Abstract
The tendon/ligament-to-bone interface has a complex organization to enable transfer of forces through the tendon/ligament to the bone. The purpose of this study is to create a co-culture environment enabling a tissue-specific tendon region and tissue-specific bone region on a degradable scaffold, using NIH 3T3 fibroblast–deposited extracellular matrix and MC 3T3 osteoblast–deposited extracellular matrix, respectively. Before full characterization of the deposited extracellular matrix coating can be analyzed, co-culture parameters including culture medium and seeding technique should be addressed. An appropriate medium formulation was developed to reduce fibroblast to osteoblast mineralization by adjusting beta-glycerophosphate concentrations. Standard growth medium with fetal bovine serum + 3 mM beta-glycerophosphate + 25 µg/mL ascorbic acid was found to be the most suitable formulation evaluated in these study conditions. Seeding and cell migration studies of co-cultured fibroblast- and osteoblast-specific scaffolds were performed to identify whether tissue regions could be created on the scaffold. Fibroblast and osteoblast regions were successfully seeded and little to no cell migration was observed up to 42 h after seeding. Finally, a preliminary analysis of basic extracellular matrix components was measured in the fibroblast, osteoblast, and transition regions. Tissue-specific DNA, glycosaminoglycan, and collagen were found in uniform amounts on the scaffolds and were not different significantly between scaffold regions. In conclusion, initial steps to create tissue-specific fibroblast and osteoblast regions on a degradable scaffold were successful in preparation for further characterization investigations as a tendon-to-bone interface scaffold.
Introduction
The tendon/ligament-to-bone interface or enthesis has a complex structure/organization to enable transfer of forces through the tendon/ligament to the bone. The body naturally achieves force transition through a four-layer gradient from tendon to fibrocartilage to mineralized fibrocartilage to bone.1–4 Examples of tissues with an enthesis include the supraspinatus tendon of the shoulder rotator cuff and the anterior cruciate ligament (ACL) in the knee.1,3 Since these enthesis locations are mostly avascular, healing after injury is very limited and often requires surgical intervention to repair the damage. It is estimated that there are 30,000–75,000 rotator cuff repair surgeries5 and 150,000 ACL surgeries6 each year in the United States. 6 In severe injuries to these sites, a graft is necessary for repair.1,6–9
Most grafts currently used clinically for repair are autograft or allograft, but each has its own complications. Autografts have additional harvesting surgeries, donor site morbidity, and increased risk for infection, while allografts have complications with possible immune rejection, problems with cellular infiltration, and incorporation into the surrounding tissue environment.6,10–12 Synthetic polymeric grafts have been used to investigate repair solutions in the ACL since the 1970s and showed good short-term success but never integrated well into the repair site and ultimately failed.13–17 Currently, research using tissue engineering principles is widely reported using different biomaterials, cell types, and growth factors.18,19 The focus has been to create different conditions on a single scaffold that can be specific to multiple tissue types, 20 such as muscle to tendon, cartilage to bone, and tendon/ligament to bone. This strategy can be implemented by changing the biomaterial’s chemical or physical structure, the cell source or a growth factor signal specific to the targeted tissue, or any combination of the above. 21
Regardless of tissue application, the scaffold will have to integrate into the tissue to transfer the loads of the musculoskeletal tissues to regain function. 1 This study seeks to identify the culture conditions that will allow a tendon-to-bone repair construct made by modifying current commercially available degradable scaffold with a tissue-specific coating of extracellular matrix (ECM) deposited by fibroblasts (FB) and osteoblasts (OB). FB will be used to deposit “tendon” ECM and OB will be used to deposit “bone” ECM on separate regions of the scaffold’s surface. Prior to a full characterization of the cell-deposited coating on the scaffold, we look to identify a co-culture medium formulation that will allow a low FB mineralization ration to OB mineralization and implement a seeding technique that will allow specific culture regions on the scaffold’s surface. Once the technique and culture medium were identified, a preliminary evaluation of basic ECM components was performed to assess the deposition behavior of the cells in co-culture.
