Abstract
Enthesis, the interfacial tissue between a tendon/ligament and bone, exhibits a complex histological transition from soft to hard tissue, which significantly complicates its repair and regeneration after injury. Because traditional surgical treatments for enthesis injury are not satisfactory, tissue engineering has emerged as a strategy for improving treatment success. Rapid advances in enthesis tissue engineering have led to the development of several strategies for promoting enthesis tissue regeneration, including biological scaffolds, cells, growth factors, and biophysical modulation. In this review, we discuss recent advances in enthesis tissue engineering, particularly the use of biological scaffolds, as well as perspectives on the future directions in enthesis tissue engineering.
Introduction
Enthesis is a key component of the tendon and ligament that forms a bridge between soft and hard tissues (tendon/ligament and bone) through the fibrous, unmineralized fibrocartilagenous, mineralized fibrocartilagenous, and bony regions.1,2 Enthesis also supports the mechanical transmission of tensile stress and force from the main body of the tendon/ligament to the bone surface.3–5 Because of its location between the tendon/ligament and the bone, the enthesis is frequently damaged during avulsion fracture and ankylosing spondylitis.6,7 The incidence rate of enthesis orthopedic injury has increased markedly in recent years, and each year, around 30 million people undergo enthesis reconstruction in the United States and Europe, which costs over 163 billion dollars.8,9 For example, in the USA, it is estimated that >100,000 reconstructions are done for anterior cruciate ligament (ACL) enthesis each year at a cost of >1.5 billion dollars. 10 Thus, effective strategies for regenerating injured enthesis are urgently needed.
Traditional surgical treatments for enthesis include the reconstruction of enthesis with broken tendon/ligament terminals.11–13 This is achieved through: (a) direct insertion of the broken enthesis into the bone tunnel, (b) mechanical anchoring of the reconstructed tissue using sutures or screws, and (c) minimizing externally-loaded forces to avoid accidental loosening or re-rupture after surgery.14–19 Because traditional surgical treatments mainly focus on reestablishing enthesis anatomy while overlooking enthesis tissue regeneration, their efficacy in enthesis repair is limited. For instance, the reestablished enthesis may remain weakly connected to the bone for an extended time, leading to the formation of scar tissue instead of the desired healthy enthesis tissue, and in severe cases degeneration and lysis may occur, resulting in the loss of the anatomical reconnection between the enthesis and the bone.20–23 Currently, the lack of effective methods of regenerating native enthesis but not the weak scar tissue is a major clinical challenge, and in recent decades, enthesis tissue engineering has emerged as a way of overcoming these limitations in the traditional surgical treatments.24,25
Enthesis tissue engineering has led to the development of several novel methods of improving enthesis regeneration. The advanced methods that are currently used in enthesis tissue engineering can be divided into the biological scaffold strategy, the cell strategy, the growth factor strategy, and the biophysical modulation strategy (Figure 1).3,26–28 These strategies can be used alone or in combination to promote enthesis regeneration. The biggest challenge in enthesis regeneration is the lack of effective ways of generating the typical four-layered histological structure at the interface between the bone and the broken enthesis tissue.
24
The biological scaffold strategy focuses on producing biocompatible enthesis scaffolds using various biomaterials and fabrication techniques to mimic the native structure of enthesis tissue as closely as possible.29–31 The use of biomimetic enthesis scaffolds also seeks to provide a favorable environment for enthesis tissue regeneration and to offer an extra platform for the growth and infiltration of regenerative enthesis tissue. To ensure that the enthesis scaffold is highly biomimetic, a multilayered enthesis scaffold fabricated using an elaborate design on the inner structure is required.
32
The cell strategy of enthesis tissue engineering relies on enthesis-associated cells to promote regeneration at the site of injury.26,33 The growth factor and biophysical modulation strategies have been found to markedly promote enthesis regeneration.34,35 Mounting evidence indicates that compared with traditional surgical treatments, these enthesis tissue engineering strategies can improve regeneration of the immature four-layered enthesis tissue
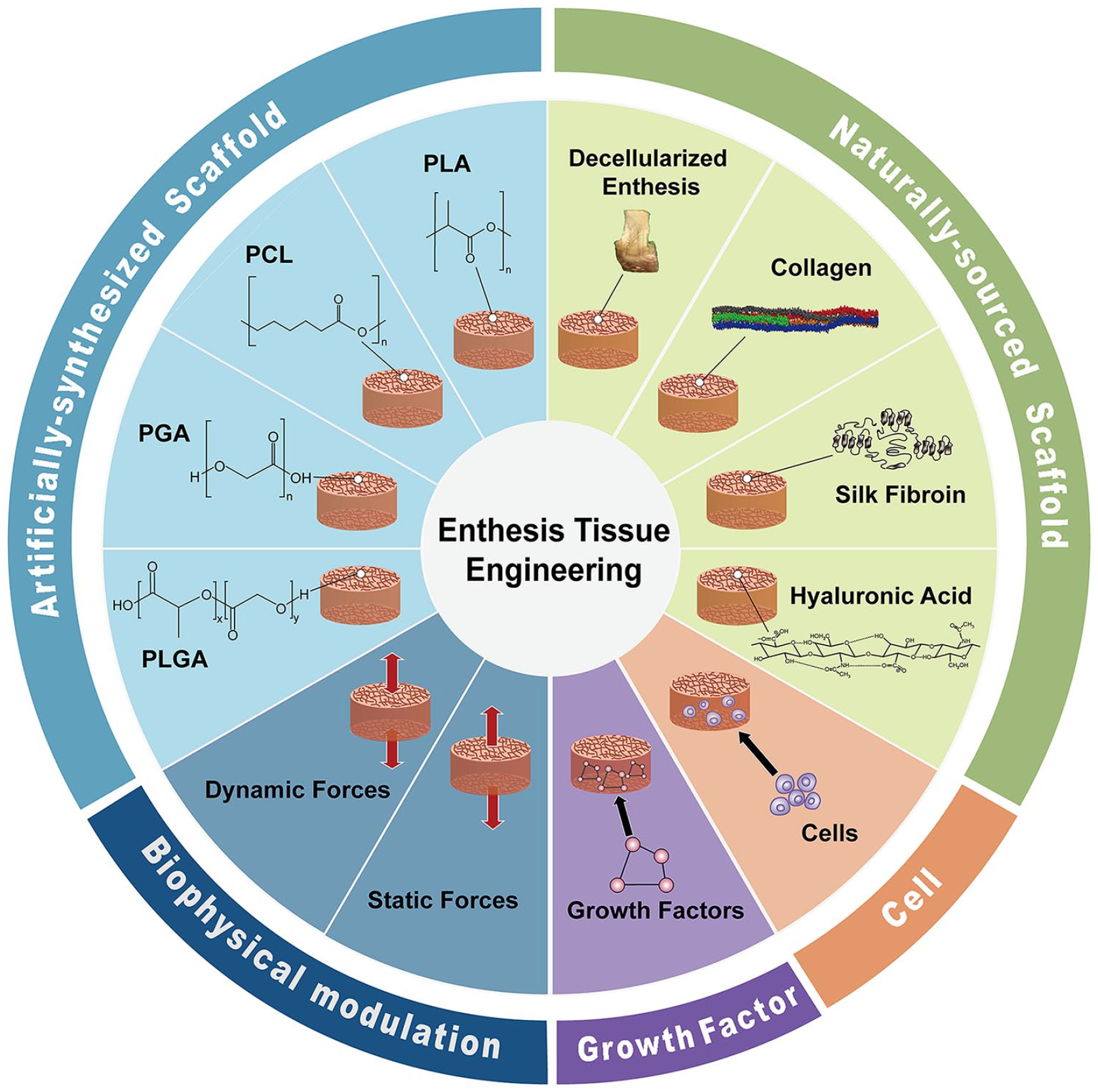
The common strategies in enthesis tissue engineering.
In this review, we first describe the characteristics of enthesis tissue, followed by a detailed discussion of the four strategies of enthesis tissue engineering, their practical applications, and their effects on enthesis regeneration. In the final part of the review, we discuss the current state and future research perspectives on enthesis tissue engineering.
Characteristics of enthesis tissue
Enthesis tissue usually refers to the tendon/ligament–bone interface and not the muscle–tendon enthesis tissue. Tendons and ligaments are highly similar tissues and with the exception of the main bodies, their bony terminals—enthesis tissue—are almost identical.40–42 Studies have also shown that injuries occurred in enthesis tissue are common enough.43–45 In this section, we try to introduce the background information on enthesis tissue and to highlight its complex histological compositions and the natural healing process of enthesis tissue in detail.
Enthesis tissue structure
Based on closeness to the bone surface, enthesis tissue is divided into the fibrous region (Zone I), unmineralized fibrocartilagenous region (Zone II), mineralized fibrocartilagenous region (Zone III), and bony region (Zone IV)40,42,46 (Figure 2(a)–(d)).
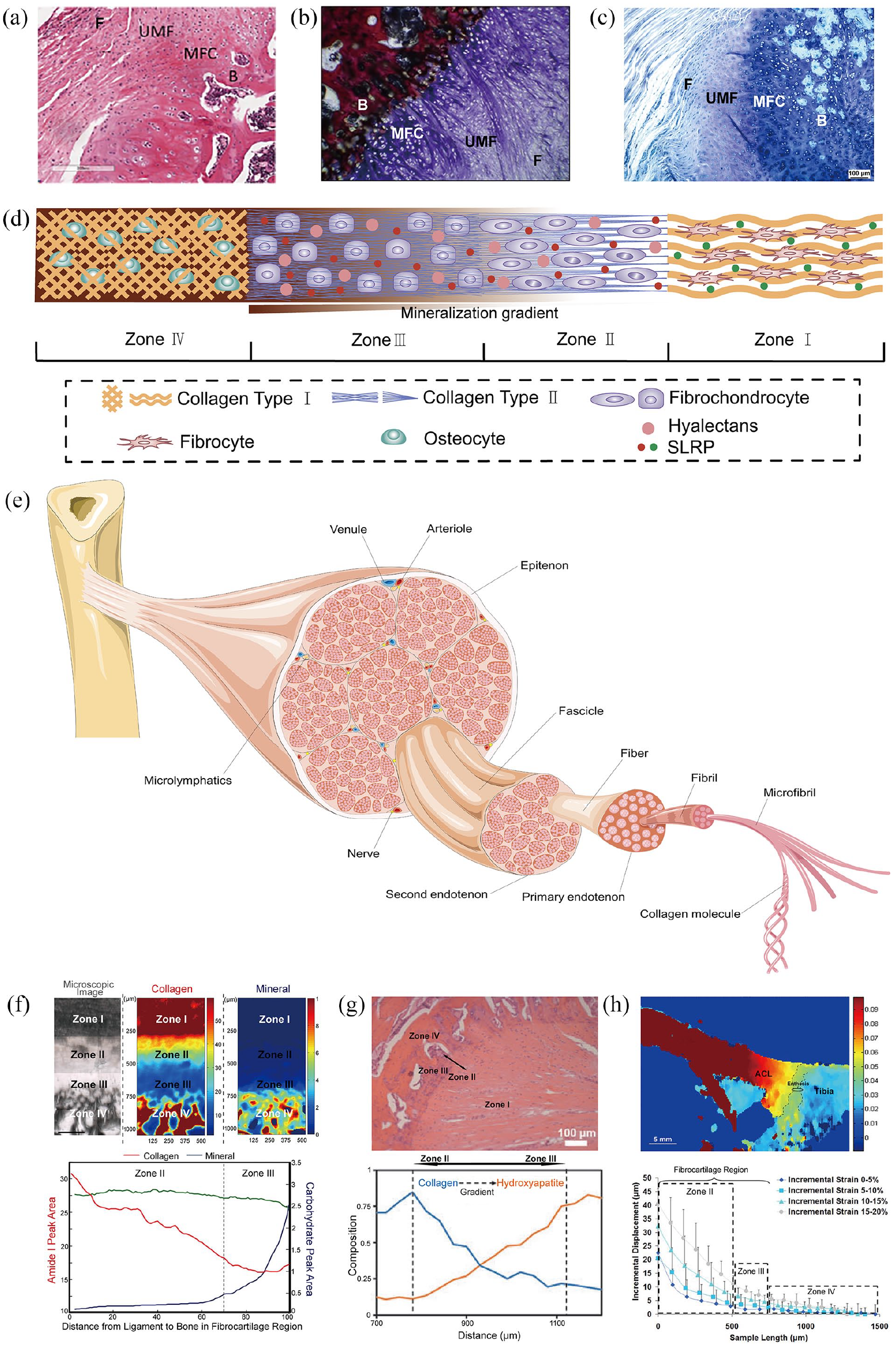
(a) Hematoxylin & Eosin image of the structure of rat enthesis, 40 (b) Sanderson staining and Van Gieson staining image of the structure of sheep enthesis, 42 (c) Giemsa staining image of the structure of sheep enthesis. 46 (d) A schematic representation of transition across the four zones. (e) The special axial hierarchical structure of Zone I. (f) The upper three figures are a microscopic image, a collagen distribution map under FTIR-I, and a mineral distribution map under FTIR-I in tibial ACL enthesis. The lower figure is a quantitative analysis of the collagen and mineral distribution in the enthesis fibrocartilage region. 74 (g) Raman spectral mapping of the gradient transition of collagen and mineral content in the fibrocartilage region of an ACL enthesis. 75 (h) Upper figure: an elastographic analysis of ACL enthesis in tension shows the distribution of mechanical stress experienced through the enthesis (yellow–green–blue). 78 Lower figure: strain response curve of an ACL enthesis under uniaxial compression. 79
The fibrous region
The fibrous region (Zone I) is made of regular special axial–hierarchical structures 47 (Figure 2(e)). Its basic structural unit is collagen type I, which first twists in clusters of five to form microfibrils, the primary fiber bundles. Next, large microfibril bundles spatially repeat along the axis and in parallel, forming the second fiber bundle, fibril. The fibril has a diameter of 100–500 nm, depending on the number of microfibrils it contains. However, based on electron microscopic examination, all fibrils have a biomolecular structure with the same periodicity (the D-band) irrespective of diameter. The D-band spatially recurs every 67 nm along the fibril and it dose can indicate how regularly microfibrils are distributed and combined. 48 Next, through the action of various biomolecules, including proteoglycans, many fibrils are crosslinked and wrapped by the primary endotenon, forming a collagen fiber. Collagen fibers are then encapsulated by a second endotenon and an epitenon, forming a fascicle that completes the Zone I structure.49,50 Both the collagen fiber and fascicle possess inner activity because the membrane and sheathe that wraps them are in free contact with each other, which allows the collagen fibers and fascicles to slightly move and glide transversally.47–51 With regard to histological nutrient exchange in Zone I, the vascular networks, together with some tiny nerves and lymphatic capillaries, are mainly distributed in the external layer (epitenon) and the second endotenon, and is rarely observed in the central region.50–52 These microvessels have a small average diameter and blood flow through them is very slow. This indicates that total blood volume in the enthesis is much lower than in other tissues like the skin and muscles, and that the nutrient/waste–product exchange in the inner region of enthesis tissue mainly depends on diffusion through the matrices, which is not sufficient for cellular metabolic needs. This nutrient shortage may be severer during accelerated cell growth, reproduction, or differentiation. Thus, the limited nutrient exchange caused by its anatomical features can explain why the enthesis has low cellular density, with 55%–70% of the enthesis being extracellular matrix (ECM), which mainly contains water. 53 This may also explain why enthesis tissue healing is slow after injury.
