Abstract
The repair of growth plate injuries is a highly complex process that involves precise spatiotemporal regulation of multiple cell types. While significant progress has been made in understanding the pathological mechanisms underlying growth plate injuries, effectively regulating this process to regenerate the injured growth plate cartilage remains a challenge. Tissue engineering technology has emerged as a promising therapeutic approach for achieving tissue regeneration through the use of functional biological materials, seed cells and biological factors, and it is now widely applied to the regeneration of bone and cartilage. However, due to the unique structure and function of growth plate cartilage, distinct strategies are required for effective regeneration. Thus, this review provides an overview of current research on the application of tissue engineering to promote growth plate regeneration. It aims to elucidates the underlying mechanisms by which tissue engineering promotes growth plate regeneration and to provide novel insights and therapeutic strategies for future research on the regeneration of growth plate.
Introduction
The growth plate, or physis, located at the end of immature long bones in children, plays a crucial role in regulating longitudinal bone growth. 1 The growth plate is divided into three compartments based on the function and morphology of the chondrocytes: the resting zone, the proliferative zone, and the hypertrophic zone. 2 Hormones regulate the relatively inactive chondroprogenitor cells (CPCs) in the resting zone, which then differentiate into chondrocytes and enter the proliferative zone. The chondrocytes in the growth plate are arranged in a regular column and secrete extracellular matrix (ECM) to maintain the stability of each differentiation stage.3,4 In the hypertrophic zone, the chondrocytes stop proliferation, increase in size, and overproduce type X collagen, while ossification of the ECM also occurs. 2 Due to its delicate nature, the growth plate is vulnerable to injury caused by fracture, infection, tumor, or iatrogenic causes, which can result in imbalanced bone growth.5 –7 In addition, the limited regenerative capacity of growth plate cartilage and the dynamic nature of immature bone make it vulnerable to bony tissue replacement, which will cause the formation of a “bone bridge.” This development leads to growth arrest and angular deformities, negatively affecting joint function and a child’s physical and mental health. 8
The conventional treatment is to resect the bone bridge formed after growth plate injuries, and then fill the defect with conventional implants such as bone cement, adipose tissue, polymer materials, etc., in order to limit the bone bridge formation. However, this approach focus on prevention methods and are unable to regenerate the injured growth plate.5 –7 Thus, it has a low success rate and may require additional surgeries, such as osteotomy or limb lengthening. Unfortunately, the unpredictability of surgical prognosis and the unsuitability of filling materials often results in bone bridge recurrence. Thus, there is an urgent need for a treatment strategy to promote the regeneration of growth plate cartilage. In recent years, tissue engineering has emerged as a potential treatment for growth plate injury. Tissue engineering create functional biomaterials by combining scaffolds, seed cells, and biological factors. Once implanted into growth plate defects, these biomaterials not only prevent the bone bridge formation but also promote the regenerative potential of growth plate cartilage. Therefore, the advanced tissue engineering technology holds significant importance for the successful treatment of growth plate injuries.
Despite this potential, current research on tissue engineering is mainly focused on defects in the bone or articular cartilage defects, with limited studies on growth plate cartilage. Due to the unique structure of growth plate cartilage, which determines its specific functions, this review aims to explore and analyze the important role of tissue engineering in growth plate regeneration and provides a systematic summary to generate new therapeutic ideas and design strategies for growth plate regeneration (Figure 1).
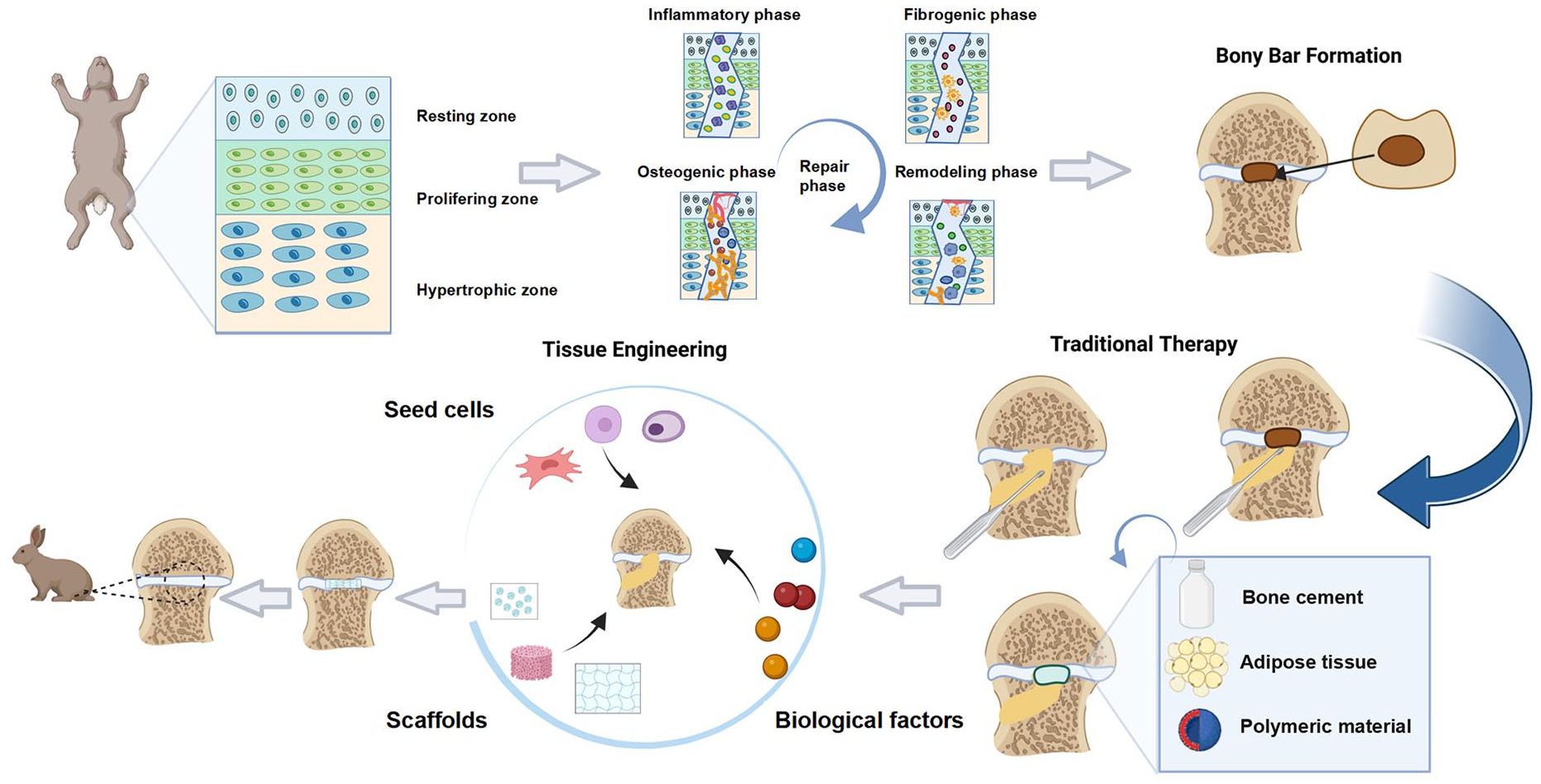
The overview of growth plate cartilage regeneration strategy. The pathological mechanisms and conventional treatment methods following growth plate injuries are presented, and the significant role of tissue engineering in the growth plates regeneration is revealed.
Pathological process of growth plate injury
Early intervention to inhibit bone bridge formation can effectively prevent the development of growth arrest and angular deformities. Furthermore, gaining a comprehensive understanding of the pathological processes involved in growth plate injuries can provide valuable insights for the exploration of novel therapeutic approaches. Animal models have been used to elucidate the four critical phases of the pathophysiology involved in growth plate injury: inflammatory, fibrogenic, osteogenic, and remodeling 9 (Figure 2).

The process of bone bridge formation after growth plate injury and related events. After the growth plate injury, cells and biological factors change dynamically with the progress of pathological process, and eventually lead to the bone bridge formation.
Inflammatory phase
The inflammatory phase is initiated within 3 days following growth plate injury. Inflammatory cells, mainly neutrophils, along with macrophages/monocytes and lymphocytes, infiltrate the injury site within approximately 8 h post-injury. 9 Chung et al. have demonstrated that neutrophils play a critical role in regulating downstream chondrogenic and osteogenic events during growth plate repair. Specifically, they have been shown to inhibit the osteogenic differentiation of mesenchymal stem cells (MSCs) while promoting chondrogenic differentiation, thus influencing bony repair events during the repair of growth plate injury. 10
During the inflammatory phase, the growth plate is influenced mainly by two stimuli: upregulated pro-inflammatory cytokines and the mechanical load generated by micro-movements at the injury site. Inflammatory cells, including macrophages and some neutrophils, infiltrate the injury site and produce high levels of pro-inflammatory cytokines such as interleukin-1 beta (IL-1β), interleukin-6 (IL-6), and tumor necrosis factor-alpha (TNF-α). During the initial inflammatory phase, IL-1β expression peaks at 8–16 h, while TNF-α, IL-6, and TGF-β1 levels peak on day 1.9,11 A study has shown that TNF-α and IL-1β inhibit aggrecan and collagen type II alpha 1 (Col2α1) expression, which impedes growth plate chondrogenesis. 12 Furthermore, the combined treatment of TNF-α and IL-1β can significantly reduce chondrocyte proliferation. This may be due to the fact that TNF-α and IL-1β induce the phosphorylation and nucleocytoplasmic shuttling of Forkhead box O3 (FOXO3) in chondrocytes, which in turn reduces the expression level of FOXO3 in the nucleus. FOXO3 is a key transcription factor involved in the metabolic disorder and hypertrophy of chondrocytes induced by pro-inflammatory cytokines. 13 Hence, the dysregulation of chondrocytes and the resulting cartilage injury induced by pro-inflammatory cytokines have emerged as a promising area for therapeutic intervention. Notably, targeting FOXO3 shows considerable potential in mitigating the detrimental effects caused by pro-inflammatory cytokines.
P38 mitogen-activated protein kinase is a crucial regulator of bone formation 14 and inflammatory response, 15 which becomes activated in growth plate injury. Zhou et al. found that TNF-α induces p38 activation in the injured growth plate during the initial inflammatory response, promoting the recruitment and proliferation of bone marrow mesenchymal stem cells (BMSCs) in vivo and in vitro, which is essential for growth plate regeneration. TNF-α also suppresses bone formation by negatively regulating gene expression of osteogenic differentiation transcription factor core-binding factor-alpha 1 (cbfa1) and osteocalcin (OCN), indicating a significant impact on the repair of growth plate injury. 15 Thus, regulating the TNF-α-p38 signaling pathway holds the potential to enhance the efficacy of repairing growth plate injuries.