Methods
Medium determination
Tendon tissue is not mineralized, yet bone tissue is, so to determine the appropriate co-culture medium formulation with respect to mineralization, NIH 3T3 mouse FBs (CRL-1658; ATCC, Manassas, VA, USA) or MC 3T3-E1 mouse calvarial OBs (CRL-2593; ATCC) cells were seeded on tissue culture plastic (TCP) in single culture with varying concentrations of the mineralizing agent, beta-glycerophosphate (β-GP) disodium pentahydrate (MPbio, Santa Ana, CA, USA), to show OB mineralization without significant FB mineralization. Both cell types were seeded in single culture at 1 × 104 cells/cm2 per well in a 12-well plate (BD Falcon, San Jose, CA, USA) in α-minimum essential medium (MEM) (Hyclone, Waltham, MA, USA) containing 10% fetal bovine serum (FBS) (Hyclone) + 1000 U/mL penicillin, 1000 µg/mL streptomycin, and 2.5 µg/mL amphotericin-B (AB/Antibiotic-Antimycotic (AM)) (Gibco, Grand Island, NY, USA), and at every medium change, 25 µg/mL
Scaffold seeding
The scaffold used for this study is X-Repair™, a commercially available poly-
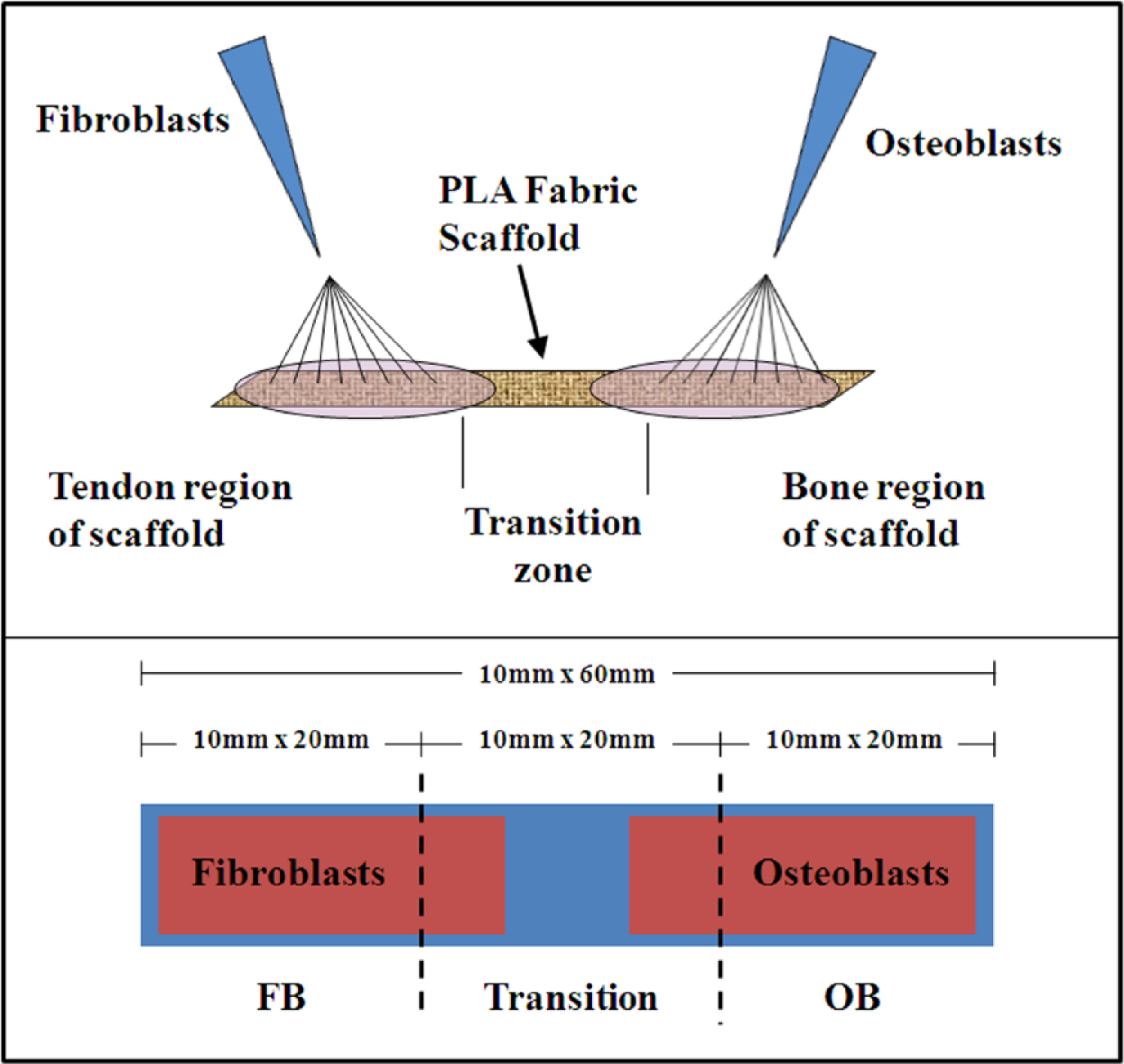
Schematic of how the PLA scaffolds are seeded with FB and OB in co-culture to make a tendon region and a bone region on the scaffold. The seeding area is shown as the red area in the bottom schematic. There is a transition zone between the two regions where both cells interact. For analysis, the scaffold is sectioned into equal thirds to measure the FB, OB, and transition regions.
Cell tracking and migration
To track the cells’ location and migration after seeding and to confirm that tendon and bone regions can be successfully seeded on a scaffold, the cells were labeled with two different fluorescent stains by a method modified by Wang et al. 2 FBs were labeled with CellTracker Green (Lonza, Allendale, NJ, USA) and the OBs were labeled with CellTracker Orange according to the manufacturer’s protocols. Briefly, cells were removed from the TCP flask, collected, and centrifuged at 350 xg for 5 min to form a pellet. FBs were resuspended in a 10 mM CellTracker Green and the OBs were resuspended in 10 mM CellTracker Orange for 30 min and placed in an incubator. Labeled cells were seeded on the scaffolds as described previously, as shown in Figure 1. The cells were allowed to attach to the scaffolds for 