With regard to cellular composition, Zone I is made of several cell types, including fibrocytes, fibroblasts, vascular endothelial cells, nerve cells, and various immune cells, including mast cells, neutrophils, and macrophages). 54 Fibrocytes are the most common cell type in Zone I, accounting for 90%–95% of all cells. 53 Fibrocytes are mostly derived from fibroblasts, which are locally embedded in intra-fibril spaces and have a larger variation potential and a higher reproduction rate than fibrocytes.49,55–57 Fibrocytes and fibroblasts are the main producers of the ECM in Zone I and they produce almost all ECM components. Without considering water, the ECM of Zone I is mainly made of collagen fibrils, which make 60%–85% of the dry weight. Collagen type I makes up most (80%–90%) of the total collagen content, followed by collagen type III (1%–10%) and collagen type II (2%). The ECM of Zone I also contains a small amount of elastin, proteoglycans, glycosaminoglycans (GAGs), and glycoproteins. These functional biomolecules can enhance the biomechanical properties of Zone I, such as its stiffness, toughness, and viscoelasticity.49,58 In enthesis tissue engineering, the factors influencing fibrogenesis might mainly function in Zone I.
The unmineralized fibrocartilagenous region
The unmineralized fibrocartilagenous region (Zone II) is characterized by an acute change in tissue composition, with the ECM components changing into collagen type II and III, which in humans, are typical components of the hyaline cartilage. 59 Zone II also contains small amounts of collagen type I and X, GAGs, and proteoglycans.60–63 The proteoglycans in Zone II are mostly aggrecan and versican, which are large chondroitin sulfate proteoglycans (hyalectans).62–64 With the exception of hyalectans, some small leucine-rich repeat proteoglycans (SLRP), such as chondroadherin and biglycan, are highly abundant in Zone II than in Zone I.65,66 Hyalectans and SLRP, which occur on the surface of fibrils, are both negatively charged proteoglycans located in the gaps of collagen type II fibrils.65,67 The molecular and electrostatic repulsive forces between proteoglycans and collagen fibrils cause collagen type II fibrils to diverge. The alignment of the collagen fibrils becomes slightly unparallel and irregular, and the fibrils can slide along each other, which markedly increases the degree of anisotropy. 68 These biomolecular features illustrate why enthesis tissue endure tensile or compressive mechanical forces in all directions. Moreover, fibrochondrocytes in Zone II are substituted for fibrocytes, which become the main cell type. However, the morphology of fibrochondrocytes is highly similar to that of fibrocytes, which are spindle-like or flat and stretched along the enthesis axis. 69
The mineralized fibrocartilagenous region
Zone II and the mineralized fibrocartilagenous region (Zone III) are both fibrocartilagenous tissues but differ in their degree of tissue calcification. In Zone III, collagen type X is the second most abundant component of collagen fibrils instead of collagen type III, and collagen type II is still predominant although its overall fibril alignment is more random and anisotropic, which may be caused by increased levels of aggrecan and versican.59,62,63 Although tissue mineralization starts to appear in this region, it is incomplete. The mineral in this area is amorphous calcium phosphate (ACP), which does not directly interact with collagen molecules but can interact with other structural biomolecules like proteoglycans, GAGs, and glycoproteins via their charged amino acid residues, particularly the negatively charged residues on carboxyl groups.70,71 Because of the low amount of non-collagenous protein and their binding capability with ACP, the mineralization process in Zone III is interrupted in the early stage. This may be why no crystalline calcium phosphate can be formed ulteriorly. 72 In Zone III, the cells are predominantly fibrochondrocytes with different morphologies and distributions. Fibrochondrocytes have a larger volume and rounder shape and are stochastically distributed in the network of collagen fibrils. 69 In enthesis tissue engineering, strategies for promoting chondrogenesis usually target both Zone II and III.
Bony region
The bony region (Zone IV) has the maximum level of histological mineralization. The predominant collagen fibrils, mineralized by hydroxyapatite, revert to collagen type I while osteocytes, osteoblasts, and osteoclasts, become randomly scattered in the bony ECM network. 73 In enthesis engineering, the enhancement of osteogenesis via various strategies can vastly improve Zone IV regeneration.
The structure transition from Zone I to Zone IV
It is clear that progression from Zone I to Zone IV is accompanied by an abrupt change from soft to hard tissue. In enthesis tissue, structural transition is compositionally distinct and graded in the interfaces between Zone I and II as well as Zone III and Zone IV, while at the same time, there is also a continuous structural gradient from Zone II to Zone III (Figure 2(f)).74,75 In the Zone II–Zone III span, hydroxyapatite levels increase continuously but relatively more rapidly in Zone III, whereas collagen levels decrease gradually, indicating the existence of a gradient transition between the two fibrocartilagenous zones (Figure 2(g)). 75 With regard to the structure–function relationship in the four zones, the typical multi-layered tissue transition and its spatial heterogeneity closely correlate with the distribution of the mechanical stress experienced through the enthesis.76–79 An elastographic analysis of tension-loaded ACL enthesis found that the highest displacement was in Zone I and that the peak value decreased gradedly from Zone I to Zone II, gradiently from Zone II to Zone III, and gradedly from Zone III to Zone IV, indicating an overall increase in tissue stiffness that corresponded with structural transition through the four zones (Figure 2(h)). 78 Moreover, an analysis of region-dependent strain response by the ACL enthesis under uniaxial unconfined compression found that incremental displacement decreased continuously from Zone II to Zone III, which may indicate that increasing tissue stiffness corresponds to structural transition in these two zones (Figure 2(h)). 79 Taken together, the characteristics of enthesis tissue discussed above are key in minimizing stress concentration and promoting gradual load transfer from the soft to hard tissue. These characteristics might also explain why this small soft–hard region (within a few hundreds of micrometers) endure relatively high local tensile/compressive stresses and forces without failure, deformation, or tear.
The natural healing process of enthesis
The natural healing process of enthesis is complex and can be divided into three phases: a) the inflammatory and necrotic phase (early period), b) the rapid proliferative phase (middle period), c) the gradual remodeling phase (final period).80–82 The inflammatory and necrotic phases usually occur in the first week of injury. Inflammation is triggered by inflammatory factors that target fibroblasts, fibrocytes, and vascular endothelial cells, resulting in cytonecrosis of the damaged tissue.83–89 During this period, the biological structure and biomechanical properties of the injured enthesis deteriorate rapidly, severely limiting regeneration capacity. Therefore, suppressing inflammation in the early period is crucial for good recovery.90,91 Because the levels of the inflammatory mediators interleukin (IL)-1β, IL-6, IL-8, tumor necrosis factor (TNF), matrix metalloproteinase (MMP)-1, MMP-3, and MMP-13 are upregulated in the early period of enthesis healing,92,93 there are several potential targets for effectively inhibiting inflammatory reactions. After inflammation subsides, the injured enthesis enters the rapid proliferation phase, which usually lasts from the first to the third or fourth week of injury and leads to marked increases in the number of nearly all cell lineages in the enthesis, including fibrocytes, fibroblasts, fibrochondrocytes, osteocytes, and vascular endothelial cells. The production and secretion of the ECM, which is mainly made of collagen type III, also accelerates. 94 This results in new enthesis tissue although at this stage it is immature and fragile, and its biomechanical properties have not met healing requirements. The last phase of healing is the long remodeling period which typically lasts several months or years. This process is characterized by a significant decrease in the total cellular density and a continuous increase in the number of fibroblasts, decrease in ECM content and overall production of collagen and GAGs, and a gradual increase in the production of collagen type I.95,96 In this period the typical four-layer physiological structure of the enthesis slowly rebuilds and the maximum stress loading capacity rises.97–99 However, after natural recovery, the enthesis tissue is far weaker than the uninjured tissue. Perfect natural healing is not achievable and there is risk that the regenerated enthesis tissue may re-rupture and undergo resorption.94,95 Moreover, enthesis healing is nonuniform and intrinsic healing process is relatively slower than extrinsic healing, probably because of the heterogeneity of the vascular network distribution in enthesis tissue.100–102
Based on the natural characteristics of enthesis tissue listed above, numerous recent studies have attempted to promote enthesis regeneration and identified several factors that may effectively improve enthesis regeneration, including bio-gels, scaffolds, growth factors, and stem cells.103–107
Tissue engineering strategies for enthesis reconstruction
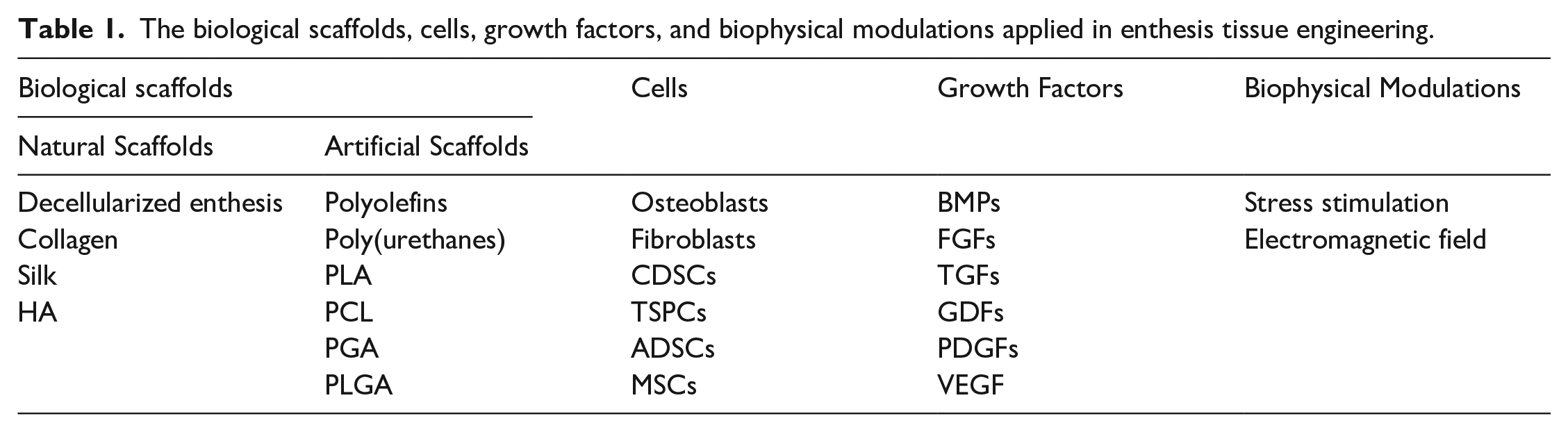
The field of tissue engineering (TE) involves several disciplines, such as cytology, histology, molecular biology, biochemistry, material science, engineering science, and clinical medicine. The concept of TE was first introduced in 1993 by Langer and Vacanti and its overall aim is to develop feasible methods of artificially rebuilding various tissues and organs.108,109 TE, which not only seeks to reconstruct histological structures, but also to restore original biological functions, has been widely applied in orthopedics and there are several strategies for repairing bone defects.110–112 However, the development of enthesis TE is not as advanced and the successful reconstruction of enthesis tissue has not yet been achieved.3,26,113–115 Studies on the use of TE in enthesis repair involve four strategies, the biological scaffold, cell, growth factor, and biophysical modulation strategies. Of these, the biological scaffold strategy has attracted the most interest (Table 1).