In the rat tibial growth plate injury model, Arasapam et al. subsequently identified two other inflammatory factors, cyclo-oxygenase-2 (COX-2) and inducible nitric oxide synthase (iNOS). These factors may enhance MSCs differentiation to chondrocytes and promote bone remodeling during bony repair of growth plate injury sites. Although inhibiting COX-2 or iNOS did not prevent downstream bony tissue repair at the injured growth plate, it effectively delayed the differentiation of MSCs into chondrocytes and the bone remodeling process. Therefore, regulating these two inflammatory factors plays a vital role in the repair of growth plate injury. 16
Additionally, mechanical loading generated by micromovement at the injury site and pro-inflammatory cytokines have opposing effects during the inflammatory phase. The frequency and type of mechanical stimuli can influence the metabolism of growth plate chondrocytes. 17 Studies have shown that IL-1β levels are significantly elevated which can cause the chondrocyte damage by promoting the degradation of extracellular matrix or by increasing oxidative stress. 18 Pichler et al. found that IL-1β promotes the proliferation of CPCs at the growth plate injury site, while mechanical loading inhibits CPCs proliferation. In vitro, the inflammatory environment during the early stage of growth plate injury suppresses osteogenesis and chondrogenesis of CPCs and significantly promotes their hypertrophy. Hypertrophy of growth plate CPCs may represent an early event of growth arrest and bone bridge formation. Therefore, reducing the impact of local pro-inflammatory cytokines on growth plate regeneration is a valuable targeted therapeutic strategy. However, considering the positive role of IL-1β discovered in this study, future research should also explore the interplay of different pro-inflammatory cytokines in the regeneration of growth plate cartilage. Additionally, efforts should be made to guide the direction of regeneration by modulating the balance between positive and negative factors.
Fibrogenic phase
The fibrogenic phase occurs 3–7 days after a growth plate injury, during which fibrous vimentin-positive mesenchymal cells, including potential MSCs, osteoprogenitor cells (OPCs), and CPCs, gather at the injury site and can differentiate into osteoblasts and chondrocytes. 19 Additionally, two growth factors, fibroblast growth factor-2 (FGF-2) and platelet-derived growth factor (PDGF), are significantly upregulated during this phase. Both have previously been shown to affect the migration and chemotaxis of various cells, including MSCs, 20 and may be involved in the recruitment of MSCs to the growth plate injury site. 9 Inhibition of the PDGF receptor results in a significant reduction of MSC infiltration and delayed healing at the growth plate injury site, indicating that PDGF may participate in the early fibrogenic reaction of growth plate injury by promoting the recruitment of MSCs. Moreover, PDGF may play a role in subsequent bone remodeling by regulating osteoclast formation or recruitment. Inhibition of PDGF receptors leads to a delay in cartilage formation and bony repair of growth plates, suggesting that PDGF may regulate MSC infiltration, proliferation, and bone or cartilage formation during the repair of growth plate injury. 21 During the fibrogenic phase, FGF-2 expression levels also significantly increase. FGF-2 stimulates the proliferation and migration of OPCs and MSCs while inhibiting chondrocyte differentiation. 22 Thus, the effects of PDGF and FGF-2 during the fibrogenic phase impact the subsequent repair process, including the osteogenic and remodeling phases.
Osteogenic phase
Approximately 7 days after a growth plate injury, the osteogenic phase is initiated with the appearance of bone trabecule and the start of bone bridge formation. During this phase, FGF-2 is expressed by MSCs and osteoblasts of newly formed bone trabecule. In addition, the level of TNF-α increases significantly on day 7 post-injury, with a large number of TNF-α-positive osteoblasts observed at the injury site. This upregulation of TNF-α during the osteogenic phase may play a crucial role in bone bridge maturation and remodeling. 9 At the same time, the levels of osteoprotegerin (OPG) and IL-6 significantly increase at the injured site. The increased OPG level suggests inhibition of active osteoclast formation, indicating that bone formation occurs early during the growth plate injury. 19 The increased expression of bone matrix proteins, such as collagen type II (Col II), collagen type X (Col X), and chondrogenic transcription factor SOX-9, suggests the involvement of chondrogenesis and endochondral osteogenesis mechanisms in the bony repair of the growth plate injury site. 16 Moreover, the increased expressions of OCN and cbfα1 indicate osteogenic differentiation and bone formation at the growth plate injury site. During the initial osteogenesis and up to 30 days post-injury, chondrocyte apoptosis was observed in adjacent areas of the growth plate injury site, close to the forming bone bridge, and in cartilage lacunae with cellular debris present, as evidenced by increased caspase-3 expression. 11 During this period, bone bridge formation not only occurred at the injured site but also involved the uninjured site.
The osteogenic phase is characterized by angiogenesis, with vascular endothelial growth factor (VEGF) playing a crucial role in regulating angiogenesis and endothelial cell growth. 23 Fischerauer et al. observed that both the hematoma and the subsequent bone trabecule formed after growth plate injury expressed VEGF and VEGF receptor-2, with VEGF expression peaking at the initial osteogenic stage. Bone bridge formation was associated with angiogenesis, which was shown to precede bone bridge formation. And the residual VEGF-positive (VEGF+) bone debris at the injury site will further stimulate bone bridge formation. 24 Inhibition of VEGF expression in the growth plate injury site led to decreased blood-vessel-like structures and vascular invasion of the adjacent uninjured site. There was also a decrease in bony repair and an increase in the proportion of MSCs at the injury site, suggesting that VEGF may lead to osteogenic differentiation of MSCs. 25 VEGF-mediated angiogenesis plays a crucial role in bone formation, endochondral ossification, and hypertrophic cartilage transformation into the bone at the growth plate injury site and also affects the differentiation tendency of MSCs. However, further research is needed to explore whether targeting VEGF suppression and clearing VEGF+ debris can promote the repair of growth plate injury. Therefore, this provides two potential strategies: using VEGF-targeting drugs or clearing VEGF+ debris to reduce VEGF expression levels, which could (1) delay vascular invasion after growth plate injury, and thereby reduce the stimulation of bone bridge formation; (2) changing the differentiation tendency of MSCs by slowing down their osteogenic differentiation, and thus reducing bone formation. However, the effectiveness of these two strategies remains to be further validated.
The Wnt/β-catenin signaling pathway is a crucial regulator of OPCs proliferation and osteoblast differentiation during fracture healing, as evidenced by various studies. 26 However, this pathway also plays a key role in growth plate development, as inactivation of β-catenin has been shown to significantly affect chondrogenesis, leading to abnormal morphology and function of growth plates. 27 In the repair of growth plate injuries, Macsai et al. observed a significant increase in β-catenin expression 4-8 days post-injury, indicating the importance of Wnt/β-catenin pathway in the fibrogenic and early osteogenic phases. 28 In addition, Chung et al. found that treatment with the β-catenin inhibitor ICG-001 decreased bone trabecule volume and promoted cartilage tissue repair at day 10 post-injury, accompanied by a decrease in osteoblast transcription factor Osterix expression, while chondrogenesis transcriptional factor SOX-9 and cartilage protein Col II mRNA expression increased. These results suggest that the Wnt/β-catenin signaling pathway is important for bone bridge formation, and its inhibition promotes cartilage regeneration after growth plate injury. In vitro studies have also shown that inhibiting the Wnt/β-catenin signaling pathway can inhibit the osteogenic potential of BMSCs and promote their chondrogenic differentiation. 29 Therefore, the Wnt/β-catenin signaling pathway is a potential target for intervention in preventing bone bridge foramtion or promoting growth plate cartilage regeneration after injury. In addition, The differentiation trend of BMSCs can be changed by regulating the Wnt/β-catenin signaling pathway, thus promoting the chondrogenesis of BMSCs, which can be a potential strategy for growth plate cartilage regeneration tissue engineering.
Remodeling phase
During the remodeling phase, which occurs from day 14 post-injury, a significant number of bone trabecule and bone marrow tissues were observed at the injury site, with minimal infiltration of residual MSCs. 30 Osteoclasts were found to be more abundant within the bone trabecule. 31 This phase was characterized by a decrease in the expression of chondrogenic genes, such as SOX-9 and Col II, and an increase in the expression of osteocalcin (OCN). 9 Moreover, the expressions of TNF-α, IGF-1, and BMP-7 increased, promoting the differentiation and recruitment of osteoclasts and subsequently bone remodeling. 32 Elevated levels of VEGF were also observed during this phase, 24 which has previously been shown to promote not only angiogenesis, but also osteoblast differentiation and osteoclast recruitment. 33 Therefore, it is possible that VEGF may participate in the process of bone remodeling by promoting osteogenesis and osteoclast recruitment.
In addition to bone bridge formation at the injury site, adjacent uninjured areas are also affected during remodeling phase. A prior microarray study showed upregulated bone morphogenetic protein (BMP) signaling during bony repair at the injury site. 28 Macsai et al. reported growth plate cartilage degeneration and enhanced bone formation in the adjacent uninjured area after growth plate injury. Furthermore, the expression of SOX-9 gene in the adjacent growth plate decreased on day 14 post-injury, 34 while the expression of chondrocyte hypertrophy related gene Col X and runt related transcription factor 2 (Runx2) increased. 35 Thus, growth plate injury could potentially affect molecular activity within the remaining adjacent growth plate structures by reducing chondrogenesis and promoting chondrocyte hypertrophy. Moreover, BMP signaling pathway activation was observed in the adjacent uninjured growth plate, and BMP-7 was significantly upregulated on day 14 and day 35. Since BMP-7 is known to possess an inhibitory effect on cartilage degeneration, 35 this finding suggests that a potential repair response was initiated by the remaining adjacent growth plate to delay the progression of cartilage degeneration induced by growth plate injury. Systemic treatment with the BMP inhibitor noggin protein has the potential to reduce bony repair at the injury site and inhibit premature chondrocyte hypertrophy, while rescuing chondrogenesis of adjacent growth plate. 30 In addition, Fischerauer et al. found that the gene expression of BMP-6 and BMPR-1a was significantly increased during the remodeling phase, when the apoptosis rate of chondrocytes was high. BMP-6 expressed by hypertrophic chondrocytes stimulates chondrogenesis and osteogenesis through BMPR-1a activation, thereby promoting post-traumatic overgrowth. 36
Additionally, Nilsson et al. reported a gradient of BMP signaling on the growth plate. In the resting zone, low levels of BMP signaling may help keep these cells in a quiescent state. In the proliferative zone, greater BMP signaling may induce the differentiation of relatively inactive chondrocytes into proliferative chondrocytes. In the hypertrophic zone, even greater BMP signaling may induce terminal differentiation to hypertrophic chondrocytes. The BMP signaling gradient identified in this study may provide a key mechanism responsible for the spatial regulation of chondrocyte proliferation and differentiation during chondrogenesis at the growth plate. 37 Therefore, regulating the BMP signaling pathway to guide the proliferation and differentiation of chondrocytes in the growth plate has potential exploratory value in the promoting the growth plate regeneration.
In addition, Su et al. proposed that osteoblast-derived neurotrophic factor-3 (NT-3) at the injury site plays an important role in promoting growth plate remodeling. NT-3 is an upstream promoter of osteogenic and angiogenic factors of BMP-2 and VEGF, capable of promoting bone formation and vascularization in the injured growth plate. 31 They found that NT-3 is highly expressed in osteoblasts at growth plate injury sites. It can induce cartilage protease to remove cartilage and augment osteoclastogenesis and resorption both directly (involving activating Erk½ and substantiating RANKL-induced increased expression of osteoclastogenic signals in differentiating osteoclasts) and indirectly (inducing osteoclastogenic signals in osteoblasts). 38 This study increases the understanding of the factors and mechanisms that control the repair of growth plate injury and remodeling. NT-3 maybe a future therapeutic target to prevent bone bridge formation and Its synergy with other factors can be further studied.
In conclusion, obtaining a comprehensive understanding of the cellular and molecular mechanisms underlying the pathological changes associated with growth plate injury holds great promise for identifying effective regenerative strategies. This knowledge is crucial for the development of tissue engineering approaches aimed at promoting growth plate cartilage regeneration. Further research in this field has the potential to significantly revolutionize the clinical management of growth plate injuries, ultimately leading to improved patient outcomes.