6 h after seeding. The scaffolds were then placed in a sterile custom-made cover glass Petri dish for fluorescence imaging. Using an inverted microscope (Nikon, Melville, NY, USA) and a motorized stage (ASI Imaging, Eugene, OR, USA), the entire scaffold was imaged by taking approximately 200 images at 4× magnification and stitching the images into a montage using BIOQUANT OSTEO software (BIOQUANT, Nashville, TN, USA). Scaffolds were imaged once at excitation/emission (ex/em) of 470 nm/515 nm and then again at ex/em of 540 nm/590 nm. Both images were then merged together with BIOQUANT. Images were replicated at 6, 18, 30, and 42 h for cell migration.
ECM deposition
ECM on the scaffolds was quantified after 28 days to characterize the amount and composition of the deposited ECM. Cells were cultured in α-MEM containing 10% FBS + AB/AM + 3 mM β-GP and 25 µg/mL AA was freshly added at every medium change. All scaffolds were cultured individually in 100 mm non-treated polystyrene Petri dishes. Scaffolds were moved to new Petri dishes every 7 days to prevent cells that migrated off the scaffolds from becoming confluent. Scaffolds (n = 4) were collected at timepoints of 1, 7, 14, 21, and 28 days and cut into three 10 × 20 mm sections: an FB section, a transition section, and an OB section, as demonstrated in Figure 1. Each section was placed in a 1.5-mL microcentrifuge tube and 1 mL of a buffered enzymatic digestion solution of 100 µg/mL proteinase-K (Promega, Madison, WI, USA) was added to every tube. All samples were then placed in an oven at 60°C overnight according to the manufacturer’s instructions to digest the ECM. The following day phenylmethanesulfonylfluoride (PMSF) was added to a concentration of 5 mM to inhibit the proteinase-K. All samples were homogenized using a sonic dismembrator and aliquots were removed for analysis. One aliquot was used to test for cell number through DNA quantification (PicoGreen assay; Invitrogen). Another aliquot was used to measure the glycosaminoglycan (GAG) content (National Diagnostics, Atlanta, GA, USA) through an Alcian Blue precipitation reaction.22,23 The last aliquot was used to quantify collagen content through the detection of the amino acid hydroxyproline (HYP).24,25 All volumes were carefully recorded for normalization during analysis.