The biological scaffolds, cells, growth factors, and biophysical modulations applied in enthesis tissue engineering.
The biological scaffold strategy
A suitable biological enthesis scaffold should have the following properties: (a) a biomimetic structure, (b) mechanically biomimetic properties, (c) mouldability, (d) biodegradability, and (e) biocompatibility (should not be cytotoxic or inflammatory).3,42,116–118 These requirements markedly limit the number of materials that can be used as biological scaffolds in enthesis TE. Enthesis scaffolds can be natural or artificial.119–127 Artificial enthesis scaffolds are fabricated using synthetic polymeric biomaterials, whereas natural ones are made of natural materials like collagen, silk fibroin, and hyaluronic acid. The most straightforward approach for developing enthesis biological scaffolds involves decellularized enthesis tissue from laboratory animals or human donors. In the following sections we discuss the common types of scaffolds in detail.
Decellularized scaffolds
Decellularized enthesis scaffolds, which are the most readily available, fall into three categories based on their origin—autogenous enthesis scaffolds, allogeneic enthesis scaffolds, and xenogeneic enthesis scaffolds.128,129 Although their tissue sources are different, the principle of scaffold-making is largely the same and involves depletion of the cellular component of raw enthesis tissue to obtain the ECM only, which can form a enthesis biological scaffold. 130 The main advantage of decellularized enthesis scaffolds are that they are easy to obtain and the tissue’s biomimetic capacity is guaranteed (especially when the scaffold is based on the same anatomical site).131,132
A study by Xu et al. which developed a porcine decellularized Achilles enthesis scaffold using a new protocol, found that the decellularized enthesis scaffold preserved the typical histological structure of enthesis tissue well (Figure 3(a)). 133 Additionally, their biomechanical assessment showed that the decellularized enthesis scaffold retained mechanical properties that were appropriate for practical applications (Figure 3(b)). 133 A study by Su et al. produced a porcine decellularized Achilles enthesis scaffold using a refined protocol and found that it promotes enthesis regeneration in vitro and in vivo (Figure 3(c)). 134 Compared with the control group (a simple decellularized tendon scaffold), more mesenchymal stem cells (MSCs) adhered, proliferated, and infiltrated the decellularized enthesis scaffold (Figure 3(d)). The MSCs in this scaffold significantly upregulated osteogenesis-associated genes like RUNX2, OPN, and ALP, as well as tenogenesis-associated genes like SCX, THBS4, and VIM (Figure 3(e)), and when implanted into rabbit tibia, the enthesis scaffold induced the regrowth of enthesis tissue (Figure 3(f)). 134
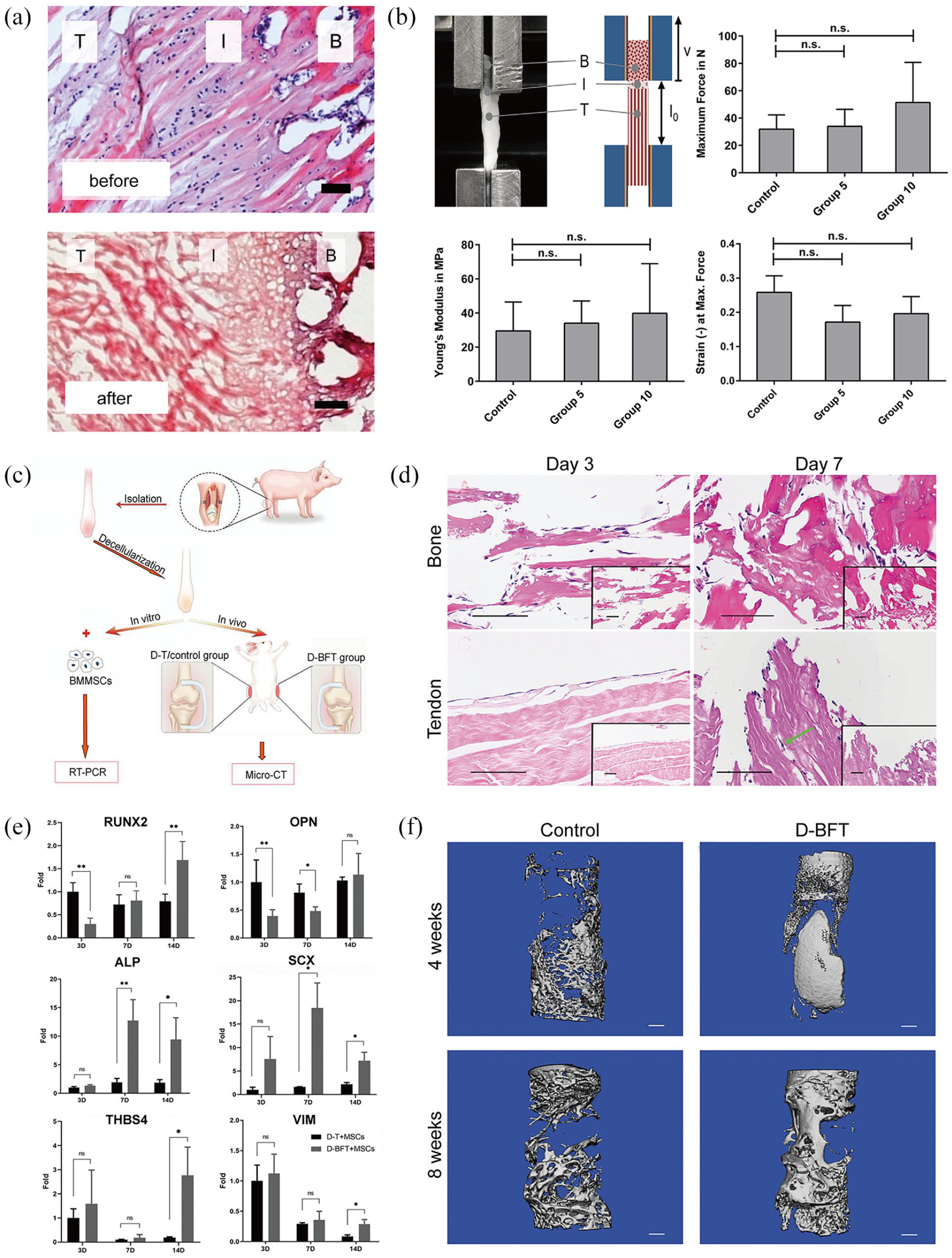
(a) Native enthesis tissue before and after decellularization, 133 (b) The decellularized enthesis scaffold exhibits good biomechanical properties (maximum force, Young’s modules, strain at maximum force), 133 (c) The process of obtaining decellularized enthesis scaffolds, 134 (d) Successful infiltration and proliferation of MSCs in the decellularized enthesis scaffold, 134 (e) Osteogenesis-associated genes (RUNX2, OPN, ALP) and tenogenesis-associated genes (SCX, THBS4, VIM) were upregulated in the decellularized enthesis scaffold, and 134 (f) More tissue was regenerated in the decellularized enthesis scaffold. 134
These experiments indicate that decellularized enthesis scaffolds have enormous potential for use in enthesis TE. However, decellularized scaffolds have limitations. Although the harvest of raw enthesis tissue is not difficult, decellularization and manufacturing protocols should be improved to increase efficiency. 134 The cellular component of raw enthesis tissue still cannot be removed radically and the structural and biomechanical properties of the enthesis’s ECM may be disrupted during decellularization.133,135 To improve enthesis scaffold decellularization, Shi et al. developed a vacuum aspiration system (VAS), which provides a negative pressure environment that maintains a continuous flow of phosphate buffered saline. Their experiments showed that the VAS considerably improves ECM preservation while minimizing the amount of remnant cells in the decellularized enthesis scaffold. Compared with traditionally-decellularized scaffolds, the mechanical properties of their optimized decellularized scaffold were significantly improved. Moreover, VAS-mediated scaffold decellularization effectively enhanced enthesis regeneration and osteogenic, chondrogenic, and tenogenic differentiation capacities were all elevated in the corresponding zones of this scaffold. 136 Therefore, VAS may significantly improve the production of decellularized enthesis scaffolds. Another key shortcoming of decellularized enthesis scaffolds is that scaffold size and its inner structure are uncontrollable and fixed individually in the different scaffolds. Currently, uniform decellularized enthesis scaffolds cannot be produced and their structures are hard to modulate.