Growth plate regeneration treatment based on tissue engineering technology
Seed cells
The growth plate cartilage regeneration process is restricted by the microenvironment. Seed cells in tissue engineering play a crucial role in initiating and promoting tissue regeneration by proliferating, differentiating into specific cell types, secreting biological factors, and facilitating cartitissue integration with the host. Therefore, chondrocytes or MSCs are commonly employed as seed cells in tissue engineering and it aims to enhance the therapeutic efficacy of growth plate regeneration.
Chondrocytes
In some studies, allogeneic chondrocyte implantation was used to repair the growth plate injury. Foster et al. reported using cultured chondrocytes in collagen gel to repair a growth plate defect in a sheep model, which prevents bone bridge formation in minor growth plate defects (less than 20% of the total growth plate). 39 Lee et al. cultured allogeneic chondrocytes embedded in agarose instead of collagen and showed that chondrocytes in agarose could also reduce the limb discrepancy and angular deformity after a larger growth arrest (50% of the growth plate). 40 Moreover, Otsuki et al. treated partial growth plate injury by allogenic costal cartilage transplantation and showed that transplantation of costal cartilage improved angular deformities and decreased bone bridge formation. It is worth mentioning that costal cartilage graft is valuable due to its abundance in cartilage ECM, which can help prevent the formation of a new bony bridge by maintaining cartilage properties. Additionally, the graft has been shown to be long-lasting than MSCs at the injury site. As a result, the costal cartilage graft is considered a safe and effective choice for use in growth plate cartilage regeneration. 41 Lee et al. extracted cells from the resting zone of 6-week-old rabbit costal growth plates cartilage. They used the 3D chondrocyte pellet culture technique to produce tissue-engineered grafts with cytological characteristics of typical growth plates. Chondrocytes maintained as 3D pellets could retain a differentiated phenotype while synthesizing and depositing cartilage matrix. The result showed that 0.03 g (wet weight) of resting zone cartilage was required to synthesize the tissue-engineered grafts, which could restore the growth potential of the defects with a diameter of 3 mm after implantation in the lower limb growth plate defect, and no apparent immune rejection occurred for up to 7 weeks post-transplantation. Since the implanted chondrocytes can be obtained from the coastal growth plate cartilage without affecting the donor site’s growth potential and mechanical properties, it is highly feasible and less harmful to use them as seed cells to repair the defects of the lower limb growth plate. 42 In addition, Liu et al. found that chondrocytes in the proliferative zone of growth plate had a higher cell growth rate and the ability to maintain phenotype, suggesting that they may be more suitable as a cell source for growth plate or articular cartilage regeneration. 43 However, the extraction process may cause damage to the donor site.
Due to concerns about the antigenicity of allogeneic chondrocytes and the risk of infectious diseases, some experiments have utilized autologous chondrocytes to repair the growth plate injury. Park et al. extracted chondrocytes from autologous growth plate of newborn dogs, cultured them until they formed micromasses (amorphous cartilage mass), and then implanted them into the proximal growth plate defect of the dog tibia. The results demonstrated a significant reduction in the formation of the bone bridge compared to the blank control group, and the implanted amorphous cartilage mass gradually formed cartilage matrix. 44 Similarly, Tomaszewski et al. utilized a cartilage-fiber structure containing autologous growth plate cartilage to repair growth plate injury, resulting in good graft morphology and integration, without inflammatory reactions and with good recovery of the growth plate structure. 45 Another study found that implanting autologous growth plate cells into growth plate defects can prevent bone bridge formation while reducing growth arrest and angular deformity. 46 Additionally, Tobita et al. extracted autologous chondrocytes from knee joint cartilage, embedded them in atelocollagen for 1 week, and then implanted them into the growth plate defect, where the implanted chondrocytes proliferated and secreted ECM. The final results indicated that the implanted chondrocytes could prevent bone bridge formation but could not provide sufficient mechanical support to prevent severe angular deformities. 47
While the advantage of autologous chondrocyte transplantation is that the host will not have an immune response to the graft, this approach has its drawbacks. For example, chondrocytes may be cultured for a relatively long period, which may miss the optimal treatment time, and the donor site may be damaged during the cell extraction process. Lee et al. showed that the extraction of chondrocytes from the costal growth plate had no significant effect on the growth potential and mechanical properties of the donor site, making it a feasible and less harmful option to use them as seed cells to repair defects of the lower limb growth plate. 43 Therefore, the safety and efficacy of autologous chondrocyte transplantation need to be further improved and evaluated.
While autologous chondrocyte transplantation offers the advantage of avoiding immune response from the host toward the graft, it does come with certain drawbacks. One such drawback is the relatively long culture period required for chondrocyte expansion, which may result in missing the optimal treatment time. Additionally, there is a risk of donor site damage during the cell extraction process. However, Lee et al. conducted a study demonstrating that extracting chondrocytes from the costal growth plate had no significant negative impact on the growth potential and mechanical properties of the donor site. This finding suggests that utilizing chondrocytes from the costal growth plate as seed cells for growth plate cartilage regeneration is a feasible and less harmful option. Nonetheless, further efforts are needed to enhance and evaluate the safety and efficacy of autologous chondrocyte transplantation.
MSCs
MSCs are the leading choice for bone and cartilage tissue engineering, which are clonogenic progenitors obtainable from different sources, including bone marrow, synovium, adipose tissue, and umbilical cord. They display multipotency, low immunogenicity, and self-renewal ability while also possessing the potential to differentiate into diverse cell types such as chondrocytes, osteocytes, and adipocytes.48,49 Furthermore, MSC is more advantageous than chondrocyte due to its ease of availability, high capability for in vitro expansion, and multi-potential differentiation. 9 Consequently, researchers have explored the utilization of MSCs from various origins to participate in the repair and regeneration of the growth plate.
BMSCs have become one of the most extensively studied and widely used cells due to their ease of isolation and culture, 50 making them a promising candidate for growth plate cartilage regeneration. Plánka et al. showed that implantation of BMSCs prevented the progression of angular deformities and decreased growth arrest caused by growth plate injury, 51 while Li et al. demonstrated that using BMSCs-loaded scaffolds could promote chondrocyte regeneration and reduce angular deformities and limb discrepancies. 52 However, McCarty et al. found that transplanted BMSCs failed to form new cartilage structures and instead promoted dense fibrous tissue formation, 53 highlighting the need to identify factors that could influence the regenerative outcomes of BMSC implantation. One potential factor may be the source of MSCs. Plánka et al. compared the therapeutic effects of allogeneic and autologous MSCs in a New Zealand rabbit femoral growth plate injury model and found no significant difference. 51 Therefore, further research is necessary to identify other factors that may affect the efficacy of BMSC implantation for growth plate cartilage regeneration. In addition, it is important to consider the potential immunomodulatory effects of allogeneic MSCs, as they can modulate immune cell activity by altering cytokine secretion and avoid host immune responses. 54 Thus, allogeneic and autologous MSCs may have similar therapeutic effects.
Another important factor that may affect the efficacy of BMSCs is their tendency to differentiate into hypertrophic chondrocytes and undergo endochondral ossification, 55 making it challenging to maintain stable cartilage tissue. Vascular invasion is necessary for the completion of ossification. 56 BMSC-derived cartilage secretome’s soluble factors have pro-angiogenic properties, which may contribute to vascular invasion. 57 To maintain the stability of BMSC-derived cartilage and prevent osteogenic differentiation, it may be necessary to regulate the expression levels of Ihh and SerpinE1, which are involved in angiogenesis mediated by BMSCs-derived cartilage secretome. 56 The differentiation state of BMSCs can also impact their regenerative ability after implantation. For example, predifferentiated BMSCs expressed higher levels of SOX-9, Col II, and aggrecan mRNA than undifferentiated cells but failed to produce a cartilaginous matrix even with continuous TGF-β1 stimulation. In contrast, undifferentiated cells were able to produce a cartilaginous matrix and correct limb length discrepancies. 58 Therefore, when using BMSCs for growth plate cartilage regeneration, it is crucial to consider their differentiation tendency and state. Better regeneration outcomes may be achieved by controlling the differentiation state of BMSCs and regulating anti-angiogenic regulators.
Adipose-derived stem cells (ADSCs) are MSCs with multidirectional differentiation potential that are isolated from autologous adipose tissue. 59 Currently, they have become one of the most widely used types of adult stem cells in the field of tissue regeneration. ADSCs can differentiate into osteocyes and chondrocytes. Compared with BMSCs, ADSCs are abundant in tissue sources, are easy to extract, and exhibit higher proliferation. Additionally, these stem cells are immune privileged, meaning allogeneic cells could potentially be used for cell-therapy. 60 However, similar to other tissue-derived adult stem cells, the capacity of ADSCs to synthesize and secrete cartilage matrix decreases during chondrogenic differentiation in vitro. 61 levels of TNF-α and matrix metalloproteinase 3 increase during the chondrogenic differentiation of ADSCs. TNF-α bound to its receptor and activated the NF-κB pathway, decreasing cartilage ECM synthesis and secretion. And TNF-α inhibitors can eliminate this effect. 61 Therefore, treating growth plate injury with ADSCs as seed cells combined with targeted inhibition of TNF-α expression has a certain prospect. In addition, ADSCs showed significantly higher proliferation and adipogenic capacity with more lipid vesicle formation and expression of the adipogenesis-related genes than BMSCs. 62 Thus, further experiments are still needed to validate the differentiation tendency of ADSCs and whether they can promote the growth plate cartilage regeneration at the site of injury.
In addition to BMSCs and ADSCs, previous studies have explored using periosteum mesenchymal stem cells (peri-MSCs) to treat growth plate injuries. These cells have low donor morbidity and good pluripotency and can differentiate into osteoblasts, chondrocytes and adipocytes.63,64 Chen et al. extracted peri-MSCs from tibial periosteum of New Zealand white rabbits, embedded them into agarose and implanted them into the growth plate defects. The results showed a significant decrease in the growth arrest and angular defromity of tibia. 65 Li et al. used chitin as scaffolds combined with peri-MSCs to repair growth plate defects in rabbits. Finally, the lower limb angular deformity and length discrepancy were effectively corrected. 66
Furthermore, the Hedgehog pathway is crucial for bone and cartilage development. Glioma-associated oncogene 1 (Gli1) is a downstream transcriptional target of the Hedgehog pathway. 67 Previous research has found that Gli1-positive (Gli1+) cells are mesenchymal stem cells that support homeostasis and injury repair, particularly in the skeletal system and teeth. 68 Zhao et al. discovered that Gli1+ MSCs in the cranial suture are the primary source of stem cells for cranial and facial bone formation and repair after injury, and their elimination causes cranial suture closure and cranial bone growth arrest.69,70 Functional and sustainable regeneration of cranial suture cartilage can be achieved by combining Gli1 + MSCs with biomaterials. 71 Additionally, Gli1 + MSCs play a crucial role in growth plate development and bone growth, and their absence affects the biological function of the growth plate and leads to bone abnormalities. 72 Moreover, Gli1 + MSCs can differentiate into osteoblasts and chondrocytes, and can rapidly expand to participate in the repair process of fractures. 72 Although Gli1 + MSCs have not yet been used in the regeneration treatment of growth plate injuries, their multipotency and ability to respond to injury suggest that they could be applied to growth plate defects in the future to evaluate their repair effects. However, further experimentation is needed to verify the differentiation trend of Gli1 + MSCs and their ability to promote growth plate cartilage regeneration at the injury site.
In conclusion, MSCs have many advantages, and their main advantage is the ability to use allogeneic MSCs without eliciting a significant immune response.73,74 However, the safety and efficacy of new treatments is the most concern for patients. Many researchers have evaluated and confirmed the safety and efficacy of MSCs therapy through many clinical trials. 75 MSCs therapy also includes certain limitations, such as the necessity for cell culture and the time spent in culture, loss of differentiation capacity ex vivo or with multiple culture passages, and reduced or halted cellular division after multiple population doublings, 76 which may result in missing the optimal treatment time and effect. In addition, MSCs from different sources have their advantages and disadvantages. Thus, selecting appropriate MSCs targets and ecological niche to assist growth plate defect regeneration will be a key factor to obtain positive results.