Statistics
The data for each test were collected and averaged. All assays including Pierce BCA, calcium, GAG, and HYP were normalized to ng DNA as indicated in each figure. Then two-way analysis of variance (ANOVA) with Holm–Sidak post hoc test was performed on the necessary groups at a significance level of
Results
This study had three main objectives: (1) to determine a suitable medium formulation to reduce FB-to-OB mineralization, (2) to demonstrate successful substrate seeding in co-culture to create multiple regions on a single scaffold, and (3) to measure and quantify the ECM deposited across the scaffold and the difference in co-cultured regions.
Medium determination
The co-culture medium was modified in order to enhance a deposition effect and a mineralization effect. AA was added to aid collagen deposition and β-GP was added for mineralization. AA concentration was held constant for all formulations at 25 µg/mL. The amount of β-GP was varied in the co-culture medium to look at differences in total protein expression and calcium deposition per cell by both FB and OB in single culture. Cell number was estimated from DNA measurements. Figure 2 shows that the FBs did not differ significantly in total protein per cell regardless of β-GP concentration. There was only a gradual increase in protein deposition between days 1 and 14. The only significant difference is between days 1 and 14 in the 5-mM group. The OB protein deposition was also not significantly affected by the β-GP. However, there were some significant time-dependent increases in total protein compared to day 1 independent of β-GP. Overall, β-GP had little effect on total protein deposition, which may be more strongly attributed to the AA.26–28

Average total protein deposition plus standard deviations of (a) FB and (b) OB cultured in 0, 1, 3, or 5 mM β-GP. Each group was measured in triplicate. There was no significant difference in FB or OB total protein deposition with regard to β-GP concentration. There are some significant time-dependent effects on protein deposition.
There was a more observable effect of β-GP concentration on mineralization in the OB cell line as shown in Figure 3. Within days 1, 7, and 14, the 5 mM β-GP concentration produced a significantly higher amount of calcium deposition than the other concentrations. Between days 1 and 14, there was a significant time-dependent increase (significance is not indicated in the OB graph) in calcium deposition for every β-GP concentration. There was no significant difference within each timepoint for β-GP concentration on FB mineralization. There was, however, a significantly higher amount of FB calcium deposition on day 14 timepoint compared to other days, with the exception of the 3 mM β-GP group. The objective of the mineralization analysis was to find the lowest ratio of FB-to-OB mineralization. The ratios of FB-to-OB mineralization at day 14 for 0, 1, 3, and 5 mM are 0.73, 1.41, 0.58, and 0.68, respectively. It is unclear why the 3-mM FB timepoint had the lowest mineralization ratio, while the 1-mM ratio had the highest. A more definitive mineralization trend may be more apparent had the cells been cultured for a longer period of time. However, based on the results, the 3-mM β-GP concentration was used in medium formulations for the subsequent experiments.

Average calcium deposition plus standard deviations of (a) FB and (b) OB cultured in 0, 1, 3, or 5 mM β-GP. Each group was measured in triplicate. There was no significant difference between β-GP concentrations in FB mineralization. There was a significant time-dependent increase in day 14 compared to other days. For OBs, there was a significant increase in mineralization of the 5-mM concentration for each timepoint compared to the other concentrations. There was also a significant increase in mineralization for all concentrations between days 1 and 14 (not indicated on graph).