Collagen scaffolds
Biomaterials based on collagen, the main component of the enthesis ECM, are widely used in medicine because of their relatively assured biocompatibility. Collagen biomaterials have been certified by the US Food and Drug Administration for decades and collagen scaffolds have been widely used in TE. 137 Collagen is easily accessible and is present several tissues types, including bone and skin.138–140 In the past, collagen was extracted from bone, tendon and fascia tissues via several steps that included digestion and hydrolysis,141,142 which preserved its amino acid sequence, molecular structure, and biomechanical properties, and therefore its biocompatibility. After extraction, collagen would be dissolved in a buffer solution that was then used to make collagen enthesis scaffolds using various methods.143–155
The electrospun collagen enthesis scaffold, an easily accessible scaffold fabricated using an electrospinning device, is one of the most commonly used TE scaffolds and it effectively supports enthesis regeneration.38,117 Based on the poly(ϵ-caprolactone) (PCL) electrospun nanofiber membrane scaffold fabricated using a simple PCL organic solution, Lin et al. added a collagen I (COL-1) solution into PCL solution and successfully fabricated a new type of a COL-1/PCL hybrid nanofiber membrane scaffold. 156 Mechanical and in vitro experiments, revealed that COL-1 markedly increased the porosity, hydrophilicity, and dissolution rate of the nanofiber membrane scaffold, indicating that COL-1 may improve the cellular affinity and biodegradability of the enthesis scaffold. 156 Moreover, tendon stem/progenitor cells (TSPCs) seeded on this COL-1/PCL hybrid scaffold exhibited a faster proliferation rate and a more widely spread cellular morphology, and highly upregulated several osteogenic differentiation genes, including Col1a1, OCN, RUNX-2, and OCT-4. Experiments in which the blending volume ratios of COL-1 and PCL solution were adjusted to fabricate different scaffolds revealed that the beneficial effects mentioned above were better in the hybrid nanofiber membrane scaffold made from a 1:2 (v/v) COL-1/PCL solution (Figure 4(a)–(d)). 156 Together, these findings highlight collagen as an ideal biomaterial for enthesis regeneration, and show that the electrospun collagen enthesis scaffold may simultaneously promote regeneration in Zone I and IV. Apart from electrospun collagen enthesis scaffold, freeze-dried collagen enthesis scaffold is frequently applied in enthesis tissue engineering (TE). Through tuning the collagen solution temperature, the solvent (water) is first frozen to successfully segregate the dissolved collagen and the solvent, the mixture is freeze-dried to remove the ice crystal and then a porous spongy-like collagen enthesis scaffold is generated. By adjusting the concentrations of the collagen solute or choosing different manufacturing protocols, both the inner structures and the overall mechanical properties of the collagen scaffold can be artificially controlled.157–159
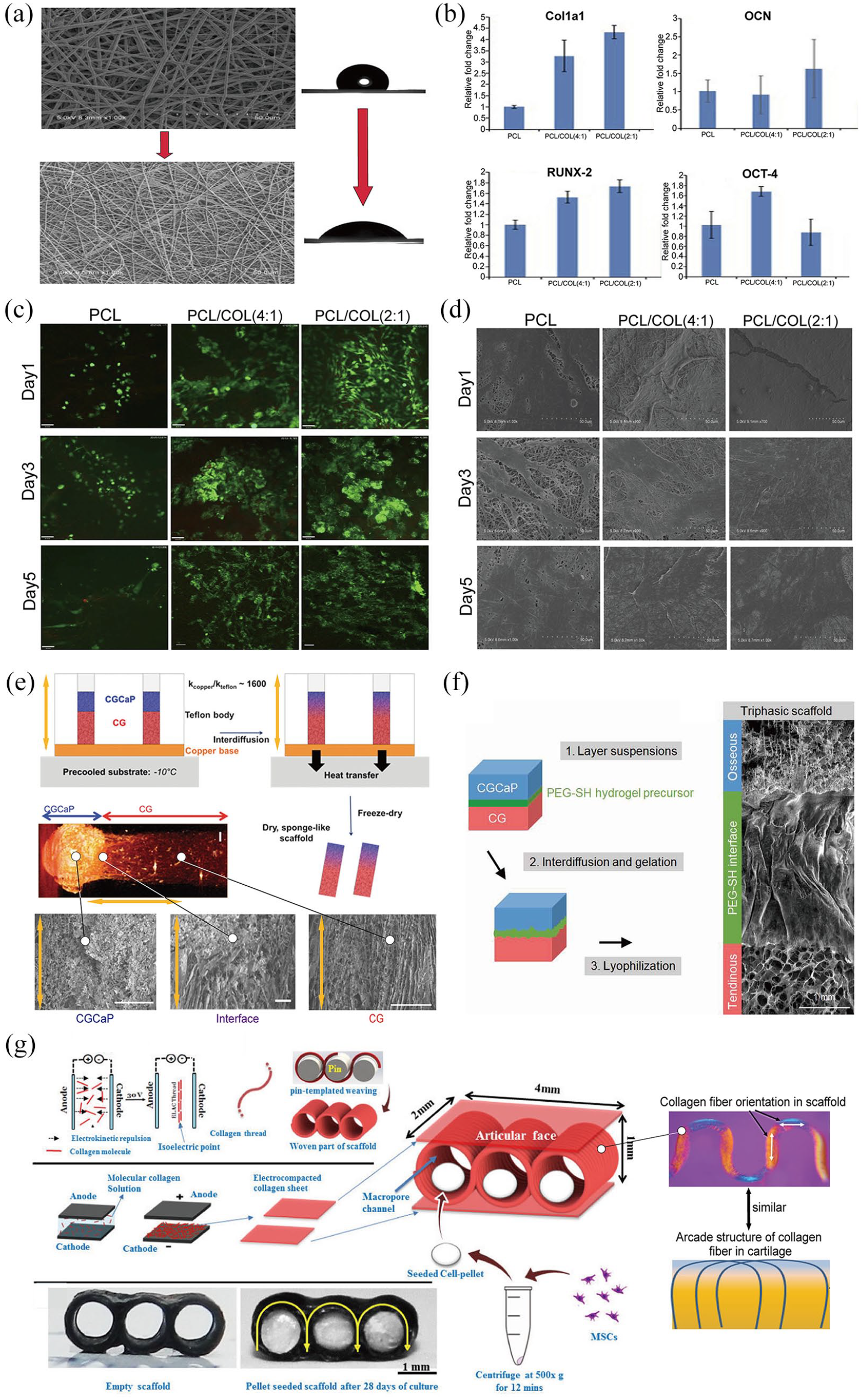
Addition of collagen material increased the porosity and hydrophilicity of the electrospun PCL scaffold (a), upregulated the osteogenic-related genes (Col1a1, OCN, RUNX-2, OCT-4) (b), enhanced the proliferation rate of TSPCs seeded in the scaffold (c) and conferred a widely-spread morphology (d). 156 (e–g) The different biomimetic collagen enthesis scaffolds.160,161,165
Based on this freeze-frying fabricating process, Caliari et al. designed and manufactured a freeze-dried biphasic collagen and a GAG (CG)/GAG and Calcium phosphate (CGCaP) enthesis scaffold. The scanning electron micrographs (SEM) of this biphasic enthesis scaffold demonstrated that the pores in the non-mineralized CG layer were longitudinally aligned and regular (corresponding to Zone I), while the pores in the mineralized CGCaP layer were relatively more isotropic and random (corresponding to Zone IV). Besides, at the interface between the two layers, the transforming collagen trabeculae were smooth and continuous (corresponding to Zone II and Zone III) (Figure 4(e)).
160
All the structures generated in this scaffold perfectly mimicked the native composition of the enthesis tissue.
Besides the above scaffolds, the utility of electrochemical collagen enthesis scaffold in enthesis TE is also remarkable. After application of an electric field to the collagen solution, there is electrolysis of water which creates a pH gradient from the anode to the cathode. Thereafter, the dissolved collagen molecules slowly migrate and assemble at the isoeletric point (pI) region. Finally, simple collagen threads, sheets or membranes are generated, which are ulteriorly fabricated into more complex scaffolds.162–164 Using this convenient fabricating process, Younesi et al. produced a plate-arcade-plate composite electrochemical collagen scaffold to emulate natural interdigitated arcade structure of the cartilage (Figure 4(g)). After seeding MSCs into the scaffold and culturing them for different periods, the studies showed that both the cell number and the scaffold weight gradually increased. H&E and Masson-trichrome staining showed that cartilage ECM slowly filled the scaffold pores with time. On the other hand, Safranin O staining demonstrated that the GAG components were produced by day 14 and were more abundant on day 28. In addition, immunostaining assay showed high generation of collagen type II and aggrecan molecules, which are the two major compositions of cartilage tissue. Moreover, Young’s modulus of the cell-seeded scaffold increased to around 60% to a cartilage-nearing value of 1.33 ± 0.37 MPa. These findings demonstrated that the composite electrochemical collagen scaffold had enormous potential for cartilage regeneration, and it could be applied in enthesis TE to help regenerate the chondral parts of enthesis tissue, which correspond to the Zone II and Zone IV. 165
Unlike the decellularized enthesis scaffold with a fixed collagen layout, the collagen enthesis scaffold is fundamentally fabricated with collagen molecules and its fibers in different methods, which has a higher degree of modulability of the scaffold inner architectures. Because the artificial collagen fibers possess excellent stiffness and toughness, the collagen enthesis scaffolds often have advanced mechanical properties. 166 Besides, since the natural collagen molecules are kept unchanged in the whole process, some cell adhesion signal targets in the collagen can be preserved, which explains why collagen scaffolds could promote cellular adhesion and proliferation as well as effective enthesis regeneration.3,117,167–169 Different collagen enthesis scaffolds may target on the different regions of enthesis tissues and exert different bioeffects on enthesis regeneration. Therefore, it is feasible to hybridize these scaffolds to fabricate a more omnipotent enthesis scaffold, such as the hybridization of electrochemical collagen enthesis scaffold and electrospun collagen enthesis scaffold.
Although collagen enthesis scaffold exhibits many advantages with relatively clear functions on enthesis regeneration, collagen is mostly extracted from experimental animal tissues (rat tail or bovine Achilles tendons) or the donor’s tissues.170–172 Before being further processed and fabricated into scaffolds, collagen needs preconditioning to inactivate the xenoantigen or alloantigen. Collagen extracted from other animal species require mandatory preconditioning processes for biocompatibility adaptation.173,174 Besides, the process of sterilization for collagen is also necessary to eliminate potential pathogens. However, it is still difficult to fully eliminate the antigens and pathogens without damaging the collagen molecules.175–177 Another challenge with collagen enthesis scaffold is the degradation rate. After being implanted, the structures and the biomechanical properties of collagen enthesis scaffolds inevitably deteriorate in a short time, which severely impacts its actual biofunctions.178–180 Therefore, if the biocompatibility and the degradation rate of collagen enthesis scaffold could be improved, its applications for enthesis regeneration will become more widespread.
Silk scaffolds
Natural silk is a protein polymer produced by insects such as silkworms, scorpions, and spiders. Out of all the available silks, silkworm silk (Bombyx Mori silk), is the most widely used in the field of TE.181–183 A key component of silk material is the silk fibroin (SF). There are at least two major types of fibroins in the silk composition, which are referred to as the light chain (25 kDa) and the heavy chain (325 kDa). These two core biocompatible fibroin molecules constitute the composite silk fibers characterized by an anti-parallel β-sheet crystal conformation in space which confer the ideal mechanical properties to the silk material. Besides, a glue-like sericin protein coat binds the SF to reinforce the molecular structure of the silk fibers. 184 Before being utilized in the fabrication of the biological scaffold, the silk should be fully degummed, dissolved, dialyzed, and then centrifuged from the SF solution, which is the common source of material for silk scaffold fabrications.184–186 Silk is a biocompatible material with ideal mechanical properties, and to uncover the possible applications of silk material in enthesis TE, there have been many attempts on silk enthesis scaffold to explore the influence on enthesis regeneration.