Skeletal stem cells (SSCs)
SSCs are a group of somatic stem cells (also known as tissue-specific stem cells), which have the potential for self-renewal and multi-directional differentiation. 77 SSCs differentiate into multipotent bone, cartilage, and stromal progenitor cells(BCSP), which then give rise to OPCs and CPCs prior to terminally differentiating into bone, cartilage, and stroma. 78 Newton et al. found special stem cell niches in the growth plate, which Promote the self-renewal of CPCs and provide a continuous supply of chondrocytes over a prolonged period. Regulation of the pool of self-renewing progenitors involves the hedgehog and mammalian target of rapamycin complex 1 (mTORC1) signaling pathways. 1 Theoretically, SSCs with appropriate cell surface markers could offer an ideal cell source for cartilage tissue engineering. Wu et al. found CD146 + SSCs in the resting and proliferating zone of growth plates, suggesting that they are progenitor cells that can generate new clones of rapidly proliferating chondrocytes. Furthermore, CD146 + SSCs exhibited higher colony-forming capacity and continuous high chondrogenic differentiation capacity in vitro. Therefore, CD146 + SSCs may be an ideal seed cell for growth plate regeneration. 79
The resting zone of the growth plate is a stem cell-rich region that gives rise to the growth plate and exhibits regenerative capabilities in response to injury. 80 In addition to CD146 + SSCs, Mizuhashi et al. found that PTHrP-positive (PTHrP+) chondrocytes present in the resting zone of the growth plate expressed a panel of markers for skeletal stem and progenitor cells and possessed the properties of SSCs in cultured conditions. PTHrP+ cells have the ability of multilineage differentiation but can not be differentiated into adipocytes in vivo. PTHrP+ cells perform two different functions: (1) These cells differentiate into proliferating chondrocytes and hypertrophic chondrocytes and eventually become osteoblasts and bone marrow stromal cells at the post-mitotic stage. (2) These cells send PTHrP signals to control chondrocyte proliferation and differentiation. And Ihh secreted by hypertrophic chondrocytes maintains the proliferation of chondrocytes and formation of columnar chondrocytes. Thus, PTHrP+ cells are able to generate columnar chondrocytes longitudinally, and some PTHrP+ cells have the long-term self-renewal ability. In addition, PTHrP+ cells can maintain the integrity of the growth plate, and partial loss of PTHrP+ cells in the resting zone can alter the integrity of the growth plate by inducing premature hypertrophic differentiation of chondrocytes in the proliferating zone. 81 Therefore, PTHrP+ cells can be further used as seed cells for tissue engineering research. However, the extraction of PTHrP+ cells and maintaining their cell viability still needs further investigation.
FoxA transcription factors are key regulators of chondrocyte hypertrophy, and FoxA1-3 is highly expressed in the hypertrophic zone of newborn mice growth plate. 82 Muruganandan et al. found FoxA2 + SSCs at the top of the growth plate cartilage resting area near the edge of the secondary ossification center. It belongs to the PTHrP negative (PTHrP-) stem cell population and represents a subgroup of long-term skeletal stem cells. During early postnatal development (P0-P28), FoxA2 + SSCs have a dual osteo-chondro-progenitor fate, contributing to the chondrogenic lineage (by forming columnar stacks of chondrocytes in the growth plate) and the osteogenic lineage. After P28, FoxA2+ SSCs remain mostly in the growth plate and exhibit chondrogenic potential. The growth plate injury activates FoxA2+ cells to undergo proliferative expansion and to provide a framework for the regenerating tissue. In conclusion, FoxA2+ cells play an important role in the development of the growth plate and the regeneration of the growth plate after injury. Meanwhile, it was also found that there was no overlap between FoxA2+ cells and PTHrP+ cells. PTHrP+ cells existed at the bottom of the resting zone, while FoxA2+ cells were located at the top of the resting zone. But FoxA2+ cells will become PTHrP+ cells over time. However, compared with PTHrP+ cells, FoxA2+ cells have multipotency and higher self-renewability and longevity than PTHrP+ cells 80 (Figure 3). Therefore, we can try to utilize FoxA2+ cells to construct a growth plate regeneration framework in the future. However, further research is needed to identify effective methods for selecting suitable FoxA2+ cells without causing damage to the donor site, as well as extracting and preserving their activity.

FoxA2+ LTSSC maintain cartilage homeostasis by contributing to long-term self-renewal, chondrogenic differentiation and cartilage repair after injury. SOC = secondary ossification center. Image from Muruganandan et al. 2022 80 (open-access article distributed under the terms of the Creative Commons Attribution Licence, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited).
In addition, previous studies have found that periosteal stem cells labeled with cathepsin K (Ctsk) are a unique type of SSCs present in the periosteum. Ctsk+ cells can self-renew and differentiate into osteocytes and chondrocytes in vitro. 64 The periosteal stem cells exhibit greater clonogenicity, growth and differentiation capacity than BMSCs. 83 Tsukasaki et al. found that periosteal stem cells can provide OPCs for intramembranous osteogenesis. At the same time, periosteal stem cells-derived Ihh controls the growth plate resting zone stem cells to ensure prolonged skeletal growth. Therefore, the precise coordination of bone growth is regulated horizontally (from periosteum/perichondrium) in addition to the vertical regulation of hedgehog signals in the growth plate (from SOC and hypertrophic zone). In the future, we can further explore its regulatory effect on chondrocytes in the resting zone of the growth plate post-injury. 84 Currently, it has been shown that the periosteal stem cells can differentiate into chondrocytes under the unstable fracture condition. These cells also possess high migratory capabilities, allowing them to be recruited to the site of bone defects in response to specific migration signals. 85 The next crucial step is to investigate the differentiation and migration of periosteal stem cells at the site of growth plate defects, with the goal of identifying novel seed cells for use in regeneration therapies. Such research will provide valuable insights into the potential of PSCs for growth plate regeneration and help to pave the way for the development of effective clinical therapies.
Other cells
In addition to ADSCs, stromal vascular fraction (SVF) can also be extracted from adipose tissue. 86 SVF contains a variety of repair cells and a mixture of cytokines. Polly et al. found that gene expression for IGF-I, TGF-β1, and FGF-2 was significantly higher in SVF cells, and these growth factors play an essential role in cartilage formation. 87 Sananta et al. implanted AD-SVF combined with freeze-dried amniotic membrane into the growth plate defect to evaluate the therapeutic effect. The results showed that the bone bridge formation rate of the AD-SVF group was lower than that of the control group, and the immature chondrocytes could be seen in the defect site, indicating that AD-SVF has a good potential for tissue engineering in inducing the regeneration of injured growth plate, reducing the deformity and the bone bridge formation with limb discrepancy. 88 The advantage of SVF over ADSCs is believed to be in two fundamental areas. Firstly, the heterogeneous cellular composition of SVF may be responsible for the better therapeutic outcome observed in comparative animal studies. Secondly, SVF is much more easily acquired without needing cell separation or culturing conditions. Therefore, AD-SVF may have more application prospects than ADSCs. However, AD-SVF has its limitations. For example, SVF is only suitable for autologous therapy because of the existence of various known cell types that lead to immune rejection. 86
Furthermore, the Wnt/β-catenin signaling pathway is essential during growth plate development. Previous studies have shown that Wnt/β-catenin signaling may be involved in the regulation of growth plate CPCs.89,90 Usami et al. Found a novel cell population that resides in the outermost layer of the growth plate facing the Ranvier’s groove. This study indicate that the ZsG+ cells in the outermost layer of the growth plate are CPCs contributing to formation of the growth plate. Meanwhile, Supply of new cells to the growth plate from this ZsG+ population is an important mechanism regulating skeletal growth. And this process is be tightly controlled by Wnt/β-catenin signaling. β-catenin may inhibit the chondrogenic differentiation of ZsG+ cells in the Ranvier’s while supporting the expansion of ZsG+ chondrogenic progenitors to the growth plate. Therefore, Wnt-responsive ZsG+ cells have the potential to become new cells for growth plate regeneration 27 (Table 1).
The summary of seed cells in tissue engineering for growth plate regeneration.

ADSCs: Adipose-derived mesenchymal stem cells; AC: articular cartilage; BMSCs: bone marrow mesenchymal stem cells; CC: costal cartilage; GPC: growth plate cartilage; GP: growth palte; Peri-MSCs: Periosteum mesenchymal stem cells; MSCs: mesenchymal stem cells.
Cell-free therapy
Recent studies have shown promising results for the use of cell therapy in preventing bone bridge formation, promoting the growth plate cartilage regeneration, and correcting limb length discrepancies and angular deformities. However, this approach is associated with several limitations, including high cost, time-consuming procedures, low survival rate in vivo and challenges in clinical translation. 91 As such, researchers have proposed alternative, cell-free strategies to address these issues and improve the efficacy of regenerative medicine.
Extracellular vesicles (EVs) are a heterogeneous group of cell-derived membranous structures comprising small-sized vesicles (exosomes (Exos)) originating from the endosomal compartment and middle-sized vesicles (microvesicles) directly budding from the cell plasma membrane. And EVs play a role in maintaining intercellular communication and tissue homeostasis. 92 MSCs have been shown to have immunomodulatory properties and a repairing effect on tissues by regulating the immune microenvironment. The immunomodulatory properties of MSCs are attributed to their paracrine mechanisms, which mainly involve Exos secretion. 93 Thus, the application of MSC-derived exosomes is one of the current hot topics in the research of cell-free therapies. 94 Exos are small membrane vesicles with a diameter of 30–150 nm comprising complex Non-coding RNA (miRNA, LncRNA and CircRNA), proteins, and endogenous factors. Compared with MSCs, Exos have the advantages of low tumorigenicity, and low immunogenicity, can be transplanted repeatedly, and are relatively easy to store. 95 Yan et al. used the Exos from umbilical cord mesenchymal stem cells (UMSCs) as the natural carrier of lncRNA H19. At the cartilage defect site, lncRNA H19 delivered from UMSCs could competitively bind to miR-29b-3p to relieve its repression of the target gene FoxO3. This is, in turn, enhances chondrocyte migration and matrix synthesis and suppress apoptosis and senescence. Thus, a novel lncRNA H19/miR-29b-3p/FoxO3 axis might be a therapeutic target for post-traumatic focal cartilage defects. 96
Subsequently, some studies have used Exos from different MSCs to regenerate growth plate cartilage. Wong et al. treated growth plate defects by intra-articular injection of MSCs-derived Exos. They found that a single intra-articular injection of MSCs-derived Exos could significantly enhance the effect of growth plate cartilage regeneration and reduce the limb length discrepancy but did not inhibit bone bridge formation. Meanwhile, there was no significant immune response in this process. The improvement of limb length discrepancy may be attributed to the stimulation of chondrocyte proliferation and the enhanced deposition of GAG and Col II, facilitated by Exos. In addition, the ability of MSCs-derived Exos to enhance cartilage tissue formation could be attributed to the positive effects of MSCs-derived Exos on chondrocytes migration, proliferation, and ECM synthesis, mediated through CD73/ecto-59-nucleotidase activity. 97 However, only a single dose of MSCs-derived Exos was tested. An increased dose or dosing frequency may change the therapeutic outcomes of MSCs-derived Exos in the regeneration of growth plate cartilage. Therefore, as a minimally invasive cell-free therapy, MSCs-derived Exos can promote the growth plate cartilage regeneration and reduce limb discrepancy after injury. 98 Another study found that after treatment with BMSCs-derived EVs, the proliferating zone at the growth plate injury site showed flat and self-oriented chondrocytes began to arrange into columnar shapes parallel to the bone growth axis, which is similar to the arragenment of the growth plate cartilage structure. The results showed that BMSCs-EVs have differentiation potential and promoted the formation of correct spatial structure in different growth plate regions, especially in proliferative zone. 99 Thus, BMSCs-EVs has potential application in the field of growth plate regeneration.