Fluorescence imaging of scaffold seeding
The second objective of this study was to investigate seeding a single scaffold in co-culture to create FB- and OB-specific regions. The FBs were stained with green tracking probe and the OBs were labeled with a red tracking probe. The montage image in Figure 4 shows the entire 10 mm × 60mm scaffold first with the green filter enabled then with the red filter. Each montage comprises approximately 200 individual 4× magnification images taken with the aid of a motorized stage and stitched together using BIOQUANT software. As can be seen in Figure 4, both FBs and OBs are attached on their respective half of the scaffold creating the tissue-specific regions. The general tissue-specific regions at low magnification are easier to distinguish. Figure 5 contains the two non-merged images with smaller selected regions shown at higher magnifications. There are very few FBs in the OB region and vice versa. On the left-hand edge of the OB side, FBs did attach and are more evident at higher exposure times. However, most cells are located in their respective regions with a decreasing gradient across the scaffold. There was no noticeable migration of cells between regions observed over the 42 h in Figure 6. The fluorescence label loses intensity over time, and therefore, the exposure time was increased by the 42-h image to intensify the colors on the scaffold. Because of the increased exposure, the 42-h image has a noticeable amount of unanticipated autofluorescence in the periphery of the image. This is due to the cyanoacrylate used to make the custom glass cover slip Petri dish for imaging and not the FB fluorescing.

Fluorescent-labeled FB (green) and OB (red) on the PLA scaffold form distinct tissue-specific regions at 6 h. The two images using the CellTracker Green and CellTracker Orange probes separately were merged to show how the cells are seeded on the entire scaffold. Image consists of approximately 200 images at 4× magnification stitched together. Scaffold size is 10 mm wide × 60 mm long. The merged image on the right shows the approximate 10 mm × 20 mm areas for FB, OB, and transition regions.

(a, b) Same two images seen from Figure 4 with higher magnification selections highlighted. (c–e) FB in the OB, transition, and FB regions, respectively. (f–h) OB in the OB, transition, and FB regions, respectively.
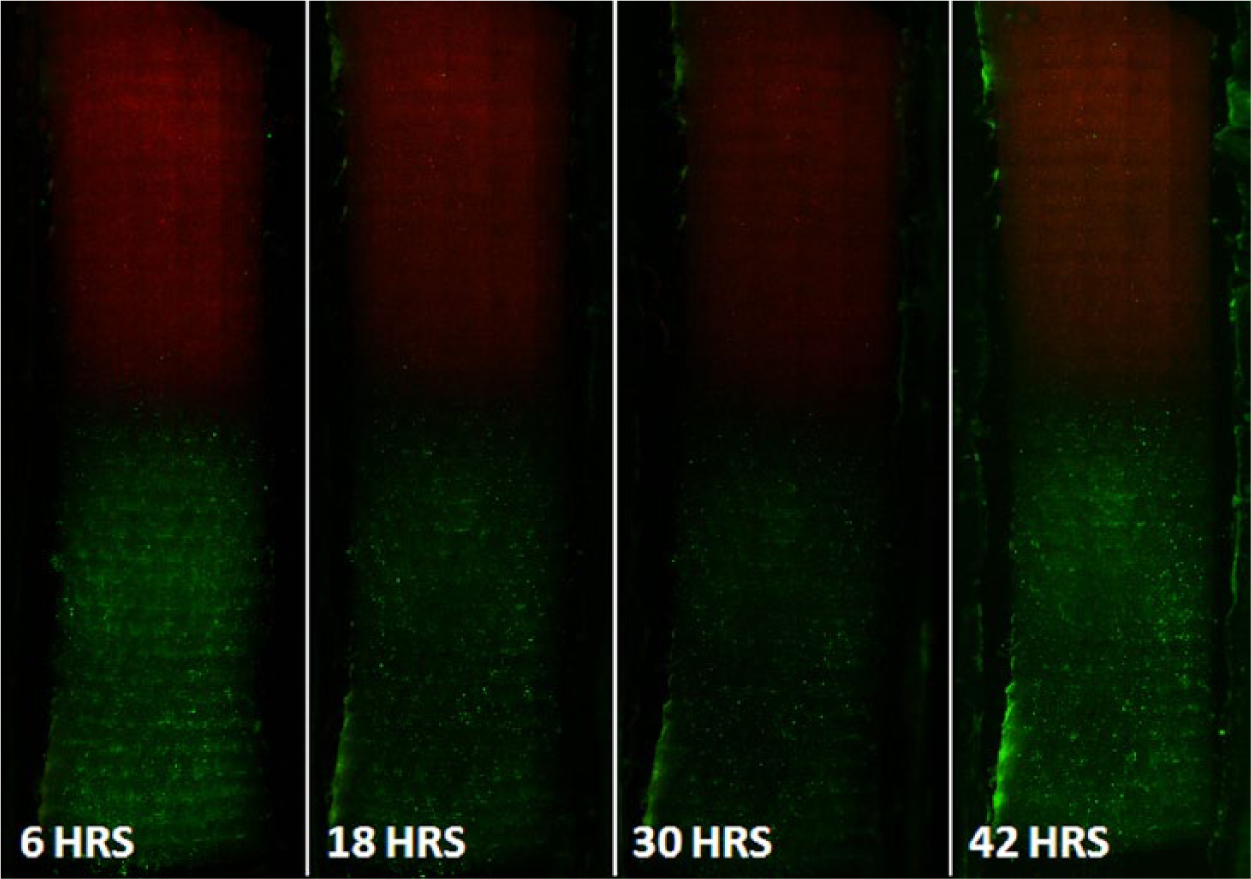
Merged fluorescent images over a 42-h period. Images show that the cells maintain the tissue-specific regions on the scaffold. No noticeable migration was observed in the measured time frame. There are green-labeled FB in the OB region and red-labeled OB in the FB region, even though no major cell migration was observed.
ECM deposition