For instance, in 2021, Chen et al. successfully manufactured a gradient-mineralized electrospun silk enthesis scaffold. Briefly, a nanofibrous silk membrane was fabricated by an electrospinning device, and then the silk membrane was placed vertically in a 10× simulated body fluid (SBF) to produce a spatial decrease in mineralization degree from the bottom to the top of the silk membrane (Figure 5(a)). SEM images showed a regular topology from the bottom to the top of the scaffold, and the energy-dispersive X-ray spectroscopy (EDS) and Fourier transform infrared spectroscopy (FTIR) analyses further confirmed the existence of the gradient mineral deposition. These experiments demonstrated that the scaffold was highly like the native structure of the enthesis tissue. Next, the osteochondral inductivity of this gradient-mineralized silk enthesis scaffold was fully analyzed. Compared to the no-mineralized silk scaffold, the gradient-mineralized silk enthesis scaffold could simultaneously promote both bone and cartilage tissue growth. Specifically, the area with higher degree of mineralization had larger osteogenic differentiation inductivity, while the area with lower degree of mineralization had more chondrogenic differentiation inductivity (Figure 5(b)). Due to these unique characteristics, the gradient-mineralized silk enthesis scaffold can be applied to restore the natural composition of enthesis tissue, which could fuel the regeneration of Zone II, Zone III and Zone IV. 187
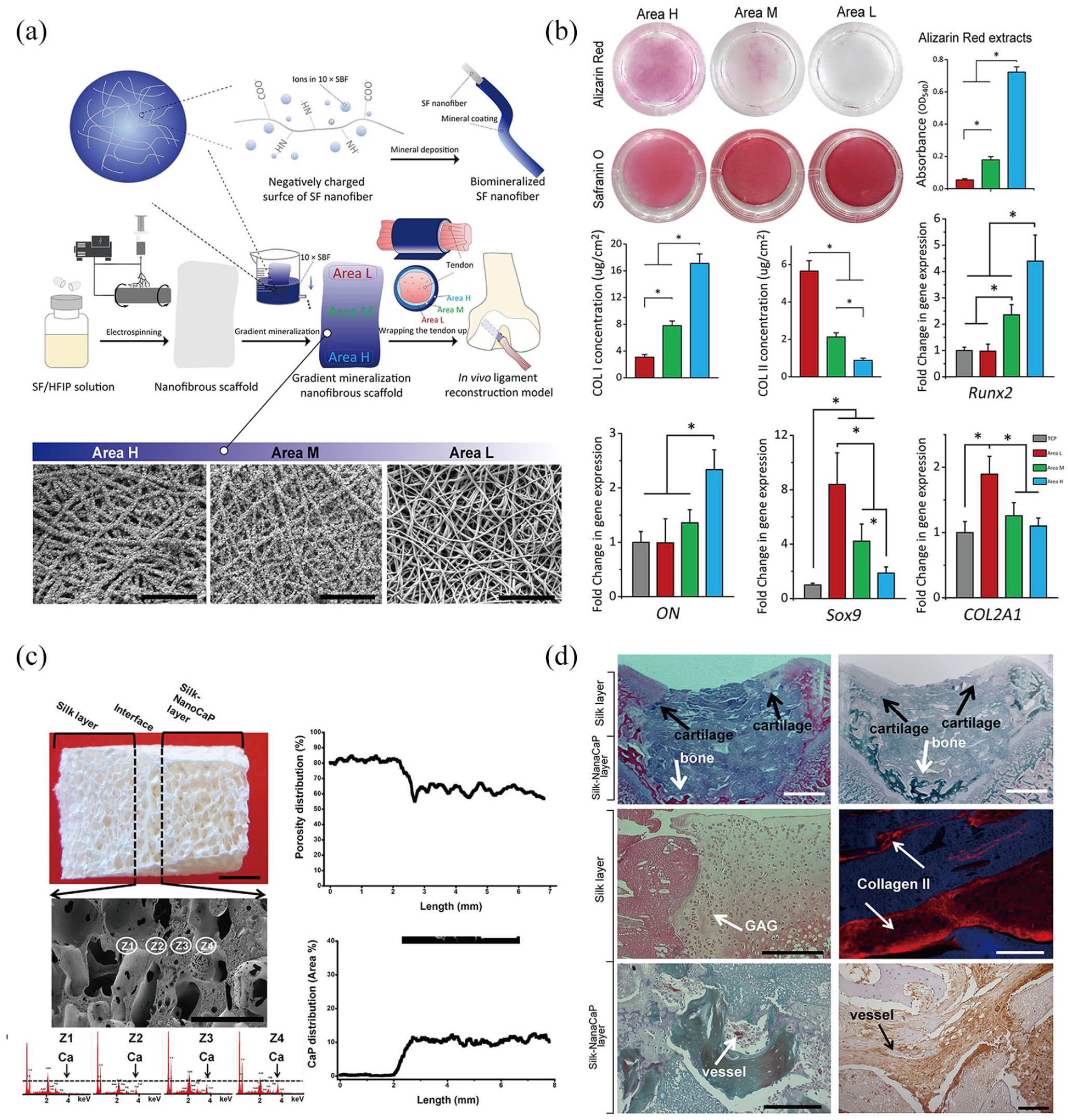
(a) The fabrication and structural layout of a gradient-mineralized electrospun silk enthesis scaffold. 187 (b) In the gradient-mineralized scaffold, areas with a higher degree of mineralization had more osteogenic differentiation inductivity (shown by Alizarin Red staining and the absorbance of staining extracts; collagen I concentration of cells seeded on the scaffold; gene expression levels of RUNX2 and ON) and lower chondrogenic differentiation inductivity (shown by Safranin O staining; collagen II concentration of cells seeded on the scaffold; gene expression levels of COL2A1 and Sox9). 187 (c) The structural characteristics of the Silk/Silk-nanoCaP bilayered salt-leached silk scaffold. 189 (d) Chondrogenesis capacity is higher in the Silk layer. Osteogenesis and angiogenesis capacities are higher in the Silk-nanoCaP layer. 189
Besides trying electrospun silk enthesis scaffold in enthesis TE, there are attempts to explore salt-leached silk enthesis scaffold for use in enthesis regeneration. Although the manufacturing protocols are diverse, the overall fabricating processes of the salt-leached silk enthesis scaffolds are nearly the same. Briefly, porogen particles (such as sodium chloride) are first added into the SF solution, soaked and dissolved in water, and then a porous silk scaffold is successfully leached out. 188
Based on the fabrication process of salt-leaching, Yan et al. manufactured a Silk/Silk-nanoCaP bilayered salt-leached enthesis scaffold. The whole scaffold had an amorphous conformation, which presented a special macro/microporous interconnective topology that highly mimicked the enthesis ECM. At the interface of the two layers, the transformation on the CaP content and scaffold porosity were gradient, and these two different layers proved to be perfectly integrated, which corresponded to the native region of Zone II and Zone III (Figure 5(c)). Both
In addition to the electrospun and salt-leached silk enthesis scaffolds, freeze-dried silk enthesis scaffold is another potential silk scaffold in enthesis TE. The overall fabricating process resembles that of the freeze-dried collagen scaffold, and a spongy-like porous silk scaffold can also be generated, which contains a splendid ECM-biomimetic inner architecture. The pore sizes of the freeze-dried silk scaffold usually vary from tens to hundreds of microns, and within reasonable range of modification, the higher freezing temperature can cause a larger pore size, demonstrating an ideal modulability.194,195
Based on the freeze-drying theories, Sang et al. designed a novel type of amorphous silk porous scaffold and fabricated it to explore its possible biofunction in angiogenesis of soft tissues. Through tuning of different freeze-drying temperatures from −20°C to −5°C, the researchers successfully produced a sequence of amorphous silk porous scaffolds. The amorphous silk porous scaffolds had similar ECM-mimicking inner microstructures, and analysis of scaffold characteristics revealed that the scaffold generated at the higher temperature had a slower degradation rate and a larger compressive modulus, which corresponded to better stability and better mechanical properties, respectively (Figure 6(a)). Furthermore,
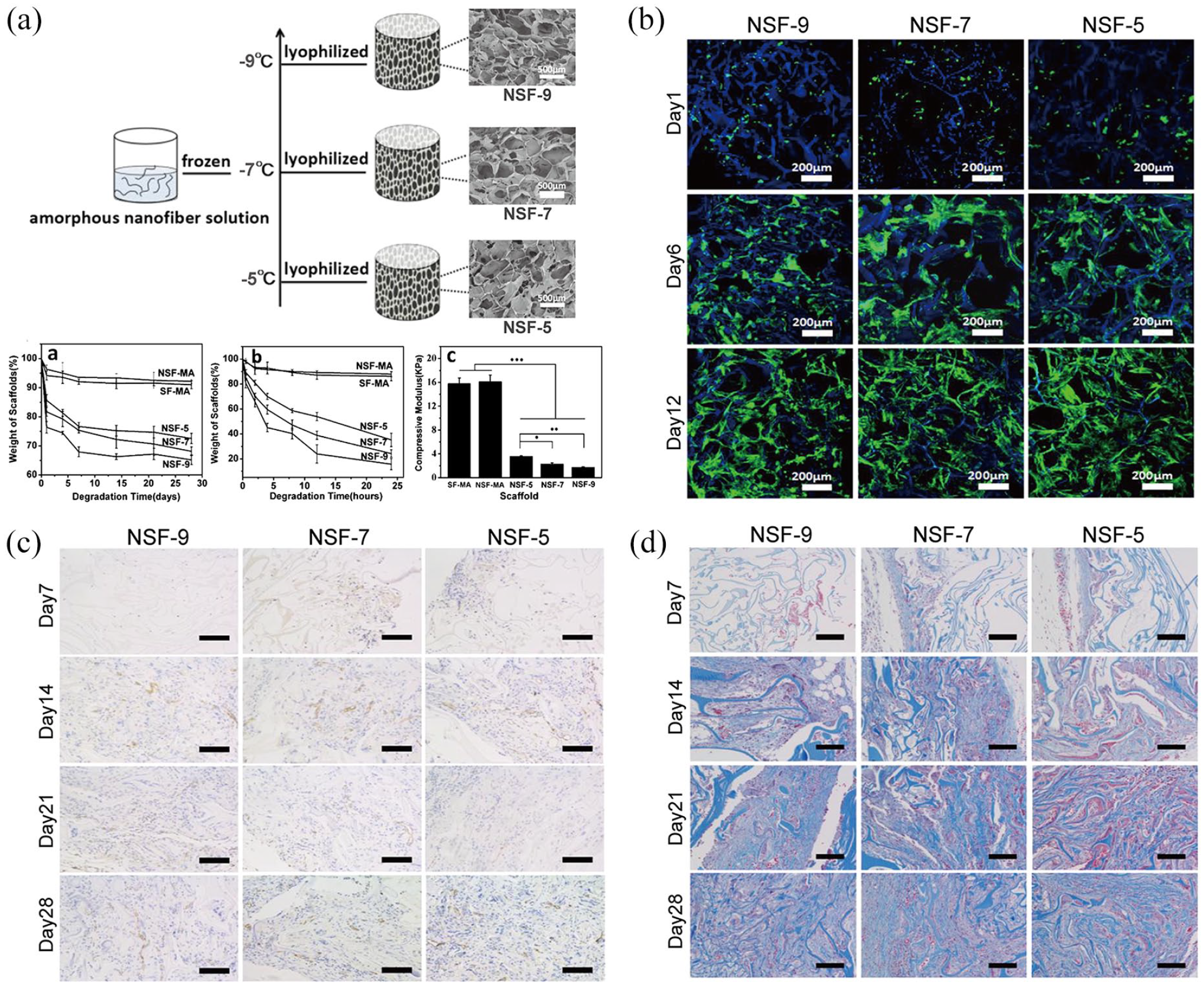
(a) A sequence of insoluble amorphous silk enthesis scaffolds freeze-dried at different temperatures and their mechanical properties. 196 The scaffold produced at a higher temperature has faster cell proliferation rate (b), larger neovascularization capacity, (c), and more regeneration of collagen content (d). 196 NSF-9, insoluble scaffolds derived from amorphous silk nanofiber solution and frozen at −9°C; NSF-7, insoluble scaffolds derived from amorphous silk nanofiber solution and frozen at −7°C; NSF-5, insoluble scaffolds derived from amorphous silk nanofiber solution and frozen at −5°C.
In summary, silk enthesis scaffold is an ideal kind of a biological scaffold with great mechanical properties in enthesis TE. The properties include good strength, great toughness, and excellent elasticity, good biodegradability and bioresorbability. Besides, its main component, silk fibroin can be easily digested by proteases, yielding degradation products including amino acids or some small peptide molecules, which can directly be absorbed by body tissues. After being implanted, silk enthesis scaffold is totally degraded and absorbed within a year with the participation of macrophages. 197 However, the silk enthesis scaffold is still limited by few challenges which restrict its applications. Compared with the silk fibroin, the element of sericin is far less compatible with the human immunosystem which could induce Type I allergic reaction in human bodies.198,199 Although majority of the sericin component in silk material was removed in the preparation process of the SF solution, there could still be few sericin remnants which could act as an allergen in the enthesis scaffold.198,199 Moreover, in the silk scaffold fabrication process, organic solvents or some harmful materials may inevitably be residual, which may increase the toxicity of the silk scaffold. 200 Therefore, there is a need to develop strategies to circumvent these problems associated with the application of silk enthesis scaffold in enthesis TE.
HA hydrogel
Apart from the above-described collagen and silk, the Hyaluronic acid (HA) biomaterial also draws a lot of attention in enthesis TE. HA is a subunit of GAGs, which has a relatively looser molecular structure and a weaker mechanical property, thus it is mainly made into hydrogel and appended into other types of scaffolds. HA is a widespread molecule in various extracellular matrices of our human bodies, which is also an essential component of the enthesis tissue. It can reinforce molecular connection of collagen fibers, increase histological viscoelasticity, and can function as a lubricant in collagen fibers to decrease friction and occurrence of fiber breakage. 201 Therefore, incorporation of enthesis scaffold with HA could increase its mechanical properties and endurance. Frizziero et al. applied a repeated HA injection into the peri-Patellar tendon region of a detrained rat, and showed that HA could increase collagen I content, decrease collagen III content and could have a positive effect on maintenance of the enthesis structure from detraining-associated damage. 202 Furthermore, HA is also an anti-inflammatory biomolecule. Addition of this element could lead to alleviation of the inflammatory response intrigued by the implanted scaffold, thus promoting the scaffold biocompatibility and exerting a positive effect in enthesis regeneration.203–207 Therefore, the HA hydrogel might be another promising natural biomaterial useful in enthesis TE.
Polyolefins and Poly (urethanes)
Conservative Polyolefins, such as poly tetrafluoroethylene and poly propylene were the initial synthetic polymeric biomaterials used in enthesis TE. Initially, researchers focused on the restoration of the anatomy using the Polyolefin materials. They used these materials to reconnect the tear and gap or to refill the large void defect of the enthesis and thus rebuild the histological completeness. There was no consideration of the cellular ingrowth or tissue regeneration, and the final outcomes did show many poor effects of these bio-inert materials: the maximum bearable tensile forces were weaker, external strengths could easily deform the implanted polymers, deterioration of injured enthesis without any regeneration, and lack of any biodegradability for these biomaterials.208,209 Because of these shortcomings, the Polyolefin materials were rendered obsolete.