Furthermore, inflammatory responses in growth plates are known to disrupt the anabolic and catabolic balance of chondrocytes, thereby degrading ECM through matrix-degrading enzymes, while Exos minimize ECM degradation after injury by modulating the local immune microenvironment. 100 Guan et al. combined BMSCs-derived Exos and ECM hydrogels for the growth plate defects treatment. BMSCs-derived Exos was found to enhance the polarization of macrophages from M1 to M2 by inhibiting the NF-κB signaling pathway, which may be beneficial for growth plate cartilage regeneration. Moreover, the anti-inflammatory effect of GMOCS-Exos can also improve the local immune microenvironment, promote the growth plate cartilage regeneration and inhibit bony repair after growth plate injury. 101 Therefore, the immunomodulatory properties of Exos can improve the inflammatory environment after growth plate injury, providing an appropriate microenvironment for growth plate cartilage regeneration. And its chondrogenic effect is essential for regenerative therapy.
Although EVs (including Exos) has the the low immunogenicity and high stability, the environmental genetic information and the protein content will affect its potential therapeutic effect to some extent. 102 In addition, the current quantitative control of EVs operations remains elusive, especially when they involve genetic operations. 103 Therefore, the above unsolved biosafety problems also restrict the clinical application of EVs. In the future, more studies, especially clinical trials, are needed to validate the safety and efficacy of exosome therapy.
Biological factors
Traditional treatments for growth plate injuries have a high recurrence rate and are associated with high uncertainty. Successful treatment should aim to prevent bone bridge formation and promote growth plate cartilage regeneration, thus restoring longitudinal bone growth function. Regeneration treatment strategies for the growth plate aim to overcome the shortcomings of traditional treatments by restoring the unique structure and function of the growth plate, which play a crucial role in treating growth plate injuries. Biological factors are currently used in growth plate regeneration to prevent bone bridge formation and promote growth plate regeneration through three key mechanisms: pro-chondrogenesis, anti-inflammatory, and anti-angiogenesis.
Growth factors
To improve the microenvironment for cartilage tissue formation and promote chondrogenesis, numerous studies have investigated the use of growth factors such as IGF-1, FGF-2, and TGF-β1. IGF-1 is a well-studied anabolic growth factor that plays a critical role in cartilage homeostasis and repair. 104 One study used localized delivery of IGF-1 from cell-free poly(lactic-co-glycolic acid) (PLGA) scaffolds to treat proximal tibial growth plate injury in rabbits, which resulted in significant cartilage regenera tion. 105 Another study created fusion proteins of cartilage-targeting single-chain human antibody fragment conjugated with IGF-1 and found that targeting IGF-1 to growth plate has superior therapeutic efficacy and minimizes adverse effects on other tissues compared to the direct use of IGF-1. 106 And the family of the FGF is critical for various cell types regarding differentiation, proliferation, migration, and growth. Among the FGF family members, FGF-2, FGF-8, and FGF-18 have recently been proposed to be the most important contributing factors in cartilage modulation. 107 As mentioned earlier, FGF-2 plays a role in the fibrogenic phase of growth plate injury by stimulating the proliferation and migration of OPCs and MSCs, and inhibiting chondrocyte differentiation. 22 In the application of engineered cartilage tissue, FGF-2 was usually used with other growth factors. Coleman et al. demonstrated that BMSCs produced greater amounts of sulfated glycosaminoglycans in the presence of FGF-2 and TGF-β1 than with TGF-β1 treated alone, which indicates that FGF-2 plays a role in growth plate regeneration. In addition, TGF-β1 plays an important role during the chondrogenesis of MSCs. 108 McCarty et al. implanted a gel foam scaffold containing autologous BMSCs and TGF-β1 in the proximal tibial growth plate defect, and results showed that the scaffold contained TGF-β1 was more effective in inhibiting bone bridge formation but failed to form new cartilage structure at the defect site. 53 In addition, SDF-1a and TGF-β3 are biological factors associated with growth plate tissue regeneration. 109 SDF-1a promotes the migration of MSCs to defect sites. And TGF-β3 acts as a chondrogenic factor to induce the differentiation of these stem cells toward the chondrogenic lineage, which has been shown to induce the transcriptional activity of SOX-9 through the Smad signaling pathway, thereby promoting the chondrogenic differentiation of MSCs. 110 Therefore, chondrogenesis-related growth factors play an important role in growth plate injury repair, and the effect of combined use may be better. However, targeted therapy should also be considered to reduce adverse reactions to other sites.
Connective tissue growth factor (CTGF) in the process of endochondral bone morphogenesis promotes growth plate chondrocytes proliferation, maturation, and hypertrophy. CTGF is the core driving force of cartilage and bone regeneration. 111 Nishida et al. created a 2 mm diameter defect on the surface of mouse articular cartilage, then implanted CTGF hydrogel with a collagen sponge and found new cartilage formation at the defect 4 weeks after surgery. In vitro, CTGF enhanced the expression of Col II and aggrecan, and induced chondrogenesis in mouse BMSCs, suggesting that CTGF may be an ideal tool for the treatment of osteoarthritis and tissue-engineered articular cartilage reconstruction. 112 Another study showed that CTGF is the direct target gene of SOX-9 in growth plate chondrocytes and nucleus pulposus cells. The deficiency of SOX-9 resulted in decreased expression of CTGF. Thus, there is a strong association between SOX-9 and CTGF in chondrocyte regulation. 113 Previous studies have shown that SOX-9 can maintain the columnar proliferation of growth plate chondrocytes and promote cell proliferation (two key features of functional growth plates). 114 Therefore, CTGF as a growth factor can be considered in the future to repair growth plate defects.
Osterix is a transcription factor with a zinc finger structure. Osterix plays a vital role in normal bone formation and bony repair as an essential transcription factor in osteoblast differentiation. Osterix activation is regulated by protein kinase-D (PKD) activation, which is required for osteoblast differentiation. 29 Previous studies showed that PKD inhibitors suppressed Osterix expression and blocked BMP-2-induced osteoblast differentiation and mineralization in vitro. 115 In addition, another study showed that PKD inhibitors prevented FGF-2-induced increased bone formation. 116 Chung et al. found that PKD inhibition after growth plate injury could delay osteogenic differentiation of MSCs and promote cartilage tissue repair and endochondral osteogenesis. The final results showed that using PKD inhibitors could partially inhibit the osteogenic signal, lead to significantly delayed bone healing of the injured growth plate, increase the number of MSCs and cartilage, and reduce the bone formation at the growth plate injury site, suggesting that PKD activity can be blocked during growth plate injury repair to reduce bone bridge formation at the injured site. 117 However, it is still necessary to further study the effect of PKD inhibition on the results of growth plate regeneration and the potential side effects. Whether PKD can be used as a potential intervention target to promote the healing of injured growth plate cartilage remains to be further verified.
Anti-inflammatory factors
In addition, anti-inflammatory therapy also plays an important role in growth plate regeneration. Yang et al. found that Punicalin could maintain the transcriptional activity of FOXO3, thus playing a positive role in improving growth inhibition and apoptosis of chondrocyte induced by pro-inflammatory factors IL-1β and TNF-α and maintaining the normal chondrocytes phenotype. 13 Wan et al. found that the levels of TNF-α and matrix metalloproteinase 3 were increased during the chondrogenic differentiation of ADSCs. TNF-α then bound to its receptor and activated the NF-κB pathway, leading to a decrease in ECM synthesis and secretion. And the inhibition of TNF-α levels significantly restores ECCM formation. 61 Arasapam et al. used the nonsteroidal anti-inflammatory drug celecoxib to block the inflammatory mediators COX-2 and iNOS in the inflammatory phase of the growth plate. They found that the inhibition of COX-2 or iNOS seemed to delay both MSCs differentiation to cartilage tissue and bone remodeling processes. However, they could not prevent the downstream bony repair at the injury site. Meanwhile, gene expression studies suggest that long-term use of celecoxib therapy may affect overall gene expression and further affect the healing process. 16 Zhou et al. used TNF-α antagonists to inhibit the inflammatory response after growth plate injury. However, the experimental results showed that the mRNA expressions of TNF-α and IL-1β did not change significantly on days 1 and 8 after growth plate injury, indicating that the inhibition of TNF-α alone could not inhibit the inflammatory response after growth plate injury. Moreover, given that the inflammatory response may play an important role in the healing process of the growth plate injury, single anti-inflammatory therapy may affect the normal healing process. Therefore, synergistic treatment should be considered in combination with other therapeutic modalities.
Anti-angiogenesis factors
In addition to anti-inflammatory and chondrogenic approaches, some studies have proposed preventing bone bridge formation by inhibiting angiogenesis. Previous studies have indicated that bone bridge formation at the site of growth plate injury primarily occurs through two pathways: intramembranous osteogenesis and endochondral osteogenesis. 118 The angiogenesis at the injury site is crucial for both pathways. 23 Anti-VEGF antibody (bevacizumab) was applied to the rat tibial growth plate injury model, and it was found that angiogenesis and bone bridge formation at the injury site decreased after treatment. However, after systemic administration, the length and shortening of the tibia caused by growth plate injury became more evident on day 60, and significant expansion of the hypertrophic zone of the intact growth plate cartilage adjacent to the growth plate injury site was observed. 25 Therefore, while VEGF-neutralizing antibodies may be a potential osteopontin agent, this study suggests the systemic treatment may have significant side effects, such as limb shortening and damage to adjacent uninjured growth plate. Therefore, Erickson et al. developed an injectable hydrogel containing VEGF-neutralizing antibodies. The rapid release of VEGF-neutralizing antibody was achieved by local injection of alginate chitosan hydrogel at the injury site. The results showed that this therapy could inhibit bone bridge formation without interfering with normal longitudinal bone growth, which is of great significance for the regeneration treatment of growth plate injury.
However, VEGF may not be the only critical regulatory factor driving angiogenesis. There may be other factors and mechanisms driving angiogenesis on growth plate injury. 119 Perlecan is a heparin sulfate proteoglycan expressed in the basement membrane and cartilage. Perlecan in cartilage enhances activation of VEGF164-VEGFR2 signaling in endothelial cells through direct binding to VEGFR2, thus promoting angiogenesis necessary for cartilage matrix remodeling and intraconal bone formation. 120 It plays a crucial role in endochondral bone formation. In addition, FGF-18 has been shown to regulate growth plate chondrocyte proliferation, hypertrophy, and cartilage vascularization, which are essential for endochondral ossification. 121 Together with FGF-2 and FGF-18, perlecan promotes chondrocyte proliferation and differentiation into growth plate cartilage in a characteristic columnar arrangement. 24 Therefore, perlecan may be a new target to prevent bone bridge formation and promote the formation of the growth plate structure.