The last objective of this study was to quantify the basic components of the ECM deposited on the static scaffolds in the FB, OB, and transition regions. These components are GAGs and collagen measured by Alcian Blue staining and HYP content, respectively. Deposited matrix distribution was measured for three regions of each scaffold: the FB, transition, and OB regions. The DNA quantified in Figure 7 indicated no significant differences between particular sections representing uniform cell dispersal over the scaffold, including the transition section. There was a significant increase in DNA after day 1 and the cells seemed to plateau between days 7 and 14. DNA was used to normalize the GAG and HYP data. GAG/DNA measurement was significantly higher in the FB region compared to the other regions on days 14 and 28. No significant collagen deposition difference between scaffold regions was determined in these static conditions. A continual and significantly increasing deposition of collagen over the entire 28-day study was found. This indicates good cell viability and activity on the scaffold. Even though not statistically significant, an interesting observation was that the ECM detected in the transition region had intermediate values in between the OB and FB regions, with the exception of day 28 for the collagen assessment, and would suggest mixed contributions from each cell type on the scaffold. Further testing in future studies would be needed to prove this hypothesis.

Quantification of DNA, GAG, and HYP deposition on the co-cultured scaffolds. The scaffolds were sectioned into equal thirds and analyzed separately. All data are mean values plus standard deviations. Groups were tested with n = 4 replicates. (a) DNA amounts were not significantly different between the scaffold regions, but it did significantly increase from day 1 and then stayed relatively constant. (b) There was a significantly higher deposition for GAG/DNA in the FB region compared to the OB region for days 14 and 28, with the transition region having intermediate values between the two. (c) There were no significant differences between groups for HYP/DNA deposition, but there was significantly increasing collagen content over time. Continually increasing collagen deposition is indicative of active and viable cells on the scaffold, in this co-culture model.
Discussion
The ultimate goal of this research is to create a more effective tissue-engineered scaffold for tendon-to-bone interfaces that will integrate with the host tissue, provide functional aid while healing, and then degrade over time as the host tissue replaces the scaffold. The approach taken to achieve this goal was a tissue engineering paradigm to create a tendon-specific and a bone-specific region on a scaffold using cell-deposited ECM on a degradable PLA fabric scaffold. For the tendon region, FBs were used and for the bone region, OBs were cultured on the scaffold. In this study, specifically, the foundations for the co-cultured scaffold including a co-culture-compatible medium formulation were determined and a seeding technique to create different specific regions on the scaffold was established. Then the seeded scaffolds were cultured and a preliminary analysis of the deposited ECM in each specific region including a transition zone was quantified with regard to basic ECM components, collagen, and GAG.