Apart from the Polyolefins, researchers have also tried Poly (urethanes) in enthesis TE due to their superior mechanical properties. Poly (urethanes) are polymers synthesized by condensation polymerization reactions between different isocyanates and polyols, which are usually accelerated by some catalysts or activated by UV radiation. Compared with Polyolefins, the tunability of the Poly (urethanes) is slightly higher. By altering the different kinds or ratios of isocyanates and polyols, the molecular architectures and mechanical properties of these polymers become tunable.210–212 The biggest advantage of the Poly (urethanes) is that the polymers have high tensile properties, which are capable of enduring large extensions without rupture. Besides, Poly (urethanes) have good elasticity and shape-memory properties, where they can recover to the primary shapes immediately following withdrawal of the tensile forces applied on the Poly (urethane) materials. 213 Due to these excellent mechanical merits, Poly (urethanes) are ideal synthetic polymeric biomaterials used to mimic the native tendon and ligament tissues. Furthermore, Poly (urethane) scaffolds have better bioactivities, with improved cellular adhesion and attachment on those materials.214,215
Although the overall properties of the Poly (urethane) scaffolds have tremendously increased compared to the conventional Polyolefin scaffolds, these scaffolds are still associated with some disadvantages. For instance, the cellular proliferating effects on their surface were not very satisfactory, and the cells seeded on Poly (urethane) scaffolds often have slow proliferation rates.216,217 Moreover, the hydrophilicity of the Poly (urethane) scaffolds is not very well, and their biodegradability is still insufficient. Besides, the overall degradation rates of the Poly (urethane) scaffolds are quite slow: some
Poly (esters)
More investigations have unearthed more advantageous groups of synthetic polymeric biomaterials, and Poly (esters) are presently the most widely used biomaterials in enthesis TE. Poly (esters) are synthetic polymers whose monomers are interconnected by ester groups produced from esterification reactions. In theory, Poly (esters) are hydrolysable due to existence of the ester groups. However, in practice, only a few kinds of Poly (esters) (e.g. the aliphatic Poly (esters)) possess better biodegradability and are capable of being used as biological scaffolds. 221 As of now, the Poly (esters) frequently-applied in TE are PLA (Polylactic acid), PGA (Polyglycolic acid), PCL (Poly(ϵ-caprolactone)), and PLGA (Polylactid-co-glycolid acid), and they are often fabricated into various scaffolds in form of Poly (esters) fibers.222–225
Among the Poly (esters), PLA draws our biggest attention because of its superior biocompatibility. PLA is a simple monomer synthesized organic polymeric biomaterial, which is only composed of lactic acid (LA) molecules. Since LA is in the form of either L (levogyrate) or D (dextrorotatory) isomer, PLA can be further divided into PLLA (poly-L-lactic acid), PDLA (poly-D-lactic acid) or PDLLA (poly-D,L-lactic acid), whose molecular architectures and mechanical properties are slightly different.226–233 As the main degradation products of PLA, LA is one of the most critical energy substrates besides glucose and aliphatic acids. These small molecules can be easily transported across the cytomembrane into the cellular plasm and participate in cellular anaerobic respiration.234–238
Based on its biosafety and biocompatibility characteristics, PLA is a superior biomaterial for enthesis TE applications. Indeed, Uehlin et al. found that aligned electro-spun PLLA nanofiber mats whose surfaces had been treated via controlled NaOH hydrolysis were a feasible scaffold for both cell adhesion and proliferation of hMSCs. This NaOH-treated PLLA scaffold had more enhanced osteogenic differentiation of hMSCs than untreated scaffolds. Furthermore, soaking this PLLA scaffold in bovine thrombin and fibrinogen not only added an extra layer of fibrin to its surface but also subsequently generated more connective topological structures within it resulting in decreased mobility of the PLLA fibers. This PLLA/fibrin hybrid scaffold had good mechanical properties, and due to the additional fibrin layer, it had an increased capacity for osteogenic differentiation (Figure 7(a)).
239
Thus, it is possibly applicable in enthesis TE as it can contribute to the mineralization process of enthesis regeneration. Relatedly, Li et al. fabricated a dual-layer organic/inorganic bipolar PLLA enthesis scaffold with an electrospinning device. The layers were from pure PLLA and nano-hydroxyapatite-PLLA fibers to simulate the non-mineralized and mineralized enthesis regions respectively (Figure 7(b)). Their simulated body fluid (SBF) mineralization-inducing
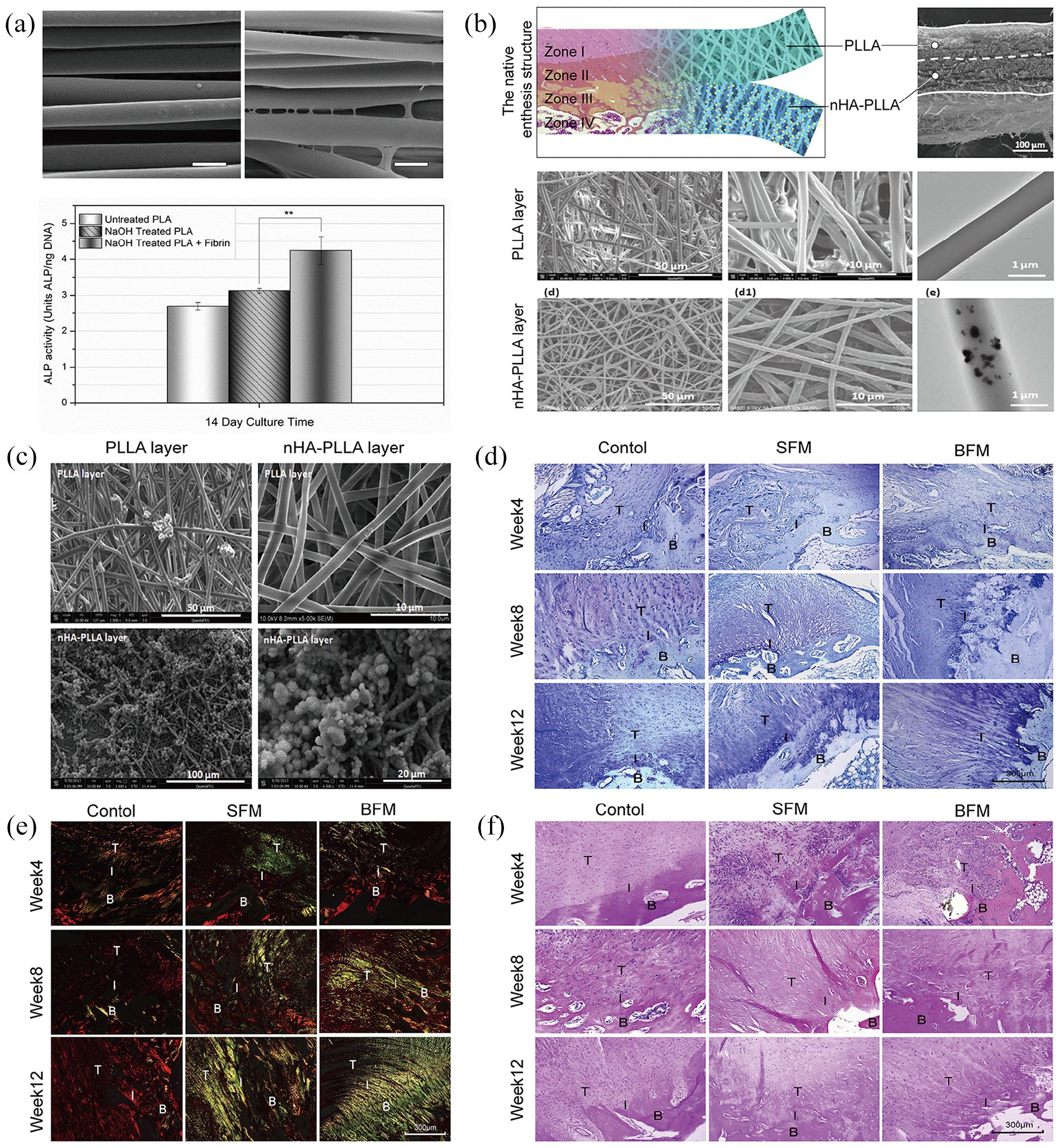
(a) With an extra layer of fibrin coated, more connective topological structures were generated, and the osteogenic differentiation capacity was elevated compared to the non-coated PLLA scaffold. 239 (b) The structure of bipolar PLLA/nHA-PLLA enthesis scaffold, 240 (c) The nHA-PLLA layer had the larger mineralization capacity compared with the PLLA layer. 240 The elevated GAG formation (d), collagen formation (e), and enthesis tissue regeneration (f) in the dual-layer enthesis scaffold were proved via the histological stainings. 240
Apart from PLA, other biodegradable materials used in enthesis regeneration are PGA, PLGA, and PCL. Low molecular weight PCL debris are safe, and permeate blood microvessels for transportation to the kidney and liver, from where they are excreted via urine and bile respectively.241,242 For both PGA and PLGA, glycolic acid is the main degradation product, which is then oxidized to glyoxylic acid that enters the human tricarboxylic acid cycle. Therefore, these biodegradable biomaterials are applicable in enthesis TE. Cao et al. manufactured a PCL/PCL-tricalcium phosphate (TCP) /PCL-TCP multi-layered porous scaffold via a three-dimensional (3D) printing device, which mimicked the natural tendon-fibrocartilage-bone histological multilayers of the enthesis tissue (Figure 8(a)). This scaffold was designed with three different layers that corresponded to the composition and thicknesses of Zone I, Zone II-Zone III, and Zone IV of native enthesis tissue and thus primarily giving it biomimetic characteristics. Indeed, it promoted fibrogenesis and chondrogenesis, cell proliferation, and ECM deposition that was spatially distinct among the three layers (Figure 8(b)). 243 Romeo et al. implanted a PGA/poly-L-lactide-co-ε-caprolactone (PLCL) nanofiber scaffold at the rotator cuff tendon enthesis site of a sheep, and after a period of 12 weeks, the native enthesis tissue structures successfully recurred in the scaffold-treated group. 42 Relatedly, Smith et al. fabricated a porous PLGA scaffold via salt-leaching and etched it with NaOH to modify its surface topography. The resultant scaffold had higher porosity and rougher nano-surface topography than unetched PLGA scaffolds, and this corresponded to the typical tissue structure of Zone IV (Figure 8(d)). 244 The adhesion of osteoblasts on it significantly increased, whereas that of fibroblasts decreased (Figure 8(e)). Thus, this special NaOH-etched PLGA scaffold is likely to improve Zone IV regeneration whereas the unetched PLGA scaffold would be beneficial for regeneration of Zone I in enthesis TE applications —fabricating a curative multi-layered PLGA scaffold for enthesis regeneration is a worthwhile venture. 244 Furthermore, Erisken et al used a new processing method of twin-screw extrusion/electrospinning (TSEE) to fabricate a special PCL scaffold with a continuously-increasing amount of β-tricalcium phosphate in its inner structure that was similar to the native structures of Zones II and III of enthesis tissues. 245 Using the same method, they fabricated a similar insulin/PCL/β-glycerophosphate scaffold, which contained simultaneous gradients of increasing and decreasing insulin and β-glycerophosphate respectively. Its insulin- and β-glycerophosphate-rich regions had high capacities for chondrogenesis and osteogenesis respectively. 246 Thus, this scaffold is also applicable in enthesis TE—it would predominantly effectively enhance regeneration in Zones II and III.