Others
MicroRNAs(miRNAs) are essential regulators of many biological processes such as cell proliferation, differentiation, migration, apoptosis, and tumorigene. 122 miRNAs play a crucial role in multiple processes of bone development. 123 Zhang et al. identified that miR-410 promoted TGF-β3-induced chondrogenic differentiation of hMSCs by directly targeting Wnt3a. Thus, miR-410 is a critical positive regulator of hMSC chondrogenic differentiation. 124 However, some miRNAs negatively regulate chondrogenic differentiation. Yang et al. found that miR-145 is a critical negative regulator of chondrogenic differentiation in mouse MSCs, which directly targets SOX-9 in the early stage of chondrogenic differentiation. 125 However, miR-145 did not change significantly during the chondrogenic differentiation of hMSCs. 126 Lee et al. found that miR-495 inhibits the expression of ECM protein by targeting SOX-9, indicating that the chondrogenic differentiation of hMSCs is inhibited. Thus, miR-495 is an essential negative regulator in the chondrogenic differentiation of hMSCs. 126 Therefore, increasing positive or reduced negative regulators may have a beneficial effect on controlling MSCs to chondrogenic differentiation.
In addition, Bone morphogenetic protein receptor 2 (BMPR2) can induce osteogenetic and chondrogenic differentiation of stem cells in vitro. 127 Thus, Inducing chondrogenic differentiation of BMRP2 and inhibiting its osteogenic differentiation and endochondral bone formation are valuable methods for inducing chondrogenic differentiation of MSCs, promoting cartilage formation, and maintaining the phenotype of chondrocytes. 128 Tian et al. found that miR-143-3p inhibits the deposition of proteoglycans and the expression of ACAN and Col2α1 in MSCs by targeting BMPR2, thus regulating early chondrogenic differentiation of MSCs. 128 Yang et al. found that MiR-100-5p inhibited the osteogenesis of hBMSCs and angiogenesis by targeting BMPR2 and suppressing the BMPR2/SMAD1/5/9 signaling pathway. Therefore, specific miRNAs can produce positive chondrogenesis and reverse the inhibition of osteogenic differentiation by targeting BMPR2. 129 This method can be applied to MSCs to make their differentiation trend toward ideal chondrogenesis.
Dicer is the critical enzyme of miRNA biological formation. Its deficiency in chondrocytes results in a reduction in the number of proliferating chondrocytes through two distinct mechanisms: decreased proliferation and accelerated differentiation into postmitotic hypertrophic chondrocytes, suggesting that miRNAs promote chondrocyte proliferation and inhibit hypertrophic differentiation. However, the study did not identify the specific miRNAs responsible for these defects. 130 Papaioannou et al. miR-140 facilitates differentiation of resting chondrocytes and inhibits hypertrophic differentiation to increase columnar proliferating chondrocytes, whose normal proliferation and survival require let-7 miRNA. 131 And there was a gradually decreasing PTHrP concentration gradient from the growth plate resting zone to the hypertrophic zone. 132 Jee et al. found that the PTHrP concentration gradient across the growth plate induces differential expression of mir-374-5p, mir-379-5p, and mir-503-5p, and the higher expression levels of these miRNAs promote proliferation and inhibit hypertrophic differentiation. 133 Zhang et al. found that expression of miR-143 was reduced, while IHH levels were elevated in the injured growth plate. By targeting IHH, miR-143 downregulation could promote growth and inhibit apoptosis of PCSCs, thereby modulating chondrogenesis effects. Thus, miR-143 may serve as a novel target for cartilage regeneration and repair. 128 In addition to miRNAs, long noncoding RNAs (lncRNAs) were also found to play essential roles in regulating growth plate chondrocyte proliferation and differentiation. LncRNA growth-arrest-specific 5 (Gas5) has been reported to be a critical control element during growth, differentiation, and development in mammalian species. 134 Liu et al. found that lncRNA Gas5 regulated proliferation and apoptosis in the growth plate by controlling FGF1 expression via miR-21 regulation. Moreover, Gas5 contributed to proapoptosis by increasing active caspase-9 and FGF1 expression but decreasing miR-21 expression. In contrast, miR-21 contributes to pro-proliferation by acting as a negative regulator of Gas5. 135 Therefore, miRNAs are essential regulators of chondrogenesis and the maintenance of chondrocyte phenotypes. By regulating the expression level of miRNAs at the injured site, it can be used as a potential therapeutic target for the control and regeneration of MSCs cartilage differentiation in the future.
Tissue engineering scaffold materials
The growth plate is responsible for the longitudinal growth of the bone and must bear the weight between the metaphysis and the diaphysis. Therefore, suitable scaffold materials should show good biocompatibility, biodegradability, and suitable mechanical properties. Many biomaterials have been used to repair growth plate injury, including natural and synthetic materials (Figure 4).

The overview of the tissue engineering for growth plate regeneration.
Natural materials
Extracellular matrix (ECM) scaffold: ECM is an ideal biological scaffold material in nature. Natural ECM is a dynamic structure composed of various components, including ECM polymers (collagen, fibrin, elastin), growth factors, GAG, hormones, and other signal molecules. 136 Natural ECM scaffolds can provide a microenvironment similar to the extracellular environment that can maintain the activity of cytokines, growth factors, and other functional proteins and contain information signals when seeding cells, which may be superior to synthetic polymers in function. 137 In vivo, the state of cell differentiation and its function are supported by tissue-specific signaling, cell-cell contact, cell polarity in tissues, and cell-ECM connections. 138 Therefore, scaffolds generated by suitable tissue-derived ECMs are suitable for regenerative therapy. However, ECM scaffold is easy to collapse due to insufficient mechanical strength, which leads to unsatisfactory treatment results. The mechanical properties of the ideal tissue engineering scaffold should match the mechanical properties of the target tissue. Therefore, understanding the collagen fiber arrangement of the ECM in each organ is essential for the design of scaffolds. 139 Previous studies have shown that cartilage ECM-derived 3D porous acellular matrix scaffolds can provide an appropriate 3D environment to support the adhesion, proliferation, and differentiation of BMSCs. 139 However, it is not enough to provide a microenvironment suitable for cells to survive and proliferate. Li et al. designed an ECM-derived oriented acellular scaffold with excellent biomechanical properties for the treatment of New Zealand white rabbit growth plate repair. BMSCs in scaffolds gradually differentiated into chondrocytes and secreted glycosaminoglycan (GAG) around the ECM. During the process, the scaffold and the regular growth plate fused well, and no inflammatory reaction occurred. The results showed that the scaffold could promote growth plate regeneration, prevent bone bridge formation, and reduce angular deformity and limb discrepancy. This growth plate-derived ECM scaffold with good properties shows the potential to repair growth plate injury in young rabbits. 52
Agarose scaffold: Agarose is a natural polysaccharide polymer consisting of D-galactose and 3, 6-anhydride l-galactose, which has many excellent properties, such as biocompatibility, reversible gel behavior, water solubility, mechanical adjustability, and physicochemical properties that support its use as a biomaterial for cell growth and drug delivery.140,141 Coleman et al. found that the injection of an in situ agarose gel into the growth plate defect resulted in a reduction of limb length discrepancy and a thicker growth plate compared to empty defect controls. However, it is not sufficient to regenerate growth plate tissue. 102 Then they repaired the growth plate defect with BMSCs-loaded agarose gel scaffolds. The results showed that the implant could correct the limb length discrepancy and prevent the premature fusion of normal growth plate tissue around the defect. 58 Chen et al. embedded the cultured MSCs into agarose and implanted them into the growth plate defect. The results showed that the implant could delay the growth arrest of the rabbit tibia, but the problem of tibia angular deformity and length discrepancy has not been solved. 65
Alginate scaffold: Alginate is a linear polysaccharide composed of homopolymeric units of 1,4-linked (-D-mannuronic acid) (M) and (-L-guluronic acid) (G) that give its structure flexibility and gelling properties. The high pore structure of alginate is conducive to cell adhesion and growth. Its pore size is between 100 and 150 mm, and the pores are connected. 142 Meanwhile, alginate as biomaterials is cost-effective, non-cytotoxic, easy to modify, and easy to cross-link. 140 Erickson et al. established an in vitro model of growth plate cartilage by using neonatal mouse growth plate chondrocytes encapsulated in alginate hydrogel beads. In the hydrogel beads, chondrocytes showed high viability, cartilage matrix deposition, and low levels of chondrocyte hypertrophy. Meanwhile, chondrocytes coated with alginate gel beads can be regulated by Pthrp-Ihh signaling. Exogenous PTHrP stimulates cell proliferation and inhibits cell hypertrophy, while Ihh signaling stimulates chondrocyte hypertrophy. Importantly, treatment of alginate bead cultures with Ihh or thyroxine resulted in the formation of a discrete region of hypertrophic cells that mimics the tissue structure of the native growth plate cartilage, thereby establishing a tunable in vitro system that mimics the tissue structure of the growth plate for modeling signal network interactions. This is necessary to induce the ribbon structure of the growth plate chondrocytes and can also be used to grow cartilage with specific geometric shapes to meet individual needs. 143 Markstedt et al. prepared a new hydrogel composed of nanocellulose and alginate. The higher the alginate content of the hydrogel, the greater the storage modulus and the lower the biotoxicity. 144 However, pure alginate saline gels carry a negative charge 145 and alginate fails to give the cell its natural binding site, 142 which may influence the may have some effect on the cells. Sturtevant and Callanan used antifreeze proteins to modify the pore structure of alginate. They prepared scaffolds with orientated aligned pores to simulate the central region of cartilage. The scaffolds had positive effects on cell viability and function. 146 Thus, alginate is still a suitable material for constructing growth plate regeneration scaffolds. In the future, through the continuous modification of alginate gel, the composite material which is most suitable for cartilage regeneration of growth plate can be found.
Chitin/Chitosan scaffold: Chitin, a major component of the exoskeleton of arthropods and molds, consists of n-acetyl-D-glucosamine monomer units linked by β-1, 4-glucoside bonds, whereas chitosan is the deacetylated form of chitin. 147 Chitosan has good biodegradability, anti-microbial ability, and mucosal adhesion. Its degradation products in vivo are non-toxic and non-immunogenicity, so it is widely used in drug release, tissue engineering, cell scaffolds, and other fields.148 –150 ECM Ishikawa et al. used chitosan-based hydrogels as scaffolds for chondrocyte culture. They found that the substantial degradation behavior of hydrogels could stimulate the embedded chondrocytes to form hyaluronic cartilage, which might be realized through the deposition induced by the change of hydrogel microstructure, the enlargement of pore size and the decrease of network density. 151 Li et al. used (peri-MSCs)-loaded chitin scaffolds to repair growth plate defects. peri-MSCs have a better proliferating ability and survival rate in chitin scaffolds, which may be because chitin components are structurally similar to GAG, which is the main division of cartilage ECM. 66 In vitro, chitin supports the expression of ECM proteins in human chondrocytes and maintains the natural round or oval shape of chondrocytes. 152 Erickson et al. prepared Ginipin-chitosan microgels and demonstrated the ability of these microgels to prevent early bone bridge formation in growth plate cartilage defects in vivo. 153 Planka et al. designed a novel scaffold consisting of Col I and chitosan nanofibers loaded with allogeneic MSCs and chondrocytes to repair growth plate defects. The scaffold has the characteristics of higher rigidity and easy operation. In addition, it has the ability of hydrodynamic seeding, which can protect the transferred cells. Meanwhile, the layering technology of peripheral implantation of MSCs and central implantation of chondrocytes is also one of the highlights. The experimental results showed that the combined transplantation of composite scaffolds with MSCs and chondrocytes could prevent growth arrest and angular deformity. Moreover, in most cases, hyaluronic cartilage-like tissue appeared in the defect and formed the typical columnar tissue of the growth plate. 154 Chitin/chitosan scaffolds have shown excellent growth plate repair ability in the current study, and their clinical transformation and application can be further explored in the future.