Other studies have taken similar approaches to repair musculoskeletal interfaces. Synthetic degradable polymers and ECM components are common scaffold choices for tendon/ligament tissue engineering due to control of factors such as mechanical properties, pore sizes, degradation properties, and scaffold geometry.6,21,29–32 For example, to have soft tissue ingrowth into a scaffold, a minimum pore size of 200 µm has been suggested, while calcified tissue needs a minimum of 100 µm. Another approach to engineering interfaces is changing the cell type involved with the scaffold. OBs are common for bone tissue engineering and specialized tendon FBs, called tenocytes, have been used for tendon. Tenocytes deposit tendon-specific ECM, but tenocytes also have low metabolic activity and low healing capabilities, which are used for maintenance and not necessarily regeneration.9,11 Therefore, other FBs, including dermal FBs, have been investigated as well. 33 After choosing individual cell types for the appropriate ECM deposition, introducing the cells into a co-culture environment could modify the ECM deposition compared to a single culture. In one published study by Wang et al., 2 they looked at how OBs and FBs interact in a co-culture system for a ligament-to-bone interface, including preliminary experiments to determine medium composition and ECM deposition of the co-cultured multiphase setup similar to our studies.
The medium formulation of Wang et al. 2 found that 1 mM β-GP was optimal for low FB mineralization and retained OB mineralization at 7 or 14 days. 2 We found that 3 mM β-GP provided low FB-to-OB mineralization. This response is most likely cell line specific. In their study, primary bovine cells were used; 2 in comparison, we used well-characterized mouse cell lines, NIH 3T3 and MC 3T3. 26 The co-culture medium should be tuned to each unique tissue engineering system because ECM and mineralization deposition could have impacts on scaffold integration with tissues. For this application, the OB-seeded bone region was targeted for higher mineralization than the FB-seeded tendon region. It was hypothesized that the higher mineral content on the bone side would help provide an environment for anchoring the scaffold in the bone. This approach is a common technique for bone tissue engineering, which uses minerals like di- and tri-calcium phosphates, or hydroxyapatites to signal osteointegration.34,35 Conversely, a low mineralized tendon region is also a functional need. Native tendon is fibrous and non-calcified lending to predominantly tensile loading while bone being highly calcified acts mostly in compression. 36 Conditioning each region properly could help achieve functional loading earlier after implantation.
Mineralization is not the only important component to the tendon-to-bone junction; subtle changes in ECM composition, like elastin, collagens, proteoglycans, and GAGs, also play a role. It was observed that the deposition of ECM transition zone on our PLA scaffold had average values that were in between the bone and tendon region values with respect to GAG and collagen. This finding would indicate that the ECM deposition in the center of the scaffold is a mix of OB and FB expression. These initial findings in validating this ECM deposition approach have shown promise in establishing tissue-specific regions with a small transition. In future studies, planned identification of more specific changes with respect to tendon- and bone-specific ECM deposition will be investigated. Other reported tendon/ligament bone studies have shown how an FB and OB co-cultured scaffold can form transitions with characteristics of native tissue between regions. Wang et al. 2 showed that in a co-cultured FB and OB environment, an increased collagen type II ECM, representative of a cartilaginous zone, was deposited without the presence of cartilage-forming cells. Their approach was taken a step further to include chondrocytes. Several successive studies by Spalazzi et al.12,37,38 using degradable multiphasic scaffolds with controlled porosity and a tri-culture of FBs, chondrocytes, and OBs measured interface-specific ECM components in vitro and in vivo to understand the role of multiple tissue types in a single scaffold. They have demonstrated that the ECM of the ligament, fibrocartilage, and bone regions found at the enthesis can be partially recreated with in vitro culture. While no functional mechanical data of the tri-phasic scaffolds were reported, these experiments are valuable to understand how cells can interact spatially to produce ECM of the enthesis. In contrast, Ma 29 proposes that because neo-tissue genesis in a tissue engineering process is not exactly the same as developmental or wound healing, it is likely unnecessary and impractical for a tissue-engineered scaffold to completely duplicate the ECM. Therefore, an effective therapy may not need to fully recreate the tendon, fibrocartilage, and bone ECM in vitro, but deposit sufficient specific ECM to direct tissue integration in vivo. The production of a functional repair of the enthesis may need to gain short-term mechanical stability of the scaffold in order to initiate mechanical loading while the long-term integration of the scaffold is directed by signaling of the tendon- and bone-specific ECM coatings. A reported study by Encalada-Diaz et al. 39 suggests that in the rotator cuff, a short-term stability with long-term integration may be the case.