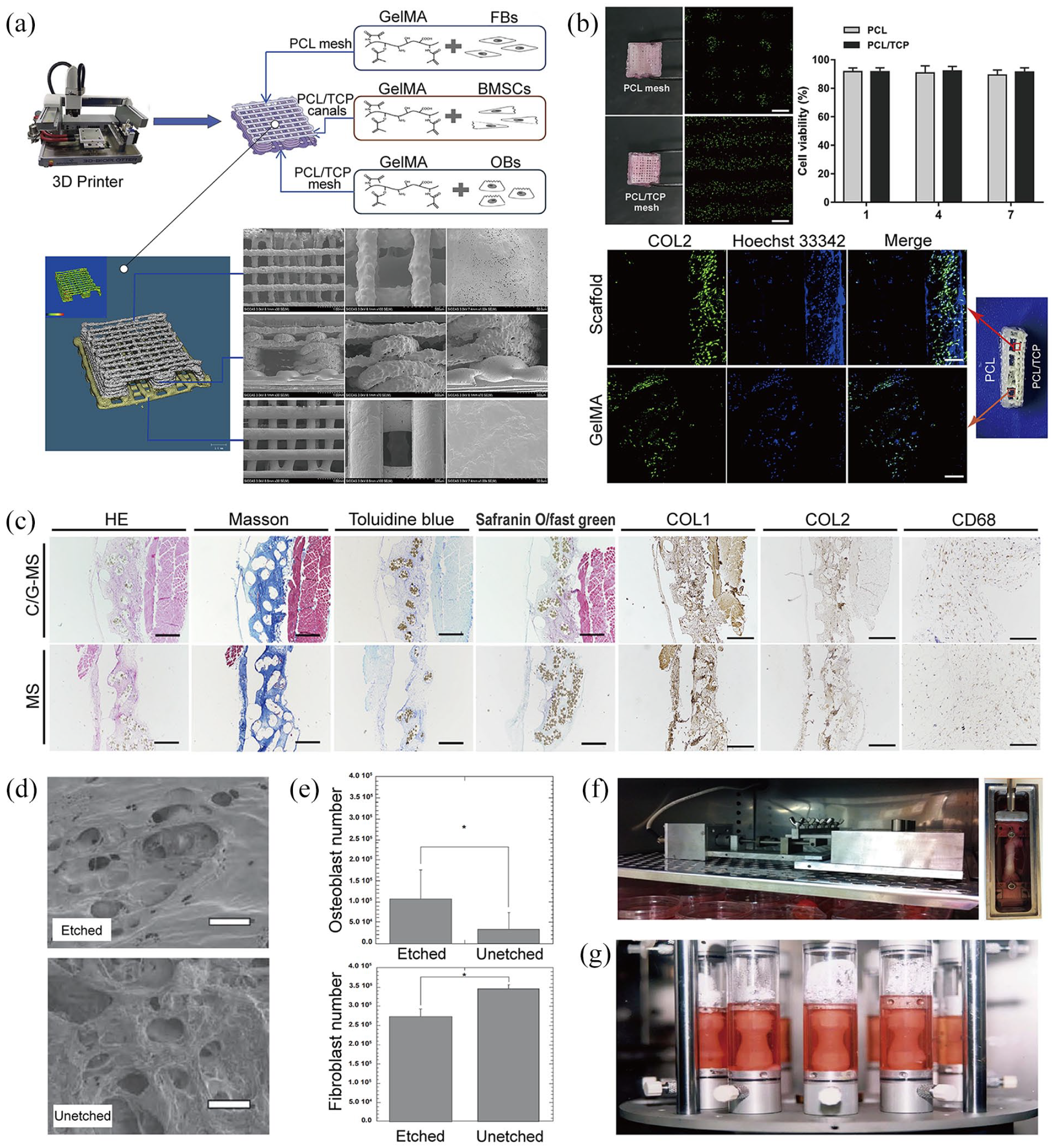
(a) A triphasic 3D-printed enthesis scaffold mimicking the natural tendon-fibrocartilage-bone structure of enthesis tissues. 243 (b) The cell proliferation and ECM deposition were high in the PCL/TCP layer. 243 (c) Histological stains showing improved regeneration of enthesis tissues due to the 3D-printed enthesis scaffold, and elevation of positive effects due to loading with GelMAs (containing either fibroblasts, BMSCs, or osteoblasts). 243 (d) After NAOH etching, the PLGA scaffold had high porosity and rough nano-surface topography that both corresponded to the tissue structure of Zone IV (Bone). 244 (e) The adhesion of osteoblasts increased, whereas that of fibroblasts simultaneously decreased in the NAOH-etched PLGA scaffold. 244 (f and g) The two different kinds of bioreactors that were used to apply cyclical stress stimulations on tendons.274,275
Taken together, synthetic polymeric scaffolds can be effectively applied in enthesis TE as they have higher degrees of tunability and reproductivity, and better biosafety profiles—since they lack potential inherent antigens/pathogens—than naturally-sourced enthesis ones.108,247,248
Biological strategies are considered a most critical aspect of enthesis TE. For example, the use of biological scaffolds provides a biomimetic platform that gives a bioactive environment for the regeneration and infiltration of the injured enthesis tissue. Thus, enthesis scaffolds should be designed and fabricated with either three layers that correspond to the tendon, fibrocartilage, and bone or four layers that correspond to the tendon, unmineralized fibrocartilage, mineralized fibrocartilage, and bone. Moreover, to better emulate the native structures of Zones II and III, enthesis scaffolds with inner structures and topologies that have gradients are indispensable.
Cell strategy
Enthesis-related cells are applicable in promoting enthesis regeneration and this is the basis of cell strategies in enthesis TE. Such cells include osteoblasts, fibroblasts, costal-cartilage-derived stem cells (CDSCs), tendon stem/progenitor cells (TSPCs), adipose-derived stem cells (ADSCs), and mesenchymal stem cells (MSCs). The specific effects of these cells in enthesis TE have been previously investigated.
Cooper et al. seeded opposite ends of a commercially purchased PLA scaffold with both osteoblasts and fibroblasts which were then co-cultured. The GAG and collagen contents of the ECM in the fibroblasts, osteoblasts, and transition regions were then assessed, and they were generally higher in the fibroblast region—the transition region between the two cellular regions mostly had intermediate values of the GAG and collagen contents at different time points. Thus, fibroblasts promoted effective ECM deposition on the enthesis scaffold and co-culturing fibroblasts with osteoblasts contributed to the formation of the natural gradient of the enthesis ECM. 249
Zuo et al. successfully harvested CDSCs from the costal cartilage tissue of newborn Sprague-Dawley rats and showed that these cells had ideal proliferation abilities and cell viability under low oxygen and nutrient
To elucidate possible functions of TSPCs in the regeneration of enthesis, Shen et al. produced a knitted silk/collagen sponge scaffold seeded with additional TSPCs and then transplanted it into a defective rabbit rotator cuff tendon enthesis. More fibroblast growth, less lymphocytes infiltration into the regenerated enthesis tissue and scaffold, and denser deposition of ECM and collagen were observed in the TSPCs-seeded scaffold than in the control. Moreover, nascent collagen fibers were more continuous and homogeneous, the interfaces between scaffold and enthesis tissues were more aligned, and higher overall mechanical properties in regenerated enthesis tissues were observed in the TSPCs-treated group than in the control. Relatedly, the expression level of fibrogenic genes (collagen I, collagen III, DCN) in TSPCs-treated scaffold was significantly upregulated, corroborating the positive effect of TSPCs on the fibrogenesis in enthesis tissue regeneration. As lymphocyte infiltration was inhibited in this scaffold, TSPCs possibly have an anti-inflammatory effect during regeneration of enthesis. 250
For ADSCs, McGoldrick et al. seeded a decellularized-enthesis scaffold with ADSCs and then used it—or a blank enthesis scaffold without ADSCs seeded—to reconstruct rat Achilles tendon enthesis tissues. The regenerated Achilles tendon enthesis in the cell-seeded group had superior biomechanical properties—ultimate failure load, ultimate tensile stress, stiffness—more tendon and fibrocartilage tissues within the scaffold, and more collagen type III in the extracellular matrices than the unseeded group. Thus, ADSCs possibly promoted enthesis regeneration in injured enthesis tissues via fibrogenesis and chondrogenesis processes. 251 In a related study, Zhao et al. co-cultured ADSCs with injured tenocytes for 48 h, ADSCs clearly promoted proliferation rates and largely reduced levels of oxidative stress in the injured tenocytes via inhibition of methylation LncRNA Morf4l1. 252 Therefore, it is hypothesized that ADSCs mainly target the regeneration of Zone I in the injured enthesis tissue, and exert their positive effect through LncRNA Morf4l1 demethylation.
Due to their totipotency, MSCs are not only widely medically utilized but are also specifically applied in repairing injured enthesis tissues, where a complex and simultaneous regeneration of multiple tissues is required. Nourissat et al. cut off a rat Achilles tendon and destroyed its enthesis. The free end of the tendon was then attached to the left bony tunnel with the fixation of sutures. They then either injected chondrocytes or MSCs into the repair site and evaluated their effects separately. These cell therapies were indeed efficient in reconstructing the injured enthesis—MSCs were even better than chondrocytes in improving high-level enthesis regeneration. 253 This was corroborated by Lim et al. who transplanted tendon autografts in rabbit ACL enthesis, and the reconstructed ACL was artificially coated with MSCs in a fibrin glue carrier. MSCs treatment group had larger areas of chondral regenerated in enthesis tissues than the control group, and a gradient transition from the cartilage to bone region was also observed. Thus, MSCs could intervene in the cartilage regenerating process of the injured enthesis tissue, which could greatly promote the maturation of enthesis tissues. 254 Using a 3D-printed triphasic enthesis scaffold, Cao et al. loaded three different gelatin methacrylates (GelMAs) encapsulating fibroblasts/BMSCs/osteoblasts onto the tendon, fibrocartilage, and bone mimicking layers. These applications of additional enthesis cells promoted fibrogenesis, chondrogenesis, and osteogenesis in the different layers of the complex enthesis scaffold (Figure 8(c)). 243 Therefore, MSCs possibly played an important role in the regeneration of enthesis tissues, which mainly acted in the cartilage regions of Zone II and III.
As another key strategy in enthesis TE, cell strategy mainly focuses on the additional applications of enthesis-related cells in enthesis tissues or scaffolds to further improve the regeneration of injured enthesis. Traditionally, enthesis-related cells were directly applied to the injured site of the enthesis tissue. However, the number of these cells, and their concomitant healing effects at the site, decreases with time. Thus, fixing enthesis-related cells on the enthesis scaffold to specifically act on the injured region may ensure longer-lasting effects. Hence, a current challenge in the successful application of cell strategy in enthesis TE is combining enthesis-related cells with the scaffold without diminishing their bioeffects.
Growth factor strategy
Biological factors accelerate enthesis regeneration; more specifically growth factors have been applied in enthesis TE. 34 Currently, commonly-used growth factors include BMPs (bone morphogenetic proteins), FGFs (fibroblast growth factors), TGFs (transforming growth factors), GDFs (growth and differentiation factors), PDGFs (platelet-derived growth factors), and VEGF (vascular endothelial growth factor). The possible functions of these factors in accelerating enthesis regeneration have been investigated.
Hashimoto et al. induced a tendon/ossicle complex by injecting BMP-2 into the rabbit flexor digitorum communis tendon enthesis and subsequently transplanted it onto the rabbit tibia surface. A month later, it had merged completely with the tibia and a direct insertion of a tendon-bone structure was histologically observed, corroborating the positive effect of BMP-2 in enthesis repairing. 255 For FGF-2, Yonemitsu et al. clearly demonstrated that FGF-2 elevated the degree of tenogenic regeneration for chronic rotator cuff enthesis tears in rats. 256 In order to explore the specific function of TGF-β3, Kovacevic et al. applied a calcium-Phosphate matrix—supplemented with TGF-β3—at the surgically-reconnected tendon-bone interface of a rat supraspinatus tendon enthesis. TGF-β3 tremendously enhanced the mechanical endurance as well as the generation of collagen in the repaired enthesis tissue. 257 Relatedly, Holladay et al. showed that the expression of tenogenic-related genes (DCN, SCX, collagen I) in TSPCs was largely upregulated by GDF-5 treatment—this indicated a potential fibrogenic effect of GDF-5 molecules in enthesis TE. 258 To elucidate the role of PDGFs in enthesis TE, Cheng et al. added PDGFs nanoparticles into a biomimetic collagen scaffold and showed that the ADSCs seeded onto this hybrid scaffold had a higher proliferation rate and a higher degree of tenogenic differentiation degree than the control. Thus, PDGFs had a stimulating effect on the regeneration of Zone I in enthesis tissue. 259 Furthermore, Cheng et al. showed that the Mg interference screw promoted enthesis fibrocartilaginous regeneration via VEGF endocellular accumulation, which corroborated the positive effect of VEGF in enthesis TE. 260
The aforementioned factors all promoted enthesis regeneration, albeit via different mechanisms. For example, FGFs mainly target the fibrous region to promote the degree of fibrogenesis in enthesis tissues, whereas BMPs target both the chondral and bony regions to promote chondrogenesis and osteogenesis respectively.255,256 TGF-β1 primarily promoted chondrogenesis in enthesis, whereas TGF-β3 simultaneously promoted fibrogenesis and chondrogenesis.261–263 Therefore, for optimum enhancement of enthesis regeneration, two or more kinds of growth factors should be applied simultaneously, successively, or even iteratively in accordance with the different phases of enthesis healing.130,264,265 Notably, heparin effectively reduced the release rate of TGF-β2 and GDF-5 from the enthesis scaffold and significantly increased their validity, which reinforced their fibrogenesis and chondrogenesis activities during enthesis regeneration. 266 Thus, heparin is a possible general potentiator of the beneficial effects of these growth factors in enthesis TE.
Taken together, the growth factor strategy is key for enthesis TE. Growth factor supplementation promotes the process of enthesis regeneration in injured enthesis tissues. Similar to the cell strategy, the growth factors are applied directly at the injured site of the enthesis tissue, which also reduces the prospects of long-lasting effects. Relatedly, the local injection of growth factors could not only cause an acute blood surge but also have unpredictable effects on normal tissues due to increased blood circulation of growth factor molecules. Therefore, growth factors should also be used in combination with enthesis scaffolds, not only to improve their targeting abilities but also to modulate their release.
Biophysical modulation strategy
Both eccentric and concentric overload training promote enthesis healing in clinical practice.