Demineralized bone matrix (DBM) scaffold: The DBM is composed of collagen scaffolds containing various growth factors and Urist invented the DBM preparation method. 155 Jin et al. extracted rabbit iliac growth plate chondrocytes and harvested them after proliferating in monolayer culture in vitro for 3 weeks. The cells were then inoculated into a DBM scaffold to construct a composite material for growth plate injury repair. The results showed that chondrocytes adhered to the surface of DBM scaffolds and proliferated, indicating good biocompatibility and adhesion. In addition, the DBM scaffold has many natural multiporous structures, so the tissue fluid can penetrate through the pores and supply the nutrition needed. However, DBM scaffolds can only prevent bone bridge formation for a short time. The bone and fibrous tissue then grew into defects after the scaffold had been absorbed, resulting in severe angular deformity. 156 Therefore, DBM scaffolds may need to be modified by doping elements or other methods to provide mechanical support and prevent bone bridge formation after the growth plate injury.
Synthetic materials
Poly(lactic-co-glycolic acid) (PLGA) scaffold: PLGA has excellent biocompatibility and biodegradability. Moreover, it can change the polymer’s mechanical properties, degradation rate, and drug release kinetics by controlling the copolymer’s composition (ratio of lactic acid to glycolic acid), molecular weight, and other factors. FDA has approved drug delivery devices using PLGA microspheres. 157 Sundararaj et al. treated growth plate injury with localized delivery of IGF-I from PLGA scaffolds. In vitro, the experiment confirmed that the release of IGF-I encapsulated in the PLGA scaffold occurred in a biphasic pattern, and the scaffold was almost wholly released during degradation. Swelling scaffolds following implantation may be advantageous in bridging gaps between the scaffold and surrounding tissue, possibly enhancing the entry of progenitor cells into the scaffold. The results showed that there was cartilage regeneration, but more disorganized. The reason might be that PLGA scaffolds failed to provide adequate mechanical support or degraded too quickly, resulting in a short support time. 105 Therefore, implanting scaffolds with higher mechanical strength and slower degradation may lead to better results. In addition, PLGA scaffolds create a microenvironment necessary for the proliferation and chondrogenic differentiation of MSCs. 158 Clark et al. evaluated the effect of IGF-1-loaded PLGA scaffold on the growth plate defects repair in the absence of MSCs. Although the final defect site did not form a columnar structure, the amount of chondrocytes increased, and the destruction of adjacent growth plate was reduced. However, there was no significant reduction in limb angular deformity. 159 In conclusion, the excellent performance of PLGA scaffolds can be used in growth plate regeneration therapy. Future consideration should be given to strengthening its mechanical support capability to provide a better regenerative microenvironment.
Alloy scaffold: Mg-based materials have the characteristics of biodegradability and biocompatibility. Hence they do not induce a systemic inflammatory reaction. In addition, the mechanical properties of Mg-based materials resemble those of cortical bone. Compared with the traditional materials used in orthopedic implants, the stress shielding response caused by Mg-based materials is reduced. 160 However, the Mg-based materials form hydrogen gas and aqueous solutions during their rapid degradation, which leads to further implant degradation. 161 Therefore, by surface modification, Song et al. designed an Mg-Ca-Zn alloy containing plasma electrolyte oxidation (PEO)-coating. Compared to non-coated alloys, PEO-coated alloys degraded slowly with diminished hydrogen gas formation, and the surface mechanical properties and stability were increased. The concentrations of Mg2+, Ca2+, and Zn2+ in local tissues were maintained within normal ranges during the degradation of Mg-Ca-Zn alloys. 162 In animal experiments, PEO-coated Mg-Ca-Zn alloy scaffold were implanted into the rabbit growth plate defects. The results showed that the implants did not cause significant immune response after implantation. In addition, the implants have excellent mechanical properties, biodegradability and stability. During the repair process, the implant binds closely to the surrounding tissue. Eventually, the defect site formed a small bone bridge, and the function of the growth plate was not interfered with by the implant. Therefore, PEO-coated Mg-Ca-Zn alloy implants have specific development potential in treating growth plate injury. Natural biological materials are mainly used in clinical trials, and most studies using synthetic materials are carried out on animal models. 163 The safety, clinical transformation, and application ability of synthetic materials must be further verified.
GMOCS scaffold: Gelatin methacryloyl (GM) hydrogels is similar to the natural ECM and it has excellent biological properties and adjustable physical properties. 164 Chondroitin sulfate (CS) is a GAG that exists primarily in the ECM of biological tissues and possesses chondrogenic properties. It exhibits good biocompatibility and have the ability to induce the cartilage formation.165,166 Guan et al. introduced doping aldehyde-functionalized chondroitin sulfate (OCS) into GM hydrogels to form biocompatible ECM (GMOCS) hydrogels by dynamic Schiff base bond formations. The dual network framework of GMOCS hydrogels ensured Exos sustained release to meet the growth plate repair requirement. BMSCs-Exos-loaded GMOCS hydrogels could regulate the immune microenvironment at the injured site, induce the high expression of anti-inflammatory factors, and construct a chondrogenic microenvironment to promote cartilage regeneration. The results showed that bone bridge formation was inhibited at the injured site, promoting the activity of chondrocytes and ECM remodeling, and thus promoting the regeneration of chondrocytes. 101 In addition, GMOCS hydrogels provided an optimal microenvironment for BMSCs, significantly enhancing the chondrogenic differentiation of BMSCs and inhibiting osteoblastic differentiation due to its OCS components. BMSCs-loaded GMOCS hydrogels can prevent the bone bridge formation and regenerate the three layers of columnar cartilage structures at the defect site, including the resting zone, the proliferative zone and the hypertrophic zone. Thus, GMOCS hydrogels can achieve sustained release of active ingredients and affect the differentiation trend of MSCs, thus provide a good niche for promoting the repair of growth plates injury and cartilage regeneration. Their safety and feasibility for clinical conversion can be further evaluated in the future 167 (Table 2).
The summary of scaffold in tissue engineering for growth plate regeneration.

AC: articular cartilage; BB: bone bridge; BMSCs: bone marrow mesenchymal stem cells; GPC: growth plate cartilage; GP: growth plate; CHC: chondrocytes; ECM: extracellular matrix; Peri-MSCs: periosteum mesenchyal stem cells; PLGA: poly lactic-co-glycolic acid; DBM: demineralized bone matrix; Exos: exsomes.
Biomimetic structure of tissue engineering scaffold
Cartilage tissue engineering has provided a promising strategy for the repair of cartilage defects. By simulating the layered structure of articular cartilage, a biomimetic microenvironment can be constructed to promote tissue regeneration. Recently, Wang et al. prepared a dual-layer biomimetic cartilage scaffold that mimicked the structural design, chemical cues, and mechanical characteristics of mature articular cartilage. This scaffold had a structure similar to natural cartilage and possessed high mechanical strength, which guided the morphology, orientation, proliferation, and differentiation of BMSCs. In vivo, the biomimetic cartilage scaffold effectively repaired new cartilage tissue in the defect area. This experiment provided a microenvironment suitable for regeneration and the results were satisfactory. 168 This study also inspires the preparation of biomimetic scaffolds for growth plate cartilage regeneration. By simulating the structural characteristics of the growth plate, a suitable microenvironment can be created for regeneration. Therefore, understanding the unique structure of growth plates is crucial for the design of bionic structures.
It was mentioned earlier that growth plate chondrocytes are divided into three compartments according to their morphology and function: resting zone, proliferative zone, and hypertrophic zone. The growth plate is heterogeneous in morphology, and its mechanical properties vary with respect to the histological zone. 169 Therefore, when simulating native growth plate cartilage tissue, the changes in its mechanical properties should be fully considered. A previous study showed that the resting zone is stiffer and less permeable than the other zones, exhibiting a dense hyaline cartilage structure and relatively few resting chondrocytes. The chondrocytes were arranged in columns in the proliferative zone below the resting zone. The stiffness of the proliferating zone is about 30% lower than that of the resting zone, while the permeability is about four times higher. 170 Eckstein et al. found that the change in stiffness from the upper hypertrophic zone to the lower hypertrophic zone on the growth plate may be mechanical cues to direct cells to endochondral ossification. Furthermore, the spatial differences in the mechanical properties of growth plates are partly caused by different ECM compositions. The resting zone is the stiffest zone due to the presence of a dense collagen structure. 169
Meanwhile, the growth plate is influenced by complex local mechanical and molecular environments. Dynamic compressive forces have been shown to stimulate growth plate cartilage growth, while shear and hydrostatic pressures are more likely to drive cartilage ossification. 171 Appropriate mechanical stimulation is essential in typical growth plate structure and function. The lack of weight bearing during growth and development will lead to changes in the actin cytoskeleton in the growth plate chondrocytes and result in the destruction of the typical columnar organization. 172 In contrast, excessive pressure on the growth plate will also destroy the typical histological characteristics and metabolic function of the growth plate. 173 Mechanical loading on the resting and proliferative zones can induce the longitudinal arrangement of cells to maintain the formation of the columnar structure of the growth plate and drive the longitudinal growth. 174 And there is a gradient of mechanoregulatory signals (hydrostatic stress and maximum tensile strain) in the growth plate, and the more significant mechanical regulatory signals of chondrocytes in the proliferative zone can maintain cartilage pheontype, keep chondrocytes from differentiating, and provide conditions favorable for cell division. However, the mechanoregulatory signal gradually decreases along the long axis of the cartilage in the hypertrophic zone and gradually provides favorable conditions for osteogenic differentiation. 175 Therefore, the unique structure and mechanical properties of the growth plate are essential for maintaining normal growth function, while ignoring these key factors may cause the effect of regeneration and repair is not ideal.
In addition to understanding the original structure and function of the growth plate, the preparation of biomimetic scaffolds also needs the support of advanced technology. 3D bioprinting is the most promising approach in regenerative medicine, ensuring that cells, materials, biomolecules, and mechanical structures are precisely integrated. 176 The technique can precisely control the microstructure and composition of scaffolds on a micro-scale, which can closely mimic the geometric structure of biological tissues in anatomy to realize the design of multi-level tissue structure. Compared to traditional tissue engineering methods, 3D bioprinting systems allow for the precise deposition of biomaterials and cells in 3D Spaces to construct more complex and precise spatial relationships. 177 Therefore, 3D bioprinting technology is an ideal method for mimicking native tissue niches in vitro. 178 Standard 3D printing techniques that are suitable for bone or cartilage tissue engineering include 3D drawing/ink writing, stereolithography (SLA), selective laser sintering (SLS), selective laser melting (SLM), fused deposition modeling (FDM), inkjet /extrusion-based bioprinting, and the new DLP 3D technology.179,180 By combining 3D bio-printing, computer-aided design (CAD), and imaging data, the designed structure has the characteristics of individuation. 181 In addition, the mechanical properties of scaffolds can be regulated by different preparation processes and chemical modifications to mimic the mechanical properties of primary tissues. The melt deposition modeling technique can be used to create gradient-structured scaffolds and the inkjet/extrusion bioprinting can be used to prepare biomimetic gradient scaffolds pre-inoculated with growth factors and stem cells. 182 In conclusion, each 3D printing technology has its own most applicable range and the continuous progress of 3D printing technology will provides strong support for biomimetic tissue engineering.