Previous studies with non-degradable fabric polyethylene terephthalate (PET) scaffolds showed very good short-term results in ACL repair, but were not able to achieve long-term integration with the surrounding tissues.13–17 Applying a tissue-specific ECM coating may help bridge the gap between the short-term success of polymer grafts and the long-term integration of biologic materials.17,40 For example, recently, Li et al. 41 have modified PET ligament scaffolds with bioglass and hydroxyapatite to try and increase osseointegration of the scaffold into the bone. They performed an in vivo bone tunnel study for 2 weeks and showed evidence of initial scaffold integration; however, long-term integration was not evaluated in their study. The degradable X-Repair PLA scaffold used in our studies potentially has an additional advantage over a non-degradable scaffold of slowly resorbing over a long-term period while still having short-term mechanical stability necessary for some functional loading. The tissue-specific ECM coatings deposited on the PLA scaffold may help integration with the surrounding tissues for successful long-term integration. Eventual investigation with a functional animal model will be necessary to fully address this hypothesis.
Clinically, this technology is very early in development, but the concept is that these commercially available scaffolds could be pretreated with precursor tissue-specific ECM, which once implanted could more quickly direct wound repair down a tissue-specific path. The bone-conditioned regions would osseointegrate more quickly than a similar non-coated scaffold and the same for the tendon-conditioned regions in its respective tendon tissues. This study has demonstrated preliminary evidence for this technology; however, the scope of this study is limited. Medium determinations could be further optimized to show more meaningful differences in FB-to-OB mineralization in culture. Perhaps, a longer culture time would show a better picture of the mineralization behavior for both cell types. Also in this study, basic ECM components including GAG and collagen were measured and while these components were found to be uniformly deposited and distributed across the scaffold indicating the culture was healthy throughout the 28-day period, both tendinous and bony tissues contain collagen and GAGs making it difficult to accurately assess the difference between the two cell-deposited ECMs. In future studies, a more specific analysis of the FB- and OB-deposited ECM will be performed to better characterize and understand the roles of the cells, especially in the transition region. Finally, immortalized murine cell lines were used for this preliminary investigation because their behavior in culture is well characterized, and the results tend to be more reproducible between experiments. Ideally, a person’s own cells could be used to facilitate conditioning of the scaffold for their personalized clinical treatment.
In conclusion, the preliminary steps taken in this study have aimed to create a cell-specific ECM environment on a mechanically robust degradable scaffold to enable better fixation of tendinous soft tissues at bony interfaces. The outlined approach is to produce tissue-preferred areas on a scaffold to target the native tissues to integrate with the scaffold. The scaffold will then assist with mechanical loading while the co-cultured ECM aids the bodies’ healing to remodel the wound. To achieve this goal, we have selected a suitable co-culture medium formulation for our initial and we were able to seed the scaffold in co-culture to create distinct regions on the scaffold. Finally, we measured ECM deposition on three regions of the scaffold. Our future work is to increase the specificity of the ECM coating through mechanical stimulation of the cell-seeded scaffolds while depositing ECM and perform more specific ECM characterization for tendon- and bone-specific markers including mineralization.
Footnotes
Acknowledgements
The authors would like to thank Synthasome, Inc. for generous donations of X-Repair PLA fabric. The authors also thank Jessica Jennings, PhD, at University of Memphis for helping with fluorescence microscopy.
Declaration of conflicting interests
The authors certify that there is no conflict of interest with any financial organization regarding the material discussed in the article.
Funding
This project was funded by the DOD PRORP-USAMRMC W81XWH-10-1-0768 grant and by the Biomaterials Applications of Memphis (BAM) Laboratories in the Biomedical Engineering Department at the University of Memphis.