267
Therefore, biophysical modulation might be a key intervention in the whole healing process of enthesis tissues.268–272 Wang et al. invented a mechanical bioreactor to test the influence of stress stimulation on enthesis tissues
Simple static tensile forces have also been applied on enthesis scaffolds to promote enthesis regeneration. Rinoldi et al. fabricated a loop-like wet-spun gelatin methacryloyl (GelMA)-alginate scaffold encapsulated with BMSCs and then stretched it in the opposite direction with a bespoke stretching device so that the scaffold was subjected to 15% static strain. This static stress stimulation promoted cell proliferation, alignment, and the expression levels of some enthesis-related genes (Collagen Type I, Collagen Type III, SCX, TNMD). 276 Thus, both dynamic and static stress stimulations improved enthesis regeneration, and stress stimulation is key for the biophysical modulation strategy.
The use of electromagnetic fields is another key aspect of the biophysical modulation strategy. Pulsed electromagnetic fields (PEMF) not only improved the early healing of rat rotator cuff enthesis after reconstruction 277 but also promoted osteogenesis at the enthesis site to create a tight tissue connection at the tendon-bone interface. 278 However, there exists a paucity of studies on electromagnetic fields for enthesis TE and its mechanism of action remains to be clarified. That said, the biophysical modulation strategy is most advantageous since biophysical stimulations of the injured enthesis are usually uncomplicated and easily controllable, non-invasive, and repeatable, making it the most economic strategy for prospective clinical applications.
Conclusions and future perspectives
In summary, the enthesis is a special complex tissue that is hard to regenerate. To achieve the successful regeneration of enthesis, the issues of tendon/ligament, cartilage and bone regenerations should all be considered at the same time, and these three kinds of tissues should be regenerated orderly in space corresponding to the composition of enthesis tissue. Due to developments in enthesis TE, four effective strategies (biological scaffold strategy, cell strategy, growth factor strategy, biophysical modulation strategy) have been unveiled so far to promote the regeneration of injured enthesis tissue significantly. And for achieving the best result of enthesis regeneration as we could, these strategies should be applied comprehensively.
Primarily, a special enthesis scaffold with the multifunction of fibrogenesis-chondrogenesis-osteogenesis is needed to be implanted
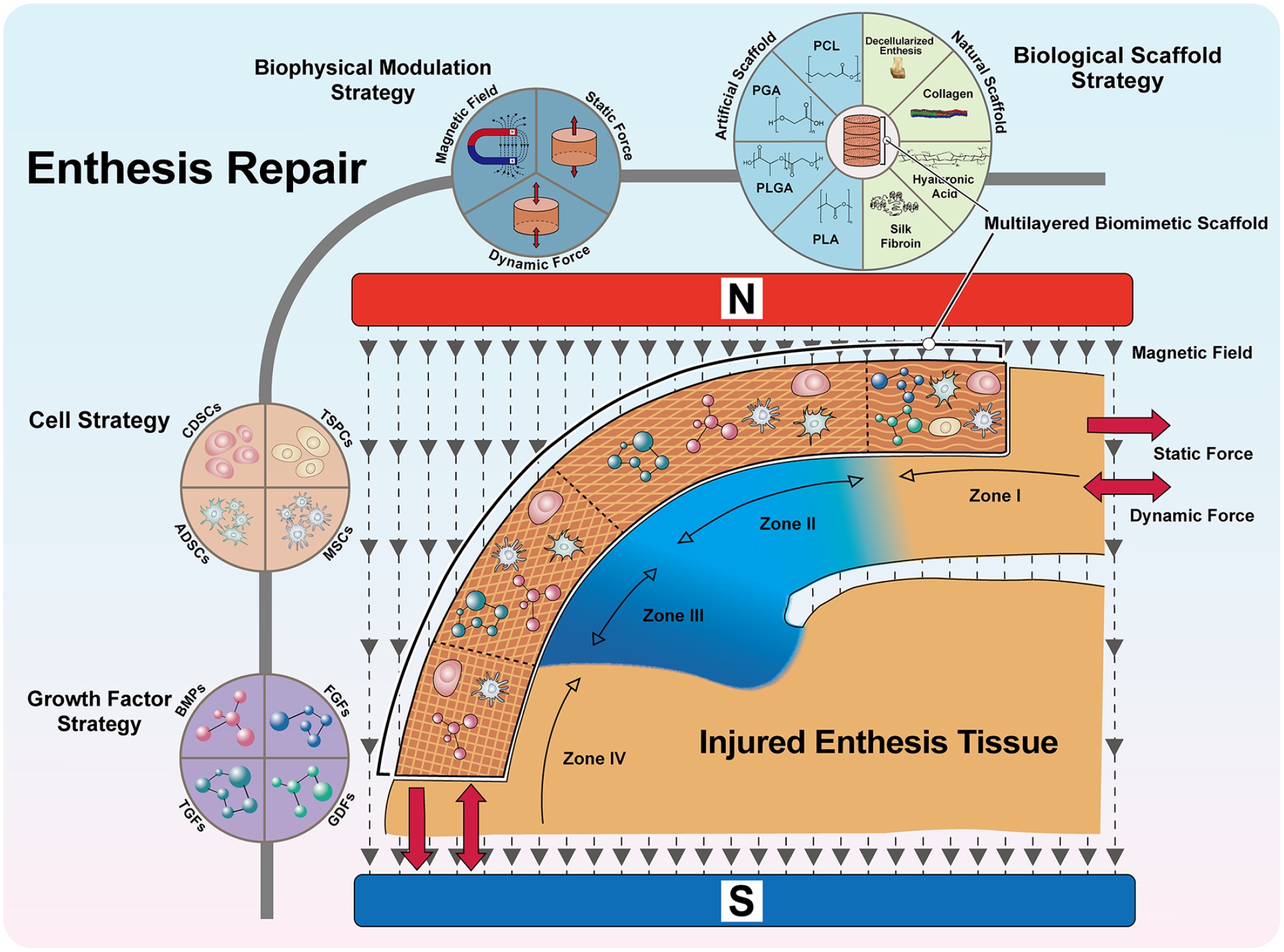
The schematic figure of different strategies’ applications in enthesis TE. To better regenerate the injured enthesis tissue, a multilayered biomimetic scaffold with the biofunction of fibrogenesis-chondrogenesis-osteogenesis should be implanted
With all the strategies synthetically applied, the problem of enthesis regeneration has been improved tremendously in comparison with the traditional surgical treatments. However, there is still no method that results in the complete reemergence of intact and native enthesis tissues, necessitating the more improvements in enthesis TE in the future. As the most important component of enthesis tissue engineering, the biological scaffold strategy mainly focuses on designing, fabricating, and verifying the enthesis scaffold in the different biomaterials. Besides the general criteria of biodegradability, biocompatibility, biosafety and reproducibility for the production of biological scaffold applied in TE, an enthesis scaffold should meet the following criteria additionally:
(a) Enthesis scaffold should be designed with a tendon/ligament-cartilage-bone multiphasic biomimetic structure, and the interfaces between the different layers should preferably correspond to the specific interfaces between the native Zones of enthesis tissue so that the scaffold biomimetic degree can get established. The structural transition in the enthesis scaffold should be distinct and graded in the interfaces between fibrogenic and chondrogenic layers as well as chondrogenic and osteogenic layers, while at the same time, there should also exist a continuous structural gradient in the chondrogenic layer to emulate the natural structure from Zone II to Zone III.
(b) As a special biological scaffold applied at the complex tissue of enthesis, the overall mechanical properties of enthesis scaffold should be optimized to endure the external tensile forces and compressive forces without deformation. The specific mechanical properties of the different layers should also strictly correspond to the native Zones of enthesis tissue, which means that enthesis scaffold should be designed into a soft-hard transited scaffold. The soft terminal corresponds to Zone I, it should have the mechanical properties similar to the tendon/ligament tissue to endure the external tensile forces. The hard terminal corresponds to Zone IV, it should have the mechanical properties similar to the bone tissue to endure both tensile and compressive forces. The immediate part corresponds to Zone II and Zone III, it should have the mechanical properties similar to the cartilage tissue to endure both tensile and compressive forces as well.
(c) Apart from the structural and biomechanical requirements listed above, there is also another requirement about the biofunction of enthesis scaffold. The spatial order of biofunctions in enthesis scaffold should be followed as fibrogenesis-chondrogenesis-osteogenesis strictly. All the choices of biomaterials, cells and growth factors for the synthesis of enthesis scaffold should be based on this requirement so that the primitive enthesis tissue could be regenerated under the induction of enthesis scaffold.
As we know, the proliferation and differentiation of cells can be influenced by the biomaterials on which they are attaching. Therefore, biomaterials that promote fibrocytes/chondrocytes/osteocytes should be used orderly to synthesize a multi-composed enthesis scaffold so that it could promote enthesis regeneration more effectively. In addition, some spatial properties in biological scaffolds (such as the porosity, pore size and the micro-topology) can also influence the cellular behaviors of the different cells and affect fibrogenesis/chondrogenesis/osteogenesis process. If these influencing factors are unveiled and applied in the design&fabrication of enthesis scaffold, the utility of enthesis scaffold in enthesis TE will come to a much higher level in the future.
For the cell strategy in enthesis TE, the MSCs are the most common cells seeded on enthesis scaffold. Due to their multiple differentiation directions, MSCs in different layers of enthesis scaffold may be transformed into corresponding differentiated cell types in an ideal case. However, in actual practice, this regular fibrogenic/chondrogenic/osteogenic differentiation in the intended layer of scaffold is difficult to achieve. In our view, orderly seeding of fibroblasts, chondroblasts, and osteoblasts on different scaffold layers might be a better strategy for promoting enthesis regeneration. What’s more, a combination of stratified enthesis scaffold and stratified cell seeding can enhance the regeneration of enthesis to a large degree.
To date, several biological molecules that promote the regeneration of enthesis have been investigated, especially focusing on growth factors. However, other molecules (e.g. some hormones and gene molecules) have not been researched. Therefore, further studies are needed to test the performance of hormone therapy and gene therapy in enthesis TE. Among the growth factors studied so far, most are used singly in enthesis TE and mostly target a single region of enthesis tissue, although enthesis regeneration involves at least three kinds of tissues (tendon/ligament, fibrocartilage and bone) at the same time. Therefore, we believe that two or more growth factors should be simultaneously laden onto the different layers of the enthesis scaffold to promote the regeneration of the intended tissues at the corresponding layers, hence improve the overall regeneration of the injured enthesis tissue.
Both the dynamic/static stress stimulation and electromagnetic field have been demonstrated as two types of valid biophysical modulation strategies for enthesis regeneration. However, they have not been sufficiently explored, and the methods utilized in the past experiments are have some practical limitations. Similarly, the role of stress stimulation in enthesis TE need to be further refined. The bioreactors used to apply mechanical forces are mostly custom-made without any unified manufacturing standard. In addition, the forces applied have not even been quantified, which makes findings from difference studies incomparable. Moreover, the specific mechanisms behind the positive phenomena of electromagnetic field stimulation in enthesis regeneration have not been fully determined. In future, the most appropriate magnetic field intensity and frequency for enthesis tissue should be tested and specified as early as possible.
In summary, the application of enthesis TE has improved enthesis tissue regeneration. The strategies discussed in this review are expected to promote regeneration of injured enthesis. Moreover, the abundance of the enthesis tissue in vertebra makes it susceptible to damage during avulsion fracture and ankylosing spondylitis. Therefore, understanding the enthesis TE will benefit research on spine-related enthesis TE.
Footnotes
Author contributions
Wangwang Luo: Writing-original draft. Yang Wang: Conceptualization. Qin Han: Revised, Supervision. Zhonghan Wang: Supervision. Jianhang Jiao: Literature collection. Xuqiang Gong: Supervision. Yang Liu: Investigation. Aobo Zhang: Investigation. Han Zhang: Literature collection. Hao Chen: Supervision, Methodology. Jincheng Wang: Supervision, Methodology. Minfei Wu: Investigation, Methodology, Funding support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by
(1) National Natural Science Foundation of China [grant numbers 82272504, 82072456]
(2) National Key R&D Program of China [No. 2018YFB1105100]
(3) Department of Science and Technology of Jilin Province, P.R.C [grant numbers YDZJ202201ZYTS131, YDZJ202201ZYTS129, 202201ZYTS505, 20220204119YY, 20220401084YY, 20210101321JC, 20210204104YY, 20200201448JC, 20200404202YY, 20200403086SF, 20200201453JC]
(4) Department of Finance in Jilin province [grant number 2020SCZT037, 2019SCZT031]
(5) Jilin Province Development and Reform Commission, P.R.C [grant numbers 2018C010 &2022C043-5]
(6) Interdisciplinary Integration and Cultivation Project of Jilin University [grant number JLUXKJC2020307]
