); pericyte-like cells with a cranial neural crest origin in human pluripotent stem cells (hPSC-CNC PCs); angiopoietin-1 (Ang-1); tunneling nanotubes (TNT); ischemia-reperfusion (I/R); oxygen-glucose deprivation/reoxygenation (OGD/R); fatty acid synthase (FASN); tissue inhibitor of metalloproteinases 2 (TIMP2); polymorphonuclear neutrophils (PMNs); glutathione (GSH); pigment epithelium-derived factor (PEDF); Gpr124/Reck-specific agonist (Wnt7aK190A); 6-bromoindirubin-3′-oxime (6-BIO); recombinant tissue plasminogen activator (rtPA); low molecular mass peptide 2 (LMP2); Buyang Huanwu decoction (BYHWD); connexin 43 (Cx43); Naoluo Xintong decoction (NLXTD); Ginseng Yangrong decoction (GSYRD); Shengui Sansheng San (SSS); DL-3-N-butylphthalide (NBP); xiaoshuan enteric-coated capsule (XSEC); enriched environment (EE); bone marrow MSC (BMSC); oligodendrocyte precursor cell (OPC); mesencephalic astrocyte-derived neurotrophic factor (MANF); gelatin methacryloyl (GelMA); microneedle (MN); Ras-associated C3 botulinum toxin substrate 1 (Rac1); pyruvate kinase M2 (PKM2); electroacupuncture (EA); miR-210 extracted from the serum of diseased mice treated with EA (EA-EXO); repetitive transcranial magnetic stimulation (RTMS); induced pluripotent stem cells (iPSCs); iPSCs generated microvasculature (iMV)
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Joint Fund of the National Natural Science Foundation of China (U2004164), Key Scientific and Technological Research Projects in Henan Province (grant number 232102311155 and 232102230106), Zhengzhou University College Student Innovation and Entrepreneurship Training Program (13210022), Zhengzhou University Major Project Cultivation Special Project (125-32214076), and Natural Science Foundation of the Jiangsu Higher Education Institutions of China (grant number 21KJB430013).