Currently, only a few studies have utilized biomimetic scaffolds for treating growth plate injuries. Yu et al. developed 3D printed biomimetic composite materials that can satisfy the mechanical needs of the growth plate and contain bioactive niches that support cartilage formation. First, they characterized the mechanical properties of rabbit growth plate tissue, and then used the modulus data to create a biomimetic scaffold that closely matched the original tissue. The scaffold was precisely fabricated using Stereolithographic digital light processing (DLP) technology, which used poly(ethylene glycol) (PEG)-based resin to create a stiff 3D printed structure composed of vertical pillars. The scaffold was then combined with cartilage-mimetic hydrogel to form the biomimetic composite material and implanted into the injured site. In vitro experiments demonstrated that the biomimetic composite material could induce chondrogenic differentiation of MSCs and stimulate ECM production. In vivo experiments showed that the biomimetic composite material allowed for significantly greater limb growth than other treatment options, although only limited cartilage-like tissues were observed at the injury site. 183
In another research, Li et al. suggested that the designed structure of the scaffold should imitate the native growth plate tissue, which has an oriented structure of chondrocytes associated with its mechanical and biological functions. They developed an ECM-derived oriented acellular scaffold to mimic the column structure of the natural growth plate. The scaffold has a biomimetic columnar structure with uniform pore sizes in both the transverse and longitudinal planes. BMSCs adhere and distribute like the columnar structure of natural chondrocytes, and oriented microtubules induce BMSC migration into the core of the scaffold. This structure may enhance the mechanical properties and protect the cells from early critical compression before sufficient ECM is deposited. Additionally, the scaffolds exhibit excellent biocompatibility, and their extracts have no toxicity to BMSCs. The results showed that the scaffold promoted new cartilage regeneration and prevented bone bridge formation, reducing angular deformity and length differences. While unable to completely prevent angulation and lower limb shortening deformities, the ECM-derived oriented growth plate biomimetic scaffold shows potential for the growth plate cartilage regeneration in young rabbits. 52
In conclusion, few biomimetic scaffolds currently exist for the repair of growth plate injury and the regeneration of growth plate cartilage. As previously mentioned, Li and Wong’s studies both mimicked the columnar structure of the growth plate by constructing scaffolds containing vertical columnar structures. The results showed that biomimetic structural had achieved preliminary results in promoting growth plate regeneration, but there was also the problem of unstable treatment effect. Therefore, it may be necessary to optimize the scaffold structure further and rationally use seed cells and biological factors to enhance the growth plate regenerative ability. In addition, more complex issues should be considered in future clinical applications, such as the pathological environment that is not conducive to cartilage regeneration after growth plate injury, the formation of bone bridges, and the differences in the mechanical properties of growth plates at different ages. Therefore, when designing biomimetic composite scaffolds in the future, we should consider the current pathological phase of growth plate injury and try to add biological factors to improve the microenvironment. At the same time, combined with computer-aided technology to characterize the mechanical properties of the growth plate and the tissue structure of different regions, and then design a biomimetic scaffold with a layered structure, which fully mimics the native growth plate and realizes an efficient growth plate regeneration scaffold. And the structure may have caused an enhancement of the mechanical properties to protect the cells from early critical compression before sufficient ECM was deposited. At the same time, the scaffolds have excellent biocompatibility, and their extracts have no toxicity to BMSCs. The results showed that the scaffold promoted the new cartilage regeneration and prevented bone bridge formation, thus reducing the angular deformity and length difference. Although not able to completely prevent angulation and lower limb shortening deformity, the ECM-derived oriented growth plate biomimetic scaffold shows potential for repairing growth plate injury and regenerating growth plate cartilage in young rabbits.
Scaffold-free strategy
Traditional scaffold-based tissue engineering techniques involve the use of biomaterials to construct a 3D structure that supports the attachment of living cells and provides a microenvironment for tissue regeneration. However, the size, porosity, shape and physicochemical properties of the scaffolds will affect the immune response and regeneration outcomes. 184 Many existing scaffolds still struggle to accurately mimic the chondrogenesis of native tissue, resulting in a mismatch between the mechanical and biochemical properties of the scaffolds and native tissues, 185 which may affect the regeneration therapy of target tissues. Although scaffolds with biocompatible materials and adjustable mechanical properties have been widely used to overcome these problems, the progress of clinical transformation and application is still partially affected. In recent years, the concept of scaffold-free tissue engineering has been paid more and more attention. The scaffold-free strategy mainly rely on the ability of cells to produce and maintain their own ECM, without interfering with cell-cell signaling, cell migration, or small molecule diffusion after implantation. 186 Currently, scaffold-free technologies can be roughly divided into cell sheet technology, transient scaffold culture method, and cell polymerization technology. 187
As an alternative to scaffold-based therapy, cell sheet technology has attracted the attention of researchers in recent years. Cell sheet technology can effectively preserve cell-cell interactions as well as ECM and cell sheet structure, resulting in a high cell density and uniform cell distribution of the constructed tissue, which more closely mimics native tissue. In addition, cell sheet technology can avoid limitations and potential problem of scaffolds.179,180 GÜLTEKİN et al. used cell sheet technology to prepare MSCs and chondrocyte sheets for growth plate repair. And the cell sheets developed in the present study facilitated growth plate cartilage regeneration and sufficiently prevented the development of a bone bridge. 188 It is worth mentioning that cartilage cell sheets made from a thermal responsive culture dish (TRCD) combined with traditional bone marrow stimulation were tested in eight patients. Cell sheet transplantation has been shown to be safe and effective in cartilage regeneration in defect sites. 189 However, cell sheet technology also has some defects, including poor stability, low mechanical strength, difficulty in operation, and other problems, which are easy to increase the difficulty of clinical surgery. 190 Therefore, the application of cell sheet technology may be limited, but it can be considered an auxiliary means of growth plate regeneration therapy in the future.
In addition to cell sheets, some studies have used scaffold-free cell-polymer to treat growth plate injuries. Chow et al. used scaffold-free bioengineered cartilage pellets (BCP) to fill the defect in the growth plate. After transplantation, the BCP exhibited continuous growth in the host and showed a resemblance to the natural growth plate. BCP has the potential to restore femoral longitudinal growth and prevent bone bridge formation or premature physeal closure. 191 However, one limitation of this technique is that the in vitro proliferation of the BCP would peak at 14 days and subsequently decline by 21 days during pellet culture, which might affect the growth potential of the BCP after its transplantation. 42 In addition, Lee et al. recently generated a cartilage tissue analog (CTA) using a suspension culture with biophysical properties similar to native hyaline cartilage. CTA was implanted into the growth plate defect, and results showed that tissue similar to the growth plate cartilage was formed in the growth plate defect while reducing bone bridge formation and angular deformities. Therefore, CTA transplantation as a treatment for growth plate injury reduces deformities by reducing bone bridge formation. Yoshida et al. created an in vitro-generated scaffold-free tissue-engineered construct (TEC) derived from synovial MSCs. A complex of Synovial MSCs and ECM formed a three-dimensional structure through active tissue contraction. Proliferative and prehypertrophic chondrocytes were formed at the injuried site after TEC implantation, and the cells presented the growth plate-like columnar arrangement. At the same time, bone bridge formation was inhibited at the injuried site. Therefore, scaffold-free TEC can be used as biomaterial for growth plate regeneration therapy. In addition, the experiment also suggested the effect of local signal gradient on growth plate cartilage regeneration. 192 Therefore, the future research direction may be to mimic the development process of normal growth plates or control the proliferation, differentiation and hypertrophy of chondrocytes by regulating the signal gradient inside growth plates, so as to promote the regeneration of growth plates.
Furthermore, scaffold-free tissue engineering can be accomplished using 3D bioprinting, whereby bioinks containing cell suspensions and additives serve as precursor materials to form the final product. Bioinks can be classified as scaffold-based or scaffold-free, with scaffold-based bioinks being the predominant form of 3D bioprinting. However, scaffold-based bioinks have certain limitations, such as byproducts. Scaffold-free bioprinting, on the other hand, is associated with challenges such as fast bioprinting process and slow integration rate between cells. 193
One approach to scaffold-free bioprinting is the Kenzan method, which utilizes a pre-designed stainless steel array of microneedles as temporary support. Tissue spheres are placed into the gaps between the microneedles using a robotic arm. Once the cells secrete ECM and establish connections, the microneedle array is removed, resulting in the formation of a complete 3D structure. This method enables the printing of vertical three-dimensional structures with temporary support, which can later be removed, achieving a scaffold-free state. 194 In another study, Breathwaite et al. used a scaffold-free 3D printer to generate 3D bio-printed structures from human bone marrow mesenchymal stem cell spheres and induced in vitro chondrogenesis and osteogenesis. The structures were manipulated and integrated under different conditions to replicate the bone-cartilage interface and other physiological processes, such as intramembrane ossification. 195 Despite these advances, scaffold-free 3D printing still faces several challenges, including improving the quality (phenotype and size uniformity of spheres) and yield of the bioink, as well as maintaining cell viability during printing. 196 Addressing these limitations is likely to be a key area of focus for future research.
Conclusion and prospect
Growth plate injuries involve a complex pathological process that often results in the formation of bone bridges. Previous studies have attempted to limit or reverse this process by targeting individual pathological events, but have not yielded satisfactory results. Therefore, it is necessary to target multiple pathological events and modulate their dynamic interactions in order to facilitate growth plate regeneration. To accomplish this, a multifaceted modulation strategy using advanced tissue engineering technology may be a promising strategy to explore.
Tissue engineering integrates scaffolds, seed cells, and biological factors to develop functional biomaterials that effectively promote the regeneration of growth plate cartilage. Seed cells and biological factors are vital components of tissue engineering, as they are instrumental in restoring the original structure of the growth plate. The regenerative process relies on the coordinated interaction of various cells and biological factors, and their functions can be further manipulated to enhance the regenerative outcomes. Additionally, to overcome the limitations of cell therapy, cell-free therapy has emerged, utilizing various MSC-derived extracellular vesicles (EVs) for regenerative purposes. While some progress has been made, the biosafety of this approach requires further validation. Scaffolds play a crucial role in providing mechanical support and creating a suitable microenvironment for growth plate regeneration. Biomimetic scaffolds, designed to mimic the native tissue environment, have shown more promising results in regeneration compared to previous scaffold designs. Furthermore, research is also exploring scaffold-free strategies as an alternative approach to promote the regeneration of growth plate cartilage.
In future research, the development of highly adaptable tissue engineering implants capable of responding to various stimuli during dynamic changes holds great importance. The emergence of 4D printing technology, which introduces the concept of “time” into 3D printing, shows promise in this regard. 4D printed scaffolds exhibit unique responsive functions and shape-shifting abilities that have the potential to accurately simulate the dynamic nature of native tissues, achieving true biomimicry. However, technical support for 4D printing is still in its early stages, and the precise biological interactions between materials and tissues are yet to be fully understood. Through the advancement of related technologies and the exploration of multidisciplinary collaborations, tissue engineering will provide crucial technical support for growth plate cartilage regeneration. This will help overcome the current obstacles in the treatment process and ultimately realize the effective regeneration of growth plate cartilage.
Footnotes
Acknowledgements
No.
Author contributions
RYG, HJZ and XNC contribute equally to this work. PFZ conceived the review and supervised the work. RYG, HJZ and XNC partcipated in the work and wrote the main manuscript text. YLB prepared figures and tables. MJF and YWW contributed to the analysis. All authors reviewed the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Jiangsu Provincial Key Research and Development Program (CN) [grant number BE2019608]; Postdoctoral Research Foundation of China [grant number 2022M721685]; Jiangsu Health Commission Medical Research Program [grant number M2020048]; National Facility for Translational Medicine (Shanghai) Open Program [grant number TMSK-2021-304]; Nanjing International Science and Technology Cooperation Program[grant number 202002050]; Nanjing Medical Science and Technology Development Key Program [grant number ZKX18041].
